Relevant Properties of Carbon Support Materials in Successful Fe-N-C Synthesis for the Oxygen Reduction Reaction: Study of Carbon Blacks and Biomass-Based Carbons
Abstract
1. Introduction
2. Materials and Methods
2.1. Oxidation of Commercial Carbon Support
2.2. Activation of Biomass
2.3. Fe-N-C Synthesis
2.4. Methods
2.5. Electrochemical Measurement
3. Results and Discussion
3.1. Physical Properties of the Carbon Supports
3.2. Application of Carbon Supports in Fe-N-C Synthesis
3.3. Impact of Carbon Support on Fe-Nx Site Incorporation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, Y.; Ji, S.; Zhao, S.; Chen, W.; Dong, J.; Cheong, W.C.; Shen, R.; Wen, X.; Zheng, L.; Rykov, A.I.; et al. Enhanced oxygen reduction with single-atomic-site iron catalysts for a zinc-air battery and hydrogen-air fuel cell. Nat. Commun. 2018, 9, 1–12. [Google Scholar] [CrossRef]
- Jaouen, F.; Jones, D.; Coutard, N.; Artero, V.; Strasser, P.; Kucernak, A. Toward platinum group metal-free catalysts for hydrogen/air proton-exchange membrane fuel cells. Johns. Matthey Technol. Rev. 2018, 62, 231–255. [Google Scholar] [CrossRef]
- Osmieri, L. Transition metal–nitrogen–carbon (M–N–C) catalysts for oxygen reduction reaction. Insights on synthesis and performance in polymer electrolyte fuel cells. ChemEngineering 2019, 3, 16. [Google Scholar] [CrossRef]
- Hu, Y.; Jensen, J.O.; Pan, C.; Cleemann, L.N.; Shypunov, I.; Li, Q. Immunity of the Fe-NC catalysts to electrolyte adsorption: Phosphate but not perchloric anions. Appl. Catal. B Environ. 2018, 234, 357–364. [Google Scholar] [CrossRef]
- Martinaiou, I.; Shahraei, A.; Grimm, F.; Zhang, H.; Wittich, C.; Klemenz, S.; Dolique, J.; Kleebe, H.-J.; Stark, R.W.; Kramm, U.I. Effect of metal species on the stability of Me-NC catalysts during accelerated stress tests mimicking the start-up and shut-down conditions. Electrochim. Acta 2017, 243, 183–196. [Google Scholar] [CrossRef]
- Banham, D.; Ye, S.; Pei, K.; Ozaki, J.I.; Kishimoto, T.; Imashiro, Y. A review of the stability and durability of non-precious metal catalysts for the oxygen reduction reaction in proton exchange membrane fuel cells. J. Power Sources 2015, 285, 334–348. [Google Scholar] [CrossRef]
- Chung, H.T.; Johnston, C.M.; Artyushkova, K.; Ferrandon, M.; Myers, D.J.; Zelenay, P. Cyanamide-derived non-precious metal catalyst for oxygen reduction. Electrochem. Commun. 2010, 12, 1792–1795. [Google Scholar] [CrossRef]
