Long-Term Assessment of Contemporary Ion-Releasing Restorative Dental Materials
Abstract
:1. Introduction
2. Materials and Methods
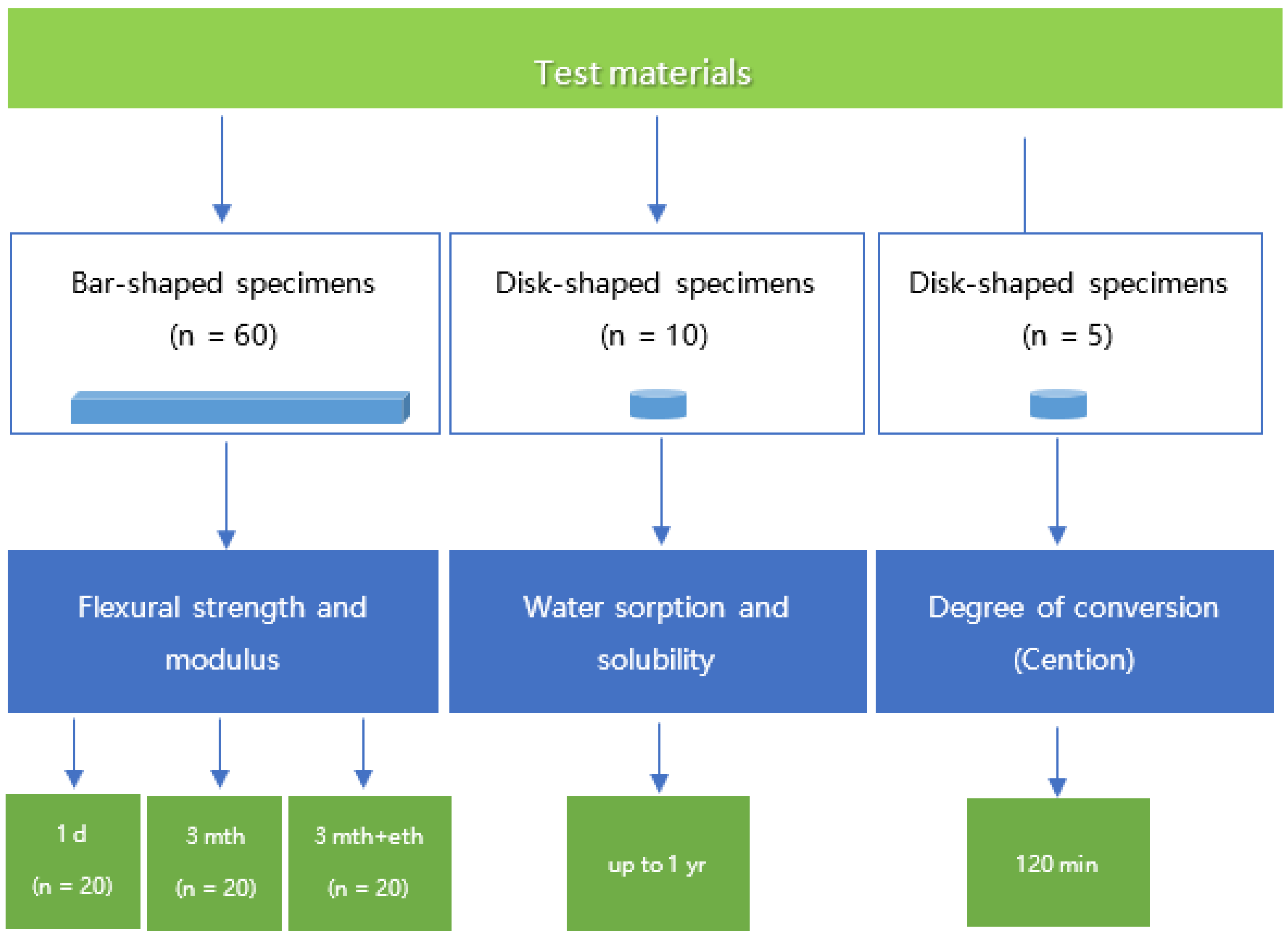
2.1. Study Protocol
2.2. Three-Point Bending Test
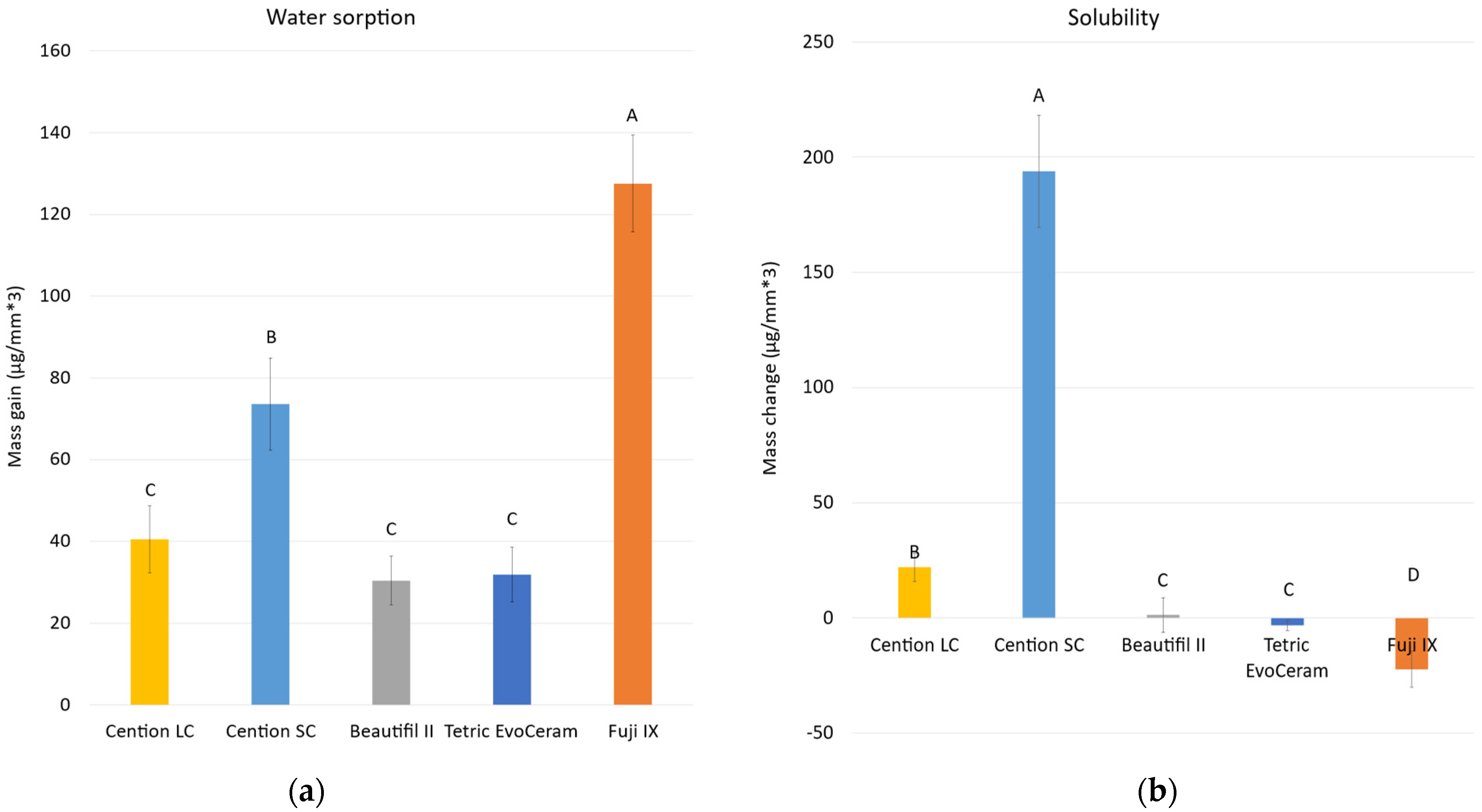
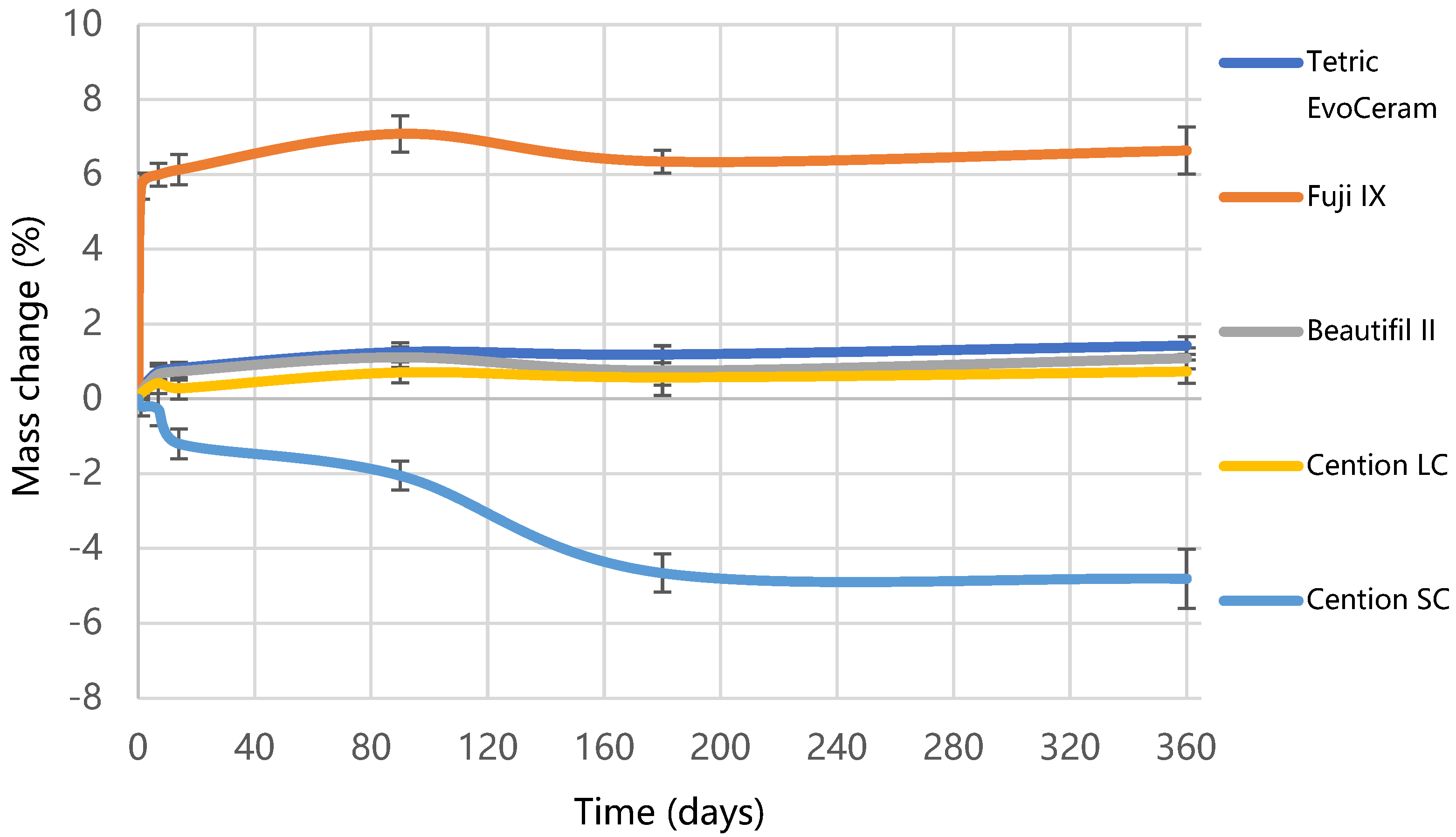
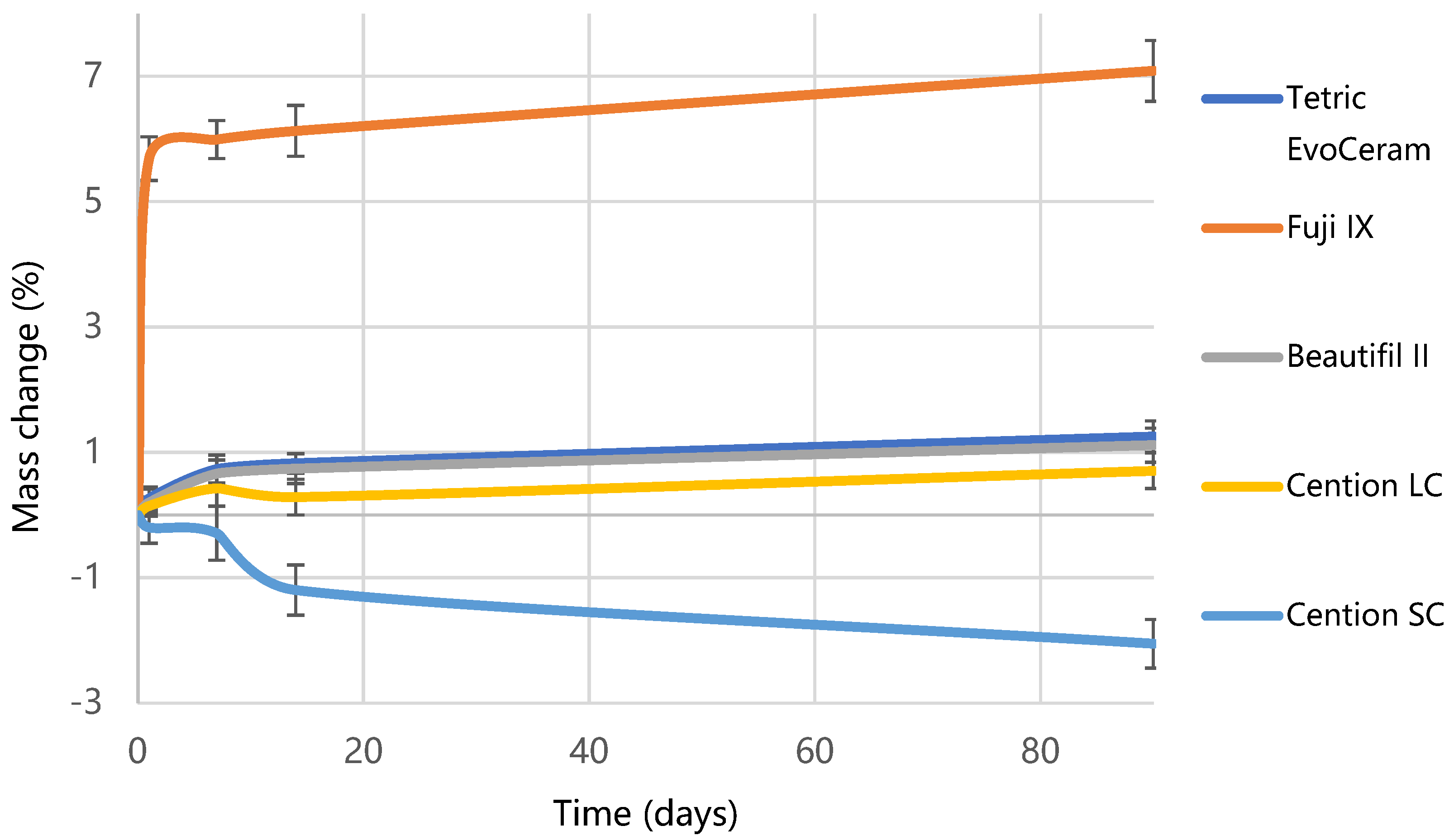
2.3. Water Sorption and Solubility
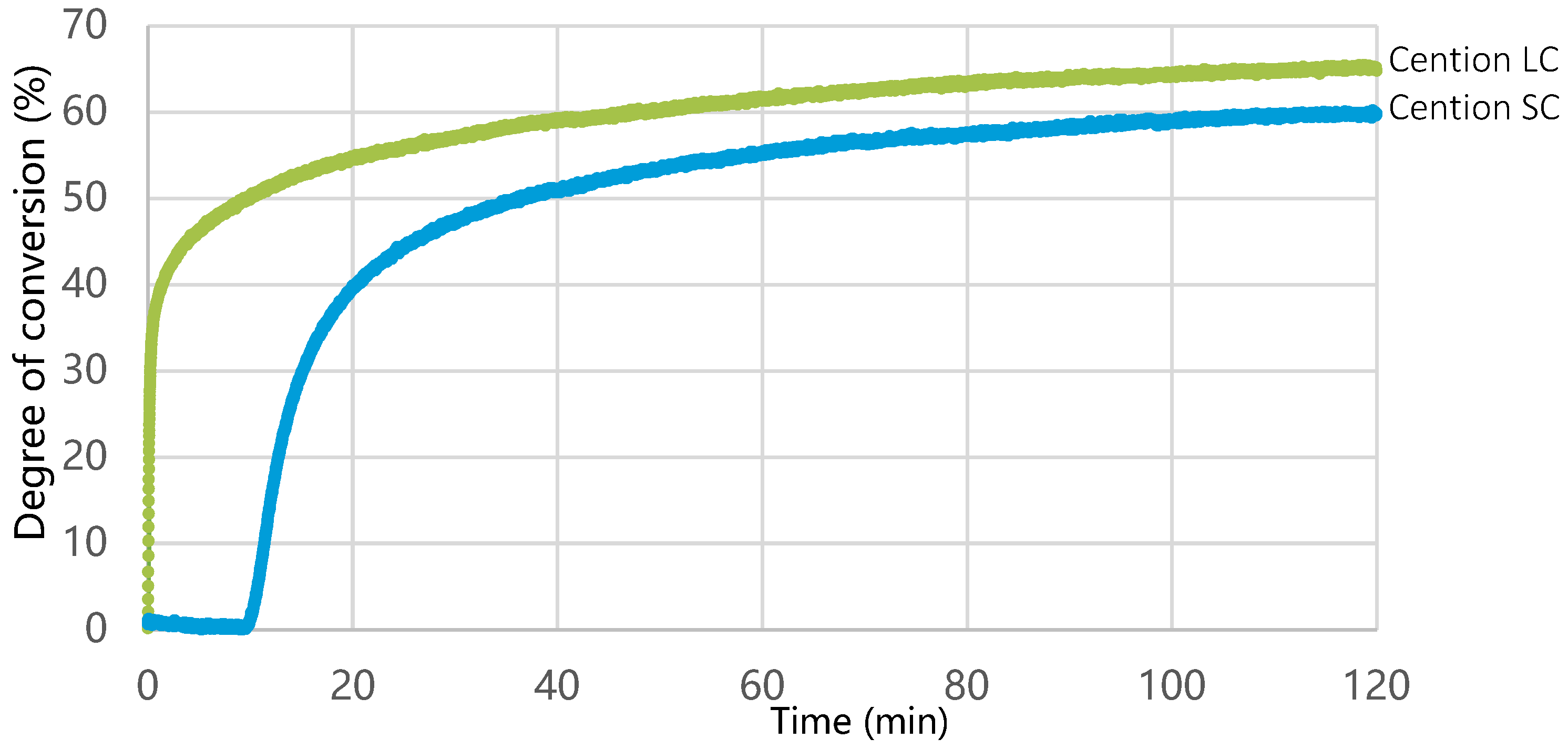
2.4. Degree of Conversion
2.5. Statistical Analysis
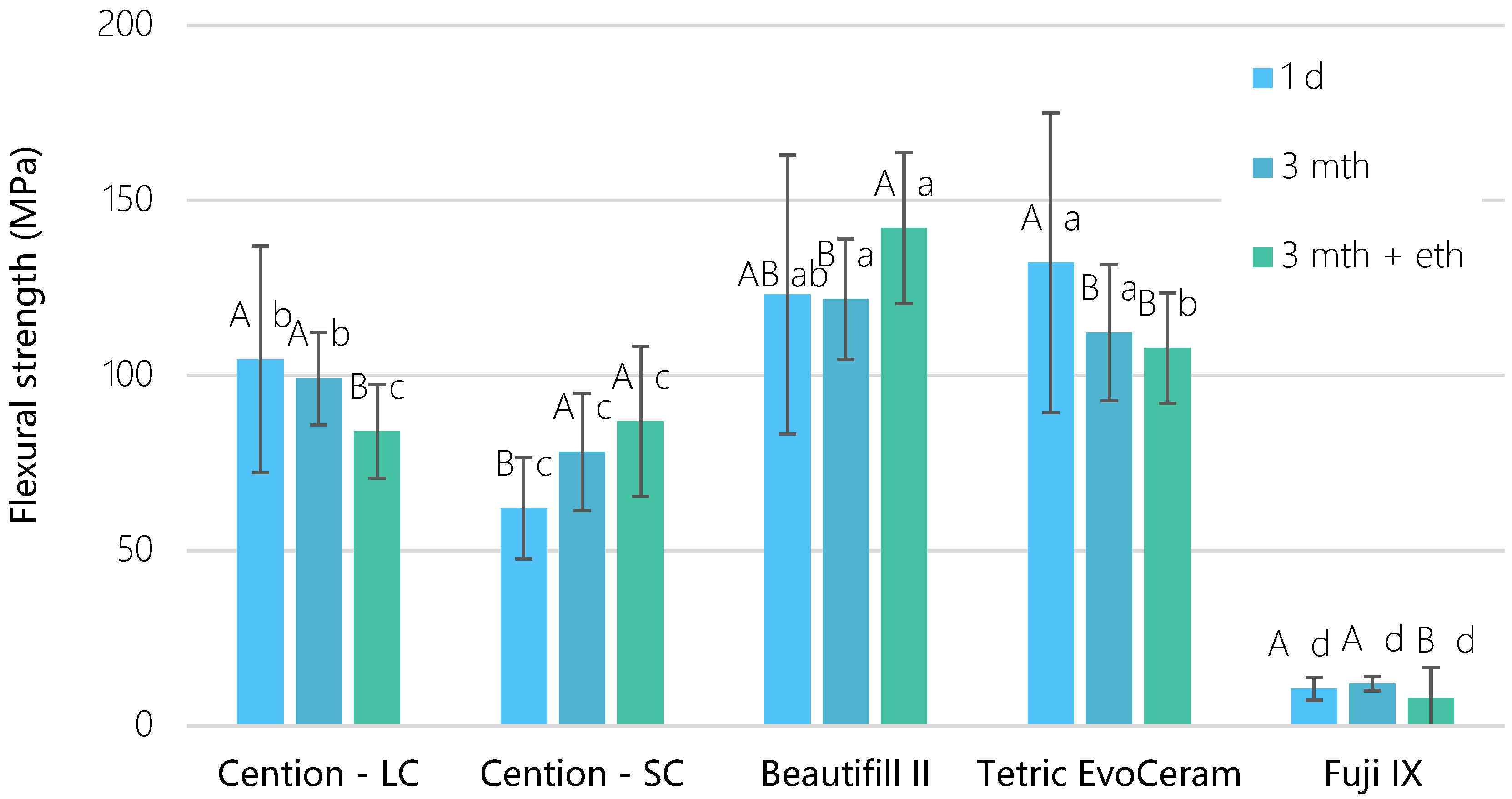
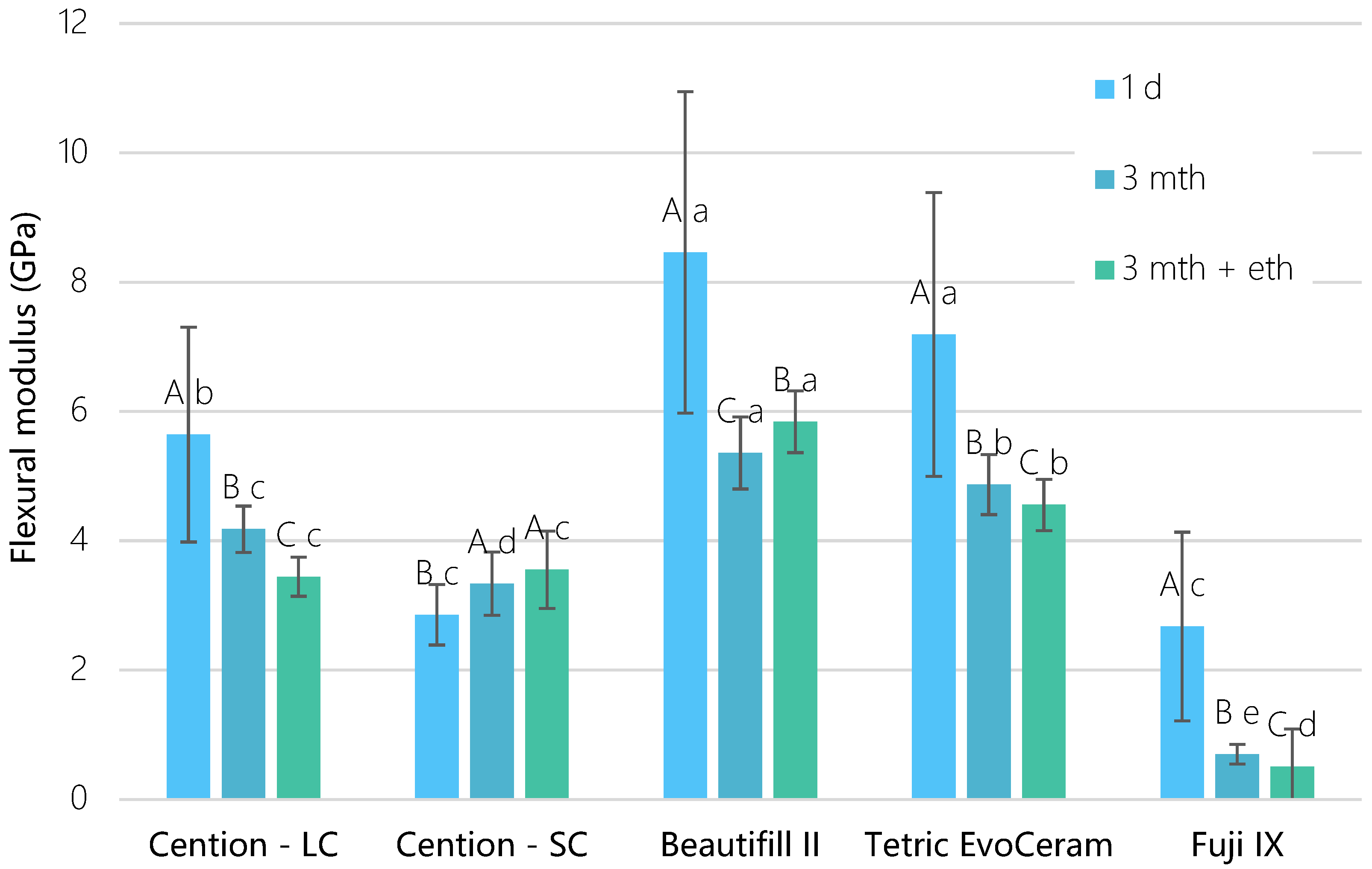
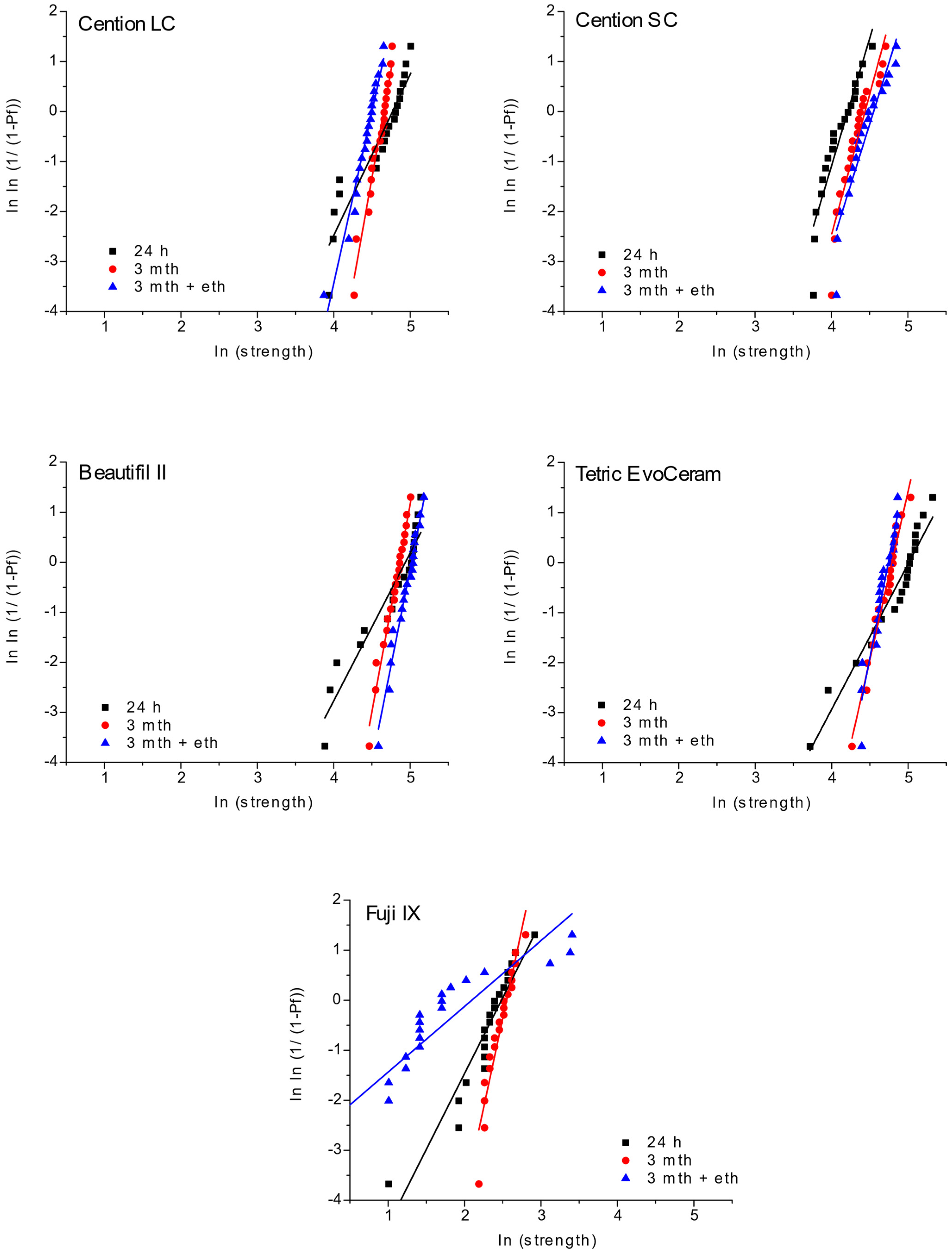
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marovic, D.; Haugen, H.J.; Negovetic Mandic, V.; Par, M.; Zheng, K.; Tarle, Z.; Boccaccini, A.R. Incorporation of Copper-Doped Mesoporous Bioactive Glass Nanospheres in Experimental Dental Composites: Chemical and Mechanical Characterization. Materials 2021, 14, 2611. [Google Scholar] [CrossRef] [PubMed]
- Marovic, D.; Tarle, Z.; Hiller, K.A.; Muller, R.; Ristic, M.; Rosentritt, M.; Skrtic, D.; Schmalz, G. Effect of silanized nanosilica addition on remineralizing and mechanical properties of experimental composite materials with amorphous calcium phosphate. Clin. Oral Investig. 