Bone Morphogenetic Proteins, Carriers, and Animal Models in the Development of Novel Bone Regenerative Therapies
Abstract
1. Introduction
2. Bone Regeneration by Bone Morphogenetic Protein Devices
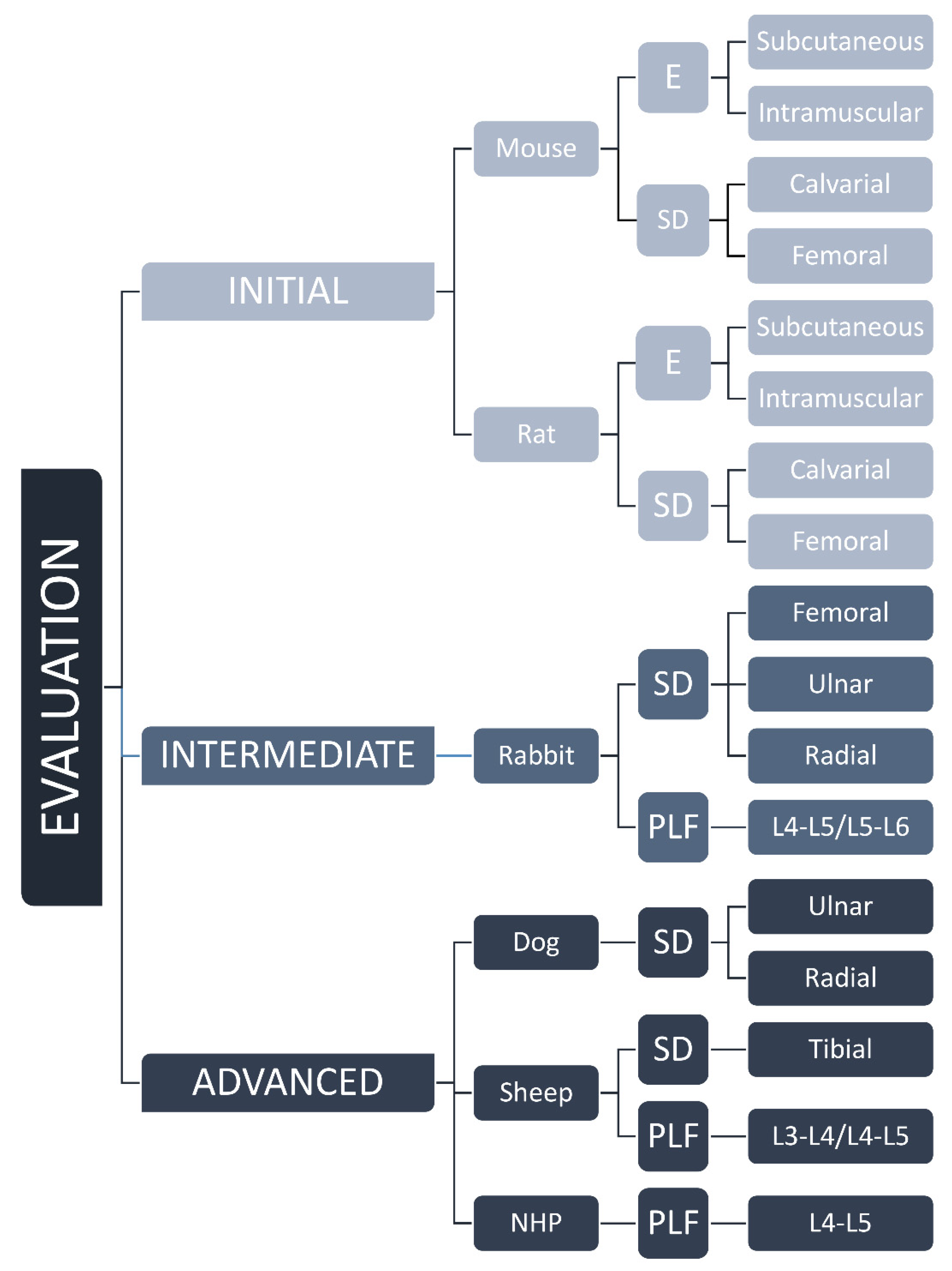
3. Animal Models
3.1. Initial Evaluation in Rodents
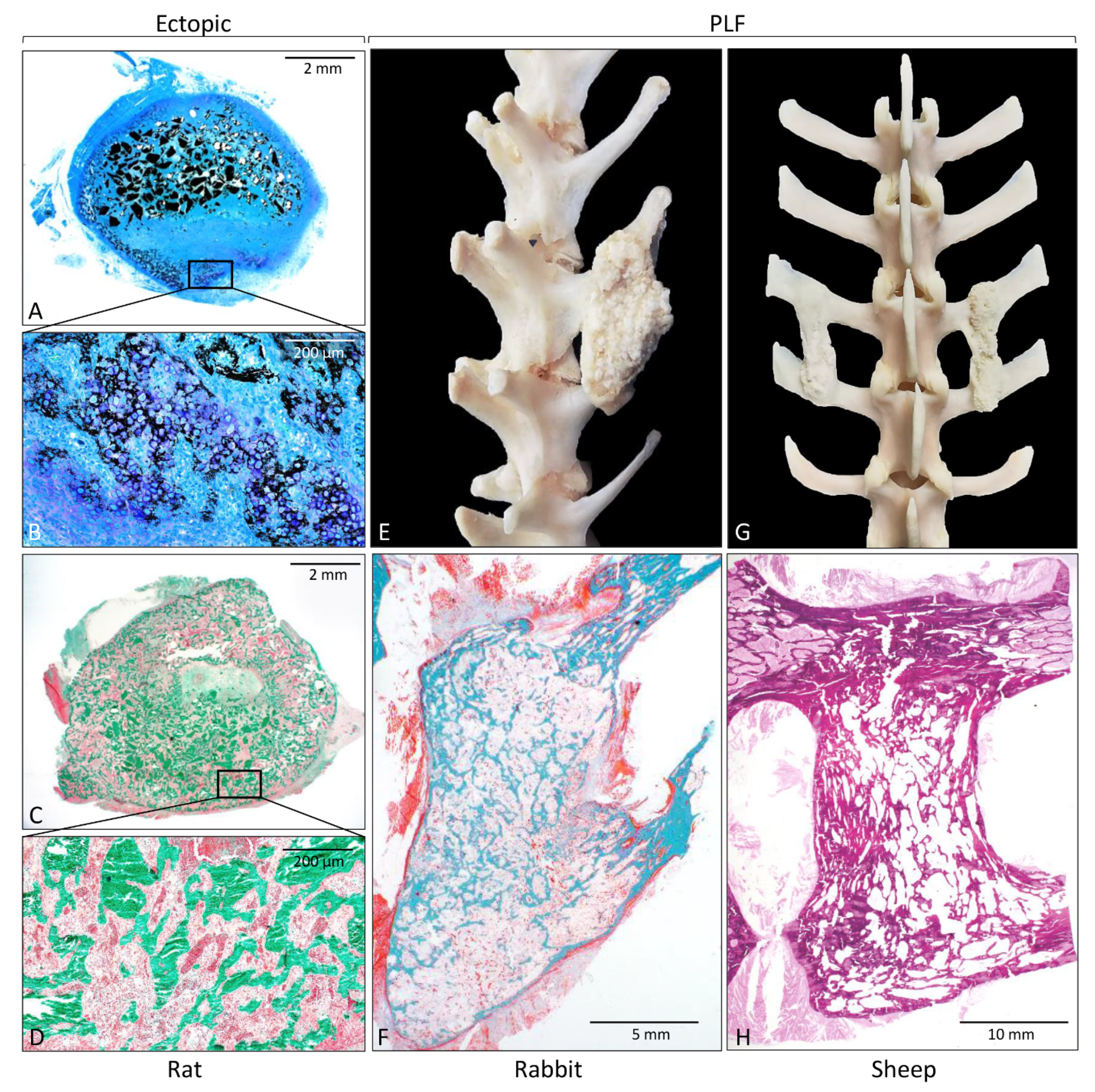
3.1.1. Ectopic Models
3.1.2. Bone Defect Models
3.2. Intermediate Evaluation in Rabbits
3.2.1. Segmental Defect Model
3.2.2. Posterolateral Spinal Fusion (PLF) Model
3.3. Advanced Evaluation of Bone Regeneration Therapies
3.3.1. Dog and Sheep Segmental Defect Model
3.3.2. Sheep PLF Model
3.3.3. Non-human Primate (NHP) PLF Model
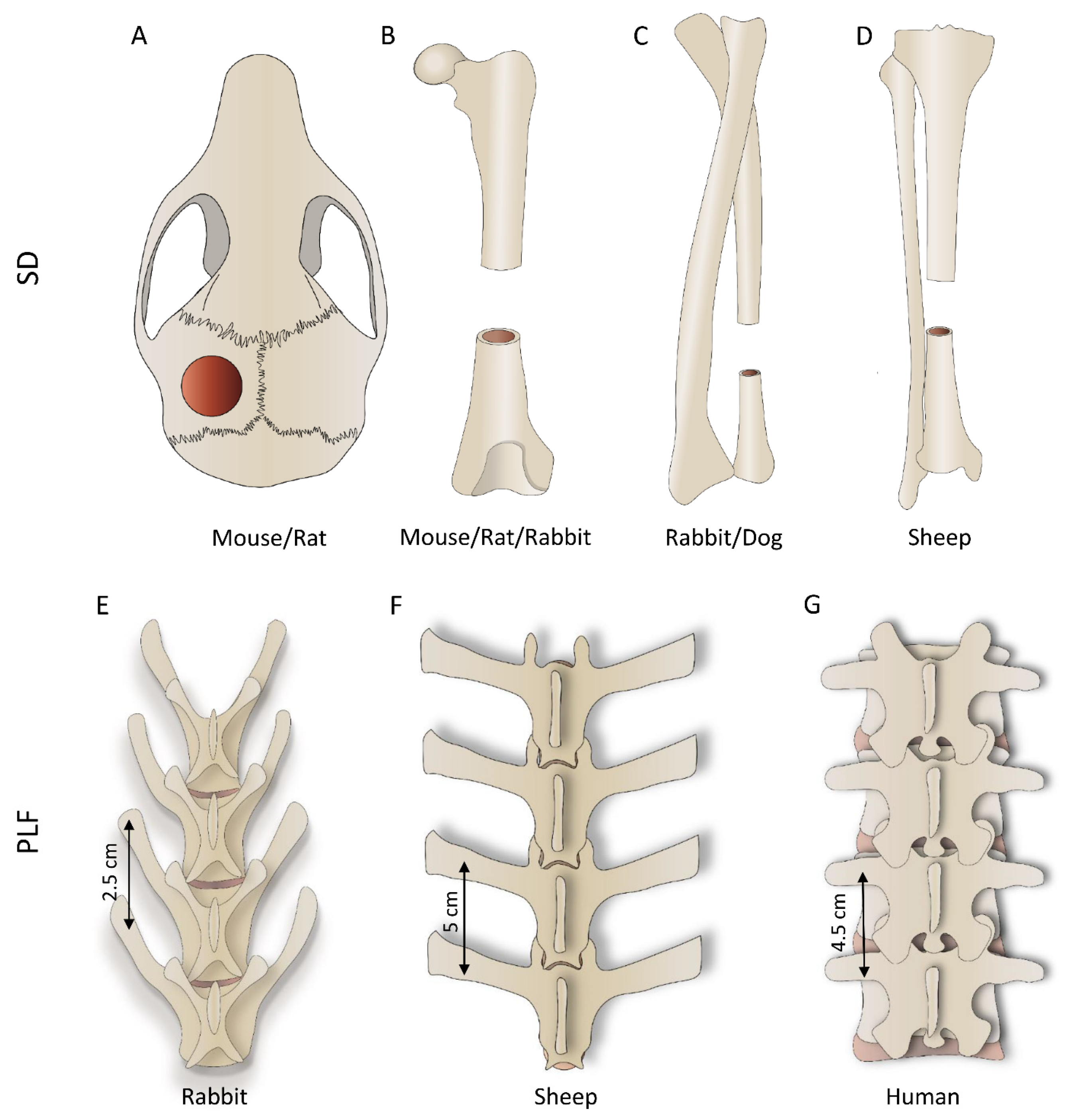
3.4. Anatomical Characteristics of the Species
3.4.1. Segmental Bone Defect
3.4.2. PLF
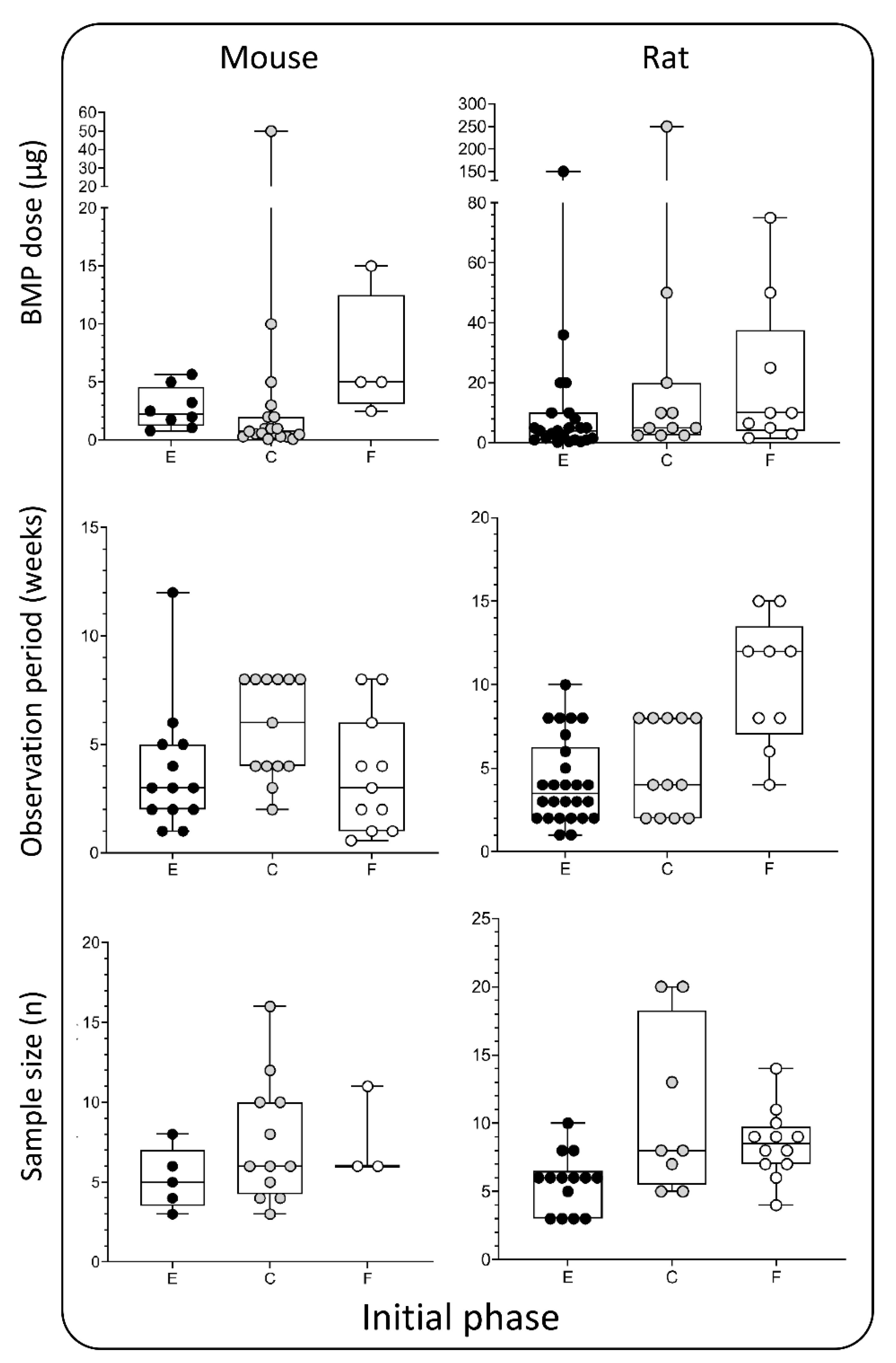
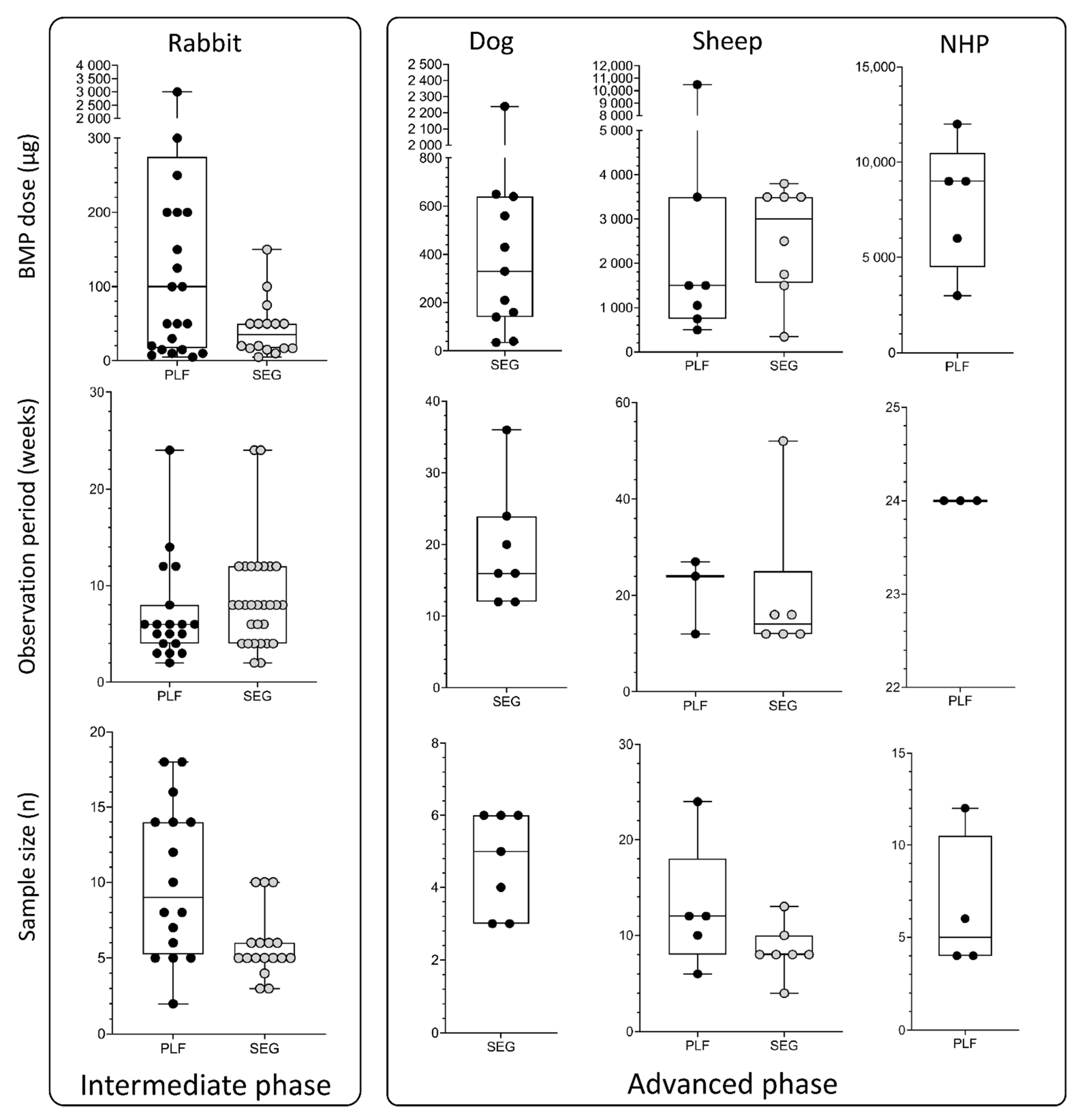
4. Appropriate Bone Morphogenetic Proteins Dosing
5. Duration of the Observation Period
6. Sample Size
7. Study Outcomes
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Dumic-Cule, I.; Pecina, M.; Jelic, M.; Jankolija, M.; Popek, I.; Grgurevic, L.; Vukicevic, S. Biological aspects of segmental bone defects management. Int. Orthop. 2015, 39, 1005–1011. [Google Scholar] [CrossRef] [PubMed]
- Lammens, J.; Marechal, M.; Delport, H.; Geris, L.; Oppermann, H.; Vukicevic, S.; Luyten, F.P. A cell-based combination product for the repair of large bone defects. Bone 2020, 138, 115511. [Google Scholar] [CrossRef] [PubMed]
- Toth, J.M.; Wang, M.; Lawson, J.; Badura, J.M.; DuBose, K.B. Radiographic, biomechanical, and histological evaluation of rhBMP-2 in a 3-level intertransverse process spine fusion: An ovine study. J. Neurosurg. Spine 2016, 25, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Dimar, J.R.; Glassman, S.D.; Burkus, K.J.; Carreon, L.Y. Clinical outcomes and fusion success at 2 years of single-level instrumented posterolateral fusions with recombinant human bone morphogenetic protein-2/compression resistant matrix versus iliac crest bone graft. Spine 2006, 31, 2534–2539. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Yu, C.H.; Yang, J.J.; Baek, H.R.; Lee, K.M.; Koo, T.Y.; Chang, B.S.; Lee, C.K. Comparative study of fusion rate induced by different dosages of Escherichia coli-derived recombinant human bone morphogenetic protein-2 using hydroxyapatite carrier. Spine J. 2012, 12, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Itoh, H.; Ebara, S.; Kamimura, M.; Tateiwa, Y.; Kinoshita, T.; Yuzawa, Y.; Takaoka, K. Experimental spinal fusion with use of recombinant human bone morphogenetic protein 2. Spine 1999, 24, 1402–1405. [Google Scholar] [CrossRef]
- Glassman, S.D.; Dimar, J.R.; Carreon, L.Y.; Campbell, M.J.; Puno, R.M.; Johnson, J.R. Initial fusion rates with recombinant human bone morphogenetic protein-2/compression resistant matrix and a hydroxyapatite and tricalcium phosphate/collagen carrier in posterolateral spinal fusion. Spine 2005, 30, 1694–1698. [Google Scholar] [CrossRef]
- Peric, M.; Dumic-Cule, I.; Grcevic, D.; Matijasic, M.; Verbanac, D.; Paul, R.; Grgurevic, L.; Trkulja, V.; Bagi, C.M.; Vukicevic, S. The rational use of animal models in the evaluation of novel bone regenerative therapies. Bone 2015, 70, 73–86. [Google Scholar] [CrossRef]
- McGovern, J.A.; Griffin, M.; Hutmacher, D.W. Animal models for bone tissue engineering and modelling disease. Dis. Model. Mech. 2018, 11. [Google Scholar] [CrossRef]
- Sampath, T.K.; Vukicevic, S. Biology of bone morphogenetic protein in bone repair and regeneration: A role for autologous blood coagulum as carrier. Bone 2020, 115602. [Google Scholar] [CrossRef]
- Carragee, E.J.; Hurwitz, E.L.; Weiner, B.K. A critical review of recombinant human bone morphogenetic protein-2 trials in spinal surgery: Emerging safety concerns and lessons learned. Spine J. 2011, 11, 471–491. [Google Scholar] [CrossRef]
- James, A.W.; LaChaud, G.; Shen, J.; Asatrian, G.; Nguyen, V.; Zhang, X.; Ting, K.; Soo, C. A review of the clinical side effects of bone morphogenetic protein-2. Tissue Eng. Part B Rev. 2016, 22, 284–297. [Google Scholar] [CrossRef]
- Govender, S.; Csimma, C.; Genant, H.K.; Valentin-Opran, A.; Amit, Y.; Arbel, R.; Aro, H.; Atar, D.; Bishay, M.; Borner, M.G.; et al. Recombinant human bone morphogenetic protein-2 for treatment of open tibial fractures: A prospective, controlled, randomized study of four hundred and fifty patients. J. Bone Jt. Surg. Am. 2002, 84, 2123–2134. [Google Scholar] [CrossRef] [PubMed]
- Boden, S.D.; Zdeblick, T.A.; Sandhu, H.S.; Heim, S.E. The use of rhBMP-2 in interbody fusion cages. Definitive evidence of osteoinduction in humans: A preliminary report. Spine 2000, 25, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Cai, X.; Huang, J.; Xu, F.; Liu, X.; Wang, Q. Recombinant human BMP-2 for the treatment of open tibial fractures. Orthopedics 2012, 35, e847–e854. [Google Scholar] [CrossRef]
- Ong, K.L.; Villarraga, M.L.; Lau, E.; Carreon, L.Y.; Kurtz, S.M.; Glassman, S.D. Off-label use of bone morphogenetic proteins in the United States using administrative data. Spine 2010, 35, 1794–1800. [Google Scholar] [CrossRef]
- Vincentelli, A.F.; Szadkowski, M.; Vardon, D.; Litrico, S.; Fuentes, S.; Steib, J.P.; le Huec, J.C.; Huppert, J.; Dubois, G.; Lenoir, T.; et al. rhBMP-2 (Recombinant Human Bone Morphogenetic Protein-2) in real world spine surgery. A phase IV, National, multicentre, retrospective study collecting data from patient medical files in French spinal centres. Orthop. Traumatol. Surg. Res. 2019, 105, 1157–1163. [Google Scholar] [CrossRef] [PubMed]
- Tannoury, C.A.; An, H.S. Complications with the use of bone morphogenetic protein 2 (BMP-2) in spine surgery. Spine J. 2014, 14, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Fu, R.; Selph, S.; McDonagh, M.; Peterson, K.; Tiwari, A.; Chou, R.; Helfand, M. Effectiveness and harms of recombinant human bone morphogenetic protein-2 in spine fusion: A systematic review and meta-analysis. Ann. Intern. Med. 2013, 158, 890–902. [Google Scholar] [CrossRef]
- Simmonds, M.C.; Brown, J.V.; Heirs, M.K.; Higgins, J.P.; Mannion, R.J.; Rodgers, M.A.; Stewart, L.A. Safety and effectiveness of recombinant human bone morphogenetic protein-2 for spinal fusion: A meta-analysis of individual-participant data. Ann. Intern. Med. 2013, 158, 877–889. [Google Scholar] [CrossRef] [PubMed]
