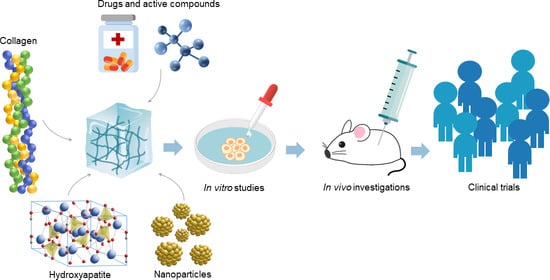
Review of the Applications of Biomedical Compositions Containing Hydroxyapatite and Collagen Modified by Bioactive Components
Abstract
1. Introduction
2. Hydroxyapatite
3. Collagen
4. Compositions of Collagen/HAp
5. Conjugates for Drug Delivery
5.1. Conjugates of Collagen/HAp/Drugs
5.1.1. Conjugates of Collagen/HAp/Statins: Preparation and Application
5.1.2. Conjugates of Collagen/HAp/Paclitaxel: Preparation and Application
5.1.3. Conjugates of Collagen/HAp/Vancomycin: Preparation and Application
5.1.4. Conjugates of Collagen/HAp/Tetracyclines: Preparation and Application
5.1.5. Conjugates of Collagen/HAp/Gentamicin: Preparation and Application
5.1.6. Conjugates of Collagen/HAp/Alendronate: Preparation and Application
5.2. Conjugates of Collagen/HAp/Metals and Nanoparticles
5.2.1. Conjugates of Collagen/HAp/Cisplatin and Platinum Nanoparticles: Preparation and Application
5.2.2. Conjugates of Collagen/HAp/Magnetite Particles (Iron Oxide Nanoparticles): Preparation and Application
5.2.3. Conjugates of Collagen/HAp/Carbon Nanotubes: Preparation and Application
5.2.4. Conjugates of Collagen/HAp/Graphene: Preparation and Application
5.2.5. Conjugates of Collagen/HAp/Silver Nanoparticles: Preparation and Application
5.2.6. Conjugates of Collagen/HAp/Gold Nanoparticles: Preparation and Application
5.2.7. Conjugates of Collagen/HAp/Bioactive Alloys: Preparation and Application
5.3. Conjugates of Collagen/HAp/Bioactive Macromolecules
5.3.1. Conjugates of Collagen/HAp/Proteins: Preparation and Application
5.3.2. Conjugates of Collagen/HAp/Phospholipids: Preparation and Application
5.3.3. Conjugates of Collagen/HAp/Pectin: Preparation and Application
5.3.4. Conjugates of Collagen/HAp/Natural Polysaccharides: Preparation and Application
5.3.5. Conjugates of Collagen/HAp/Synthetic Polymers: Preparation and Application
5.3.6. Conjugates of Collagen/HAp/Flavonoids: Preparation and Application
5.3.7. Conjugates of Collagen/HAp/Growth Factors: Preparation and Application
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Goonoo, N.; Bhaw-Luximon, A.; Bowlin, G.L.; Jhurry, D. An Assessment of Biopolymer- and Synthetic Polymer-Based Scaffolds for Bone and Vascular Tissue Engineering. Polym. Int. 2013, 62, 523–533. [Google Scholar] [CrossRef]
- Zhao, P.; Gu, H.; Mi, H.; Rao, C.; Fu, J.; Turng, L. Fabrication of Scaffolds in Tissue Engineering: A Review. Front. Mech. Eng. 2018, 13, 107–119. [Google Scholar] [CrossRef]
- Kaviyarasu, K.; Maria Magdalane, C.; Kanimozhi, K.; Kennedy, J.; Siddhardha, B.; Subba Reddy, E.; Rotte, N.K.; Sharma, C.S.; Thema, F.T.; Letsholathebe, D.; et al. Elucidation of Photocatalysis, Photoluminescence and Antibacterial Studies of ZnO Thin Films by Spin Coating Method. J. Photochem. Photobiol. B Biol. 2017, 173, 466–475. [Google Scholar] [CrossRef]
- Magdalane, C.M.; Kaviyarasu, K.; Vijaya, J.J.; Siddhardha, B.; Jeyaraj, B.; Kennedy, J.; Maaza, M. Evaluation on the Heterostructured CeO2/Y2O3 Binary Metal Oxide Nanocomposites for UV/Vis Light Induced Photocatalytic Degradation of Rhodamine—B Dye for Textile Engineering Application. J. Alloys Compd. 2017, 727, 1324–1337. [Google Scholar] [CrossRef]
- Velasco, M.A.; Narváez-Tovar, C.A.; Garzón-Alvarado, D.A. Design, Materials, and Mechanobiology of Biodegradable Scaffolds for Bone Tissue Engineering. Biomed. Res. Int. 2015, 2015, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Dhandayuthapani, B.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Polymeric Scaffolds in Tissue Engineering Application: A Review. Int. J. Polym. Sci. 2011, 2011, 1–19. [Google Scholar] [CrossRef]
- Khan, F.; Tanaka, M. Designing Smart Biomaterials for Tissue Engineering. Int. J. Mol. Sci. 2017, 19, 17. [Google Scholar] [CrossRef] [PubMed]
- Kloxin, A.M.; Kasko, A.M.; Salinas, C.N.; Anseth, K.S. Photodegradable Hydrogels for Dynamic Tuning of Physical and Chemical Properties. Science 2009, 324, 59–63. [Google Scholar] [CrossRef]
- Kumbar, S.G.; James, R.; Nukavarapu, S.P.; Laurencin, C.T. Electrospun Nanofiber Scaffolds: Engineering Soft Tissues. Biomed. Mater. 2008, 3, 034002. [Google Scholar] [CrossRef]
- Dimitriou, R.; Jones, E.; McGonagle, D.; Giannoudis, P.V. Bone Regeneration: Current Concepts and Future Directions. BMC Med. 2011, 9, 66. [Google Scholar] [CrossRef]
- Ho-Shui-Ling, A.; Bolander, J.; Rustom, L.E.; Johnson, A.W.; Luyten, F.P.; Picart, C. Bone Regeneration Strategies: Engineered Scaffolds, Bioactive Molecules and Stem Cells Current Stage and Future Perspectives. Biomaterials 2018, 180, 143–162. [Google Scholar] [CrossRef]
- Fu, S.; Ni, P.; Wang, B.; Chu, B.; Zheng, L.; Luo, F.; Luo, J.; Qian, Z. Injectable and Thermo-Sensitive PEG-PCL-PEG Copolymer/Collagen/n-HA Hydrogel Composite for Guided Bone Regeneration. Biomaterials 2012, 33, 4801–4809. [Google Scholar] [CrossRef]
- Trimeche, M. Biomaterials for Bone Regeneration: An Overview; Elsevier: Amsterdam, The Netherlands, 2017; Volume 1, pp. 1–5. [Google Scholar] [CrossRef]
- Jahan, K.; Tabrizian, M. Composite Biopolymers for Bone Regeneration Enhancement in Bony Defects. Biomater. Sci. 2016, 4, 25–39. [Google Scholar] [CrossRef]
- Neffe, A.T.; Julich-Gruner, K.K.; Lendlein, A. Combinations of biopolymers and synthetic polymers for bone regeneration. In Biomaterials for Bone Regeneration; Elsevier: Amsterdam, The Netherlands, 2014; pp. 87–110. [Google Scholar]
- White, A.A.; Best, S.M.; Kinloch, I.A. Hydroxyapatite-Carbon Nanotube Composites for Biomedical Applications: A Review. Int. J. Appl. Ceram. Technol. 2007, 4, 1–13. [Google Scholar] [CrossRef]
- Jiang, D.; Premachandra, G.S.; Johnston, C.; Hem, S.L. Structure and Adsorption Properties of Commercial Calcium Phosphate Adjuvant. Vaccine 2004, 23, 693–698. [Google Scholar] [CrossRef]
- Liu, P.; Tao, J.; Cai, Y.; Pan, H.; Xu, X.; Tang, R. Role of Fetal Bovine Serum in the Prevention of Calcification in Biological Fluids. J. Cryst. Growth 2008, 310, 4672–4675. [Google Scholar] [CrossRef]
- Yoshida, K.; Kobayashi, M.; Hyuga, H.; Kondo, N.; Kita, H.; Hashimoto, K.; Toda, Y. Reaction Sintering of β-Tricalcium Phosphates and Their Mechanical Properties. J. Eur. Ceram. Soc. 2007, 27, 3215–3220. [Google Scholar] [CrossRef]
- Destainville, A.; Champion, E.; Bernache-Assollant, D.; Laborde, E. Synthesis, Characterization and Thermal Behavior of Apatitic Tricalcium Phosphate. Mater. Chem. Phys. 2003, 80, 269–277. [Google Scholar] [CrossRef]
- Mobasherpour, I.; Heshajin, M.S.; Kazemzadeh, A.; Zakeri, M. Synthesis of Nanocrystalline Hydroxyapatite by Using Precipitation Method. J. Alloys Compd. 2007, 430, 330–333. [Google Scholar] [CrossRef]
- Barros, L.A.F.; Ferreira, E.E.; Peres, A.E.C. Floatability of Apatites and Gangue Minerals of an Igneous Phosphate Ore. Miner. Eng. 2008, 21, 994–999. [Google Scholar] [CrossRef]
- Cai, S.; Wang, Y.; Lv, H.; Peng, Z.; Yao, K. Synthesis of Carbonated Hydroxyapatite Nanofibers by Mechanochemical Methods. Ceram. Int. 2005, 31, 135–138. [Google Scholar] [CrossRef]
- Veiderma, M.; Tõnsuaadu, K.; Knubovets, R.; Peld, M. Impact of Anionic Substitutions on Apatite Structure and Properties. J. Organomet. Chem. 2005, 690, 2638–2643. [Google Scholar] [CrossRef]
- Haberko, K.; Haberko, M.; Pyda, W.; Pędzich, Z.; Chłopek, J.; Mozgawa, W.; Bućko, M. Sposób otrzymywania Naturalnego Hydroksyapatytu z Kości Zwierzęcych. Polish Patent No. P-359960/2003, 5 May 2003. [Google Scholar]
- Suchanek, W.L.; Byrappa, K.; Shuk, P.; Riman, R.E.; Janas, V.F.; TenHuisen, K.S. Preparation of Magnesium-Substituted Hydroxyapatite Powders by the Mechanochemical–Hydrothermal Method. Biomaterials 2004, 25, 4647–4657. [Google Scholar] [CrossRef]
- Shellis, R.P.; Wilson, R.M. Apparent Solubility Distributions of Hydroxyapatite and Enamel Apatite. J. Colloid Interface Sci. 2004, 278, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Landi, E.; Tampieri, A.; Mattioli-Belmonte, M.; Celotti, G.; Sandri, M.; Gigante, A.; Fava, P.; Biagini, G. Biomimetic Mg- and Mg,CO3-Substituted Hydroxyapatites: Synthesis Characterization and in Vitro Behaviour. J. Eur. Ceram. Soc. 2006, 26, 2593–2601. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Mechanism of Solid-State Conversion of Non-Stoichiometric Hydroxyapatite to Diphase Calcium Phosphate. Russ. Chem. Bull. 2003, 52, 2369–2375. [Google Scholar] [CrossRef]
- Bouhaouss, A.; Laghzizil, A.; Bensaoud, A.; Ferhat, M.; Lorent, G.; Livage, J. Mechanism of Ionic Conduction in Oxy and Hydroxyapatite Structures. Int. J. Inorg. Mater. 2001, 3, 743–747. [Google Scholar] [CrossRef]
- Ooi, C.Y.; Hamdi, M.; Ramesh, S. Properties of Hydroxyapatite Produced by Annealing of Bovine Bone. Ceram. Int. 2007, 33, 1171–1177. [Google Scholar] [CrossRef]
- Descamps, M.; Hornez, J.C.; Leriche, A. Effects of Powder Stoichiometry on the Sintering of β-Tricalcium Phosphate. J. Eur. Ceram. Soc. 2007, 27, 2401–2406. [Google Scholar] [CrossRef]
- Camiré, C.L.; Gbureck, U.; Hirsiger, W.; Bohner, M. Correlating Crystallinity and Reactivity in an α-Tricalcium Phosphate. Biomaterials 2005, 26, 2787–2794. [Google Scholar] [CrossRef]
- Peck, W.H.; Tumpane, K.P. Low Carbon Isotope Ratios in Apatite: An Unreliable Biomarker in Igneous and Metamorphic Rocks. Chem. Geol. 2007, 245, 305–314. [Google Scholar] [CrossRef]
- Rønsbo, J.G. Apatite in the Ilímaussaq Alkaline Complex: Occurrence, Zonation and Compositional Variation. Lithos 2008, 106, 71–82. [Google Scholar] [CrossRef]
- Belousova, E.A.; Griffin, W.L.; O’Reilly, S.Y.; Fisher, N.I. Apatite as an Indicator Mineral for Mineral Exploration: Trace-Element Compositions and Their Relationship to Host Rock Type. J. Geochem. Explor. 2002, 76, 45–69. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Myoui, A. Bone Tissue Engineering with Porous Hydroxyapatite Ceramics. J. Artif. Organs 2005, 8, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Parekh, B.; Joshi, M.; Vaidya, A. Characterization and Inhibitive Study of Gel-Grown Hydroxyapatite Crystals at Physiological Temperature. J. Cryst. Growth 2008, 310, 1749–1753. [Google Scholar] [CrossRef]
- Rajabi-Zamani, A.H.; Behnamghader, A.; Kazemzadeh, A. Synthesis of Nanocrystalline Carbonated Hydroxyapatite Powder via Nonalkoxide Sol–Gel Method. Mater. Sci. Eng. C 2008, 28, 1326–1329. [Google Scholar] [CrossRef]
- Быстрoва, А.В.; Bystrova, A.V. Мoделирoвание и Анализ Данных Синхрoтoрoннoгo Облучения Для Мoдифицирoваннoй Структуры Гидрoксиапатита. Математическая Биoлoгия и Биoинфoрматика 2014, 9, 171–182. [Google Scholar] [CrossRef]
- Barralet, J.; Knowles, J.C.; Best, S.; Bonfield, W. Thermal Decomposition of Synthesised Carbonate Hydroxyapatite. J. Mater. Sci. Mater. Med. 2002, 13, 529–533. [Google Scholar] [CrossRef]
- Dikeman, C.D.M. Encyclopedia of Meat Sciences; Devine, C., Dikeman, M., Jensen, W., Eds.; Academic Press: Cambridge, MA, USA, 2004; ISBN 9780080924441. [Google Scholar]
- Stull, D.D. Meat Processing. In The SAGE Encyclopedia of Food Issues; Kerry, J.P., Kerry, J.F., Ledward, D.A., Eds.; SAGE Publications, Inc.: Thousand Oaks, CA, USA, 2002; ISBN 9781855736665. [Google Scholar]
- Bernache-Assollant, D.; Ababou, A.; Champion, E.; Heughebaert, M. Sintering of Calcium Phosphate Hydroxyapatite Ca10(PO4)6(OH)2 I. Calcination and Particle Growth. J. Eur. Ceram. Soc. 2003, 23, 229–241. [Google Scholar] [CrossRef]
- Wang, T.; Dorner-Reisel, A. Thermo-Analytical Investigations of the Decomposition of Oxyhydroxyapatite. Mater. Lett. 2004, 58, 3025–3028. [Google Scholar] [CrossRef]
- Qu, H.; Wei, M. The Effect of Fluoride Contents in Fluoridated Hydroxyapatite on Osteoblast Behavior. Acta Biomater. 2006, 2, 113–119. [Google Scholar] [CrossRef]
- Dyshlovenko, S.; Pateyron, B.; Pawlowski, L.; Murano, D. Numerical Simulation of Hydroxyapatite Powder Behaviour in Plasma Jet. Surf. Coat. Technol. 2004, 179, 110–117. [Google Scholar] [CrossRef]
- Ślósarczyk, A. Bioceramika Hydroksyapatytowa; Ceramika 5; Polski Biuletyn Ceramiczny 13: Kraków, Poland, 1997; ISBN 8371080158/9788371080159. [Google Scholar]
- Zorn, F.; Weber, F.; Almeida, A.; Taubert, I.; Wagenknecht, R.; Eberle, W. Process for the Production of Spongiosa Bone Ceramic Having Low Calcium Oxide Content. Patent WO1996014886A1, 10 October 1941. [Google Scholar]
- Peters, F.; Schwarz, K.; Epple, M. The Structure of Bone Studied with Synchrotron X-ray Diffraction, X-ray Absorption Spectroscopy and Thermal Analysis. Thermochim. Acta 2000, 361, 131–138. [Google Scholar] [CrossRef]
- Fathi, M.H.; Hanifi, A.; Mortazavi, V. Preparation and Bioactivity Evaluation of Bone-like Hydroxyapatite Nanopowder. J. Mater. Process. Technol. 2008, 202, 536–542. [Google Scholar] [CrossRef]
