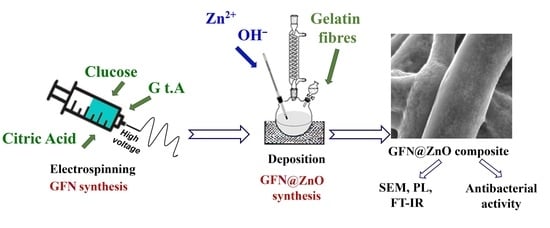
Fabrication of Gelatin-ZnO Nanofibers for Antibacterial Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Gelatin Fibers
2.3. GNF@ZnO Composite Synthesis
2.4. Characterization
2.5. Antibacterial Activity of the Obtained GNF@ZnO Composites
3. Results
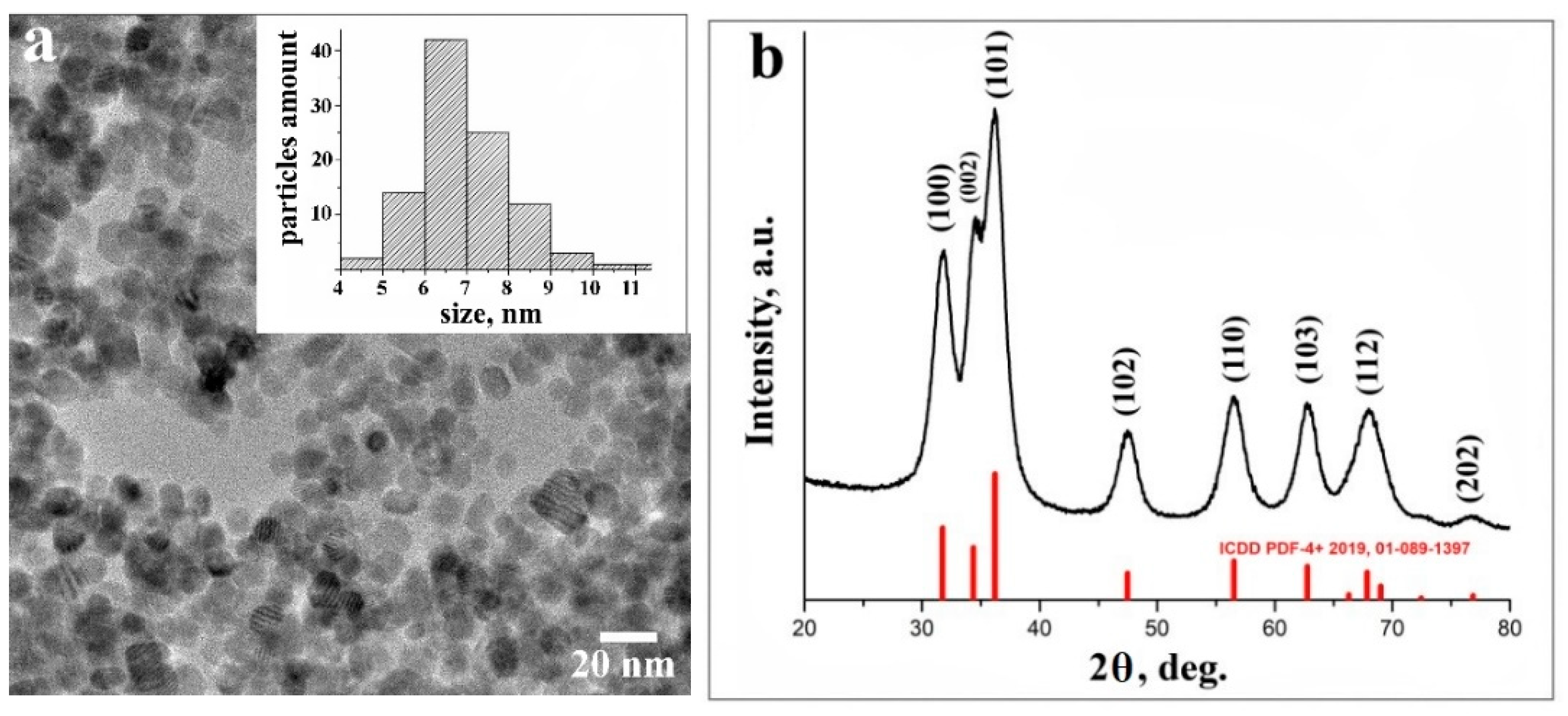
3.1. Characterization of ZnO NPs
3.1.1. TEM and XRD Analyses
3.1.2. Textural Properties
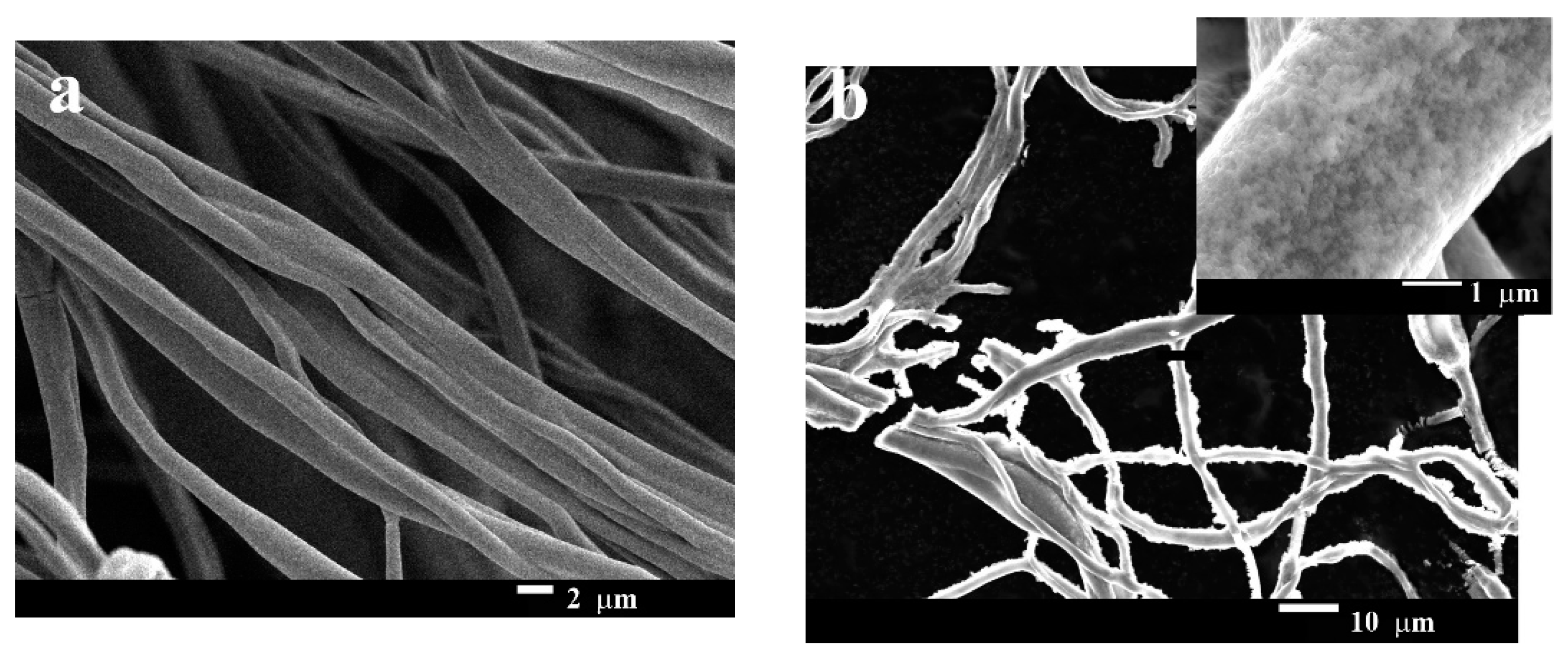
3.2. Characterization of GNF@ZnO Composites
3.2.1. Morphology of the Gelatin Nanofibers and GNF@ZnO Composites
3.2.2. Elemental Composition of Studied GNF@ZnO Composites
3.2.3. Thermal Analysis of GNF@ZnO Composites
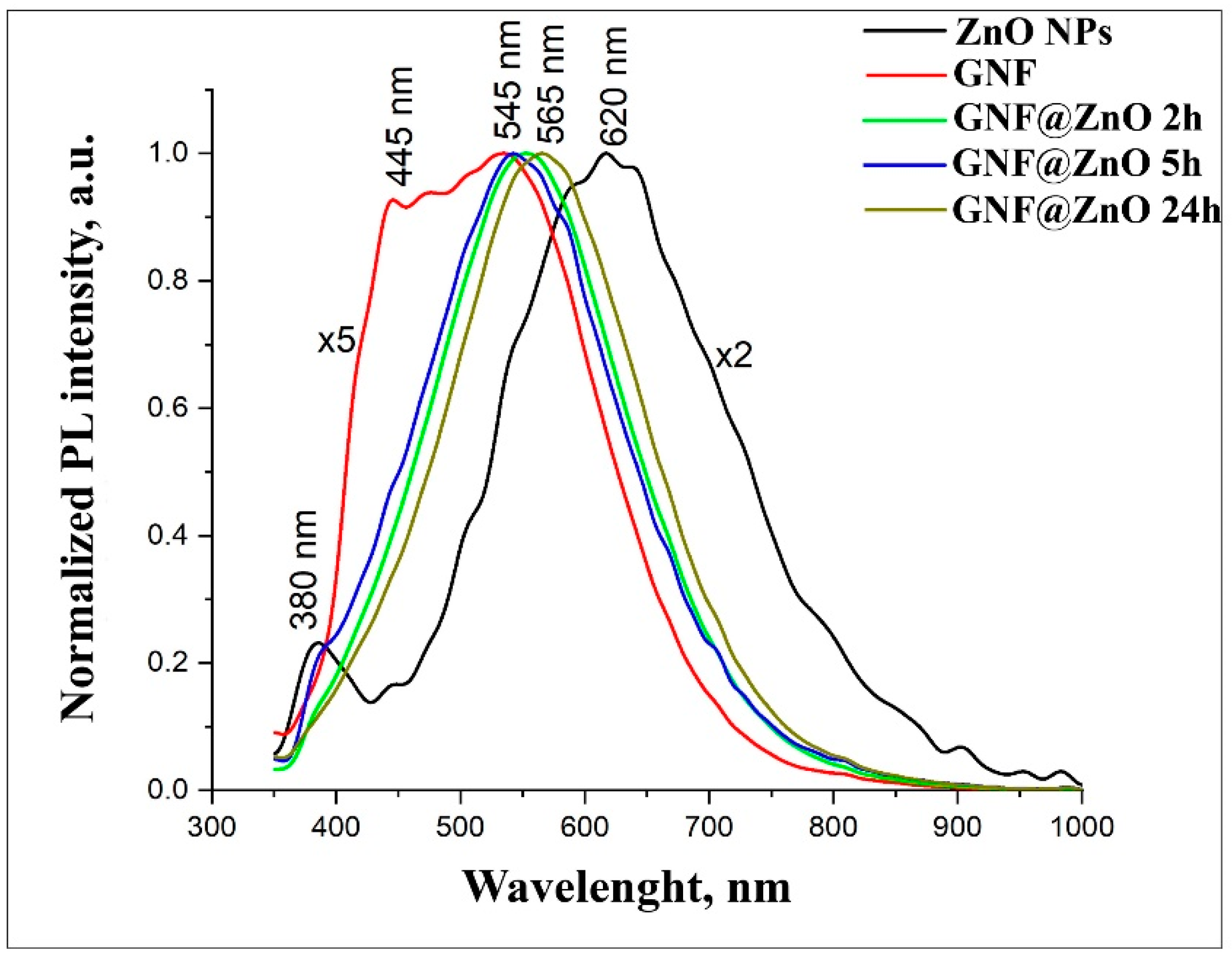
3.2.4. Photoluminescence Properties of GNF@ZnO Composites
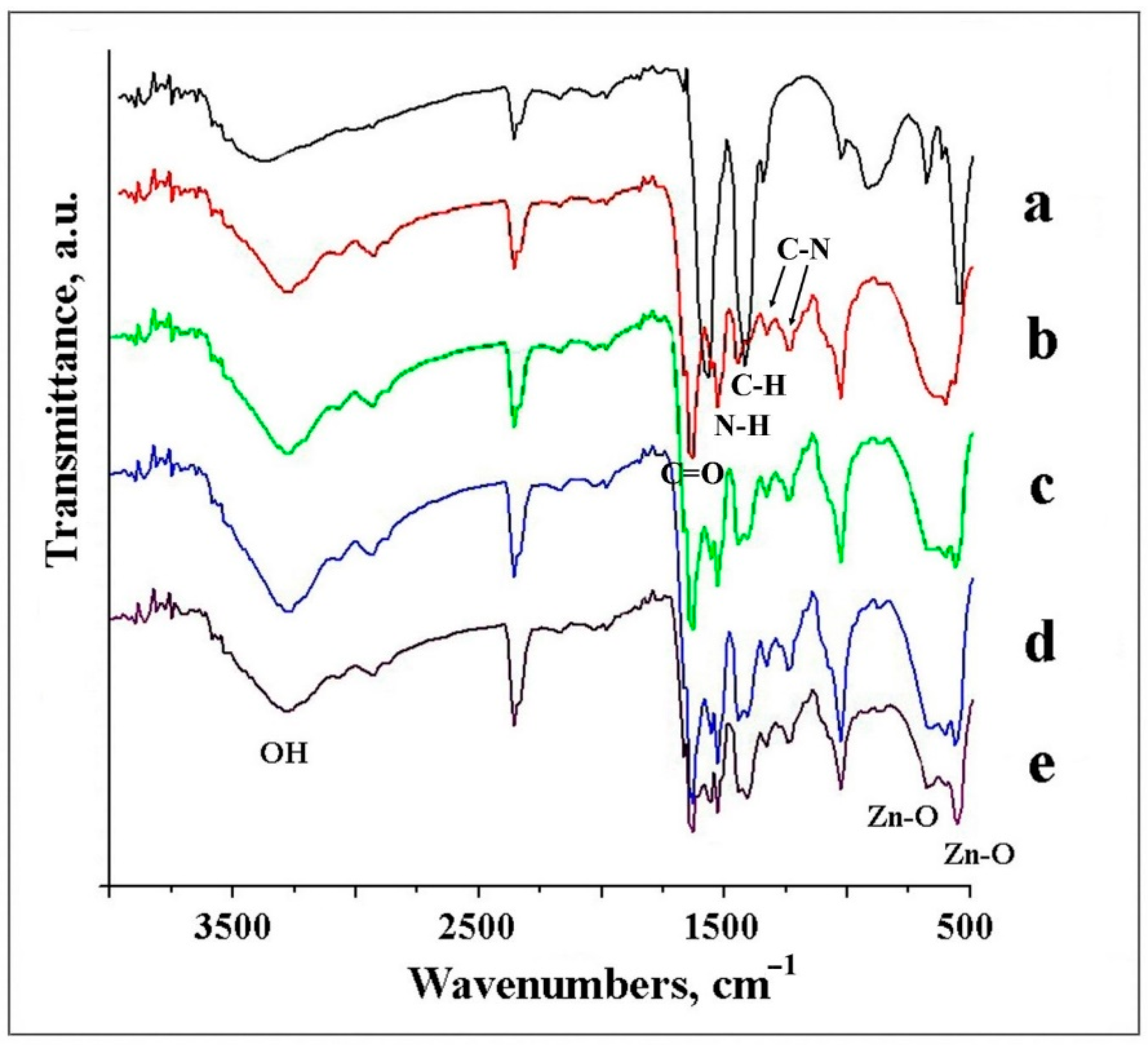
3.2.5. FTIR Analysis of Studied Samples
3.2.6. Antibacterial Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khranovskyy, V.; Yakimova, R.; Karlsson, F.; Syed, A.S.; Holtz, P.-O.; Nigussa Urgessa, Z.; Samuel Oluwafemi, O.; Reinhardt Botha, J. Comparative PL study of individual ZnO nanorods, grown by APMOCVD and CBD techniques. Phys. B Condens. Matter 2012, 407, 1538–1542. [Google Scholar] [CrossRef]
- Zheng, H.; Jiang, Y.; Yang, S.; Zhang, Y.; Yan, X.; Hu, J.; Shi, Y.; Zou, B. ZnO nanorods array as light absorption antenna for high-gain UV photodetectors. J. Alloy. Compd. 2020, 812, 152158. [Google Scholar] [CrossRef]
- Graniel, O.; Iatsunskyi, I.; Coy, E.; Humbert, C.; Barbillon, G.; Michel, T.; Maurin, D.; Balme, S.; Miele, P.; Bechelany, M. Au-covered hollow urchin-like ZnO nanostructures for surface-enhanced Raman scattering sensing. J. Mater. Chem. C 2019, 7, 15066–15073. [Google Scholar] [CrossRef]