- Hiremath, G.K.; Steinmetz, M.P.; Krishnaney, A.A. Is it safe to use recombinant human bone morphogenetic protein in posterior cervical fusion? Spine 2009, 34, 885–889. [Google Scholar] [CrossRef]
- Vukicevic, S.; Oppermann, H.; Verbanac, D.; Jankolija, M.; Popek, I.; Curak, J.; Brkljacic, J.; Pauk, M.; Erjavec, I.; Francetic, I.; et al. The clinical use of bone morphogenetic proteins revisited: A novel biocompatible carrier device OSTEOGROW for bone healing. Int. Orthop. 2014, 38, 635–647. [Google Scholar] [CrossRef] [PubMed]
- Vukicevic, S.; Grgurevic, L. BMP-6 and mesenchymal stem cell differentiation. Cytokine Growth Factor Rev. 2009, 20, 441–448. [Google Scholar] [CrossRef]
- Song, K.; Krause, C.; Shi, S.; Patterson, M.; Suto, R.; Grgurevic, L.; Vukicevic, S.; van Dinther, M.; Falb, D.; Ten Dijke, P.; et al. Identification of a key residue mediating bone morphogenetic protein (BMP)-6 resistance to noggin inhibition allows for engineered BMPs with superior agonist activity. J. Biol. Chem. 2010, 285, 12169–12180. [Google Scholar] [CrossRef]
- El Bialy, I.; Jiskoot, W.; Nejadnik, M.R. Formulation, delivery and stability of bone morphogenetic proteins for effective bone regeneration. Pharm. Res. 2017, 34, 1152–1170. [Google Scholar] [CrossRef]
- Seeherman, H.; Wozney, J.M. Delivery of bone morphogenetic proteins for orthopedic tissue regeneration. Cytokine Growth Factor Rev. 2005, 16, 329–345. [Google Scholar] [CrossRef]
- Haidar, Z.S.; Hamdy, R.C.; Tabrizian, M. Delivery of recombinant bone morphogenetic proteins for bone regeneration and repair. Part B: Delivery systems for BMPs in orthopaedic and craniofacial tissue engineering. Biotechnol. Lett. 2009, 31, 1825–1835. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, V.; Sinha, M. A review on carrier systems for bone morphogenetic protein-2. J. Biomed. Mater. Res. B Appl. Biomater. 2017, 105, 904–925. [Google Scholar] [CrossRef]
- Jung, U.W.; Choi, S.Y.; Pang, E.K.; Kim, C.S.; Choi, S.H.; Cho, K.S. The effect of varying the particle size of beta tricalcium phosphate carrier of recombinant human bone morphogenetic protein-4 on bone formation in rat calvarial defects. J. Periodontol. 2006, 77, 765–772. [Google Scholar] [CrossRef]
- Lee, S.H.; Shin, H. Matrices and scaffolds for delivery of bioactive molecules in bone and cartilage tissue engineering. Adv. Drug Deliv. Rev. 2007, 59, 339–359. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Kaito, T.; Furuya, M.; Seno, S.; Okuzaki, D.; Kikuta, J.; Tsukazaki, H.; Matsuda, H.; Yoshikawa, H.; Ishii, M. In vivo dynamic analysis of BMP-2-induced ectopic bone formation. Sci. Rep. 2020, 10, 4751. [Google Scholar] [CrossRef]
- Kim, C.S.; Kim, J.I.; Kim, J.; Choi, S.H.; Chai, J.K.; Kim, C.K.; Cho, K.S. Ectopic bone formation associated with recombinant human bone morphogenetic proteins-2 using absorbable collagen sponge and beta tricalcium phosphate as carriers. Biomaterials 2005, 26, 2501–2507. [Google Scholar] [CrossRef]
- Johnson, M.R.; Boerckel, J.D.; Dupont, K.M.; Guldberg, R.E. Functional restoration of critically sized segmental defects with bone morphogenetic protein-2 and heparin treatment. Clin. Orthop. Relat. Res. 2011, 469, 3111–3117. [Google Scholar] [CrossRef][Green Version]
- Williams, J.C.; Maitra, S.; Anderson, M.J.; Christiansen, B.A.; Reddi, A.H.; Lee, M.A. BMP-7 and bone regeneration: Evaluation of dose-response in a rodent segmental defect model. J. Orthop. Trauma 2015, 29, e336–e341. [Google Scholar] [CrossRef]
- Krishnan, L.; Priddy, L.B.; Esancy, C.; Li, M.T.; Stevens, H.Y.; Jiang, X.; Tran, L.; Rowe, D.W.; Guldberg, R.E. Hydrogel-based Delivery of rhBMP-2 Improves Healing of Large Bone Defects Compared with Autograft. Clin. Orthop. Relat. Res. 2015, 473, 2885–2897. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Wang, J.; Cao, L.; Qian, X.; Xing, W.; Lu, J.; Liu, C. Segmental bone regeneration using rhBMP-2-loaded collagen/chitosan microspheres composite scaffold in a rabbit model. Biomed. Mater. 2012, 7, 035002. [Google Scholar] [CrossRef] [PubMed]
- Jenis, L.G.; Wheeler, D.; Parazin, S.J.; Connolly, R.J. The effect of osteogenic protein-1 in instrumented and noninstrumented posterolateral fusion in rabbits. Spine J. 2002, 2, 173–178. [Google Scholar] [CrossRef]
- Minamide, A.; Kawakami, M.; Hashizume, H.; Sakata, R.; Yoshida, M.; Tamaki, T. Experimental study of carriers of bone morphogenetic protein used for spinal fusion. J. Orthop. Sci. 2004, 9, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Namikawa, T.; Terai, H.; Hoshino, M.; Miyamoto, S.; Takaoka, K. Ectopic bone formation in mice associated with a lactic acid/dioxanone/ethylene glycol copolymer-tricalcium phosphate composite with added recombinant human bone morphogenetic protein-2. Biomaterials 2006, 27, 3927–3933. [Google Scholar] [CrossRef]
- Lee, J.W.; Lee, S.; Lee, S.H.; Yang, H.S.; Im, G.I.; Kim, C.S.; Park, J.H.; Kim, B.S. Improved spinal fusion efficacy by long-term delivery of bone morphogenetic protein-2 in a rabbit model. Acta Orthop. 2011, 82, 756–760. [Google Scholar] [CrossRef]
- Kang, S.W.; Bae, J.H.; Park, S.A.; Kim, W.D.; Park, M.S.; Ko, Y.J.; Jang, H.S.; Park, J.H. Combination therapy with BMP-2 and BMSCs enhances bone healing efficacy of PCL scaffold fabricated using the 3D plotting system in a large segmental defect model. Biotechnol. Lett. 2012, 34, 1375–1384. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Jiang, P.; Xue, S.; Wang, T.; Li, H.; Wang, J. Repair of a critical-size segmental rabbit femur defect using bioglass-beta-TCP monoblock, a vascularized periosteal flap and BMP-2. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 2148–2156. [Google Scholar] [CrossRef] [PubMed]
- Paralkar, V.M.; Borovecki, F.; Ke, H.Z.; Cameron, K.O.; Lefker, B.; Grasser, W.A.; Owen, T.A.; Li, M.; DaSilva-Jardine, P.; Zhou, M.; et al. An EP2 receptor-selective prostaglandin E2 agonist induces bone healing. Proc. Natl. Acad. Sci. USA 2003, 100, 6736–6740. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Zamparini, F.; Esposti, M.D.; Chiellini, F.; Fava, F.; Fabbri, P.; Taddei, P.; Prati, C. Highly porous polycaprolactone scaffolds doped with calcium silicate and dicalcium phosphate dihydrate designed for bone regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 102, 341–361. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, M.G.; Zamparini, F.; Esposti, M.D.; Chiellini, F.; Aparicio, C.; Fava, F.; Fabbri, P.; Taddei, P.; Prati, C. Polylactic acid-based porous scaffolds doped with calcium silicate and dicalcium phosphate dihydrate designed for biomedical application. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 82, 163–181. [Google Scholar] [CrossRef]