- Liu, D.-M. Fabrication and Characterization of Porous Hydroxyapatite Granules. Biomaterials 1996, 17, 1955–1957. [Google Scholar] [CrossRef]
- Kim, I.-S.; Kumta, P.N. Sol–Gel Synthesis and Characterization of Nanostructured Hydroxyapatite Powder. Mater. Sci. Eng. B 2004, 111, 232–236. [Google Scholar] [CrossRef]
- Brodsky, B.; Werkmeister, J.A.; Ramshaw, J.A.M. Collagens and Gelatins. In Biopolymers Online; Fahnestock, S.R., Steinbüchel, A., Eds.; Wiley: Hoboken, NJ, USA, 2003. [Google Scholar]
- Ramshaw, J.A.M.; Peng, Y.Y.; Glattauer, V.; Werkmeister, J.A. Collagens as Biomaterials. J. Mater. Sci. Mater. Med. 2009, 20, 3–8. [Google Scholar] [CrossRef]
- An, B.; Lin, Y.-S.; Brodsky, B. Collagen Interactions: Drug Design and Delivery. Adv. Drug Deliv. Rev. 2016, 97, 69–84. [Google Scholar] [CrossRef]
- Jehle, K.S.; Rohatgi, A.; Baig, M.K. Use of Porcine Dermal Collagen Graft and Topical Negative Pressure on Infected Open Abdominal Wounds. J. Wound Care 2007, 16, 36–37. [Google Scholar] [CrossRef]
- Fleck, C.A.; Simman, R. Modern Collagen Wound Dressings: Function and Purpose. J. Am. Col. Certif. Wound Spec. 2010, 2, 50–54. [Google Scholar] [CrossRef]
- Ramshaw, J.A.; Werkmeister, J.A.; Dumsday, G.J. Bioengineered Collagens. Bioengineered 2014, 5, 227–233. [Google Scholar] [CrossRef]
- Kew, S.J.; Gwynne, J.H.; Enea, D.; Abu-Rub, M.; Pandit, A.; Zeugolis, D.; Brooks, R.A.; Rushton, N.; Best, S.M.; Cameron, R.E. Regeneration and Repair of Tendon and Ligament Tissue Using Collagen Fibre Biomaterials. Acta Biomater. 2011, 7, 3237–3247. [Google Scholar] [CrossRef]
- Dzobo, K.; Thomford, N.E.; Senthebane, D.A.; Shipanga, H.; Rowe, A.; Dandara, C.; Pillay, M.; Motaung, K.S.C.M. Advances in Regenerative Medicine and Tissue Engineering: Innovation and Transformation of Medicine. Stem Cells Int. 2018, 2018, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Ullah, S.; Chen, X. Fabrication, Applications and Challenges of Natural Biomaterials in Tissue Engineering. Appl. Mater. Today 2020, 20, 100656. [Google Scholar] [CrossRef]
- Thavornyutikarn, B.; Chantarapanich, N.; Sitthiseripratip, K.; Thouas, G.A.; Chen, Q. Bone Tissue Engineering Scaffolding: Computer-Aided Scaffolding Techniques. Prog. Biomater. 2014, 3, 61–102. [Google Scholar] [CrossRef] [PubMed]
- Giwa, S.; Lewis, J.K.; Alvarez, L.; Langer, R.; Roth, A.E.; Church, G.M.; Markmann, J.F.; Sachs, D.H.; Chandraker, A.; Wertheim, J.A.; et al. The Promise of Organ and Tissue Preservation to Transform Medicine. Nat. Biotechnol. 2017, 35, 530–542. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, M. Hydroxyapatite/Collagen Bone-Like Nanocomposite. Biol. Pharm. Bull. 2013, 36, 1666–1669. [Google Scholar] [CrossRef]
- Ramesh, N.; Moratti, S.C.; Dias, G.J. Hydroxyapatite-Polymer Biocomposites for Bone Regeneration: A Review of Current Trends. J. Biomed. Mater. Res. Part B Appl. Biomater. 2018, 106, 2046–2057. [Google Scholar] [CrossRef]
- Nakata, R.; Tachibana, A.; Tanabe, T. Preparation of Keratin Hydrogel/Hydroxyapatite Composite and Its Evaluation as a Controlled Drug Release Carrier. Mater. Sci. Eng. C 2014, 41, 59–64. [Google Scholar] [CrossRef]
- Chocholata, P.; Kulda, V.; Babuska, V. Fabrication of Scaffolds for Bone-Tissue Regeneration. Materials 2019, 12, 568. [Google Scholar] [CrossRef]
- Bakhtiar, H.; Mazidi, A.; Asl, M.S.; Ellini, M.R.; Moshiri, A.; Nekoofar, M.H.; Dummer, P.M.H. The Role of Stem Cell Therapy in Regeneration of Dentine-Pulp Complex: A Systematic Review. Prog. Biomater. 2018, 7, 249–268. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xu, S.; Zhou, S.; Xu, W.; Leary, M.; Choong, P.; Qian, M.; Brandt, M.; Xie, Y.M. Topological Design and Additive Manufacturing of Porous Metals for Bone Scaffolds and Orthopaedic Implants: A Review. Biomaterials 2016, 83, 127–141. [Google Scholar] [CrossRef]
- Park, K. Controlled Drug Delivery Systems: Past Forward and Future Back. J. Control. Release 2014, 190, 3–8. [Google Scholar] [CrossRef]
- Kohrs, N.J.; Liyanage, T.; Venkatesan, N.; Najarzadeh, A.; Puleo, D.A. Drug Delivery Systems and Controlled Release. In Encyclopedia of Biomedical Engineering; Elsevier: Amsterdam, The Netherlands, 2019; pp. 316–329. [Google Scholar]
- Wang, J.; Ni, Q.; Wang, Y.; Zhang, Y.; He, H.; Gao, D.; Ma, X.; Liang, X.-J. Nanoscale Drug Delivery Systems for Controllable Drug Behaviors by Multi-Stage Barrier Penetration. J. Control. Release 2021, 331, 282–295. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Y.; Shi, L. Controlled Drug Delivery Systems in Eradicating Bacterial Biofilm-Associated Infections. J. Control. Release 2020. [Google Scholar] [CrossRef]
- Huang, X.; Brazel, C.S. On the Importance and Mechanisms of Burst Release in Matrix-Controlled Drug Delivery Systems. J. Control. Release 2001, 73, 121–136. [Google Scholar] [CrossRef]
- Alizadeh-Osgouei, M.; Li, Y.; Wen, C. A Comprehensive Review of Biodegradable Synthetic Polymer-Ceramic Composites and Their Manufacture for Biomedical Applications. Bioact. Mater. 2019, 4, 22–36. [Google Scholar] [CrossRef]
- Mondal, S.; Pal, U. 3D Hydroxyapatite Scaffold for Bone Regeneration and Local Drug Delivery Applications. J. Drug Deliv. Sci. Technol. 2019, 53, 101131. [Google Scholar] [CrossRef]
- Liu, D.; Liang, L.; Regenstein, J.M.; Zhou, P. Extraction and Characterisation of Pepsin-Solubilised Collagen from Fins, Scales, Skins, Bones and Swim Bladders of Bighead Carp (Hypophthalmichthys Nobilis). Food Chem. 2012, 133, 1441–1448. [Google Scholar] [CrossRef]
- Pastorino, L.; Dellacasa, E.; Scaglione, S.; Giulianelli, M.; Sbrana, F.; Vassalli, M.; Ruggiero, C. Oriented Collagen Nanocoatings for Tissue Engineering. Colloids Surfaces B Biointerfaces 2014, 114, 372–378. [Google Scholar] [CrossRef]
- Prabhakaran, M.P.; Vatankhah, E.; Ramakrishna, S. Electrospun Aligned PHBV/Collagen Nanofibers as Substrates for Nerve Tissue Engineering. Biotechnol. Bioeng. 2013, 110, 2775–2784. [Google Scholar] [CrossRef]
- Kijeńska, E.; Prabhakaran, M.P.; Swieszkowski, W.; Kurzydlowski, K.J.; Ramakrishna, S. Electrospun Bio-Composite P(LLA-CL)/Collagen I/Collagen III Scaffolds for Nerve Tissue Engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100B, 1093–1102. [Google Scholar] [CrossRef]
- Wei, K.; Li, Y.; Lei, X.; Yang, H.; Teramoto, A.; Yao, J.; Abe, K.; Ko, F.K. Emulsion Electrospinning of a Collagen-Like Protein/PLGA Fibrous Scaffold: Empirical Modeling and Preliminary Release Assessment of Encapsulated Protein. Macromol. Biosci. 2011. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-C.; Tan, F.; Marra, K.G.; Jan, S.-S.; Liu, D.-C. Synthesis and Characterization of Collagen/Hyaluronan/Chitosan Composite Sponges for Potential Biomedical Applications. Acta Biomater. 2009, 5, 2591–2600. [Google Scholar] [CrossRef]
- Karadas, O.; Yucel, D.; Kenar, H.; Torun Kose, G.; Hasirci, V. Collagen Scaffolds with in Situ-Grown Calcium Phosphate for Osteogenic Differentiation of Wharton’s Jelly and Menstrual Blood Stem Cells. J. Tissue Eng. Regen. Med. 2012. [Google Scholar] [CrossRef] [PubMed]
- Fauzi, M.B.; Lokanathan, Y.; Aminuddin, B.S.; Ruszymah, B.H.I.; Chowdhury, S.R. Ovine Tendon Collagen: Extraction, Characterisation and Fabrication of Thin Films for Tissue Engineering Applications. Mater. Sci. Eng. C 2016, 68, 163–171. [Google Scholar] [CrossRef]
- Jadhav, S.B.; Jain, G.K. Statins and Osteoporosis: New Role for Old Drugs. J. Pharm. Pharmacol. 2006, 58, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-B. The Use of Simvastatin in Bone Regeneration. Med. Oral Patol. Oral Cir. Bucal 2009, 14, e485–e488. [Google Scholar]
- Alam, S.; Ueki, K.; Nakagawa, K.; Marukawa, K.; Hashiba, Y.; Yamamoto, E.; Sakulsak, N.; Iseki, S. Statin-Induced Bone Morphogenetic Protein (BMP) 2 Expression during Bone Regeneration: An Immunohistochemical Study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 2009, 107, 22–29. [Google Scholar] [CrossRef]
- Sonobe, M.; Hattori, K.; Tomita, N.; Yoshikawa, T.; Aoki, H.; Takakura, Y.; Suguro, T. Stimulatory Effects of Statins on Bone Marrow-Derived Mesenchymal Stem Cells. Study of a New Therapeutic Agent for Fracture. Biomed. Mater. Eng. 2005, 15, 261–267. [Google Scholar]
- Gao, S.; Shiota, M.; Fujii, M.; Chen, K.; Shimogishi, M.; Sato, M.; Kasugai, S. Combination of Simvastatin and Hydroxyapatite Fiber Induces Bone Augmentation. Open J. Regen. Med. 2013, 2, 53–60. [Google Scholar] [CrossRef]
- Sun, T.; Zhan, B.; Qin, D.; Wang, B.; Gao, Y. Evaluation on the Anti-Oxidation Effects of a Thermosensitive Chitosan-Based Hydrogel Loaded Antioxidin-RL. J. Control. Release 2017, 259, e129–e130. [Google Scholar] [CrossRef]
- Zong, Y.; Wu, J.; Shen, K. Nanoparticle Albumin-Bound Paclitaxel as Neoadjuvant Chemotherapy of Breast Cancer: A Systematic Review and Meta-Analysis. Oncotarget 2017, 8, 17360–17372. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, D.N.; Roriz, V.M.; de Oliveira, G.J.P.L.; Duarte, W.R.; Pinto, L.N.M.P.; Vianna, L.M.d.S.; Carneiro, F.P.; Ferreira, V.M.M. Local Effect of Simvastatin Combined with Different Osteoconductive Biomaterials and Collagen Sponge on New Bone Formation in Critical Defects in Rat Calvaria. Acta Cir. Bras. 2020, 35. [Google Scholar] [CrossRef]
- Monjo, M.; Rubert, M.; Wohlfahrt, J.C.; Rønold, H.J.; Ellingsen, J.E.; Lyngstadaas, S.P. In Vivo Performance of Absorbable Collagen Sponges with Rosuvastatin in Critical-Size Cortical Bone Defects. Acta Biomater. 2010, 6, 1405–1412. [Google Scholar] [CrossRef]
- Shepherd, J.; Hunninghake, D.B.; Stein, E.A.; Kastelein, J.J.P.; Harris, S.; Pears, J.; Hutchinson, H.G. Safety of Rosuvastatin. Am. J. Cardiol. 2004, 94, 882–888. [Google Scholar] [CrossRef]
- Zhu, L.; Chen, L. Progress in Research on Paclitaxel and Tumor Immunotherapy. Cell. Mol. Biol. Lett. 2019, 24, 40. [Google Scholar] [CrossRef]
- Yardley, D.A. Nab-Paclitaxel Mechanisms of Action and Delivery. J. Control. Release 2013, 170, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Weaver, B.A. How Taxol/Paclitaxel Kills Cancer Cells. Mol. Biol. Cell 2014, 25, 2677–2681. [Google Scholar] [CrossRef]
- Bernabeu, E.; Cagel, M.; Lagomarsino, E.; Moretton, M.; Chiappetta, D.A. Paclitaxel: What Has Been Done and the Challenges Remain Ahead. Int. J. Pharm. 2017, 526, 474–495. [Google Scholar] [CrossRef]
- Lopez-Heredia, M.A.; Bernard Kamphuis, G.J.; Thüne, P.C.; Cumhur Öner, F.; Jansen, J.A.; Frank Walboomers, X. An Injectable Calcium Phosphate Cement for the Local Delivery of Paclitaxel to Bone. Biomaterials 2011, 32, 5411–5416. [Google Scholar] [CrossRef]
- Venkatasubbu, G.; Ramasamy, S.; Kumar, J.; Reddy, P. Acute and Subchronic Toxicity Analysis of Surface Modified Paclitaxel Attached Hydroxyapatite and Titanium Dioxide Nanoparticles. Int. J. Nanomed. 2015, 137. [Google Scholar] [CrossRef] [PubMed]
- Venkatasubbu, G.D.; Ramasamy, S.; Reddy, G.P.; Kumar, J. In Vitro and In Vivo Anticancer Activity of Surface Modified Paclitaxel Attached Hydroxyapatite and Titanium Dioxide Nanoparticles. Biomed. Microdevices 2013, 15, 711–726. [Google Scholar] [CrossRef]
- Lai, Y.-L.; Lai, S.-B.; Yen, S.-K. Paclitaxel/Hydroxyapatite Composite Coatings on Titanium Alloy for Biomedical Applications. Mater. Sci. Eng. C 2017, 79, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Venkatasubbu, G.D.; Ramasamy, S.; Avadhani, G.S.; Ramakrishnan, V.; Kumar, J. Surface Modification and Paclitaxel Drug Delivery of Folic Acid Modified Polyethylene Glycol Functionalized Hydroxyapatite Nanoparticles. Powder Technol. 2013, 235, 437–442. [Google Scholar] [CrossRef]
- Srivastav, A.; Chandanshive, B.; Dandekar, P.; Khushalani, D.; Jain, R. Biomimetic Hydroxyapatite a Potential Universal Nanocarrier for Cellular Internalization & Drug Delivery. Pharm. Res. 2019, 36, 60. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, T.; Ikoma, T.; Monkawa, A.; Yunoki, S.; Abe, T.; Sakane, M.; Tanaka, M. Preparation of Hydroxyapatite-Alginate Gels as a Carrier for Controlled Release of Paclitaxel. Key Eng. Mater. 2007, 330–332, 1053–1056. [Google Scholar] [CrossRef]
- Watanabe, K.; Nishio, Y.; Makiura, R.; Nakahira, A.; Kojima, C. Paclitaxel-Loaded Hydroxyapatite/Collagen Hybrid Gels as Drug Delivery Systems for Metastatic Cancer Cells. Int. J. Pharm. 2013, 446, 81–86. [Google Scholar] [CrossRef]
- Gardete, S.; Tomasz, A. Mechanisms of Vancomycin Resistance in Staphylococcus Aureus. J. Clin. Investig. 2014, 124, 2836–2840. [Google Scholar] [CrossRef]
- Grupper, M.; Nicolau, D.P. Obesity and Skin and Soft Tissue Infections. Curr. Opin. Infect. Dis. 2017, 30, 180–191. [Google Scholar] [CrossRef]
- Tian, Y.; Huang, X.; Wu, L.-J.; Yi, L.; Li, M.; Gu, S.-C.; Guo, D.-J.; Zhan, Q.-Y. Pneumonia Caused by Community-Acquired Methicillin-Resistant Staphylococcus Aureus. Chin. Med. J. 2018, 131, 2002–2004. [Google Scholar] [CrossRef]
