Recent Advances in Metal-Based Nanoparticle-Mediated Biological Effects in Arabidopsis thaliana: A Mini Review
Abstract
:1. Introduction
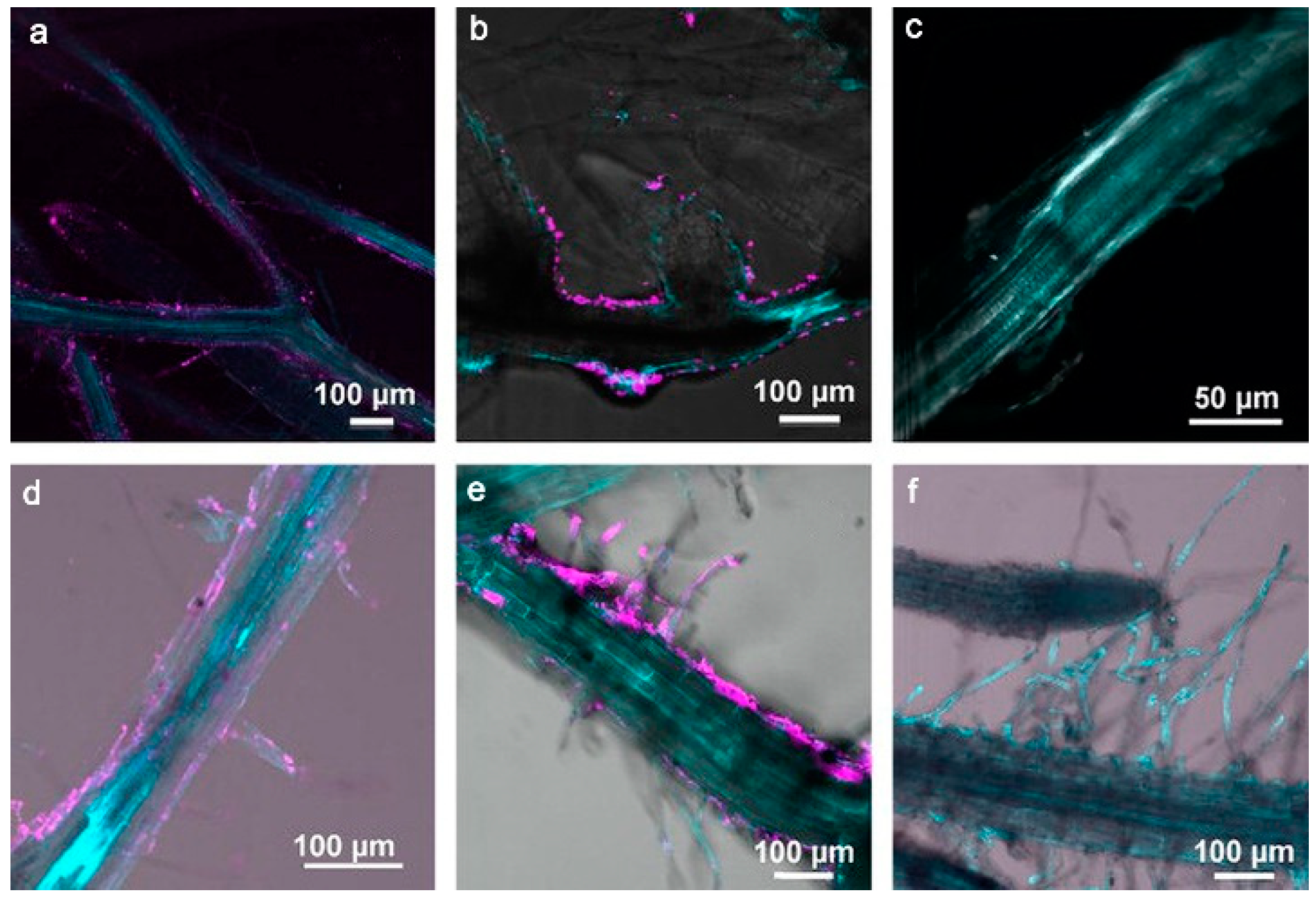
2. Absorption and Transport of MNPs in Arabidopsis thaliana
2.1. Absorption and Transport of Monometallic Nanoparticles in Arabidopsis thaliana
2.2. Absorption and Transport of Metal Oxide Nanoparticles in Arabidopsis thaliana
2.3. Absorption and Transport of Other Metal-Based Nanoparticles in Arabidopsis thaliana
3. Phytotoxic Effects of MNPs on Arabidopsis thaliana
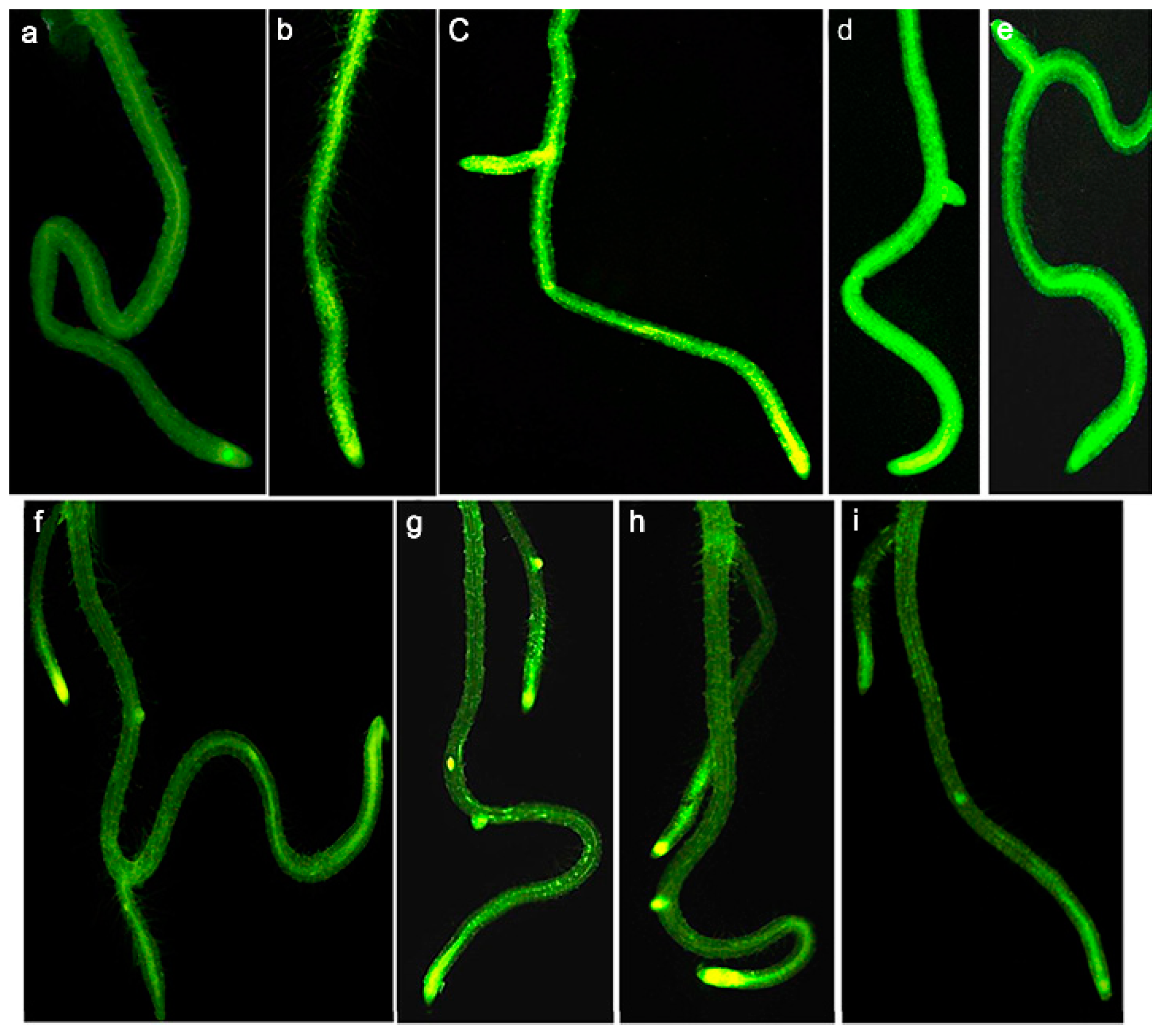
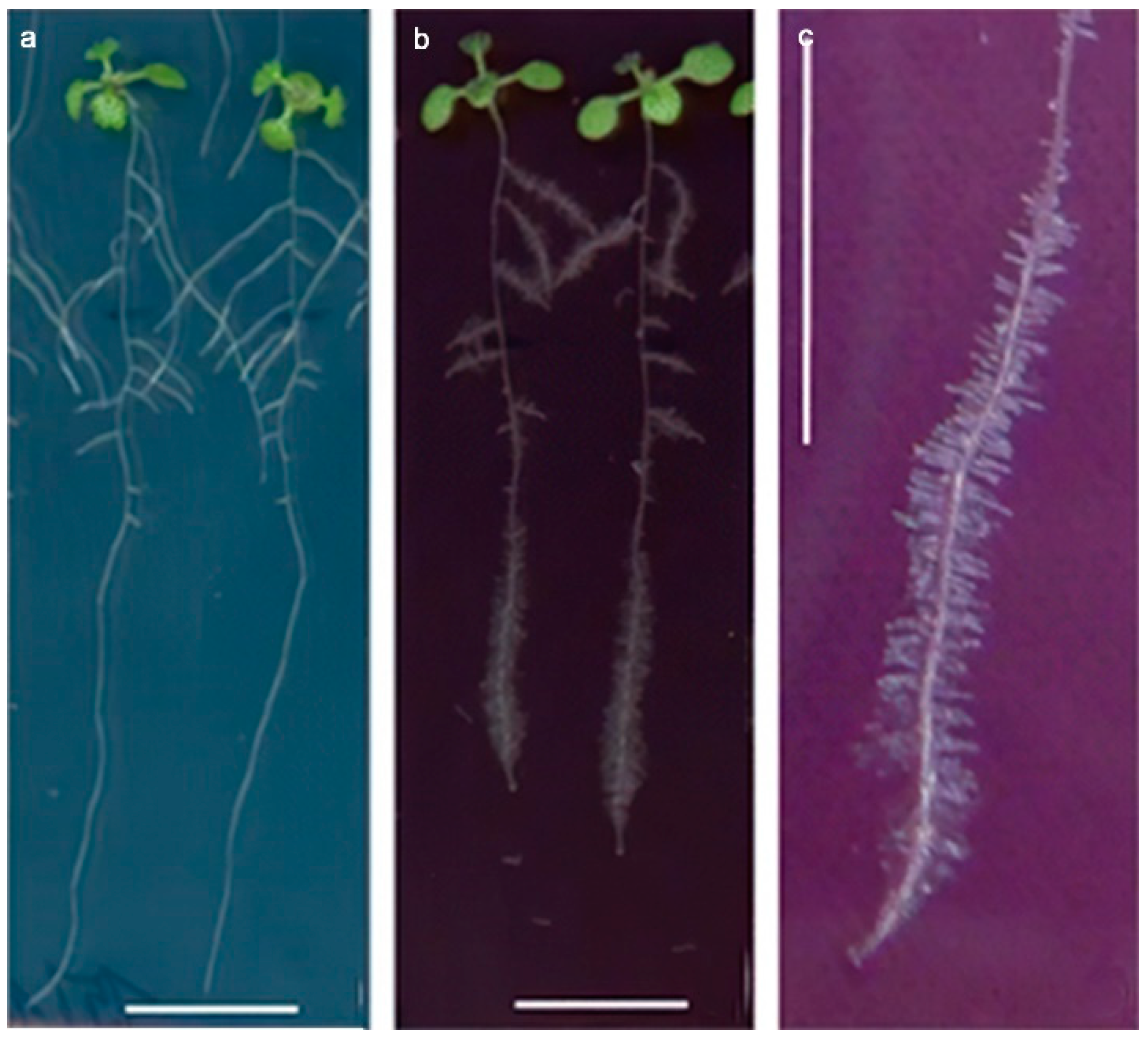
3.1. Toxic Effects at the Morphological Level
3.2. Toxic Effects at the Physiological Level
3.3. Toxic Effects at the Molecular Level
4. Conclusions and Outlook
- (i)
- Currently available literature that investigated the effects of MNPs on Arabidopsis used experimental settings that differed considerably from actual environment settings. For instance, the medium used was water or sandy soil rather than actual soil. The treatment time was comparatively shorter than that observed in nature, and Arabidopsis was exposed in a relatively single manner to MNPs, i.e., the roots were mainly exposed to the soil in the presence of MNPs, and thus, the effects of using leaf sprays or hydroponics remain to be determined.
- (ii)
- The physicochemical properties, treatments and growth stages of the MNPs used in the experiments differed among studies. Therefore, the source of nanomaterials, preparation methods, testing equipment selection and design of exposure conditions should be standardized.
- (iii)
- The internalization of metal ions produces different levels of toxicity to plants than direct ingestion of metal ions. The toxic effect seems to be concentration-dependent. Further, as the toxic effects from MNPs could be from the released metal ions alone or the combined effect of nanoparticles themselves, the source of toxicity should be identified.
- (iv)
- As studies at different levels of the plant’s response, i.e., subcellular, physiological and biochemical levels, are being performed, the interactions between MNPs and Arabidopsis should be designed in a way that combines the traditional toxicological research methods with histological techniques (transcriptomics, metabolomics and proteomics) to provide more accurate and in-depth elucidation of the mechanisms of MNP-based phytotoxicity.
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rocha-Santos, T.A.P. Sensors and biosensors based on magnetic nanoparticles. TrAC Trends Anal. Chem. 2014, 62, 28–36. [Google Scholar] [CrossRef]
- Prashant, K.J.; Huang, X.; Ivan, H.E.-S.; Mostafa, A.E.-S. Noble metals on the nanoscale: Optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Acc. Chem. Res. 2007, 41, 1578–1586. [Google Scholar]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huh, A.J.; Kwon, Y.J. “Nanoantibiotics”: A new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. J. Control. Release 2011, 156, 128–145. [Google Scholar] [CrossRef]
- Wu, J.; Wang, X.; Wang, Q.; Lou, Z.; Li, S.; Zhu, Y.; Qin, L.; Wei, H. Nanomaterials with enzyme-like characteristics (nanozymes): Next-generation artificial enzymes (II). Chem. Soc. Rev. 2019, 48, 1004–1076. [Google Scholar] [CrossRef]
- Tan, C.; Cao, X.; Wu, X.-J.; He, Q.; Yang, J.; Zhang, X.; Chen, J.; Zhao, W.; Han, S.; Nam, G.-H.; et al. Recent advances in ultrathin two-dimensional nanomaterials. Chem. Rev. 2017, 117, 6225–6331. [Google Scholar] [CrossRef]
- Haghighi, M.; Teixeira da Silva, J.A. The effect of N-TiO2 on tomato, onion, and radish seed germination. J. Crop Sci. Biotechnol. 2014, 17, 221–227. [Google Scholar] [CrossRef]
- Zhang, Z.; Ke, M.; Qu, Q.; Peijnenburg, W.; Lu, T.; Zhang, Q.; Ye, Y.; Xu, P.; Du, B.; Sun, L.; et al. Impact of copper nanoparticles and ionic copper exposure on wheat (Triticum aestivum L.) root morphology and antioxidant response. Environ. Pollut. 2018, 239, 689–697. [Google Scholar] [CrossRef]
- Lee, C.W.; Mahendra, S.; Zodrow, K.; Li, D.; Tsai, Y.-C.; Braam, J.; Alvarez, P.J. Alvarez. developmental phytotoxicity of metal oxide nanoparticles to Arabidopsis thaliana. Environ. Toxicol. Chem. 2010, 29, 669–675. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, H.; Zheng, X.; Mu, H. The impacts of silver nanoparticles and silver ions on wastewater biological phosphorous removal and the mechanisms. J. Hazard. Mater. 2012, 239–240, 88–94. [Google Scholar] [CrossRef]
- Zhu, H.; Han, J.; Xiao, J.Q.; Jin, Y. Uptake, translocation, and accumulation of manufactured iron oxide nanoparticles by pumpkin plants. J. Environ. Monit. 2008, 10, 713–717. [Google Scholar] [CrossRef]