2014, 18, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Marovic, D.; Tarle, Z.; Hiller, K.A.; Muller, R.; Rosentritt, M.; Skrtic, D.; Schmalz, G. Reinforcement of experimental composite materials based on amorphous calcium phosphate with inert fillers. Dent. Mater. 2014, 30, 1052–1060. [Google Scholar] [CrossRef]
- Par, M.; Attin, T.; Tarle, Z.; Taubock, T.T. A New Customized Bioactive Glass Filler to Functionalize Resin Composites: Acid-Neutralizing Capability, Degree of Conversion, and Apatite Precipitation. J. Clin. Med. 2020, 9, 1173. [Google Scholar] [CrossRef]
- Par, M.; Spanovic, N.; Mohn, D.; Attin, T.; Taubock, T.T.; Tarle, Z. Curing potential of experimental resin composites filled with bioactive glass: A comparison between Bis-EMA and UDMA based resin systems. Dent. Mater. 2020, 36, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Vallittu, P.K.; Boccaccini, A.R.; Hupa, L.; Watts, D.C. Bioactive dental materials-Do they exist and what does bioactivity mean? Dent. Mater. 2018, 34, 693–694. [Google Scholar] [CrossRef]
- Darvell, B. Bioactivity—symphony or cacophony? A personal view of a tangled field. Prosthesis 2021, 3, 8. [Google Scholar] [CrossRef]
- Hench, L.L. The story of Bioglass®. J. Mater. Sci. Mater. Med. 2006, 17, 967–978. [Google Scholar] [CrossRef]
- Spagnuolo, G. Bioactive Dental Materials: The Current Status. Materials 2022, 15, 2016. [Google Scholar] [CrossRef]