- Viter, R.; Savchuk, M.; Starodub, N.; Balevicius, Z.; Tumenas, S.; Ramanaviciene, A.; Jevdokimovs, D.; Erts, D.; Iatsunskyi, I.; Ramanavicius, A. Photoluminescence immunosensor based on bovine leukemia virus proteins immobilized on the ZnO nanorods. Sens. Actuators B Chem. 2019, 285, 601–606. [Google Scholar] [CrossRef]
- Myndrul, V.; Coy, E.; Bechelany, M.; Iatsunskyi, I. Photoluminescence label-free immunosensor for the detection of Aflatoxin B1 using polyacrylonitrile/zinc oxide nanofibers. Mater. Sci. Eng. C 2021, 118, 111401. [Google Scholar] [CrossRef]
- Tamashevski, A.; Harmaza, Y.; Slobozhanina, E.; Viter, R.; Iatsunskyi, I. Photoluminescent Detection of Human T-Lymphoblastic Cells by ZnO Nanorods. Molecules 2020, 25, 3168. [Google Scholar] [CrossRef]
- Babayevska, N.; Peplińska, B.; Jarek, M.; Yate, L.; Tadyszak, K.; Gapiński, J.; Iatsunskyi, I.; Jurga, S. Synthesis, structure, EPR studies and up-conversion luminescence of ZnO:Er3+–Yb3+ @Gd2O3 nanostructures. Rsc Adv. 2016, 6, 89305–89312. [Google Scholar] [CrossRef]
- Silina, E.Y.; Gernaey, V.K.; Semenova, D.; Iatsunskyi, I. Application of Organic-Inorganic Hybrids in Chemical Analysis, Bio- and Environmental Monitoring. Appl. Sci. 2020, 10, 1458. [Google Scholar] [CrossRef]
- Baranowska-Korczyc, A.; Kościński, M.; Coy, E.L.; Grześkowiak, B.F.; Jasiurkowska-Delaporte, M.; Peplińska, B.; Jurga, S. ZnS coating for enhanced environmental stability and improved properties of ZnO thin films. Rsc Adv. 2018, 8, 24411–24421. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Nagarajan, S.; Belaid, H.; Farha, C.; Iatsunskyi, I.; Coy, E.; Soussan, L.; Huon, V.; Bares, J.; Belkacemi, K.; et al. Fabrication of 3D printed antimicrobial polycaprolactone scaffolds for tissue engineering applications. Mater. Sci. Eng. C 2021, 118, 111525. [Google Scholar] [CrossRef]
