Properties of Microplasma Coating on AZ91 Magnesium Alloy Prepared from Electrolyte with and without the Borax Addition
Abstract
:1. Introduction
2. Materials and Methods
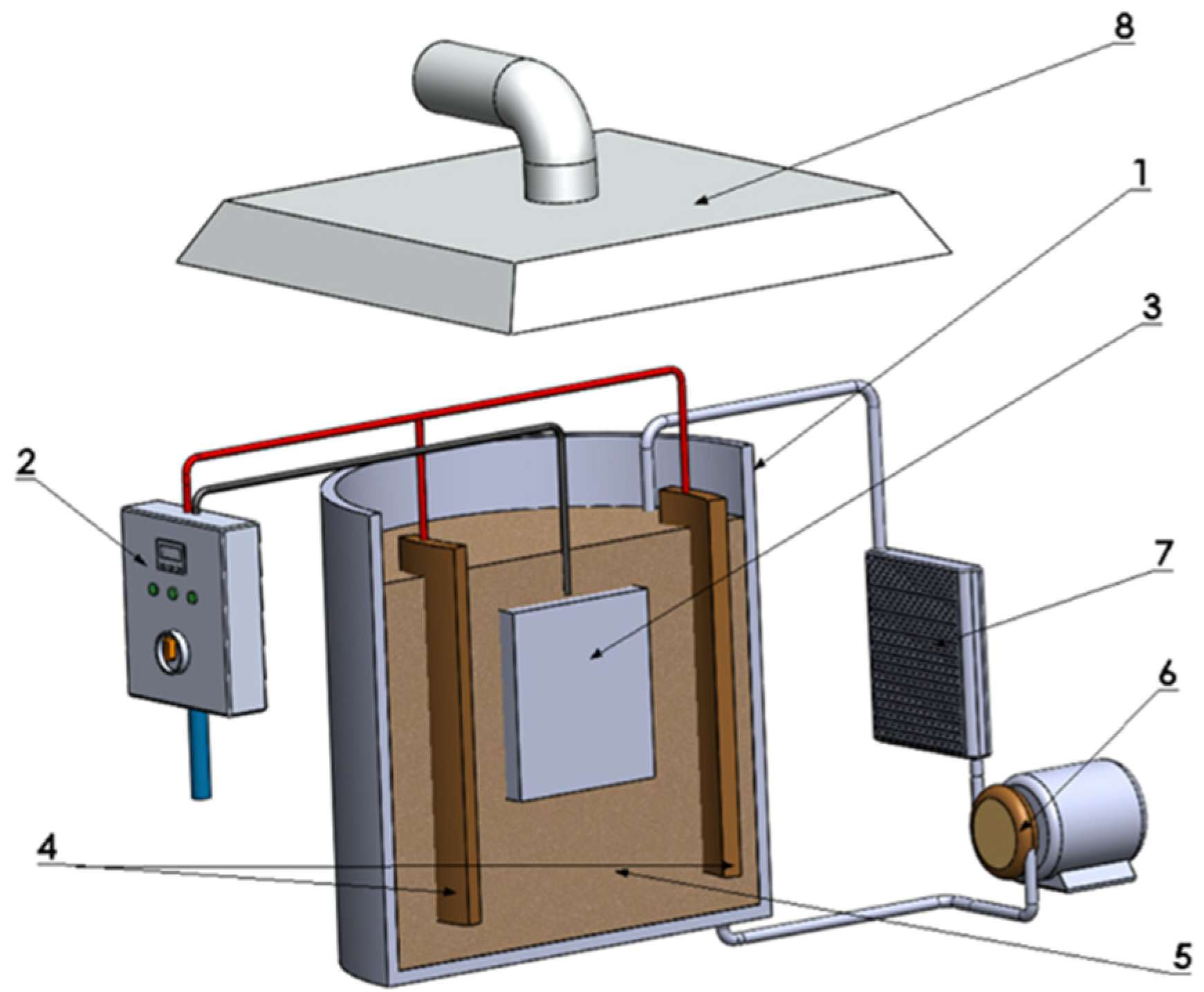
2.1. Preparation of Samples, Electrolytes, and PEO Coatings
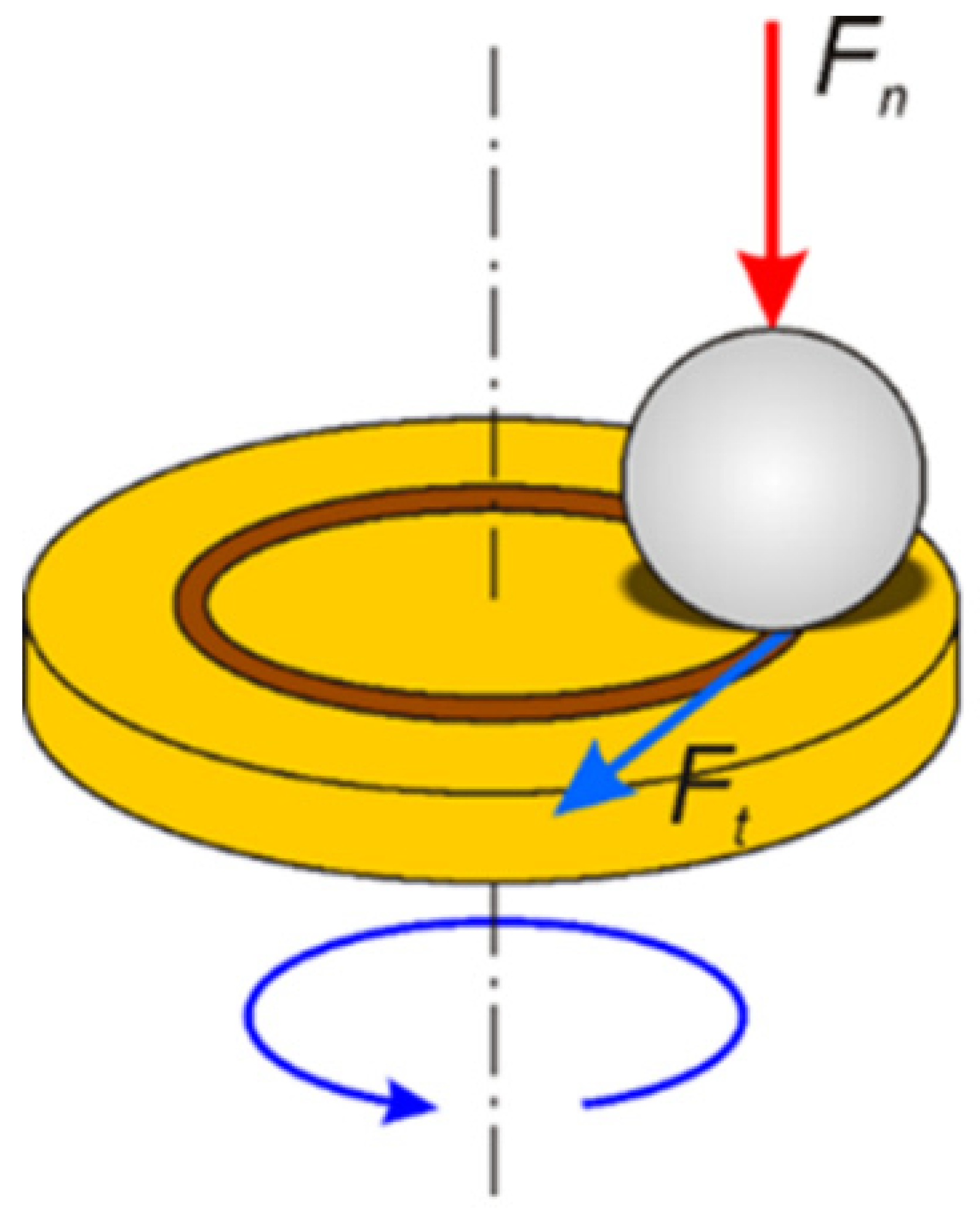
2.2. Characterization of the Substrates and PEO Coatings
3. Results and Discussion
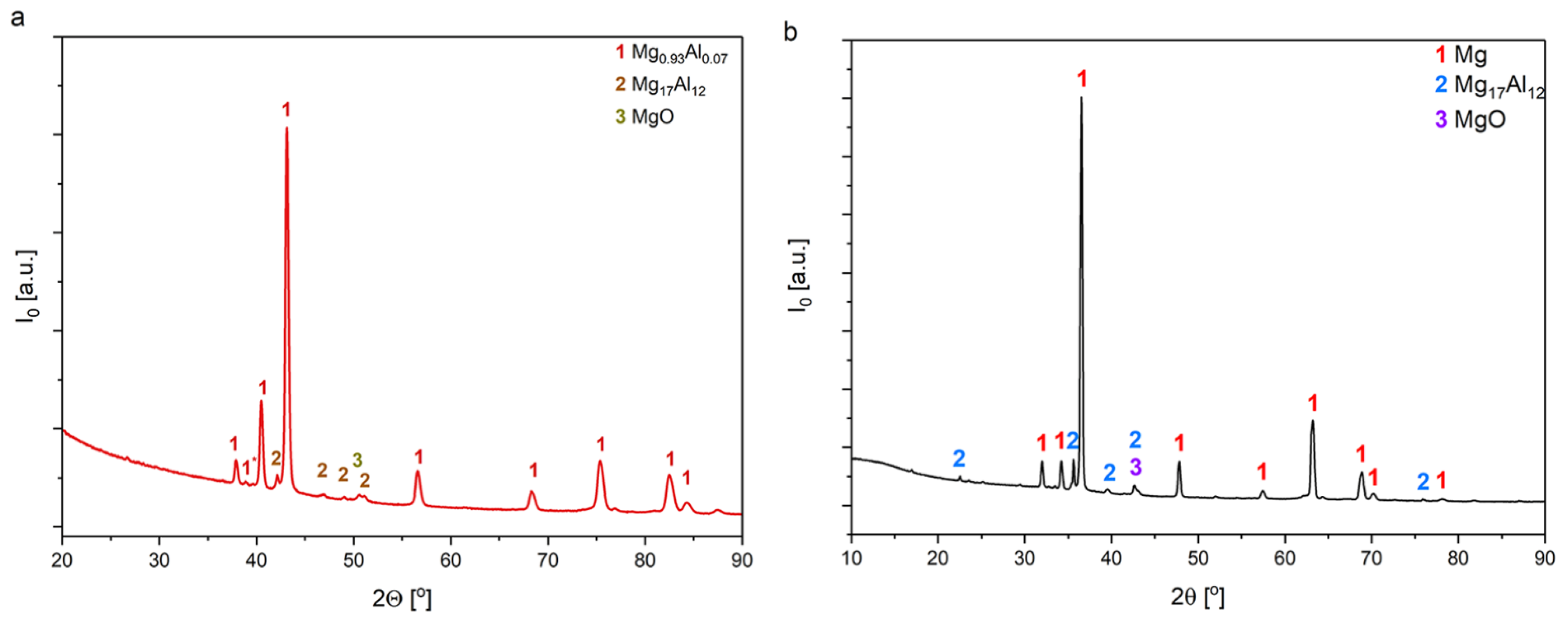
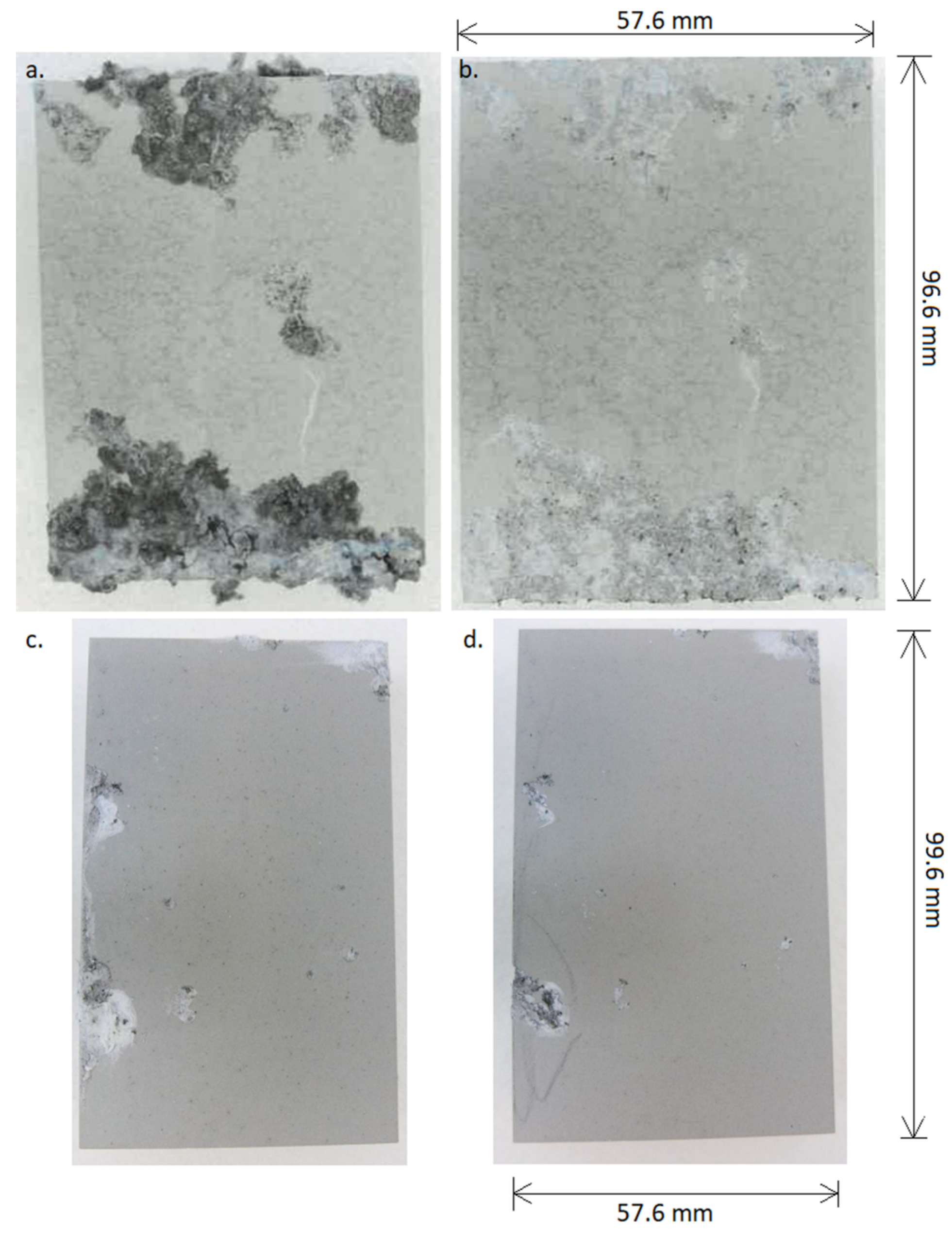
3.1. The Coatings Morphology and Structure
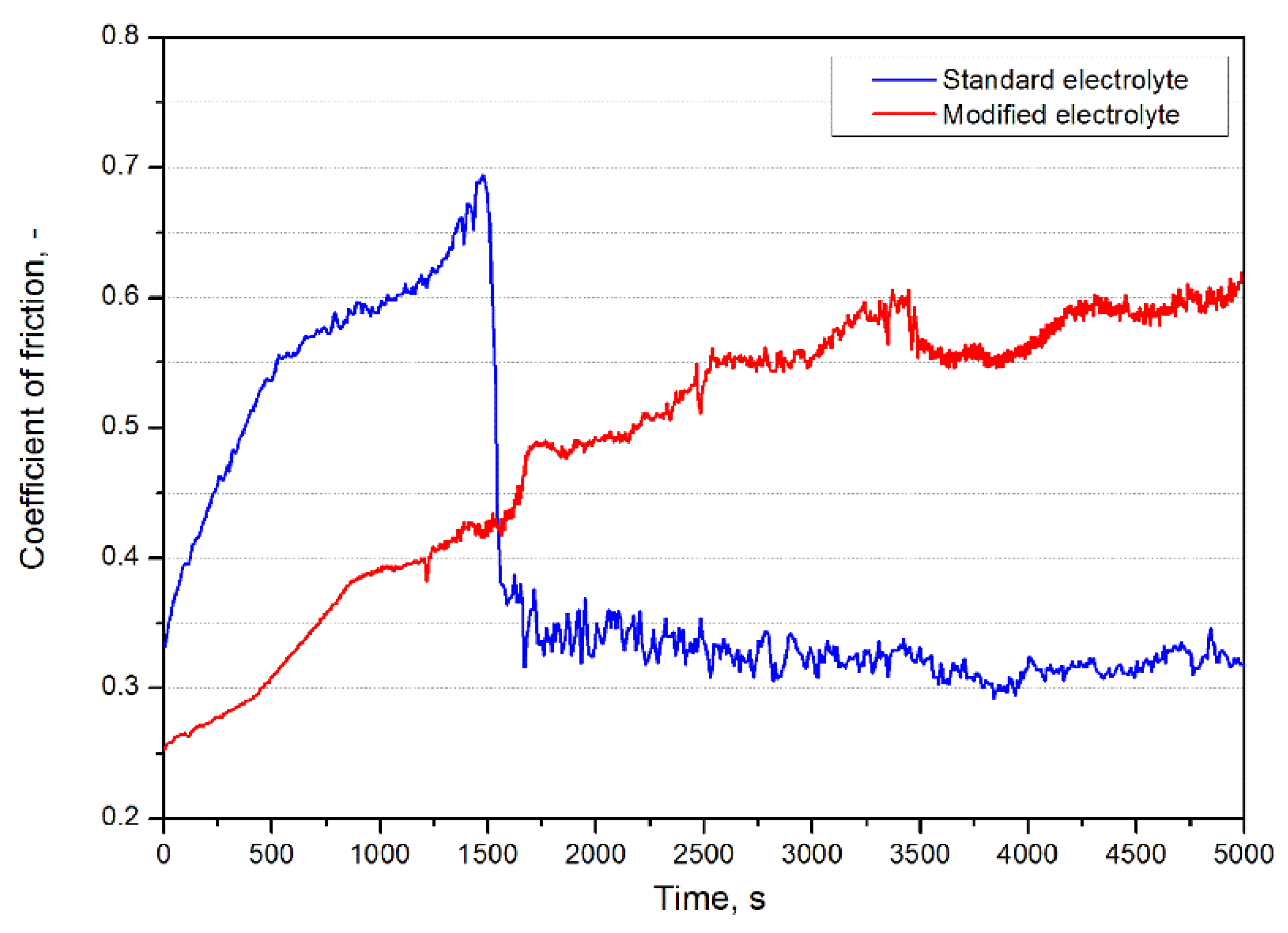
3.2. Wear Resistance of the Coatings
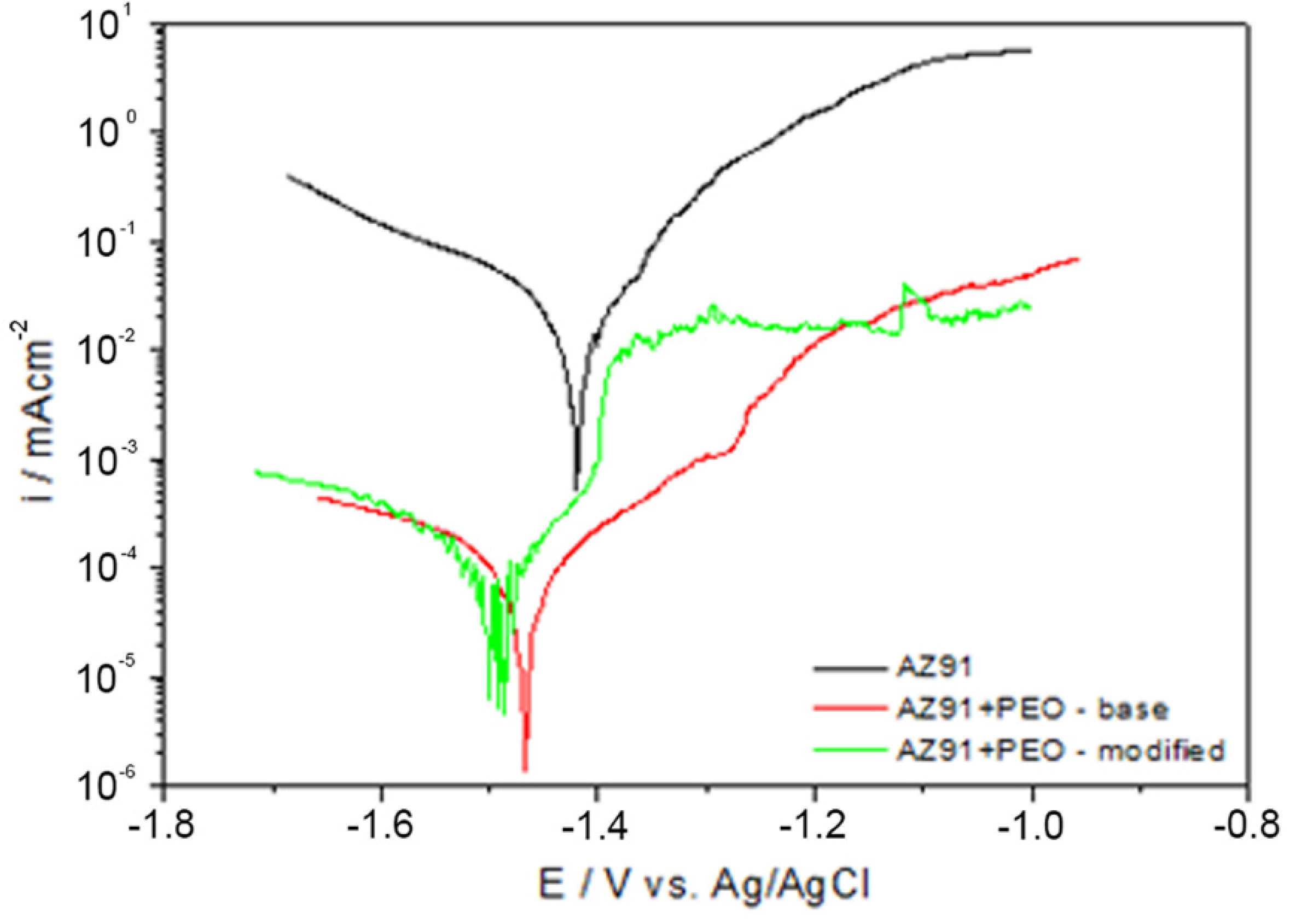
3.3. The Corrosion Resistance of the Coatings
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- You, C.S.; Huang, Y.; Kainer, K.U.; Hort, N. Recent research and developments on wrought magnesium alloys. J. Magnes. Alloy. 2017, 5, 239–253. [Google Scholar] [CrossRef]