- Francois, P.; Fouquet, V.; Attal, J.-P.; Dursun, E. Commercially Available Fluoride-Releasing Restorative Materials: A Review and a Proposal for Classification. Materials 2020, 13, 2313. [Google Scholar] [CrossRef]
- Watson, T.F.; Atmeh, A.R.; Sajini, S.; Cook, R.J.; Festy, F. Present and future of glass-ionomers and calcium-silicate cements as bioactive materials in dentistry: Biophotonics-based interfacial analyses in health and disease. Dent. Mater. 2014, 30, 50–61. [Google Scholar] [CrossRef] [Green Version]
- Nicholson, J.W.; Wilson, A.D. The effect of storage in aqueous solutions on glass-ionomer and zinc polycarboxylate dental cements. J. Mater. Sci. Mater. Med. 2000, 11, 357–360. [Google Scholar] [CrossRef]
- Gemalmaz, D.; Yoruc, B.; Ozcan, M.; Alkumru, H.N. Effect of early water contact on solubility of glass ionomer luting cements. J. Prosthet. Dent. 1998, 80, 474–478. [Google Scholar] [CrossRef]
- Lohbauer, U. Dental Glass Ionomer Cements as Permanent Filling Materials?—Properties, Limitations Future Trends. Materials 2009, 3, 76–96. [Google Scholar] [CrossRef]
- Xie, D.; Brantley, W.A.; Culbertson, B.M.; Wang, G. Mechanical properties and microstructures of glass-ionomer cements. Dent. Mater. 2000, 16, 129–138. [Google Scholar] [CrossRef]