- Qin, Y.; Sun, R.; Wu, C.; Wang, L.; Zhang, C. Exosome: A novel approach to stimulate bone regeneration through regulation of osteogenesis and angiogenesis. Int. J. Mol. Sci. 2016, 17, 712. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Gardin, C.; Zamparini, F.; Ferroni, L.; Esposti, M.D.; Parchi, G.; Ercan, B.; Manzoli, L.; Fava, F.; Fabbri, P.; et al. Mineral-doped poly(L-lactide) acid scaffolds enriched with exosomes improve osteogenic commitment of human adipose-derived mesenchymal stem cells. Nanomaterials 2020, 10, 432. [Google Scholar] [CrossRef]
- Roldan, J.C.; Detsch, R.; Schaefer, S.; Chang, E.; Kelantan, M.; Waiss, W.; Reichert, T.E.; Gurtner, G.C.; Deisinger, U. Bone formation and degradation of a highly porous biphasic calcium phosphate ceramic in presence of BMP-7, VEGF and mesenchymal stem cells in an ectopic mouse model. J. Craniomaxillofac. Surg. 2010, 38, 423–430. [Google Scholar] [CrossRef]
- Liang, G.; Yang, Y.; Oh, S.; Ong, J.L.; Zheng, C.; Ran, J.; Yin, G.; Zhou, D. Ectopic osteoinduction and early degradation of recombinant human bone morphogenetic protein-2-loaded porous beta-tricalcium phosphate in mice. Biomaterials 2005, 26, 4265–4271. [Google Scholar] [CrossRef]
- Kuboki, Y.; Takita, H.; Kobayashi, D.; Tsuruga, E.; Inoue, M.; Murata, M.; Nagai, N.; Dohi, Y.; Ohgushi, H. BMP-induced osteogenesis on the surface of hydroxyapatite with geometrically feasible and nonfeasible structures: Topology of osteogenesis. J. Biomed. Mater. Res. 1998, 39, 190–199. [Google Scholar] [CrossRef]
- Tsuruga, E.; Takita, H.; Itoh, H.; Wakisaka, Y.; Kuboki, Y. Pore size of porous hydroxyapatite as the cell-substratum controls BMP-induced osteogenesis. J. Biochem. 1997, 121, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.I.; Asahina, I.; Ohmamiuda, K.; Takahashi, K.; Yokota, S.; Enomoto, S. Evaluation of ceramics composed of different hydroxyapatite to tricalcium phosphate ratios as carriers for rhBMP-2. Biomaterials 2001, 22, 1643–1651. [Google Scholar] [CrossRef]
- Vehof, J.W.; Takita, H.; Kuboki, Y.; Spauwen, P.H.; Jansen, J.A. Histological characterization of the early stages of bone morphogenetic protein-induced osteogenesis. J. Biomed. Mater. Res. 2002, 61, 440–449. [Google Scholar] [CrossRef]
- Tazaki, J.; Murata, M.; Akazawa, T.; Yamamoto, M.; Ito, K.; Arisue, M.; Shibata, T.; Tabata, Y. BMP-2 release and dose-response studies in hydroxyapatite and beta-tricalcium phosphate. Biomed. Mater. Eng. 2009, 19, 141–146. [Google Scholar] [PubMed]
- Park, J.C.; So, S.S.; Jung, I.H.; Yun, J.H.; Choi, S.H.; Cho, K.S.; Kim, C.S. Induction of bone formation by Escherichia coli-expressed recombinant human bone morphogenetic protein-2 using block-type macroporous biphasic calcium phosphate in orthotopic and ectopic rat models. J. Periodontal Res. 2011, 46, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Strobel, L.A.; Rath, S.N.; Maier, A.K.; Beier, J.P.; Arkudas, A.; Greil, P.; Horch, R.E.; Kneser, U. Induction of bone formation in biphasic calcium phosphate scaffolds by bone morphogenetic protein-2 and primary osteoblasts. J. Tissue Eng. Regen. Med. 2014, 8, 176–185. [Google Scholar] [CrossRef]
- Ma, J.; Yang, F.; Both, S.K.; Prins, H.J.; Helder, M.N.; Pan, J.; Cui, F.Z.; Jansen, J.A.; van den Beucken, J.J. Bone forming capacity of cell- and growth factor-based constructs at different ectopic implantation sites. J. Biomed. Mater. Res. A 2015, 103, 439–450. [Google Scholar] [CrossRef]
- Kim, J.W.; Choi, K.H.; Yun, J.H.; Jung, U.W.; Kim, C.S.; Choi, S.H.; Cho, K.S. Bone formation of block and particulated biphasic calcium phosphate lyophilized with Escherichia coli-derived recombinant human bone morphogenetic protein 2 in rat calvarial defects. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2011, 112, 298–306. [Google Scholar] [CrossRef]
- Jang, J.W.; Yun, J.H.; Lee, K.I.; Jang, J.W.; Jung, U.W.; Kim, C.S.; Choi, S.H.; Cho, K.S. Osteoinductive activity of biphasic calcium phosphate with different rhBMP-2 doses in rats. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 113, 480–487. [Google Scholar] [CrossRef]
- Lee, J.H.; Ryu, M.Y.; Baek, H.R.; Lee, K.M.; Seo, J.H.; Lee, H.K.; Ryu, H.S. Effects of porous beta-tricalcium phosphate-based ceramics used as an E. coli-derived rhBMP-2 carrier for bone regeneration. J. Mater. Sci. Mater. Med. 2013, 24, 2117–2127. [Google Scholar] [CrossRef]
- Kuroiwa, Y.; Niikura, T.; Lee, S.Y.; Oe, K.; Iwakura, T.; Fukui, T.; Matsumoto, T.; Matsushita, T.; Nishida, K.; Kuroda, R. Escherichia coli-derived BMP-2-absorbed beta-TCP granules induce bone regeneration in rabbit critical-sized femoral segmental defects. Int. Orthop. 2019, 43, 1247–1253. [Google Scholar] [CrossRef]
- Louis-Ugbo, J.; Kim, H.S.; Boden, S.D.; Mayr, M.T.; Li, R.C.; Seeherman, H.; D’Augusta, D.; Blake, C.; Jiao, A.; Peckham, S. Retention of 125I-labeled recombinant human bone morphogenetic protein-2 by biphasic calcium phosphate or a composite sponge in a rabbit posterolateral spine arthrodesis model. J. Orthop. Res. 2002, 20, 1050–1059. [Google Scholar] [CrossRef]
- Suh, D.Y.; Boden, S.D.; Louis-Ugbo, J.; Mayr, M.; Murakami, H.; Kim, H.S.; Minamide, A.; Hutton, W.C. Delivery of recombinant human bone morphogenetic protein-2 using a compression-resistant matrix in posterolateral spine fusion in the rabbit and in the non-human primate. Spine 2002, 27, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Dohzono, S.; Imai, Y.; Nakamura, H.; Wakitani, S.; Takaoka, K. Successful spinal fusion by E. coli-derived BMP-2-adsorbed porous beta-TCP granules: A pilot study. Clin. Orthop. Relat. Res. 2009, 467, 3206–3212. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, M.H.; Oliver, R.A.; Christou, C.; Yu, Y.; Bertollo, N.; Irie, H.; Walsh, W.R. Lumbar spinal fusion with beta-TCP granules and variable Escherichia coli-derived rhBMP-2 dose. Spine J. 2014, 14, 1758–1768. [Google Scholar] [CrossRef] [PubMed]