- Damberga, D.; Fedorenko, V.; Grundšteins, K.; Altundal, Ş.; Šutka, A.; Ramanavičius, A.; Coy, E.; Mrówczyński, R.; Iatsunskyi, I.; Viter, R. Influence of PDA Coating on the Structural, Optical and Surface Properties of ZnO Nanostructures. Nanomaterials 2020, 10, 2438. [Google Scholar] [CrossRef] [PubMed]
- Rychter, M.; Baranowska-Korczyc, A.; Milanowski, B.; Jarek, M.; Maciejewska, B.M.; Coy, E.L.; Lulek, J. Cilostazol-Loaded Poly(ε-Caprolactone) Electrospun Drug Delivery System for Cardiovascular Applications. Pharm. Res. 2018, 35, 32. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Liu, M.; Zhang, Q.; Huang, Q.; Huang, H.; Tian, J.; Jiang, R.; Wen, Y.; Zhang, X.; Wei, Y. Preparation of water dispersible and biocompatible nanodiamond-poly(amino acid) composites through the ring-opening polymerization. Mater. Sci. Eng. C 2018, 91, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Beek, W.J.E.; Wienk, M.M.; Kemerink, M.; Yang, X.; Janssen, R.A.J. Hybrid Zinc Oxide Conjugated Polymer Bulk Heterojunction Solar Cells. J. Phys. Chem. B 2005, 109, 9505–9516. [Google Scholar] [CrossRef]
- Inbasekar, C.; Fathima, N.N. Collagen stabilization using ionic liquid functionalised cerium oxide nanoparticle. Int. J. Biol. Macromol. 2020, 147, 24–28. [Google Scholar] [CrossRef]
- Nagarajan, S.; Belaid, H.; Pochat-Bohatier, C.; Teyssier, C.; Iatsunskyi, I.; Coy, E.; Balme, S.; Cornu, D.; Miele, P.; Kalkura, N.S.; et al. Design of Boron Nitride/Gelatin Electrospun Nanofibers for Bone Tissue Engineering. Acs Appl. Mater. Interfaces 2017, 9, 33695–33706. [Google Scholar] [CrossRef]
- Li, M.; Mondrinos, M.J.; Gandhi, M.R.; Ko, F.K.; Weiss, A.S.; Lelkes, P.I. Electrospun protein fibers as matrices for tissue engineering. Biomaterials 2005, 26, 5999–6008. [Google Scholar] [CrossRef]
- Azizi-Lalabadi, M.; Ehsani, A.; Ghanbarzadeh, B.; Divband, B. Polyvinyl alcohol/gelatin nanocomposite containing ZnO, TiO2 or ZnO/TiO2 nanoparticles doped on 4A zeolite: Microbial and sensory qualities of packaged white shrimp during refrigeration. Int. J. Food Microbiol. 2020, 312, 108375. [Google Scholar] [CrossRef]
