Investigations of High-Strength Mg–Al–Ca–Mn Alloys with a Broad Range of Ca+Al Contents
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Thermodynamic Calculations
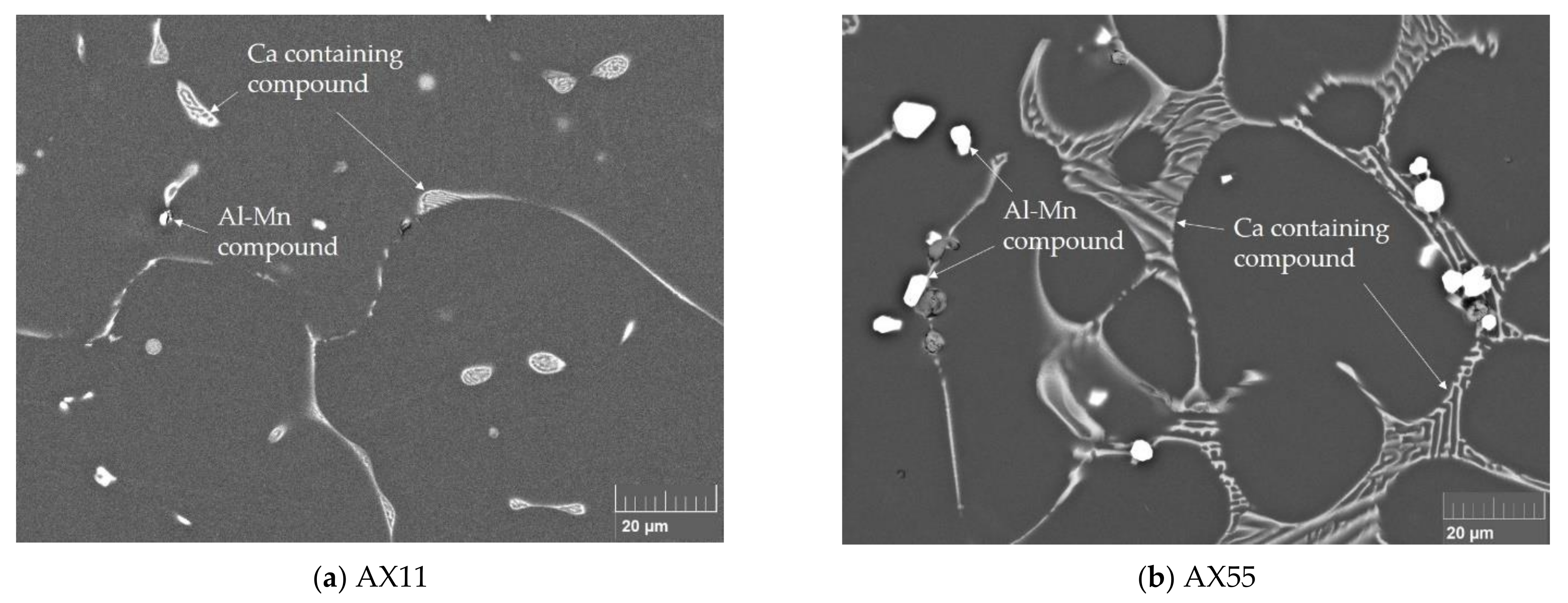
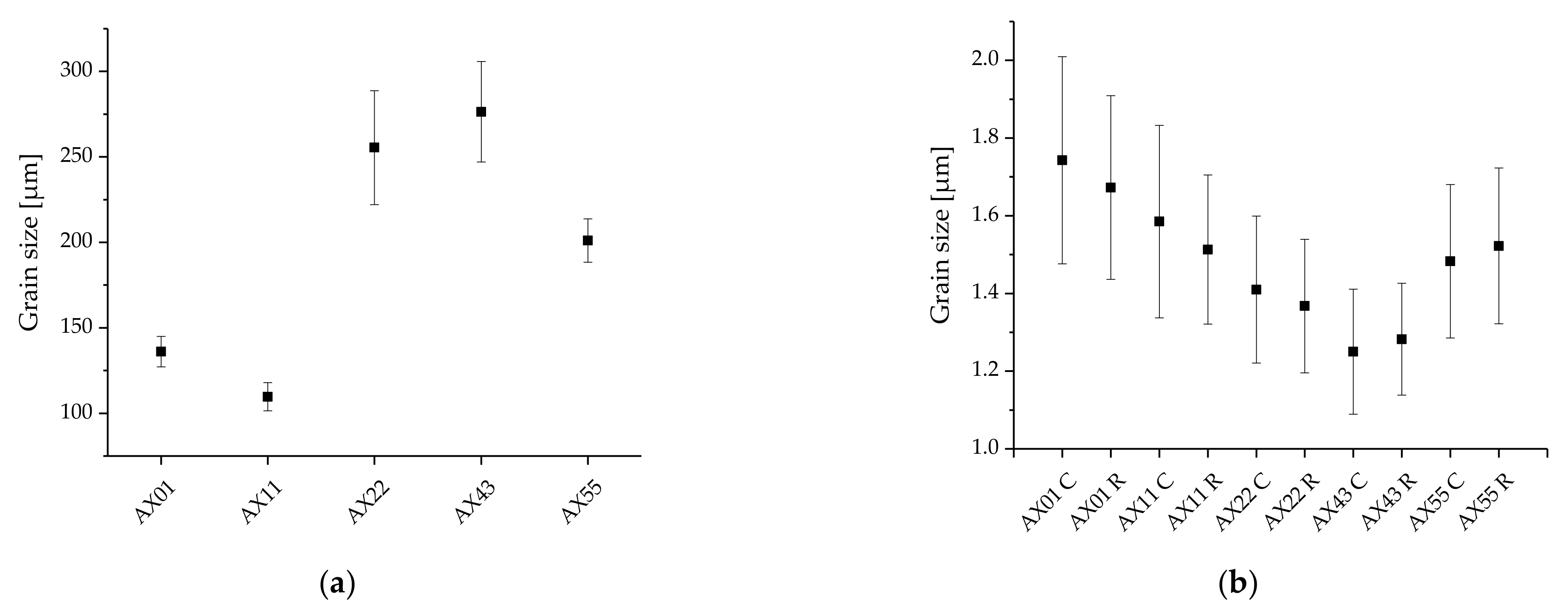
3.2. Microstructure
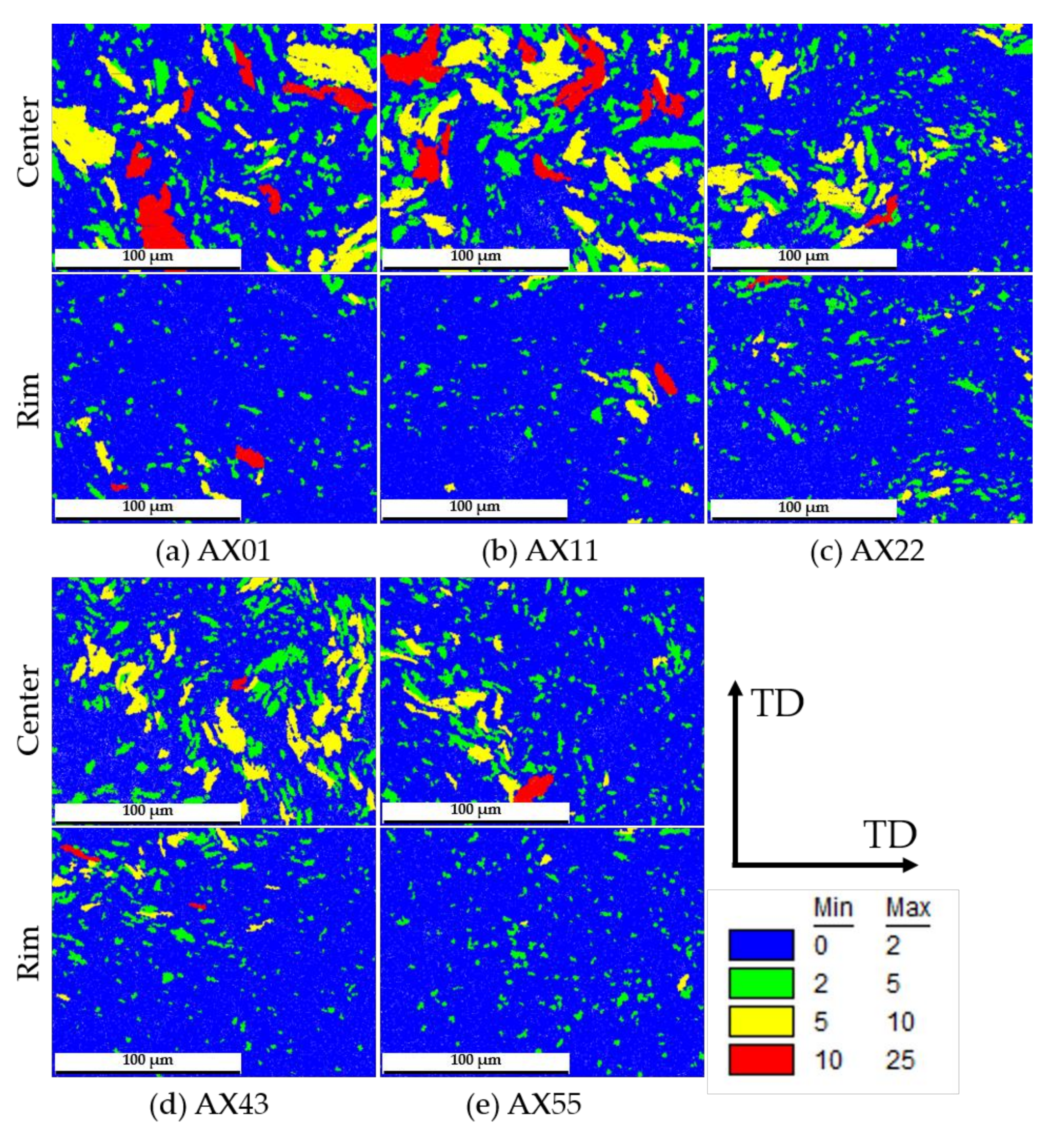
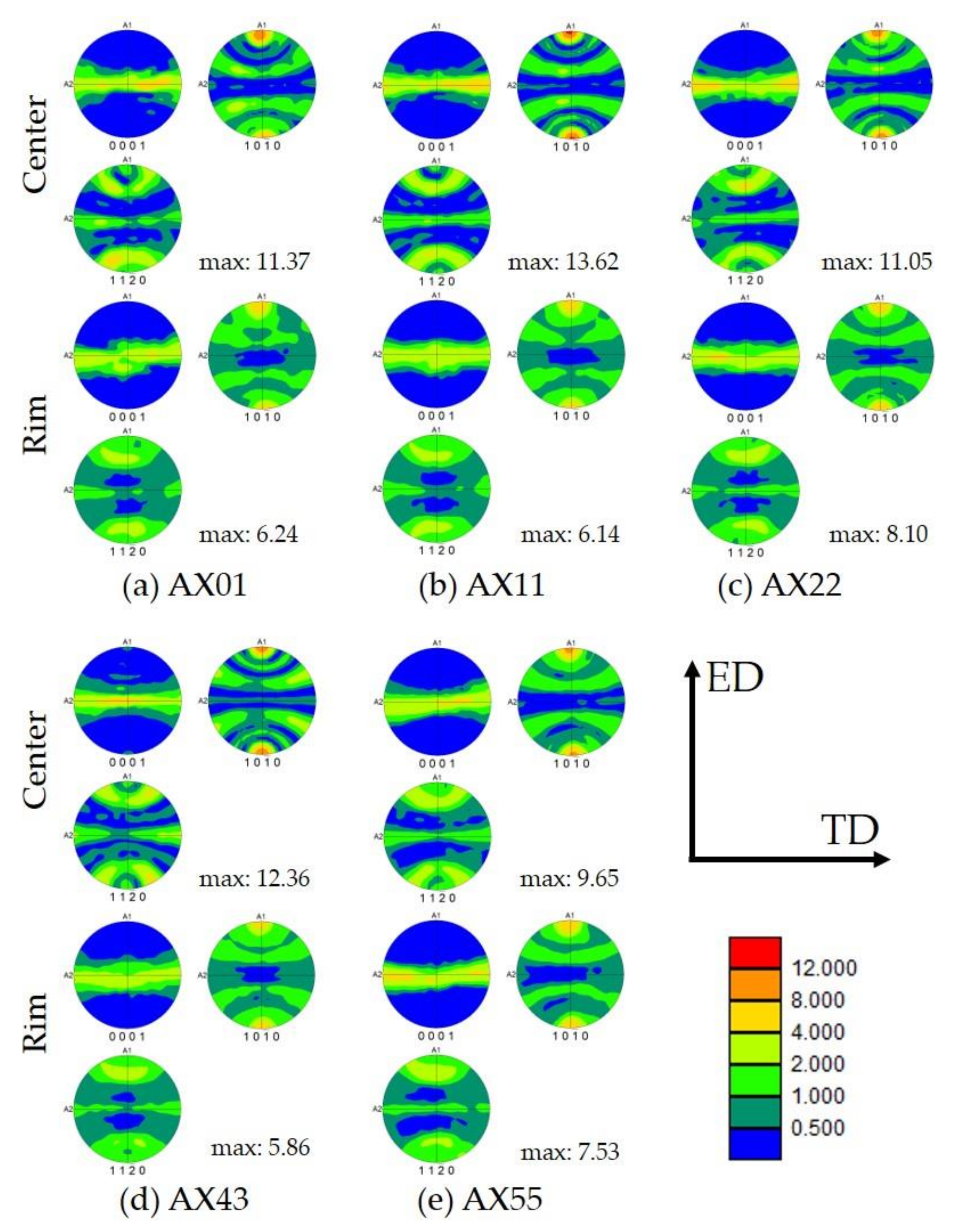