- Kurepa, J.; Paunesku, T.; Vogt, S.; Arora, H.; Rabatic, B.M.; Lu, J.; Wanzer, M.B.; Woloschak, G.E.; Smalle, J.A. Uptake and distribution of ultrasmall anatase TiO2 Alizarin red S nanoconjugates in Arabidopsis thaliana. Nano Lett. 2010, 10, 2296–2302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corredor, E.; Testillano, P.S.; Coronado, M.J.; Gonzalez-Melendi, P.; Fernandez-Pacheco, R.; Marquina, C.; Ibarra, M.R.; de la Fuente, J.M.; Rubiales, D.; Perez-de-Luque, A.; et al. Nanoparticle penetration and transport in living pumpkin plants: In situ subcellular identification. BMC Plant Biol. 2009, 9, 45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shams, G.; Ranjbar, M.; Amiri, A. Effect of silver nanoparticles on concentration of silver heavy element and growth indexes in cucumber (Cucumis sativus L. negeen). J. Nanopart. Res. 2013, 15, 1630. [Google Scholar] [CrossRef]
- Daohui Lin, B.X. Root Uptake and phytotoxicity of ZnO nanoparticles. Environ. Sci. Technol. 2008, 42, 5580–5585. [Google Scholar]
- Song, U.; Jun, H.; Waldman, B.; Roh, J.; Kim, Y.; Yi, J.; Lee, E.J. Functional analyses of nanoparticle toxicity: A comparative study of the effects of TiO2 and Ag on tomatoes (Lycopersicon esculentum). Ecotoxicol. Environ. Saf. 2013, 93, 60–67. [Google Scholar] [CrossRef]
- Zhao, L.; Sun, Y.; Hernandez-Viezcas, J.A.; Servin, A.D.; Hong, J.; Niu, G.; Peralta-Videa, J.R.; Duarte-Gardea, M.; Gardea-Torresdey, J.L. Influence of CeO2 and ZnO nanoparticles on cucumber physiological markers and bioaccumulation of Ce and Zn: A life cycle study. J. Agric. Food Chem. 2013, 61, 11945–11951. [Google Scholar] [CrossRef]
- Matthed, D.; Whiteside, K.K.T.; Peter, R.A. The brighter side of soils: Quantum dots track organic nitrogen through fungi and plants. Ecology 2009, 90, 100–108. [Google Scholar]
- Ruttkay-Nedecky, B.; Krystofova, O.; Nejdl, L.; Adam, V. Nanoparticles based on essential metals and their phytotoxicity. J. Nanobiotechnol. 2017, 15, 33. [Google Scholar] [CrossRef]
- Khodakovskaya, M.V.; de Silva, K.; Nedosekin, D.A.; Dervishi, E.; Biris, A.S.; Shashkov, E.V.; Galanzha, E.I.; Zharov, V.P. Complex genetic, photothermal, and photoacoustic analysis of nanoparticle-plant interactions. Proc. Natl. Acad. Sci. USA 2011, 108, 1028–1033. [Google Scholar] [CrossRef] [Green Version]
- Zhai, G.; Walters, K.S.; Peate, D.W.; Alvarez, P.J.; Schnoor, J.L. Transport of gold nanoparticles through plasmodesmata and precipitation of gold ions in woody poplar. Environ. Sci. Technol. Lett. 2014, 1, 146–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, A.F.; Rylott, E.L.; Anderson, C.W.; Bruce, N.C. Investigating the toxicity, uptake, nanoparticle formation and genetic response of plants to gold. PLoS ONE 2014, 9, e93793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rico, C.M.; Majumdar, S.; Duarte-Gardea, M.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Interaction of nanoparticles with edible plants and their possible implications in the food chain. J. Agric. Food Chem. 2011, 59, 3485–3498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kohan-Baghkheirati, E.; Geisler-Lee, J. Gene expression, protein function and pathways of Arabidopsis thaliana responding to silver nanoparticles in comparison to silver ions, cold, salt, drought, and heat. Nanomaterials 2015, 5, 436–467. [Google Scholar] [CrossRef]
- Katsumiti, A.; Gilliland, D.; Arostegui, I.; Cajaraville, M.P. Mechanisms of toxicity of Ag nanoparticles in comparison to bulk and ionic Ag on mussel hemocytes and gill cells. PLoS ONE 2015, 10, e0129039. [Google Scholar] [CrossRef]
- Scherer, M.D.; Sposito, J.C.V.; Falco, W.F.; Grisolia, A.B.; Andrade, L.H.C.; Lima, S.M.; Machado, G.; Nascimento, V.A.; Goncalves, D.A.; Wender, H.; et al. Cytotoxic and genotoxic effects of silver nanoparticles on meristematic cells of Allium cepa roots: A close analysis of particle size dependence. Sci. Total Environ. 2019, 660, 459–467. [Google Scholar] [CrossRef]
- Koo, Y.; Lukianova-Hleb, E.Y.; Pan, J.; Thompson, S.M.; Lapotko, D.O.; Braam, J. In planta response of arabidopsis to photothermal impact mediated by gold nanoparticles. Small 2016, 12, 623–630. [Google Scholar] [CrossRef]
- Geisler-Lee, J.; Wang, Q.; Yao, Y.; Zhang, W.; Geisler, M.; Li, K.; Huang, Y.; Chen, Y.; Kolmakov, A.; Ma, X. Phytotoxicity, accumulation and transport of silver nanoparticles by Arabidopsis thaliana. Nanotoxicology 2013, 7, 323–337. [Google Scholar] [CrossRef]
- Avellan, A.; Schwab, F.; Masion, A.; Chaurand, P.; Borschneck, D.; Vidal, V.; Rose, J.; Santaella, C.; Levard, C. Nanoparticle uptake in plants: Gold nanomaterial localized in roots of Arabidopsis thaliana by X-ray computed nanotomography and hyperspectral imaging. Environ. Sci. Technol. 2017, 51, 8682–8691. [Google Scholar] [CrossRef] [Green Version]