- Chin, S.J.; Moore, G.A.; Zhang, M.; Clarke, H.D.; Spangehl, M.J.; Young, S.W. The AAHKS Clinical Research Award: Intraosseous Regional Prophylaxis Provides Higher Tissue Concentrations in High BMI Patients in Total Knee Arthroplasty: A Randomized Trial. J. Arthroplast. 2018, 33, S13–S18. [Google Scholar] [CrossRef]
- Baddour, L.M.; Wilson, W.R.; Bayer, A.S.; Fowler, V.G.; Tleyjeh, I.M.; Rybak, M.J.; Barsic, B.; Lockhart, P.B.; Gewitz, M.H.; Levison, M.E.; et al. Infective Endocarditis in Adults: Diagnosis, Antimicrobial Therapy, and Management of Complications. Circulation 2015, 132, 1435–1486. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.; Qu, H.; Zhang, G.; Zhang, X. Osteogenic and Antibacterial Properties of Vancomycin-Laden Mesoporous Bioglass/PLGA Composite Scaffolds for Bone Regeneration in Infected Bone Defects. Artif. Cells Nanomed. Biotechnol. 2017, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Jian, C.; Xu, F.; Bao, T.; Lan, S.; Wu, G.; Qi, B.; Bai, Z.; Yu, A. Vancomycin–Impregnated Electrospun Polycaprolactone (PCL) Membrane for the Treatment of Infected Bone Defects: An Animal Study. J. Biomater. Appl. 2018, 32, 1187–1196. [Google Scholar] [CrossRef]
- Zanjanizadeh Ezazi, N.; Shahbazi, M.-A.; Shatalin, Y.V.; Nadal, E.; Mäkilä, E.; Salonen, J.; Kemell, M.; Correia, A.; Hirvonen, J.; Santos, H.A. Conductive Vancomycin-Loaded Mesoporous Silica Polypyrrole-Based Scaffolds for Bone Regeneration. Int. J. Pharm. 2018, 536, 241–250. [Google Scholar] [CrossRef]
- Ionita, D.; Bajenaru-Georgescu, D.; Totea, G.; Mazare, A.; Schmuki, P.; Demetrescu, I. Activity of Vancomycin Release from Bioinspired Coatings of Hydroxyapatite or TiO 2 Nanotubes. Int. J. Pharm. 2017, 517, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Parent, M.; Magnaudeix, A.; Delebassée, S.; Sarre, E.; Champion, E.; Viana Trecant, M.; Damia, C. Hydroxyapatite Microporous Bioceramics as Vancomycin Reservoir: Antibacterial Efficiency and Biocompatibility Investigation. J. Biomater. Appl. 2016, 31, 488–498. [Google Scholar] [CrossRef]
- Thapa, R.K.; Kiick, K.L.; Sullivan, M.O. Encapsulation of Collagen Mimetic Peptide-Tethered Vancomycin Liposomes in Collagen-Based Scaffolds for Infection Control in Wounds. Acta Biomater. 2020, 103, 115–128. [Google Scholar] [CrossRef]
- Suchý, T.; Šupová, M.; Klapková, E.; Horný, L.; Rýglová, Š.; Žaloudková, M.; Braun, M.; Sucharda, Z.; Ballay, R.; Veselý, J.; et al. The Sustainable Release of Vancomycin and Its Degradation Products From Nanostructured Collagen/Hydroxyapatite Composite Layers. J. Pharm. Sci. 2016, 105, 1288–1294. [Google Scholar] [CrossRef]
- Lian, X.; Mao, K.; Liu, X.; Wang, X.; Cui, F. In Vivo Osteogenesis of Vancomycin Loaded Nanohydroxyapatite/Collagen/Calcium Sulfate Composite for Treating Infectious Bone Defect Induced by Chronic Osteomyelitis. J. Nanomater. 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Lian, X.; Liu, H.; Wang, X.; Xu, S.; Cui, F.; Bai, X. Antibacterial and Biocompatible Properties of Vancomycin-Loaded Nano-Hydroxyapatite/Collagen/Poly (Lactic Acid) Bone Substitute. Prog. Nat. Sci. Mater. Int. 2013, 23, 549–556. [Google Scholar] [CrossRef]
- Egawa, S.; Hirai, K.; Matsumoto, R.; Yoshii, T.; Yuasa, M.; Okawa, A.; Sugo, K.; Sotome, S. Efficacy of Antibiotic-Loaded Hydroxyapatite/Collagen Composites Is Dependent on Adsorbability for Treating Staphylococcus Aureus Osteomyelitis in Rats. J. Orthop. Res. 2020, 38, 843–851. [Google Scholar] [CrossRef]
- Suchý, T.; Šupová, M.; Klapková, E.; Adamková, V.; Závora, J.; Žaloudková, M.; Rýglová, Š.; Ballay, R.; Denk, F.; Pokorný, M.; et al. The Release Kinetics, Antimicrobial Activity and Cytocompatibility of Differently Prepared Collagen/Hydroxyapatite/Vancomycin Layers: Microstructure vs. Nanostructure. Eur. J. Pharm. Sci. 2017, 100, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Coelho, C.C.; Sousa, S.R.; Monteiro, F.J. Heparinized Nanohydroxyapatite/Collagen Granules for Controlled Release of Vancomycin. J. Biomed. Mater. Res. Part A 2015, 103, 3128–3138. [Google Scholar] [CrossRef] [PubMed]
- Chopra, I.; Roberts, M. Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Microbiol. Mol. Biol. Rev. 2001, 65, 232–260. [Google Scholar] [CrossRef]
- Bahrami, F.; Morris, D.L.; Pourgholami, M.H. Tetracyclines: Drugs with Huge Therapeutic Potential. Mini Rev. Med. Chem. 2012, 12, 44–52. [Google Scholar] [CrossRef]
- Grossman, T.H. Tetracycline Antibiotics and Resistance. Cold Spring Harb. Perspect. Med. 2016, 6, a025387. [Google Scholar] [CrossRef] [PubMed]
- Schwake-Anduschus, C.; Langenkämper, G. Chlortetracycline and Related Tetracyclines: Detection in Wheat and Rye Grain. J. Sci. Food Agric. 2018, 98, 4542–4549. [Google Scholar] [CrossRef]
- Chopra, I. Glycylcyclines: Third-Generation Tetracycline Antibiotics. Curr. Opin. Pharmacol. 2001, 1, 464–469. [Google Scholar] [CrossRef]
- Harja, M.; Ciobanu, G. Studies on Adsorption of Oxytetracycline from Aqueous Solutions onto Hydroxyapatite. Sci. Total Environ. 2018, 628–629, 36–43. [Google Scholar] [CrossRef]
- Henehan, M.; Montuno, M.; De Benedetto, A. Doxycycline as an Anti-Inflammatory Agent: Updates in Dermatology. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 1800–1808. [Google Scholar] [CrossRef]
- Garrido-Mesa, N.; Zarzuelo, A.; Gálvez, J. Minocycline: Far beyond an Antibiotic. Br. J. Pharmacol. 2013, 169, 337–352. [Google Scholar] [CrossRef]
- Alikhan, A.; Kurek, L.; Feldman, S.R. The Role of Tetracyclines in Rosacea. Am. J. Clin. Dermatol. 2010, 11, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Gomes, P.S.; Santos, J.D.; Fernandes, M.H. Cell-Induced Response by Tetracyclines on Human Bone Marrow Colonized Hydroxyapatite and Bonelike®. Acta Biomater. 2008, 4, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Rabiee, S.M. Development of Hydroxyapatite Bone Cement for Controlled Drug Release via Tetracycline Hydrochloride. Bull. Mater. Sci. 2013, 36, 171–174. [Google Scholar] [CrossRef]
- Rusu, L.C.; Nedelcu, I.A.; Georgiana Albu, M.; Sonmez, M.; Voicu, G.; Radulescu, M.; Ficai, D.; Ficai, A.; Negrutiu, M.-L.; Sinescu, C. Tetracycline Loaded Collagen/Hydroxyapatite Composite Materials for Biomedical Applications. J. Nanomater. 2015, 2015, 1–5. [Google Scholar] [CrossRef]
- Song, W.; Yu, X.; Markel, D.C.; Shi, T.; Ren, W. Coaxial PCL/PVA Electrospun Nanofibers: Osseointegration Enhancer and Controlled Drug Release Device. Biofabrication 2013, 5, 035006. [Google Scholar] [CrossRef]
- Semyari, H.; Salehi, M.; Taleghani, F.; Ehterami, A.; Bastami, F.; Jalayer, T.; Semyari, H.; Hamed Nabavi, M.; Semyari, H. Fabrication and Characterization of Collagen–Hydroxyapatite-Based Composite Scaffolds Containing Doxycycline via Freeze-Casting Method for Bone Tissue Engineering. J. Biomater. Appl. 2018, 33, 501–513. [Google Scholar] [CrossRef]
- Mederle, N.; Marin, S.; Marin, M.M.; Danila, E.; Mederle, O.; Albu Kaya, M.G.; Ghica, M.V. Innovative Biomaterials Based on Collagen-Hydroxyapatite and Doxycycline for Bone Regeneration. Adv. Mater. Sci. Eng. 2016, 2016, 1–5. [Google Scholar] [CrossRef][Green Version]
- Martin, V.; Ribeiro, I.A.; Alves, M.M.; Gonçalves, L.; Claudio, R.A.; Grenho, L.; Fernandes, M.H.; Gomes, P.; Santos, C.F.; Bettencourt, A.F. Engineering a Multifunctional 3D-Printed PLA-Collagen-Minocycline-NanoHydroxyapatite Scaffold with Combined Antimicrobial and Osteogenic Effects for Bone Regeneration. Mater. Sci. Eng. C 2019, 101, 15–26. [Google Scholar] [CrossRef]
- Crcek, M.; Zdovc, J.; Kerec Kos, M. A Review of Population Pharmacokinetic Models of Gentamicin in Paediatric Patients. J. Clin. Pharm. Ther. 2019, 44, 659–674. [Google Scholar] [CrossRef] [PubMed]
- Hayward, R.S.; Harding, J.; Molloy, R.; Land, L.; Longcroft-Neal, K.; Moore, D.; Ross, J.D.C. Adverse Effects of a Single Dose of Gentamicin in Adults: A Systematic Review. Br. J. Clin. Pharmacol. 2018, 84, 223–238. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chen, Y.; Wu, P.; Chen, B. Update on New Medicinal Applications of Gentamicin: Evidence-Based Review. J. Formos. Med. Assoc. 2014, 113, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.C.; Srinivasjois, R.; Moon, K. One Dose per Day Compared to Multiple Doses per Day of Gentamicin for Treatment of Suspected or Proven Sepsis in Neonates. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef]
- Byrnes, M.; Dorman, R. Use of Gentamicin as Empiric Coverage for Ventilator-Associated Pneumonia: The “Con” Perspective. Surg. Infect. 2016, 17, 334–336. [Google Scholar] [CrossRef]
- Shahpari, O.; Mousavian, A.; Elahpour, N.; Malahias, M.-A.; Ebrahimzadeh, M.H.; Moradi, A. The Use of Antibiotic Impregnated Cement Spacers in the Treatment of Infected Total Joint Replacement: Challenges and Achievements. Arch. Bone Jt. Surg. 2020, 8, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Moskowitz, J.S.; Blaisse, M.R.; Samuel, R.E.; Hsu, H.-P.; Harris, M.B.; Martin, S.D.; Lee, J.C.; Spector, M.; Hammond, P.T. The Effectiveness of the Controlled Release of Gentamicin from Polyelectrolyte Multilayers in the Treatment of Staphylococcus Aureus Infection in a Rabbit Bone Model. Biomaterials 2010, 31, 6019–6030. [Google Scholar] [CrossRef]
- Oshima, S.; Sato, T.; Honda, M.; Suetsugu, Y.; Ozeki, K.; Kikuchi, M. Fabrication of Gentamicin-Loaded Hydroxyapatite/Collagen Bone-Like Nanocomposite for Anti-Infection Bone Void Fillers. Int. J. Mol. Sci. 2020, 21, 551. [Google Scholar] [CrossRef]
- Amaro Martins, V.C.; Goissis, G. Nonstoichiometric Hydroxyapatite-Anionic Collagen Composite as Support for the Double Sustained Release of Gentamicin and Norfloxacin/Ciprofloxacin. Artif. Organs 2000, 24, 224–230. [Google Scholar] [CrossRef]
- Ionescu (Filip), O.; Ciocilteu, M.V.; Manda, C.V.; Neacsu, I.A.; Ficai, A.; Amzoiu, E.; Stiolica, A.T.; Croitoru, O.; Neamtu, J. Bone-Graft Delivery Systems of Type PLGA- Gentamicin and Collagen-Hydroxyapatite-Gentamicine. Mater. Plast. 2019, 56, 534–537. [Google Scholar] [CrossRef]
- Suchý, T.; Šupová, M.; Sauerová, P.; Hubálek Kalbáčová, M.; Klapková, E.; Pokorný, M.; Horný, L.; Závora, J.; Ballay, R.; Denk, F.; et al. Evaluation of Collagen/Hydroxyapatite Electrospun Layers Loaded with Vancomycin, Gentamicin and Their Combination: Comparison of Release Kinetics, Antimicrobial Activity and Cytocompatibility. Eur. J. Pharm. Biopharm. 2019, 140, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Boanini, E.; Gazzano, M.; Rubini, K.; Bigi, A. Composite Nanocrystals Provide New Insight on Alendronate Interaction with Hydroxyapatite Structure. Adv. Mater. 2007, 19, 2499–2502. [Google Scholar] [CrossRef]
- Boanini, E.; Torricelli, P.; Gazzano, M.; Giardino, R.; Bigi, A. Alendronate-Hydroxyapatite Nanocomposites and Their Interaction with Osteoclasts and Osteoblast-like Cells. Biomaterials 2008, 29, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Bigi, A.; Boanini, E.; Capuccini, C.; Fini, M.; Mihailescu, I.N.; Ristoscu, C.; Sima, F.; Torricelli, P. Biofunctional Alendronate-Hydroxyapatite Thin Films Deposited by Matrix Assisted Pulsed Laser Evaporation. Biomaterials 2009, 30, 6168–6177. [Google Scholar] [CrossRef]
- Ma, X.; He, Z.; Han, F.; Zhong, Z.; Chen, L.; Li, B. Preparation of Collagen/Hydroxyapatite/Alendronate Hybrid Hydrogels as Potential Scaffolds for Bone Regeneration. Colloids Surfaces B Biointerfaces 2016, 143, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Sugata, Y.; Sotome, S.; Yuasa, M.; Hirano, M.; Shinomiya, K.; Okawa, A. Effects of the Systemic Administration of Alendronate on Bone Formation in a Porous Hydroxyapatite/Collagen Composite and Resorption by Osteoclasts in a Bone Defect Model in Rabbits. J. Bone Jt. Surg. 2011, 93, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.-Y.; Cui, Y.-W.; Zhang, L.-C. Recent Development in Beta Titanium Alloys for Biomedical Applications. Metals 2020, 10, 1139. [Google Scholar] [CrossRef]
- Galindo, T.G.P.; Chai, Y.; Tagaya, M. Hydroxyapatite Nanoparticle Coating on Polymer for Constructing Effective Biointeractive Interfaces. J. Nanomater. 2019, 2019, 1–23. [Google Scholar] [CrossRef]
- Okada, M.; Matsumoto, T. Synthesis and Modification of Apatite Nanoparticles for Use in Dental and Medical Applications. Jpn. Dent. Sci. Rev. 2015, 51, 85–95. [Google Scholar] [CrossRef]
- Cardoso, V.S.; Quelemes, P.V.; Amorin, A.; Primo, F.L.; Gobo, G.G.; Tedesco, A.C.; Mafud, A.C.; Mascarenhas, Y.P.; Corrêa, J.R.; Kuckelhaus, S.A.; et al. Collagen-Based Silver Nanoparticles for Biological Applications: Synthesis and Characterization. J. Nanobiotechnology 2014, 12, 36. [Google Scholar] [CrossRef]
- Galluzzi, L.; Vitale, I.; Michels, J.; Brenner, C.; Szabadkai, G.; Harel-Bellan, A.; Castedo, M.; Kroemer, G. Systems Biology of Cisplatin Resistance: Past, Present and Future. Cell Death Dis. 2014, 5, e1257. [Google Scholar] [CrossRef]
