Structural Stability of the SUPER304H Steel Used in Energetics
Abstract
1. Introduction
2. Materials
3. Methods
3.1. Grinding and Polishing
3.2. Etching
3.3. Microscopy and Related Analyses
3.4. Bending
3.5. Welding
3.6. Mechanical Properties
3.7. Heat Treatment
3.8. Laboratory Aging
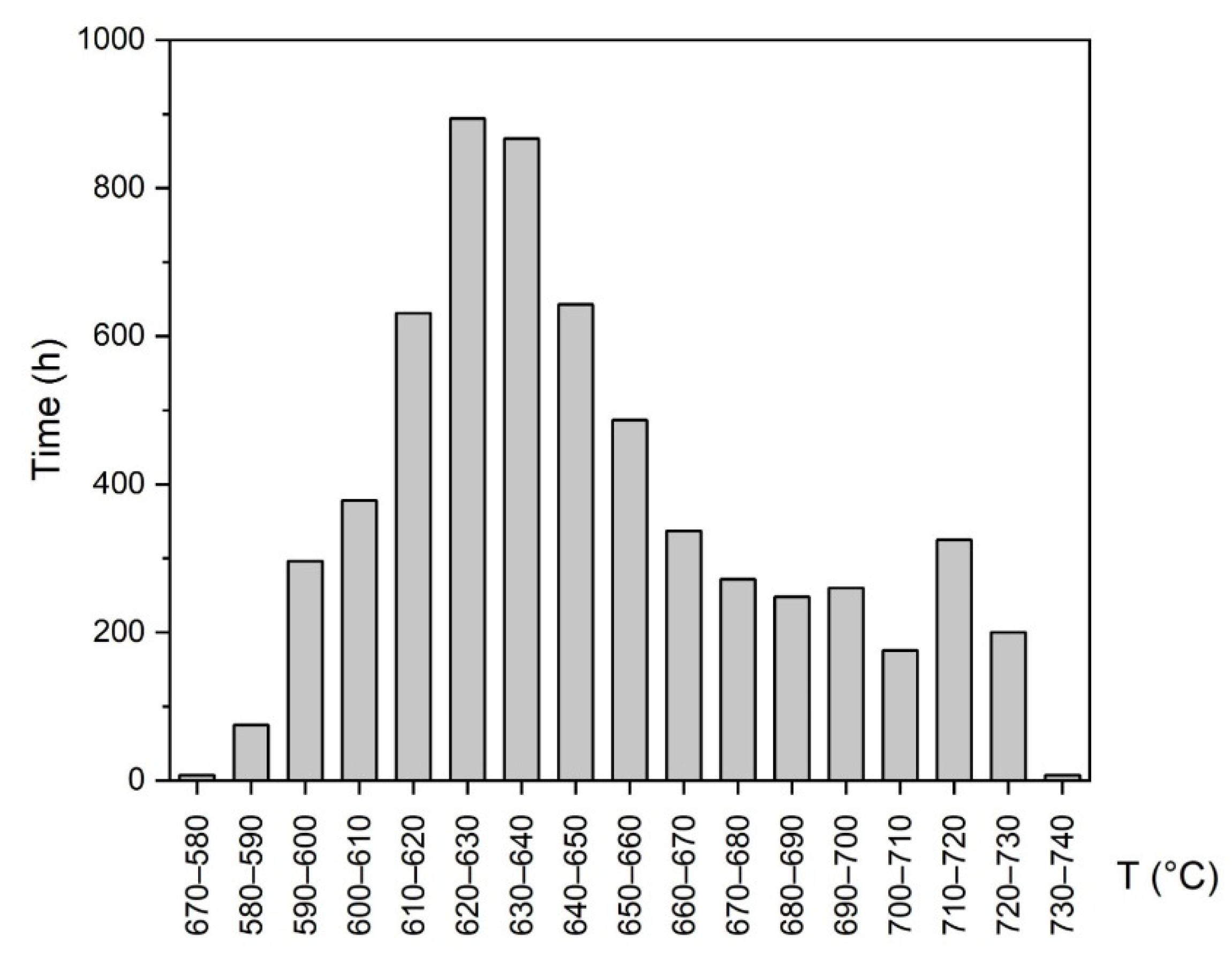
3.9. Power Plant Material Exposure
4. Results