3.3. Texture Measurements of As-Extruded Alloys
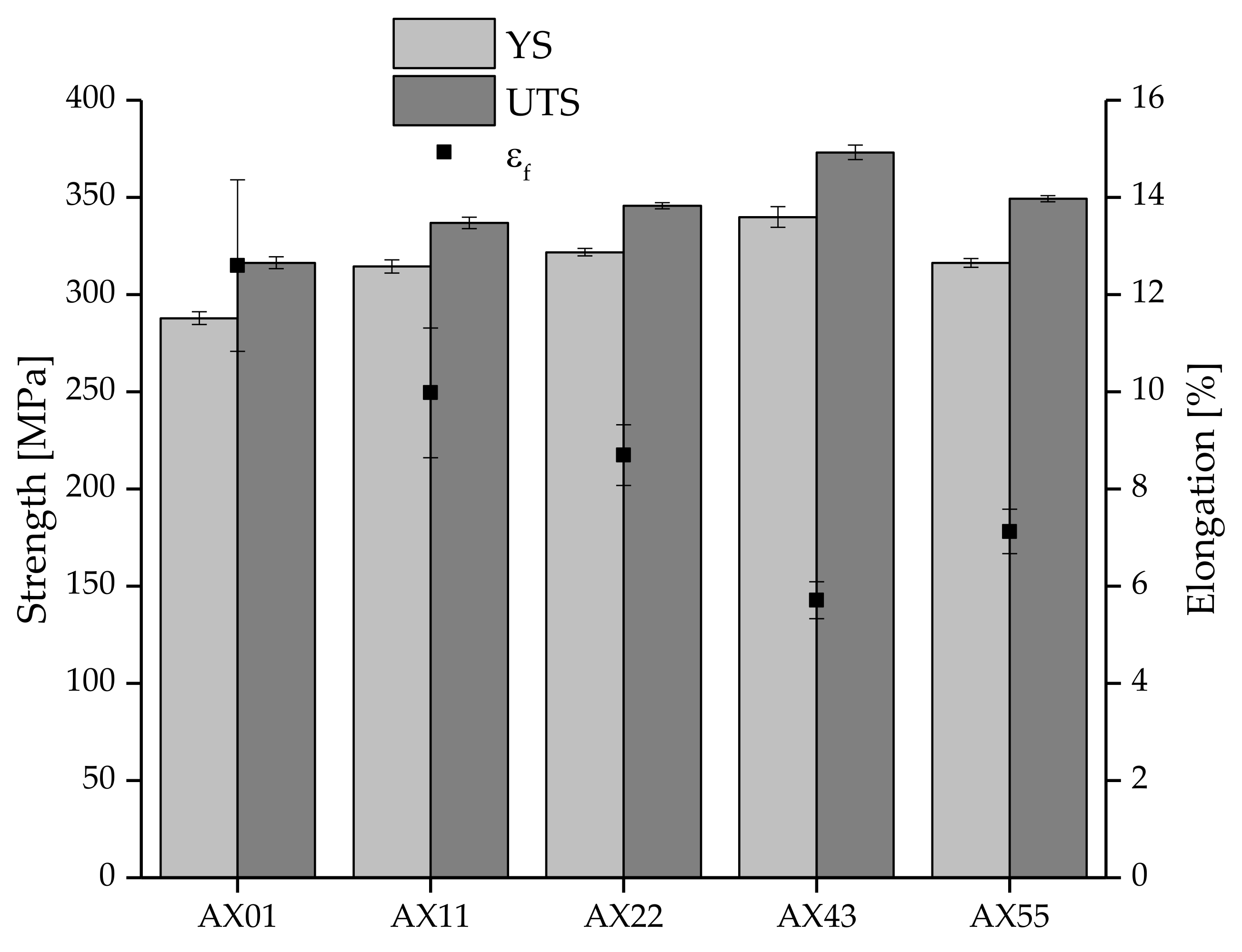
3.4. Mechanical Properties
3.5. Thermal Stability
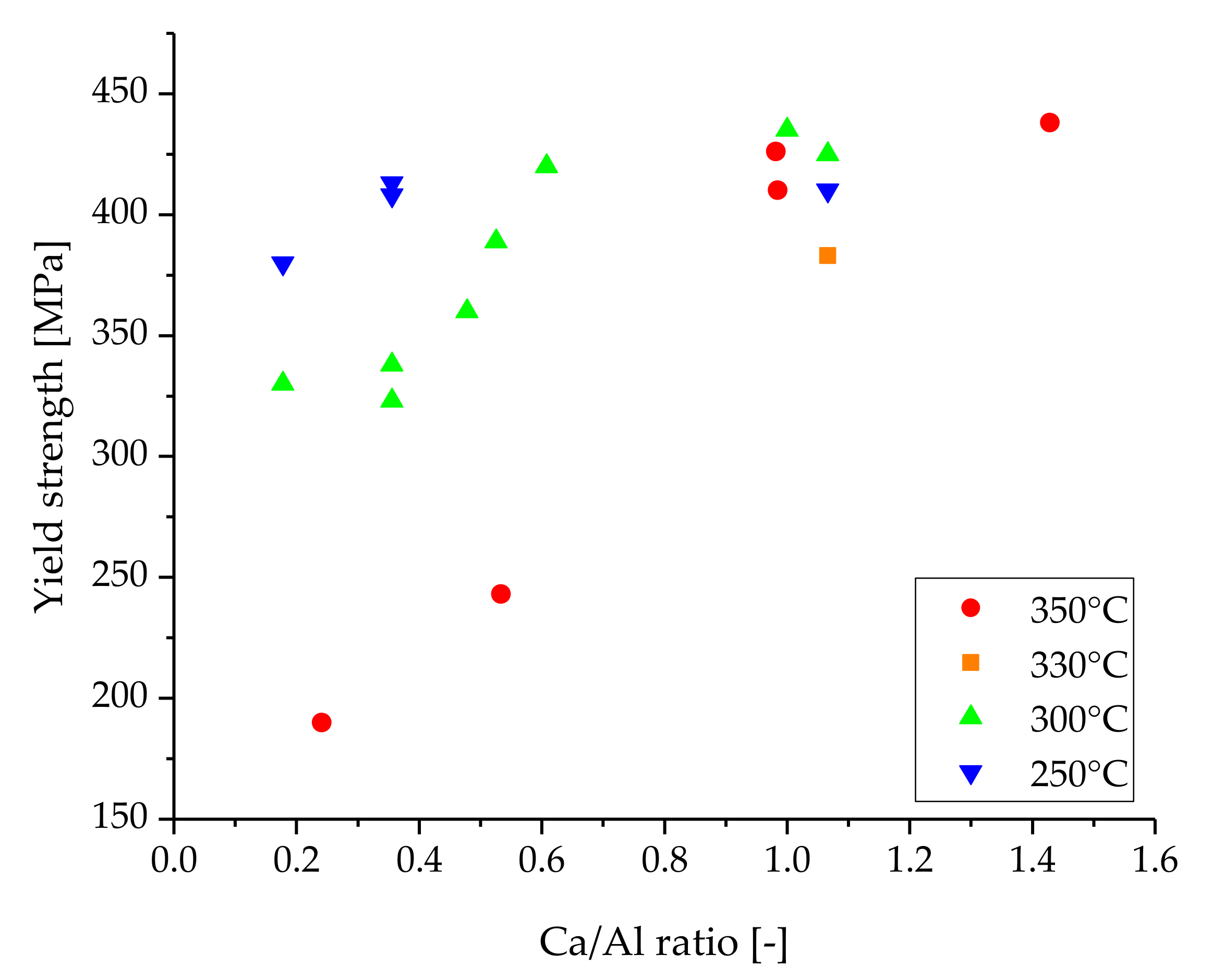
4. Discussion
5. Conclusions
- By maintaining a constant Ca/Al ratio, a wide selection of alloys with similar structure and attractive mechanical properties (YS > 285 MPa, elongation > 5.5%) can be created.