- Artyushkova, K.; Rojas-Carbonell, S.; Santoro, C.; Weiler, E.; Serov, A.; Awais, R.; Gokhale, R.R.; Atanassov, P. Correlations between Synthesis and Performance of Fe-Based PGM-Free Catalysts in Acidic and Alkaline Media: Evolution of Surface Chemistry and Morphology. ACS Appl. Energy Mater. 2019, 2, 5406–5418. [Google Scholar] [CrossRef]
- Shui, J.; Chen, C.; Grabstanowicz, L.; Zhao, D.; Liu, D.J. Highly efficient nonprecious metal catalyst prepared with metal–organic framework in a continuous carbon nanofibrous network. Proc. Natl. Acad. Sci. USA 2015, 112, 10629–10634. [Google Scholar] [CrossRef]
- Medard, C.; Lefevre, M.; Dodelet, J.P.; Jaouen, F.; Lindbergh, G. Oxygen reduction by Fe-based catalysts in PEM fuel cell conditions: Activity and selectivity of the catalysts obtained with two Fe precursors and various carbon supports. Electrochim. Acta 2006, 51, 3202–3213. [Google Scholar] [CrossRef]
- Li, J.C.; Tang, D.M.; Hou, P.X.; Li, G.X.; Cheng, M.; Liu, C.; Cheng, H.M. The effect of carbon support on the oxygen reduction activity and durability of single-atom iron catalysts. MRS Commun. 2018, 8, 1158–1166. [Google Scholar] [CrossRef]
- Kumar, K.; Gairola, P.; Lions, M.; Ranjbar-Sahraie, N.; Mermoux, M.; Dubau, L.; Zitolo, A.; Maillard, F. Physical and chemical considerations for improving catalytic activity and stability of non-precious-metal oxygen reduction reaction catalysts. ACS Catal. 2018, 8, 11264–11276. [Google Scholar] [CrossRef]
- Sgarbi, R.; Kumar, K.; Jaouen, F.; Zitolo, A.; Ticianelli, E.A.; Maillard, F. Oxygen reduction reaction mechanism and kinetics on M-NxCy and M@N-C active sites present in model M-N-C catalysts under alkaline and acidic conditions. J. Solid State Electrochem. 2019, 1–12. [Google Scholar] [CrossRef]
- Li, J.; Jiao, L.; Wegener, E.; Richard, L.L.; Liu, E.; Zitolo, A.; Sougrati, M.T.; Mukerjee, S.; Zhao, Z.; Huang, Y.; et al. Evolution Pathway from Iron Compounds to Fe1(II)–N4 Sites through Gas-Phase Iron during Pyrolysis. J. Am. Chem. Soc. 2019, 142, 1417–1423. [Google Scholar] [CrossRef]
- Choi, C.H.; Baldizzone, C.; Polymeros, G.; Pizzutilo, E.; Kasian, O.; Schuppert, A.K.; Sahraie, N.R.; Sougrati, M.T.; Mayrhofer, K.J.J.; Jaouen, F. Minimizing operando demetallation of Fe-NC electrocatalysts in acidic medium. ACS Catal. 2016, 6, 3136–3146. [Google Scholar] [CrossRef]
