Recent Advances on the Application of Layered Double Hydroxides in Concrete—A Review
Abstract
1. Introduction
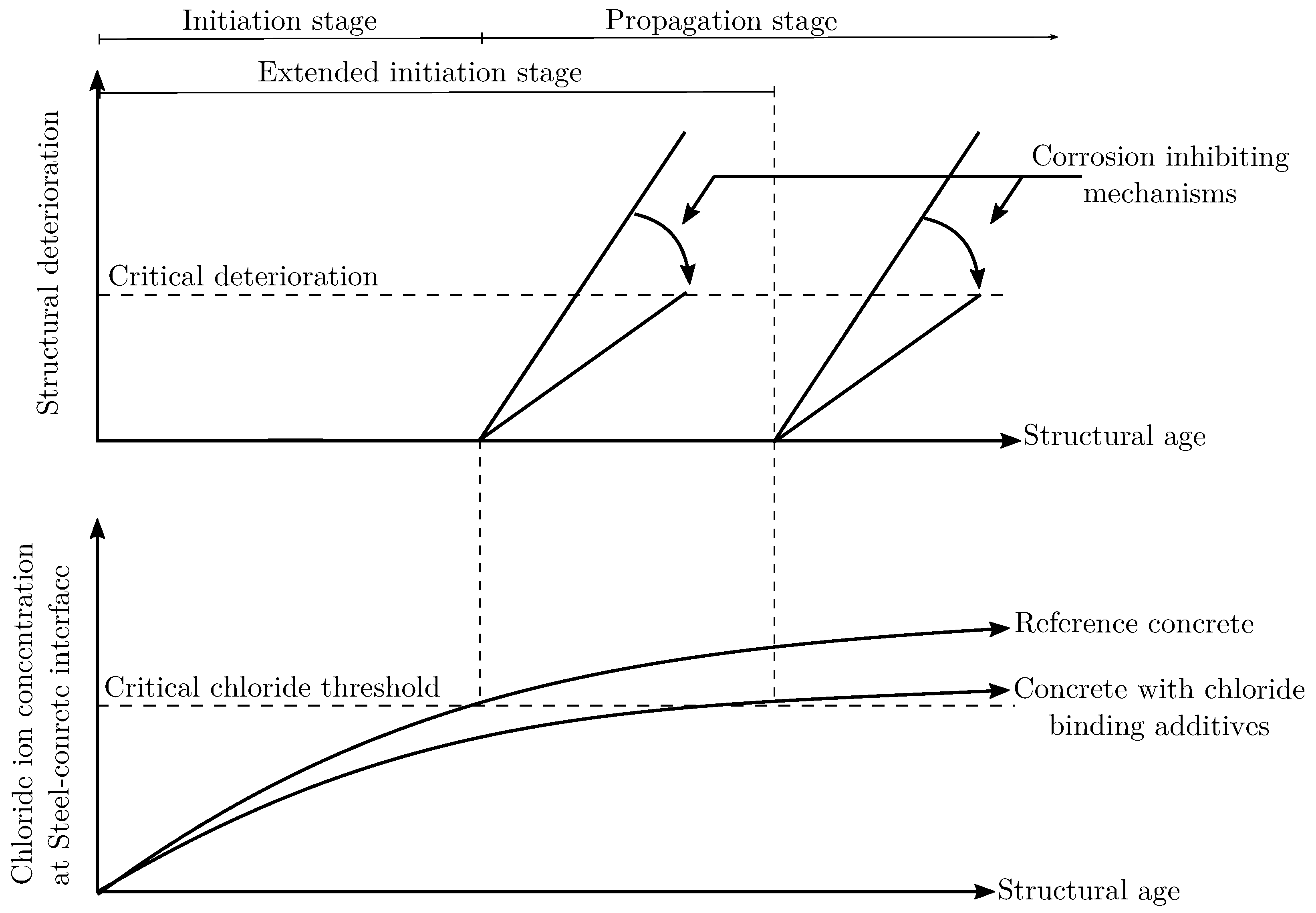
2. Chloride Induced Steel Corrosion in Concrete
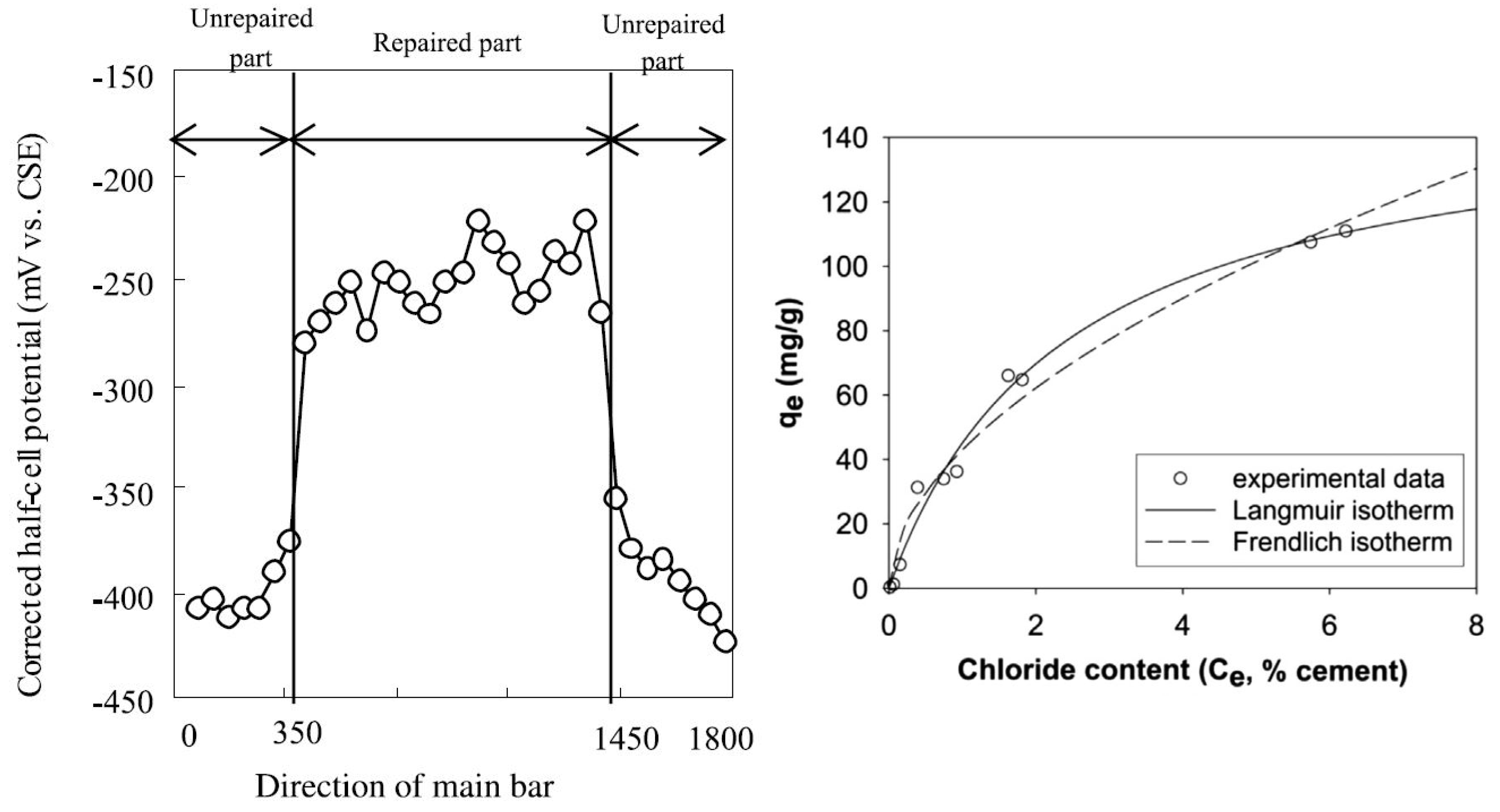
3. Chloride Binding in Concrete
4. Layered Double Hydroxides
4.1. Preparation Methods
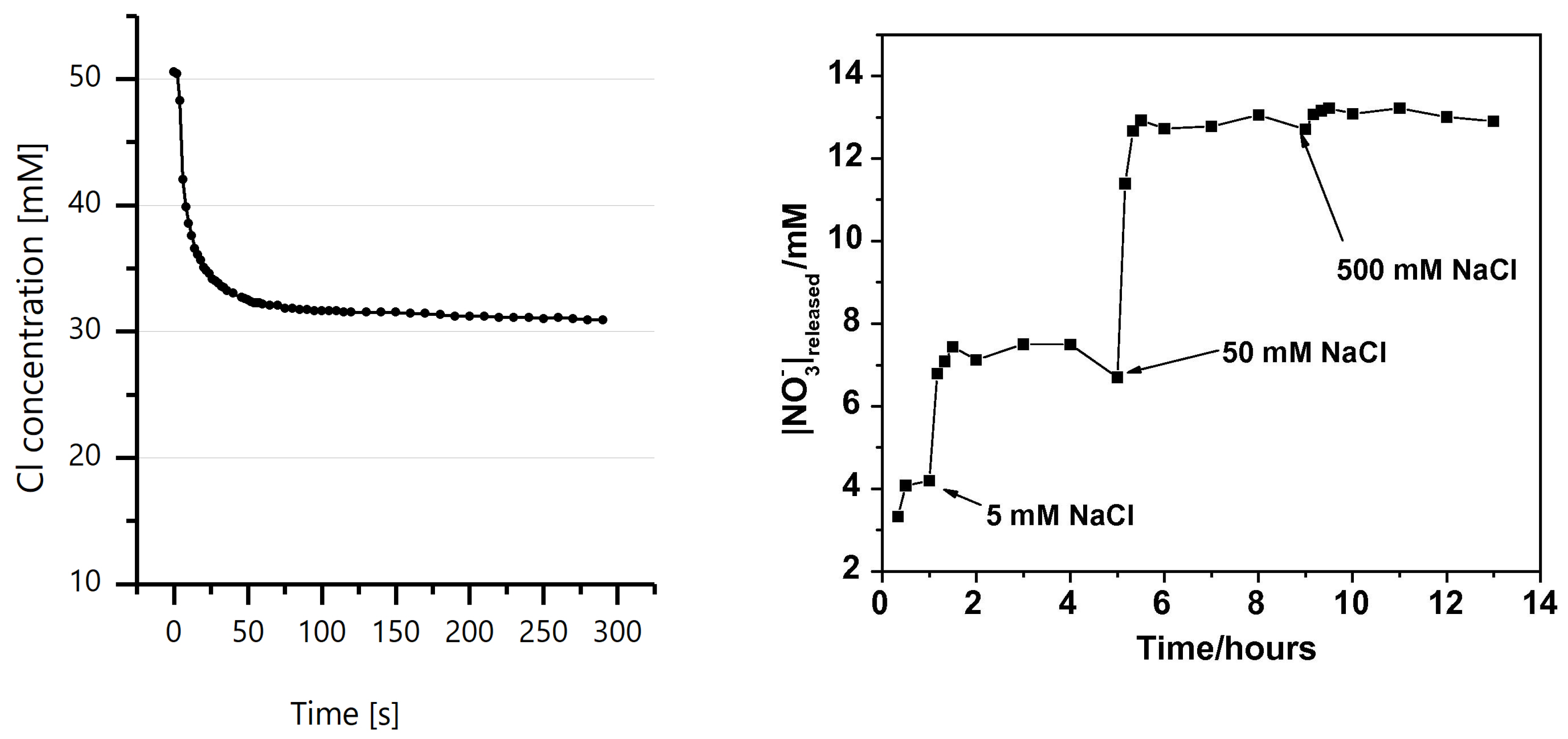
4.2. Ion Exchange Property Of LDH
5. ompatibility of Ldh with Cementitious Environments
5.1. Effect on Mechanical Properties
5.2. Effect of LDH Dosage on Microstructure
5.3. Effect ON Hydration
6. Ion Exchange Property and Self Protection of Concrete
6.1. Chloride Entrapment and Corrosion Performance
6.2. Role of Ldh in Carbonation Control
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| LDH | Layered Double Hydroxide |
| CSE | Copper sulphate electrode |
| pAB | P-aminobenzoate |
| CA | tricalcium aluminate |
| CAF | tetracalcium alumino-ferrite |
| AFm | aluminate monosulphate |
| CSH | calcium silicate hydrate |
References
- DESA; UN. World Population Prospects 2019: Highlights; United Nations Department for Economic and Social Affairs: New York, NY, USA, 2019. [Google Scholar]
- Mehta, P.K. Greening of the concrete industry for sustainable development. Concr. Int. 2002, 24, 23–28. [Google Scholar]