- The combination of extrusion process and intermetallic phase formation results in a bi-modal grain structure with a large fraction (up to 89%) of fine recrystallized grains (<2 µm). The intermetallic phases enable this fine grain structure by inhibiting grain growth and facilitating recrystallization.
- Alloy production can be realized with the use of abundant raw materials (Mg, Al, Ca, and Mn).
- No heat treatments are necessary during processing.
- The obtained mechanical properties are well maintained after exposure for 24 h at elevated temperatures (250 °C), as the intermetallic phases present are thermally stable.
- No narrow tolerances for chemical composition (except a Ca/Al ratio of >0.9) need to be maintained; therefore, alloy production and recycling are simplified.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Alloy | Avg. Grain Size (Recrystallized) | Recrystallized Area Fraction | Max. Texture Intensity | Avg. Schmid Factor | |
|---|---|---|---|---|---|
| [µm2] | [%] | [m.r.u.] | [1] | ||
| AX01 | Center | 3.04 ± 0.07 | 67.7 | 11.37 | 0.15 |
| Rim | 2.80 ± 0.06 | 89.0 | 6.24 | 0.20 | |
| AX11 | Center | 2.51 ± 0.06 | 55.1 | 13.62 | 0.15 |
| Rim | 2.29 ± 0.04 | 88.7 | 6.14 | 0.35 | |
| AX22 | Center | 1.99 ± 0.04 | 67.0 | 11.05 | 0.17 |
| Rim | 1.87 ± 0.03 | 81.2 | 8.10 | 0.19 | |
| AX43 | Center | 1.56 ± 0.03 | 62.8 | 12.36 | 0.17 |
| Rim | 1.64 ± 0.02 | 86.0 | 5.86 | 0.35 | |
| AX55 | Center | 2.20 ± 0.04 | 78.1 | 9.65 | 0.16 |
| Rim | 2.32 ± 0.04 | 88.3 | 7.53 | 0.17 | |
| Alloy | YS [MPa] | UTS [MPa] | εf [%] |
|---|---|---|---|
| AX01 | 287.9 ± 3.3 | 316.3 ± 3.1 | 12.6 ± 1.8 |
| AX11 | 314.5 ± 3.4 | 336.9 ± 3.0 | 10.0 ± 1.3 |
| AX22 | 321.8 ± 2.0 | 345.7 ± 1.6 | 8.7 ± 0.6 |
| AX43 | 339.9 ± 5.3 | 373.2 ± 3.7 | 5.7 ± 0.4 |
| AX55 | 316.3 ± 2.2 | 349.3 ± 1.6 | 7.1 ± 0.5 |
| Alloy | YS [MPa] | UTS [MPa] | εf [%] | Ca/Al [-] | Stock Material | Extrusion Ratio | Forming Temperature [°C] | Ram Speed [mm/s] | Source | |
|---|---|---|---|---|---|---|---|---|---|---|
| AXM3304 | 426 | 439 | 3.9 | 0.9 | as-cast | 1:12 | 350 | 13 | [20] | |
| AXM3404 | 438 | 457 | 2.5 | 1.3 | as-cast | 1:12 | 350 | 13 | [20] | |
| AXM4104 | 190 | 294 | 29.9 | 0.3 | as-cast | 1:12 | 350 | 13 | [20] | |
| AXM4204 | 243 | 314 | 16.6 | 0.5 | as-cast | 1:12 | 350 | 13 | [20] | |
| AXM4304 | 410 | 420 | 5.6 | 0.9 | as-cast | dir., | 1:20 | 350 | 0.1 | [27] |
| XAZM1100 | 410 | 1 | 1.0 | cast & hom. | indir., | 1:20 | 250 | 0.8 | [28] | |
| 425 | 11 | 1.0 | cast & hom. | indir., | 1:20 | 300 | 0.8 | [28] | ||
| 383 | 12.5 | 1.0 | cast & hom. | indir., | 1:20 | 330 | 0.8 | [28] | ||
| AZXM310503 | 380 | 420 | 5.1 | 0.2 | cast & hom. | 1:20 | 250 | 0.1 | [29] | |
| 330 | 360 | 13.5 | 0.2 | cast & hom. | 1:20 | 300 | 0.8 | [29] | ||
| AZXM31103 | 413 | 432 | 7.5 | 0.3 | cast & hom. | 1:20 | 250 | 0.1 | [29] | |
| 323 | 356 | 11.8 | 0.3 | cast & hom. | 1:20 | 300 | 0.8 | [29] | ||
| AXM3103 | 408 | 432 | 6.5 | 0.3 | cast & hom. | 1:20 | 250 | 0.1 | [29] | |
| 338 | 372 | 9.6 | 0.3 | cast & hom. | 1:20 | 300 | 0.8 | [29] | ||
| AXM5303 | 420 | 451 | 4.1 | 0.6 | as-cast | indir., | 1:12 | 300 | 0.1 | [30] |
| AXM6303 | 389 | 423 | 3.6 | 0.5 | as-cast | indir., | 1:12 | 300 | 0.1 | [30] |
| AXM7303 | 360 | 414 | 5.4 | 0.5 | as-cast | indir., | 1:12 | 300 | 0.1 | [30] |