- Kramm, U.I.; Herranz, J.; Larouche, N.; Arruda, T.M.; Lefevre, M.; Jaouen, F.; Bogdanoff, P.; Fiechter, S.; Abs-Wurmbach, I.; Dodelet, J.P.; et al. Structure of the catalytic sites in Fe/N/C-catalysts for O2-reduction in PEM fuel cells. Phys. Chem. Chem. Phys. 2012, 14, 11673–11688. [Google Scholar] [CrossRef]
- Zhang, H.; Chung, H.T.; Cullen, D.A.; Wagner, S.; Kramm, U.I.; More, K.L.; Zelenay, P.; Wu, G. High-performance fuel cell cathodes exclusively containing atomically dispersed iron active sites. Energy Environ. Sci. 2019, 12, 2548–2558. [Google Scholar] [CrossRef]
- Tian, J.; Birry, L.; Jaouen, F.; Dodelet, J.P. Fe-based catalysts for oxygen reduction in proton exchange membrane fuel cells with cyanamide as nitrogen precursor and/or pore-filler. Electrochim. Acta 2011, 56, 3276–3285. [Google Scholar] [CrossRef]
- Setyowati, V.A.; Huang, H.C.; Liu, C.C.; Wang, C.H. Effect of iron precursors on the structure and oxygen reduction activity of iron–nitrogen–carbon catalysts. Electrochim. Acta 2011, 211, 933–940. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, J.; Chung, D.Y.; Yoo, J.M.; Lee, H.S.; Kim, M.J.; Mun, S.B.; Kwon, S.G.; Sung, Y.E.; Hyeon, T. Design principle of Fe–N–C electrocatalysts: How to optimize multimodal porous structures? J. Am. Chem. Soc. 2019, 141, 2035–2045. [Google Scholar] [CrossRef]
- Lefèvre, M.; Proietti, E.; Jaouen, F.; Dodelet, J.P. Iron-based catalysts with improved oxygen reduction activity in polymer electrolyte fuel cells. Science 2009, 324, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Jaouen, F.; Lefèvre, M.; Dodelet, J.-P.; Cai, M. Heat-Treated Fe/N/C Catalysts for O2 Electroreduction: Are Active Sites Hosted in Micropores? J. Phys. Chem. B 2006, 110, 5553–5558. [Google Scholar] [CrossRef]
- Jaouen, F.; Herranz, J.; Lefevre, M.; Dodelet, J.P.; Kramm, U.I.; Herrmann, I.; Bogdanoff, P.; Maruyama, J.; Nagaoka, T.; Garsuch, A.; et al. Cross-laboratory experimental study of non-noble-metal electrocatalysts for the oxygen reduction reaction. ACS Appl. Mater. Interfaces 2009, 1, 1623–1639. [Google Scholar] [CrossRef]
- Jaouen, F.; Charreteur, F.; Dodelet, J.P. Fe-based catalysts for oxygen reduction in PEMFCs: Importance of the disordered phase of the carbon support. J. Electrochem. Soc. 2006, 153, A89. [Google Scholar] [CrossRef]