- Notodihardjo, F.Z.; Kakudo, N.; Kushida, S.; Suzuki, K.; Kusumoto, K. Bone regeneration with BMP-2 and hydroxyapatite in critical-size calvarial defects in rats. J. Craniomaxillofac. Surg. 2012, 40, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.C.; Robu, I.S.; Patel, R.; Leu, M.C.; Velez, M.; Chu, T.M. The effects of 3D bioactive glass scaffolds and BMP-2 on bone formation in rat femoral critical size defects and adjacent bones. Biomed. Mater. 2014, 9, 045013. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Wang, D.; Zhang, X.; Lu, W.; Han, Y.; Ou, Y.; Zhou, K.; Fen, W.; Liu, J.; Peng, L.; et al. Experimental study of nano-hydroxyapatite/recombinant human bone morphogenetic protein-2 composite artificial bone. Artif. Cells Blood Substit. Immobil. Biotechnol. 2010, 38, 150–156. [Google Scholar] [CrossRef]
- Huang, T.Y.; Wu, C.C.; Weng, P.W.; Chen, J.M.; Yeh, W.L. Effect of ErhBMP-2-loaded beta-tricalcium phosphate on ulna defects in the osteoporosis rabbit model. Bone Rep. 2021, 14, 100739. [Google Scholar] [CrossRef]
- Wu, Y.; Hou, J.; Yin, M.; Wang, J.; Liu, C. Enhanced healing of rabbit segmental radius defects with surface-coated calcium phosphate cement/bone morphogenetic protein-2 scaffolds. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 44, 326–335. [Google Scholar] [CrossRef]
- Tazaki, J.; Akazawa, T.; Murata, M.; Yamamoto, M.; Tabata, Y.; Yoshimoto, R.; Arisue, M. BMP-2 Dose-response and release studies in functionally graded HAp. Key Eng. Mater. 2006, 309–311, 965–968. [Google Scholar] [CrossRef]
- Vukicevic, S.; Stokovic, N.; Pecina, M. Is ceramics an appropriate bone morphogenetic protein delivery system for clinical use? Int. Orthop. 2019, 43, 1275–1276. [Google Scholar] [CrossRef]
- Stokovic, N.; Ivanjko, N.; Erjavec, I.; Milosevic, M.; Oppermann, H.; Shimp, L.; Sampath, K.T.; Vukicevic, S. Autologous bone graft substitute containing rhBMP6 within autologous blood coagulum and synthetic ceramics of different particle size determines the quantity and structural pattern of bone formed in a rat sucutaneous assay. Bone 2020, 115654. [Google Scholar] [CrossRef]
- Stokovic, N.; Ivanjko, N.; Pecin, M.; Erjavec, I.; Karlovic, S.; Smajlovic, A.; Capak, H.; Milosevic, M.; Bubic Spoljar, J.; Vnuk, D.; et al. Evaluation of synthetic ceramics as compression resistant matrix to promote osteogenesis of autologous blood coagulum containing recombinant human bone morphogenetic protein 6 in rabbit posterolateral lumbar fusion model. Bone 2020, 140, 115544. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Bioceramics of calcium orthophosphates. Biomaterials 2010, 31, 1465–1485. [Google Scholar] [CrossRef]
- Stokovic, N.; Ivanjko, N.; Milesevic, M.; Matic Jelic, I.; Bakic, K.; Rumenovic, V.; Oppermann, H.; Shimp, L.; Sampath, T.K.; Pecina, M.; et al. Synthetic ceramic macroporous blocks as a scaffold in ectopic bone formation induced by recombinant human bone morphogenetic protein 6 within autologous blood coagulum in rats. Int. Orthop. 2020, 45, 1097–1107. [Google Scholar] [CrossRef] [PubMed]
- Bae, E.B.; Park, K.H.; Shim, J.H.; Chung, H.Y.; Choi, J.W.; Lee, J.J.; Kim, C.H.; Jeon, H.J.; Kang, S.S.; Huh, J.B. Efficacy of rhBMP-2 Loaded PCL/beta-TCP/bdECM Scaffold Fabricated by 3D Printing Technology on Bone Regeneration. Biomed. Res. Int. 2018, 2018, 2876135. [Google Scholar] [CrossRef]
- Chu, T.M.; Warden, S.J.; Turner, C.H.; Stewart, R.L. Segmental bone regeneration using a load-bearing biodegradable carrier of bone morphogenetic protein-2. Biomaterials 2007, 28, 459–467. [Google Scholar] [CrossRef]
- Rodriguez-Evora, M.; Delgado, A.; Reyes, R.; Hernandez-Daranas, A.; Soriano, I.; San Roman, J.; Evora, C. Osteogenic effect of local, long versus short term BMP-2 delivery from a novel SPU-PLGA-betaTCP concentric system in a critical size defect in rats. Eur. J. Pharm. Sci. 2013, 49, 873–884. [Google Scholar] [CrossRef]
- Yoneda, M.; Terai, H.; Imai, Y.; Okada, T.; Nozaki, K.; Inoue, H.; Miyamoto, S.; Takaoka, K. Repair of an intercalated long bone defect with a synthetic biodegradable bone-inducing implant. Biomaterials 2005, 26, 5145–5152. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lu, Y.; Tian, X.; Cui, G.; Zhao, Y.; Yang, Q.; Yu, S.; Xing, G.; Zhang, B. Segmental bone regeneration using an rhBMP-2-loaded gelatin/nanohydroxyapatite/fibrin scaffold in a rabbit model. Biomaterials 2009, 30, 6276–6285. [Google Scholar] [CrossRef] [PubMed]
- Luca, L.; Rougemont, A.L.; Walpoth, B.H.; Boure, L.; Tami, A.; Anderson, J.M.; Jordan, O.; Gurny, R. Injectable rhBMP-2-loaded chitosan hydrogel composite: Osteoinduction at ectopic site and in segmental long bone defect. J. Biomed. Mater. Res. A 2011, 96, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Fujita, N.; Matsushita, T.; Ishida, K.; Sasaki, K.; Kubo, S.; Matsumoto, T.; Kurosaka, M.; Tabata, Y.; Kuroda, R. An analysis of bone regeneration at a segmental bone defect by controlled release of bone morphogenetic protein 2 from a biodegradable sponge composed of gelatin and beta-tricalcium phosphate. J. Tissue Eng. Regen. Med. 2012, 6, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Hokugo, A.; Takahashi, Y.; Nakano, T.; Hiraoka, M.; Tabata, Y. Combination of BMP-2-releasing gelatin/beta-TCP sponges with autologous bone marrow for bone regeneration of X-ray-irradiated rabbit ulnar defects. Biomaterials 2015, 56, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Namikawa, T.; Terai, H.; Suzuki, E.; Hoshino, M.; Toyoda, H.; Nakamura, H.; Miyamoto, S.; Takahashi, N.; Ninomiya, T.; Takaoka, K. Experimental spinal fusion with recombinant human bone morphogenetic protein-2 delivered by a synthetic polymer and beta-tricalcium phosphate in a rabbit model. Spine 2005, 30, 1717–1722. [Google Scholar] [CrossRef]
- Vukicevic, S.; Grgurevic, L.; Erjavec, I.; Pecin, M.; Bordukalo-Niksic, T.; Stokovic, N.; Lipar, M.; Capak, H.; Maticic, D.; Windhager, R.; et al. Autologous blood coagulum is a physiological carrier for BMP6 to induce new bone formation and promote posterolateral lumbar spine fusion in rabbits. J. Tissue Eng. Regen. Med. 2020, 14, 147–159. [Google Scholar] [CrossRef]
- Grgurevic, L.; Oppermann, H.; Pecin, M.; Erjavec, I.; Capak, H.; Pauk, M.; Karlovic, S.; Kufner, V.; Lipar, M.; Bubic Spoljar, J.; et al. Recombinant human bone morphogenetic protein 6 delivered within autologous blood coagulum restores critical size segmental defects of ulna in rabbits. JBMR Plus 2019, 3, e10085. [Google Scholar] [CrossRef]
- Grgurevic, L.; Erjavec, I.; Gupta, M.; Pecin, M.; Bordukalo-Niksic, T.; Stokovic, N.; Vnuk, D.; Farkas, V.; Capak, H.; Milosevic, M.; et al. Autologous blood coagulum containing rhBMP6 induces new bone formation to promote anterior lumbar interbody fusion (ALIF) and posterolateral lumbar fusion (PLF) of spine in sheep. Bone 2020. [Google Scholar] [CrossRef]