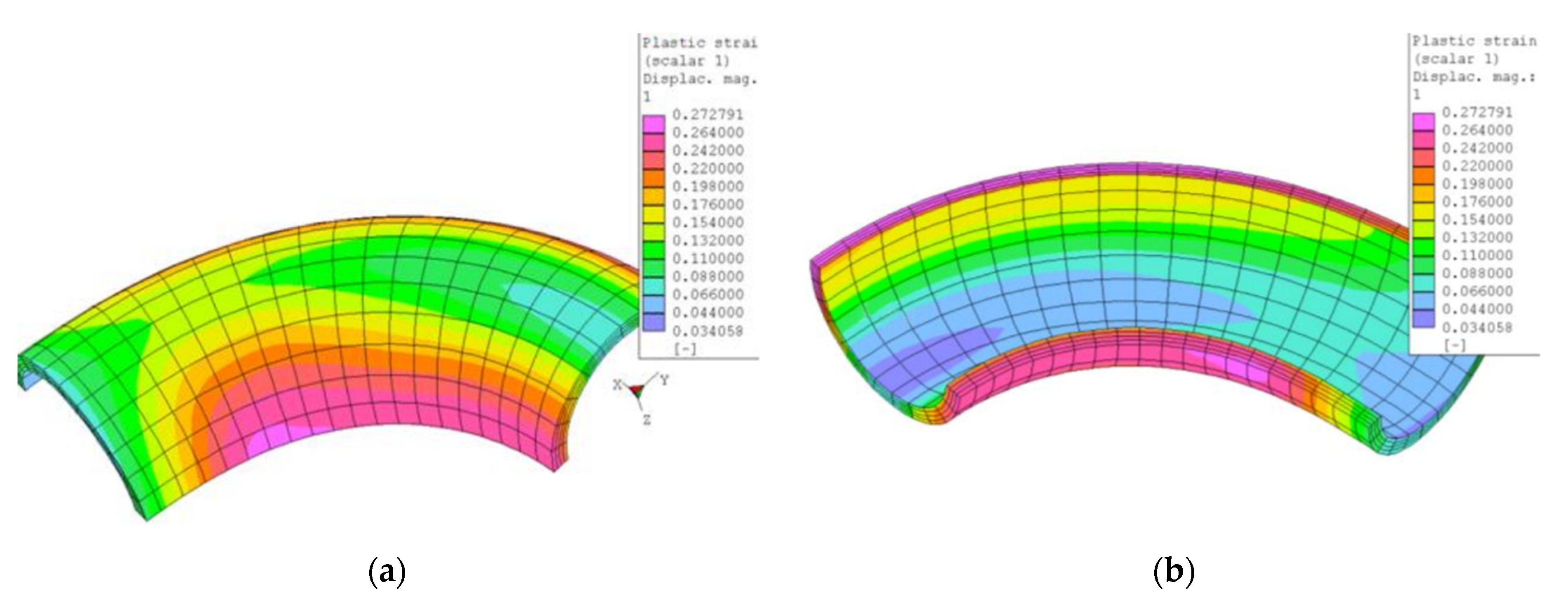
4.1. Bending
4.2. Welding
4.3. Tensile Test
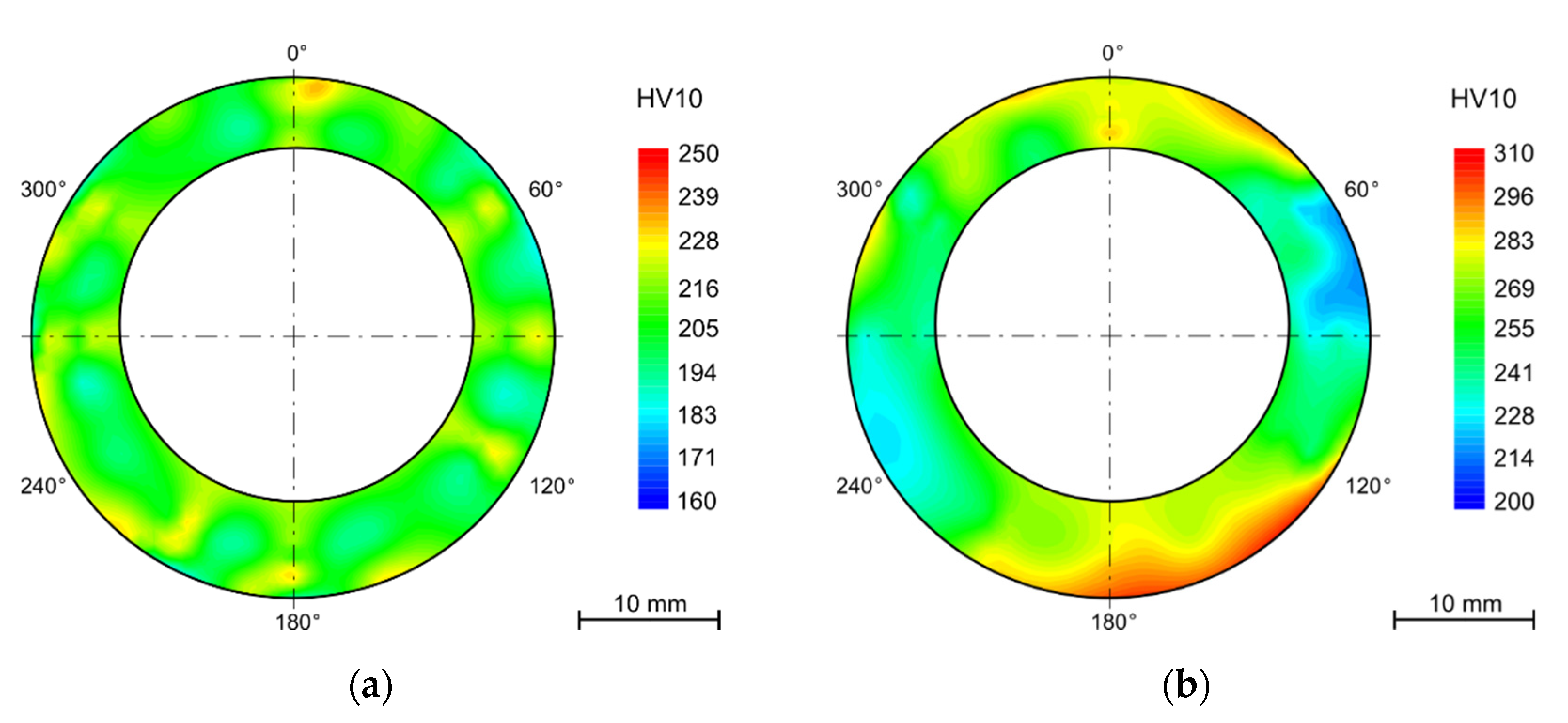
4.4. Hardness
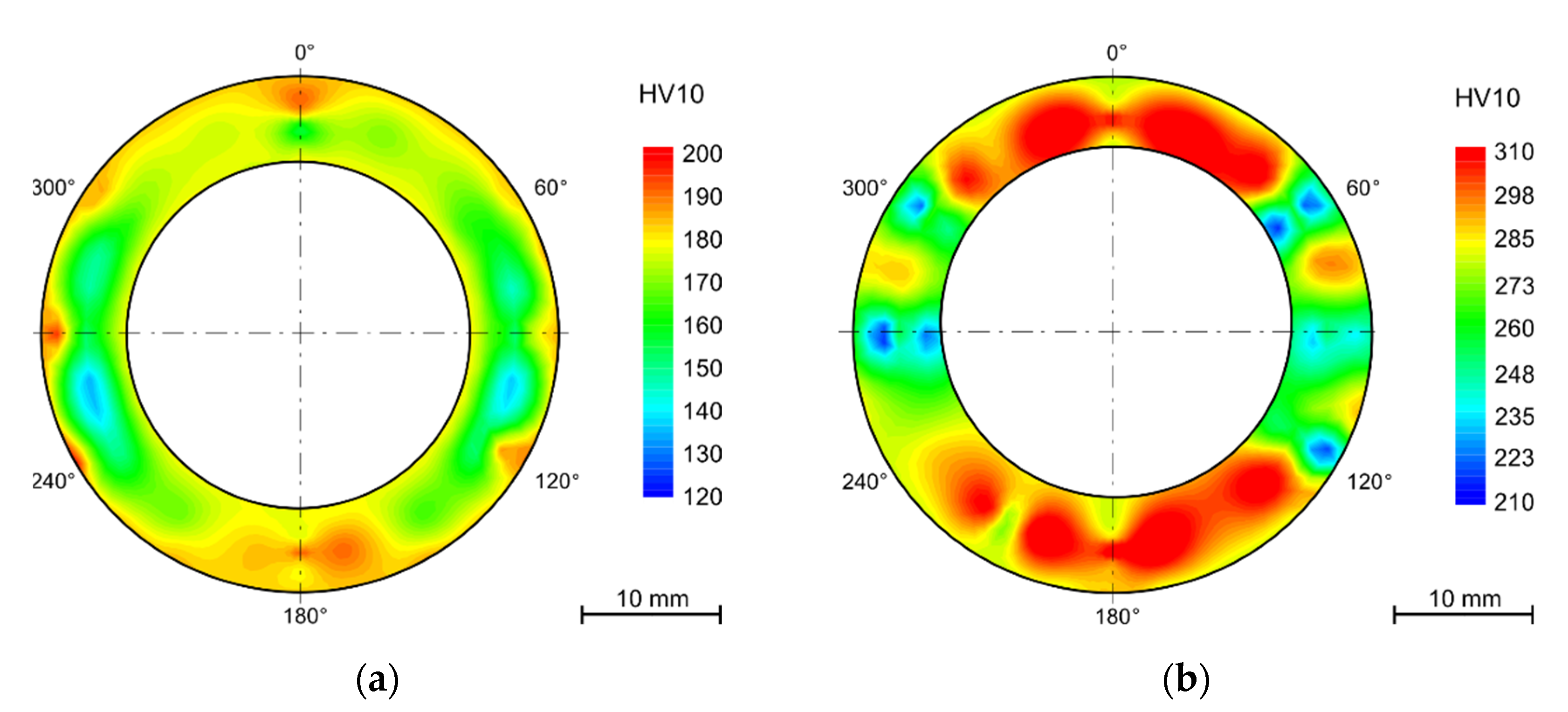