- Leonard, N.; Nallathambi, V.; Barton, S.C. Carbon supports for non-Precious metal oxygen reducing catalysts. J. Electrochem. Soc. 2013, 160, F788. [Google Scholar] [CrossRef]
- Volperts, A.; Plavniece, A.; Dobele, G.; Zhurinsh, A.; Kruusenberg, I.; Kaare, K.; Locs, J.; Tamasauskaite-Tamasiunaite, L.; Norkus, E. Biomass based activated carbons for fuel cells. Renew. Energy 2019, 141, 40–45. [Google Scholar] [CrossRef]
- De, S.; Balu, A.M.; Van der Waal, J.C.; Luque, R. Biomass-derived porous carbon materials: Synthesis and catalytic applications. ChemCatChem 2015, 7, 1608–1629. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, Y.; Lin, C.; Yang, W.; Meng, Y.; Guo, Y.; Li, M.; Xiao, D. Hierarchically porous nitrogen-rich carbon derived from wheat straw as an ultra-high-rate anode for lithium ion batteries. J. Mater. Chem. 2014, 2, 9684–9690. [Google Scholar] [CrossRef]
- Koyutürk, B.; Evans, J.; Multhaupt, H.; Selve, S.; Simke, J.R.J.; Wark, M.; Fellinger, T.P. Sol–gel chemistry in molten Brønsted acids towards “activated” carbons and beyond. Nanoscale 2019, 11, 13154–13160. [Google Scholar] [CrossRef]
- Kiciński, W.; Dyjak, S. Transition metal impurities in carbon-based materials: Pitfalls, artifacts and deleterious effects. Carbon 2020, 168, 748–845. [Google Scholar] [CrossRef]
- Schmies, H.; Hornberger, E.; Anke, B.; Jurzinsky, T.; Nong, H.N.; Dionigi, F.; Kuhl, S.; Drnec, J.; Lerch, M.; Cremers, C.; et al. Impact of carbon support functionalization on the electrochemical stability of Pt fuel cell catalysts. Chem. Mater. 2018, 30, 7287–7295. [Google Scholar] [CrossRef]
- Chen, F.; Ma, L.; Ren, J.; Zhang, M.; Luo, X.; Li, B.; Song, Z.; Zhou, X. Wheat Straw-Derived N-, O-, and S-Tri-doped Porous Carbon with Ultrahigh Specific Surface Area for Lithium-Sulfur Batteries. Materials 2018, 11, 989. [Google Scholar] [CrossRef]
- Choi, J.Y.; Hsu, R.S.; Chen, Z. Highly active porous carbon-supported nonprecious metal−N electrocatalyst for oxygen reduction reaction in PEM fuel cells. J. Phys. Chem. 2010, 114, 8048–8053. [Google Scholar] [CrossRef]
- Wu, G.; More, K.L.; Johnston, C.M.; Zelenay, P. High-performance electrocatalysts for oxygen reduction derived from polyaniline, iron, and cobalt. Science 2011, 332, 443–447. [Google Scholar] [CrossRef]