- Vukicevic, S.; Peric, M.; Oppermann, H.; Stokovic, N.; Ivanjko, N.; Erjavec, I.; Kufner, V.; Vnuk, D.; Bubic Spoljar, J.; Pecin, M.; et al. Bone morphogenetic proteins: From discovery to development of a novel autologous bone graft substitute consisting of recombinant human BMP6 delivered in autologous blood coagulum carrier. Rad CASA Med. Sci. 2020, 544, 26–41. [Google Scholar]
- Pecin, M.; Stokovic, N.; Ivanjko, N.; Smajlovic, A.; Kreszinger, M.; Capak, H.; Vrbanac, Z.; Oppermann, H.; Maticic, D.; Vukicevic, S. A novel autologous bone graft substitute containing rhBMP6 in autologous blood coagulum with synthetic ceramics for reconstruction of a large humerus segmental gunshot defect in a dog: The first veterinary patient to receive a novel osteoinductive therapy. Bone Rep. 2021, 14, 100759. [Google Scholar] [CrossRef]
- Chiari, C.; Grgurevic, L.; Bordukalo-Niksic, T.; Oppermann, H.; Valentinitsch, A.; Nemecek, E.; Staats, K.; Schreiner, M.; Trost, C.; Kolb, A.; et al. Recombinant human BMP6 applied within autologous blood coagulum accelerates bone healing: Randomized controlled trial in high tibial osteotomy patients. J. Bone Miner. Res. 2020, 35, 1893–1903. [Google Scholar] [CrossRef]
- Durdevic, D.; Vlahovic, T.; Pehar, S.; Miklic, D.; Oppermann, H.; Bordukalo-Niksic, T.; Gavrankapetanovic, I.; Jamakosmanovic, M.; Milosevic, M.; Martinovic, S.; et al. A novel autologous bone graft substitute comprised of rhBMP6 blood coagulum as carrier tested in a randomized and controlled Phase I trial in patients with distal radial fractures. Bone 2020, 140, 115551. [Google Scholar] [CrossRef]
- Ji, W.; Kerckhofs, G.; Geeroms, C.; Marechal, M.; Geris, L.; Luyten, F.P. Deciphering the combined effect of bone morphogenetic protein 6 and calcium phosphate on bone formation capacity of periosteum derived cells-based tissue engineering constructs. Acta Biomater. 2018, 80, 97–107. [Google Scholar] [CrossRef]
- Eyckmans, J.; Roberts, S.J.; Schrooten, J.; Luyten, F.P. A clinically relevant model of osteoinduction: A process requiring calcium phosphate and BMP/Wnt signalling. J. Cell. Mol. Med. 2010, 14, 1845–1856. [Google Scholar] [CrossRef] [PubMed]
- Eyckmans, J.; Roberts, S.J.; Bolander, J.; Schrooten, J.; Chen, C.S.; Luyten, F.P. Mapping calcium phosphate activated gene networks as a strategy for targeted osteoinduction of human progenitors. Biomaterials 2013, 34, 4612–4621. [Google Scholar] [CrossRef] [PubMed]
- Bolander, J.; Chai, Y.C.; Geris, L.; Schrooten, J.; Lambrechts, D.; Roberts, S.J.; Luyten, F.P. Early BMP, Wnt and Ca(2+)/PKC pathway activation predicts the bone forming capacity of periosteal cells in combination with calcium phosphates. Biomaterials 2016, 86, 106–118. [Google Scholar] [CrossRef]
- Bolander, J.; Ji, W.; Geris, L.; Bloemen, V.; Chai, Y.C.; Schrooten, J.; Luyten, F.P. The combined mechanism of bone morphogenetic protein- and calcium phosphate-induced skeletal tissue formation by human periosteum derived cells. Eur. Cell Mater. 2016, 31, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Katagiri, H.; Mendes, L.F.; Luyten, F.P. Reduction of BMP6-induced bone formation by calcium phosphate in wild-type compared with nude mice. J. Tissue Eng. Regen. Med. 2019, 13, 846–856. [Google Scholar] [CrossRef]
- Chai, Y.C.; Roberts, S.J.; Desmet, E.; Kerckhofs, G.; van Gastel, N.; Geris, L.; Carmeliet, G.; Schrooten, J.; Luyten, F.P. Mechanisms of ectopic bone formation by human osteoprogenitor cells on CaP biomaterial carriers. Biomaterials 2012, 33, 3127–3142. [Google Scholar] [CrossRef]
- Mumcuoglu, D.; Fahmy-Garcia, S.; Ridwan, Y.; Nicke, J.; Farrell, E.; Kluijtmans, S.G.; van Osch, G.J. Injectable BMP-2 delivery system based on collagen-derived microspheres and alginate induced bone formation in a time- and dose-dependent manner. Eur. Cell Mater. 2018, 35, 242–254. [Google Scholar] [CrossRef]
- Lin, X.; Hunziker, E.B.; Liu, T.; Hu, Q.; Liu, Y. Enhanced biocompatibility and improved osteogenesis of coralline hydroxyapatite modified by bone morphogenetic protein 2 incorporated into a biomimetic coating. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 96, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Reves, B.T.; Jennings, J.A.; Bumgardner, J.D.; Haggard, W.O. Osteoinductivity Assessment of BMP-2 Loaded Composite Chitosan-Nano-Hydroxyapatite Scaffolds in a Rat Muscle Pouch. Materials 2011, 4, 1360–1374. [Google Scholar] [CrossRef]
- Bhakta, G.; Rai, B.; Lim, Z.X.; Hui, J.H.; Stein, G.S.; van Wijnen, A.J.; Nurcombe, V.; Prestwich, G.D.; Cool, S.M. Hyaluronic acid-based hydrogels functionalized with heparin that support controlled release of bioactive BMP-2. Biomaterials 2012, 33, 6113–6122. [Google Scholar] [CrossRef]
- Kisiel, M.; Martino, M.M.; Ventura, M.; Hubbell, J.A.; Hilborn, J.; Ossipov, D.A. Improving the osteogenic potential of BMP-2 with hyaluronic acid hydrogel modified with integrin-specific fibronectin fragment. Biomaterials 2013, 34, 704–712. [Google Scholar] [CrossRef]
- Luca, L.; Rougemont, A.L.; Walpoth, B.H.; Gurny, R.; Jordan, O. The effects of carrier nature and pH on rhBMP-2-induced ectopic bone formation. J. Control. Release 2010, 147, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Sommer, N.G.; Hahn, D.; Okutan, B.; Marek, R.; Weinberg, A.M. Animal models in orthopedic research: The proper animal model to answer fundamental questions on bone healing depending on pathology and implant material. In Animal Models in Medicine and Biology; Tvrdá, E., Yenisetti, C.S., Eds.; IntechOpen: London, UK, 2020. [Google Scholar]
- Simpson, A.H.; Murray, I.R. Osteoporotic fracture models. Curr. Osteoporos Rep. 2015, 13, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Jacenko, O.; Olsen, B.R. Transgenic mouse models in studies of skeletal disorders. J. Rheumatol. Suppl. 1995, 43, 39–41. [Google Scholar]
- Fan, J.; Im, C.S.; Cui, Z.K.; Guo, M.; Bezouglaia, O.; Fartash, A.; Lee, J.Y.; Nguyen, J.; Wu, B.M.; Aghaloo, T.; et al. Delivery of phenamil enhances BMP-2-induced osteogenic differentiation of adipose-derived stem cells and bone formation in calvarial defects. Tissue Eng. Part A 2015, 21, 2053–2065. [Google Scholar] [CrossRef]
- Gronowicz, G.; Jacobs, E.; Peng, T.; Zhu, L.; Hurley, M.; Kuhn, L.T. Calvarial bone regeneration is enhanced by sequential delivery of FGF-2 and BMP-2 from layer-by-layer coatings with a biomimetic calcium phosphate barrier layer. Tissue Eng. Part A 2017, 23, 1490–1501. [Google Scholar] [CrossRef] [PubMed]