| XAZM11 | 435 | 449 | 4.2 | 1.0 | cast & hom. | 1:23 | 300 | 0.4 | [31] | |
References
- Yu, H.; Xin, Y.; Wang, M.; Liu, Q. Hall-petch relationship in mg alloys: A review. J. Mater. Sci. Technol. 2018, 34, 248–256. [Google Scholar] [CrossRef]
- Li, C.; Sun, H.; Fang, W. Effect of extrusion temperatures on microstructures and mechanical properties of Mg-3Zn-0.2Ca-0.5Y alloy. Procedia Eng. 2014, 81, 610–615. [Google Scholar] [CrossRef] [Green Version]
- Ding, S.X.; Chang, C.P.; Kao, P.W. Effects of processing parameters on the grain refinement of magnesium alloy by equal-channel angular extrusion. Metall. Mater. Trans. A 2009, 40, 415. [Google Scholar] [CrossRef]
- Hofstetter, J.; Rüedi, S.; Baumgartner, I.; Kilian, H.; Mingler, B.; Povoden-Karadeniz, E.; Pogatscher, S.; Uggowitzer, P.J.; Löffler, J.F. Processing and microstructure—Property relations of High-Strength Low-Alloy (HSLA) Mg–Zn–Ca alloys. Acta Mater. 2015, 98, 423–432. [Google Scholar] [CrossRef]
- Xu, C.; Nakata, T.; Qiao, X.; Zheng, M.; Wu, K.; Kamado, S. Effect of LPSO and SFs on microstructure evolution and mechanical properties of Mg-Gd-Y-Zn-Zr alloy. Sci. Rep. 2017, 7, 40846. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Shen, Y.; Jiang, B.; Tang, A.; Chai, Y.; Song, J.; Yang, T.; Huang, G.; Pan, F. A Good balance between ductility and stretch formability of dilute Mg-Sn-Y sheet at room temperature. Mater. Sci. Eng. A 2018, 736, 404–416. [Google Scholar] [CrossRef]
- Nie, K.; Zhu, Z.; Munroe, P.; Deng, K.; Han, J. The Effect of Zn/Ca ratio on the microstructure, texture and mechanical properties of dilute Mg–Zn–Ca–Mn alloys that exhibit superior strength. J. Mater. Sci. 2020, 55, 3588–3604. [Google Scholar] [CrossRef]
- Kammer, C. Magnesium-Taschenbuch; Aluminium-Verlag: Düsseldorf, Germany, 2000; ISBN 978-3-87017-264-0. [Google Scholar]
- Avedesian, M.M.; Baker, H. ASM Specialty Handbook: Magnesium and Magnesium Alloys; ASM International: Almere, The Netherlands, 1999; ISBN 978-0-87170-657-7. [Google Scholar]
- Lee, Y.C.; Dahle, A.K.; StJohn, D.H. Grain refinement of magnesium. In Essential Readings in Magnesium Technology; Mathaudhu, S.N., Luo, A.A., Neelameggham, N.R., Nyberg, E.A., Sillekens, W.H., Eds.; Springer International Publishing: Basel, Switzerland, 2016; pp. 247–254. ISBN 978-3-319-48099-2. [Google Scholar]
- Zuo, Y.B.; Fu, X.; Mou, D.; Zhu, Q.F.; Li, L.; Cui, J.Z. Study on the role of Ca in the grain refinement of Mg–Ca binary alloys. Mater. Res. Innov. 2015, 19, S1-94–S1-97. [Google Scholar] [CrossRef]
- Yuan, W.; Panigrahi, S.K.; Su, J.-Q.; Mishra, R.S. Influence of grain size and texture on hall—Petch relationship for a magnesium alloy. Scr. Mater. 2011, 65, 994–997. [Google Scholar] [CrossRef]
- Khudododova, G.D.; Kulyasova, O.B.; Islamgaliev, R.K.; Valiev, R.Z. Microstructure and mechanical properties of the Mg–Zn–Ca biodegradable alloy after severe plastic deformation. IOP Conf. Ser. Mater. Sci. Eng. 2019, 672, 012030. [Google Scholar] [CrossRef]
- You, B.-S.; Park, W.-W.; Chung, I.-S. Effect of calcium additions on the oxidation behavior in magnesium alloys. Scr. Mater. 2000, 42, 1089–1094. [Google Scholar] [CrossRef]
- Ha, S.-H.; Lee, J.-K.; Jo, H.-H.; Jung, S.-B.; KIM Shae, K. Behavior of CaO and calcium in pure magnesium. Rare Met. 2006, 25, 150–154. [Google Scholar] [CrossRef]