- Bigi, A.; Cojazzi, G.; Panzavolta, S.; Roveri, N.; Rubini, K. Stabilization of gelatin films by crosslinking with genipin. Biomaterials 2002, 23, 4827–4832. [Google Scholar] [CrossRef]
- Nourbakhsh, S.; Talebian, A.; Faramarzi, S. Preparation and Characterization of Gelatin/ZnO Nano-Composite Film. Mater. Today Proc. 2017, 4, 7038–7043. [Google Scholar] [CrossRef]
- Zhang, Y.; Nayak, T.; Hong, H.; Cai, W. Biomedical Applications of Zinc Oxide Nanomaterials. Curr. Mol. Med. 2013, 13, 1633–1645. [Google Scholar] [CrossRef] [PubMed]
- Siimon, K.; Reemann, P.; Põder, A.; Pook, M.; Kangur, T.; Kingo, K.; Jaks, V.; Mäeorg, U.; Järvekülg, M. Effect of glucose content on thermally cross-linked fibrous gelatin scaffolds for tissue engineering. Mater. Sci. Eng. C 2014, 42, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Woźniak-Budych, M.J.; Przysiecka, Ł.; Langer, K.; Peplińska, B.; Jarek, M.; Wiesner, M.; Nowaczyk, G.; Jurga, S. Green synthesis of rifampicin-loaded copper nanoparticles with enhanced antimicrobial activity. J. Mater. Sci. Mater. Med. 2017, 28, 42. [Google Scholar] [CrossRef] [PubMed]
- Borkowski, A.; Ławniczak, Ł.; Cłapa, T.; Narożna, D.; Selwet, M.; Pęziak, D.; Markiewicz, B.; Chrzanowski, Ł. Different antibacterial activity of novel theophylline-based ionic liquids—Growth kinetic and cytotoxicity studies. Ecotoxicol. Env. Saf. 2016, 130, 54–64. [Google Scholar] [CrossRef]
- Koch, S.; Späth, S.; Shmeliov, A.; Nicolosi, V.; Mandel, K. Air bubble promoted large scale synthesis of luminescent ZnO nanoparticles. J. Mater. Chem. C 2015, 3, 12430–12435. [Google Scholar] [CrossRef]
- An, Z.; Xue, J.; Cao, H.; Zhu, C.; Wang, H. A facile synthesis of silicon carbide nanoparticles with high specific surface area by using corn cob. Adv. Powder Technol. 2019, 30, 164–169. [Google Scholar] [CrossRef]