- Huntzinger, D.N.; Eatmon, T.D. A life-cycle assessment of Portland cement manufacturing: Comparing the traditional process with alternative technologies. J. Clean. Prod. 2009, 17, 668–675. [Google Scholar] [CrossRef]
- Uwasu, M.; Hara, K.; Yabar, H. World cement production and environmental implications. Environ. Dev. 2014, 10, 36–47. [Google Scholar] [CrossRef]
- Kim, Y.; Worrell, E. CO2 emission trends in the cement industry: An international comparison. Mitig. Adapt. Strateg. Glob. Chang. 2002, 7, 115–133. [Google Scholar] [CrossRef]
- Chen, C.; Habert, G.; Bouzidi, Y.; Jullien, A. Environmental impact of cement production: Detail of the different processes and cement plant variability evaluation. J. Clean. Prod. 2010, 18, 478–485. [Google Scholar] [CrossRef]
- Tuutti, K. Corrosion of Steel in Concrete—(Technical Report); Cement-och Betonginst CBI Sweden: Stockholm, Sweden, 1982. [Google Scholar]
- Bertolini, L.; Elsener, B.; Pedeferri, P.; Redaelli, E.; Polder, R. Corrosion of Steel in Concrete; Wiley-Vch.: Weinheim, Germany, 2013; Volume 392. [Google Scholar]
- Broomfield, J.P. Corrosion of Steel in Concrete: Understanding, Investigation and Repair; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Poursaee, A. Corrosion of steel in concrete structures. In Corrosion of Steel in Concrete Structures; Elsevier: Amsterdam, The Netherlands, 2016; pp. 19–33. [Google Scholar]