- Paolone, G. Direct composites in anteriors: A matter of substrate. Int. J. Esthet. Dent. 2017, 12, 468–481. [Google Scholar]
- Marovic, D.; Taubock, T.T.; Attin, T.; Panduric, V.; Tarle, Z. Monomer conversion and shrinkage force kinetics of low-viscosity bulk-fill resin composites. Acta Odontol. Scand. 2015, 73, 474–480. [Google Scholar] [CrossRef]
- Marovic, D.; Par, M.; Macan, M.; Klaric, N.; Plazonic, I.; Tarle, Z. Aging-Dependent Changes in Mechanical Properties of the New Generation of Bulk-Fill Composites. Materials 2022, 15, 902. [Google Scholar] [CrossRef]
- Garoushi, S.; Vallittu, P.K.; Lassila, L. Characterization of fluoride releasing restorative dental materials. Dent. Mater. J. 2018, 37, 293–300. [Google Scholar] [CrossRef] [Green Version]
- Eweis, A.H.; Yap, A.U.; Yahya, N.A. Impact of dietary solvents on flexural properties of bulk-fill composites. Saudi Dent. J. 2018, 30, 232–239. [Google Scholar] [CrossRef]
- Ozer, F.; Irmak, O.; Yakymiv, O.; Mohammed, A.; Pande, R.; Saleh, N.; Blatz, M. Three-year Clinical Performance of Two Giomer Restorative Materials in Restorations. Oper. Dent. 2021, 46, E60–E67. [Google Scholar] [CrossRef] [PubMed]
- Par, M.; Gubler, A.; Attin, T.; Tarle, Z.; Tarle, A.; Taubock, T.T. Ion release and hydroxyapatite precipitation of resin composites functionalized with two types of bioactive glass. J. Dent. 2022, 118, 103950. [Google Scholar] [CrossRef]
