Novel Cold Crucible Ultrasonic Atomization Powder Production Method for 3D Printing
Abstract
1. Introduction
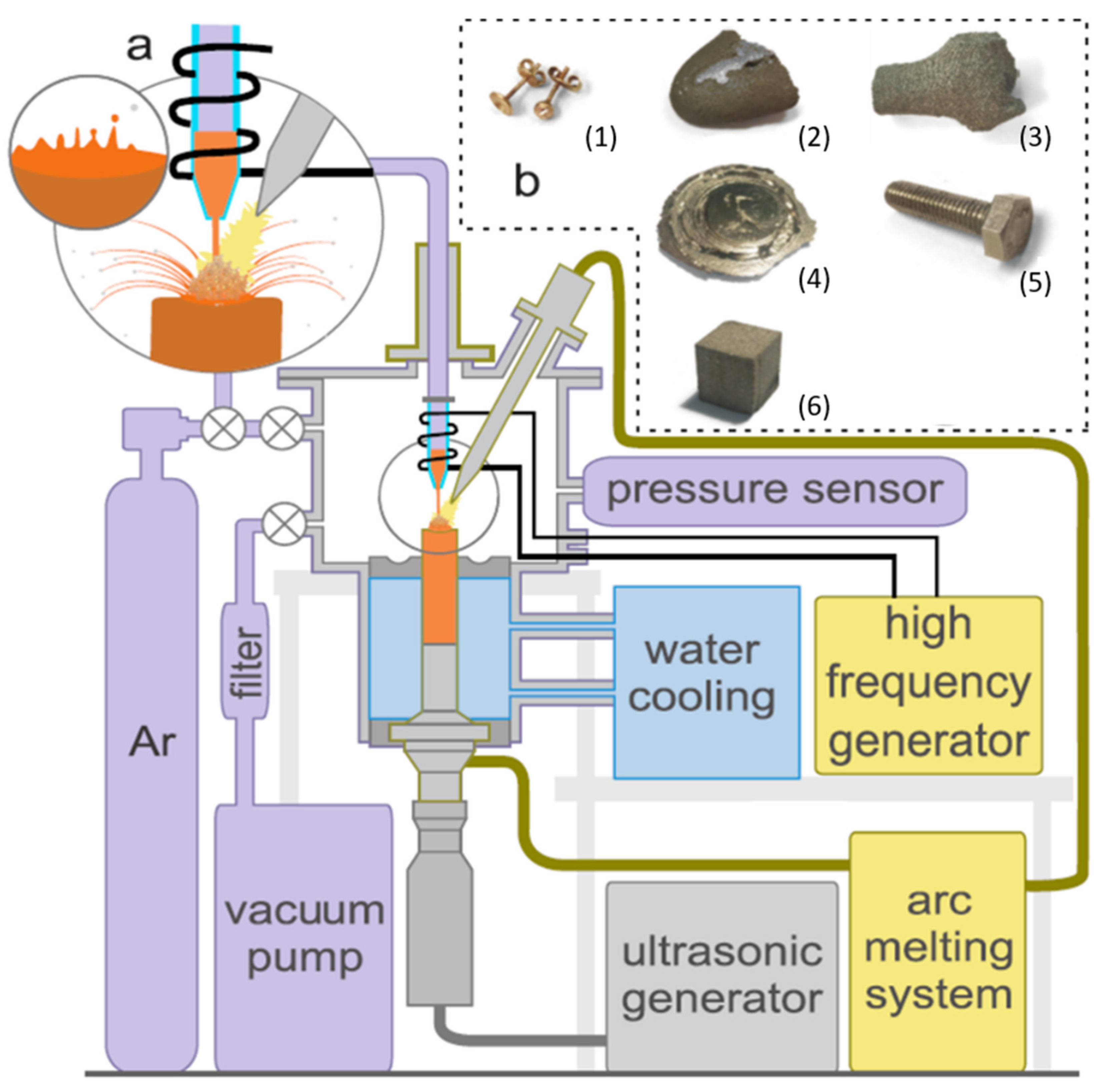
2. Materials and Methods
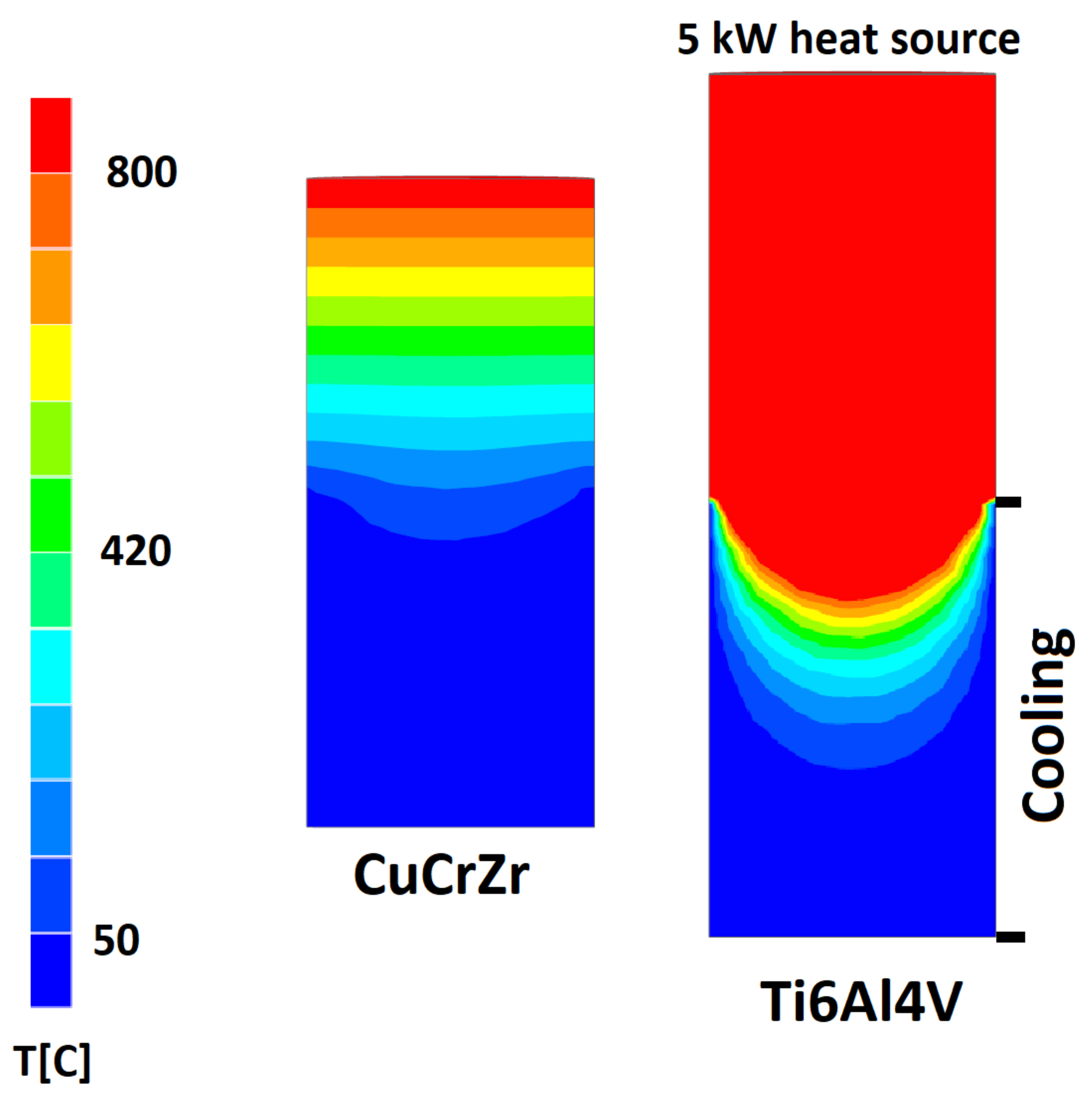
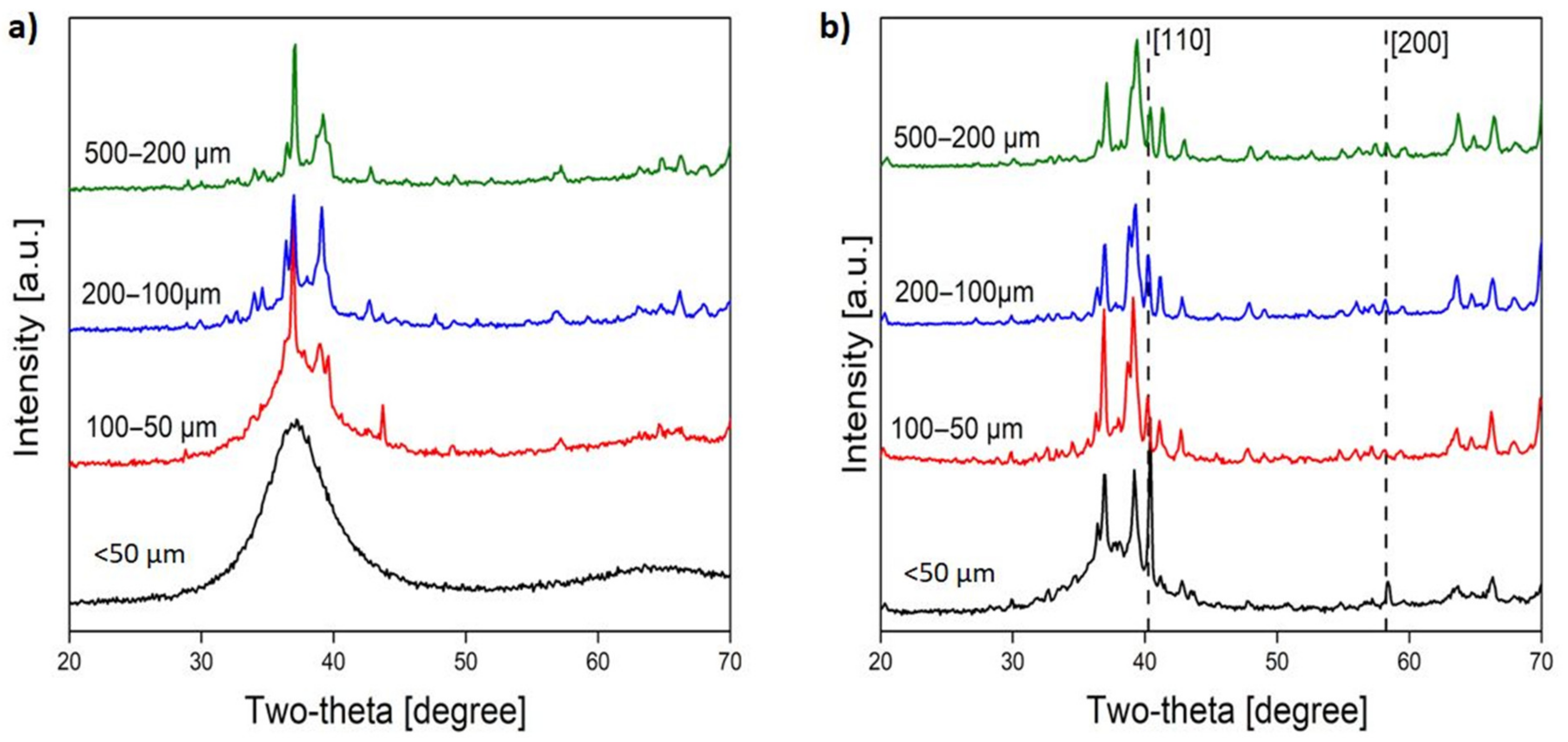
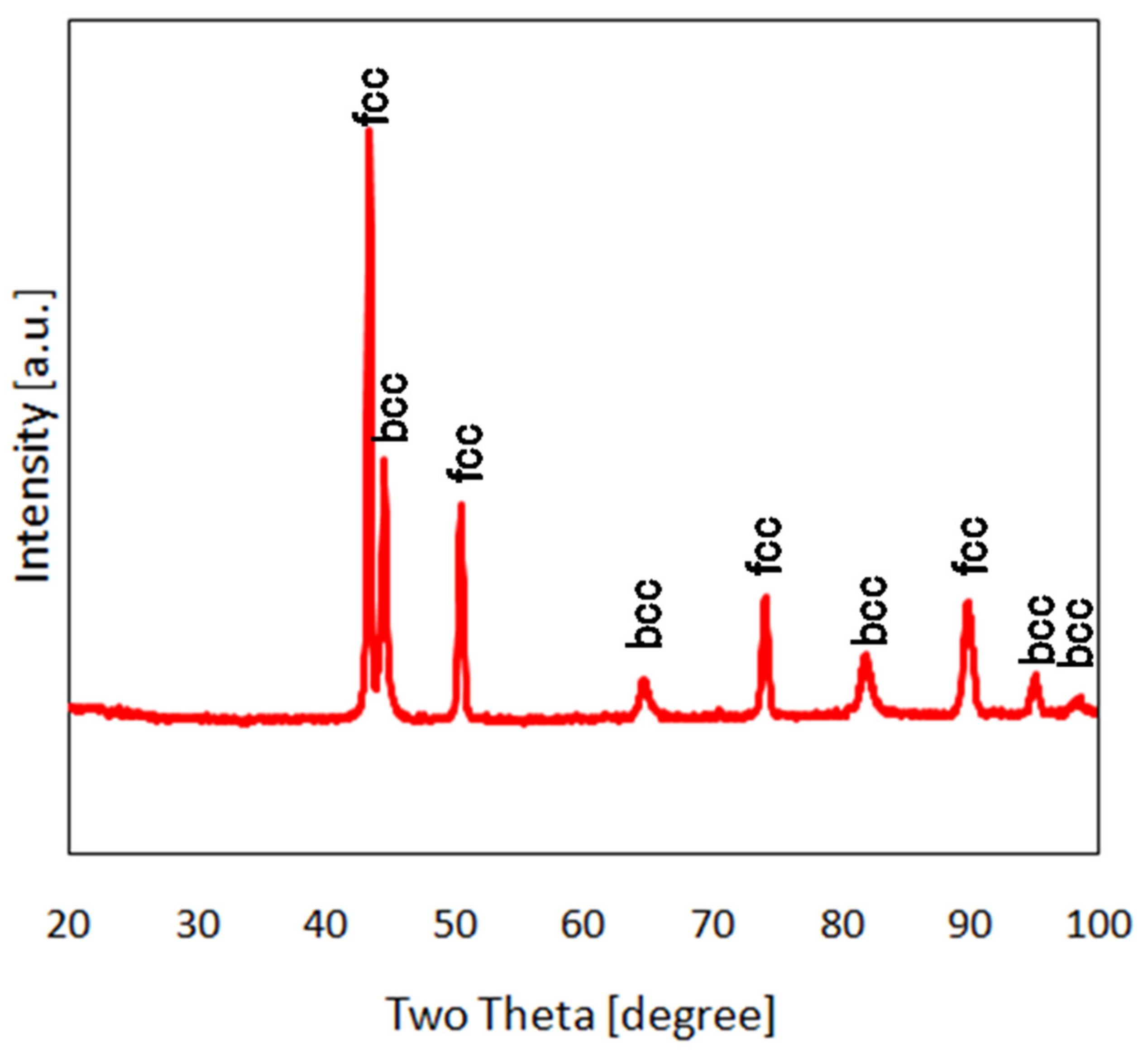
3. Results and Discussion
- (1)
- 0 < 50 μm was 11.38 ± 0.2%;
- (2)
- 50 < 100 μm was 13.40 ± 0.1%;
- (3)
- 100 < 200 μm was 14.62 ± 0.1%;
- (4)
- 200 < 500 μm was 31,48% ± 0.1%.
4. Conclusions
- (1)
- The powder particles tend to be spherical, regardless of the selected composition or particle size, with an exceptional Hall flowability of 10–12 s for AISI 304.
- (2)
- The particle size distribution is dependent on the alloying elements, indicating the importance of surface tension in atomization mechanisms.
- (3)
- Slight contamination and strong evaporation of manganese, zinc and zirconium were observed.
- (4)
- The cooling rates achieved during the process were high enough to provide the vitrification of the AMZ4 Zr-based alloy. The tungsten alloying seems to decrease the glass-forming ability of this system.