- Darband, G.B.; Aliofkhazraei, M.; Hamghalam, P.; Valizade, N. Plasma electrolytic oxidation of magnesium and its alloys: Mechanism, properties and applications. J. Magnes. Alloy. 2017, 5, 74–132. [Google Scholar] [CrossRef]
- Hoche, H.; Rosenkranz, C.; Delp, A.; Lohrengel, M.M.; Broszeit, E.; Berger, C. Investigation of the macroscopic and microscopic electrochemical corrosion behaviour of PVD-coated magnesium die cast alloy AZ91. Surf. Coat. Technol. 2005, 193, 178–184. [Google Scholar] [CrossRef]
- Brunelli, K.; Dabala, M.; Calliari, I.; Magrini, M. Effect of HCl pre-treatment on corrosion resistance of cerium-based conversion coatings on magnesium and magnesium alloys. Corros. Sci. 2005, 47, 989–1000. [Google Scholar] [CrossRef]
- Ghali, E.; Dietzel, W.; Kainer, K.U. General and localized corrosion of magnesium alloys: A critical review. J. Mater. Eng. Perform. 2004, 13, 7–23. [Google Scholar] [CrossRef]
- Długosz, P.; Bochniak, W.; Ostachowski, P.; Molak, R.; Duarte Guigou, M.; Hebda, M. The Influence of Conventional or KOBO Extrusion Process on the Properties of AZ91 (MgAl9Zn1) Alloy. Materials 2021, 14, 6543. [Google Scholar] [CrossRef]