- Lippens, B.C.; De Boer, J.H. Studies on pore systems in catalysts: V. The t method. J. Catal. 1965, 4, 319–323. [Google Scholar] [CrossRef]
- Pantea, D.; Darmstadt, H.; Kaliaguine, S.; Roy, C. Electrical conductivity of conductive carbon blacks: Influence of surface chemistry and topology. Appl. Surf. Sci. 2003, 217, 181–193. [Google Scholar] [CrossRef]
- Sadezky, A.; Muckenhuber, H.; Grothe, H.; Niessner, R.; Pöschl, U. Raman microspectroscopy of soot and related carbonaceous materials: Spectral analysis and structural information. Carbon 2005, 43, 1731–1742. [Google Scholar] [CrossRef]
- Primbs, M.; Sun, Y.; Roy, A.; Malko, D.; Mehmood, A.; Sougrati, M.T.; Blanchard, P.Y.; Granozzi, G.; Kosmal, T.; Daniel, G.; et al. Establishing reactivity descriptors for platinum group metal (PGM)-free Fe–N–C catalysts for PEM fuel cells. Energy Environ. Sci. 2020, 13, 2480–2500. [Google Scholar] [CrossRef]
- Shafeeyan, M.S.; Daud, W.M.A.W.; Houshmand, A.; Arami-Niya, A. Ammonia modification of activated carbon to enhance carbon dioxide adsorption: Effect of pre-oxidation. Appl. Surf. Sci 2011, 257, 3936–3942. [Google Scholar] [CrossRef]
- Kruk, M.; Jaroniec, M.; Bereznitski, Y. Adsorption study of porous structure development in carbon blacks. J. Colloid Interface Sci. 1996, 182, 282–288. [Google Scholar] [CrossRef]
- Shan, D.; Yang, J.; Liu, W.; Yan, J.; Fan, Z. Biomass-derived three-dimensional honeycomb-like hierarchical structured carbon for ultrahigh energy density asymmetric supercapacitors. J. Mater. Chem. A 2016, 4, 13589–13602. [Google Scholar] [CrossRef]
- Rehman, A.; Park, S.J. Microporous carbons derived from melamine and isophthalaldehyde: One-pot condensation and activation in a molten salt medium for efficient gas adsorption. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hilton, R.; Bick, P.; Tekeei, A.; Leimkuehler, E.; Pfeifer, P.; Suppes, G.J. Mass balance and performance analysis of potassium hydroxide activated carbon. Ind. Eng. Chem. Res. 2012, 51, 9129–9135. [Google Scholar] [CrossRef]
- Lillo-Ródenas, M.A.; Cazorla-Amorós, D.; Linares-Solano, A. Understanding chemical reactions between carbons and NaOH and KOH: An insight into the chemical activation mechanism. Carbon 2003, 41, 267–275. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Knauer, M.; Carrara, M.; Rothe, D.; Niessner, R.; Ivleva, N.P. Changes in Structure and Reactivity of Soot during Oxidation and Gasification by Oxygen, Studied by Micro-Raman Spectroscopy and Temperature Programmed Oxidation. Aerosol Sci. Technol. 2009, 43, 1–8. [Google Scholar] [CrossRef]