- (5)
- The proposed atomization technique was shown to be a useful tool for the development of new metallic materials for powder metallurgy and additive manufacturing. Moreover, it is possible to experiment with just a single or a few cubic centimeters of a particular material. This method is suitable for fabrication of any metallic material in small quantities, which is very convenient in experimental work.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Spierings, A.B.; Voegtlin, M.; Bauer, T.; Wegener, K. Powder flowability characterisation methodology for powder-bed-based metal additive manufacturing. Prog. Addit. Manuf. 2016, 1, 9–20. [Google Scholar] [CrossRef]
- Baitimerov, R.; Lykov, P.; Zherebtsov, D.; Radionova, L.; Shultc, A.; Prashanth, K.G. Influence of powder characteristics on processability of AlSi12 alloy fabricated by selective laser melting. Materials 2018, 11, 742. [Google Scholar] [CrossRef]
- Antony, L.V.; Reddy, R.G. Processes for production of high-purity metal powders. JOM 2003, 55, 14–18. [Google Scholar] [CrossRef]
- Dawes, J.; Bowerman, R.; Trepleton, R. Introduction to the Additive Manufacturing Powder Metallurgy Supply Chain. JMTRAP 2015, 59, 243–256. [Google Scholar] [CrossRef]
- Klotz, U.E.; Tiberto, D.; Held, F. Optimization of 18-karat yellow gold alloys for the additive manufacturing of jewelry and watch parts. Gold Bull. 2017, 50, 111–121. [Google Scholar] [CrossRef]
- Martín, A.; Cepeda-Jiménez, C.M.; Pérez-Prado, M.T. Gas atomization of γ-TiAl Alloy Powder for Additive Manufacturing. Adv. Eng. Mater. 2020, 22, 1900594. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.; Kim, K.; Lee, K.; Jeong, Y.; Kim, S.; Hwang, J.; Park, J. Development of Atomized Powder Fabrication Technology for High Uranium Density LEU Dispersion Targets. In Proceedings of the Transactions of the Korean Nuclear Society Spring Meeting, Jeju, Korea, 23–24 May 2019. [Google Scholar]
- Zeoli, N.; Gu, S. Numerical modelling of droplet break-up for gas atomisation. Comput. Mater. Sci. 2006, 38, 282–292. [Google Scholar] [CrossRef]
- Xie, J.W.; Zhao, Y.Y.; Dunkley, J.J. Effects of processing conditions on powder particle size and morphology in centrifugal atomisation of tin. Powder Metall. 2004, 47, 168–172. [Google Scholar] [CrossRef]
- Boulos, M. Plasma power can make better powders. Met. Powder Rep. 2004, 59, 16–21. [Google Scholar] [CrossRef]
- Lang, R.J. Ultrasonic atomization of liquids. J. Acoust. Soc. Am. 1962, 34, 6–8. [Google Scholar] [CrossRef]
- Lierke, E.G.; Griesshammer, G. The formation of metal powders by ultrasonic atomization of molten metals. Ultrasonics 1967, 5, 224–228. [Google Scholar] [CrossRef]
- Bauckhage, K.; Andersen, O.; Hansmann, S.; Reich, W.; Schreckenberg, P. Production of fine powders by ultrasonic standing wave atomization. Powder Technol. 1996, 86, 77–86. [Google Scholar] [CrossRef]
- Wisutmethangoon, S.; Plookphol, T.; Sungkhaphaitoon, P. Production of SAC305 powder by ultrasonic atomization. Powder Technol. 2011, 209, 105–111. [Google Scholar] [CrossRef]