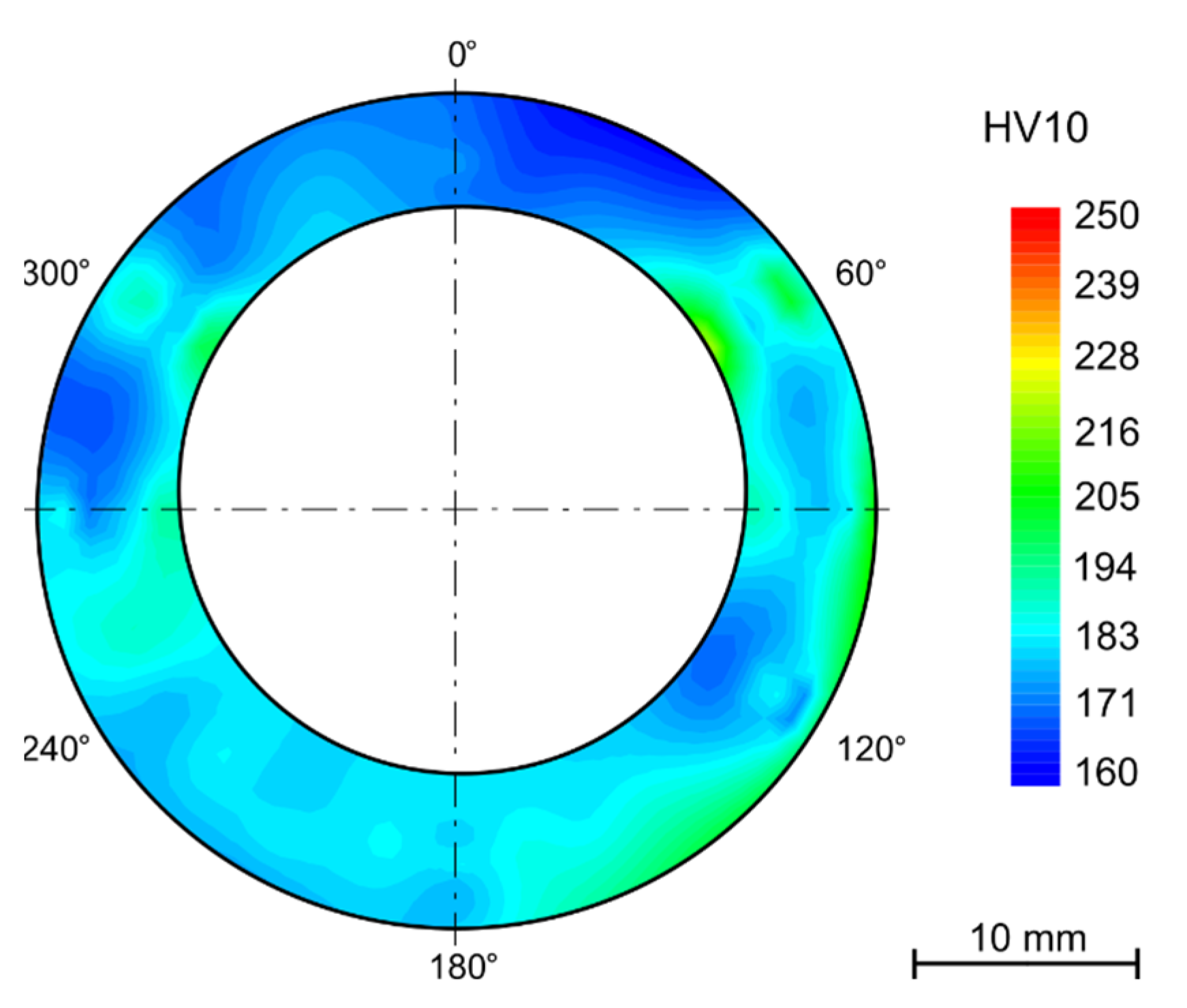
4.4.1. Bends
4.4.2. Welds
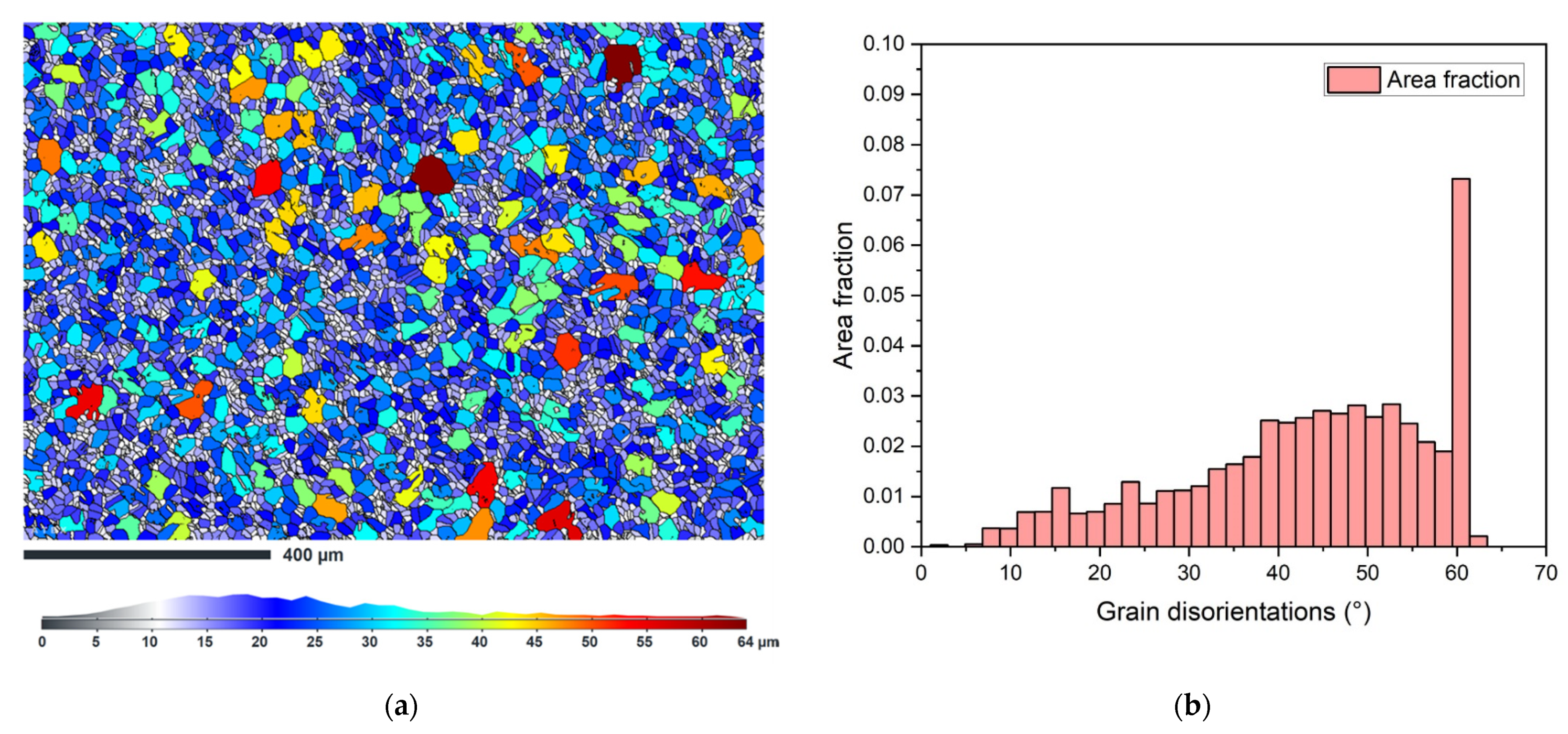
4.5. Grain Size
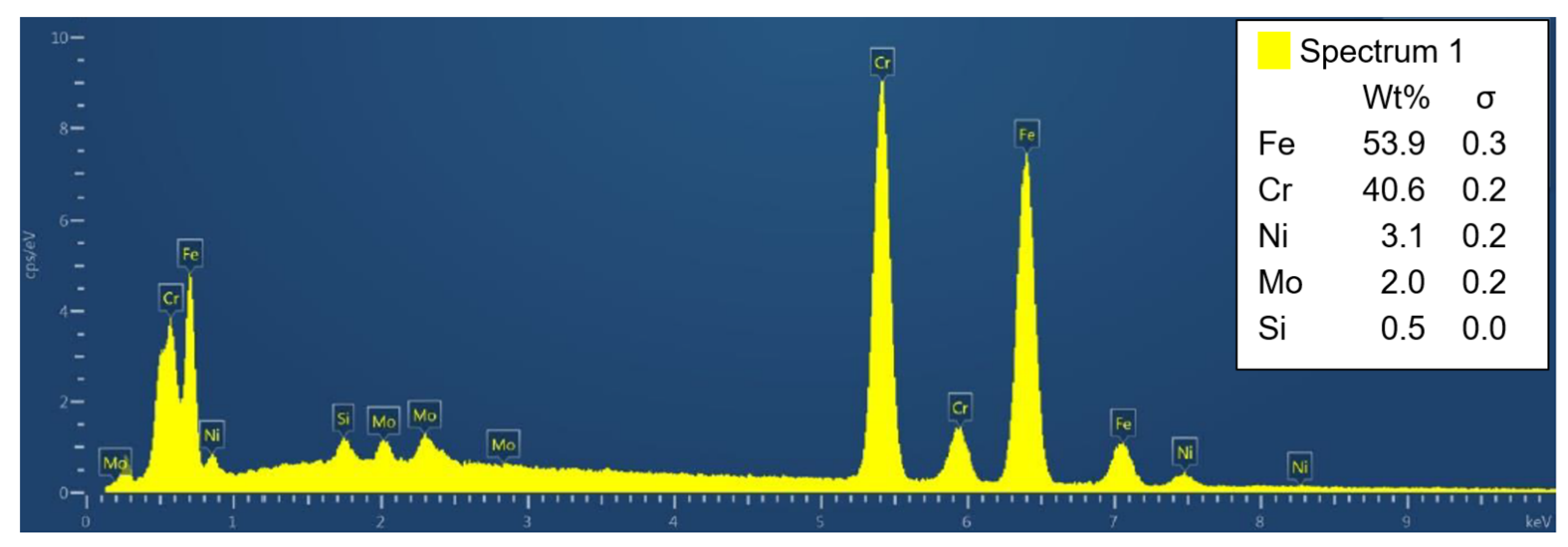