- Bakhshian Nik, A.; Zare, H.; Razavi, S.; Mohammadi, H.; Torab Ahmadi, P.; Yazdani, N.; Bayandori, M.; Rabiee, N.; Izadi Mobarakeh, J. Smart drug delivery: Capping strategies for mesoporous silica nanoparticles. Microporous Mesoporous Mater. 2020, 299, 110115. [Google Scholar] [CrossRef]
- Majid, S.; Ahmad, K.S.; Sharif, S. Optical and structural properties of single source precursor based pure and Cu-doped antimony sulphide thin films by physical vapour deposition assisted technique. Chem. Phys. 2020, 539, 110979. [Google Scholar] [CrossRef]
- Damberga, D.; Viter, R.; Fedorenko, V.; Iatsunskyi, I.; Coy, E.; Graniel, O.; Balme, S.; Miele, P.; Bechelany, M. Photoluminescence Study of Defects in ZnO-Coated Polyacrylonitrile Nanofibers. J. Phys. Chem. C 2020, 124, 9434–9441. [Google Scholar] [CrossRef]
- Fedorenko, V.; Viter, R.; Mrówczyński, R.; Damberga, D.; Coy, E.; Iatsunskyi, I. Synthesis and photoluminescence properties of hybrid 1D core–shell structured nanocomposites based on ZnO/polydopamine. Rsc Adv. 2020, 10, 29751–29758. [Google Scholar] [CrossRef]
- Myndrul, V.; Vysloužilová, L.; Klápšt’ová, A.; Coy, E.; Jancelewicz, M.; Iatsunskyi, I. Formation and Photoluminescence Properties of ZnO Nanoparticles on Electrospun Nanofibers Produced by Atomic Layer Deposition. Coatings 2020, 10, 1199. [Google Scholar] [CrossRef]
- Viter, R.; Abou Chaaya, A.; Iatsunskyi, I.; Nowaczyk, G.; Kovalevskis, K.; Erts, D.; Miele, P.; Smyntyna, V.; Bechelany, M. Tuning of ZnO 1D nanostructures by atomic layer deposition and electrospinning for optical gas sensor applications. Nanotechnology 2015, 26, 105501. [Google Scholar] [CrossRef] [PubMed]
- Azhniuk, Y.M.; Dzhagan, V.M.; Raevskaya, A.E.; Stroyuk, A.L.; Kuchmiy, S.Y.; Valakh, M.Y.; Zahn, D.R.T. Optical studies of CdSe/HgSe and CdSe/Ag2Se core/shell nanoparticles embedded in gelatin. J. Phys. Condens. Matter 2008, 20, 455203. [Google Scholar] [CrossRef]