- Pohlman, R.; Heisler, K.; Cichos, M. Powdering aluminium and aluminium alloys by ultrasound. Ultrasonics 1974, 12, 11–15. [Google Scholar] [CrossRef]
- Kruus, P. Production of zinc dust using ultrasound. Ultrasonics 1988, 26, 216–217. [Google Scholar] [CrossRef]
- Caccioppoli, G.; Clausen, B.; Bonjour, C.; Pralong, P. Fabrication of metal powders by ultrasonic atomization: Description of an atomizer based on a new ultrasonic device. Matériaux 2002, 100, 1–4. [Google Scholar]
- Alavi, S.H.; Harimkar, S.P. Ultrasonic vibration-assisted laser atomization of stainless steel. Powder Technol. 2017, 321, 89–93. [Google Scholar] [CrossRef]
- Goto, Y.; Kito, Y. Metallic Powder Production from a Plate Arc Electrode Vibrated in an Ultrasonic Frequency. IEEJ Trans. Fundam. Mater. 1992, 112, 695–700. [Google Scholar] [CrossRef]
- Wang, W.H.; Dong, C.; Shek, C.H. Bulk metallic glasses. Mater. Sci. Eng. R Rep. 2004, 44, 45–89. [Google Scholar] [CrossRef]
- Suryanarayana, C.; Inoue, A. Bulk Metallic Glasses; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar] [CrossRef]
- Li, X. Additive Manufacturing of Advanced Multi-Component Alloys: Bulk Metallic Glasses and High Entropy Alloys. Adv. Eng. Mater. 2018, 20, 1700874. [Google Scholar] [CrossRef]
- Gao, X.; Yu, Z.; Hu, W.; Lu, Y.; Zhu, Z.; Ji, Y.; Lu, Y.; Qin, Z.; Lu, X. In situ strengthening of CrMnFeCoNi high-entropy alloy with Al realized by laser additive manufacturing. JALCOM 2020, 847, 156563. [Google Scholar] [CrossRef]
- Zhou, K.; Wang, Z.; He, F.; Liu, S.; Li, J.; Kai, J.J.; Wang, J. A precipitation-strengthened high-entropy alloy for additive manufacturing. Addit. Manuf. 2020, 35, 101410. [Google Scholar] [CrossRef]
- Fedorov, M.; Wróbel, J.S.; Fernández-Caballero, A.; Kurzydłowski, K.J.; Nguyen-Manh, D. Phase stability and magnetic properties in fcc Fe-Cr-Mn-Ni alloys from first-principles modeling. Phys. Rev. B 2020, 101, 174416. [Google Scholar] [CrossRef]
- Sheikhaliev, S.M. Production of metal powders by ultrasonic atomization of melts. Sov. Powder Metall. Metal Ceram. 1983, 22, 793–798. [Google Scholar] [CrossRef]
- Leong, Z.; Wróbel, J.S.; Dudarev, S.L.; Goodall, R.; Todd, I.; Nguyen-Manh, D. The effect of electronic structure on the phases present in high entropy alloys. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Yasuda, K.; Honma, H.; Xu, Z.; Asakura, Y.; Koda, S. Ultrasonic atomization amount for different frequencies. Jpn. J. Appl. Phys. 2011, 50, 07HE23. [Google Scholar] [CrossRef]
- Rajan, R.; Pandit, A.B. Correlations to predict droplet size in ultrasonic atomisation. Ultrasonics 2001, 39, 235–255. [Google Scholar] [CrossRef]
- Smitll, A.F.; Hales, R. Diffusion of manganese in type 316 austenitic stainless steel. Met. Sci. 1975, 9, 181–184. [Google Scholar] [CrossRef]
- Block-Bolten, A.; Eagar, T.W. Metal vaporization from weld pools. Metall. Trans. B 1984, 15, 461–469. [Google Scholar] [CrossRef]
- Oerlikon.com. Available online: https://www.oerlikon.com/ecomaXL/files/metco/oerlikon_DSM-0272_AM_316L-AusteniticSteel.pdf&download=1 (accessed on 2 December 2020).