- Schardt, S.; Weidler, N.; Wallace, W.D.Z.; Martinaiou, I.; Stark, R.W.; Kramm, U.I. Influence of the structure-forming agent on the performance of Fe-NC Catalysts. Catalysts 2018, 8, 260. [Google Scholar]
- Baldelli, A.; Rogak, S.N. Morphology and Raman spectra of aerodynamically classified soot samples. Atmos. Meas. Tech. 2019, 12, 4339–4346. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertso, J. Interpretation of Raman spectra of disordered and amorphous carbon. Phys. Rev. B 2000, 61, 14095. [Google Scholar] [CrossRef]
- Pawlyta, M.; Rouzaud, J.N.; Duber, S. Raman microspectroscopy characterization of carbon blacks: Spectral analysis and structural information. Carbon 2015, 84, 479–490. [Google Scholar] [CrossRef]
- Wu, G.; Santandreu, A.; Kellogg, W.; Gupta, S.; Ogoke, O.; Zhang, H.; Wang, H.L.; Dai, L. Carbon nanocomposite catalysts for oxygen reduction and evolution reactions: From nitrogen doping to transition-metal addition. Nano Energy 2016, 29, 83–110. [Google Scholar] [CrossRef]
- Li, J.; Jia, Q.; Mukerjee, S.; Sougrati, M.T.; Drazic, G.; Zitolo, A.; Jaouen, F. The Challenge of Achieving a High Density of Fe-Based Active Sites in a Highly Graphitic Carbon Matrix. Catalysts 2019, 9, 144. [Google Scholar] [CrossRef]
- Thomas, A.; Fischer, A.; Goettmann, F.; Antonietti, M.; Müller, J.O.; Schlögl, R.; Carlsson, J.M. Graphitic carbon nitride materials: Variation of structure and morphology and their use as metal-free catalysts. J. Mater. Chem. 2008, 18, 4893–4908. [Google Scholar] [CrossRef]
- Lee, J.H.; Park, M.J.; Jung, J.; Ryu, J.; Cho, E.; Nam, S.W.; Kim, J.Y.; Yoon, C.W. Facile synthesis of hollow Fe–N–C hybrid nanostructures for oxygen reduction reactions. Inorg. Chim. Acta 2014, 422, 3–7. [Google Scholar] [CrossRef]
- Onodera, T.; Mizukami, T.; Suzuki, S.; Kawaji, J.; Yamaga, K.; Yamamoto, T. Analysis of forming process of nitrogen-doped carbon catalyst derived from Fe 1, 10-phenanthroline compound and its oxygen reduction reaction activity. Catal. Commun. 2014, 43, 66–71. [Google Scholar] [CrossRef]