- Tiskaya, M.; Al-Eesa, N.A.; Wong, F.S.L.; Hill, R.G. Characterization of the bioactivity of two commercial composites. Dent. Mater. 2019, 35, 1757–1768. [Google Scholar] [CrossRef] [PubMed]
- Par, M.; Gubler, A.; Attin, T.; Tarle, Z.; Taubock, T.T. Anti-demineralizing protective effects on enamel identified in experimental and commercial restorative materials with functional fillers. Sci. Rep. 2021, 11, 11806. [Google Scholar] [CrossRef] [PubMed]
- Par, M.; Gubler, A.; Attin, T.; Tarle, Z.; Tarle, A.; Taubock, T.T. Experimental Bioactive Glass-Containing Composites and Commercial Restorative Materials: Anti-Demineralizing Protection of Dentin. Biomedicines 2021, 9, 1616. [Google Scholar] [CrossRef] [PubMed]
- Tecco, S.; Grusovin, M.G.; Sciara, S.; Bova, F.; Pantaleo, G.; Cappare, P. The association between three attitude-related indexes of oral hygiene and secondary implant failures: A retrospective longitudinal study. Int. J. Dent. Hyg. 2018, 16, 372–379. [Google Scholar] [CrossRef]
- Gherlone, E.; Polizzi, E.; Tete, G.; Cappare, P. Dentistry and Covid-19 pandemic: Operative indications post-lockdown. New Microbiol. 2021, 44, 1–11. [Google Scholar]