- Herberg, S.; Aguilar-Perez, A.; Howie, R.N.; Kondrikova, G.; Periyasamy-Thandavan, S.; Elsalanty, M.E.; Shi, X.; Hill, W.D.; Cray, J.J. Mesenchymal stem cell expression of SDF-1beta synergizes with BMP-2 to augment cell-mediated healing of critical-sized mouse calvarial defects. J. Tissue Eng. Regen. Med. 2017, 11, 1806–1819. [Google Scholar] [CrossRef]
- Huang, K.C.; Yano, F.; Murahashi, Y.; Takano, S.; Kitaura, Y.; Chang, S.H.; Soma, K.; Ueng, S.W.N.; Tanaka, S.; Ishihara, K.; et al. Sandwich-type PLLA-nanosheets loaded with BMP-2 induce bone regeneration in critical-sized mouse calvarial defects. Acta Biomater. 2017, 59, 12–20. [Google Scholar] [CrossRef]
- La, W.G.; Kwon, S.H.; Lee, T.J.; Yang, H.S.; Park, J.; Kim, B.S. The effect of the delivery carrier on the quality of bone formed via bone morphogenetic protein-2. Artif. Organs 2012, 36, 642–647. [Google Scholar] [CrossRef]
- Maisani, M.; Sindhu, K.R.; Fenelon, M.; Siadous, R.; Rey, S.; Mantovani, D.; Chassande, O. Prolonged delivery of BMP-2 by a non-polymer hydrogel for bone defect regeneration. Drug Deliv. Transl. Res. 2018, 8, 178–190. [Google Scholar] [CrossRef]
- Reyes, R.; Rodriguez, J.A.; Orbe, J.; Arnau, M.R.; Evora, C.; Delgado, A. Combined sustained release of BMP2 and MMP10 accelerates bone formation and mineralization of calvaria critical size defect in mice. Drug Deliv. 2018, 25, 750–756. [Google Scholar] [CrossRef]
- Seo, B.B.; Koh, J.T.; Song, S.C. Tuning physical properties and BMP-2 release rates of injectable hydrogel systems for an optimal bone regeneration effect. Biomaterials 2017, 122, 91–104. [Google Scholar] [CrossRef]
- Terauchi, M.; Inada, T.; Kanemaru, T.; Ikeda, G.; Tonegawa, A.; Nishida, K.; Arisaka, Y.; Tamura, A.; Yamaguchi, S.; Yui, N. Potentiating bioactivity of BMP-2 by polyelectrolyte complexation with sulfonated polyrotaxanes to induce rapid bone regeneration in a mouse calvarial defect. J. Biomed. Mater. Res. A 2017, 105, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.S.; La, W.G.; Park, J.; Kim, C.S.; Im, G.I.; Kim, B.S. Efficient bone regeneration induced by bone morphogenetic protein-2 released from apatite-coated collagen scaffolds. J. Biomater. Sci. Polym. Ed. 2012, 23, 1659–1671. [Google Scholar] [CrossRef] [PubMed]
- Alaee, F.; Hong, S.H.; Dukas, A.G.; Pensak, M.J.; Rowe, D.W.; Lieberman, J.R. Evaluation of osteogenic cell differentiation in response to bone morphogenetic protein or demineralized bone matrix in a critical sized defect model using GFP reporter mice. J. Orthop. Res. 2014, 32, 1120–1128. [Google Scholar] [CrossRef]
- Bougioukli, S.; Jain, A.; Sugiyama, O.; Tinsley, B.A.; Tang, A.H.; Tan, M.H.; Adams, D.J.; Kostenuik, P.J.; Lieberman, J.R. Combination therapy with BMP-2 and a systemic RANKL inhibitor enhances bone healing in a mouse critical-sized femoral defect. Bone 2016, 84, 93–103. [Google Scholar] [CrossRef]
- Zwingenberger, S.; Langanke, R.; Vater, C.; Lee, G.; Niederlohmann, E.; Sensenschmidt, M.; Jacobi, A.; Bernhardt, R.; Muders, M.; Rammelt, S.; et al. The effect of SDF-1alpha on low dose BMP-2 mediated bone regeneration by release from heparinized mineralized collagen type I matrix scaffolds in a murine critical size bone defect model. J. Biomed. Mater. Res. A 2016, 104, 2126–2134. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.; Histing, T.; Holstein, J.H.; Klein, M.; Laschke, M.W.; Matthys, R.; Ignatius, A.; Wildemann, B.; Lienau, J.; Peters, A.; et al. Rodent animal models of delayed bone healing and non-union formation: A comprehensive review. Eur. Cell Mater. 2013, 26, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Diab, T.; Pritchard, E.M.; Uhrig, B.A.; Boerckel, J.D.; Kaplan, D.L.; Guldberg, R.E. A silk hydrogel-based delivery system of bone morphogenetic protein for the treatment of large bone defects. J. Mech. Behav. Biomed. Mater. 2012, 11, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Peng, K.T.; Hsieh, M.Y.; Lin, C.T.; Chen, C.F.; Lee, M.S.; Huang, Y.Y.; Chang, P.J. Treatment of critically sized femoral defects with recombinant BMP-2 delivered by a modified mPEG-PLGA biodegradable thermosensitive hydrogel. BMC Musculoskelet. Disord. 2016, 17, 286. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.H.; Song, H.R.; Kim, H.J.; Lim, H.C.; Park, J.H.; Liu, Y.; Teoh, S.H. Discontinuous release of bone morphogenetic protein-2 loaded within interconnected pores of honeycomb-like polycaprolactone scaffold promotes bone healing in a large bone defect of rabbit ulna. Tissue Eng. Part A 2011, 17, 2389–2397. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.J.; Kang, S.H.; Kwon, H.J.; Cho, S.W.; Kim, H.J. Bone healing properties of autoclaved autogenous bone grafts incorporating recombinant human bone morphogenetic protein-2 and comparison of two delivery systems in a segmental rabbit radius defect. Maxillofac Plast. Reconstr. Surg. 2014, 36, 94–102. [Google Scholar] [CrossRef]
- Boden, S.D.; Schimandle, J.H.; Hutton, W.C. 1995 Volvo Award in basic sciences. The use of an osteoinductive growth factor for lumbar spinal fusion. Part II: Study of dose, carrier, and species. Spine 1995, 20, 2633–2644. [Google Scholar] [CrossRef]
- Valdes, M.; Moore, D.C.; Palumbo, M.; Lucas, P.R.; Robertson, A.; Appel, J.; Ehrlich, M.G.; Keeping, H.S. rhBMP-6 stimulated osteoprogenitor cells enhance posterolateral spinal fusion in the New Zealand white rabbit. Spine J. 2007, 7, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Konishi, S.; Nakamura, H.; Seki, M.; Nagayama, R.; Yamano, Y. Hydroxyapatite granule graft combined with recombinant human bone morphogenic protein-2 for solid lumbar fusion. J. Spinal Disord. Tech. 2002, 15, 237–244. [Google Scholar] [CrossRef]
- Itoh, T.; Mochizuki, M.; Nishimura, R.; Matsunaga, S.; Kadosawa, T.; Kokubo, S.; Yokota, S.; Sasaki, N. Repair of ulnar segmental defect by recombinant human bone morphogenetic protein-2 in dogs. J. Vet. Med. Sci. 1998, 60, 451–458. [Google Scholar] [CrossRef][Green Version]
- Tuominen, T.; Jamsa, T.; Tuukkanen, J.; Nieminen, P.; Lindholm, T.C.; Lindholm, T.S.; Jalovaara, P. Native bovine bone morphogenetic protein improves the potential of biocoral to heal segmental canine ulnar defects. Int. Orthop. 2000, 24, 289–294. [Google Scholar] [CrossRef][Green Version]