- Milewska-Hendel, A.; Zubko, M.; Stroz, D.; Kurczynska, E.U. Effect of nanoparticles surface charge on the Arabidopsis thaliana (L.) roots development and their movement into the root cells and protoplasts. Int. J. Mol. Sci. 2019, 20, 1650. [Google Scholar] [CrossRef] [Green Version]
- Milani, N.; McLaughlin, M.J.; Stacey, S.P.; Kirby, J.K.; Hettiarachchi, G.M.; Beak, D.G.; Cornelis, G. Dissolution kinetics of macronutrient fertilizers coated with manufactured zinc oxide nanoparticles. J. Agric. Food Chem. 2012, 60, 3991–3998. [Google Scholar] [CrossRef] [PubMed]
- Mir, A.H.; Qamar, A.; Qadir, I.; Naqvi, A.H.; Begum, R. Accumulation and trafficking of zinc oxide nanoparticles in an invertebrate model, Bombyx mori, with insights on their effects on immuno-competent cells. Sci. Rep. 2020, 10, 1617. [Google Scholar] [CrossRef] [PubMed]
- Bafaro, E.; Liu, Y.; Xu, Y.; Dempski, R.E. The emerging role of zinc transporters in cellular homeostasis and cancer. Signal Transduct. Target. Ther. 2017, 2, 17029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, A.; Sinilal, B.; Dhandapani, G.; Meagher, R.B.; Sahi, S.V. Effects of deficiency and excess of zinc on morphophysiological traits and spatiotemporal regulation of zinc-responsive genes reveal incidence of cross talk between micro- and macronutrients. Environ. Sci. Technol. 2013, 47, 5327–5335. [Google Scholar] [CrossRef]
- Hussain, D.; Haydon, M.J.; Wang, Y.; Wong, E.; Sherson, S.M.; Young, J.; Camakaris, J.; Harper, J.F.; Cobbett, C.S. P-type ATPase heavy metal transporters with roles in essential zinc homeostasis in Arabidopsis. Plant Cell 2004, 16, 1327–1339. [Google Scholar] [CrossRef] [Green Version]
- Nair, P.M.G.; Chung, I.M. Regulation of morphological, molecular and nutrient status in Arabidopsis thaliana seedlings in response to ZnO nanoparticles and Zn ion exposure. Sci. Total Environ. 2017, 575, 187–198. [Google Scholar] [CrossRef]
- Jia, H.; Chen, S.; Wang, X.; Shi, C.; Liu, K.; Zhang, S.; Li, J. Copper oxide nanoparticles alter cellular morphology via disturbing the actin cytoskeleton dynamics in Arabidopsis roots. Nanotoxicology 2020, 14, 127–144. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, L.; Zhao, J.; Wang, X.; White, J.C.; Xing, B. CuO Nanoparticle interaction with Arabidopsis thaliana: Toxicity, parent-progeny transfer, and gene expression. Environ. Sci. Technol. 2016, 50, 6008–6016. [Google Scholar] [CrossRef]
- Hayat, S.; Pichtel, J.; Faizan, M. Sustainable agriculture reviews 41 nanotechnology for plant growth and development. In Nanotechnology for Plant Growth and Development; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Garcia-Sanchez, S.; Bernales, I.; Cristobal, S. Early response to nanoparticles in the Arabidopsis transcriptome compromises plant defence and root-hair development through salicylic acid signalling. BMC Genom. 2015, 16, 341. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Kurepa, J.; Smalle, J.A. Ultra-small TiO2 nanoparticles disrupt microtubular networks in Arabidopsis thaliana. Plant Cell Environ. 2011, 34, 811–820. [Google Scholar] [CrossRef]
- Ma, C.; Chhikara, S.; Xing, B.; Musante, C.; White, J.C.; Dhankher, O.P. Physiological and molecular response of Arabidopsis thaliana (L.) to nanoparticle cerium and indium oxide exposure. ACS Sustain. Chem. Eng. 2013, 1, 768–778. [Google Scholar] [CrossRef]
- Birbaum, K.; Brogioli, R.; Schellenberg, M.; Martinoia, E.; Wendelin, E.; Stark, J.W.; Detlef, G.; Ludwig, K.L. No evidence for cerium dioxide nanoparticle translocation in maize plants. Environ. Sci. Technol. 2010, 44, 8718–8723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martha, L.L.-M.; Rose, G.D.L.; Jose, A.H.-V.; Castillo-Michel, H.; Botez, C.E.; Jose, R.P.-V.; Jorge, L.G.-T. Evidence of the differential biotransformation and genotoxicity of ZnO and CeO2 nanoparticles on soybean (Glycine max) plants. Environ. Sci. Technol. 2010, 44, 7315–7320. [Google Scholar]
- Priester, J.H.; Ge, Y.; Mielke, R.E.; Horst, A.M.; Moritz, S.C.; Espinosa, K.; Gelb, J.; Walker, S.L.; Nisbet, R.M.; An, Y.J.; et al. Soybean susceptibility to manufactured nanomaterials with evidence for food quality and soil fertility interruption. Proc. Natl. Acad. Sci. USA 2012, 109, E2451–E2456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, P.; Ma, Y.; Zhang, Z.; He, X.; Zhang, J.; Guo, Z.; Tai, R.; Zhao, Y.; Chai, Z. Biotransformation of ceria nanoparticles in cucumber plants. ACS Nano 2012, 6, 9943–9950. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zhao, C.; Qian, K.; Sun, M.; Hao, Y.; Han, L.; Wang, C.; Ma, C.; White, J.C.; Xing, B. Physiological responses of pumpkin to zinc oxide quantum dots and nanoparticles. Environ. Pollut. 2022, 296, 118723. [Google Scholar] [CrossRef]