- Manohar, S.; Leung, N. Cisplatin Nephrotoxicity: A Review of the Literature. J. Nephrol. 2018, 31, 15–25. [Google Scholar] [CrossRef]
- Perše, M.; Večerić-Haler, Ž. Cisplatin-Induced Rodent Model of Kidney Injury: Characteristics and Challenges. Biomed. Res. Int. 2018, 2018, 1–29. [Google Scholar] [CrossRef]
- Dasari, S.; Bernard Tchounwou, P. Cisplatin in Cancer Therapy: Molecular Mechanisms of Action. Eur. J. Pharmacol. 2014, 740, 364–378. [Google Scholar] [CrossRef]
- Amable, L. Cisplatin Resistance and Opportunities for Precision Medicine. Pharmacol. Res. 2016, 106, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.-Y.; Chen, M.-H.; Kuo, W.-T.; Sun, Y.-J.; Lin, F.-H. The Characterization and Evaluation of Cisplatin-Loaded Magnetite–Hydroxyapatite Nanoparticles (MHAp/CDDP) as Dual Treatment of Hyperthermia and Chemotherapy for Lung Cancer Therapy. Ceram. Int. 2015, 41, 2399–2410. [Google Scholar] [CrossRef]
- Barroug, A.; Glimcher, M.J. Hydroxyapatite Crystals as a Local Delivery System for Cisplatin: Adsorption and Release of Cisplatin in Vitro. J. Orthop. Res. 2002, 20, 274–280. [Google Scholar] [CrossRef]
- Palazzo, B.; Iafisco, M.; Laforgia, M.; Margiotta, N.; Natile, G.; Bianchi, C.L.; Walsh, D.; Mann, S.; Roveri, N. Biomimetic Hydroxyapatite-Drug Nanocrystals as Potential Bone Substitutes with Antitumor Drug Delivery Properties. Adv. Funct. Mater. 2007, 17, 2180–2188. [Google Scholar] [CrossRef]
- Andronescu, E.; Ficai, A.; Albu, M.G.; Mitran, V.; Sonmez, M.; Ficai, D.; Ion, R.; Cimpean, A. Collagen-Hydroxyapatite/Cisplatin Drug Delivery Systems for Locoregional Treatment of Bone Cancer. Technol. Cancer Res. Treat. 2013, 12, 275–284. [Google Scholar] [CrossRef]
- Ficai, A.; Sonmez, M.; Albu, M.G.; Mihaiescu, D.E.; Ficai, D.; Bleotu, C. Antitumoral Materials with Regenerative Function Obtained Using a Layer-by-Layer Technique. Drug Des. Dev. Ther. 2015, 1269. [Google Scholar] [CrossRef]
- Rafieepour, A.; Azari, M.R.; Peirovi, H.; Khodagholi, F.; Jaktaji, J.P.; Mehrabi, Y.; Naserzadeh, P.; Mohammadian, Y. Investigation of the Effect of Magnetite Iron Oxide Particles Size on Cytotoxicity in A 549 Cell Line. Toxicol. Ind. Health 2019, 35, 703–713. [Google Scholar] [CrossRef]
- Mos, Y.M.; Zorzano, K.B.; Buisman, C.J.N.; Weijma, J. Magnetite Synthesis from Ferrous Iron Solution at PH 6.8 in a Continuous Stirred Tank Reactor. Water Sci. Technol. 2018, 77, 1870–1878. [Google Scholar] [CrossRef] [PubMed]
- Abe, H.; Naka, T.; Sato, K.; Suzuki, Y.; Nakano, M. Shape-Controlled Syntheses of Magnetite Microparticles and Their Magnetorheology. Int. J. Mol. Sci. 2019, 20, 3617. [Google Scholar] [CrossRef] [PubMed]
- Roca, A.G.; Morales, M.P.; Serna, C.J. Synthesis of Monodispersed Magnetite Particles From Different Organometallic Precursors. IEEE Trans. Magn. 2006, 42, 3025–3029. [Google Scholar] [CrossRef]
- Wallyn, J.; Anton, N.; Vandamme, T.F. Synthesis, Principles, and Properties of Magnetite Nanoparticles for In Vivo Imaging Applications—A Review. Pharmaceutics 2019, 11, 601. [Google Scholar] [CrossRef] [PubMed]
- Hariyanto, Y.A.; Taufiq, A.; Sunaryono; Soontaranon, S. Investigation on the Three-Dimensional Nanostructure and the Optical Properties of Hydroxyapatite/Magnetite Nanocomposites Prepared from Natural Resources. J. Korean Phys. Soc. 2019, 75, 708–715. [Google Scholar] [CrossRef]
- Iwasaki, T.; Nakatsuka, R.; Murase, K.; Takata, H.; Nakamura, H.; Watano, S. Simple and Rapid Synthesis of Magnetite/Hydroxyapatite Composites for Hyperthermia Treatments via a Mechanochemical Route. Int. J. Mol. Sci. 2013, 14, 9365–9378. [Google Scholar] [CrossRef]
- Tampieri, A.; Iafisco, M.; Sandri, M.; Panseri, S.; Cunha, C.; Sprio, S.; Savini, E.; Uhlarz, M.; Herrmannsdörfer, T. Magnetic Bioinspired Hybrid Nanostructured Collagen–Hydroxyapatite Scaffolds Supporting Cell Proliferation and Tuning Regenerative Process. ACS Appl. Mater. Interfaces 2014, 6, 15697–15707. [Google Scholar] [CrossRef]
- Heidari, F.; Bahrololoom, M.E.; Vashaee, D.; Tayebi, L. In Situ Preparation of Iron Oxide Nanoparticles in Natural Hydroxyapatite/Chitosan Matrix for Bone Tissue Engineering Application. Ceram. Int. 2015, 41, 3094–3100. [Google Scholar] [CrossRef]
- Zaborowska, M.; Bodin, A.; Bäckdahl, H.; Popp, J.; Goldstein, A.; Gatenholm, P. Microporous Bacterial Cellulose as a Potential Scaffold for Bone Regeneration. Acta Biomater. 2010, 6, 2540–2547. [Google Scholar] [CrossRef]
- Andronescu, E.; Ficai, M.; Voicu, G.; Ficai, D.; Maganu, M.; Ficai, A. Synthesis and Characterization of Collagen/Hydroxyapatite: Magnetite Composite Material for Bone Cancer Treatment. J. Mater. Sci. Mater. Med. 2010, 21, 2237–2242. [Google Scholar] [CrossRef]
- Yu, M.F.; Lourie, O.; Dyer, M.J.; Moloni, K.; Kelly, T.F.; Ruoff, R.S. Strength and Breaking Mechanism of Multiwalled Carbon Nanotubes under Tensile Load. Science 2000, 287, 637–640. [Google Scholar] [CrossRef]
- Lau, K.-T.; Hui, D. Effectiveness of Using Carbon Nanotubes as Nano-Reinforcements for Advanced Composite Structures. Carbon 2002, 40, 1605–1606. [Google Scholar] [CrossRef]
- Thostenson, E.T.; Ren, Z.; Chou, T.-W. Advances in the Science and Technology of Carbon Nanotubes and Their Composites: A Review. Compos. Sci. Technol. 2001, 61, 1899–1912. [Google Scholar] [CrossRef]
- Liao, S.; Xu, G.; Wang, W.; Watari, F.; Cui, F.; Ramakrishna, S.; Chan, C.K. Self-Assembly of Nano-Hydroxyapatite on Multi-Walled Carbon Nanotubes. Acta Biomater. 2007, 3, 669–675. [Google Scholar] [CrossRef] [PubMed]
- Zanello, L.P.; Zhao, B.; Hu, H.; Haddon, R.C. Bone Cell Proliferation on Carbon Nanotubes. Nano Lett. 2006, 6, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.-Y.; Han, T.H.; Hong, J.; Kim, J.E.; Lee, S.H.; Kim, H.W.; Kim, S.O. Noncovalent Functionalization of Graphene with End-Functional Polymers. J. Mater. Chem. 2010, 20, 1907. [Google Scholar] [CrossRef]
- Lin, T.; Bajpai, V.; Ji, T.; Dai, L. Chemistry of Carbon Nanotubes. Aust. J. Chem. 2003, 56, 635. [Google Scholar] [CrossRef]
- Tasis, D.; Tagmatarchis, N.; Bianco, A.; Prato, M. Chemistry of Carbon Nanotubes. Chem. Rev. 2006, 106, 1105–1136. [Google Scholar] [CrossRef]
- Liu, Z.-J.; Yuan, Z.-Y.; Zhou, W.; Peng, L.-M.; Xu, Z. Co/Carbon-Nanotube Monometallic System: The Effects of Oxidation by Nitric Acid. Phys. Chem. Chem. Phys. 2001, 3, 2518–2521. [Google Scholar] [CrossRef]
- Kealley, C.; Elcombe, M.; Van Riessen, A.; Ben-Nissan, B. Neutron Characterisation of Hydroxyapatite Bioceramics. Key Eng. Mater. 2006, 309–311, 61–64. [Google Scholar] [CrossRef]
- Wei, G.; Gong, C.; Hu, K.; Wang, Y.; Zhang, Y. Biomimetic Hydroxyapatite on Graphene Supports for Biomedical Applications: A Review. Nanomaterials 2019, 9, 1435. [Google Scholar] [CrossRef] [PubMed]
- Jing, Z.; Wu, Y.; Su, W.; Tian, M.; Jiang, W.; Cao, L.; Zhao, L.; Zhao, Z. Carbon Nanotube Reinforced Collagen/Hydroxyapatite Scaffolds Improve Bone Tissue Formation In Vitro and In Vivo. Ann. Biomed. Eng. 2017, 45, 2075–2087. [Google Scholar] [CrossRef] [PubMed]
- Türk, S.; Altınsoy, I.; Çelebi Efe, G.; Ipek, M.; Özacar, M.; Bindal, C. 3D Porous Collagen/Functionalized Multiwalled Carbon Nanotube/Chitosan/Hydroxyapatite Composite Scaffolds for Bone Tissue Engineering. Mater. Sci. Eng. C 2018, 92, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, E.E.; Della Colleta, H.H.M.; Ferlauto, A.S.; Moreira, R.L.; Resende, R.R.; Oliveira, S.; Kitten, G.T.; Lacerda, R.G.; Ladeira, L.O. Nanostructured 3-D Collagen/Nanotube Biocomposites for Future Bone Regeneration Scaffolds. Nano Res. 2009, 2, 462–473. [Google Scholar] [CrossRef]
- Li, M.; Xiong, P.; Yan, F.; Li, S.; Ren, C.; Yin, Z.; Li, A.; Li, H.; Ji, X.; Zheng, Y.; et al. An Overview of Graphene-Based Hydroxyapatite Composites for Orthopedic Applications. Bioact. Mater. 2018, 3, 1–18. [Google Scholar] [CrossRef]
- Crowder, S.W.; Prasai, D.; Rath, R.; Balikov, D.A.; Bae, H.; Bolotin, K.I.; Sung, H.-J. Three-Dimensional Graphene Foams Promote Osteogenic Differentiation of Human Mesenchymal Stem Cells. Nanoscale 2013, 5, 4171. [Google Scholar] [CrossRef]
- Baradaran, S.; Moghaddam, E.; Basirun, W.J.; Mehrali, M.; Sookhakian, M.; Hamdi, M.; Moghaddam, M.R.N.; Alias, Y. Mechanical Properties and Biomedical Applications of a Nanotube Hydroxyapatite-Reduced Graphene Oxide Composite. Carbon 2014, 69, 32–45. [Google Scholar] [CrossRef]
- Klébert, S.; Balázsi, C.; Balázsi, K.; Bódis, E.; Fazekas, P.; Keszler, A.M.; Szépvölgyi, J.; Károly, Z. Spark Plasma Sintering of Graphene Reinforced Hydroxyapatite Composites. Ceram. Int. 2015, 41, 3647–3652. [Google Scholar] [CrossRef]
- Li, M.; Wang, Y.; Liu, Q.; Li, Q.; Cheng, Y.; Zheng, Y.; Xi, T.; Wei, S. In Situ Synthesis and Biocompatibility of Nano Hydroxyapatite on Pristine and Chitosan Functionalized Graphene Oxide. J. Mater. Chem. B 2013, 1, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Singh Yadav, B.; Kumar Sahu, R.; Kumar Pramanick, A.; Mishra, T.; Alam, A.; Bharti, M.; Mukherjee, S.; Kumar, S.; Nayar, S. Collagen Functionalized Graphene Sheets Decorated with in Situ Synthesized Nano Hydroxyapatite Electrospun into Fibers. Mater. Today Commun. 2019, 18, 167–175. [Google Scholar] [CrossRef]
- Zakharov, N.A.; Ezhova, Z.A.; Koval, E.M.; Tkachev, A.G.; Kuznetsov, N.T. Calcium Hydroxyapatite in Hydroxyapatite/Graphene Oxide/Collagen Nanohybrids. Russ. J. Inorg. Chem. 2015, 60, 1467–1480. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Z.; Su, G.; Sun, X.; Wang, Y.; Fang, Z.; Chen, M.; Zhang, Q. Graphene Oxide Incorporated Collagen/Nano-Hydroxyapatite Composites with Improved Mechanical Properties for Bone Repair Materials. J. Biomater. Tissue Eng. 2017, 7, 1000–1007. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Liu, D.; Yang, Q.; Huang, C.; Yang, C.; Zhang, Q. Preparation and Cytological Study of Collagen/Nano-Hydroxyapatite/Graphene Oxide Composites. Acta Bioeng. Biomech. 2018, 20, 65–74. [Google Scholar] [CrossRef]
- Liang, C.; Luo, Y.; Yang, G.; Xia, D.; Liu, L.; Zhang, X.; Wang, H. Graphene Oxide Hybridized NHAC/PLGA Scaffolds Facilitate the Proliferation of MC3T3-E1 Cells. Nanoscale Res. Lett. 2018, 13. [Google Scholar] [CrossRef] [PubMed]
- Yılmaz, E.; Çakıroğlu, B.; Gökçe, A.; Findik, F.; Gulsoy, H.O.; Gulsoy, N.; Mutlu, Ö.; Özacar, M. Novel Hydroxyapatite/Graphene Oxide/Collagen Bioactive Composite Coating on Ti16Nb Alloys by Electrodeposition. Mater. Sci. Eng. C 2019, 101, 292–305. [Google Scholar] [CrossRef] [PubMed]
- Oyefusi, A.; Olanipekun, O.; Neelgund, G.M.; Peterson, D.; Stone, J.M.; Williams, E.; Carson, L.; Regisford, G.; Oki, A. Hydroxyapatite Grafted Carbon Nanotubes and Graphene Nanosheets: Promising Bone Implant Materials. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 132, 410–416. [Google Scholar] [CrossRef]
- Akhavan, O.; Ghaderi, E. Toxicity of Graphene and Graphene Oxide Nanowalls Against Bacteria. ACS Nano 2010, 4, 5731–5736. [Google Scholar] [CrossRef]
- Nair, M.; Nancy, D.; Krishnan, A.G.; Anjusree, G.S.; Vadukumpully, S.; Nair, S.V. Graphene Oxide Nanoflakes Incorporated Gelatin–Hydroxyapatite Scaffolds Enhance Osteogenic Differentiation of Human Mesenchymal Stem Cells. Nanotechnology 2015, 26, 161001. [Google Scholar] [CrossRef]
- Liu, Y.; Dang, Z.; Wang, Y.; Huang, J.; Li, H. Hydroxyapatite/Graphene-Nanosheet Composite Coatings Deposited by Vacuum Cold Spraying for Biomedical Applications: Inherited Nanostructures and Enhanced Properties. Carbon 2014, 67, 250–259. [Google Scholar] [CrossRef]
- Mohammadrezaei, D.; Golzar, H.; Rezai Rad, M.; Omidi, M.; Rashedi, H.; Yazdian, F.; Khojasteh, A.; Tayebi, L. In Vitro Effect of Graphene Structures as an Osteoinductive Factor in Bone Tissue Engineering: A Systematic Review. J. Biomed. Mater. Res. Part A 2018, 106, 2284–2343. [Google Scholar] [CrossRef] [PubMed]
- Balani, K.; Anderson, R.; Laha, T.; Andara, M.; Tercero, J.; Crumpler, E.; Agarwal, A. Plasma-Sprayed Carbon Nanotube Reinforced Hydroxyapatite Coatings and Their Interaction with Human Osteoblasts in Vitro. Biomaterials 2007, 28, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Mathur, P.; Jha, S.; Ramteke, S.; Jain, N.K. Pharmaceutical Aspects of Silver Nanoparticles. Artif. Cells Nanomed. Biotechnol. 2018, 46, 115–126. [Google Scholar] [CrossRef]
- Markowska, K.; Grudniak, A.M.; Wolska, K.I. Silver Nanoparticles as an Alternative Strategy against Bacterial Biofilms. Acta Biochim. Pol. 2013, 60, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, E.; Milani, M.; Fekri Aval, S.; Kouhi, M.; Akbarzadeh, A.; Tayefi Nasrabadi, H.; Nikasa, P.; Joo, S.W.; Hanifehpour, Y.; Nejati-Koshki, K.; et al. Silver Nanoparticles: Synthesis Methods, Bio-Applications and Properties. Crit. Rev. Microbiol. 2014, 1–8. [Google Scholar] [CrossRef]