- Hu, Y.; Zhang, C.; Zhang, S.; Xiong, Z.; Xu, J. Development of a porous poly(L-lactic acid)/hydroxyapatite/collagen scaffold as a BMP delivery system and its use in healing canine segmental bone defect. J. Biomed. Mater. Res. A 2003, 67, 591–598. [Google Scholar] [CrossRef]
- Jones, C.B.; Sabatino, C.T.; Badura, J.M.; Sietsema, D.L.; Marotta, J.S. Improved healing efficacy in canine ulnar segmental defects with increasing recombinant human bone morphogenetic protein-2/allograft ratios. J. Orthop. Trauma 2008, 22, 550–559. [Google Scholar] [CrossRef]
- Harada, Y.; Itoi, T.; Wakitani, S.; Irie, H.; Sakamoto, M.; Zhao, D.; Nezu, Y.; Yogo, T.; Hara, Y.; Tagawa, M. Effect of Escherichia coli-produced recombinant human bone morphogenetic protein 2 on the regeneration of canine segmental ulnar defects. J. Bone Miner. Metab. 2012, 30, 388–399. [Google Scholar] [CrossRef]
- Minier, K.; Toure, A.; Fusellier, M.; Fellah, B.; Bouvy, B.; Weiss, P.; Gauthier, O. BMP-2 delivered from a self-crosslinkable CaP/hydrogel construct promotes bone regeneration in a critical-size segmental defect model of non-union in dogs. Vet. Comp. Orthop. Traumatol. 2014, 27, 411–421. [Google Scholar] [PubMed]
- Lammens, J.; Marechal, M.; Delport, H.; Geris, L.; Luyten, F.P. A flowchart for the translational research of cell-based therapy in the treatment of long bone defects. J. Regen. Med. 2021, 10, 1. [Google Scholar]
- Cipitria, A.; Reichert, J.C.; Epari, D.R.; Saifzadeh, S.; Berner, A.; Schell, H.; Mehta, M.; Schuetz, M.A.; Duda, G.N.; Hutmacher, D.W. Polycaprolactone scaffold and reduced rhBMP-7 dose for the regeneration of critical-sized defects in sheep tibiae. Biomaterials 2013, 34, 9960–9968. [Google Scholar] [CrossRef] [PubMed]
- den Boer, F.C.; Wippermann, B.W.; Blokhuis, T.J.; Patka, P.; Bakker, F.C.; Haarman, H.J. Healing of segmental bone defects with granular porous hydroxyapatite augmented with recombinant human osteogenic protein-1 or autologous bone marrow. J. Orthop. Res. 2003, 21, 521–528. [Google Scholar] [CrossRef]
- Pluhar, G.E.; Turner, A.S.; Pierce, A.R.; Toth, C.A.; Wheeler, D.L. A comparison of two biomaterial carriers for osteogenic protein-1 (BMP-7) in an ovine critical defect model. J. Bone Jt. Surg. Br. 2006, 88, 960–966. [Google Scholar] [CrossRef]
- Reichert, J.C.; Epari, D.R.; Wullschleger, M.E.; Berner, A.; Saifzadeh, S.; Noth, U.; Dickinson, I.C.; Schuetz, M.A.; Hutmacher, D.W. Bone tissue engineering. Reconstruction of critical sized segmental bone defects in the ovine tibia. Orthopade 2012, 41, 280–287. [Google Scholar] [CrossRef]
- Akamaru, T.; Suh, D.; Boden, S.D.; Kim, H.S.; Minamide, A.; Louis-Ugbo, J. Simple carrier matrix modifications can enhance delivery of recombinant human bone morphogenetic protein-2 for posterolateral spine fusion. Spine 2003, 28, 429–434. [Google Scholar] [CrossRef]
- Zwingenberger, S.; Yao, Z.; Jacobi, A.; Vater, C.; Valladares, R.D.; Li, C.; Nich, C.; Rao, A.J.; Christman, J.E.; Antonios, J.K.; et al. Enhancement of BMP-2 induced bone regeneration by SDF-1alpha mediated stem cell recruitment. Tissue Eng. Part A 2014, 20, 810–818. [Google Scholar] [PubMed]
- Yamamoto, M.; Takahashi, Y.; Tabata, Y. Enhanced bone regeneration at a segmental bone defect by controlled release of bone morphogenetic protein-2 from a biodegradable hydrogel. Tissue Eng. 2006, 12, 1305–1311. [Google Scholar] [CrossRef] [PubMed]
| Mouse Ectopic Model | |||||
|---|---|---|---|---|---|
| Author (Year) | Carrier | BMP Dose (µg) | Time (Weeks or Days) | Sample Size (n) | |
| Kato et al. (2006) | PLA-DX-PEG, PLA-DX-PEG/TCP, TCP | 2 and 5 | 3 and 6 weeks | 6 | |
| Roldan et al. (2010) | BCP | 12 weeks | 8 | ||
| Liang et al. (2014) | TCP | 50 | 3, 7, 14, 21, and 28 days | 5 | |
| Bolander et al. (2016) | CaP granules/Collagen | 1.06 and 1.77 | 5 weeks | 4 | |
| Ji et al. (2018) | CaP-based materials | 0.81, 3.24, and 5.67 | 2 and 5 weeks | 3 | |
| Hashimoto et al. (2020) | Collagen | 2.5 | 7, 10, 14, and 21 days | ||
| Mouse Calvarial Defect Model | |||||
| Author (Year) | Calvarial Defect Size (mm) | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| La et al. (2012) | 4 | TCP, Heparin—conjugated fibrin | 0.3 | 8 | 10 |
| Yang et al. (2012) | 4 | Collagen, Apatite—coated collagen | 0.5, 0.75, 1, 2, and 3 | 8 | 6 |
| Fan et al. (2015) | 3 | PLGA/Apatite layer | 0.3, 0.6, and 1 | 6 | 8, 12 |
| Gronowitz et al. (2017) | 3.5 | Collagen/HA | 2 | 3 | 4 |
| Herberg et al. (2017) | 5 | Acellular dermis | 0.542 | 4 | 10 |
| Huang et al. (2017) | 3.5 | PLA | 50 | 2, 4, 6, and 8 | 16 |
| Seo et al. (2017) | 5 | Poly(phosphazene) hydrogels Poly(phosphazene) hydrogels/BCP | 5 and 10 | 8 | 3 |
| Terauchi et al. (2017) | 3.5 | Sulphopropyl ether—modified polyrotaxanes/Collagen | 0.1 | 4 | 5, 6 |
| Maisani et al. (2018) | 3.5 | Hydrogel | 1 | 8 | 6 |
| Reyes et al. (2018) | 4 | PLGA | 0.1, 0.3, and 0.6 | 4 and 8 | 4 |
| Mouse Femoral Defect Model | |||||
| Author (Year) | Femoral Defect Size (mm) | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| Alaee et al. (2014) | 2 | Collagen | 5 | 4 days, 1, 2, 3, 4, and 8 | 6 |
| Bougrouli et al. (2016) | 2 | Collagen | 5 | 1, 2, 4, and 8 | 6 |
| Zwingenbergen et al. (2016) | 3 | Heparin/functionalized mineralized collagen matrix | 2.5 and 15 | 6 | 11 |
| Rat Ectopic Model | ||||
|---|---|---|---|---|
| Author (Year) | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| Kuboki et al. (1998) | HA | 1, 2, 3, and 4 | ||
| Tsuruga et al. (1998) | HA | 4 | 1, 2, 3, and 4 | 3 |
| Alam et al. (2000) | TCP, HA, BCP | 1.5 and 10 | 2 and 4 | 3 |
| Vehof et al. (2002) | HA | 8 | 3, 5, 7, and 9 | 3 |
| Kim Chang-Sung et al. (2004) | TCP, Collagen | 5 | 2 and 8 | 10 |
| Tazaki et al. (2006) | HA | 0.5, 1, and 5 | 3 | |
| Tazaki et al. (2008) | HA, TCP | 0.5, 1, and 5 | 3 | 3 |
| Luca et al. (2010) | Chitosan/Hyaluronan hydrogel | 150 | 3 | 3, 6 |
| Reves et al. (2011) | Chitosan-nano-HA | 36 | 4 | 6 |
| Park et al. (2011) | BCP | 2.5 | 2 and 8 | 5–8 |
| Bhakta et al. (2012) | Hyaluronan-based hydrogel | 5 | 8 | 6 |
| Strobel et al. (2012) | BCP | 1.6 | 2, 4, and 6 | 6 |
| Kisiel et al. (2013) | Hyaluronan hydrogel/Fibronectin fragments | 4 | 7 | 6 |
| Ma et al. (2014) | BCP | 20 | 8 | 6 |
| Mumcuoglu et al. (2018) | Collagen-based microspheres/Alginate | 0.3, 1, and 10 | 10 | 8 |
| Lin et al. (2019) | Coralline HA | 20 | 5 | 6 |
| Rat Calvarial Defect Model | ||||
| Author (Year) | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| Jung et al. (2006) | TCP | 2.5 | 2 and 8 | 20 |