- Kolackova, M.; Moulick, A.; Kopel, P.; Dvorak, M.; Adam, V.; Klejdus, B.; Huska, D. Antioxidant, gene expression and metabolomics fingerprint analysis of Arabidopsis thaliana treated by foliar spraying of ZnSe quantum dots and their growth inhibition of Agrobacterium tumefaciens. J. Hazard. Mater. 2019, 365, 932–941. [Google Scholar] [CrossRef]
- Navarro, D.A.; Bisson, M.A.; Aga, D.S. Investigating uptake of water-dispersible CdSe/ZnS quantum dot nanoparticles by Arabidopsis thaliana plants. J. Hazard. Mater. 2012, 211–212, 427–435. [Google Scholar] [CrossRef]
- Laaveri, T.; Sterne, J.; Rombo, L.; Kantele, A. Systematic review of loperamide: No proof of antibiotics being superior to loperamide in treatment of mild/moderate travellers’ diarrhoea. Travel Med. Infect. Dis. 2016, 14, 299–312. [Google Scholar] [CrossRef]
- Shen, Y.; Borgatta, J.; Ma, C.; Singh, G.; Tamez, C.; Schultes, N.P.; Zhang, Z.; Dhankher, O.P.; Elmer, W.H.; He, L.; et al. Role of foliar biointerface properties and nanomaterial chemistry in controlling Cu transfer into wild-type and mutant Arabidopsis thaliana leaf tissue. J. Agric. Food Chem. 2022, 70, 4267–4278. [Google Scholar] [CrossRef]
- He, J.; Zhang, L.; He, S.Y.; Ryser, E.T.; Li, H.; Zhang, W. Stomata facilitate foliar sorption of silver nanoparticles by Arabidopsis thaliana. Environ. Pollut. 2022, 292, 118448. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Hashmi, S.S.; Palma, J.M.; Corpas, F.J. Influence of metallic, metallic oxide, and organic nanoparticles on plant physiology. Chemosphere 2022, 290, 133329. [Google Scholar] [CrossRef] [PubMed]
- Sosan, A.; Svistunenko, D.; Straltsova, D.; Tsiurkina, K.; Smolich, I.; Lawson, T.; Subramaniam, S.; Golovko, V.; Anderson, D.; Sokolik, A.; et al. Engineered silver nanoparticles are sensed at the plasma membrane and dramatically modify the physiology of Arabidopsis thaliana plants. Plant J. 2016, 85, 245–257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, T.; Wu, J.; Xu, S.; Deng, C.; Wu, L.; Wu, Y.; Bian, P. A potential involvement of plant systemic response in initiating genotoxicity of Ag-nanoparticles in Arabidopsis thaliana. Ecotoxicol. Environ. Saf. 2019, 170, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Bao, D.; Oh, Z.G.; Chen, Z. Characterization of silver nanoparticles internalized by arabidopsis plants using single particle ICP-MS analysis. Front. Plant Sci. 2016, 7, 32. [Google Scholar] [CrossRef] [Green Version]
- Ke, M.; Qu, Q.; Peijnenburg, W.; Li, X.; Zhang, M.; Zhang, Z.; Lu, T.; Pan, X.; Qian, H. Phytotoxic effects of silver nanoparticles and silver ions to Arabidopsis thaliana as revealed by analysis of molecular responses and of metabolic pathways. Sci. Total Environ. 2018, 644, 1070–1079. [Google Scholar] [CrossRef]
- Wang, L.; Sun, J.; Lin, L.; Fu, Y.; Alenius, H.; Lindsey, K.; Chen, C. Silver nanoparticles regulate Arabidopsis root growth by concentration-dependent modification of reactive oxygen species accumulation and cell division. Ecotoxicol. Environ. Saf. 2020, 190, 110072. [Google Scholar] [CrossRef]
- Ke, M.; Li, Y.; Qu, Q.; Ye, Y.; Peijnenburg, W.; Zhang, Z.; Xu, N.; Lu, T.; Sun, L.; Qian, H. Offspring toxicity of silver nanoparticles to Arabidopsis thaliana flowering and floral development. J. Hazard. Mater. 2020, 386, 121975. [Google Scholar] [CrossRef]
- Sun, J.; Wang, L.; Li, S.; Yin, L.; Huang, J.; Chen, C. Toxicity of silver nanoparticles to Arabidopsis: Inhibition of root gravitropism by interfering with auxin pathway. Environ. Toxicol. Chem. 2017, 36, 2773–2780. [Google Scholar] [CrossRef]
- Siegel, J.; Zaruba, K.; Svorcik, V.; Kroumanova, K.; Burketova, L.; Martinec, J. Round-shape gold nanoparticles: Effect of particle size and concentration on Arabidopsis thaliana root growth. Nanoscale Res. Lett. 2018, 13, 95. [Google Scholar] [CrossRef] [Green Version]
- Kumar, V.; Guleria, P.; Kumar, V.; Yadav, S.K. Gold nanoparticle exposure induces growth and yield enhancement in Arabidopsis thaliana. Sci. Total Environ. 2013, 461–462, 462–468. [Google Scholar] [CrossRef] [PubMed]
- Sperdouli, I.; Moustaka, J.; Antonoglou, O.; Adamakis, I.S.; Dendrinou-Samara, C.; Moustakas, M. Leaf age-dependent effects of foliar-sprayed CuZn nanoparticles on photosynthetic efficiency and ROS generation in Arabidopsis thaliana. Materials 2019, 12, 2498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vankova, R.; Landa, P.; Podlipna, R.; Dobrev, P.I.; Prerostova, S.; Langhansova, L.; Gaudinova, A.; Motkova, K.; Knirsch, V.; Vanek, T. ZnO nanoparticle effects on hormonal pools in Arabidopsis thaliana. Sci. Total Environ. 2017, 593–594, 535–542. [Google Scholar] [CrossRef]