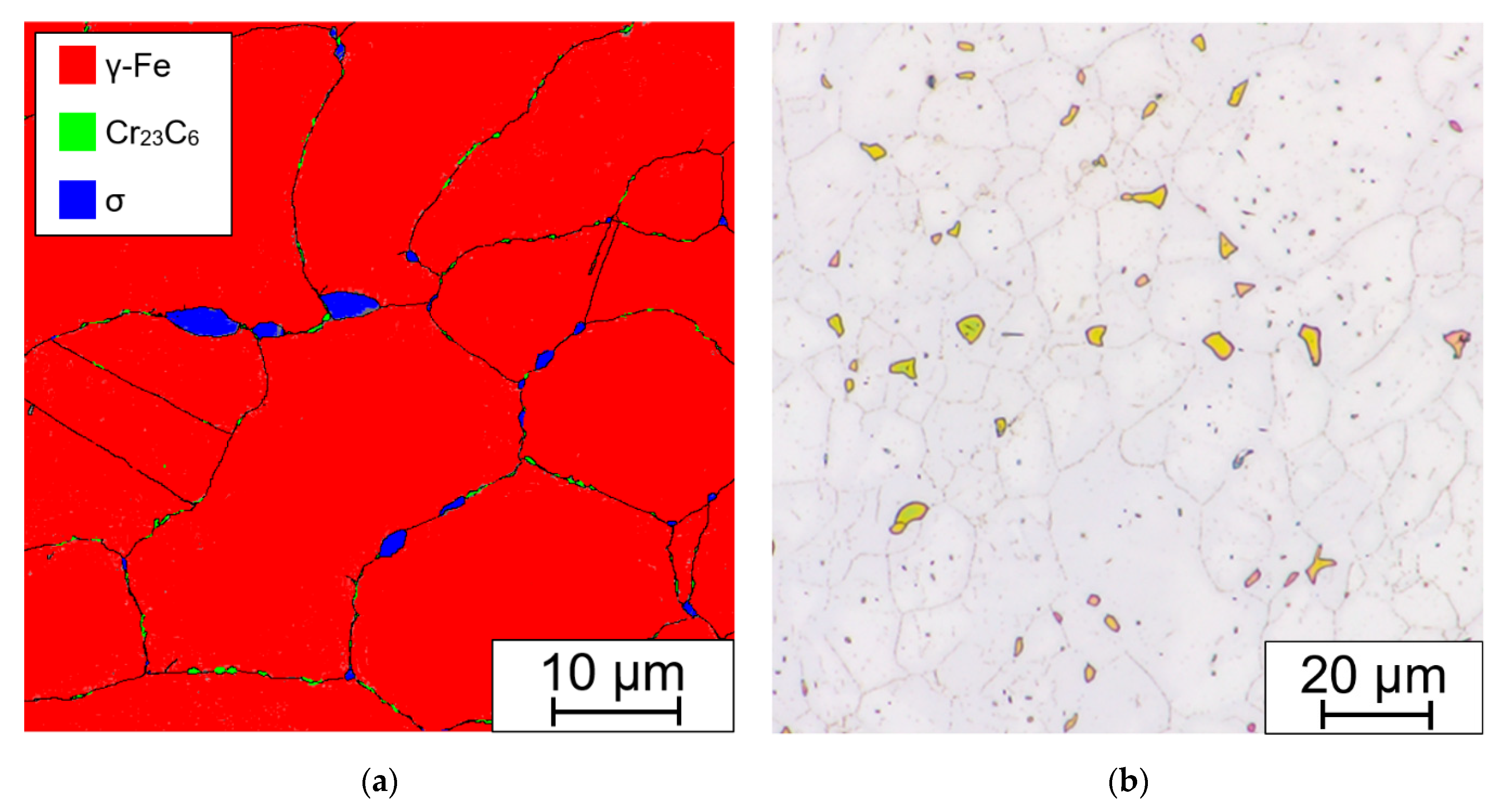
4.6. Microstructure
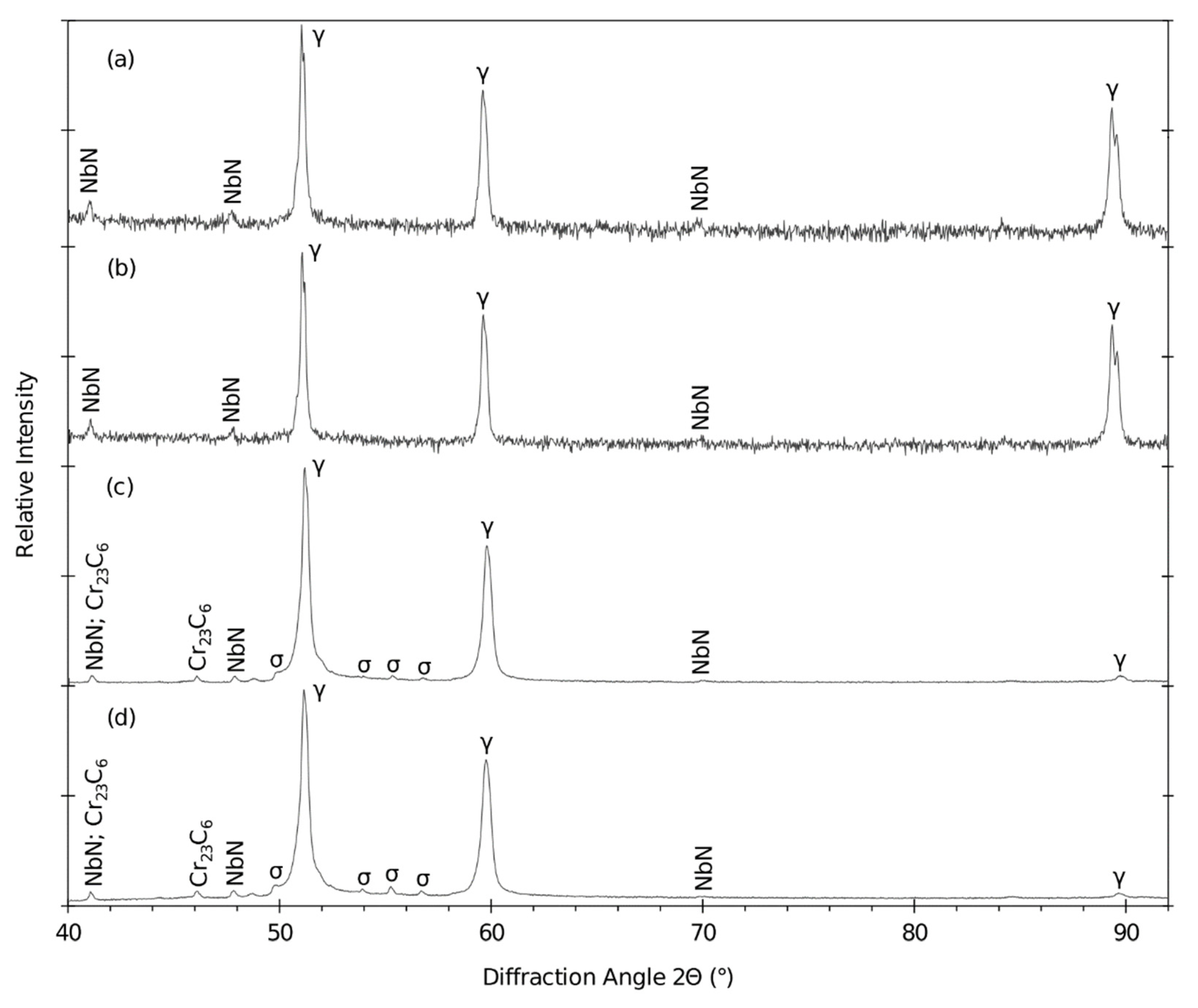
4.7. XRPD and EBSD
5. Discussion
6. Conclusions
- Hardness varies between the tubes’ wall in the as received state despite heat treatment after rolling. The hardness is higher for the outer and inner surfaces (approx. 200 HV10) and lower in the middle of the wall thickness (160 HV10).