- Strickland, K.; Miner, E.; Jia, Q.; Tylus, U.; Ramaswamy, N.; Liang, W.; Sougrati, M.T.; Jaouen, F.; Mukerjee, S. Highly active oxygen reduction non-platinum group metal electrocatalyst without direct metal–nitrogen coordination. Nat. Commun. 2015, 6, 1–8. [Google Scholar] [CrossRef]
- Zhong, L.; Jensen, J.O.; Cleemann, L.N.; Pan, C.; Li, Q. Electrochemical probing into the active sites of graphitic-layer encapsulated iron oxygen reduction reaction electrocatalysts. Sci. Bull. 2018, 63, 24–30. [Google Scholar] [CrossRef]
- Yang, W.; Liu, X.; Yue, X.; Jia, J.; Guo, S. Bamboo-like carbon nanotube/Fe3C nanoparticle hybrids and their highly efficient catalysis for oxygen reduction. J. Am. Chem. Soc. 2015, 137, 1436–1439. [Google Scholar] [CrossRef]
- Huang, Z.; Pan, H.; Yang, W.; Zhou, H.; Gao, N.; Fu, C.; Li, S.; Li, H.; Kuang, Y. In situ self-template synthesis of Fe–N-doped double-shelled hollow carbon microspheres for oxygen reduction reaction. ACS Nano 2018, 12, 208–216. [Google Scholar] [CrossRef]
- Lai, Q.; Su, Q.; Gao, Q.; Liang, Y.; Wang, Y.; Yang, Z.; Zhang, X.; He, J.; Tong, H. In situ self-sacrificed template synthesis of Fe-N/G catalysts for enhanced oxygen reduction. ACS Appl. Mater. Interfaces 2015, 7, 18170–18178. [Google Scholar] [CrossRef]
- Chung, H.T.; Cullen, D.A.; Higgins, D.; Sneed, B.T.; Holby, E.F.; More, K.L.; Zelenay, P. Direct atomic-level insight into the active sites of a high-performance PGM-free ORR catalyst. Science 2017, 357, 479–484. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Maurice, J.L.; Gohier, A.; Lee, C.S.; Pribat, D.; Cojocaru, C.S. Iron catalysts for the growth of carbon nanofibers: Fe, Fe3C or both? Chem. Mater. 2011, 23, 5379–5387. [Google Scholar] [CrossRef]
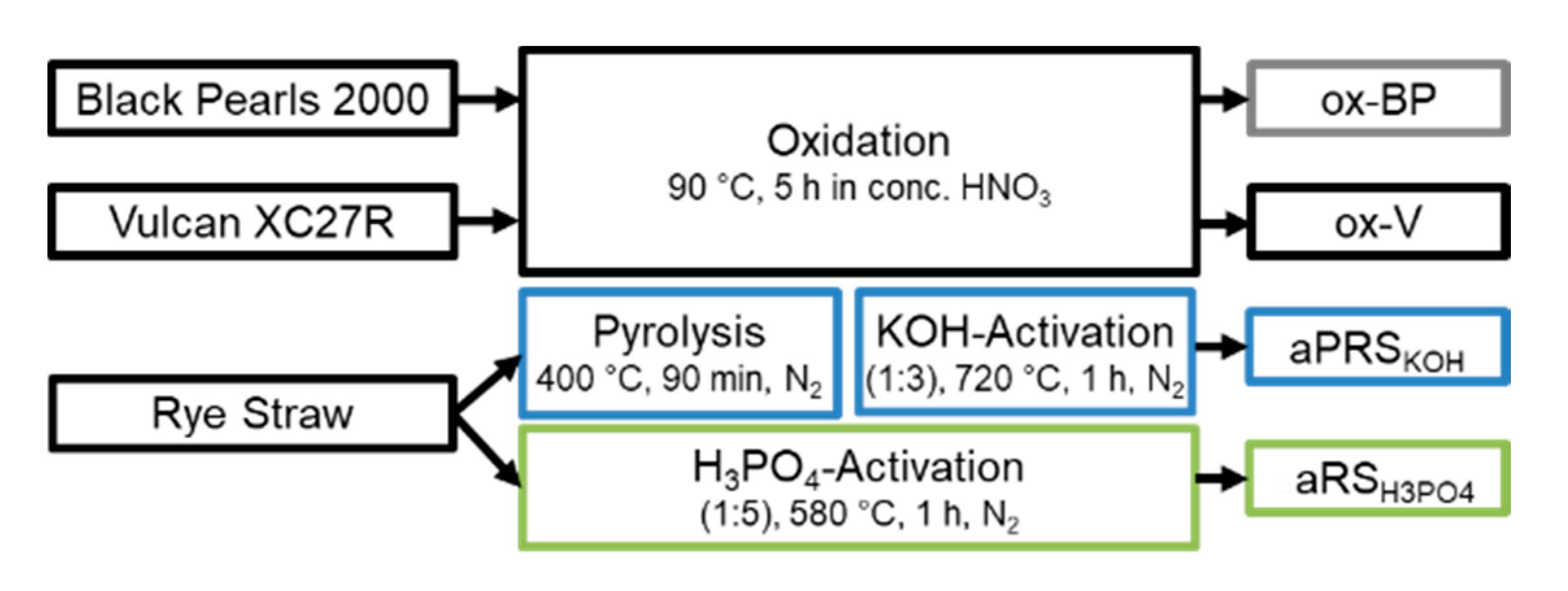

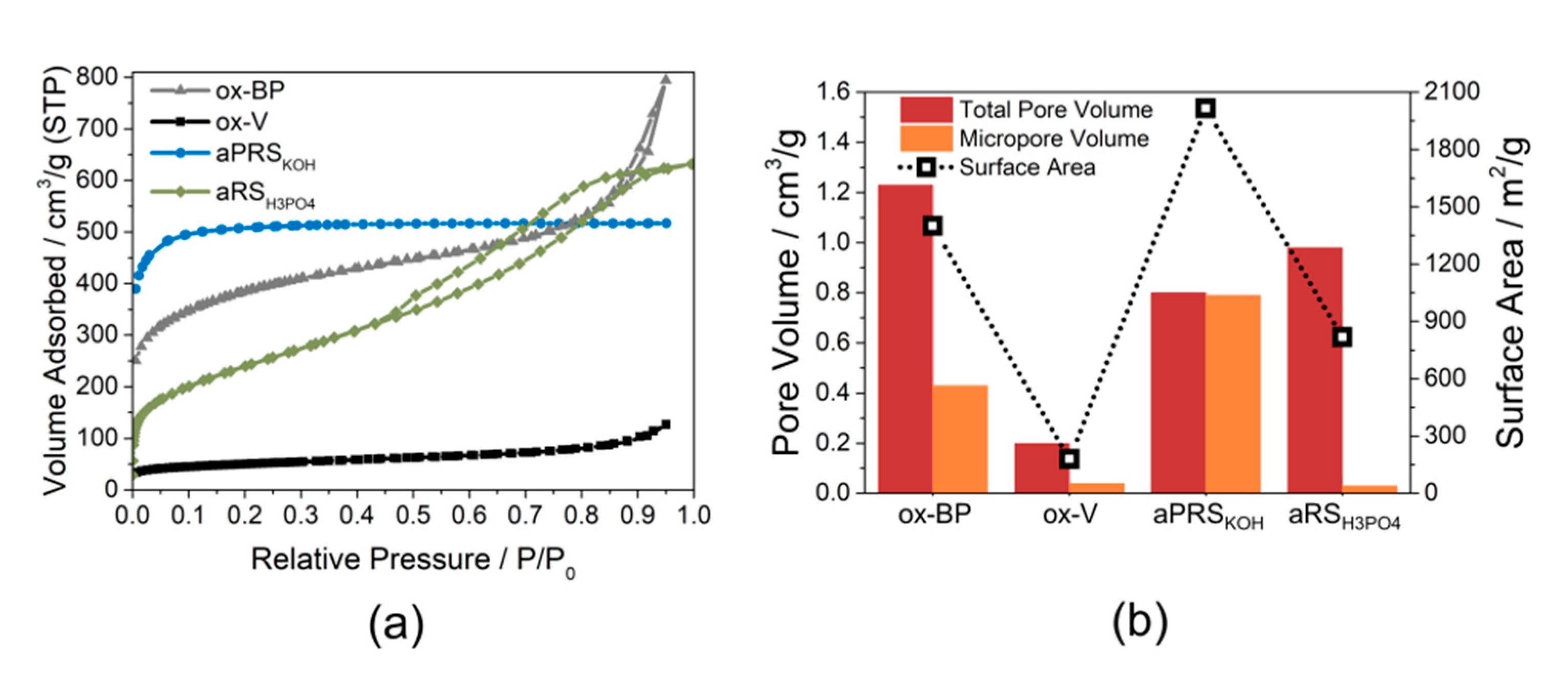
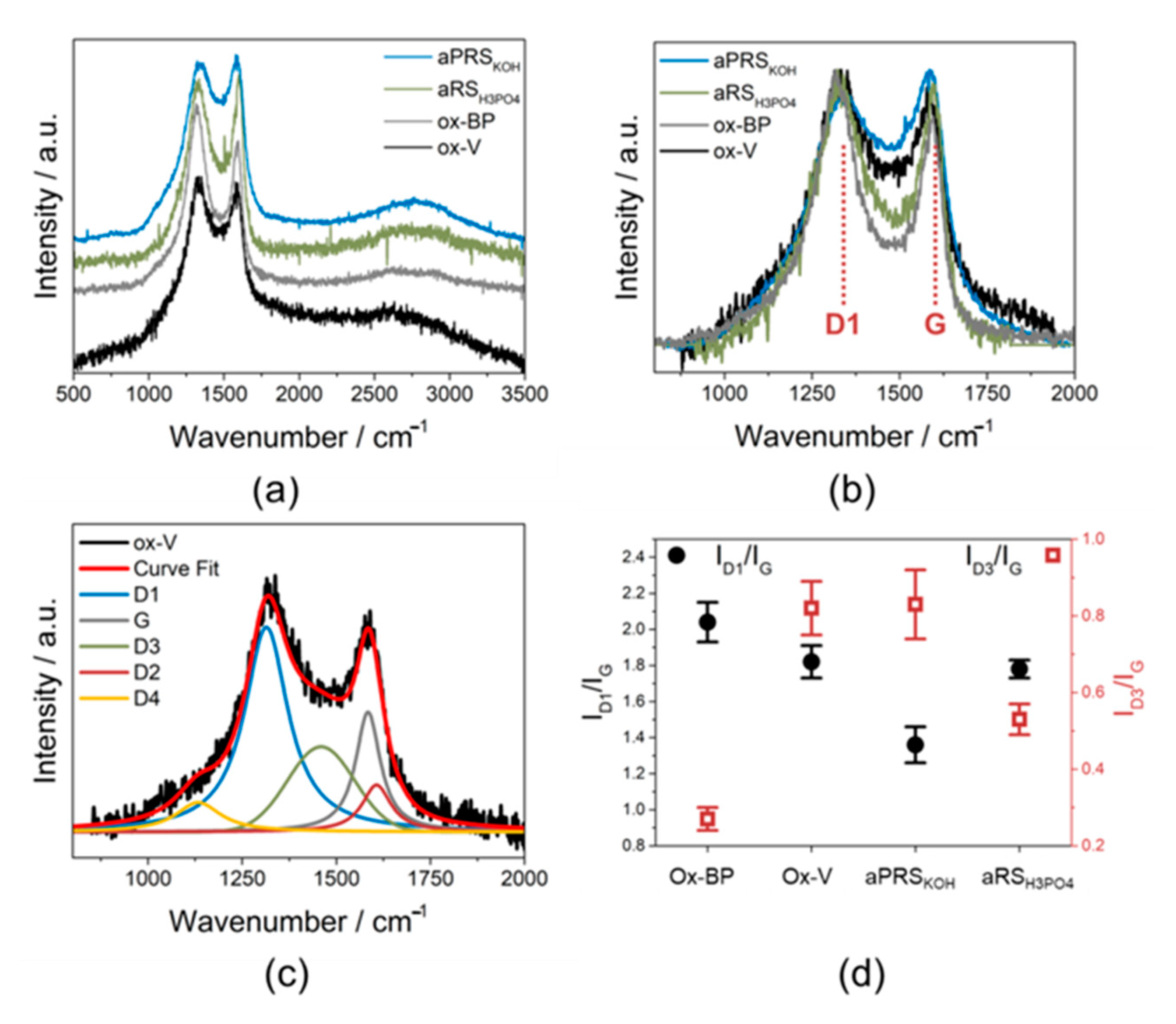
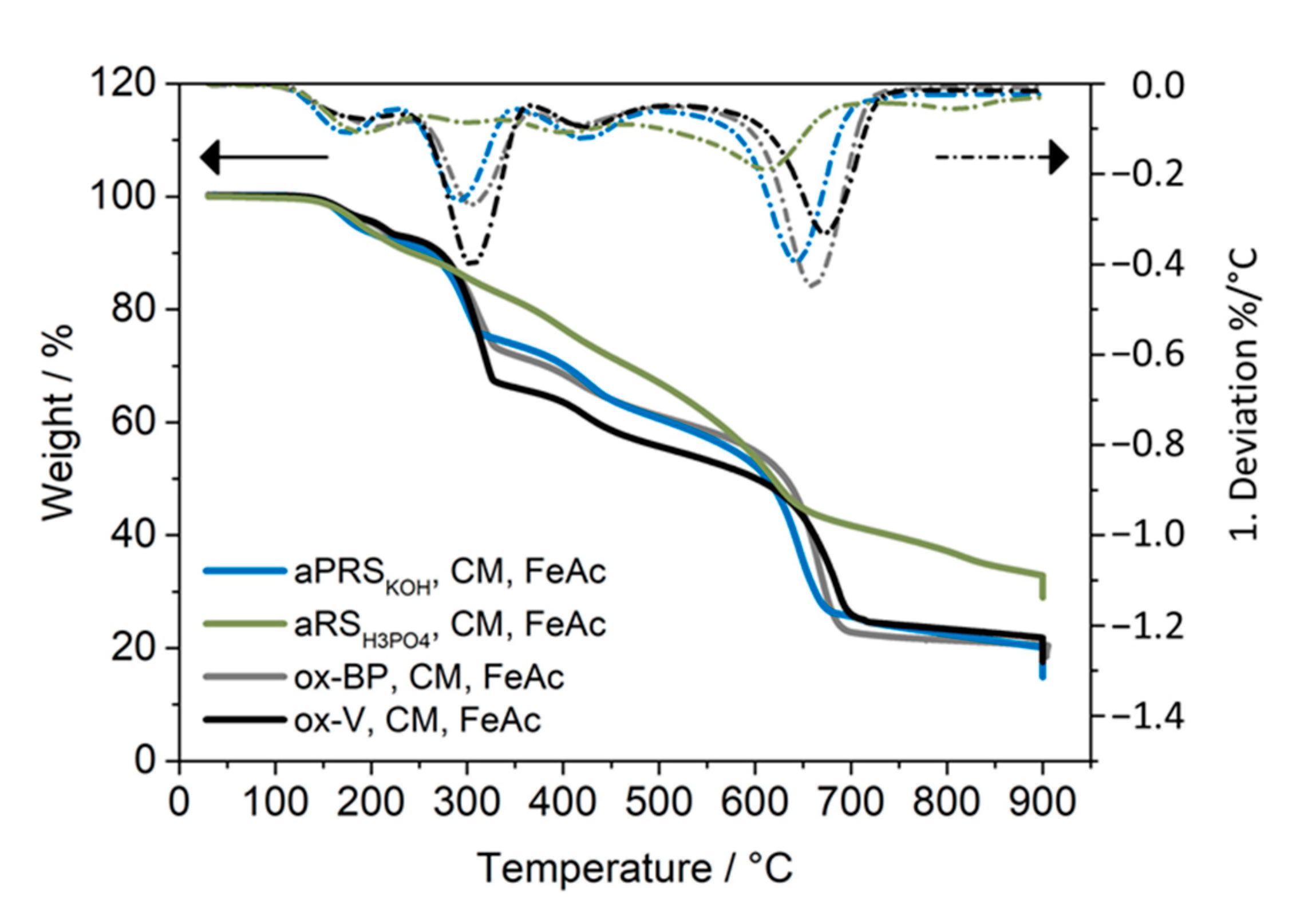


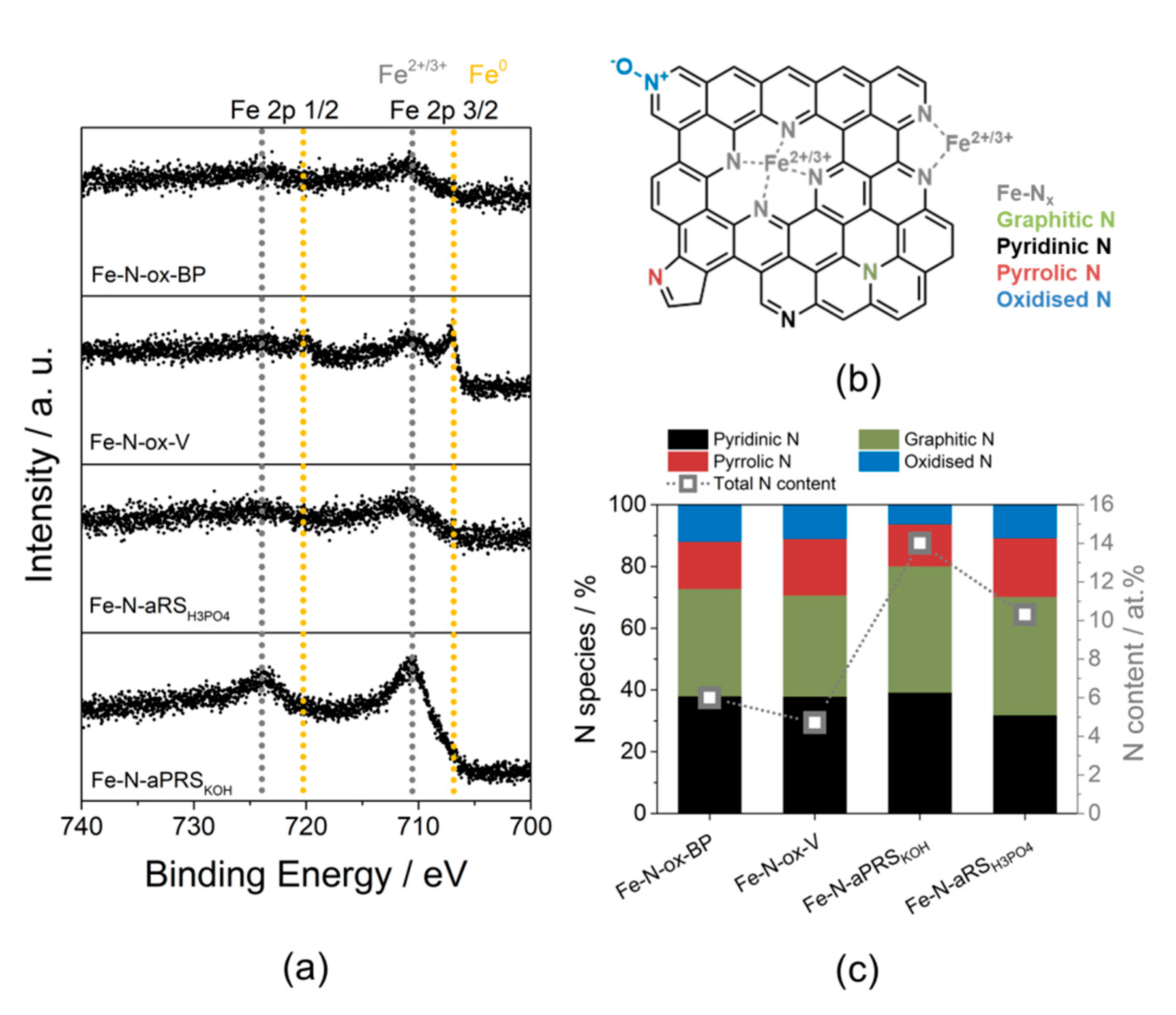

| Sample | SSA/m2/g | PV/cm3/g | MiPV/cm3/g | MiA m2/g |
|---|---|---|---|---|
| ox-BP | 1400 | 1.23 | 0.43 | 991 |
| Fe-N-ox-BP | 1280 | 1.19 | 0.39 | 902 |
| ox-V | 179 | 0.20 | 0.04 | 89 |
| Fe-N-ox-V | 172 | 0.22 | 0.04 | 95 |
| aPRSKOH | 2015 | 0.80 | 0.79 | 1998 |
| Fe-N-aPRSKOH | 863 | 0.56 | 0.28 | 675 |
| aRSH3PO4 | 818 | 0.98 | 0.03 | 37 |
| Fe-N-aRSH3PO4 | 977 | 0.88 | 0.11 | 214 |
| Parameter | Ox-BP | Ox-V | aPRSKOH | aRSH3PO4 |