- Li, X.; Chen, S.; Li, J.; Wang, X.; Zhang, J.; Kawazoe, N.; Chen, G. 3D Culture of Chondrocytes in Gelatin Hydrogels with Different Stiffness. Polymers 2016, 8, 269. [Google Scholar] [CrossRef] [PubMed]
- Babayevska, N.; Florczak, P.; Woźniak-Budych, M.; Jarek, M.; Nowaczyk, G.; Zalewski, T.; Jurga, S. Functionalized multimodal ZnO@Gd 2 O 3 nanosystems to use as perspective contrast agent for MRI. Appl. Surf. Sci. 2017, 404, 129–137. [Google Scholar] [CrossRef]
- Zhuang, C.; Tao, F.; Cui, Y. Anti-degradation gelatin films crosslinked by active ester based on cellulose. Rsc Adv. 2015, 5, 52183–52193. [Google Scholar] [CrossRef]
- Amjadi, S.; Emaminia, S.; Heyat Davudian, S.; Pourmohammad, S.; Hamishehkar, H.; Roufegarinejad, L. Preparation and characterization of gelatin-based nanocomposite containing chitosan nanofiber and ZnO nanoparticles. Carbohydr. Polym. 2019, 216, 376–384. [Google Scholar] [CrossRef]
- Narayanan, P.M.; Wilson, W.S.; Abraham, A.T.; Sevanan, M. Synthesis, Characterization, and Antimicrobial Activity of Zinc Oxide Nanoparticles Against Human Pathogens. Bionanoscience 2012, 2, 329–335. [Google Scholar] [CrossRef]
- Loo, Y.Y.; Rukayadi, Y.; Nor-Khaizura, M.-A.-R.; Kuan, C.H.; Chieng, B.W.; Nishibuchi, M.; Radu, S. In Vitro Antimicrobial Activity of Green Synthesized Silver Nanoparticles Against Selected Gram-negative Foodborne Pathogens. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Potekhina, N.V.; Streshinskaya, G.M.; Tul’skaya, E.M.; Kozlova, Y.I.; Senchenkova, S.N.; Shashkov, A.S. Phosphate-containing cell wall polymers of bacilli. Biochemistry 2011, 76, 745–754. [Google Scholar] [CrossRef]