- Raupach, M.; Elsener, B.; Polder, R.; Mietz, J. Corrosion of Reinforcement in Concrete: Monitoring, Prevention and Rehabilitation Techniques; Woodhead Publishing: Sawston, UK, 2014; Volume 38. [Google Scholar]
- Lewis, D.; Copenhagen, W. Corrosion of reinforcing steel in concrete in marine atmospheres. Corrosion 1959, 15, 60–66. [Google Scholar] [CrossRef]
- Hausmann, D. Steel corrosion in concrete–How does it occur? Mater. Prot. 1967, 6, 19–23. [Google Scholar]
- Page, C. Mechanism of corrosion protection in reinforced concrete marine structures. Nature 1975, 258, 514. [Google Scholar] [CrossRef]
- Cai, H.; Liu, X. Freeze-thaw durability of concrete: Ice formation process in pores. Cem. Concr. Res. 1998, 28, 1281–1287. [Google Scholar] [CrossRef]
- Berto, L.; Saetta, A.; Talledo, D. Constitutive model of concrete damaged by freeze–thaw action for evaluation of structural performance of RC elements. Constr. Build. Mater. 2015, 98, 559–569. [Google Scholar] [CrossRef]
- Huovinen, S. Abrasion of Concrete by Ice in Arctic Sea Structures; Technical Research Centre of Finland: Espoo, Finland, 1990. [Google Scholar]
- Jacobsen, S.; Scherer, G.W.; Schulson, E.M. Concrete–ice abrasion mechanics. Cem. Concr. Res. 2015, 73, 79–95. [Google Scholar] [CrossRef]