- Khan, A.R.; Wakeel, A.; Muhammad, N.; Liu, B.; Wu, M.; Liu, Y.; Ali, I.; Zaidi, S.H.R.; Azhar, W.; Song, G.; et al. Involvement of ethylene signaling in zinc oxide nanoparticle-mediated biochemical changes in Arabidopsis thaliana leaves. Environ. Sci. Nano 2019, 6, 341–355. [Google Scholar] [CrossRef]
- Landa, P.; Prerostova, S.; Petrova, S.; Knirsch, V.; Vankova, R.; Vanek, T. The Transcriptomic response of Arabidopsis thaliana to zinc oxide: A comparison of the impact of nanoparticle, bulk, and ionic zinc. Environ. Sci. Technol. 2015, 49, 14537–14545. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; He, R.; Zhao, J.; Nie, G.; Xu, L.; Xing, B. Oxidative stress-induced toxicity of CuO nanoparticles and related toxicogenomic responses in Arabidopsis thaliana. Environ. Pollut. 2016, 212, 605–614. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azhar, W.; Khan, A.R.; Muhammad, N.; Liu, B.; Song, G.; Hussain, A.; Yasin, M.U.; Khan, S.; Munir, R.; Gan, Y. Ethylene mediates CuO NP-induced ultrastructural changes and oxidative stress in Arabidopsis thaliana leaves. Environ. Sci. Nano 2020, 7, 938–953. [Google Scholar] [CrossRef]
- Jia, H.; Yang, J.; Liu, H.; Liu, K.; Ma, P.; Chen, S.; Shi, W.; Wei, T.; Ren, X.; Guo, J.; et al. Hydrogen sulfide—Cysteine cycle plays a positive role in Arabidopsis responses to Copper Oxide nanoparticles stress. Environ. Exp. Bot. 2018, 155, 195–205. [Google Scholar] [CrossRef]
- Shull, T.E.; Kurepa, J.; Smalle, J.A. Anatase TiO2 nanoparticles induce autophagy and chloroplast degradation in thale cress (Arabidopsis thaliana). Environ. Sci. Technol. 2019, 53, 9522–9532. [Google Scholar] [CrossRef]
- Liu, H.; Ma, C.; Chen, G.; White, J.C.; Wang, Z.; Xing, B.; Dhankher, O.P. Titanium dioxide nanoparticles alleviate tetracycline toxicity to Arabidopsis thaliana (L.). ACS Sustain. Chem. Eng. 2017, 5, 3204–3213. [Google Scholar] [CrossRef]
- Yang, X.; Pan, H.; Wang, P.; Zhao, F.J. Particle-specific toxicity and bioavailability of cerium oxide (CeO2) nanoparticles to Arabidopsis thaliana. J. Hazard. Mater. 2017, 322, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Bombin, S.; LeFebvre, M.; Sherwood, J.; Xu, Y.; Bao, Y.; Ramonell, K.M. Developmental and reproductive effects of iron oxide nanoparticles in Arabidopsis thaliana. Int. J. Mol. Sci. 2015, 16, 24174–24193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dietz, K.J.; Herth, S. Plant nanotoxicology. Trends. Plant Sci. 2011, 16, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Aslani, F.; Bagheri, S.; Muhd Julkapli, N.; Juraimi, A.S.; Hashemi, F.S.; Baghdadi, A. Effects of engineered nanomaterials on plants growth: An overview. Sci. World J. 2014, 2014, 641759. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, D.K.; Tripathi, A.; Shweta; Singh, S.; Singh, Y.; Vishwakarma, K.; Yadav, G.; Sharma, S.; Singh, V.K.; Mishra, R.K.; et al. Uptake, Accumulation and toxicity of silver nanoparticle in autotrophic plants, and heterotrophic microbes: A concentric review. Front. Microbiol. 2017, 8, 7. [Google Scholar] [CrossRef]
- Paul, K.; Großkinsky, D.K.; Vass, I.; Roitsch, T. Silver nanoparticles affect Arabidopsis thaliana leaf tissue integrity and suppress pseudomonas syringae infection symptoms in a dose-dependent manner. BioNanoScience 2022, 12, 332–338. [Google Scholar] [CrossRef]
- Ferrari, E.; Barbero, F.; Busquets-Fite, M.; Franz-Wachtel, M.; Kohler, H.R.; Puntes, V.; Kemmerling, B. Growth-promoting gold nanoparticles decrease stress responses in arabidopsis seedlings. Nanomaterials 2021, 11, 3161. [Google Scholar] [CrossRef]
- Kaveh, R.; Li, Y.S.; Ranjbar, S.; Tehrani, R.; Brueck, C.L.; Van Aken, B. Changes in Arabidopsis thaliana gene expression in response to silver nanoparticles and silver ions. Environ. Sci. Technol. 2013, 47, 10637–10644. [Google Scholar] [CrossRef]
- Syu, Y.Y.; Hung, J.H.; Chen, J.C.; Chuang, H.W. Impacts of size and shape of silver nanoparticles on Arabidopsis plant growth and gene expression. Plant Physiol. Biochem. 2014, 83, 57–64. [Google Scholar] [CrossRef]
- Dai, Y.; Wang, Z.; Zhao, J.; Xu, L.; Xu, L.; Yu, X.; Wei, Y.; Xing, B. Interaction of cuo nanoparticles with plant cells: Internalization, oxidative stress, electron transport chain disruption, and toxicogenomic responses. Environ. Sci. Nano 2018, 5, 2269–2281. [Google Scholar] [CrossRef]