- The acceleration of the sigma phase growth is clearly caused by the plastic deformation after cold working. Solution annealing is then a crucial operation. The suggested parameters 1130 °C/15 with cooling in water were fully sufficient.
- Isotermic aging compared to operation conditions (note that the exposure time in the power plant also includes planned outage periods) shows only slight differences in hardness (approximately 20 HV10 in the middle of the wall thickness) and other observed properties. The short period of exceeding the 700 °C temperature during the service operation had no major impact on the stability of SUPER304H steel.
- The suggested solution annealing parameters of 1130 °C/15 min/water were fully sufficient for the treatment of the bent samples, but in the case of the weld joints, it caused unfavorable grain growth close to the HAZ root area.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Viswanathan, R.; Purgert, R.; Goodstine, S.U.S. Program on Materials Technology for USC Coal-Fired Boilers. In Proceedings of the Advances in Materials Technology for Fossil Power Plants; ASM International: Marco Island, FL, USA, 2008; Volume 5, p. 16. [Google Scholar]
- Vinoth Kumar, M.; Balasubramanian, V.; Gourav Rao, A. Effect of Filler Addition on Solidification Behaviour and Hot Tensile Properties of GTA-Welded Tube Joints of Super 304H Austenitic Stainless Steel. Int. J. Mech. Mater. Eng. 2015, 10, 25. [Google Scholar] [CrossRef]
- da Fonseca, G.S.; de Oliveira, P.M.; Diniz, M.G.; Bubnoff, D.V.; Castro, J.A. de Sigma Phase in Superduplex Stainless Steel: Formation, Kinetics and Microstructural Path. Mat. Res. 2017, 20, 249–255. [Google Scholar] [CrossRef]
- Tang, B.; Zhu, L. Precipitation Behaviour of Z-Phase in Domestic S30432 Steel at High-Temperature and Stress. Mater. Mech. Eng. 2015, 39. [Google Scholar]
- Le, T.G.; Yoon, K.B.; Ma, Y.W. Metal Temperature Estimation and Microstructure Evaluation of Long-Term Service-Exposed SUPER304H Steel Boiler Tubes. Met. Mater. Int. 2020, 27, 5121–5132. [Google Scholar] [CrossRef]
- Zhou, Q.; Liu, J.; Gao, Y. An Insight into Oversaturated Deformation-Induced Sigma Precipitation in SUPER304H Austenitic Stainless Steel. Mater. Des. 2019, 181, 108056. [Google Scholar] [CrossRef]
- Golański, G.; Zieliński, A.; Purzyńska, H. Precipitation Processes in Creep-Resistant Austenitic Steels. In Austenitic Stainless Steels—New Aspects; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Sakai, K.; Morita, S.; Yamamoto, T.; Tsumura, T. Design and Operating Experience of the Latest 1,000-MW Coal-Fired Boiler. Hitachi Rev. 1998, 47, 5. [Google Scholar]
- Ni, J.; Li, H.; Li, M.; Wang, Z. Research on Microstructure Transformation of SUPER304H Stainless Steel in the Process of Aging at 700; Atlantis Press: Dordrecht, The Netherlands, 2016; pp. 1974–1978. [Google Scholar] [CrossRef]
- Pomikálek, L.; Hermanová, Š.; Dobrovodská, L. Effect of Welding on the Properties of HR3C, Super 304H, TP347HFG and P92 Steels; Tanger: Brno, Czech Republic, 2013; Volume 22, p. 6. [Google Scholar]
- Liang, Z.; Zhao, Q.; Deng, J.; Wang, Y. Influence of Aging Treatment on the Microstructure and Mechanical Properties of T92/Super 304H Dissimilar Metal Welds. Mater. High Temp. 2018, 35, 327–334. [Google Scholar] [CrossRef]
- Sumitomo Metal Industries. Inspection Certificate; Sumitomo Metal Industries: Amgasaki, Japan, 2012. [Google Scholar]
- Voestalpine Böhler Welding. Böhler Welding Product Catalogue. Available online: http://www.voestalpine.com/welding (accessed on 28 November 2021).