- Akashi, A.; Matsuya, Y.; Unemori, M.; Akamine, A. The relationship between water absorption characteristics and the mechanical strength of resin-modified glass-ionomer cements in long-term water storage. Biomaterials 1999, 20, 1573–1578. [Google Scholar] [CrossRef]
- Marovic, D.; Par, M.; Crnadak, A.; Sekelja, A.; Negovetic Mandic, V.; Gamulin, O.; Rakic, M.; Tarle, Z. Rapid 3 s Curing: What Happens in Deep Layers of New Bulk-Fill Composites? Materials 2021, 14, 515. [Google Scholar] [CrossRef]
- Haugen, H.J.; Marovic, D.; Par, M.; Thieu, M.K.L.; Reseland, J.E.; Johnsen, G.F. Bulk Fill Composites Have Similar Performance to Conventional Dental Composites. Int. J. Mol. Sci. 2020, 21, 5136. [Google Scholar] [CrossRef]
- Bahari, M.; Kahnamoui, M.A.; Chaharom, M.E.E.; Kimyai, S.; Sattari, Z. Effect of curing method and thermocycling on flexural strength and microhardness of a new composite resin with alkaline filler. Dent. Res. J. 2021, 18, 96. [Google Scholar] [CrossRef]
- Yap, A.U.; Choo, H.S.; Choo, H.Y.; Yahya, N.A. Flexural Properties of Bioactive Restoratives in Cariogenic Environments. Oper. Dent. 2021, 46, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Ilie, N. Comparative Effect of Self- or Dual-Curing on Polymerization Kinetics and Mechanical Properties in a Novel, Dental-Resin-Based Composite with Alkaline Filler. Materials 2018, 11, 108. [Google Scholar] [CrossRef] [Green Version]