- Tumburu, L.; Andersen, C.P.; Rygiewicz, P.T.; Reichman, J.R. Phenotypic and genomic responses to titanium dioxide and cerium oxide nanoparticles in Arabidopsis germinants. Environ. Toxicol. Chem. 2015, 34, 70–83. [Google Scholar] [CrossRef] [PubMed]
- Francisco, J.; Romera, E.A. Iron-deficiency stress responses in cucumber (Cucumis sativus L.) roots. Plant Physiol. 1994, 105, 1133–1138. [Google Scholar]
- Kupper, H.; Kupper, F.; Spiller, M. Environmental relevance of heavy metal-substituted chlorophylls using the example of water plants. J. Exp. Bot. 1996, 47, 259–266. [Google Scholar] [CrossRef] [Green Version]
- Qian, H.; Peng, X.; Han, X.; Ren, J.; Sun, L.; Fu, Z. Comparison of the toxicity of silver nanoparticles and silver ions on the growth of terrestrial plant model Arabidopsis thaliana. J. Environ. Sci. 2013, 25, 1947–1956. [Google Scholar] [CrossRef]
- Yoon, H.; Kang, Y.G.; Chang, Y.S.; Kim, J.H. Effects of zerovalent iron nanoparticles on photosynthesis and biochemical adaptation of soil-grown Arabidopsis thaliana. Nanomaterials 2019, 9, 1543. [Google Scholar] [CrossRef] [Green Version]
- Yan, A.; Chen, Z. Impacts of silver nanoparticles on plants: A focus on the phytotoxicity and underlying mechanism. Int. J. Mol. Sci. 2019, 20, 1003. [Google Scholar] [CrossRef]
- Nair, P.M.; Chung, I.M. Physiological and molecular level effects of silver nanoparticles exposure in rice (Cucumis sativus L.) seedlings. Chemosphere 2014, 112, 105–113. [Google Scholar] [CrossRef]
- Sopjani, M.; Foller, M.; Haendeler, J.; Gotz, F.; Lang, F. Silver ion-induced suicidal erythrocyte death. J. Appl. Toxicol. 2009, 29, 531–536. [Google Scholar] [CrossRef]
- Wijnhoven, S.W.P.; Peijnenburg, W.J.G.M.; Herberts, C.A.; Hagens, W.I.; Oomen, A.G.; Heugens, E.H.W.; Roszek, B.; Bisschops, J.; Gosens, I.; Van De Meent, D.; et al. Nano-silver—A review of available data and knowledge gaps in human and environmental risk assessment. Nanotoxicology 2009, 3, 109–138. [Google Scholar] [CrossRef]
- Jin, Y.; Chen, S.; Fan, X.; Song, H.; Li, X.; Xu, J.; Qian, H. Diuron treatment reveals the different roles of two cyclic electron transfer pathways in photosystem II in Arabidopsis thaliana. Pestic. Biochem. Physiol. 2017, 137, 15–20. [Google Scholar] [CrossRef]
- Du, W.; Sun, Y.; Ji, R.; Zhu, J.; Wu, J.; Guo, H. TiO2 and ZnO nanoparticles negatively affect wheat growth and soil enzyme activities in agricultural soil. J. Environ. Monit. 2011, 13, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Timmusk, S.; Seisenbaeva, G.; Behers, L. Titania (TiO2) nanoparticles enhance the performance of growth-promoting rhizobacteria. Sci. Rep. 2018, 8, 617. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, B.; Shabbir, A.; Jaleel, H.; Khan, M.M.A.; Sadiq, Y. Efficacy of titanium dioxide nanoparticles in modulating photosynthesis, peltate glandular trichomes and essential oil production and quality in Mentha piperita L. Curr. Plant Biol. 2018, 13, 6–15. [Google Scholar] [CrossRef]
- Wu, F.; Fang, Q.; Yan, S.; Pan, L.; Tang, X.; Ye, W. Effects of zinc oxide nanoparticles on arsenic stress in rice (Cucumis sativus L.): Germination, early growth, and arsenic uptake. Environ. Sci. Pollut. Res. Int. 2020, 27, 26974–26981. [Google Scholar] [CrossRef]
- Rai-Kalal, P.; Jajoo, A. Priming with zinc oxide nanoparticles improve germination and photosynthetic performance in wheat. Plant Physiol. Biochem. 2021, 160, 341–351. [Google Scholar] [CrossRef]
- Wan, J.; Wang, R.; Wang, R.; Ju, Q.; Wang, Y.; Xu, J. Comparative physiological and transcriptomic analyses reveal the toxic effects of ZnO nanoparticles on plant growth. Environ. Sci. Technol. 2019, 53, 4235–4244. [Google Scholar] [CrossRef]
- Tumburu, L.; Andersen, C.P.; Rygiewicz, P.T.; Reichman, J.R. Molecular and physiological responses to titanium dioxide and cerium oxide nanoparticles in Arabidopsis. Environ. Toxicol. Chem. 2017, 36, 71–82. [Google Scholar] [CrossRef]
- Cui, D.; Wang, J.; Wang, H.; Yang, Y.; Zhao, M. The cytotoxicity of endogenous CdS and Cd2+ ions during CdS NPs biosynthesis. J. Hazard. Mater. 2021, 409, 124485. [Google Scholar] [CrossRef]
- Pagano, L.; Marmiroli, M.; Villani, M.; Magnani, J.; Rossi, R.; Zappettini, A.; White, J.C.; Marmiroli, N. Engineered nanomaterial exposure affects organelle genetic material replication in Arabidopsis thaliana. ACS Nano 2022, 16, 2249–2260. [Google Scholar] [CrossRef]
- Ali, S.; Mehmood, A.; Khan, N.; Tan, B. Uptake, translocation, and consequences of nanomaterials on plant growth and stress adaptation. J. Nanomater. 2021, 2021, 1–17. [Google Scholar] [CrossRef]
- Salehi, H.; Chehregani, A.; Lucini, L.; Majd, A.; Gholami, M. Morphological, proteomic and metabolomic insight into the effect of cerium dioxide nanoparticles to Phaseolus vulgaris L. under soil or foliar application. Sci. Total. Environ. 2018, 616–617, 1540–1551. [Google Scholar] [CrossRef] [PubMed]