- Ryan, A. Ice Wear and Abrasion of Marine Concrete: Design of Experimental Apparatus and Procedures. Ph.D. Thesis, Memorial University of Newfoundland, St. John’s, NL, Canada, 2018. [Google Scholar]
- Marchand, J.; Odler, I.; Skalny, J.P. Sulfate Attack on Concrete; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Attiogbe, E.K.; Rizkalla, S.H. Response of concrete to sulfuric acid attack. ACI Mater. J. 1988, 85, 481–488. [Google Scholar]
- Glass, G.; Buenfeld, N. Chloride-induced corrosion of steel in concrete. Prog. Struct. Eng. Mater. 2000, 2, 448–458. [Google Scholar] [CrossRef]
- Angst, U.M. Challenges and opportunities in corrosion of steel in concrete. Mater. Struct. 2018, 51, 4. [Google Scholar] [CrossRef]
- Gomes, C.; Mir, Z.; Sampaio, R.S.; Bastos, A.; Maia, F.; Rocha, C.; Tedim, J.; Ferreira, M. Effect of Layered Double Hydroxides on the Performance and Service Life of Reinforced Concrete. In Durable Concrete for Infrastructure under Severe Conditions Smart Admixtures, Self-Responsiveness and Nano-Additions Proceedings 10–11 September 2019; Ghent. Ghent University: Ghent, Belgium, 2019; pp. 51–54. [Google Scholar]
- Gomes, C.; Mir, Z.; Sampaio, R.S.; Bastos, A.; Tedim, J.; Maia, F.; Rocha, C.; Ferreira, M. On the use of ZnAl layered double hydroxide (LDH) to extend the service life of reinforced concrete. Front. Mater. in review.