- Lee, S.; Jun, B.-H. Silver Nanoparticles: Synthesis and Application for Nanomedicine. Int. J. Mol. Sci. 2019, 20, 865. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Zheng, J. Antibacterial Activity of Silver Nanoparticles: Structural Effects. Adv. Healthc. Mater. 2018, 7, 1701503. [Google Scholar] [CrossRef]
- Predoi, D.; Iconaru, S.L.; Albu, M.; Petre, C.C.; Jiga, G. Physicochemical and Antimicrobial Properties of Silver-Doped Hydroxyapatite Collagen Biocomposite. Polym. Eng. Sci. 2017, 57, 537–545. [Google Scholar] [CrossRef]
- Ciobanu, C.S.; Popa, C.L.; Petre, C.C.; Jiga, G.; Trusca, R.; Predoi, D. Characterisations of Collagen-Silver-Hydroxyapatite Nanocomposites. AIP Conf. Proc. 2016, 1736, 020148. [Google Scholar]
- Kohout, C.; Santi, C.; Polito, L. Anisotropic Gold Nanoparticles in Biomedical Applications. Int. J. Mol. Sci. 2018, 19, 3385. [Google Scholar] [CrossRef] [PubMed]
- Boisselier, E.; Astruc, D. Gold Nanoparticles in Nanomedicine: Preparations, Imaging, Diagnostics, Therapies and Toxicity. Chem. Soc. Rev. 2009, 38, 1759. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Pandit, S.; Mokkapati, V.R.S.S.; Garg, A.; Ravikumar, V.; Mijakovic, I. Gold Nanoparticles in Diagnostics and Therapeutics for Human Cancer. Int. J. Mol. Sci. 2018, 19, 1979. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Crawford, B.M.; Vo-Dinh, T. Gold Nanoparticles-Mediated Photothermal Therapy and Immunotherapy. Immunotherapy 2018, 10, 1175–1188. [Google Scholar] [CrossRef]
- Rajchakit, U.; Sarojini, V. Recent Developments in Antimicrobial-Peptide-Conjugated Gold Nanoparticles. Bioconjug. Chem. 2017, 28, 2673–2686. [Google Scholar] [CrossRef]
- Gao, W.; Thamphiwatana, S.; Angsantikul, P.; Zhang, L. Nanoparticle Approaches against Bacterial Infections. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2014, 6, 532–547. [Google Scholar] [CrossRef]
- Liu, P.-F.; Lo, C.-W.; Chen, C.-H.; Hsieh, M.-F.; Huang, C.-M. Use of Nanoparticles as Therapy for Methicillin-Resistant Staphylococcus Aureus Infections. Curr. Drug Metab. 2009, 10, 875–884. [Google Scholar] [CrossRef]
- Kumar, V.B.; Khajuria, D.K.; Karasik, D.; Gedanken, A. Silver and Gold Doped Hydroxyapatite Nanocomposites for Enhanced Bone Regeneration. Biomed. Mater. 2019, 14, 055002. [Google Scholar] [CrossRef]
- Ross, R.D.; Roeder, R.K. Binding Affinity of Surface Functionalized Gold Nanoparticles to Hydroxyapatite. J. Biomed. Mater. Res. Part A 2011, 99A, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Xu, X.; Feng, X.; Ma, L.; Deng, X.; Wu, S.; Liu, X.; Yang, C. Gold Nanoparticles-Loaded Hydroxyapatite Composites Guide Osteogenic Differentiation of Human Mesenchymal Stem Cells through Wnt/β-Catenin Signaling Pathway. Int. J. Nanomed. 2019, 14, 6151–6163. [Google Scholar] [CrossRef] [PubMed]
- Vukomanović, M.; Logar, M.; Škapin, S.D.; Suvorov, D. Hydroxyapatite/Gold/Arginine: Designing the Structure to Create Antibacterial Activity. J. Mater. Chem. B 2014, 2, 1557–1564. [Google Scholar] [CrossRef]
- Mondal, S.; Hoang, G.; Manivasagan, P.; Moorthy, M.S.; Vy Phan, T.T.; Kim, H.H.; Nguyen, T.P.; Oh, J. Rapid Microwave-Assisted Synthesis of Gold Loaded Hydroxyapatite Collagen Nano-Bio Materials for Drug Delivery and Tissue Engineering Application. Ceram. Int. 2019, 45, 2977–2988. [Google Scholar] [CrossRef]
- Aryal, S.; Bahadur, K.C.R.; Bhattarai, S.R.; Prabu, P.; Kim, H.Y. Immobilization of Collagen on Gold Nanoparticles: Preparation, Characterization, and Hydroxyapatite Growth. J. Mater. Chem. 2006, 16, 4642. [Google Scholar] [CrossRef]
- Prasad, K.; Bazaka, O.; Chua, M.; Rochford, M.; Fedrick, L.; Spoor, J.; Symes, R.; Tieppo, M.; Collins, C.; Cao, A.; et al. Metallic Biomaterials: Current Challenges and Opportunities. Materials 2017, 10, 884. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, G.; Harja, M. Cerium-Doped Hydroxyapatite/Collagen Coatings on Titanium for Bone Implants. Ceram. Int. 2019, 45, 2852–2857. [Google Scholar] [CrossRef]
- Ciobanu, G.; Ciobanu, O. Investigation on the Effect of Collagen and Vitamins on Biomimetic Hydroxyapatite Coating Formation on Titanium Surfaces. Mater. Sci. Eng. C 2013, 33, 1683–1688. [Google Scholar] [CrossRef] [PubMed]
- Uezono, M.; Takakuda, K.; Kikuchi, M.; Suzuki, S.; Moriyama, K. Hydroxyapatite/Collagen Nanocomposite-Coated Titanium Rod for Achieving Rapid Osseointegration onto Bone Surface. J. Biomed. Mater. Res. Part B Appl. Biomater. 2013, 101, 1031–1038. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Yu, L.; Wei, M. Sectioning Studies of Biomimetic Collagen-Hydroxyapatite Coatings on Ti-6Al-4V Substrates Using Focused Ion Beam. Appl. Surf. Sci. 2018, 444, 590–597. [Google Scholar] [CrossRef]
- Sun, T.; Lee, W.C.; Wang, M. A Comparative Study of Apatite Coating and Apatite/Collagen Composite Coating Fabricated on NiTi Shape Memory Alloy through Electrochemical Deposition. Mater. Lett. 2011, 65, 2575–2577. [Google Scholar] [CrossRef]
- Hu, R.; Lin, C.; Wang, H.; Tao, T. Modulation Effects of Collagen I{cyrillic, Ukrainian} on the Structure of Electrochemically Deposited Hydroxyapatite Coating. Mater. Lett. 2010, 64, 915–917. [Google Scholar] [CrossRef]
- Manara, S.; Paolucci, F.; Palazzo, B.; Marcaccio, M.; Foresti, E.; Tosi, G.; Sabbatini, S.; Sabatino, P.; Altankov, G.; Roveri, N. Electrochemically-Assisted Deposition of Biomimetic Hydroxyapatite-Collagen Coatings on Titanium Plate. Inorg. Chim. Acta 2008, 361, 1634–1645. [Google Scholar] [CrossRef]
- Li, T.; Li, J.S.; Kou, H.C.; Li, F.P.; Lu, T.L. Preparation of Collagen/Hydroxyapatite Composite Coating on Porous Titanium Substrate and Its Cellular Response. Mater. Sci. Forum 2015, 815, 429–433. [Google Scholar] [CrossRef]
- Ou, K.L.; Chung, R.J.; Tsai, F.Y.; Liang, P.Y.; Huang, S.W.; Chang, S.Y. Effect of Collagen on the Mechanical Properties of Hydroxyapatite Coatings. J. Mech. Behav. Biomed. Mater. 2011, 4, 618–624. [Google Scholar] [CrossRef]
- He, J.; Huang, T.; Gan, L.; Zhou, Z.; Jiang, B.; Wu, Y.; Wu, F.; Gu, Z. Collagen-Infiltrated Porous Hydroxyapatite Coating and Its Osteogenic Properties: In Vitro and in Vivo Study. J. Biomed. Mater. Res. Part A 2012, 100, 1706–1715. [Google Scholar] [CrossRef]
- Lee, S.W.; Hahn, B.D.; Kang, T.Y.; Lee, M.J.; Choi, J.Y.; Kim, M.K.; Kim, S.G. Hydroxyapatite and Collagen Combination-Coated Dental Implants Display Better Bone Formation in the Peri-Implant Area than the Same Combination plus Bone Morphogenetic Protein-2-Coated Implants, Hydroxyapatite Only Coated Implants, and Uncoated Implants. J. Oral Maxillofac. Surg. 2014, 72, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Teng, S.H.; Lee, E.J.; Park, C.S.; Choi, W.Y.; Shin, D.S.; Kim, H.E. Bioactive Nanocomposite Coatings of Collagen/Hydroxyapatite on Titanium Substrates. J. Mater. Sci. Mater. Med. 2008, 19, 2453–2461. [Google Scholar] [CrossRef] [PubMed]
- Tapsir, Z.; Saidin, S. Synthesis and Characterization of Collagen-Hydroxyapatite Immobilized on Polydopamine Grafted Stainless Steel. Surf. Coat. Technol. 2016, 285, 11–16. [Google Scholar] [CrossRef]
- Benmarouane, A.; Citterio, H.; Millet, P.; Buslaps, T.; Lodini, A. Evaluation of Residual Stresses at the Interface with Implant by Synchrotron Radiation. Mater. Sci. Forum 2013, 772, 135–139. [Google Scholar] [CrossRef]
- Salomé, M.; Peyrin, F.; Cloetens, P.; Odet, C.; Laval-Jeantet, A.-M.; Baruchel, J.; Spanne, P. A Synchrotron Radiation Microtomography System for the Analysis of Trabecular Bone Samples. Med. Phys. 1999, 26, 2194–2204. [Google Scholar] [CrossRef]
- Ao, H.; Zong, J.; Nie, Y.; Wan, Y.; Zheng, X. An in Vivo Study on the Effect of Coating Stability on Osteointegration Performance of Collagen/Hyaluronic Acid Multilayer Modified Titanium Implants. Bioact. Mater. 2018, 3, 97–101. [Google Scholar] [CrossRef]
- Ao, H.Y.; Xie, Y.T.; Yang, S.B.; Wu, X.D.; Li, K.; Zheng, X.B.; Tang, T.T. Covalently Immobilised Type I Collagen Facilitates Osteoconduction and Osseointegration of Titanium Coated Implants. J. Orthop. Transl. 2016, 5, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.-L.; Kim, D.-E. Self-Healing Characteristics of Collagen Coatings with Respect to Surface Abrasion OPEN. Nat. Publ. Gr. 2016. [Google Scholar] [CrossRef]
- Haïat, G.; Gao, X.; Fraulob, M. Biomechanical Behaviours of the Bone-Implant Interface: A Review. J. R. Soc. Interface 2019. [Google Scholar] [CrossRef]
- Tapsir, Z.; Jamaludin, F.H.; Pingguan-Murphy, B.; Saidin, S. Immobilisation of Hydroxyapatite-Collagen on Polydopamine Grafted Stainless Steel 316L: Coating Adhesion and in Vitro Cells Evaluation. J. Biomater. Appl. 2018, 32, 987–995. [Google Scholar] [CrossRef] [PubMed]
- Edgar, S.; Hopley, B.; Genovese, L.; Sibilla, S.; Laight, D.; Shute, J. Effects of Collagen-Derived Bioactive Peptides and Natural Antioxidant Compounds on Proliferation and Matrix Protein Synthesis by Cultured Normal Human Dermal Fibroblasts. Sci. Rep. 2018, 8, 10474. [Google Scholar] [CrossRef] [PubMed]
- Farokhi, M.; Mottaghitalab, F.; Samani, S.; Shokrgozar, M.A.; Kundu, S.C.; Reis, R.L.; Fatahi, Y.; Kaplan, D.L. Silk Fibroin/Hydroxyapatite Composites for Bone Tissue Engineering. Biotechnol. Adv. 2018, 36, 68–91. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Yuan, Y.; Liu, C. Biomaterial-Guided Immobilization and Osteoactivity of Bone Morphogenetic Protein-2. Appl. Mater. Today 2020, 19, 100599. [Google Scholar] [CrossRef]
- Hettiaratchi, M.H.; Chou, C.; Servies, N.; Smeekens, J.M.; Cheng, A.; Esancy, C.; Wu, R.; McDevitt, T.C.; Guldberg, R.E.; Krishnan, L. Competitive Protein Binding Influences Heparin-Based Modulation of Spatial Growth Factor Delivery for Bone Regeneration. Tissue Eng. Part A 2017, 23, 683–695. [Google Scholar] [CrossRef]
- Grgurevic, L.; Christensen, G.L.; Schulz, T.J.; Vukicevic, S. Bone Morphogenetic Proteins in Inflammation, Glucose Homeostasis and Adipose Tissue Energy Metabolism. Cytokine Growth Factor Rev. 2016, 27, 105–118. [Google Scholar] [CrossRef]
- MacArthur, M.W.; Thornton, J.M. Deviations from Planarity of the Peptide Bond in Peptides and Proteins. J. Mol. Biol. 1996, 264, 1180–1195. [Google Scholar] [CrossRef]
- Mizokami, A.; Kawakubo-Yasukochi, T.; Hirata, M. Osteocalcin and Its Endocrine Functions. Biochem. Pharmacol. 2017, 132, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Moser, S.C.; van der Eerden, B.C.J. Osteocalcin—A Versatile Bone-Derived Hormone. Front. Endocrinol. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Knepper-Nicolai, B.; Reinstorf, A.; Hofinger, I.; Flade, K.; Wenz, R.; Pompe, W. Influence of Osteocalcin and Collagen I on the Mechanical and Biological Properties of Biocement D. Biomol. Eng. 2002, 19, 227–231. [Google Scholar] [CrossRef]
- Rammelt, S.; Neumann, M.; Hanisch, U.; Reinstorf, A.; Pompe, W.; Zwipp, H.; Biewener, A. Osteocalcin Enhances Bone Remodeling around Hydroxyapatite/Collagen Composites. J. Biomed. Mater. Res. Part A 2005, 73, 284–294. [Google Scholar] [CrossRef] [PubMed]
- Wehrle-Martinez, A.S.; Dittmer, K.E.; Aberdein, D.; Thompson, K.G. Osteocalcin and Osteonectin Expression in Canine Osteosarcoma. Vet. Pathol. 2016, 53, 781–787. [Google Scholar] [CrossRef]
- Ciceri, P.; Elli, F.; Cappelletti, L.; Tosi, D.; Savi, F.; Bulfamante, G.; Cozzolino, M. Osteonectin (SPARC) Expression in Vascular Calcification: In Vitro and Ex Vivo Studies. Calcif. Tissue Int. 2016, 99, 472–480. [Google Scholar] [CrossRef]
- Liao, S.; Ngiam, M.; Chan, C.K.; Ramakrishna, S. Fabrication of Nano-Hydroxyapatite/Collagen/Osteonectin Composites for Bone Graft Applications. Biomed. Mater. 2009, 4, 025019. [Google Scholar] [CrossRef]
- Baranowski, A.; Klein, A.; Ritz, U.; Ackermann, A.; Anthonissen, J.; Kaufmann, K.B.; Brendel, C.; Götz, H.; Rommens, P.M.; Hofmann, A. Surface Functionalization of Orthopedic Titanium Implants with Bone Sialoprotein. PLoS ONE 2016, 11, e0153978. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Song, L.; Li, J.; Wang, Y.; Yang, C.; Kou, X.; Xiao, B.; Zhang, W.; Li, L.; Liu, S.; et al. Bone Sialoprotein-αvβ3 Integrin Axis Promotes Breast Cancer Metastasis to the Bone. Cancer Sci. 2019, 110, 3157–3172. [Google Scholar] [CrossRef]
- Baht, G.; Hunter, G.; Goldberg, H. Bone Sialoprotein–Collagen Interaction Promotes Hydroxyapatite Nucleation. Matrix Biol. 2008, 27, 600–608. [Google Scholar] [CrossRef]
- Fudge, D.S.; Winegard, T.; Ewoldt, R.H.; Beriault, D.; Szewciw, L.; McKinley, G.H. From Ultra-Soft Slime to Hard -Keratins: The Many Lives of Intermediate Filaments. Integr. Comp. Biol. 2009, 49, 32–39. [Google Scholar] [CrossRef]