|---|---|---|---|---|
| Morphology (SEM, Figure 1) | Small C-particles (<100 nm) | Small C-particles (<100 nm) | Large C-particles (≥100 nm) | Large C-particles (≥100 nm) |
| Surface area (N2 sorption, Figure 2) | Moderate SA (≥800 m2/g) | Low SA (<800 m2/g) | Moderate SA (≥800 m2/g) | Moderate SA (≥800 m2/g) |
| Pore volume (N2 sorption, Table 1) | Moderate PV (≥0.8 cm3/g) | Low PV (<0.8 cm3/g) | Moderate PV (≥0.8 cm3/g) | Moderate PV (≥0.8 cm3/g) |
| Porosity (N2 sorption, Table 1) | Micro + Meso | - | Micro | Meso |
| Carbon structure (Raman, Figure 3) | Less amorphous (ID3/IG < 0.8) | More amorphous (ID3/IG ≥ 0.8) | More amorphous (ID3/IG ≥ 0.8) | Less amorphous (ID3/IG < 0.8) |
| Incorporation of Fe-Nx-sites | Successful | Mainly Fe particle | Mainly Fe3C particle | Successful |
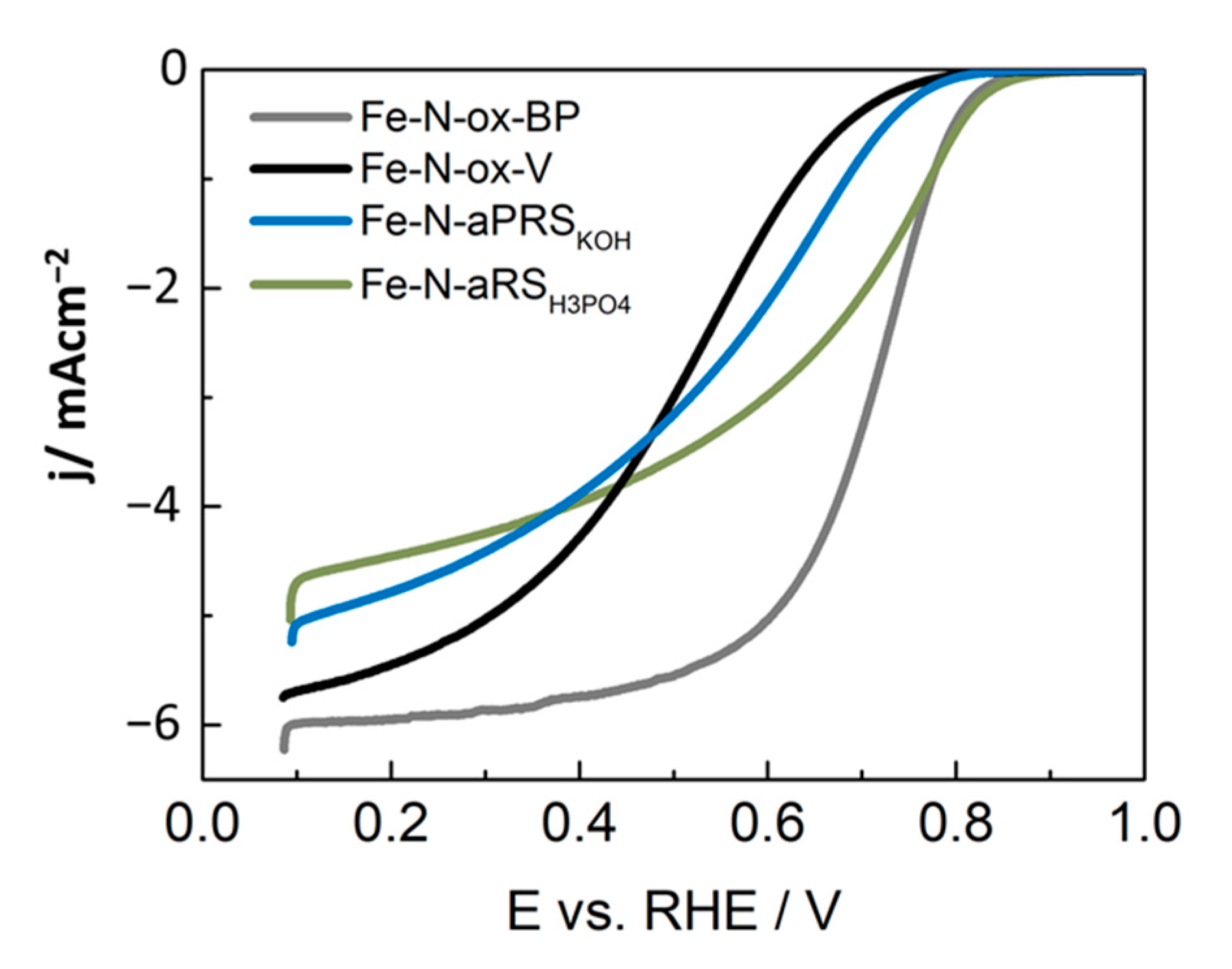
| ORR activity of Fe-N-C@ 0.80 VRHE (LSV, Figure 9) | Improved (1.27 A/g) | Low (0.13 A/g) | Low (0.19 A/g) | Improved (1.52 A/g) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hülstede, J.; Schonvogel, D.; Schmies, H.; Wagner, P.; Schröter, F.; Dyck, A.; Wark, M. Relevant Properties of Carbon Support Materials in Successful Fe-N-C Synthesis for the Oxygen Reduction Reaction: Study of Carbon Blacks and Biomass-Based Carbons. Materials 2021, 14, 45. https://doi.org/10.3390/ma14010045
Hülstede J, Schonvogel D, Schmies H, Wagner P, Schröter F, Dyck A, Wark M. Relevant Properties of Carbon Support Materials in Successful Fe-N-C Synthesis for the Oxygen Reduction Reaction: Study of Carbon Blacks and Biomass-Based Carbons. Materials. 2021; 14(1):45. https://doi.org/10.3390/ma14010045
Chicago/Turabian StyleHülstede, Julia, Dana Schonvogel, Henrike Schmies, Peter Wagner, Frank Schröter, Alexander Dyck, and Michael Wark. 2021. "Relevant Properties of Carbon Support Materials in Successful Fe-N-C Synthesis for the Oxygen Reduction Reaction: Study of Carbon Blacks and Biomass-Based Carbons" Materials 14, no. 1: 45. https://doi.org/10.3390/ma14010045
APA StyleHülstede, J., Schonvogel, D., Schmies, H., Wagner, P., Schröter, F., Dyck, A., & Wark, M. (2021). Relevant Properties of Carbon Support Materials in Successful Fe-N-C Synthesis for the Oxygen Reduction Reaction: Study of Carbon Blacks and Biomass-Based Carbons. Materials, 14(1), 45. https://doi.org/10.3390/ma14010045