- Raki, L.; Beaudoin, J.; Mitchell, L. Layered double hydroxide-like materials: Nanocomposites for use in concrete. Cem. Concr. Res. 2004, 34, 1717–1724. [Google Scholar] [CrossRef]
- Yang, Z.; Fischer, H.; Polder, R. Modified hydrotalcites as a new emerging class of smart additive of reinforced concrete for anticorrosion applications: A literature review. Mater. Corros. 2013, 64, 1066–1074. [Google Scholar] [CrossRef]
- Yang, Z.; Fischer, H.; Polder, R. Possibilities for improving corrosion protection of reinforced concrete by modified hydrotalcites–a literature review. In Advances in Modeling Concrete Service Life; RILEM Bookseries vol 3; Springer: Dordrecht, The Netherlands, 2012; pp. 95–105. [Google Scholar]
- USGS. Cement: 2007 Minerals Yearbook—US. Geological Survey; USGS: Reston, VA, USA, 2010.
- Gagg, C.R. Cement and concrete as an engineering material: An historic appraisal and case study analysis. Eng. Fail. Anal. 2014, 40, 114–140. [Google Scholar] [CrossRef]
- Poursaee, A.; Hansson, C. Reinforcing steel passivation in mortar and pore solution. Cem. Concr. Res. 2007, 37, 1127–1133. [Google Scholar] [CrossRef]
- Ghods, P.; Isgor, O.; McRae, G.; Li, J.; Gu, G. Microscopic investigation of mill scale and its proposed effect on the variability of chloride-induced depassivation of carbon steel rebar. Corros. Sci. 2011, 53, 946–954. [Google Scholar] [CrossRef]
- Andrade, C.; Merino, P.; Novoa, X.; Perez, M.; Soler, L. Passivation of reinforcing steel in concrete. In Materials Science Forum; Trans Tech Publications Ltd: Stafa-Zurich, Switzerland, 1995; Volume 192, pp. 891–898. [Google Scholar]
- Saremi, M.; Mahallati, E. A study on chloride-induced depassivation of mild steel in simulated concrete pore solution. Cem. Concr. Res. 2002, 32, 1915–1921. [Google Scholar] [CrossRef]
- Ghods, P.; Isgor, O.; McRae, G.; Miller, T. The effect of concrete pore solution composition on the quality of passive oxide films on black steel reinforcement. Cem. Concr. Compos. 2009, 31, 2–11. [Google Scholar] [CrossRef]
- Ghods, P.; Isgor, O.; Carpenter, G.; Li, J.; McRae, G.; Gu, G. Nano-scale study of passive films and chloride-induced depassivation of carbon steel rebar in simulated concrete pore solutions using FIB/TEM. Cem. Concr. Res. 2013, 47, 55–68. [Google Scholar] [CrossRef]
- Lian, C.; Zhuge, Y.; Beecham, S. The relationship between porosity and strength for porous concrete. Constr. Build. Mater. 2011, 25, 4294–4298. [Google Scholar] [CrossRef]
- Angst, U.M.; Geiker, M.R.; Michel, A.; Gehlen, C.; Wong, H.; Isgor, O.B.; Elsener, B.; Hansson, C.M.; François, R.; Hornbostel, K.; et al. The steel–concrete interface. Mater. Struct. 2017, 50, 143. [Google Scholar] [CrossRef]
- Alonso, C.; Andrade, C.; Castellote, M.; Castro, P. Chloride threshold values to depassivate reinforcing bars embedded in a standardized OPC mortar. Cem. Concr. Res. 2000, 30, 1047–1055. [Google Scholar] [CrossRef]
- Angst, U.; Elsener, B.; Larsen, C.K.; Vennesland, Ø. Critical chloride content in reinforced concrete—A review. Cem. Concr. Res. 2009, 39, 1122–1138. [Google Scholar] [CrossRef]
- Suda, K.; Misra, S.; Motohashi, K. Corrosion products of reinforcing bars embedded in concrete. Corros. Sci. 1993, 35, 1543–1549. [Google Scholar] [CrossRef]
- Molina, F.; Alonso, C.; Andrade, C. Cover cracking as a function of rebar corrosion: Part 2—Numerical model. Mater. Struct. 1993, 26, 532–548. [Google Scholar] [CrossRef]
- Sola, E.; Ožbolt, J.; Balabanić, G.; Mir, Z. Experimental and numerical study of accelerated corrosion of steel reinforcement in concrete: Transport of corrosion products. Cem. Concr. Res. 2019, 120, 119–131. [Google Scholar] [CrossRef]