- Tu, H.; Yu, W.; Duan, L. Structural Studies and Macro-Performances of Hydroxyapatite-Reinforced Keratin Thin Films for Biological Applications. J. Mater. Sci. 2016, 51, 9573–9588. [Google Scholar] [CrossRef]
- Dias, G.J.; Mahoney, P.; Hung, N.A.; Sharma, L.A.; Kalita, P.; Smith, R.A.; Kelly, R.J.; Ali, A. Osteoconduction in Keratin-Hydroxyapatite Composite Bone-Graft Substitutes. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 2034–2044. [Google Scholar] [CrossRef]
- Fan, J.; Yu, M.-Y.; Lei, T.; Wang, Y.-H.; Cao, F.-Y.; Qin, X.; Liu, Y. In Vivo Biocompatibility and Improved Compression Strength of Reinforced Keratin/Hydroxyapatite Scaffold. Tissue Eng. Regen. Med. 2018, 15, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Arslan, Y.E.; Sezgin Arslan, T.; Derkus, B.; Emregul, E.; Emregul, K.C. Fabrication of Human Hair Keratin/Jellyfish Collagen/Eggshell-Derived Hydroxyapatite Osteoinductive Biocomposite Scaffolds for Bone Tissue Engineering: From Waste to Regenerative Medicine Products. Colloids Surfaces B Biointerfaces 2017, 154, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Schneiders, W.; Reinstorf, A.; Pompe, W.; Grass, R.; Biewener, A.; Holch, M.; Zwipp, H.; Rammelt, S. Effect of Modification of Hydroxyapatite/Collagen Composites with Sodium Citrate, Phosphoserine, Phosphoserine/RGD-Peptide and Calcium Carbonate on Bone Remodelling. Bone 2007, 40, 1048–1059. [Google Scholar] [CrossRef] [PubMed]
- Bilem, I.; Chevallier, P.; Plawinski, L.; Sone, E.D.; Durrieu, M.C.; Laroche, G. RGD and BMP-2 Mimetic Peptide Crosstalk Enhances Osteogenic Commitment of Human Bone Marrow Stem Cells. Acta Biomater. 2016, 36, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Rezania, A.; Healy, K.E. The Effect of Peptide Surface Density on Mineralization of a Matrix Deposited by Osteogenic Cells. J. Biomed. Mater. Res. 2000, 52, 595–600. [Google Scholar] [CrossRef]
- Hoesli, C.A.; Garnier, A.; Juneau, P.-M.; Chevallier, P.; Duchesne, C.; Laroche, G. A Fluorophore-Tagged RGD Peptide to Control Endothelial Cell Adhesion to Micropatterned Surfaces. Biomaterials 2014, 35, 879–890. [Google Scholar] [CrossRef]
- Dahlin, C.; Johansson, A.; Hoffman, M.; Molenberg, A. Early Biocompatibility of Poly (Ethylene Glycol) Hydrogel Barrier Materials for Guided Bone Regeneration. An in Vitro Study Using Human Gingival Fibroblasts (HGF-1). Clin. Oral Implant. Res. 2014, 25, 16–20. [Google Scholar] [CrossRef]
- Raggio, C.L.; Boyan, B.D.; Boskey, A.L. In Vivo Hydroxyapatite Formation Induced by Lipids. J. Bone Miner. Res. 2009, 1, 409–415. [Google Scholar] [CrossRef]
- Xu, S.; Yu, J.J. Beneath the Minerals, a Layer of Round Lipid Particles Was Identified to Mediate Collagen Calcification in Compact Bone Formation. Biophys. J. 2006, 91, 4221–4229. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wang, Y.; Chen, X. Mineralization Regulation and Biological Influence of Bioactive Glass-Collagen-Phosphatidylserine Composite Scaffolds. Sci. China Life Sci. 2012, 55, 236–240. [Google Scholar] [CrossRef][Green Version]
- Meertens, L.; Carnec, X.; Lecoin, M.P.; Ramdasi, R.; Guivel-Benhassine, F.; Lew, E.; Lemke, G.; Schwartz, O.; Amara, A. The TIM and TAM Families of Phosphatidylserine Receptors Mediate Dengue Virus Entry. Cell Host Microbe 2012, 12, 544–557. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; Kanwar, S.S. Phosphatidylserine: A Cancer Cell Targeting Biomarker. Semin. Cancer Biol. 2018, 52, 17–25. [Google Scholar] [CrossRef]
- Yang, C.; Fang, C. Microporous Nano-Hydroxyapatite/Collagen/Phosphatidylserine Scaffolds Embedding Collagen Microparticles for Controlled Drug Delivery in Bone Tissue Engineering. Mater. Res. 2015, 18, 1077–1081. [Google Scholar] [CrossRef][Green Version]
- Yang, C.R.; Wang, Y.J.; Chen, X.F. Preparation and Evaluation of Biomimetric Nano-Hydroxyapatite-Based Composite Scaffolds for Bone-Tissue Engineering. Chin. Sci. Bull. 2012, 57, 2787–2792. [Google Scholar] [CrossRef]
- Xu, C.; Su, P.; Chen, X.; Meng, Y.; Yu, W.; Xiang, A.P.; Wang, Y. Biocompatibility and Osteogenesis of Biomimetic Bioglass-Collagen-Phosphatidylserine Composite Scaffolds for Bone Tissue Engineering. Biomaterials 2011, 32, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wu, H.; Chen, S.; Kang, G. Three-Dimensional Bioglass-Collagen-Phosphatidylserine Scaffolds Designed with Functionally Graded Structure and Mechanical Features. Biomed. Eng. Biomed. Tech. 2018, 63, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Mohnen, D. Pectin Structure and Biosynthesis. Curr. Opin. Plant Biol. 2008, 11, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Lara-Espinoza, C.; Carvajal-Millán, E.; Balandrán-Quintana, R.; López-Franco, Y.; Rascón-Chu, A. Pectin and Pectin-Based Composite Materials: Beyond Food Texture. Molecules 2018, 23, 942. [Google Scholar] [CrossRef] [PubMed]
- Wikiera, A.; Irla, M.; Mika, M. Health-Promoting Properties of Pectin. Postepy Hig. Med. Dosw. 2014, 68, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.F.; Barrias, C.C.; Bártolo, P.J.; Granja, P.L. Cell-Instructive Pectin Hydrogels Crosslinked via Thiol-Norbornene Photo-Click Chemistry for Skin Tissue Engineering. Acta Biomater. 2018, 66, 282–293. [Google Scholar] [CrossRef]
- Wenpo, F.; Gaofeng, L.; Shuying, F.; Yuanming, Q.; Keyong, T. Preparation and Characterization of Collagen–Hydroxyapatite/Pectin Composite. Int. J. Biol. Macromol. 2015, 74, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Muxika, A.; Etxabide, A.; Uranga, J.; Guerrero, P.; de la Caba, K. Chitosan as a Bioactive Polymer: Processing, Properties and Applications. Int. J. Biol. Macromol. 2017, 105, 1358–1368. [Google Scholar] [CrossRef]
- Costa, E.M.; Silva, S.; Veiga, M.; Tavaria, F.K.; Pintado, M.M. Chitosan’s Biological Activity upon Skin-Related Microorganisms and Its Potential Textile Applications. World J. Microbiol. Biotechnol. 2018, 34, 93. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef]
- Antony, R.; Arun, T.; Manickam, S.T.D. A Review on Applications of Chitosan-Based Schiff Bases. Int. J. Biol. Macromol. 2019, 129, 615–633. [Google Scholar] [CrossRef]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial Properties of Chitosan and Mode of Action: A State of the Art Review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef]
- Patrulea, V.; Ostafe, V.; Borchard, G.; Jordan, O. Chitosan as a Starting Material for Wound Healing Applications. Eur. J. Pharm. Biopharm. 2015, 97, 417–426. [Google Scholar] [CrossRef]
- Ahmed, F.; Soliman, F.M.; Adly, M.A.; Soliman, H.A.M.; El-Matbouli, M.; Saleh, M. Recent Progress in Biomedical Applications of Chitosan and Its Nanocomposites in Aquaculture: A Review. Res. Vet. Sci. 2019, 126, 68–82. [Google Scholar] [CrossRef]
- Kaczmarek, B.; Sionkowska, A.; Gołyńska, M.; Polkowska, I.; Szponder, T.; Nehrbass, D.; Osyczka, A.M. In Vivo Study on Scaffolds Based on Chitosan, Collagen, and Hyaluronic Acid with Hydroxyapatite. Int. J. Biol. Macromol. 2018, 118, 938–944. [Google Scholar] [CrossRef]
- Sionkowska, A.; Kaczmarek, B. Preparation and Characterization of Composites Based on the Blends of Collagen, Chitosan and Hyaluronic Acid with Nano-Hydroxyapatite. Int. J. Biol. Macromol. 2017, 102, 658–666. [Google Scholar] [CrossRef]
- Roffi, A.; Kon, E.; Perdisa, F.; Fini, M.; Di Martino, A.; Parrilli, A.; Salamanna, F.; Sandri, M.; Sartori, M.; Sprio, S.; et al. A Composite Chitosan-Reinforced Scaffold Fails to Provide Osteochondral Regeneration. Int. J. Mol. Sci. 2019, 20, 2227. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, L.; Hu, M.; Liu, H.; Wen, W.; Xiao, H.; Niu, Y. Synthesis and Characterization of Collagen-Chitosan-Hydroxyapatite Artificial Bone Matrix. J. Biomed. Mater. Res. Part A 2008, 86, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, B.; Sionkowska, A.; Osyczka, A.M. Physicochemical Properties of Scaffolds Based on Mixtures of Chitosan, Collagen and Glycosaminoglycans with Nano-Hydroxyapatite Addition. Int. J. Biol. Macromol. 2018, 118, 1880–1883. [Google Scholar] [CrossRef] [PubMed]
- Munhoz, M.A.S.; Hirata, H.H.; Plepis, A.M.G.; Martins, V.C.A.; Cunha, M.R. Use of Collagen/Chitosan Sponges Mineralized with Hydroxyapatite for the Repair of Cranial Defects in Rats. Injury 2018, 49, 2154–2160. [Google Scholar] [CrossRef]
- Teng, S.-H.; Liang, M.-H.; Wang, P.; Luo, Y. Biomimetic Composite Microspheres of Collagen/Chitosan/Nano-Hydroxyapatite: In-Situ Synthesis and Characterization. Mater. Sci. Eng. C 2016, 58, 610–613. [Google Scholar] [CrossRef]
- Rahman, M.S.; Rana, M.M.; Spitzhorn, L.-S.; Akhtar, N.; Hasan, M.Z.; Choudhury, N.; Fehm, T.; Czernuszka, J.T.; Adjaye, J.; Asaduzzaman, S.M. Fabrication of Biocompatible Porous Scaffolds Based on Hydroxyapatite/Collagen/Chitosan Composite for Restoration of Defected Maxillofacial Mandible Bone. Prog. Biomater. 2019, 8, 137–154. [Google Scholar] [CrossRef]
- Elango, J.; Saravanakumar, K.; Rahman, S.U.; Henrotin, Y.; Regenstein, J.M.; Wu, W.; Bao, B. Chitosan-Collagen 3D Matrix Mimics Trabecular Bone and Regulates RANKL-Mediated Paracrine Cues of Differentiated Osteoblast and Mesenchymal Stem Cells for Bone Marrow Macrophage-Derived Osteoclastogenesis. Biomolecules 2019, 9, 173. [Google Scholar] [CrossRef]
- Pallela, R.; Venkatesan, J.; Janapala, V.R.; Kim, S.-K. Biophysicochemical Evaluation of Chitosan-Hydroxyapatite-Marine Sponge Collagen Composite for Bone Tissue Engineering. J. Biomed. Mater. Res. Part A 2012, 100, 486–495. [Google Scholar] [CrossRef]
- Qi, X.; Huang, Y.; Han, D.; Zhang, J.; Cao, J.; Jin, X.; Huang, J.; Li, X.; Wang, T. Three-Dimensional Poly (ε-Caprolactone)/Hydroxyapatite/Collagen Scaffolds Incorporating Bone Marrow Mesenchymal Stem Cells for the Repair of Bone Defects. Biomed. Mater. 2016, 11, 025005. [Google Scholar] [CrossRef]
- Wu, H.; Song, Y.; Li, J.; Lei, X.; Zhang, S.; Gao, Y.; Cheng, P.; Liu, B.; Miao, S.; Bi, L.; et al. Blockade of Adrenergic Β-receptor Activation through Local Delivery of Propranolol from a 3D Collagen/Polyvinyl Alcohol/Hydroxyapatite Scaffold Promotes Bone Repair in Vivo. Cell Prolif. 2020, 53. [Google Scholar] [CrossRef]
- Coutu, D.L.; Cuerquis, J.; El Ayoubi, R.; Forner, K.-A.; Roy, R.; François, M.; Griffith, M.; Lillicrap, D.; Yousefi, A.-M.; Blostein, M.D.; et al. Hierarchical Scaffold Design for Mesenchymal Stem Cell-Based Gene Therapy of Hemophilia B. Biomaterials 2011, 32, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Prosecká, E.; Rampichová, M.; Litvinec, A.; Tonar, Z.; Králíčková, M.; Vojtová, L.; Kochová, P.; Plencner, M.; Buzgo, M.; Míčková, A.; et al. Collagen/Hydroxyapatite Scaffold Enriched with Polycaprolactone Nanofibers, Thrombocyte-Rich Solution and Mesenchymal Stem Cells Promotes Regeneration in Large Bone Defect in Vivo. J. Biomed. Mater. Res. Part A 2015, 103, 671–682. [Google Scholar] [CrossRef] [PubMed]
- Phipps, M.C.; Clem, W.C.; Catledge, S.A.; Xu, Y.; Hennessy, K.M.; Thomas, V.; Jablonsky, M.J.; Chowdhury, S.; Stanishevsky, A.V.; Vohra, Y.K.; et al. Mesenchymal Stem Cell Responses to Bone-Mimetic Electrospun Matrices Composed of Polycaprolactone, Collagen I and Nanoparticulate Hydroxyapatite. PLoS ONE 2011, 6, e16813. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, D.B.; Middleton, J.C.; Tannenbaum, R.; Wick, T.M. Mechanical Properties and Osteogenic Potential of Hydroxyapatite-PLGA-Collagen Biomaterial for Bone Regeneration. J. Biomater. Sci. Polym. Ed. 2016, 27, 1139–1154. [Google Scholar] [CrossRef]
- Li, J.; Yuan, X.; He, F.; Mak, A.F.T. Hybrid Coating of Hydroxyapatite and Collagen within Poly(D,L-lactic- Co -glycolic Acid) Scaffold. J. Biomed. Mater. Res. Part B Appl. Biomater. 2008, 86, 381–388. [Google Scholar] [CrossRef]
- Degirmenbasi, N.; Kalyon, D.M.; Birinci, E. Biocomposites of Nanohydroxyapatite with Collagen and Poly(Vinyl Alcohol). Colloids Surfaces B Biointerfaces 2006, 48, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Ficai, M.; Andronescu, E.; Ficai, D.; Voicu, G.; Ficai, A. Synthesis and Characterization of COLL–PVA/HA Hybrid Materials with Stratified Morphology. Colloids Surfaces B Biointerfaces 2010, 81, 614–619. [Google Scholar] [CrossRef]
- Kumar, V.; Suman, U.; Rubal; Yadav, S.K. Flavonoid secondary metabolite: Biosynthesis and role in growth and development in plants. In Recent Trends and Techniques in Plant Metabolic Engineering; Springer: Singapore, 2018; pp. 19–45. ISBN 9789811322518. [Google Scholar]
- Sakaki, J.; Melough, M.; Lee, S.G.; Pounis, G.; Chun, O.K. Polyphenol-Rich Diets in Cardiovascular Disease Prevention. In Analysis in Nutrition Research; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 259–298. ISBN 9780128145562. [Google Scholar]
- Rauter, A.P.; Ennis, M.; Hellwich, K.H.; Herold, B.J.; Horton, D.; Moss, G.P.; Schomburg, I. Nomenclature of Flavonoids (IUPAC Recommendations 2017). Pure Appl. Chem. 2018, 90, 1429–1486. [Google Scholar] [CrossRef]