- Li, H.; Subhash, G.; Kecskes, L.J.; Dowding, R.J. Mechanical behavior of tungsten preform reinforced bulk metallic glass composites. Mater. Sci. Eng. A 2005, 403, 134–143. [Google Scholar] [CrossRef]
- Uslan, I.; Saritas, S.; Davies, T.J. Effects of variables on size and characteristics of gas atomized aluminium powders. Powder Metall. 1999, 42, 157–163. [Google Scholar] [CrossRef]



| µm | Ti | 304 | AMZ4 | AMZ4 + W | Ni–Mn–Ga | Fe–Cr–Mn–Ni | Au-Alloy |
|---|---|---|---|---|---|---|---|
| <50 | 5.8% | 3.2% | 10.2% | 1.7% | 2.3% | 13.1% | 21.1% |
| 50–100 | 32.7% | 41.9% | 23.3% | 11.8% | 49.9% | 42.1% | 42.4% |
| 100–200 | 46.2% | 36.3% | 20.9% | 22.7% | 38.0% | 40.1% | 22.3% |
| 200–500 | 15.3% | 18.6% | 45.6% | 63.8% | 9.8% | 4.7% | 14.2% |
| Material | Bulk (wt. %) | Powder (wt. %) |
|---|---|---|
| Ti Grade 2 | Ti 99.84 ± 0.02 Fe 0.16 ± 0.02 | Ti 99.53 ± 0.03 Fe 0.16 ± 0.02 Cu 0.15 ± 0.01 Ni 0.08 ± 0.01 Mn 0.08 ± 0.02 |
| 304 AISI | Fe 69.94 ± 0.13 Cr 19.89 ± 0.10 Ni 7.69 ± 0.09 Mn 1.48 ± 0.05 Cu 0.87 ± 0.03 Mo 0.13 ± 0.03 | Fe 70.12 ± 0.12 Cr 18.42 ± 0.09 Ni 8.23 ± 0.09 Mn 1.63 ± 0.05 Cu 0.85 ± 0.03 Mo 0.16 ± 0.04 Si 0.59 ± 0.02 |
| AMZ4 | Zr 75.12 ± 0.18 Cu 22.94 ± 0.18 Nb 1.94 ± 0.05 | Zr 73.81 ± 0.36 Cu 23.85 ± 0.34 Nb 1.94 ± 0.09 Fe 0.40 ± 0.16 |
| AMZ4+W | Zr 56.83 ± 0.14 W 21.60 ± 0.14 Cu 20.29 ± 0.10 Nb 1.28 ± 0.02 | Zr 55.67 ± 0.14 W 22.53 ± 0.15 Cu 20.49 ± 0.10 Nb 1.31 ± 0.02 |
| Ni–Mn–Ga | Ni 33.98 ± 0.11 Mn 35.17 ± 0.12 Ga 30.85 ± 0.06 | Ni 35.80 ± 0.12 Mn 31.80 ± 0.12 Ga 32.40 ± 0.06 |
| Fe–Cr–Mn–Ni | Fe 28.68 ± 0.15 Cr 24.83 ± 0.14 Mn 24.97 ± 0.13 Ni 21.52 ± 0.14 | Fe 28.52 ± 0.15 Cr 24.68 ± 0.13 Mn 21.74 ± 0.12 Ni 21.89 ± 0.13 Cu1.80 ± 0.05 W1.37 ± 0.05 |
| Au-alloy | Au 63.20 ± 0.17 Cu 24.63 ± 0.14 Ag 9.75 ± 0.10 Zn 2.42 ± 0.05 | Au 63.24 ± 0.10 Cu 27.61 ± 0.09 Ag 8.25 ± 0.05 Zn 0.90 ± 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Żrodowski, Ł.; Wróblewski, R.; Choma, T.; Morończyk, B.; Ostrysz, M.; Leonowicz, M.; Łacisz, W.; Błyskun, P.; Wróbel, J.S.; Cieślak, G.; et al. Novel Cold Crucible Ultrasonic Atomization Powder Production Method for 3D Printing. Materials 2021, 14, 2541. https://doi.org/10.3390/ma14102541
Żrodowski Ł, Wróblewski R, Choma T, Morończyk B, Ostrysz M, Leonowicz M, Łacisz W, Błyskun P, Wróbel JS, Cieślak G, et al. Novel Cold Crucible Ultrasonic Atomization Powder Production Method for 3D Printing. Materials. 2021; 14(10):2541. https://doi.org/10.3390/ma14102541
Chicago/Turabian StyleŻrodowski, Łukasz, Rafał Wróblewski, Tomasz Choma, Bartosz Morończyk, Mateusz Ostrysz, Marcin Leonowicz, Wojciech Łacisz, Piotr Błyskun, Jan S. Wróbel, Grzegorz Cieślak, and et al. 2021. "Novel Cold Crucible Ultrasonic Atomization Powder Production Method for 3D Printing" Materials 14, no. 10: 2541. https://doi.org/10.3390/ma14102541
APA StyleŻrodowski, Ł., Wróblewski, R., Choma, T., Morończyk, B., Ostrysz, M., Leonowicz, M., Łacisz, W., Błyskun, P., Wróbel, J. S., Cieślak, G., Wysocki, B., Żrodowski, C., & Pomian, K. (2021). Novel Cold Crucible Ultrasonic Atomization Powder Production Method for 3D Printing. Materials, 14(10), 2541. https://doi.org/10.3390/ma14102541