- Thatoi, H.; Das, S.; Mishra, J.; Rath, B.P.; Das, N. Bacterial chromate reductase, a potential enzyme for bioremediation of hexavalent chromium: A review. J. Env. Manag. 2014, 146, 383–399. [Google Scholar] [CrossRef] [PubMed]
- Ramstedt, M.; Leone, L.; Persson, P.; Shchukarev, A. Cell Wall Composition of Bacillus subtilis Changes as a Function of pH and Zn2+ Exposure: Insights from Cryo-XPS Measurements. Langmuir 2014, 30, 4367–4374. [Google Scholar] [CrossRef] [PubMed]
- Karton-Lifshin, N.; Segal, E.; Omer, L.; Portnoy, M.; Satchi-Fainaro, R.; Shabat, D. A Unique Paradigm for a Turn-ON Near-Infrared Cyanine-Based Probe: Noninvasive Intravital Optical Imaging of Hydrogen Peroxide. J. Am. Chem. Soc. 2011, 133, 10960–10965. [Google Scholar] [CrossRef] [PubMed]
- Thanusha, A.V.; Dinda, A.K.; Koul, V. Evaluation of nano hydrogel composite based on gelatin/HA/CS suffused with Asiatic acid/ZnO and CuO nanoparticles for second degree burns. Mater. Sci. Eng. C 2018, 89, 378–386. [Google Scholar]
- Azizi-Lalabadi, M.; Ehsani, A.; Divband, B.; Alizadeh-Sani, M. Antimicrobial activity of Titanium dioxide and Zinc oxide nanoparticles supported in 4A zeolite and evaluation the morphological characteristic. Sci. Rep. 2019, 9, 17439. [Google Scholar] [CrossRef]
- Surendra, T.V.; Roopan, S.M.; Al-Dhabi, N.A.; Arasu, M.V.; Sarkar, G.; Suthindhiran, K. Vegetable Peel Waste for the Production of ZnO Nanoparticles and its Toxicological Efficiency, Antifungal, Hemolytic, and Antibacterial Activities. Nanoscale Res. Lett. 2016, 11, 546. [Google Scholar] [CrossRef]


| Composite | GNF@ZnO 2 h, Atomic, % | GNF@ZnO 5 h, Atomic, % | GNF@ZnO 24 h, Atomic, % | |
|---|---|---|---|---|
| Elements | ||||
| C | 53.01 | 50.04 | 27.11 | |
| O | 31.03 | 33.95 | 32.34 | |
| Zn | 15.87 | 16.01 | 40.55 | |
| total | 100.00 | 100.00 | 100.00 | |
| Sample | 1st Weight Loss, % | 2nd and 3th WEIGHT Loss, % | Content of ZnO a, wt.% | Content of ZnO b, wt.% |
|---|---|---|---|---|