- Cheng, A.X.; Han, X.J.; Wu, Y.F.; Lou, H.X. The Function and Catalysis of 2-Oxoglutarate-Dependent Oxygenases Involved in Plant Flavonoid Biosynthesis. Int. J. Mol. Sci. 2014, 15, 1080–1095. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An Overview. J. Nutr. Sci. 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Bondonno, N.P.; Dalgaard, F.; Kyrø, C.; Murray, K.; Bondonno, C.P.; Lewis, J.R.; Croft, K.D.; Gislason, G.; Scalbert, A.; Cassidy, A.; et al. Flavonoid Intake Is Associated with Lower Mortality in the Danish Diet Cancer and Health Cohort. Nat. Commun. 2019, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C. Molecular and Cellular Basis of the Anticancer Activity of the Prenylated Flavonoid Icaritin in Hepatocellular Carcinoma. Chem. Biol. Interact. 2020, 325, 109124. [Google Scholar] [CrossRef] [PubMed]
- Kopustinskiene, D.M.; Jakstas, V.; Savickas, A.; Bernatoniene, J. Flavonoids as Anticancer Agents. Nutrients 2020, 12, 457. [Google Scholar] [CrossRef]
- George, V.C.; Dellaire, G.; Rupasinghe, H.P.V. Plant Flavonoids in Cancer Chemoprevention: Role in Genome Stability. J. Nutr. Biochem. 2017, 45, 1–14. [Google Scholar] [CrossRef]
- Bondonno, C.P.; Croft, K.D.; Ward, N.; Considine, M.J.; Hodgson, J.M. Dietary Flavonoids and Nitrate: Effects on Nitric Oxide and Vascular Function. Nutr. Rev. 2015, 73, 216–235. [Google Scholar] [CrossRef]
- Fusi, F.; Trezza, A.; Tramaglino, M.; Sgaragli, G.; Saponara, S.; Spiga, O. The Beneficial Health Effects of Flavonoids on the Cardiovascular System: Focus on K+ Channels. Pharmacol. Res. 2020, 152, 104625. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Hernández Bautista, R.J.; Sandhu, M.A.; Hussein, O.E. Beneficial Effects of Citrus Flavonoids on Cardiovascular and Metabolic Health. Oxid. Med. Cell. Longev. 2019, 2019. [Google Scholar] [CrossRef]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-Inflammatory Effects of Flavonoids. Food Chem. 2019, 299. [Google Scholar] [CrossRef]
- Choy, K.W.; Murugan, D.; Leong, X.F.; Abas, R.; Alias, A.; Mustafa, M.R. Flavonoids as Natural Anti-Inflammatory Agents Targeting Nuclear Factor-Kappa B (NFκB) Signaling in Cardiovascular Diseases: A Mini Review. Front. Pharmacol. 2019, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhu, L.; Fukuda, K.; Ouyang, S.; Chen, X.; Wang, C.; Zhang, C.J.; Martin, B.; Gu, C.; Qin, L.; et al. The Flavonoid Cyanidin Blocks Binding of the Cytokine Interleukin-17A to the IL-17RA Subunit to Alleviate Inflammation in Vivo. Sci. Signal. 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhao, S.; Lu, J.; Kang, X.; Zhang, G.; Zhao, F.; Nie, J.; Yang, X.; Xin, X.; Zhang, H.; et al. Quantitative Proteomics Analysis of the Treatment of Asthma Rats with Total Flavonoid Extract from Chamomile. Biotechnol. Lett. 2020, 42, 905–916. [Google Scholar] [CrossRef]
- Smith, L.J.; Kalhan, R.; Wise, R.A.; Sugar, E.A.; Lima, J.J.; Irvin, C.G.; Dozor, A.J.; Holbrook, J.T.; Hanania, N.; Sockrider, M.; et al. Effect of a Soy Isoflavone Supplement on Lung Function and Clinical Outcomes in Patients with Poorly Controlled Asthma: A Randomized Clinical Trial. JAMA J. Am. Med. Assoc. 2015, 313, 2033–2043. [Google Scholar] [CrossRef] [PubMed]
- Testa, R.; Bonfigli, A.R.; Genovese, S.; De Nigris, V.; Ceriello, A. The Possible Role of Flavonoids in the Prevention of Diabetic Complications. Nutrients 2016, 8, 310. [Google Scholar] [CrossRef]
- Sarian, M.N.; Ahmed, Q.U.; Mat So’Ad, S.Z.; Alhassan, A.M.; Murugesu, S.; Perumal, V.; Syed Mohamad, S.N.A.; Khatib, A.; Latip, J. Antioxidant and Antidiabetic Effects of Flavonoids: A Structure-Activity Relationship Based Study. Biomed Res. Int. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, Y.A.; Garud, M.S.; Oza, M.J.; Barve, K.H.; Gaikwad, A.B. Diabetes, diabetic complications, and flavonoids. In Fruits, Vegetables, and Herbs: Bioactive Foods in Health Promotion; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 77–104. ISBN 9780128029893. [Google Scholar]
- Maher, P. The Potential of Flavonoids for the Treatment of Neurodegenerative Diseases. Int. J. Mol. Sci. 2019, 20, 3056. [Google Scholar] [CrossRef] [PubMed]
- De Andrade Teles, R.B.; Diniz, T.C.; Costa Pinto, T.C.; de Oliveira Júnior, R.G.; Gama e Silva, M.; de Lavor, É.M.; Fernandes, A.W.C.; de Oliveira, A.P.; de Almeida Ribeiro, F.P.R.; da Silva, A.A.M.; et al. Flavonoids as Therapeutic Agents in Alzheimer’s and Parkinson’s Diseases: A Systematic Review of Preclinical Evidences. Oxid. Med. Cell. Longev. 2018, 2018, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Jones, Q.R.D.; Warford, J.; Rupasinghe, H.P.V.; Robertson, G.S. Target-Based Selection of Flavonoids for Neurodegenerative Disorders. Trends Pharmacol. Sci. 2012, 33, 602–610. [Google Scholar] [CrossRef]
- Costa, S.L.; Silva, V.D.A.; dos Santos Souza, C.; Santos, C.C.; Paris, I.; Muñoz, P.; Segura-Aguilar, J. Impact of Plant-Derived Flavonoids on Neurodegenerative Diseases. Neurotox. Res. 2016, 30, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.J.; Jeon, S.B.; Oh, H.; Lee, B.H.; Lee, S.Y.; Oh, S.H.; Jung, J.Y.; Choi, C. Comparison of the Antiviral Activity of Flavonoids against Murine Norovirus and Feline Calicivirus. Food Control 2016, 60, 25–30. [Google Scholar] [CrossRef]
- Zakaryan, H.; Arabyan, E.; Oo, A.; Zandi, K. Flavonoids: Promising Natural Compounds against Viral Infections. Arch. Virol. 2017, 162, 2539–2551. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Kaleem, M.; Ahmed, Z.; Shafiq, H. Therapeutic Potential of Flavonoids and Their Mechanism of Action against Microbial and Viral Infections-A Review. Food Res. Int. 2015, 77, 221–235. [Google Scholar] [CrossRef]
- Dai, W.; Bi, J.; Li, F.; Wang, S.; Huang, X.; Meng, X.; Sun, B.; Wang, D.; Kong, W.; Jiang, C.; et al. Antiviral Efficacy of Flavonoids against Enterovirus 71 Infection in Vitro and in Newborn Mice. Viruses 2019, 11, 625. [Google Scholar] [CrossRef]
- Al Mamun, M.A.; Hosen, M.J.; Islam, K.; Khatun, A.; Alam, M.M.; Al-Bari, M.A.A. Tridax Procumbens Flavonoids Promote Osteoblast Differentiation and Bone Formation. Biol. Res. 2015, 48, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Xiong, Y.; Zhang, Y.; Jia, L.; Zhang, W.; Xu, X. Rutin Promotes Osteogenic Differentiation of Periodontal Ligament Stem Cells through the GPR30-Mediated PI3K/AKT/MTOR Signaling Pathway. Exp. Biol. Med. 2020, 245, 552–561. [Google Scholar] [CrossRef]
- Kim, I.R.; Kim, S.E.; Baek, H.S.; Kim, B.J.; Kim, C.H.; Chung, I.K.; Park, B.S.; Shin, S.H. The Role of Kaempferol-Induced Autophagy on Differentiation and Mineralization of Osteoblastic MC3T3-E1 Cells. BMC Complement. Altern. Med. 2016, 16, 1–10. [Google Scholar] [CrossRef]
- Liu, L.; Wang, D.; Qin, Y.; Xu, M.; Zhou, L.; Xu, W.; Liu, X.; Ye, L.; Yue, S.; Zheng, Q.; et al. Astragalin Promotes Osteoblastic Differentiation in MC3T3-E1 Cells and Bone Formation in Vivo. Front. Endocrinol. 2019, 10, 228. [Google Scholar] [CrossRef]
- Kim, Y.J.; Bae, Y.C.; Suh, K.T.; Jung, J.S. Quercetin, a Flavonoid, Inhibits Proliferation and Increases Osteogenic Differentiation in Human Adipose Stromal Cells. Biochem. Pharmacol. 2006, 72, 1268–1278. [Google Scholar] [CrossRef]
- Srivastava, S.; Bankar, R.; Roy, P. Assessment of the Role of Flavonoids for Inducing Osteoblast Differentiation in Isolated Mouse Bone Marrow Derived Mesenchymal Stem Cells. Phytomedicine 2013, 20, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.G.; Son, K.M.; Park, H.C.; Zhu, T.; Kwon, J.H.; Yang, H.C. Stimulating Effects of Quercetin and Phenamil on Differentiation of Human Dental Pulp Cells. Eur. J. Oral Sci. 2013, 121, 559–565. [Google Scholar] [CrossRef]
- Saulite, L.; Jekabsons, K.; Klavins, M.; Muceniece, R.; Riekstina, U. Effects of Malvidin, Cyanidin and Delphinidin on Human Adipose Mesenchymal Stem Cell Differentiation into Adipocytes, Chondrocytes and Osteocytes. Phytomedicine 2019, 53, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhang, J.; Fong, C.; Yao, X.; Yang, M. Herba Epimedii Flavonoids Suppress Osteoclastic Differentiation and Bone Resorption by Inducing G2/M Arrest and Apoptosis. Biochimie 2012, 94, 2514–2522. [Google Scholar] [CrossRef]
- Harada, S.; Tominari, T.; Matsumoto, C.; Hirata, M.; Takita, M.; Inada, M.; Miyaura, C. Nobiletin, a Polymethoxy Flavonoid, Suppresses Bone Resorption by Inhibiting NFκB-Dependent Prostaglandin E Synthesis in Osteoblasts and Prevents Bone Loss Due to Estrogen Deficiency. J. Pharmacol. Sci. 2011, 115, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Tominari, T.; Hirata, M.; Matsumoto, C.; Inada, M.; Miyaura, C. Polymethoxy Flavonoids, Nobiletin and Tangeretin, Prevent Lipopolysaccharide-Induced Inflammatory Bone Loss in an Experimental Model for Periodontitis. J. Pharmacol. Sci. 2012, 119, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Wattel, A.; Kamel, S.; Mentaverri, R.; Lorget, F.; Prouillet, C.; Petit, J.P.; Fardelonne, P.; Brazier, M. Potent Inhibitory Effect of Naturally Occurring Flavonoids Quercetin and Kaempferol on in Vitro Osteoclastic Bone Resorption. Biochem. Pharmacol. 2003, 65, 35–42. [Google Scholar] [CrossRef]
- Chiba, H.; Uehara, M.; Wu, J.; Wang, X.; Masuyama, R.; Suzuki, K.; Kanazawa, K.; Ishimi, Y. Hesperidin, a Citrus Flavonoid, Inhibits Bone Loss and Decreases Serum and Hepatic Lipids in Ovariectomized Mice. J. Nutr. 2003, 133, 1892–1897. [Google Scholar] [CrossRef] [PubMed]
- Lei, Z.; Ouyang, L.; Gong, Y.; Wang, Z.; Yu, B. Effect of Eriodictyol on Collagen-Induced Arthritis in Rats by Akt/Hif-1α Pathway. Drug Des. Dev. Ther. 2020, 14, 1633–1639. [Google Scholar] [CrossRef]
- Li, R.; Li, J.; Cai, L.; Hu, C.; Zhang, L. Suppression of Adjuvant Arthritis by Hesperidin in Rats and Its Mechanisms. J. Pharm. Pharmacol. 2008, 60, 221–228. [Google Scholar] [CrossRef]
- Manchope, M.F.; Artero, N.A.; Fattori, V.; Mizokami, S.S.; Pitol, D.L.; Issa, J.P.M.; Fukada, S.Y.; Cunha, T.M.; Alves-Filho, J.C.; Cunha, F.Q.; et al. Naringenin Mitigates Titanium Dioxide (TiO2)-Induced Chronic Arthritis in Mice: Role of Oxidative Stress, Cytokines, and NFκB. Inflamm. Res. 2018, 67, 997–1012. [Google Scholar] [CrossRef]
- Ahmad, S.F.; Zoheir, K.M.A.; Abdel-Hamied, H.E.; Ashour, A.E.; Bakheet, S.A.; Attia, S.M.; Abd-Allah, A.R.A. Amelioration of Autoimmune Arthritis by Naringin through Modulation of T Regulatory Cells and Th1/Th2 Cytokines. Cell. Immunol. 2014, 287, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Kuo, W.S.; Weng, C.T.; Chen, J.H.; Wu, C.L.; Shiau, A.L.; Hsieh, J.L.; So, E.C.; Wu, P.T.; Chen, S.Y. Amelioration of Experimentally Induced Arthritis by Reducing Reactive Oxygen Species Production through the Intra-Articular Injection of Water-Soluble Fullerenol. Nanomaterials 2019, 9, 909. [Google Scholar] [CrossRef]
- Impellizzeri, D.; Esposito, E.; Di Paola, R.; Ahmad, A.; Campolo, M.; Peli, A.; Morittu, V.M.; Britti, D.; Cuzzocrea, S. Erratum to Palmitoylethanolamide and Luteolin Ameliorate Development of Arthritis Caused by Injection of Collagen Type II in Mice [Arthritis Res Ther. 2013;15: R192] doi:10.1186/Ar4382. Arthritis Res. Ther. 2016, 18. [Google Scholar] [CrossRef]
- Gul, A.; Kunwar, B.; Mazhar, M.; Faizi, S.; Ahmed, D.; Shah, M.R.; Simjee, S.U. Rutin and Rutin-Conjugated Gold Nanoparticles Ameliorate Collagen-Induced Arthritis in Rats through Inhibition of NF-ΚB and INOS Activation. Int. Immunopharmacol. 2018, 59, 310–317. [Google Scholar] [CrossRef]
- Forte, L.; Torricelli, P.; Boanini, E.; Gazzano, M.; Rubini, K.; Fini, M.; Bigi, A. Antioxidant and Bone Repair Properties of Quercetin-Functionalized Hydroxyapatite: An in Vitro Osteoblast-Osteoclast-Endothelial Cell Co-Culture Study. Acta Biomater. 2016, 32, 298–308. [Google Scholar] [CrossRef]
- Sistanipour, E.; Meshkini, A.; Oveisi, H. Catechin-Conjugated Mesoporous Hydroxyapatite Nanoparticle: A Novel Nano-Antioxidant with Enhanced Osteogenic Property. Colloids Surfaces B Biointerfaces 2018, 169, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Pang, S.; Li, X.; Wu, D.; Li, H.; Wang, X. Tuning Inflammation Response via Adjusting Microstructure of Hydroxyapatite and Biomolecules Modification. Colloids Surfaces B Biointerfaces 2019, 177, 496–505. [Google Scholar] [CrossRef]
- Shahzad, M.; Millhouse, E.; Culshaw, S.; Edwards, C.A.; Ramage, G.; Combet, E. Selected Dietary (Poly)Phenols Inhibit Periodontal Pathogen Growth and Biofilm Formation. Food Funct. 2015, 6, 719–729. [Google Scholar] [CrossRef]