- Baek, S.-M.; Kim, H.J.; Jeong, H.Y.; Sohn, S.-D.; Shin, H.-J.; Choi, K.-J.; Lee, K.-S.; Lee, J.G.; Yim, C.D.; You, B.S.; et al. Effect of alloyed ca on the microstructure and corrosion properties of extruded AZ61 Mg alloy. Corros. Sci. 2016, 112, 44–53. [Google Scholar] [CrossRef]
- Yim, C.D.; Kim, Y.M.; You, B.S. Effect of Ca addition on the corrosion resistance of gravity cast AZ31 magnesium alloy. Mater. Trans. 2007, 48, 1023–1028. [Google Scholar] [CrossRef] [Green Version]
- Deng, M.; Wang, L.; Höche, D.; Lamaka, S.V.; Wang, C.; Snihirova, D.; Jin, Y.; Zhang, Y.; Zheludkevich, M.L. Approaching “stainless magnesium” by Ca micro-alloying. Mater. Horiz. 2021, 8, 589–596. [Google Scholar] [CrossRef]
- Luo, A.A.; Powell, B.R.; Balogh, M.P. Creep and microstructure of magnesium-aluminum-calcium based alloys. Metall. Mater. Trans. A 2002, 33, 567–574. [Google Scholar] [CrossRef]
- Li, Z.T.; Zhang, X.D.; Zheng, M.Y.; Qiao, X.G.; Wu, K.; Xu, C.; Kamado, S. Effect of Ca/Al ratio on microstructure and mechanical properties of Mg-Al-Ca-Mn alloys. Mater. Sci. Eng. A 2017, 682, 423–432. [Google Scholar] [CrossRef]
- Jayaraj, J.; Mendis, C.L.; Ohkubo, T.; Oh-ishi, K.; Hono, K. Enhanced precipitation hardening of Mg–Ca alloy by Al addition. Scr. Mater. 2010, 63, 831–834. [Google Scholar] [CrossRef]
- Esmaily, M.; Svensson, J.E.; Fajardo, S.; Birbilis, N.; Frankel, G.S.; Virtanen, S.; Arrabal, R.; Thomas, S.; Johansson, L.G. Fundamentals and advances in magnesium alloy corrosion. Prog. Mater. Sci. 2017, 89, 92–193. [Google Scholar] [CrossRef]
- Cihova, M.; Schäublin, R.; Hauser, L.B.; Gerstl, S.S.A.; Simson, C.; Uggowitzer, P.J.; Löffler, J.F. Rational design of a lean magnesium-based alloy with high age-hardening response. Acta Mater. 2018, 158, 214–229. [Google Scholar] [CrossRef]
- Nakata, T.; Xu, C.; Ajima, R.; Shimizu, K.; Hanaki, S.; Sasaki, T.T.; Ma, L.; Hono, K.; Kamado, S. Strong and ductile age-hardening Mg-Al-Ca-Mn alloy that can be extruded as fast as aluminum alloys. Acta Mater. 2017, 130, 261–270. [Google Scholar] [CrossRef]
- Hono, K.; Mendis, C.L.; Sasaki, T.T.; Oh-ishi, K. Towards the development of heat-treatable high-strength wrought Mg alloys. Scr. Mater. 2010, 63, 710–715. [Google Scholar] [CrossRef]
- Pan, H.; Qin, G.; Huang, Y.; Ren, Y.; Sha, X.; Han, X.; Liu, Z.-Q.; Li, C.; Wu, X.; Chen, H.; et al. Development of low-alloyed and rare-earth-free magnesium alloys having ultra-high strength. Acta Mater. 2018, 149, 350–363. [Google Scholar] [CrossRef] [Green Version]
- Xu, S.W.; Oh-ishi, K.; Kamado, S.; Uchida, F.; Homma, T.; Hono, K. High-strength extruded Mg–Al–Ca–Mn alloy. Scr. Mater. 2011, 65, 269–272. [Google Scholar] [CrossRef]
- Pan, H.; Kang, R.; Li, J.; Xie, H.; Zeng, Z.; Huang, Q.; Yang, C.; Ren, Y.; Qin, G. Mechanistic investigation of a low-alloy Mg–Ca-based extrusion alloy with high strength–ductility synergy. Acta Mater. 2020, 186, 278–290. [Google Scholar] [CrossRef]
- Zeng, Z.R.; Zhu, Y.M.; Nie, J.F.; Xu, S.W.; Davies, C.H.J.; Birbilis, N. Effects of calcium on strength and microstructural evolution of extruded alloys based on Mg-3Al-1Zn-0.3Mn. Metall. Mater. Trans. A 2019, 50, 4344–4363. [Google Scholar] [CrossRef]
- Li, Z.T.; Qiao, X.G.; Xu, C.; Kamado, S.; Zheng, M.Y.; Luo, A.A. Ultrahigh strength Mg-Al-Ca-Mn extrusion alloys with various aluminum contents. J. Alloy. Compd. 2019, 792, 130–141. [Google Scholar] [CrossRef]