- Beausir, B.; Fundenberger, J.-J. Analysis Tools for Electron and X-ray Diffraction, ATEX—Software; Université de Lorraine—Metz, France. 2017. Available online: www.atex-software.eu (accessed on 28 November 2021).











| Material | C | Si | Mn | P | S | Cu | Cr | Ni | Nb | N | B | Al |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SUPER304H | 0.03 | <0.3 | <1.0 | <0.040 | <0.010 | 3.0 | 18.0 | 9.0 | 0.45 | 0.85 | 0.005 | 0.017 |
| Thermanit 304 H Cu | 0.1 | 0.4 | 3.2 | - * | - | 3.0 | 18.0 | 16.0 | 0.4 | 0.2 | - | - |
| Layer | Welding Speed (mm/min) | Current (A) | Arc Voltage (V) | Preheat/Interpass (°C) | Arc Energy (J/mm) | Heat Input * (J/mm) |
|---|---|---|---|---|---|---|
| 1 | 33.0 | 99.6 | 8.8 | 14 | 1597 | 958 |
| 2 | 28.8 | 89.3 | 9.5 | 128 | 1767 | 1060 |
| 3 | 25.8 | 86.5 | 9.5 | 148 | 1911 | 1147 |
| Mean Lineal Length L (Mm) | After Bending | 650 °C/7560 h | 650 °C/15,000 h | 650 °C/18,500 h |
|---|---|---|---|---|
| Direction 0° | 0.0116 | 0.0143 | 0.0082 | 0.0082 |
| Direction 90° | 0.0112 | 0.0128 | 0.0087 | 0. 0087 |
| Avg number G | 9.5 | 9.5 | 9.5 | 10.5 |
| Location | BM-Left | HAZ-Left | HAZ-Right | BM-Right | ||
|---|---|---|---|---|---|---|
| Further Area | FL | FL | Further Area | |||
| crown | 6.5 | 6 | 5.5 | not visible | 6 | 6 |
| center | 6.5 | 7 | 2 | 3 | 5 | 6.5 |
| root | 6.5 | 6.5 | 3 | not visible | 3 (4) | 7 |
| Location | BM-Left | HAZ-Left | HAZ-Right | BM-Right | ||
|---|---|---|---|---|---|---|
| Further Area | FL | FL | Further Area | |||
| crown | 7.5 (1.5) | 7 | 6.5 | not visible | 6.5 | 7.5 |
| center | 6.5 | 6.5 | 5 | 5 | 6.5 | 7.5 |
| root | 7.5 (6) | 1 | 3 | 4 | not visible (4) | 8 |
| Location | BM-Left | HAZ-Left | HAZ-Right | BM-Right | ||
|---|---|---|---|---|---|---|
| Further Area | FL | FL | Further Area | |||
| crown | 8.5 | 7 | 5 | 6 | 7 | 8 |
| center | 8.5 | 6.5 | 5 | 4.5 | 6 | 8.5 |
| root | 8.5 | 7 | 3.5 | 3.5 | 5.5 | 8 |
| Location | BM-Left | HAZ-Left | HAZ-Right | BM-Right | ||
|---|---|---|---|---|---|---|
| Further Area | FL | FL | Further Area | |||
| crown | 8 | 6.5 | 6.5 | 6.5 | 5 | 7 (3.5) |
| center | 7.5 | 4 * | 2 | 3.5 | 5 | 7.5 |
| root | 7.5 (6) | 4.5 (1) | not visible | 4 | 1 | 7.5 |
| Sample | Phase | Weight Fraction (%) | Lattice Parameter (Å) |
|---|---|---|---|
| N-650 °C-7560 h | Austenite | 93.24 | a = 3.604 |
| NbN | 1.13 | a = 4.436 | |
| Cr23C6 | 0.78 | a = 10.619 | |
| Fe-Cr sigma | 4.85 | a = 8.806 c = 4.581 | |
| N-650 °C-15,000 h | Austenite | 93.24 | a = 3.604 |
| NbN | 1.13 | a = 4.439 | |
| Cr23C6 | 0.78 | a = 10.62 | |
| Fe-Cr sigma | 4.85 | a = 8.810 c = 4.585 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pilsová, L.; Horváth, J.; Mára, V. Structural Stability of the SUPER304H Steel Used in Energetics. Materials 2022, 15, 455. https://doi.org/10.3390/ma15020455
Pilsová L, Horváth J, Mára V. Structural Stability of the SUPER304H Steel Used in Energetics. Materials. 2022; 15(2):455. https://doi.org/10.3390/ma15020455
Chicago/Turabian StylePilsová, Lucie, Jakub Horváth, and Vladimír Mára. 2022. "Structural Stability of the SUPER304H Steel Used in Energetics" Materials 15, no. 2: 455. https://doi.org/10.3390/ma15020455
APA StylePilsová, L., Horváth, J., & Mára, V. (2022). Structural Stability of the SUPER304H Steel Used in Energetics. Materials, 15(2), 455. https://doi.org/10.3390/ma15020455