- Sola, E. Experimental and Numerical Study of Chloride Induced Corrosion in Reinforced Concrete. Ph.D. Thesis, University of Stuttgart, Stuttgart, Germany, 2017. [Google Scholar]
- Ožbolt, J.; Oršanić, F.; Balabanić, G. Modeling influence of hysteretic moisture behavior on distribution of chlorides in concrete. Cem. Concr. Compos. 2016, 67, 73–84. [Google Scholar] [CrossRef]
- Yuan, Q.; Shi, C.; De Schutter, G.; Audenaert, K.; Deng, D. Chloride binding of cement-based materials subjected to external chloride environment–a review. Constr. Build. Mater. 2009, 23, 1–13. [Google Scholar] [CrossRef]
- Arya, C.; Buenfeld, N.; Newman, J. Factors influencing chloride-binding in concrete. Cem. Concr. Res. 1990, 20, 291–300. [Google Scholar] [CrossRef]
- Page, C.L. Initiation of chloride-induced corrosion of steel in concrete: Role of the interfacial zone. Mater. Corros. 2009, 60, 586–592. [Google Scholar] [CrossRef]
- Justnes, H. A review of chloride binding in cementitious systems. Nord. Concr. Res. Publ. 1998, 21, 48–63. [Google Scholar]
- Neville, A. Chloride attack of reinforced concrete: An overview. Mater. Struct. 1995, 28, 63. [Google Scholar] [CrossRef]
- Florea, M.; Brouwers, H. Chloride binding related to hydration products: Part I: Ordinary Portland Cement. Cem. Concr. Res. 2012, 42, 282–290. [Google Scholar] [CrossRef]
- Suryavanshi, A.; Scantlebury, J.; Lyon, S. Mechanism of Friedel’s salt formation in cements rich in tri-calcium aluminate. Cem. Concr. Res. 1996, 26, 717–727. [Google Scholar] [CrossRef]
- Zibara, H. Binding of External Chlorides by Cement Pastes. Ph.D. Thesis, National Library of Canada= Bibliothèque nationale du Canada, Ottawa, ON, Canada, 2001. [Google Scholar]
- Traetteberg, A. The mechanism of chloride penetration in concrete. SINTEF Rep. STF65 A 1977, 77070, 1977. [Google Scholar]
- Luping, T.; Nilsson, L.O. Chloride binding capacity and binding isotherms of OPC pastes and mortars. Cem. Concr. Res. 1993, 23, 247–253. [Google Scholar] [CrossRef]
- Al-Hussaini, M.; Sangha, C.; Plunkett, B.; Walden, P. The effect of chloride ion source on the free chloride ion percentages in OPC mortars. Cem. Concr. Res. 1990, 20, 739–745. [Google Scholar] [CrossRef]
- Juenger, M.C.; Siddique, R. Recent advances in understanding the role of supplementary cementitious materials in concrete. Cem. Concr. Res. 2015, 78, 71–80. [Google Scholar] [CrossRef]
- Kayali, O.; Ahmed, M.; Khan, M. Friedel’s salt and hydrotalcite-layered double hydroxides and the protection against chloride induced corrosion. Civ. Envir. Res 2013, 5, 111–117. [Google Scholar]
- Dhir, R.; El-Mohr, M.; Dyer, T. Chloride binding in GGBS concrete. Cem. Concr. Res. 1996, 26, 1767–1773. [Google Scholar] [CrossRef]
- Iyi, N.; Okamoto, K.; Kaneko, Y.; Matsumoto, T. Effects of anion species on deintercalation of carbonate ions from hydrotalcite-like compounds. Chem. Lett. 2005, 34, 932–933. [Google Scholar] [CrossRef]
- Ma, J.; Duan, P.; Ren, D.; Zhou, W. Effects of layered double hydroxides incorporation on carbonation resistance of cementitious materials. J. Mater. Res. Technol. 2019, 8, 292–298. [Google Scholar] [CrossRef]
- Alcantara, A.; Aranda, P.; Darder, M.; Ruiz-Hitzky, E. Bionanocomposites based on alginate–zein/layered double hydroxide materials as drug delivery systems. J. Mater. Chem. 2010, 20, 9495–9504. [Google Scholar] [CrossRef]
- Bejoy, N. Hydrotalcite. Resonance 2001, 6, 57–61. [Google Scholar] [CrossRef]
- Kwak, S.Y.; Jeong, Y.J.; Park, J.S.; Choy, J.H. Bio-LDH nanohybrid for gene therapy. Solid State Ionics 2002, 151, 229–234. [Google Scholar] [CrossRef]