- Song, G.; Atrens, A. Understanding Magnesium Corrosion—A Framework for Improved Alloy Performance. Adv. Eng. Mater. 2003, 5, 837–858. [Google Scholar] [CrossRef]
- Zhao, M.C.; Liu, M.; Song, G.; Atrens, A. Influence of the β-phase morphology on the corrosion of the Mg alloy AZ91. Corros. Sci. 2008, 50, 1939–1953. [Google Scholar] [CrossRef]
- Szewczyk-Nykiel, A.; Długosz, P.; Darłak, P.; Hebda, M. Corrosion Resistance of Cordierite-Modified Light MMCs. J. Mater. Eng. Perform. 2017, 26, 2555–2562. [Google Scholar] [CrossRef] [Green Version]
- Song, G.; Atrens, A.; Wu, Z.; Zhang, B. Corrosion behaviour of AZ21, AZ501 and AZ91 in sodium chloride. Corros. Sci. 1998, 40, 1769–1791. [Google Scholar] [CrossRef]
- Yu, Z.; Tang, A.; Li, C.; Liu, J.; Pan, F. Effect of manganese on the microstructure and mechanical properties of magnesium alloys. Int. J. Mater. Res. 2019, 110, 1016–1024. [Google Scholar] [CrossRef]
- Zhou, W.; Aung, N.N.; Choudhary, A.; Kanouni, M. Evaluation of corrosion resistance of magnesium alloys in radiator coolants. Corros. Eng. Sci. Technol. 2011, 46, 386–391. [Google Scholar] [CrossRef]
- Kleszcz, K.; Hebda, M.; Kyzioł, A.; Krawiec, H.; Kyzioł, K. Towards prevention of biofilm formation: Ti6Al7Nb modified with nanocomposite layers of chitosan and Ag/Au nanoparticles. Appl. Surf. Sci. 2021, 557, 149795. [Google Scholar] [CrossRef]
- Jabłoński, P.; Hebda, M.; Pytlak, P.; Kyzioł, A.; Krawiec, H.; Grzesik, Z.; Kyzioł, K. Impact of chitosan/noble metals-based coatings on the plasmochemically activated surface of NiTi alloy. Mater. Chem. Phys. 2020, 248, 122931. [Google Scholar] [CrossRef]
- Khaselev, O.; Weiss, D.; Yahalom, J. Anodizing of Pure Magnesium in KOH-Aluminate Solutions under Sparking. J. Electrochem. Soc. 1999, 146, 1757–1761. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Zhang, M.Y.; Wu, K. Microarc oxidation coating formed on SiCw/AZ91 magnesium matrix composite and its corrosion resistance. Mater. Lett. 2005, 59, 1727–1731. [Google Scholar] [CrossRef]
- Lu, X.; Mohedano, M.; Blawert, C.; Matykina, E.; Arrabal, R.; Kainer, K.U.; Zheludkevich, M.L. Plasma electrolytic oxidation coatings with particle additions—A review. Surf. Coat. Technol. 2016, 307, 1165–1182. [Google Scholar] [CrossRef]
- Ur Rehman, Z.; Choi, D. Investigation of ZrO2 nanoparticles concentration and processing time effect on the localized PEO coatings formed on AZ91 alloy. J. Magnes. Alloy. 2019, 7, 555–655. [Google Scholar] [CrossRef]
- Pelczar, D.; Długosz, P.; Darłak, P.; Szewczyk-Nykiel, A.; Nykiel, M.; Hebda, M. The Effect of BN or SiC Addition on PEO Properties of Coatings Formed on AZ91 Magnesium Alloy. Arch. Metall. Mater. 2022, 67, 147–154. [Google Scholar] [CrossRef]
- Liang, J.; Guo, B.G.; Tian, J.; Liu, W.H.; Zhou, J.F.; Liu, W.M.; Xu, T. Effects of NaAlO2 on structure and corrosion resistance of microarc oxidation coatings formed on AM60B magnesium alloy in phosphate–KOH electrolyte. Surf. Coat. Technol. 2005, 199, 121–126. [Google Scholar] [CrossRef]
- Pezzato, L.; Rigon, M.; Martucci, A.; Brunelli, K.; Dabala, M. Plasma Electrolytic Oxidation (PEO) as pre-treatment for sol-gel coating on aluminum and magnesium alloys. Surf. Coat. Technol. 2019, 366, 114–123. [Google Scholar] [CrossRef]
- Shihai, C.; Jiunmin, H.; Weijing, L.; Suk-Bong, K.; Jung-Moo, L. Study on wear behavior of plasma electrolytic oxidation coatings on aluminum alloy. Rare Met. 2006, 25, 141–145. [Google Scholar] [CrossRef]
- Aliofkhazraei, M.; Rouhaghdam, A.S.; Gupta, P. Nano-Fabrication by Cathodic Plasma Electrolysis. Crit. Rev. Solid State Mater. Sci. 2011, 36, 174–190. [Google Scholar] [CrossRef]
- Einkhah, F.; Lee, K.M.; Sani, M.A.F.; Yoo, B.; Shin, D.H. Structure and corrosion behavior of oxide layer with Zr compounds on AZ31 Mg alloy processed by two-step plasma electrolytic oxidation. Surf. Coat. Technol. 2014, 238, 75–79. [Google Scholar] [CrossRef]
- Duan, H.P.; Yan, C.W.; Wang, F.H. Effect of Electrolyte Additives on Performance of Plasma Electrolytic Oxidation Films Formed on Magnesium Alloy AZ91D. Electrochim. Acta 2007, 52, 3785–3793. [Google Scholar] [CrossRef]
- Zhang, L.J.; Fan, J.J.; Zhang, Z.; Cao, F.H.; Zhang, J.Q.; Cao, C.N. Study on the anodic film formation process of AZ91D magnesium alloy. Electrochim. Acta 2007, 52, 5325–5333. [Google Scholar] [CrossRef]
- Bai, A.; Chen, Z.J. Effect of electrolyte additives on anti-corrosion ability of micro-arc oxide coatings formed on magnesium alloy AZ91D. Surf. Coat. Technol. 2009, 203, 1956–1963. [Google Scholar] [CrossRef]
- EN 1753:2019 Magnesium and Magnesium Alloys—Magnesium Alloy Ingots and Castings. Available online: https://standards.iteh.ai/catalog/standards/cen/da0f1cb6-f514-4599-b1b3-ef746eb29780/en-1753-2019 (accessed on 25 September 2019).