- Forte, L.; Torricelli, P.; Boanini, E.; Rubini, K.; Fini, M.; Bigi, A. Quercetin and Alendronate Multi-Functionalized Materials as Tools to Hinder Oxidative Stress Damage. J. Biomed. Mater. Res. Part A 2017, 105, 3293–3303. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.W.K.; Rabie, A.B.M. Effect of Naringin Collagen Graft on Bone Formation. Biomaterials 2006, 27, 1824–1831. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Guirado, J.L.; López-López, P.J.; Domínguez, M.F.; Gosálvez, M.M.; Prados-Frutos, J.C.; Gehrke, S.A. Histologic Evaluation of New Bone in Post-Extraction Sockets Induced by Melatonin and Apigenin: An Experimental Study in American Fox Hound Dogs. Clin. Oral Implant. Res. 2018, 29. [Google Scholar] [CrossRef]
- Wang, W.; Sun, L.; Zhang, P.; Song, J.; Liu, W. An Anti-Inflammatory Cell-Free Collagen/Resveratrol Scaffold for Repairing Osteochondral Defects in Rabbits. Acta Biomater. 2014, 10, 4983–4995. [Google Scholar] [CrossRef]
- Li, D.; Yuan, T.; Zhang, X.; Xiao, Y.; Wang, R.; Fan, Y.; Zhang, X. Icariin: A Potential Promoting Compound for Cartilage Tissue Engineering. Osteoarthr. Cartil. 2012, 20, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Samadian, H.; Vaez, A.; Ehterami, A.; Salehi, M.; Farzamfar, S.; Sahrapeyma, H.; Norouzi, P. Sciatic Nerve Regeneration by Using Collagen Type I Hydrogel Containing Naringin. J. Mater. Sci. Mater. Med. 2019, 30. [Google Scholar] [CrossRef]
- Gomathi, K.; Gopinath, D.; Ahmed, M.R.; Jayakumar, R. Quercetin Incorporated Collagen Matrices for Dermal Wound Healing Processes in Rat. Biomaterials 2003, 24, 2767–2772. [Google Scholar] [CrossRef]
- Kook, Y.J.; Tian, J.; Jeon, Y.S.; Choi, M.J.; Song, J.E.; Park, C.H.; Reis, R.L.; Khang, G. Nature-Derived Epigallocatechin Gallate/Duck’s Feet Collagen/Hydroxyapatite Composite Sponges for Enhanced Bone Tissue Regeneration. J. Biomater. Sci. Polym. Ed. 2018, 29, 984–996. [Google Scholar] [CrossRef]
- Pati, F.; Song, T.H.; Rijal, G.; Jang, J.; Kim, S.W.; Cho, D.W. Ornamenting 3D Printed Scaffolds with Cell-Laid Extracellular Matrix for Bone Tissue Regeneration. Biomaterials 2015, 37, 230–241. [Google Scholar] [CrossRef]
- Song, J.E.; Tian, J.; Kook, Y.J.; Thangavelu, M.; Choi, J.H.; Khang, G. A BMSCs-Laden Quercetin/Duck’s Feet Collagen/Hydroxyapatite Sponge for Enhanced Bone Regeneration. J. Biomed. Mater. Res. Part A 2020, 108, 784–794. [Google Scholar] [CrossRef]
- Song, J.E.; Jeon, Y.S.; Tian, J.; Kim, W.K.; Choi, M.J.; Carlomagno, C.; Khang, G. Evaluation of Silymarin/Duck’s Feet-Derived Collagen/Hydroxyapatite Sponges for Bone Tissue Regeneration. Mater. Sci. Eng. C 2019, 97, 347–355. [Google Scholar] [CrossRef]
- Zhao, H.; Tang, J.; Zhou, D.; Weng, Y.; Qin, W.; Liu, C.; Lv, S.; Wang, W.; Zhao, X. Electrospun Icariin-Loaded Core-Shell Collagen, Polycaprolactone, Hydroxyapatite Composite Scaffolds for the Repair of Rabbit Tibia Bone Defects. Int. J. Nanomed. 2020, 15, 3039–3056. [Google Scholar] [CrossRef]
- Phillips, A.M. Overview of the Fracture Healing Cascade. Injury 2005, 36, S5–S7. [Google Scholar] [CrossRef] [PubMed]
- AI-Aql, Z.S.; Alagl, A.S.; Graves, D.T.; Gerstenfeld, L.C.; Einhorn, T.A. Molecular Mechanisms Controlling Bone Formation during Fracture Healing and Distraction Osteogenesis. J. Dent. Res. 2008, 87, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Fazzalari, N.L. Bone Fracture and Bone Fracture Repair. Osteoporos. Int. 2011, 22, 2003–2006. [Google Scholar] [CrossRef] [PubMed]
- Lauzon, M.-A.; Bergeron, É.; Marcos, B.; Faucheux, N. Bone Repair: New Developments in Growth Factor Delivery Systems and Their Mathematical Modeling. J. Control. Release 2012, 162, 502–520. [Google Scholar] [CrossRef] [PubMed]
- Kanematsu, A.; Yamamoto, S.; Ozeki, M.; Noguchi, T.; Kanatani, I.; Ogawa, O.; Tabata, Y. Collagenous Matrices as Release Carriers of Exogenous Growth Factors. Biomaterials 2004, 25, 4513–4520. [Google Scholar] [CrossRef] [PubMed]
- Morin, R.; Kaplan, D.; Perez-Ramirez, B. Bone Morphogenetic Protein-2 Binds As Multilayers To A Collagen Delivery Matrix: An Equilibrium Thermodynamic Analysis. Biomacromolecules 2006, 7, 131–138. [Google Scholar] [CrossRef]
- Geiger, M. Collagen Sponges for Bone Regeneration with RhBMP-2. Adv. Drug Deliv. Rev. 2003, 55, 1613–1629. [Google Scholar] [CrossRef] [PubMed]
- Poynton, A.R.; Lane, J.M. Safety Profile for the Clinical Use of Bone Morphogenetic Proteins in the Spine. Spine 2002, 27, S40–S48. [Google Scholar] [CrossRef] [PubMed]
- Rihn, J.A.; Patel, R.; Makda, J.; Hong, J.; Anderson, D.G.; Vaccaro, A.R.; Hilibrand, A.S.; Albert, T.J. Complications Associated with Single-Level Transforaminal Lumbar Interbody Fusion. Spine J. 2009, 9, 623–629. [Google Scholar] [CrossRef]
- Shahlaie, K.; Kim, K.D. Occipitocervical Fusion Using Recombinant Human Bone Morphogenetic Protein-2. Spine 2008, 33, 2361–2366. [Google Scholar] [CrossRef] [PubMed]
- Carragee, E.J.; Mitsunaga, K.A.; Hurwitz, E.L.; Scuderi, G.J. Retrograde Ejaculation after Anterior Lumbar Interbody Fusion Using RhBMP-2: A Cohort Controlled Study. Spine J. 2011, 11, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.F.; Jones, C.B.; Sietsema, D.L. Complications of RhBMP-2 Utilization for Posterolateral Lumbar Fusions Requiring Reoperation: A Single Practice, Retrospective Case Series Report. Spine J. 2013, 13, 1244–1252. [Google Scholar] [CrossRef]
- Owens, K.; Glassman, S.D.; Howard, J.M.; Djurasovic, M.; Witten, J.L.; Carreon, L.Y. Perioperative Complications with RhBMP-2 in Transforaminal Lumbar Interbody Fusion. Eur. Spine J. 2011, 20, 612–617. [Google Scholar] [CrossRef] [PubMed]
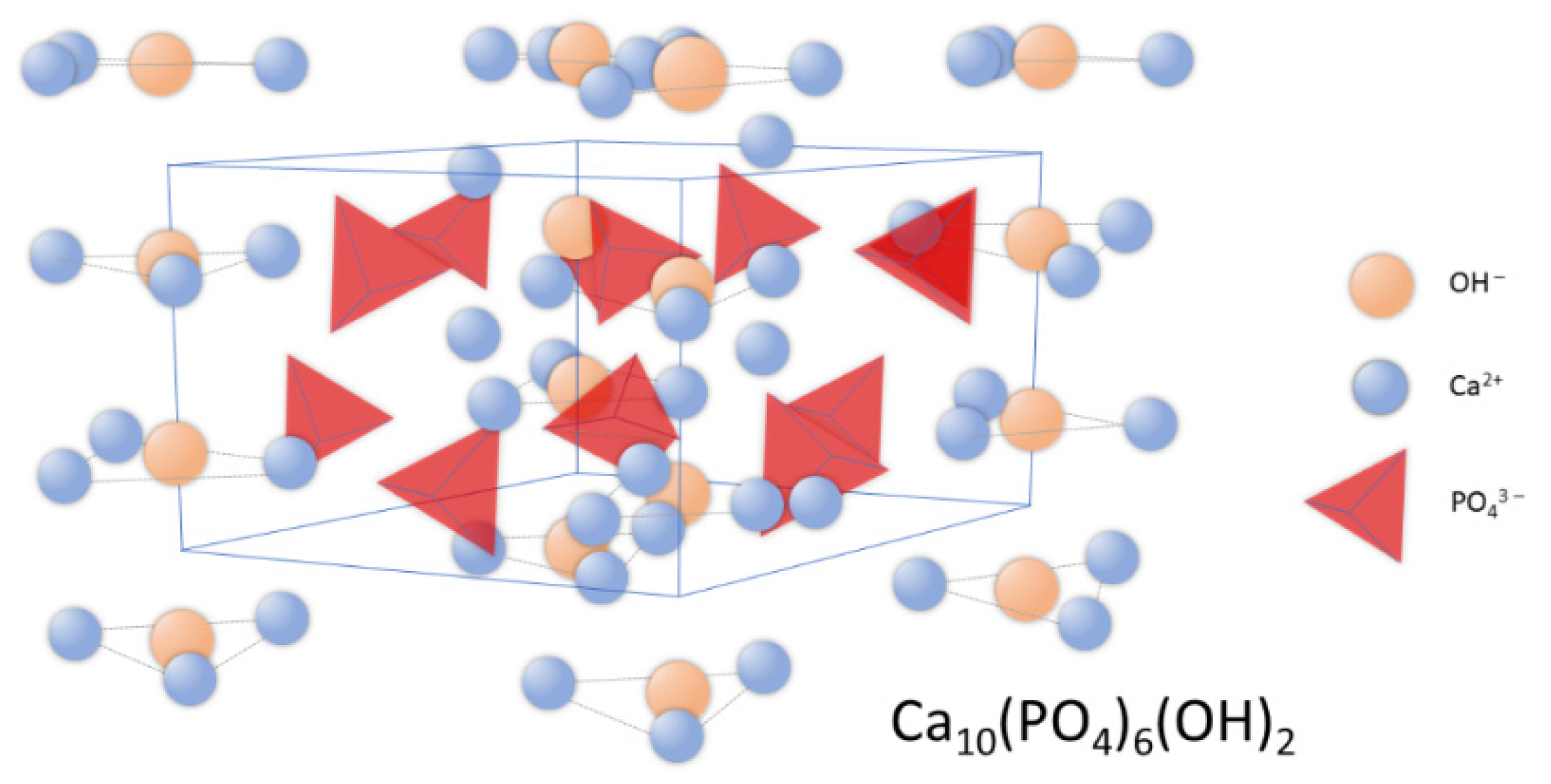
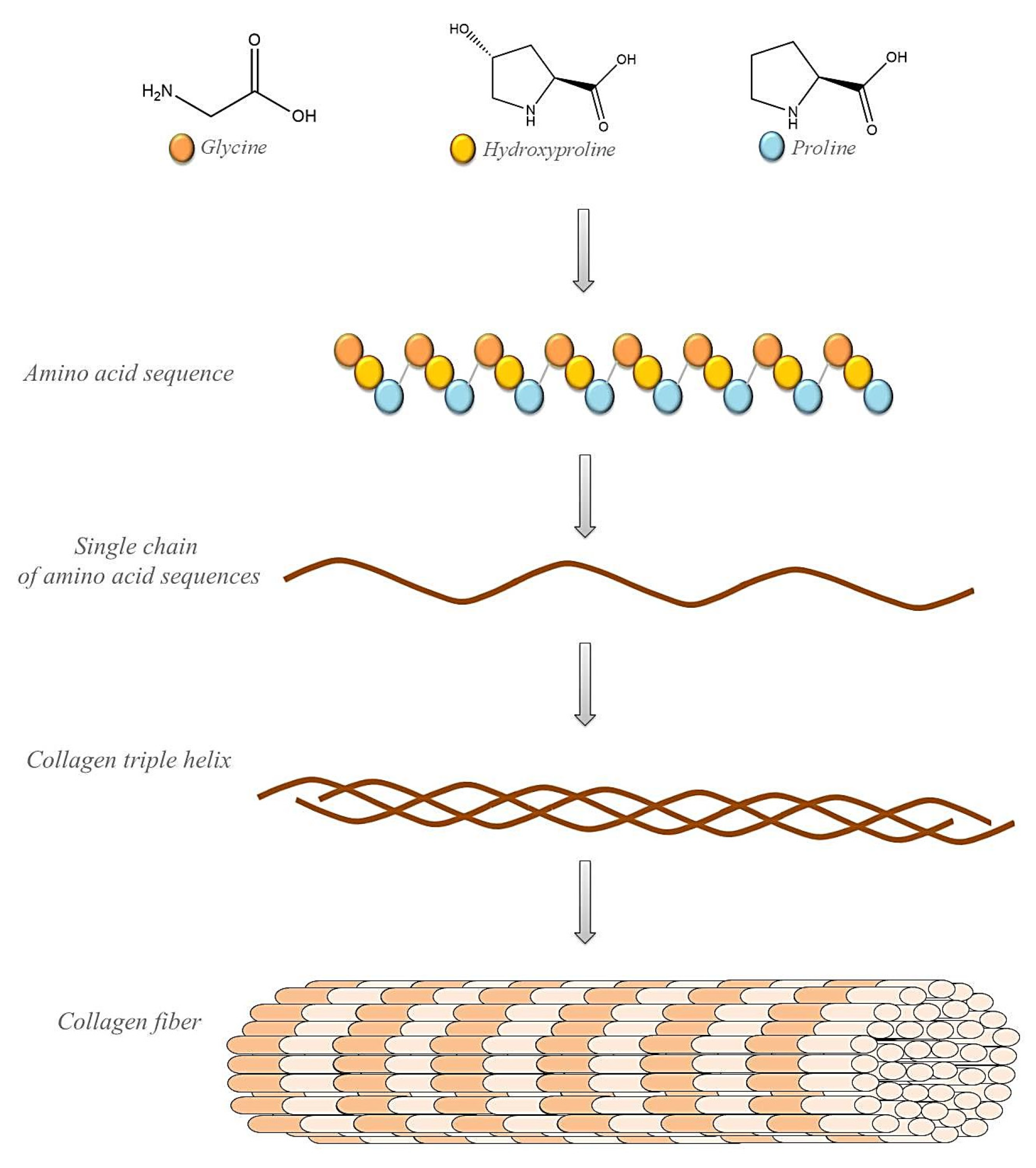

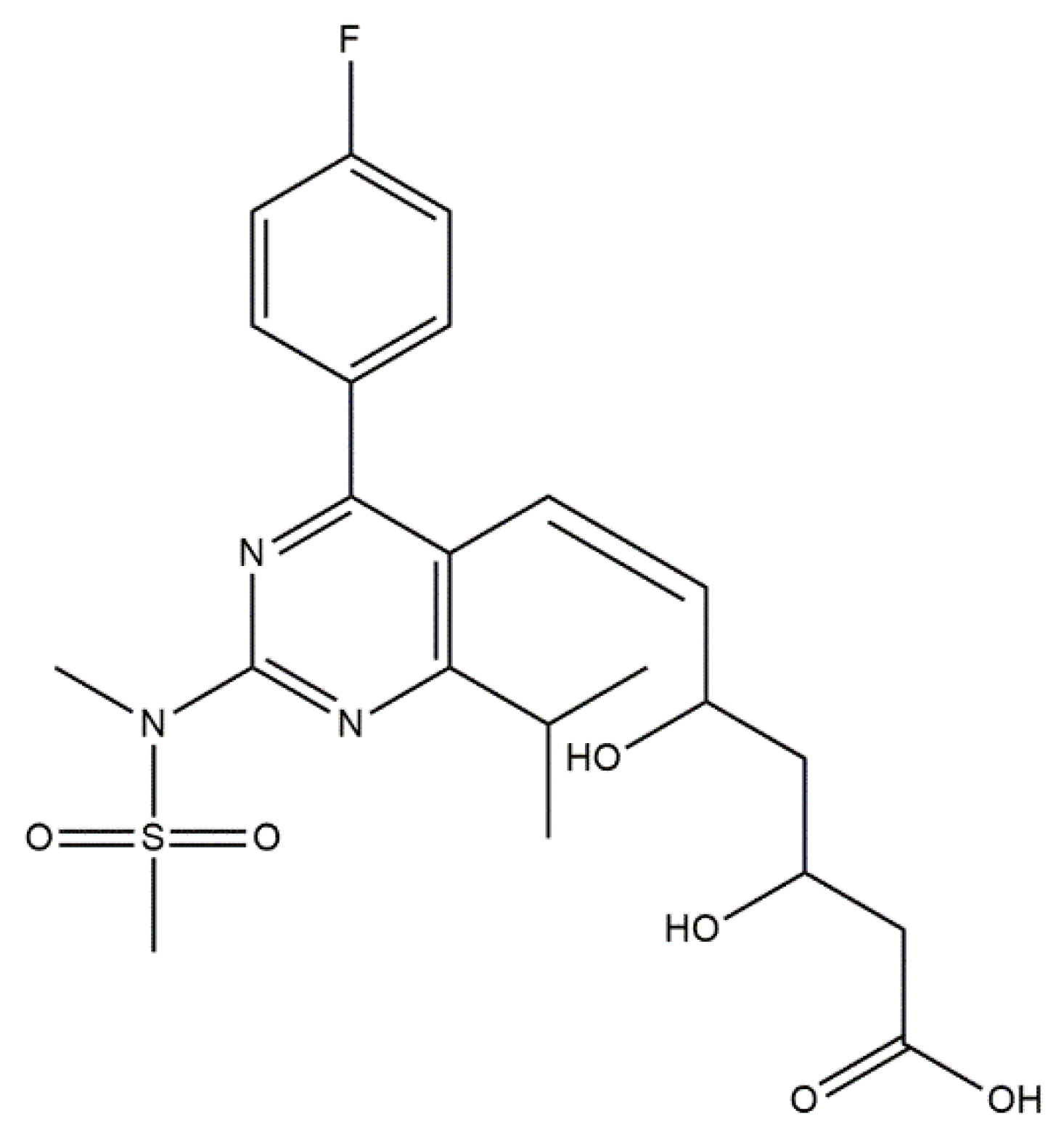
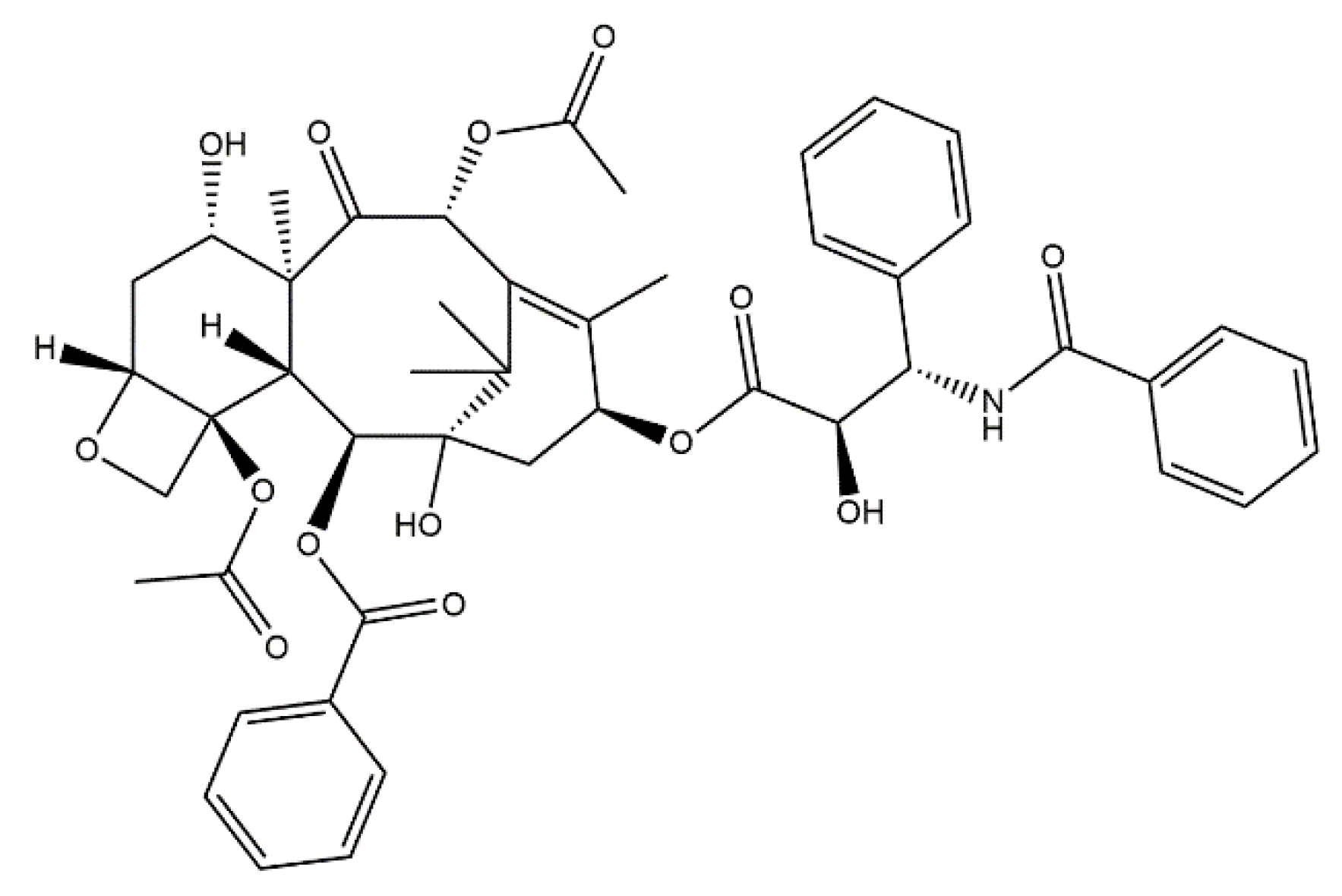
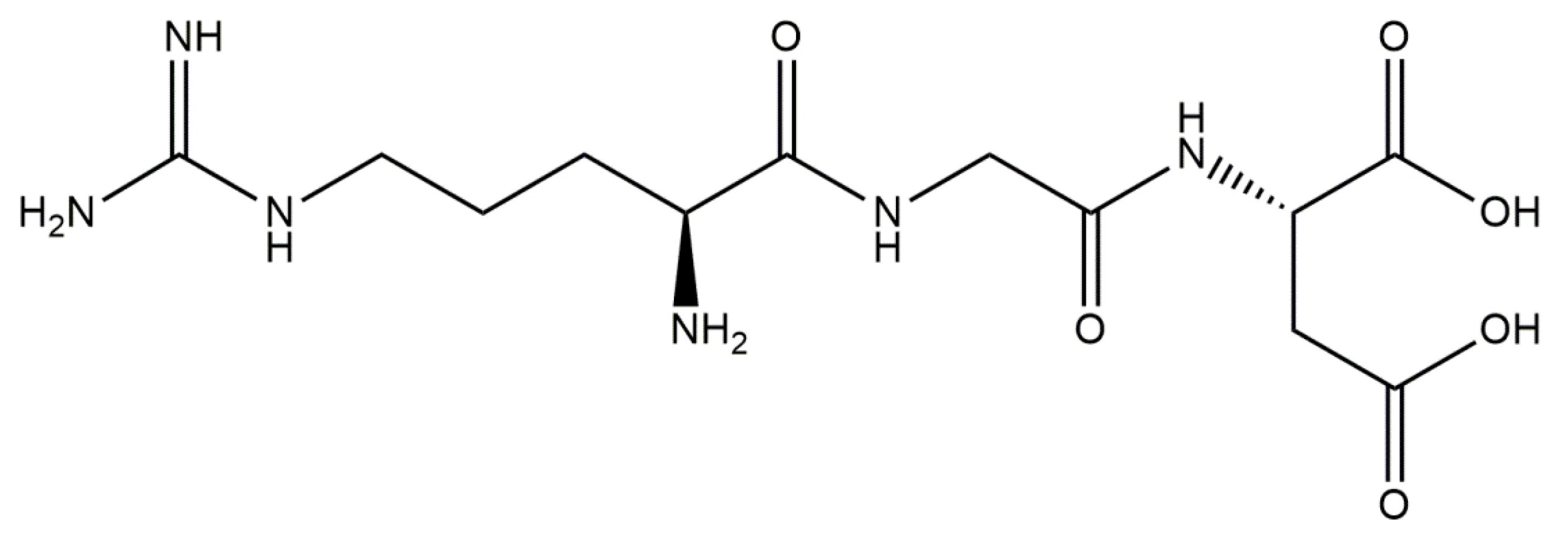
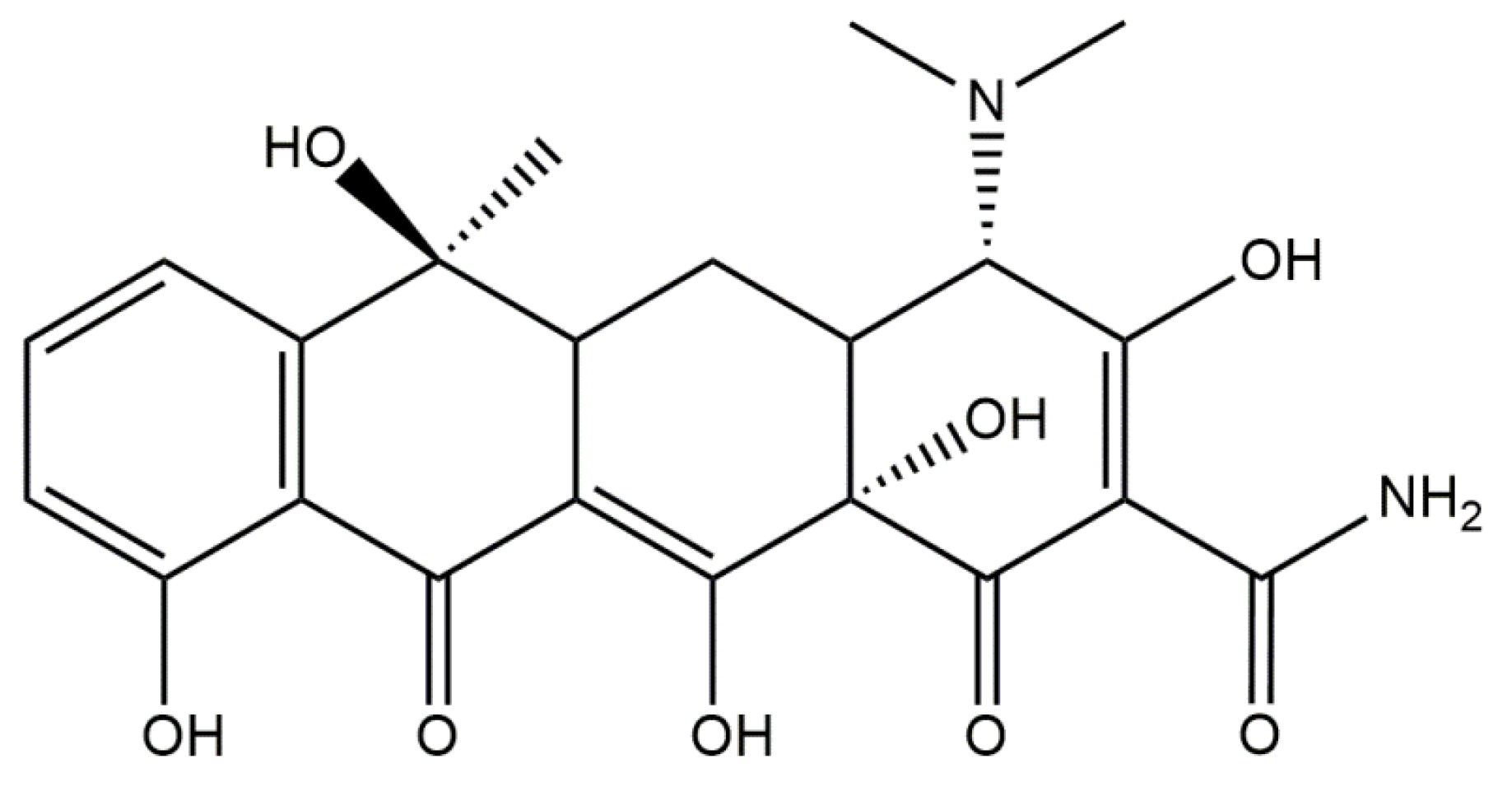


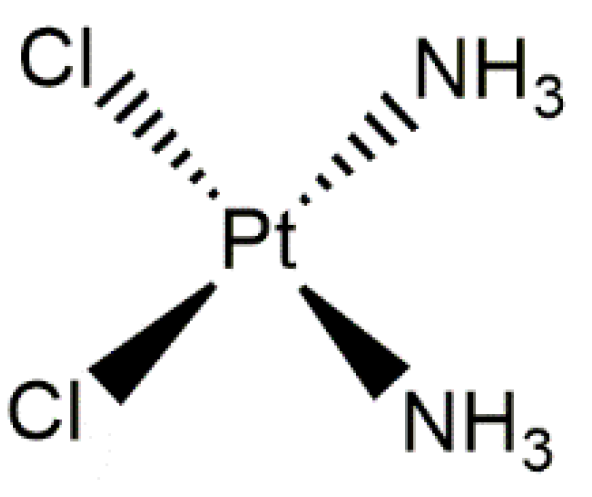
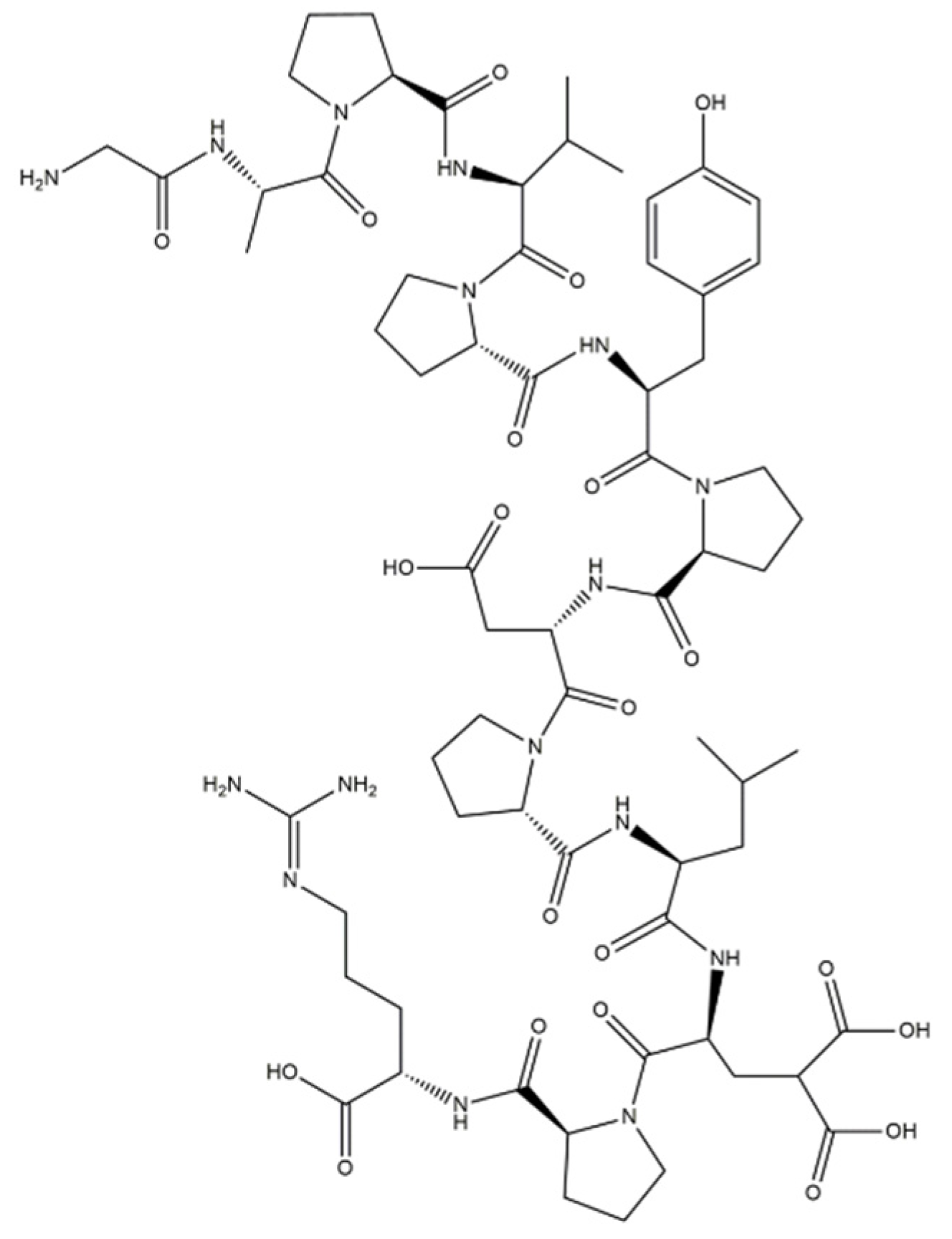
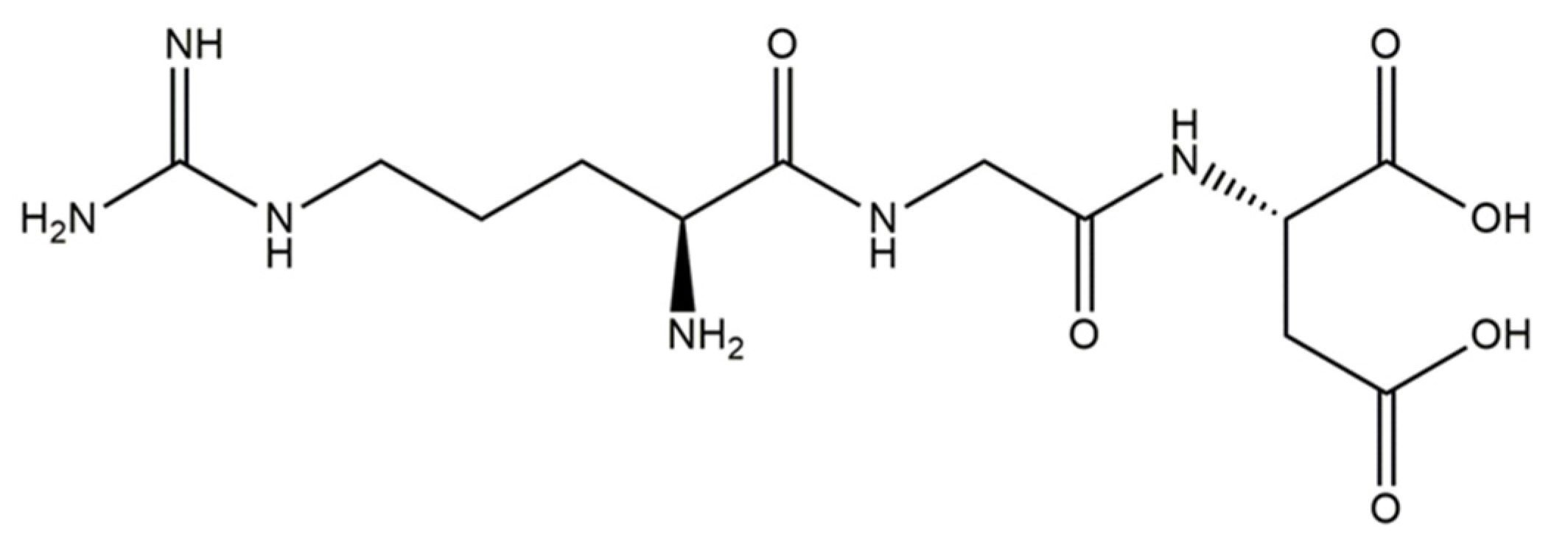
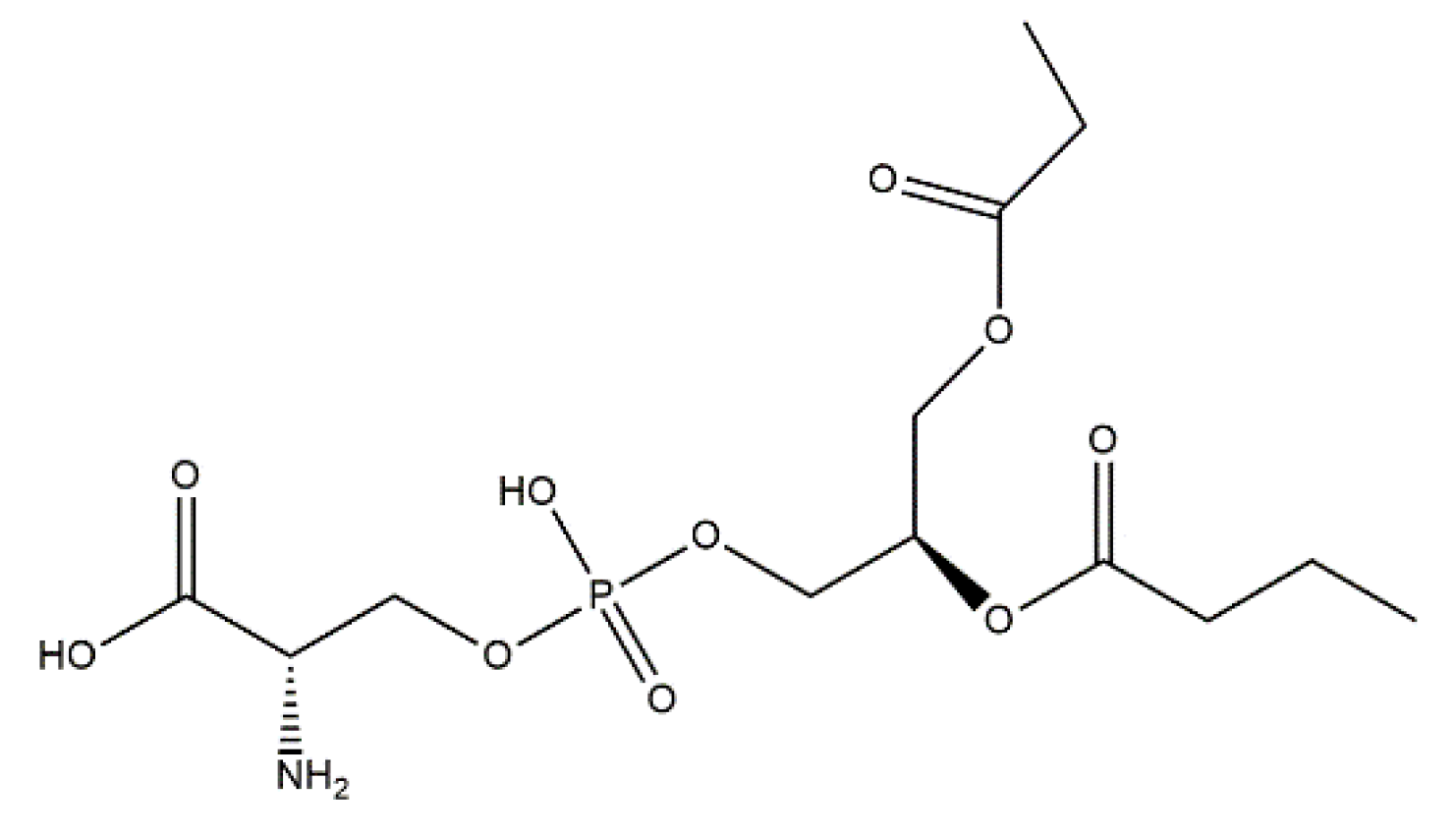

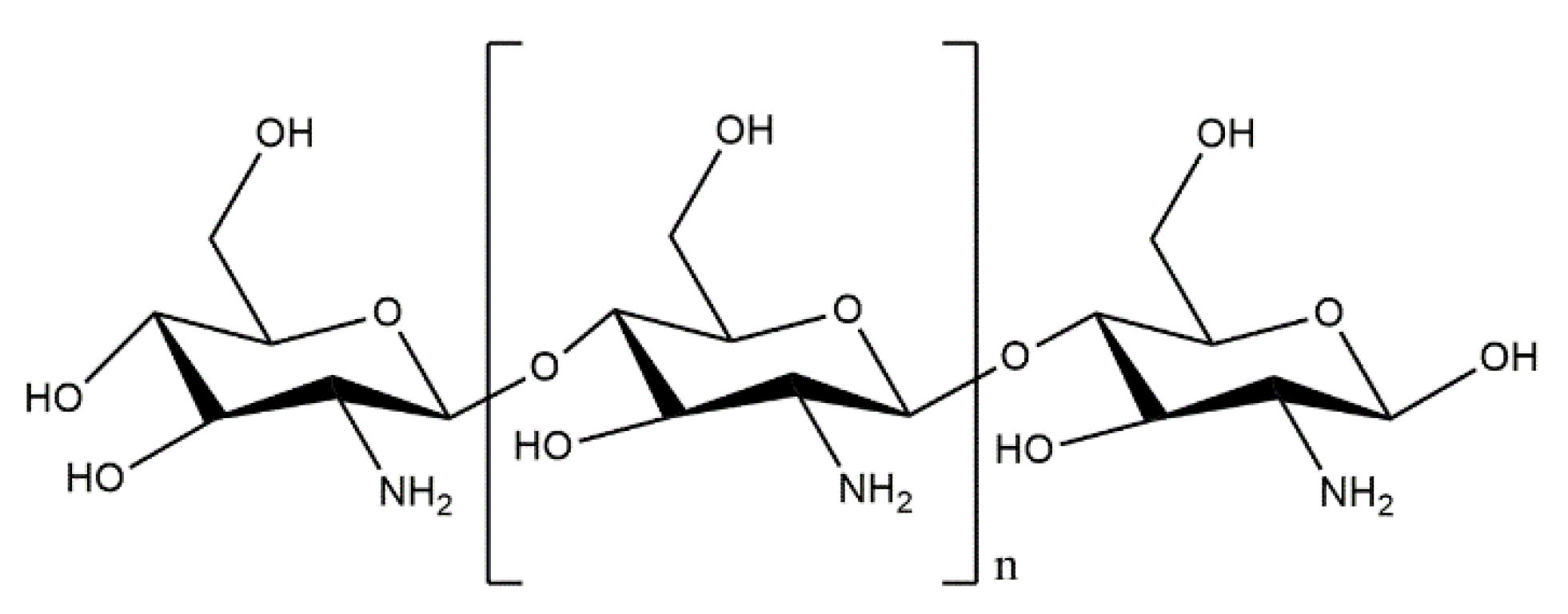
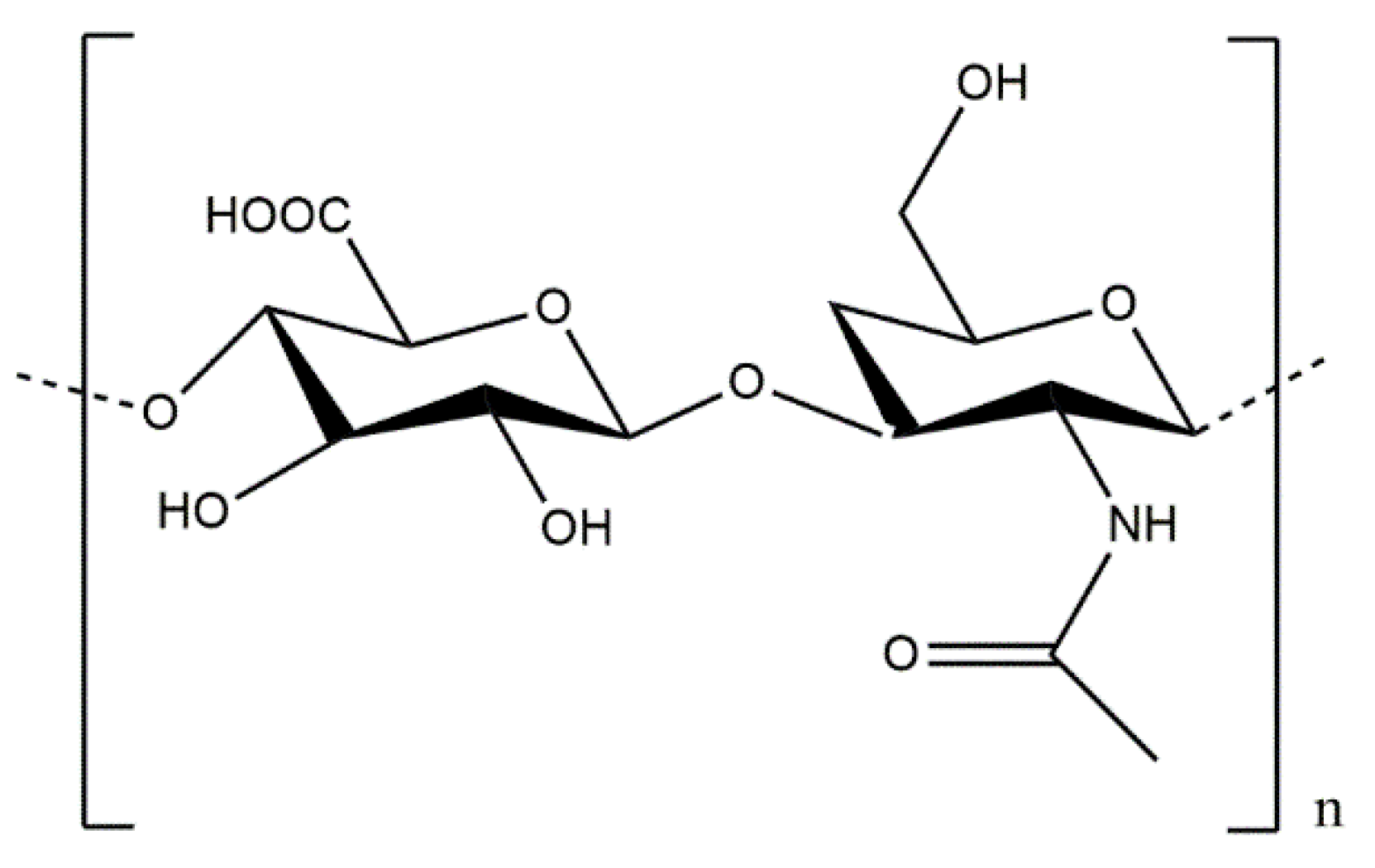
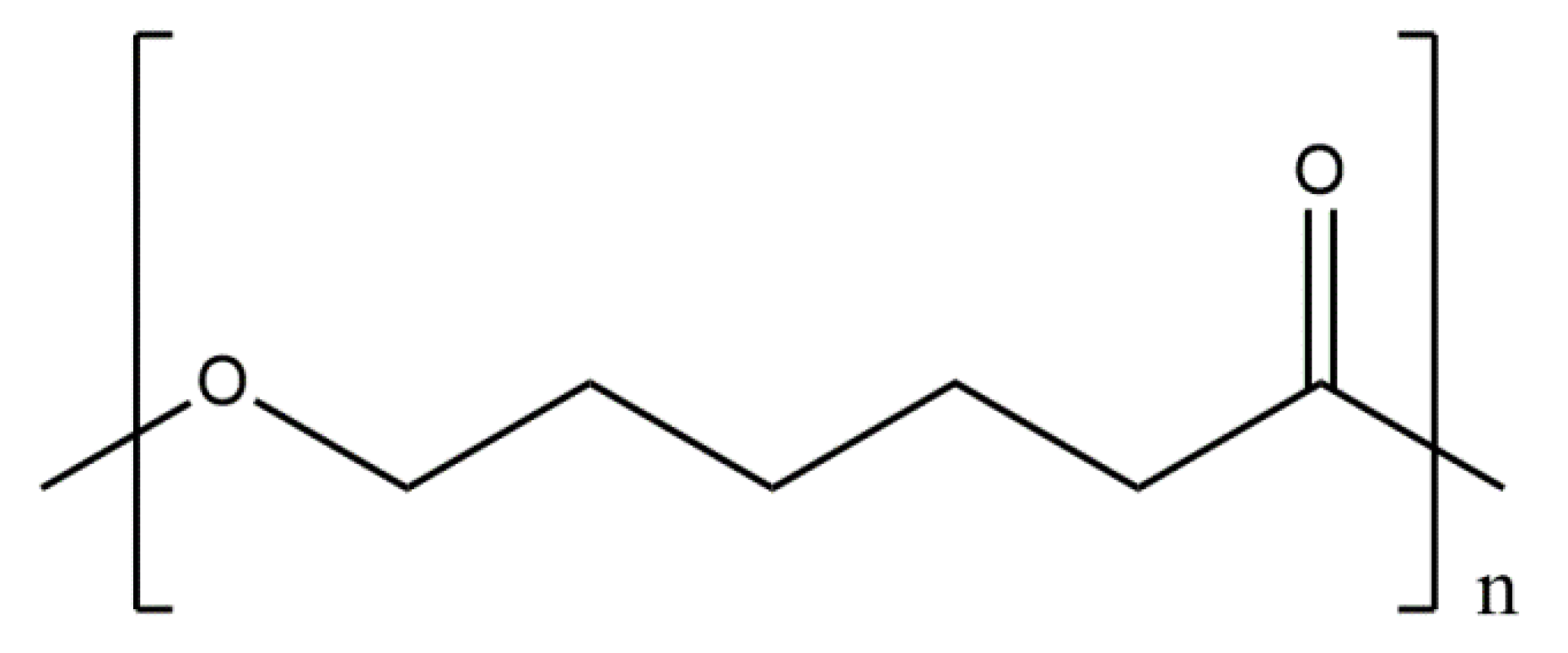

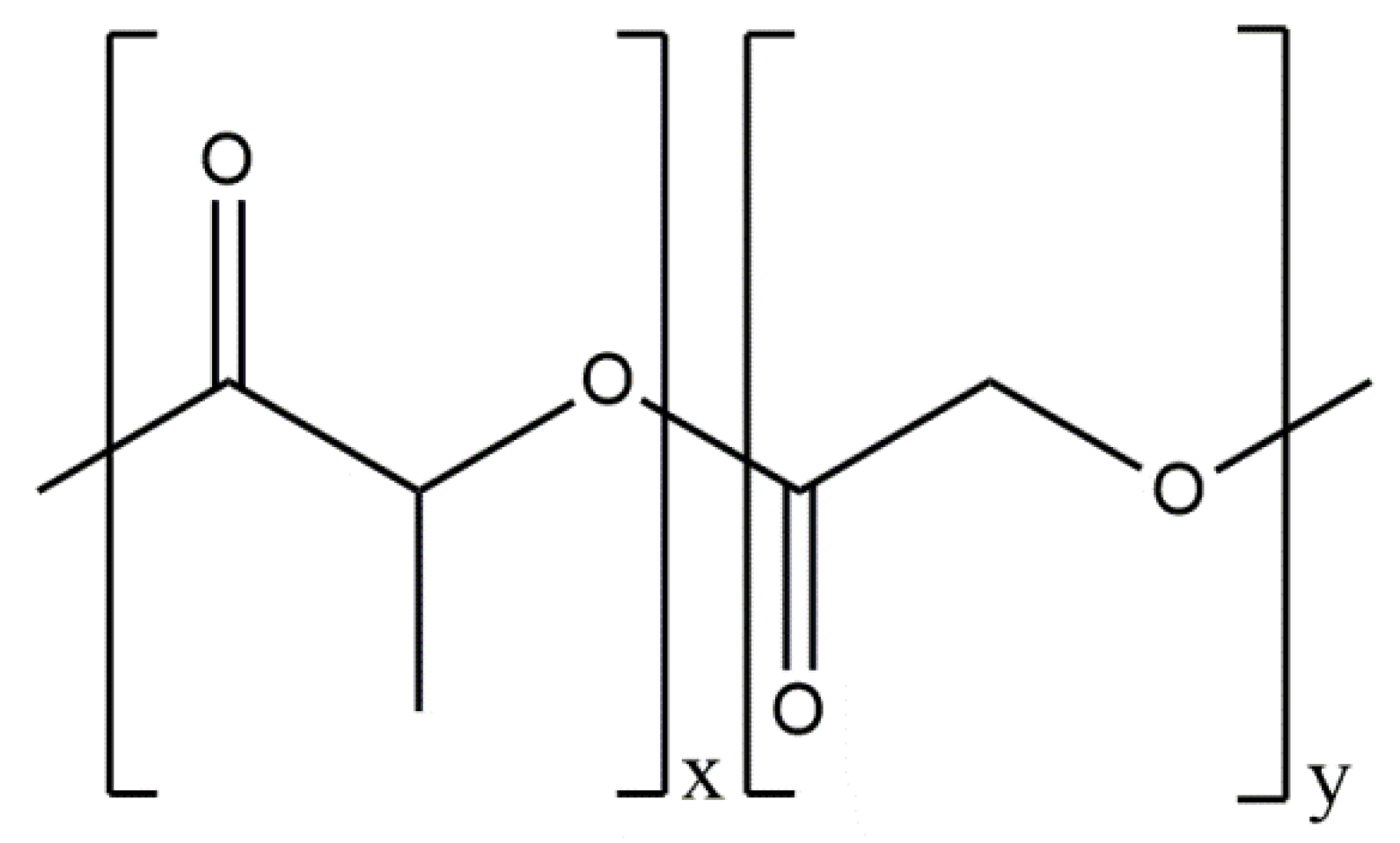
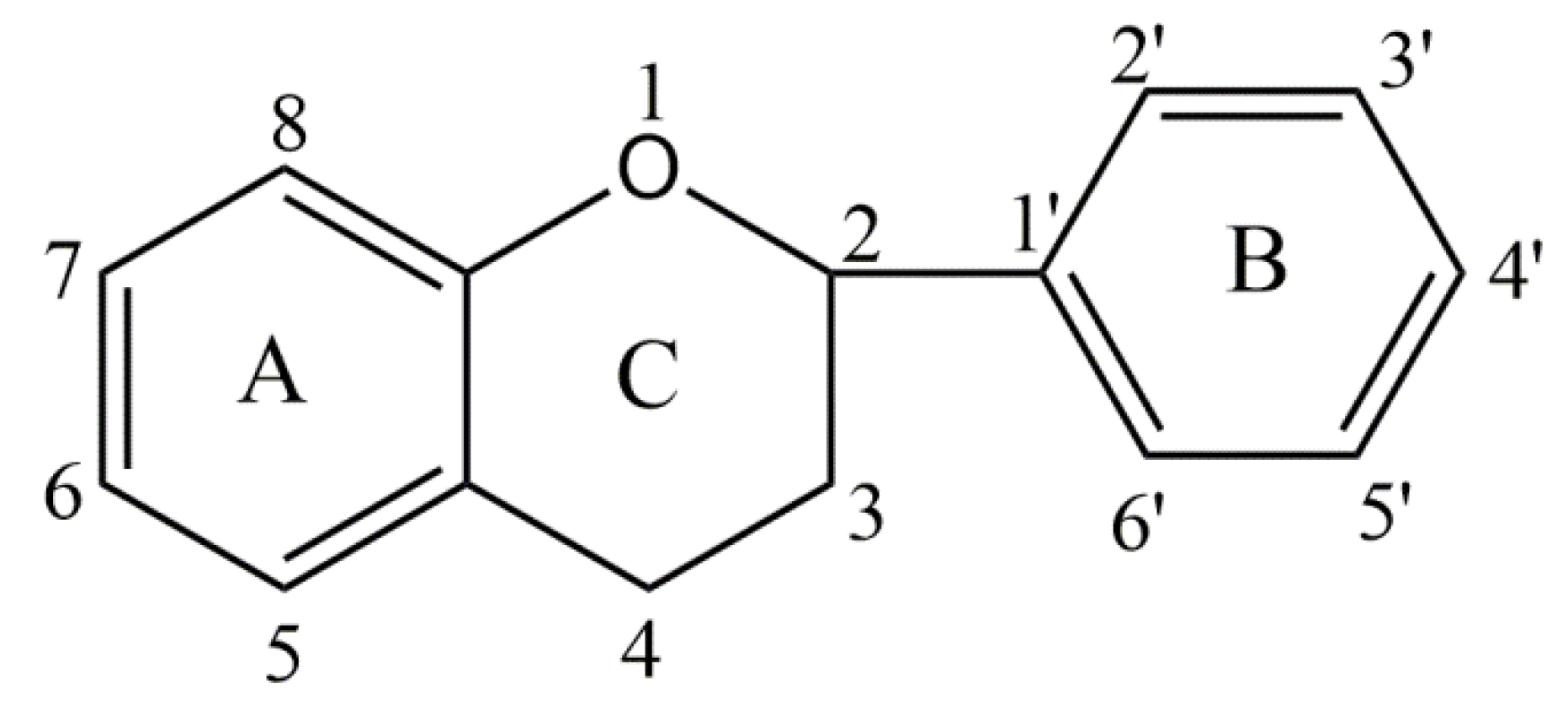
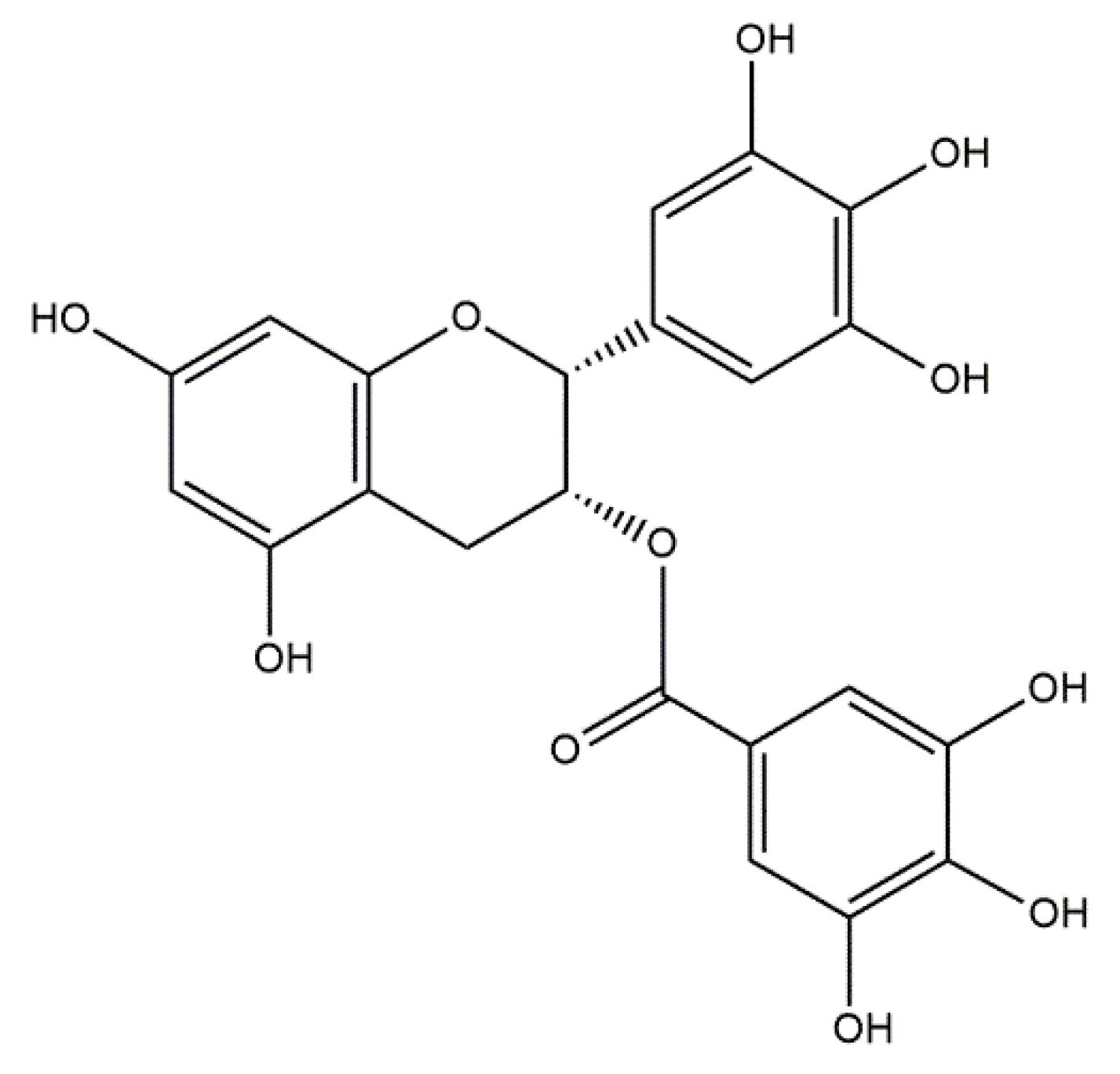


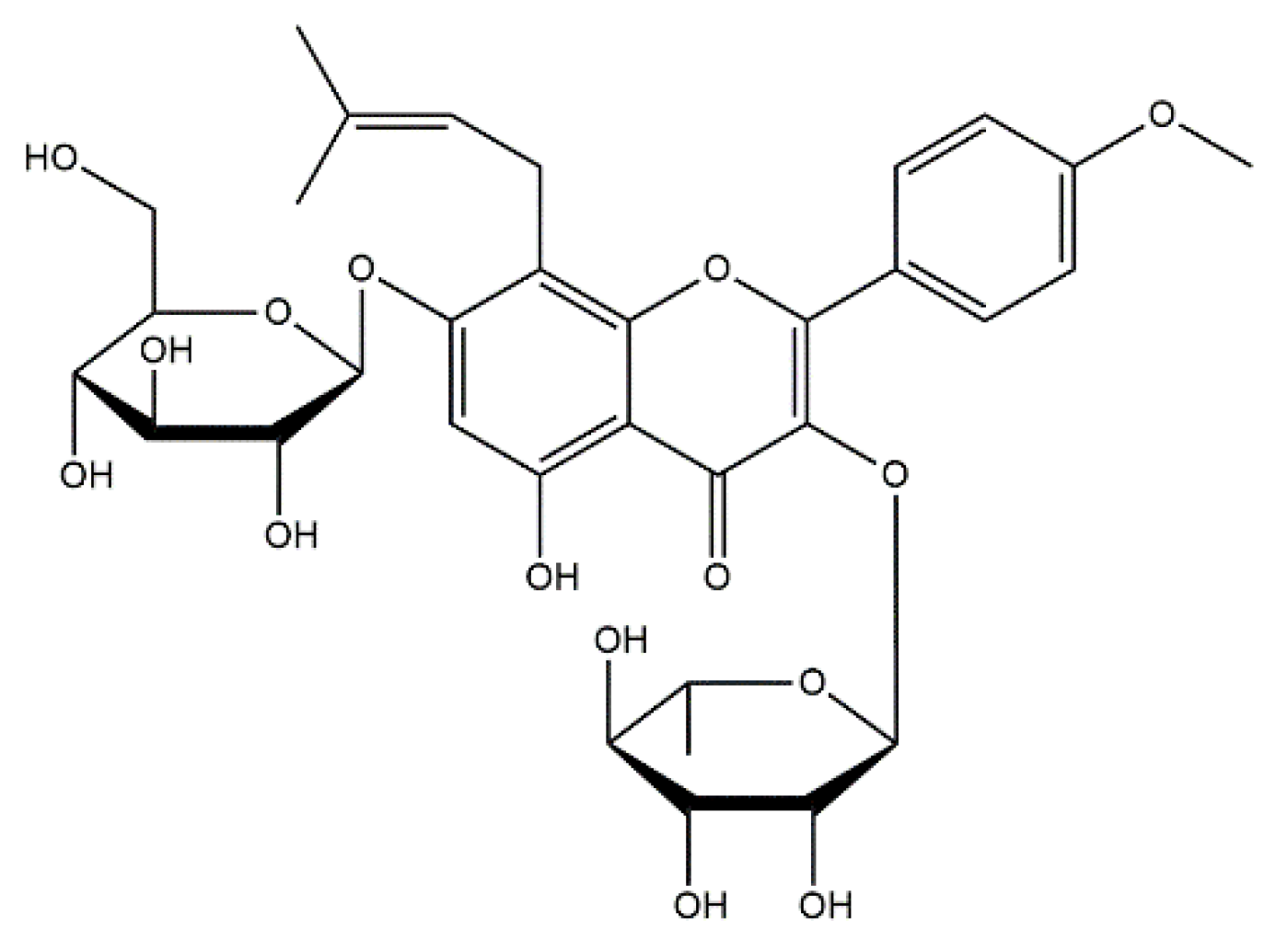
| Type | Molecular Composition | Tissue Distribution | Genes (Genomic Localization) | Supramolecular Structure and Organization |
|---|---|---|---|---|
| I | [a1(I)]2a2(I) | bone, dermis, tendon, ligaments, cornea | COL1A1 (17q21.31–q22) COL1A2 (7q22.1) | Fibril-forming collagens |
| II | [a1(II)]3 | cartilage, vitreous body, nucleus pulposus | COL2A1 (12q13.11–q13.2) | |
| III | [a1(III)]3 | skin, vessel wall, reticular fibers of most tissues (lungs, liver, spleen, etc.) | COL3A1 (2q31) | |
| V | a1(V),a2(V),a3(V) | lung, cornea, bone, fetal membranes; together with type I collagen | COL5A1 (9q34.2–q34.3) COL5A2 (2q31) COL5A3 (19p13.2) | |
| XI | a1(XI)a2(XI)a3(XI) | cartilage, vitreous body | COL11A1 (1p21) COL11A2 (6p21.3) COL11A3 = COL2A1 | |
| IV | [a1(IV)]2a2(IV); a1–a6 | basement membranes | COL4A1 (13q34) COL4A2 (13q34) COL4A3 (2q36–q37) COL4A4 (2q36–q37) COL4A5 (Xq22.3) COL4A6 (Xp22.3) | Basement-membrane collagens |
| VI | a1(VI),a2(VI),a3(VI) | widespread: dermis, cartilage, placenta, lungs, vessel wall, intervertebral disc | COL6A1 (21q22.3) COL6A2 (21q22.3) COL6A3 (2q37) | Microfibrillar collagen |
| VII | [a1(VII)]3 | skin, dermal–epidermal junctions, oral mucosa, cervix | COL7A1 (3p21.3) | Anchoring fibrils |
| VIII | [a1(VIII)]2a2(VIII) | endothelial cells, Descemet’s membrane | COL8A1 (3q12–q13.1) COL8A2 (1p34.3–p32.3) | Hexagonal network-forming collagens |
| X | [a3(X)]3 | hypertrophic cartilage | COL10A1 (6q21–q22.3) | |
| IX | a1(IX)a2(IX)a3(IX) | cartilage, vitreous humor, cornea | COL9A1 (6q13) COL9A2 (1p33–p32.2) | FACIT collagens |
| XII | [a1(XII)]3 | perichondrium, ligaments, tendon | COL12A1 (6q12–q13) | |
| XIV | [a1(XIV)]3 | dermis, tendon, vessel wall, placenta, lungs, liver | COL9A1 (8q23) | |
| XIX | [a1(XIX)]3 | human rhabdomyosarcoma | COL19A1 (6q12–q14) | |
| XX | [a1(XX)]3 | corneal epithelium, embryonic skin, sternal cartilage, tendon | COL21A1 (6p12.3–11.2) | |
| XXI | [a1(XXI)]3 | blood vessel wall | COL21A1 (6p12.3–11.2) | |
| XIII | [a1(XIII)]3 | epidermis, hair follicle, endomysium, intestine, chondrocytes, lungs, liver | COL13A1 (10q22) | Transmembrane collagens |
| XVII | [a1(XVII)]3 | dermal–epidermal junctions | COL17A1 (10q24.3) | Multiplexins |
| XV | [a1(XV)]3 | fibroblasts, smooth muscle cells, kidney, pancreas | COL15A1 (9q21–q22) | |
| XVI | [a1(XVI)]3 | fibroblasts, amnion, keratinocytes | COL16A1 (1p34) | |
| XVIII | [a1(XVIII)]3 | lungs, liver | COL18A1 (21q22.3) |
| Substrate | Deposition of HAp | Deposition of Collagen | HAp Precursors | Collagen Type | Ref. |
|---|---|---|---|---|---|
| Ti plate | Single-step electrochemically-assisted deposition | Ca(NO3)2, NH4H2PO4 | Type I extracted from equine Achilles tendon | [240] | |
| Ti-6Al-4V of medical grade | (1) Self-assembly in SBF; hydrothermal method | (2) Iimmersion in collagen solution | SBF; CaHPO4×2H2O, Ca(OH)2 | Type I extracted from porcine skin | [242] |
| Plate-shape dental implants (CP Ti) | Single-step self-assembly | CaCl2·2H2O, NaH2PO4×H2O, NaHCO3 (supersaturated calcification solution) | Type I from bovine Achilles tendon | [234] | |
| NiTi shape memory alloy disks | Single-step electrochemically assisted deposition | Double-strength simulated body fluid (2SBF) | Type I | [238] | |
| Ti-6Al-4V disks | (1) Liquid precursor plasma-spraying process | (2) Immersion in collagen solution | Liquid HAp precursor prepared through a wet-chemical route | Type I derived from bovine skin | [243] |
| CP Ti discs | Spin-coating of the HAp/Coll sols | Ca(OH)2, H3PO4 | Type I derived from calf skin | [245] | |
| Ti disc or implant | (1) Aerosol deposition | (2) Immersion in collagen solution | Commercial HAp powder | Type I from calf skin | [244] |
| CP Ti screw-shaped dental implants | Single-step self-assembly | CaCl2·2H2O, NaH2PO4×H2O, NaHCO3 (supersaturated calcification solution) | Type I from bovine Achilles tendon | [235] | |
| CP Ti rod | Dipping rod several times into the suspension | Ca(OH)2, H3PO4 | Not specified | [236] | |
| Ti-6Al-4V discs | Single-step self-assembly | SBF | Type I extracted from rat tails | [237] | |
| Sintered porous Ti substrate | (1) Immersion in SBF | (2) Immersion in collagen solution | SBF | Humanlike collagen | [241] |
| Ti plate | Single-step electrochemically assisted deposition | CaCl2, NaH2PO4, NaCl | Type I | [239] | |
| Polydopamine grafted stainless steel (SS316L) | (2) Immersion in SBF | (1) Immersion in collagen solution | SBF | Type I | [246] |
| Bone Healing Stages | Cytokines | Biological Process Steps |
|---|---|---|
| Inflammation | IL-1, IL-6 TGF-β BMP-2 PDGF GDF-8 RANKL and M-CSF OPG | Hematoma Inflammation MSCs recruitment |
| Cartilage formation | TGF-β2 TGF-β3 BMP-5, BMP-6 VEGFs Angiopoietin-1, Angiopoietin-2 | Chondrogenesis Endochondral ossification Cell proliferation Intramembranous ossification Vascular ingrowth New angiogenesis |
| Cartilage resorption and primary bone formation | VEGF Angiopoietin BMP-3, BMP-4, BMP-7, BMP-8 RANKL & M-CSF | Chondrocyte apoptosis Matrix proteolysis Cartilage resorption Active osteogenesis Bone cell recruitment Woven bone formation Neo-angiogenesis |
| Secondary bone formation and remodeling | IL-1, IL-6 RANKL and M-CSF | Bone remodeling Osteoblast activity Marrow establishment |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sobczak-Kupiec, A.; Drabczyk, A.; Florkiewicz, W.; Głąb, M.; Kudłacik-Kramarczyk, S.; Słota, D.; Tomala, A.; Tyliszczak, B. Review of the Applications of Biomedical Compositions Containing Hydroxyapatite and Collagen Modified by Bioactive Components. Materials 2021, 14, 2096. https://doi.org/10.3390/ma14092096
Sobczak-Kupiec A, Drabczyk A, Florkiewicz W, Głąb M, Kudłacik-Kramarczyk S, Słota D, Tomala A, Tyliszczak B. Review of the Applications of Biomedical Compositions Containing Hydroxyapatite and Collagen Modified by Bioactive Components. Materials. 2021; 14(9):2096. https://doi.org/10.3390/ma14092096
Chicago/Turabian StyleSobczak-Kupiec, Agnieszka, Anna Drabczyk, Wioletta Florkiewicz, Magdalena Głąb, Sonia Kudłacik-Kramarczyk, Dagmara Słota, Agnieszka Tomala, and Bożena Tyliszczak. 2021. "Review of the Applications of Biomedical Compositions Containing Hydroxyapatite and Collagen Modified by Bioactive Components" Materials 14, no. 9: 2096. https://doi.org/10.3390/ma14092096
APA StyleSobczak-Kupiec, A., Drabczyk, A., Florkiewicz, W., Głąb, M., Kudłacik-Kramarczyk, S., Słota, D., Tomala, A., & Tyliszczak, B. (2021). Review of the Applications of Biomedical Compositions Containing Hydroxyapatite and Collagen Modified by Bioactive Components. Materials, 14(9), 2096. https://doi.org/10.3390/ma14092096