| Kim et al. (2011) | BCP | 50 and 250 | 2 and 8 | 20 |
| Park et al. (2011) | BCP | 2.5 | 2 and 8 | 5–8 |
| Notodihardjo et al. (2011) | HA | 10 | 4 | 5 |
| Jang et al. (2012) | BCP | 2.5, 5, 10, and 20 | 2 and 8 | 8 |
| Lee JH et al. (2013) | TCP, HA, BCP | 5 | 4 and 8 | 13 |
| Bae et al. (2017) | PCL/TCP | 5 | 4 | 7 |
| Rat Femoral Defect Model | ||||
| Author (Year) | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| Chu et al. (2006) | Poly(propylene fumarate)/TCP/DCP | 10 | 6 and 15 | 4, 7 |
| Johnson et al. (2011) | Collagen, Collagen/Heparin, Heparin | 3 | 12 | 7, 9 |
| Diab et al. (2011) | PCL/Silk fibroin hydrogel | 5 | 12 | 10 |
| Lee et al. (2012) | BCP | 1000 | 4 and 8 | 6 |
| Rodriguez-Evora et al. (2013) | Segmented polyurethane/PLGA/ TCP ceramics | 1.6 and 6.5 | 12 | 9 |
| Wai-Ching et al. (2014) | Bioactive glass/DCP | 10 | 15 | 8, 9 |
| Williams et al. (2015) | Collagen | 25, 50, 75 and 100 | 8 | 8, 11 |
| Krishnan et al. (2015) | Nanofiber mesh alginate | 5 µg | 12 | 14 |
| Rabbit Segmental Defect Model | |||||
|---|---|---|---|---|---|
| Author (Year) | Model | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| Yoneda et al. (2004) | Femur (1.5 cm) | PLA-DX-PEG/TCP | 50 | 24 | 5 |
| Yamamoto et al. (2006) | Ulna (2 cm) | Gelatin hydrogel | 17 | 6 | 3 |
| Liu et al. (2009) | Radius (1.5 cm) | Gelatin/nanoHA/Fibrin | 100 | 4, 8, and 12 | 5 |
| Luca et al. (2010) | Radius (1.5 cm) | Chitosan hydrogel/TCP | 150 | 8 | 1 (pilot) |
| Zhu et al. (2010) | Radius | nanoHA | 4, 8, and 12 | 10 | |
| Bae et al. (2011) | Ulna (1.5 cm) | PCL/fibrin | 75 | 8 | 5 |
| Fujita et al. (2011) | Ulna (2 cm) | Gelatin/TCP | 17 | 4 and 8 | 6, 10 |
| Sun-Woong et al. (2012) | Ulna (2 cm) | PCL | 15 | 12 | 6 |
| Hou et al. (2012) | Radius (1.5 cm) | Collagen, Collagen/Chitosan | 50 | 2, 4, 8, and 12 | 3, 5 |
| Choi et al. (2014) | Radius (2 cm) | Collagen, Fibrin glue | 50 | 6 and 12 | 4 |
| Wu et al. (2014) | Radius (1.5 cm) | CaP cement, Hydroxypropylmethyl cellulose/CaP cement | 50 | 2, 4, 8, and 12 | 5 |
| Yamamoto et al. (2015) | Ulna (2 cm) | Gelatin/TCP | 17 | 6 | 6 |
| Peng et al. (2016) | Femur (1 cm) | PEG-PLGA hydrogel | 5, 10, and 20 | 12 | 6 |
| Pan et al. (2017) | Femur (2 cm) | Bioglass/TCP | 20 | 4 and 8 | 5 |
| Kuroiwa et al. (2018) | Femur (2 cm) | TCP | 50 | 12 and 24 | 10 |
| Grgurevic et al. (2019) | Ulna (1.5 cm) | Autologous blood coagulum | 25, 50, and 100 | 23 | 5 |
| Huang et al. (2021) | Ulna (2 cm) | TCP | 20 | 8 | 5 |
| Rabbit PLF Model | ||||
|---|---|---|---|---|
| Author (Year) | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| Boden et al. (1995) | DBM, Biocoral/ Collagen | 100 and 300 | 5 | 14–16 |
| Itoh et al. (1999) | Collagen | 10, 50, and 200 | 24 | 6 |
| Louis-Ugbo et al. (2001) | BCP, Collagen/BCP | 3000/mL | 5 | 18 |
| Jenis et al. (2002) | Collagen | - | 3 and 12 | 8 |
| Konishi et al. (2002) | Autograft/HA | 200 | 2, 4, and 6 | 2–7 |
| Suh et al. (2002) | Collagen/BCP, BCP | 860 | 5 | 14 |
| Minamide et al. (2003) | TCP cement, True bone ceramics, Collagen | 100 | 3 and 6 | 5–10 |
| Namikawa et al. (2005) | TCP/PLA-DX-PEG | 7.5, 15, and 30 | 6 | 5 |
| Valdes et al. (2007) | - | 6 | 18 | |
| Dohzono et al. (2009) | TCP | 5, 15, 50, and 150 | 4 and 8 | 5–8 |
| Lee JW et al. (2011) | Heparin—conjugated PLGA nanospheres, PLGA nanospheres | 20 | 12 | 12 |
| Lee JH et al. (2012) | HA | 10, 50, 200, and 500 | 3 and 6 | 14 |
| Vukicevic et al. (2019) | Autologous blood coagulum, Autologous blood coagulum/Allograft | 125, 250, 500, and 1000 | 14 | 4 |
| Dog Segmental Defect Model | |||||
|---|---|---|---|---|---|
| Author (Year) | Model | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| Itoh (1998) | Ulna (2 cm) | PLGA/Gelatin | 40, 160, and 640 | 16 | 4 |
| Tuominen (2000) | Ulna (2 cm) | Coral | - | 16 and 36 | 3, 6 |
| Hu (2003) | Radius (2 cm) | HA/Collagen/PLA | - | 24 | 6 |
| Jones (2008) | Ulna (2.5 cm) | Collagen/Allograft, Collagen/BCP ceramics | 210, 430, and 650 | 12 | 6 |
| Harada (2012) | Ulna (2.5 cm) | TCP | 35, 140, 560, and 2240 | 12 | 3 |
| Minier (2014) | Ulna (2 cm) | CaP/Hydrogel | 330 | 20 | 5 |
| Sheep Segmental Defect Model | |||||
| Author (Year) | Model | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| Den Boer et al. (2003) | Tibia (3 cm) | HA | 2500 | 12 | 8 |
| Pluhar et al. (2006) | Tibia (5 cm) | Carboxymethylcellulose/Bovine collagen, Collagen | 3500 | 16 | 10 |
| Reichert et al. (2012) | Tibia (3 cm) | mPCL-TCP | 3500 | 12 and 52 | 8 |
| Cipitria et al. (2013) | Tibia (3 cm) | mPCL-TCP | 1750 and 3500 | 12 | 8 |
| Lammens et al. (2020) | Tibia (3 and 4.5 cm) | CaP ceramics | 344, 1500, and 3800 | 16 | 4, 8, 13 |
| Sheep PLF Model | ||||
|---|---|---|---|---|
| Author (Year) | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| Pelletier et al. (2014) | TCP | 1050, 3500, and 10,500 | 12 | 12 |
| Toth et al. (2016) | Collagen/BCP, Collagen-ceramic sponge | 750 and 1500/cm3 | 24 | 12–24 |
| Grgurevic et al. (2020) | Autologous blood coagulum, Autologous blood coagulum/Allograft | 500 and 1500 | 27 | 6–10 |
| NHP PLF Model | ||||
| Author (Year) | Carrier | BMP Dose (µg) | Time (Weeks) | Sample Size (n) |
| Boden et al. (1999) | BCP | 6000, 9000, and 12,000 | 24 | 4–12 |
| Suh et al. (2002) | Ceramic/Collagen | 9000 | 24 | 4 |
| Akamaru et al. (2003) | Collagen/BCP Collagen/Allograft | 3000 | 24 | 6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stokovic, N.; Ivanjko, N.; Maticic, D.; Luyten, F.P.; Vukicevic, S. Bone Morphogenetic Proteins, Carriers, and Animal Models in the Development of Novel Bone Regenerative Therapies. Materials 2021, 14, 3513. https://doi.org/10.3390/ma14133513
Stokovic N, Ivanjko N, Maticic D, Luyten FP, Vukicevic S. Bone Morphogenetic Proteins, Carriers, and Animal Models in the Development of Novel Bone Regenerative Therapies. Materials. 2021; 14(13):3513. https://doi.org/10.3390/ma14133513
Chicago/Turabian StyleStokovic, Nikola, Natalia Ivanjko, Drazen Maticic, Frank P. Luyten, and Slobodan Vukicevic. 2021. "Bone Morphogenetic Proteins, Carriers, and Animal Models in the Development of Novel Bone Regenerative Therapies" Materials 14, no. 13: 3513. https://doi.org/10.3390/ma14133513
APA StyleStokovic, N., Ivanjko, N., Maticic, D., Luyten, F. P., & Vukicevic, S. (2021). Bone Morphogenetic Proteins, Carriers, and Animal Models in the Development of Novel Bone Regenerative Therapies. Materials, 14(13), 3513. https://doi.org/10.3390/ma14133513