- Gomes de Araújo-Neto, V.; Sebold, M.; Fernandes de Castro, E.; Feitosa, V.P.; Giannini, M. Evaluation of physico-mechanical properties and filler particles characterization of conventional, bulk-fill, and bioactive resin-based composites. J. Mech. Behav. Biomed. Mater. 2021, 115, 104288. [Google Scholar] [CrossRef]
- Kelić, K.; Par, M.; Peroš, K.; Šutej, I.; Tarle, Z. Fluoride-Releasing Restorative Materials: The Effect of a Resinous Coat on Ion Release. Acta Stomatol. Croat. 2020, 54, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Roulet, J.F.; Gummadi, S.; Hussein, H.S.; Abdulhameed, N.; Shen, C. In vitro wear of dual-cured bulkfill composites and flowable bulkfill composites. J. Esthet. Restor. Dent. 2020, 32, 512–520. [Google Scholar] [CrossRef]
- Pribadi, N.; Budiarti, D.; Kurniawan, H.J.; Widjiastuti, I. The NF-kB and Collagen Type 1 Expression in Dental Pulp after Treated Calcium Hydroxide Combined with Propolis. Eur. J. Dent. 2021, 15, 122–126. [Google Scholar] [CrossRef]
- Aldhafyan, M.; Silikas, N.; Watts, D.C. Influence of curing modes on conversion and shrinkage of dual-cure resin-cements. Dent. Mater. 2022, 38, 194–203. [Google Scholar] [CrossRef]
- ISO 4049:2009; Dentistry—Polymer-Based Filling, Restorative and Luting Materials. International Organization for Standardization: Geneva, Switzerland, 2000.