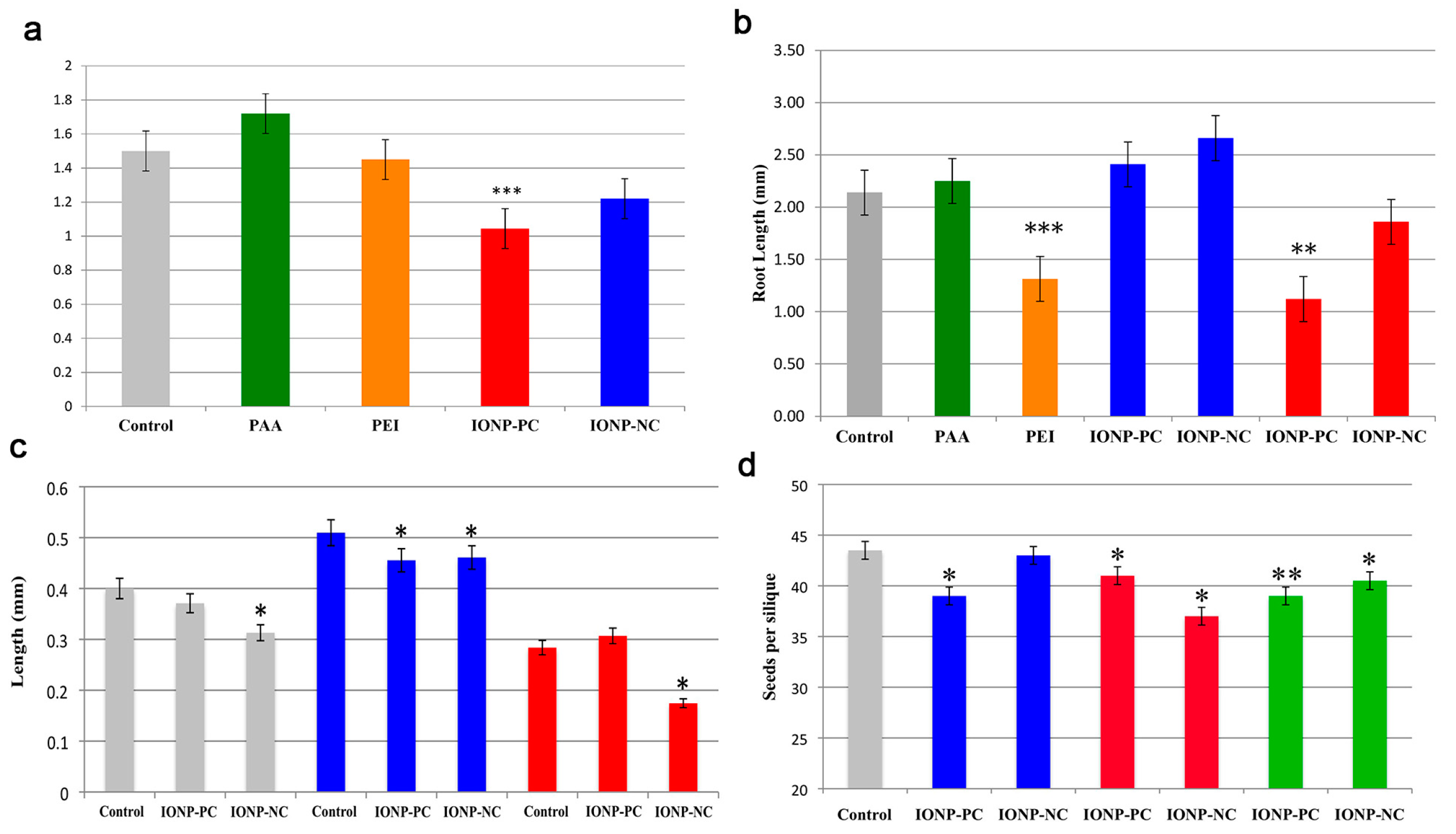
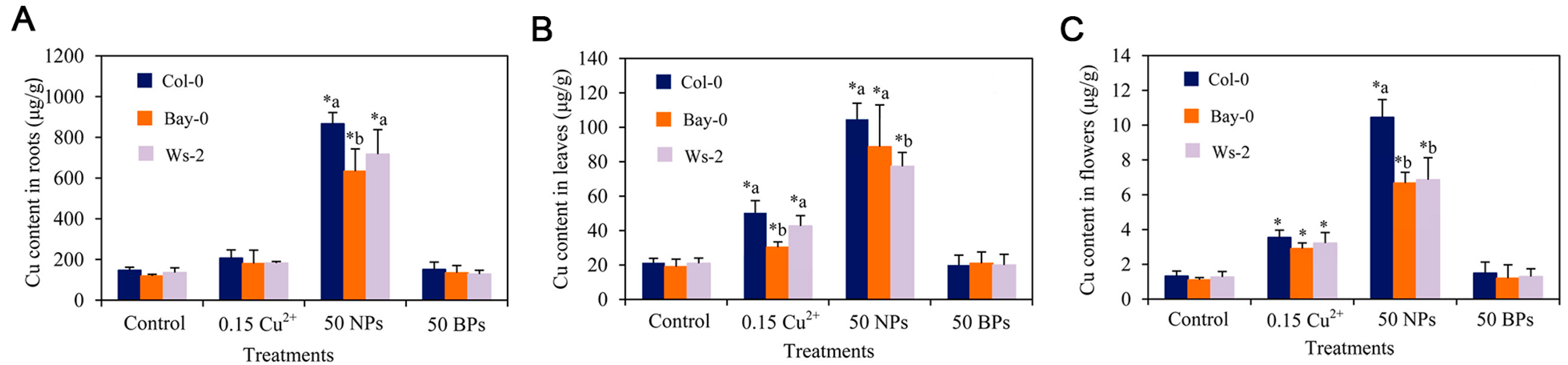

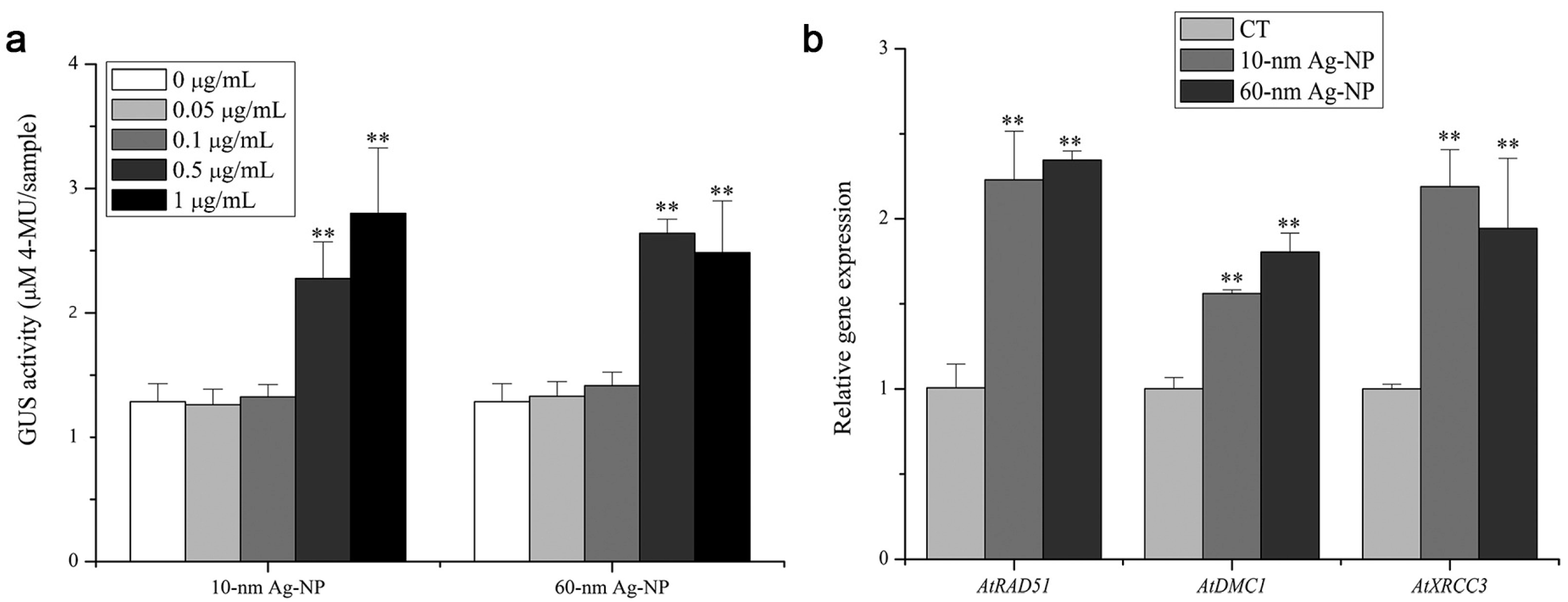

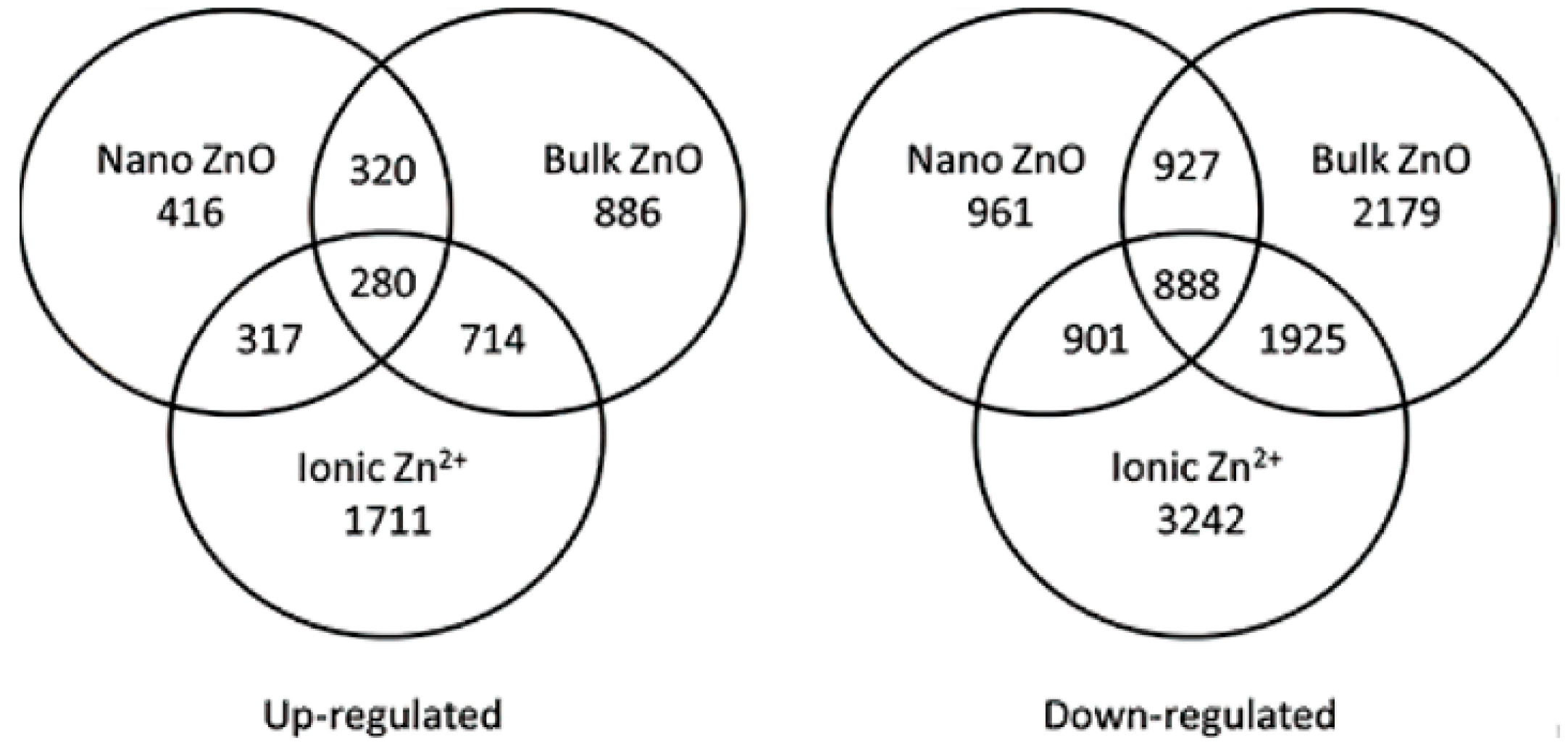

| MNPs | Size | Concentration | Impact | Reference |
|---|---|---|---|---|
| AgNPs | 20, 40, 80 nm | 7.0 × 1010, | Inhibited seedling root elongation and showed a linear dose–response relationship. | [28] |
| 9 × 109, | ||||
| 1.1 × 109 | ||||
| particles/mL | ||||
| AgNPs | 41 ± 1.5 nm | Greater than 300 mg/L | The inhibitory effect was saturated at 3000 mg/L, inhibiting growth and photosynthetic efficiency. | [54] |
| AgNPs | 10, 60 nm | 0, 0.05, 0.1, 0.5 and 1 μg/mL | After exposure to 60 nm Ag NPs, the Ag content in the aerial tissues was significantly increased. | [55] |
| AgNPs | 10 nm | 0.02 mg/L | Ag NPs no longer existed as intact individual particles but were aggregated and/or biotransformed in the plant. | [56] |
| AgNPs | 10 nm | 1.0, 2.5 mg/L | Induced glycolysis and affected the TCA cycle and aspartate family pathway. Glycine, serine and threonine metabolism were reduced. | [57] |
| AgNPs | 25.6 ± 5.1 nm | 0, 10, 30, 40, 50, 60, 70, 80, 90, 100, 150, 200 and 300 mg/L | Low Ag NPs levels induced ROS, accelerated root tip cell proliferation and promoted root growth. Relatively high concentrations of Ag NPs inhibited cell division, thereby limiting root growth. | [58] |
| AgNPs | 10–12 nm | 12.5 mg/kg | Affected the quality of pod and the growth of offspring seed, delayed flowering time by altering relevant pathways (photoperiod, autonomous and vernalization pathways) and inhibited pollen formation and development. Any negative effects on flower development could be transferred to the offspring. | [59] |
| PVP-coated AgNPs | 25 nm | 10, 30, 50, 100, 150 mg/L | Suppression of root to gravity with dose-dependent effects. | [60] |
| Au NPs | 60 nm | 1 ppm | Upon entering the leaves, it acted as a photothermal agent and remotely activated local biological processes in the plant on demand. | [27] |
| Au NPs | 5 nm | 25 µg/mL | Different surface charges affected Arabidopsis root development. | [30] |
| Au NPs | 13.4 ± 1.3, 12.1 ± 0.8 nm | 10 mg/L | Separated border-like cell sheets (isolated from the root) and associated mucus accumulated and trapped NPs independent of particle charge, in contrast to the marginal cells on the root crown that exhibited charge specificity. | [29] |
| Au NPs | 10–18 nm | 100 mg/mL | Au NPs had significant effects on the lateral roots of Arabidopsis. At the highest concentration, the minimal Au NPs inhibited the length of primary roots but contrarily also promoted the growth of hairy roots. | [61] |
| Au NPs | 24 nm | 10, 80 µg/mL | Exposure to Au NPs at 24 nm at concentrations of 10 and 80 μg/mL significantly increased seed germination, nutritional growth and free-radical scavenging activity. | [62] |
| CdSe/ZnS QDs | 6.3 ± 0.7 nm | 5 µg/mL | The ratio of reduced glutathione (GSH) to oxidized glutathione (GSSG) was reduced in the plants. | [49] |
| ZnSe QDs | - | 100, 250 μM | Caused oxidative stress in the leaves. | [48] |
| CuZn NPs | 20–30 nm | 30 mg/L | The photosystem II (PSII) function of young leaves was negatively affected. | [63] |
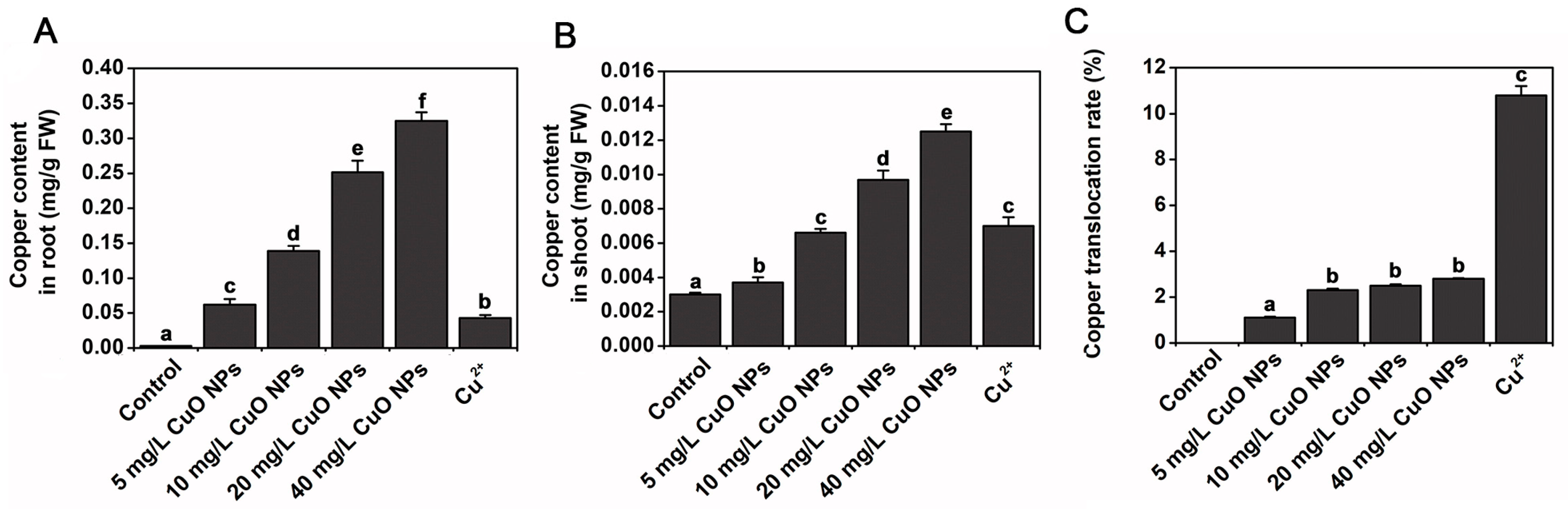
| ZnO NPs | 30 nm | 0.16–100 mg/L | High doses of ZnO NPs resulted in upregulation of the stress hormone abscisic acid, mainly in the apical regions and leaves. | [64] |
| ZnO NPs | 30 nm | 50, 100, 200 and 300 mg/L | Caused sugar and chlorophyll changes, DAB and NBT staining and antioxidant defense systems. | [65] |
| ZnO NPs | 20–45 nm | 0, 20, 50, 100, 200 mg/L | Arabidopsis’s fresh weight and primary root length were reduced, except when at a concentration of 20 mg/L. | [36] |
| ZnO NPs | 20 nm | 4 mg/L | There were 816 upregulated transcripts and 2179 downregulated transcripts. | [66] |
| CuO NPs | 40 nm | 10, 20, 40 mg/L | Interference with dynamic changes in actin led to abnormal apical cell development and inhibition of growth hormone transport, causing secondary damage to plant cells. | [37] |
| CuO NPs | 30–50 nm | 10, 20 mg/L | After 10 and 20 mg/L treatment for 2 h, the root cells of Arabidopsis were severely damaged. | [67] |
| CuO NPs | 38 nm | 50, 100, 200, 300, 400 mg/L | Affected rosette size, biomass, chlorophyll content, lipid peroxidation, ROS accumulation and cellular ultrastructure in Arabidopsis. | [68] |
| CuO NPs | 20–40 nm | 20, 50 mg/mL | The growth of Arabidopsis seedlings of different ecotypes (Col-0, Bay-0 and Ws-2) and the germination of their pollen and harvested seeds were inhibited. | [38] |