| GNF@ZnO 2 h | 7.1 | 55.1 | 37.8 | 40.7 |
| GNF@ZnO 5 h | 7.0 | 51.7 | 41.3 | 44.4 |
| GNF@ZnO 24 h | 4.4 | 32.2 | 63.4 | 66.3 |
| Samples | S. aureus (G+) | B. pumilus (G+) | E. coli (G−) | P. fluorescens (G−) |
|---|---|---|---|---|
| ZnO NPs x | 19 ± 0.82 | 16 ± 0.32 | 11.3 ± 0.47 | 17.7 ± 0.47 |
| GNF@ZnO 2h x | 10 ± 0.63 | 0 | 9.2 ± 0.75 | 10 ± 0 |
| GNF@ZnO 5h x | 7.8 ± 0.4 | 0 | 8.9 ± 0.49 | 11.1 ± 0.8 |
| GNF@ZnO 24h x | 11 ± 0 | 0 | 0 | 9.8 ± 0.75 |
| GNF x | 0 | 0 | 0 | 0 |
| gelatin + ZnO * | 4.9 ± 0.6 | 5.3 ± 0.2 | ||
| G.ZnO NP ** G.CHNF.ZnO NPs ** | 30.62 ± 0.56 33.13 ± 0.67 | 15.06 ± 0.17 25.06 ± 0.24 | ||
| ZnO/4A z *** | 6.21 ± 0.02 | 6.86 ± 0.03 | 6.34 ± 0.03 | |
| ZnO NPs (~50 nm) **** | 6.0 | 6.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Babayevska, N.; Przysiecka, Ł.; Nowaczyk, G.; Jarek, M.; Järvekülg, M.; Kangur, T.; Janiszewska, E.; Jurga, S.; Iatsunskyi, I. Fabrication of Gelatin-ZnO Nanofibers for Antibacterial Applications. Materials 2021, 14, 103. https://doi.org/10.3390/ma14010103
Babayevska N, Przysiecka Ł, Nowaczyk G, Jarek M, Järvekülg M, Kangur T, Janiszewska E, Jurga S, Iatsunskyi I. Fabrication of Gelatin-ZnO Nanofibers for Antibacterial Applications. Materials. 2021; 14(1):103. https://doi.org/10.3390/ma14010103
Chicago/Turabian StyleBabayevska, Nataliya, Łucja Przysiecka, Grzegorz Nowaczyk, Marcin Jarek, Martin Järvekülg, Triin Kangur, Ewa Janiszewska, Stefan Jurga, and Igor Iatsunskyi. 2021. "Fabrication of Gelatin-ZnO Nanofibers for Antibacterial Applications" Materials 14, no. 1: 103. https://doi.org/10.3390/ma14010103
APA StyleBabayevska, N., Przysiecka, Ł., Nowaczyk, G., Jarek, M., Järvekülg, M., Kangur, T., Janiszewska, E., Jurga, S., & Iatsunskyi, I. (2021). Fabrication of Gelatin-ZnO Nanofibers for Antibacterial Applications. Materials, 14(1), 103. https://doi.org/10.3390/ma14010103