- Cai, P.; Zheng, H.; Wang, C.; Ma, H.; Hu, J.; Pu, Y.; Liang, P. Competitive adsorption characteristics of fluoride and phosphate on calcined Mg–Al–CO3 layered double hydroxides. J. Hazard. Mater. 2012, 213, 100–108. [Google Scholar] [CrossRef]
- Kameda, T.; Takeuchi, H.; Yoshioka, T. Uptake of heavy metal ions from aqueous solution using Mg–Al layered double hydroxides intercalated with citrate, malate, and tartrate. Sep. Purif. Technol. 2008, 62, 330–336. [Google Scholar] [CrossRef]
- Lv, L.; He, J.; Wei, M.; Evans, D.; Duan, X. Uptake of chloride ion from aqueous solution by calcined layered double hydroxides: Equilibrium and kinetic studies. Water Res. 2006, 40, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Douglas, G.; Wendling, L.; Pleysier, R.; Trefry, M. Hydrotalcite formation for contaminant removal from Ranger mine process water. Mine Water Environ. 2010, 29, 108–115. [Google Scholar] [CrossRef]
- Alvarez, D.; Collazo, A.; Hernández, M.; Nóvoa, X.R.; Pérez, C. Corrosion protective properties of hydrotalcites doped hybrid sol-gel coatings on aluminium substrates. In Materials Science Forum; Trans Tech Publications Ltd: Stafa-Zurich, Switzerland, 2010; Volume 636, pp. 996–1003. [Google Scholar]
- Wajima, T.; Shimizu, T.; Ikegami, Y. New simple process of making agricultural cultivation solution from seawater. Bull. Soc. Sea Water Sci. Jpn. 2006, 60, 201–202. [Google Scholar]
- Mohapatra, L.; Parida, K. Zn–Cr layered double hydroxide: Visible light responsive photocatalyst for photocatalytic degradation of organic pollutants. Sep. Purif. Technol. 2012, 91, 73–80. [Google Scholar] [CrossRef]
- Peng, C.; Yu, J.; Zhao, Z.; Dai, J.; Fu, J.; Zhao, M.; Wang, W. Synthesis and properties of a clean and sustainable deicing additive for asphalt mixture. PLoS ONE 2015, 10, e0115721. [Google Scholar] [CrossRef] [PubMed]
- Rives, V. Layered Double Hydroxides: Present and Future; Nova Publishers: Hauppauge, NY, USA, 2001. [Google Scholar]
- Duan, X.; Evans, D.G. Layered Double Hydroxides; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2006; Volume 119. [Google Scholar]
- Bontchev, R.P.; Liu, S.; Krumhansl, J.L.; Voigt, J.; Nenoff, T.M. Synthesis, characterization, and ion exchange properties of hydrotalcite Mg6Al2(OH)16(A)x(A‘)2−x. 4H2O(A, A‘= Cl−, Br−, I−, and NO, 2 ⩾ x⩾ 0) derivatives. Chem. Mater. 2003, 15, 3669–3675. [Google Scholar] [CrossRef]
- Newman, S.P.; Jones, W. Comparative study of some layered hydroxide salts containing exchangeable interlayer anions. J. Solid State Chem. 1999, 148, 26–40. [Google Scholar] [CrossRef]
- Newman, S.P.; Jones, W. Synthesis, characterization and applications of layered double hydroxides containing organic guests. New J. Chem. 1998, 22, 105–115. [Google Scholar] [CrossRef]
- Lv, L.; Sun, P.; Gu, Z.; Du, H.; Pang, X.; Tao, X.; Xu, R.; Xu, L. Removal of chloride ion from aqueous solution by ZnAl-NO3 layered double hydroxides as anion-exchanger. J. Hazard. Mater. 2009, 161, 1444–1449. [Google Scholar] [CrossRef]
- De Sá, F.P.; Cunha, B.N.; Nunes, L.M. Effect of pH on the adsorption of Sunset Yellow FCF food dye into a layered double hydroxide (CaAl-LDH-NO3). Chem. Eng. J. 2013, 215, 122–127. [Google Scholar] [CrossRef]
- He, J.; Wei, M.; Li, B.; Kang, Y.; Evans, D.G.; Duan, X. Preparation of layered double hydroxides. In Layered Double Hydroxides; Springer: Berlin/Heidelberg, Germany, 2006; pp. 89–119. [Google Scholar]
- Zuo, J.; Wu, B.; Luo, C.; Dong, B.; Xing, F. Preparation of MgAl layered double hydroxides intercalated with nitrite ions and corrosion protection of steel bars in simulated carbonated concrete pore solution. Corros. Sci. 2019, 152, 120–129. [Google Scholar] [CrossRef]