| CuO NPs | - | 5, 10 μg/mL | Elevated endogenous H2S and Cys content inhibited Arabidopsis root elongation in a dose-dependent manner. | [69] |
| CuO NPs | - | 10, 20 μg/mL | Strongly inhibited the growth of Arabidopsis. | [67] |
| TiO2 | 5–15 nm | 0.1, 0.5 mM | Showed phytotoxicity and could induce autophagy and protect plant cells from nanoparticle-induced damage, especially oxidative damage to chloroplasts. | [70] |
| TiO2 | 5–15 nm | 50, 100 mg/L | Reduced TC toxicity and increased the expression of both γ-glutamyl cysteine synthase (ECS) and glutathione synthase (GS) in Arabidopsis. | [71] |
| CeO2 | 10–30 nm | 0–2000 ppm | Exposure to CeO2 NPs at 250 ppm significantly increased plant biomass. At 500–2000 ppm CeO2 NPs, plant growth was reduced by up to 85% in a dose-dependent manner, and chlorophyll production was reduced by nearly 60% and 85% at 1000 and 2000 ppm, respectively. At 1000 ppm, MDA formation was increased by 2.5-fold. | [42] |
| CeO2 | 15–30 nm | 100, 200, 500, 1000, 2000 and 3000 mg/L | High concentrations of CeO2-NPs inhibited plant growth and adversely affected plants’ antioxidant system and photosystem. | [72] |
| In2O3 | 20–70 nm | 0–2000 ppm | Resulted in a 3.8–4.6-fold increase in glutathione synthetase (GS) transcription products. | [42] |
| Fe2O3 | 30 nm | 3, 25 mg/L | The 3 mg/L treatment had no significant effect on seedling and root length, and the 25 mg/L treatment resulted in a reduction in seedling and root length. | [73] |
| Types of MMNPs | Exposure Concentration | Exposure Time | Experimental Results | Reference |
|---|---|---|---|---|
| Ag | 5 mg/L | 10 d | Raise: respond to abiotic stress (mental stress, oxidation stress, salt stress, osmotic stress, hunger stress and water stress) | [55] |
| Lower: respond to pathogen stress and hormone stimulation (abscisic acid, auxin and ethylene) | ||||
| Ag | 12.5 mg/L | 45 d | Raise: organic acids, sugars, amino acids | [24] |
| Lower: amino acids, phenols | ||||
| Au | 100 g/L | - | Lower: expression of miR164, miR167, miR395, miR414, miR398 and miR408 | [62] |
| ZnO | 4 mg/L | 7 d | Raise: respond to abiotic stress (oxidative stress, salt stress, osmotic stress and water stress) and biological stress (pathogen defense), and participate in Zn2+ binding, transport and steady state | [65] |
| Lower: participate in cell tissue and biogenesis (tubulin, arabinogalactan glycoprotein), DNA or RNA metabolism (histone) | ||||
| ZnO | 100 mg/L | 7 d | Raise: lateral roots develop in response to abiotic stress (oxidative stress, salt stress, osmotic stress and water stress) and biological stress (wound stimulation and pathogen defense) | [36] |
| Lower: participate in cell tissue and biogenesis (translation, nucleosome assembly, tubulin), electron transfer | ||||
| CuO | 10 mg/L | 7 d | Raise: response to abiotic stress and biotic stress, Cu2+ binding and transport, plant hormone signal transduction | [38] |
| Lower: participation in metal homeostasis and transport and root hair development | ||||
| TiO2 | 50 μg/mL | 7 d | Raise: | [70] |
| Small particle size: increased expression level of AtRAD54 | ||||
| Big particle size: unable to increase AtRAD54 expression | ||||
| TiO2 | 20 μg/mL | 7 d | Raise: | [82] |
| Small particle size: AtRAD51, AtDMC1, AtXRCC3 | ||||
| Big particle size: AtRAD51, AtDMC1, AtXRCC3 | ||||
| CeO2 | 1 g/L | - | Raise: glutathione metabolism (oxidative stress response) and metal stress response genes | [102] |
| Al2O3 | 10 mg/L | 10d | Raise: respond to abiotic stress and biotic stress, cell wall development, nitrogen and phosphorus transport and root development | [91] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geng, M.; Li, L.; Ai, M.; Jin, J.; Hu, D.; Song, K. Recent Advances in Metal-Based Nanoparticle-Mediated Biological Effects in Arabidopsis thaliana: A Mini Review. Materials 2022, 15, 4539. https://doi.org/10.3390/ma15134539
Geng M, Li L, Ai M, Jin J, Hu D, Song K. Recent Advances in Metal-Based Nanoparticle-Mediated Biological Effects in Arabidopsis thaliana: A Mini Review. Materials. 2022; 15(13):4539. https://doi.org/10.3390/ma15134539
Chicago/Turabian StyleGeng, Min, Linlin Li, Mingjun Ai, Jun Jin, Die Hu, and Kai Song. 2022. "Recent Advances in Metal-Based Nanoparticle-Mediated Biological Effects in Arabidopsis thaliana: A Mini Review" Materials 15, no. 13: 4539. https://doi.org/10.3390/ma15134539
APA StyleGeng, M., Li, L., Ai, M., Jin, J., Hu, D., & Song, K. (2022). Recent Advances in Metal-Based Nanoparticle-Mediated Biological Effects in Arabidopsis thaliana: A Mini Review. Materials, 15(13), 4539. https://doi.org/10.3390/ma15134539