- Li, M.; Xie, D.; Li, J.; Xie, H.; Huang, Q.; Pan, H.; Qin, G. Realizing ultra-fine grains and ultra-high strength in conventionally extruded Mg-Ca-Al-Zn-Mn alloys: The multiple roles of nano-precipitations. Mater. Charact. 2021, 175, 111049. [Google Scholar] [CrossRef]
- Cross, A.J.; Prior, D.J.; Stipp, M.; Kidder, S. The recrystallized grain size piezometer for quartz: An EBSD-based calibration. Geophys. Res. Lett. 2017, 44, 6667–6674. [Google Scholar] [CrossRef]
- Liang, S.M.; Chen, R.S.; Blandin, J.J.; Suery, M.; Han, E.H. Thermal analysis and solidification pathways of Mg–Al–Ca system alloys. Mater. Sci. Eng. A 2008, 480, 365–372. [Google Scholar] [CrossRef]
- Suzuki, A.; Saddock, N.D.; Jones, J.W.; Pollock, T.M. Solidification paths and eutectic intermetallic phases in Mg–Al–Ca ternary alloys. Acta Mater. 2005, 53, 2823–2834. [Google Scholar] [CrossRef]
- Suzuki, A.; Saddock, N.D.; Jones, J.W.; Pollock, T.M. Structure and transition of eutectic (Mg,Al)2Ca laves phase in a die-cast Mg–Al–Ca base alloy. Scr. Mater. 2004, 51, 1005–1010. [Google Scholar] [CrossRef]
- Chen, P.; Li, B.; Culbertson, D.; Jiang, Y. Negligible effect of twin-slip interaction on hardening in deformation of a Mg-3Al-1Zn alloy. Mater. Sci. Eng. A 2018, 729, 285–293. [Google Scholar] [CrossRef]
- Jiang, M.G.; Xu, C.; Yan, H.; Fan, G.H.; Nakata, T.; Lao, C.S.; Chen, R.S.; Kamado, S.; Han, E.H.; Lu, B.H. Unveiling the formation of basal texture variations based on twinning and dynamic recrystallization in AZ31 magnesium alloy during extrusion. Acta Mater. 2018, 157, 53–71. [Google Scholar] [CrossRef]


| Alloy | Al [wt.%] | Ca [wt.%] | Mn [wt.%] | Mg | Ca/Al Ratio * [-] |
|---|---|---|---|---|---|
| AX01 | 0.50–0.70 | 0.55–0.70 | 0.20–0.30 | Balance | 1.04 |
| AX11 | 1.00–1.10 | 1.00–1.10 | 0.20–0.30 | Balance | 1.00 |
| AX22 | 2.20–2.40 | 2.10–2.40 | 0.35–0.45 | Balance | 0.98 |
| AX43 | 3.40–3.80 | 3.20–3.50 | 0.35–0.46 | Balance | 0.93 |
| AX55 | 4.50–5.00 | 4.40–5.00 | 0.35–0.47 | Balance | 0.99 |
| Condition | YS [MPa] | UTS [MPa] | εf [%] |
|---|---|---|---|
| As extruded | 314.5 ± 3.4 | 336.9 ± 3.0 | 10.0 ± 1.3 |
| 150 °C/9 h | 304.8 ± 4.5 | 326.2 ± 3.4 | 11.4 ± 0.8 |
| 250 °C/24 h | 288.9 ± 1.3 | 321.4 ± 0.7 | 9.7 ± 1.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gneiger, S.; Papenberg, N.P.; Arnoldt, A.R.; Schlögl, C.M.; Fehlbier, M. Investigations of High-Strength Mg–Al–Ca–Mn Alloys with a Broad Range of Ca+Al Contents. Materials 2021, 14, 5439. https://doi.org/10.3390/ma14185439
Gneiger S, Papenberg NP, Arnoldt AR, Schlögl CM, Fehlbier M. Investigations of High-Strength Mg–Al–Ca–Mn Alloys with a Broad Range of Ca+Al Contents. Materials. 2021; 14(18):5439. https://doi.org/10.3390/ma14185439
Chicago/Turabian StyleGneiger, Stefan, Nikolaus P. Papenberg, Aurel R. Arnoldt, Carina M. Schlögl, and Martin Fehlbier. 2021. "Investigations of High-Strength Mg–Al–Ca–Mn Alloys with a Broad Range of Ca+Al Contents" Materials 14, no. 18: 5439. https://doi.org/10.3390/ma14185439
APA StyleGneiger, S., Papenberg, N. P., Arnoldt, A. R., Schlögl, C. M., & Fehlbier, M. (2021). Investigations of High-Strength Mg–Al–Ca–Mn Alloys with a Broad Range of Ca+Al Contents. Materials, 14(18), 5439. https://doi.org/10.3390/ma14185439