- Stansbury, J.W. Dimethacrylate network formation and polymer property evolution as determined by the selection of monomers and curing conditions. Dent. Mater. 2012, 28, 13–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barszczewska-Rybarek, I.M. A Guide through the Dental Dimethacrylate Polymer Network Structural Characterization and Interpretation of Physico-Mechanical Properties. Materials 2019, 12, 4057. [Google Scholar] [CrossRef] [Green Version]
- Sirovica, S.; Solheim, J.H.; Skoda, M.W.A.; Hirschmugl, C.J.; Mattson, E.C.; Aboualizadeh, E.; Guo, Y.; Chen, X.; Kohler, A.; Romanyk, D.L.; et al. Origin of micro-scale heterogeneity in polymerisation of photo-activated resin composites. Nat. Commun. 2020, 11, 1849. [Google Scholar] [CrossRef] [Green Version]
- Da Silva, E.M.; Poskus, L.T.; Guimaraes, J.G.; de Araujo Lima Barcellos, A.; Fellows, C.E. Influence of light polymerization modes on degree of conversion and crosslink density of dental composites. J. Mater. Sci. Mater. Med. 2008, 19, 1027–1032. [Google Scholar] [CrossRef]
- Soh, M.S.; Yap, A.U. Influence of curing modes on crosslink density in polymer structures. J. Dent. 2004, 32, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Sideridou, I.D.; Karabela, M.M.; Bikiaris, D.N. Aging studies of light cured dimethacrylate-based dental resins and a resin composite in water or ethanol/water. Dent. Mater. 2007, 23, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Yap, A.U.; Wang, X.; Wu, X.; Chung, S.M. Comparative hardness and modulus of tooth-colored restoratives: A depth-sensing microindentation study. Biomaterials 2004, 25, 2179–2185. [Google Scholar] [CrossRef]
- Bhatia, H.P.; Singh, S.; Sood, S.; Sharma, N. A Comparative Evaluation of Sorption, Solubility, and Compressive Strength of Three Different Glass Ionomer Cements in Artificial Saliva: An in vitro Study. Int. J. Clin. Pediatr. Dent. 2017, 10, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, R.; Alshali, R.Z.; Silikas, N. The effect of desiccation on water sorption, solubility and hygroscopic volumetric expansion of dentine replacement materials. Dent. Mater. 2018, 34, e205–e213. [Google Scholar] [CrossRef] [Green Version]
- Cefaly, D.F.; Franco, E.B.; Mondelli, R.F.; Francisconi, P.A.; Navarro, M.F. Diametral tensile strength and water sorption of glass-ionomer cements used in Atraumatic Restorative Treatment. J. Appl. Oral Sci. 2003, 11, 96–101. [Google Scholar] [CrossRef] [Green Version]
- Muller, J.A.; Rohr, N.; Fischer, J. Evaluation of ISO 4049: Water sorption and water solubility of resin cements. Eur. J. Oral Sci. 2017, 125, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Par, M.; Spanovic, N.; Bjelovucic, R.; Marovic, D.; Schmalz, G.; Gamulin, O.; Tarle, Z. Long-term water sorption and solubility of experimental bioactive composites based on amorphous calcium phosphate and bioactive glass. Dent. Mater. J. 2019, 38, 555–564. [Google Scholar] [CrossRef] [Green Version]
- Alshali, R.Z.; Salim, N.A.; Satterthwaite, J.D.; Silikas, N. Long-term sorption and solubility of bulk-fill and conventional resin-composites in water and artificial saliva. J. Dent. 2015, 43, 1511–1518. [Google Scholar] [CrossRef] [PubMed]
- Ferracane, J.L.; Berge, H.X.; Condon, J.R. In vitro aging of dental composites in water—effect of degree of conversion, filler volume, and filler/matrix coupling. J. Biomed. Mater. Res. 1998, 42, 465–472. [Google Scholar] [CrossRef]
- Par, M.; Tarle, Z.; Hickel, R.; Ilie, N. Mechanical properties of experimental composites containing bioactive glass after artificial aging in water and ethanol. Clin. Oral Investig. 2019, 23, 2733–2741. [Google Scholar] [CrossRef] [PubMed]
- Szczesio-Wlodarczyk, A.; Sokolowski, J.; Kleczewska, J.; Bociong, K. Ageing of Dental Composites Based on Methacrylate Resins—A Critical Review of the Causes and Method of Assessment. Polymers 2020, 12, 882. [Google Scholar] [CrossRef] [PubMed]
- Heintze, S.D.; Ilie, N.; Hickel, R.; Reis, A.; Loguercio, A.; Rousson, V. Laboratory mechanical parameters of composite resins and their relation to fractures and wear in clinical trials—A systematic review. Dent. Mater. 2017, 33, e101–e114. [Google Scholar] [CrossRef]
| Type | Product Name (Manufacturer) | Composition | Curing Mechanism |
|---|---|---|---|
| Alkasite | Cention (Ivoclar Vivadent) | Powder: inert barium alumino-boro-silicate glass, ytterbium fluoride, a calcium fluoro-alumino-silicate glass, and a reactive SiO2-CaO-CaF2-Na2O glass Liquid: UDMA, aromatic aliphatic UDMA, DCP, and PEG-400-DMA Initiator system: hydroperoxide, Ivocerin, and acyl phosphine oxide Filler content: 58–59 vol% | Dual-cure |
| Giomer | Beautifil II (Shofu Dental GmbH) | Fillers: s-PRG (aluminofluoro-borosilicate glass), Al2O3 Resin: bis-GMA, TEGDMA Filler content: 69 vol% | Light-cure |
| Glass-ionomer | Fuji IX GP Fast (GC Europe) | Powder: fluoro-alumino-silicate glass Liquid: Polybasic carboxylic acid (copolymer of acrylic and maleic acid), tartaric acid, water | Self-cure |
| Composite (control) | Tetric EvoCeram (Ivoclar Vivadent) | Fillers: Barium glass filler, ytterbium fluoride, mixed oxide, prepolymers Resin: bis-GMA, UDMA, bis-EMA Filler content: 53–55 vol% | Light-cure |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marovic, D.; Par, M.; Posavec, K.; Marić, I.; Štajdohar, D.; Muradbegović, A.; Tauböck, T.T.; Attin, T.; Tarle, Z. Long-Term Assessment of Contemporary Ion-Releasing Restorative Dental Materials. Materials 2022, 15, 4042. https://doi.org/10.3390/ma15124042
Marovic D, Par M, Posavec K, Marić I, Štajdohar D, Muradbegović A, Tauböck TT, Attin T, Tarle Z. Long-Term Assessment of Contemporary Ion-Releasing Restorative Dental Materials. Materials. 2022; 15(12):4042. https://doi.org/10.3390/ma15124042
Chicago/Turabian StyleMarovic, Danijela, Matej Par, Karlo Posavec, Ivana Marić, Dominik Štajdohar, Alen Muradbegović, Tobias T. Tauböck, Thomas Attin, and Zrinka Tarle. 2022. "Long-Term Assessment of Contemporary Ion-Releasing Restorative Dental Materials" Materials 15, no. 12: 4042. https://doi.org/10.3390/ma15124042
APA StyleMarovic, D., Par, M., Posavec, K., Marić, I., Štajdohar, D., Muradbegović, A., Tauböck, T. T., Attin, T., & Tarle, Z. (2022). Long-Term Assessment of Contemporary Ion-Releasing Restorative Dental Materials. Materials, 15(12), 4042. https://doi.org/10.3390/ma15124042