- Xin, S.; Song, L.; Zhao, R.; Hu, X. Influence of cathodic current on composition, structure and properties of Al2O3 coatings on aluminum alloy prepared by micro-arc oxidation process. Thin Solid Films 2006, 515, 326–332. [Google Scholar] [CrossRef]



| Symbol | Chemical Composition [wt%] | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Al | Zn | Mn | Si | Fe | Cu | Ni | Ti | Mg | |
| AZ91 | 8.50 | 0.75 | 0.13 | 0.02 | 0.02 | 0.01 | 0.002 | 0.025 | Balance |
| EN 1753: 2019 standard | 8.50 ÷ 9.50 | 0.30 ÷ 1.00 | <0.00 ÷ 0.15 | <0.30 | <0.03 | <0.025 | <0.001 | – | Balance |
| Base PEO Coating | Modified PEO Coating | ||
|---|---|---|---|
| Electrolyte composition | NaOH (g·cm−3) | 4 | 4 |
| Na2SiO3·5H2O (g·cm−3) | 15 | 15 | |
| NaF (g·cm−3) | - | 2 | |
| Na2B4O7 (g·cm−3) | - | 2 | |
| PEO process conditions | Time (s) | 360–420 | |
| Electrolysis voltage (V) | 405 | ||
| Average anodic current density (A·dm−2) | 5 | ||
| Controlled current frequency (Hz) | 1000 | ||
| Current rise rate (A·μs−1) | 200 | ||
| Level of current filling pulse (%) | 30 | ||
| Type of Sample | Designation of Samples |
|---|---|
| AZ91 magnesium alloy—reference sample | AZ91 |
| AZ91 magnesium alloy covered without the borax addition PEO layer (see Table 2) | Base PEO coating |
| AZ91 magnesium alloy covered with the borax addition PEO layer (see Table 2) | Modified PEO coating |
| Parameter | Value and Unit |
|---|---|
| Ball material | WC [−] |
| Ball diameter | 6 [mm] |
| Load | 2 [N] |
| The radius of the sliding circle | 6 [mm] |
| Sliding speed | 0.1 [m/s] |
| Sliding distance | 500 [m] |
| Duration of the test | 5 000 [s] |
| Number of cycles | 13 263 [−] |
| Point No. from Figure 4. | Base PEO Coating | Modified PEO Coating | ||||||
|---|---|---|---|---|---|---|---|---|
| O | Mg | Al | Si | O | Mg | Al | Si | |
| 1 | 57.2 | 31.3 | 3.1 | 8.4 | 50.8 | 34.6 | 2.3 | 12.3 |
| 2 | 48.1 | 43.7 | 3.7 | 4.5 | 37.2 | 50.9 | 6.2 | 5.7 |
| 3 | - | 91.5 | 8.5 | - | - | 91.4 | 8.6 | - |
| 4 | - | 90.0 | 10.0 | - | - | 93.0 | 7.0 | - |
| Polarization Parameter | AZ91 | Base PEO Coating | Modified PEO Coating |
|---|---|---|---|
| Ecorr (V) | −1.404 | −1.467 | −1.494 |
| icorr (A cm−2) | 2.0 × 10−5 | 1.2 × 10−7 | 1.7 × 10−7 |
| Rp (Ω·cm2) | 1297 | 3.18 × 105 | 2.58 × 105 |
| corrosion rate (mm/year) | 1.396 | 0.0692 | 0.0060 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bisztyga-Szklarz, M.; Rząd, E.; Boroń, Ł.; Klimczyk, P.; Polczyk, T.; Łętocha, A.; Rajska, M.; Hebda, M.; Długosz, P. Properties of Microplasma Coating on AZ91 Magnesium Alloy Prepared from Electrolyte with and without the Borax Addition. Materials 2022, 15, 1354. https://doi.org/10.3390/ma15041354
Bisztyga-Szklarz M, Rząd E, Boroń Ł, Klimczyk P, Polczyk T, Łętocha A, Rajska M, Hebda M, Długosz P. Properties of Microplasma Coating on AZ91 Magnesium Alloy Prepared from Electrolyte with and without the Borax Addition. Materials. 2022; 15(4):1354. https://doi.org/10.3390/ma15041354
Chicago/Turabian StyleBisztyga-Szklarz, Magdalena, Ewa Rząd, Łukasz Boroń, Piotr Klimczyk, Tomasz Polczyk, Aneta Łętocha, Maria Rajska, Marek Hebda, and Piotr Długosz. 2022. "Properties of Microplasma Coating on AZ91 Magnesium Alloy Prepared from Electrolyte with and without the Borax Addition" Materials 15, no. 4: 1354. https://doi.org/10.3390/ma15041354
APA StyleBisztyga-Szklarz, M., Rząd, E., Boroń, Ł., Klimczyk, P., Polczyk, T., Łętocha, A., Rajska, M., Hebda, M., & Długosz, P. (2022). Properties of Microplasma Coating on AZ91 Magnesium Alloy Prepared from Electrolyte with and without the Borax Addition. Materials, 15(4), 1354. https://doi.org/10.3390/ma15041354