- Yoon, S.; Moon, J.; Bae, S.; Duan, X.; Giannelis, E.P.; Monteiro, P.M. Chloride adsorption by calcined layered double hydroxides in hardened Portland cement paste. Mater. Chem. Phys. 2014, 145, 376–386. [Google Scholar] [CrossRef]
- Ke, X.; Bernal, S.A.; Provis, J.L. Uptake of chloride and carbonate by Mg-Al and Ca-Al layered double hydroxides in simulated pore solutions of alkali-activated slag cement. Cem. Concr. Res. 2017, 100, 1–13. [Google Scholar] [CrossRef]
- Wong, F.; Buchheit, R. Utilizing the structural memory effect of layered double hydroxides for sensing water uptake in organic coatings. Prog. Org. Coatings 2004, 51, 91–102. [Google Scholar] [CrossRef]
- Wei, J.; Gao, Z.; Song, Y.; Yang, W.; Wang, J.; Li, Z.; Mann, T.; Zhang, M.; Liu, L. Solvothermal synthesis of Li–Al layered double hydroxides and their electrochemical performance. Mater. Chem. Phys. 2013, 139, 395–402. [Google Scholar] [CrossRef]
- Bergaya, F.; Lagaly, G. Handbook of Clay Science; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Hochstetter, C. Untersuchung über die Zusammensetzung einiger Mineralien. J. Für Prakt. Chem. 1842, 27, 375–378. [Google Scholar] [CrossRef]
- Matschei, T.; Lothenbach, B.; Glasser, F. The AFm phase in Portland cement. Cem. Concr. Res. 2007, 37, 118–130. [Google Scholar] [CrossRef]
- Renaudin, G.; Francois, M.; Evrard, O. Order and disorder in the lamellar hydrated tetracalcium monocarboaluminate compound. Cem. Concr. Res. 1999, 29, 63–69. [Google Scholar] [CrossRef]
- Provis, J.L.; Bernal, S.A. Geopolymers and related alkali-activated materials. Annu. Rev. Mater. Res. 2014, 44, 299–327. [Google Scholar] [CrossRef]
- Wang, S.D.; Scrivener, K.L. Hydration products of alkali activated slag cement. Cem. Concr. Res. 1995, 25, 561–571. [Google Scholar] [CrossRef]
- Ke, X.; Bernal, S.A.; Provis, J.L. Chloride binding capacity of hydrotalcite in near-neutral and alkaline environments. In Proceedings of the 34thCementand Concrete Science Conference, University of Sheffield, Sheffield, UK, 14–17 September 2014. [Google Scholar]
- Kayali, O.; Khan, M.; Ahmed, M.S. The role of hydrotalcite in chloride binding and corrosion protection in concretes with ground granulated blast furnace slag. Cem. Concr. Compos. 2012, 34, 936–945. [Google Scholar] [CrossRef]
- Tedim, J.; Kuznetsova, A.; Salak, A.; Montemor, F.; Snihirova, D.; Pilz, M.; Zheludkevich, M.; Ferreira, M. Zn–Al layered double hydroxides as chloride nanotraps in active protective coatings. Corros. Sci. 2012, 55, 1–4. [Google Scholar] [CrossRef]
- Forano, C.; Hibino, T.; Leroux, F.; Taviot-Guého, C. Chapter 13.1 layered double hydroxides in Handbook of Clay science. Dev. Clay Sci. 2006, 1, 1021–1095. [Google Scholar]
- Kuang, Y.; Zhao, L.; Zhang, S.; Zhang, F.; Dong, M.; Xu, S. Morphologies, preparations and applications of layered double hydroxide micro-/nanostructures. Materials 2010, 3, 5220–5235. [Google Scholar] [CrossRef]
- Qu, Z.; Yu, Q.; Brouwers, H. Relationship between the particle size and dosage of LDHs and concrete resistance against chloride ingress. Cem. Concr. Res. 2018, 105, 81–90. [Google Scholar] [CrossRef]
- Xu, S.; Chen, Z.; Zhang, B.; Yu, J.; Zhang, F.; Evans, D.G. Facile preparation of pure CaAl-layered double hydroxides and their application as a hardening accelerator in concrete. Chem. Eng. J. 2009, 155, 881–885. [Google Scholar] [CrossRef]
- Duan, P.; Chen, W.; Ma, J.; Shui, Z. Influence of layered double hydroxides on microstructure and carbonation resistance of sulphoaluminate cement concrete. Constr. Build. Mater. 2013, 48, 601–609. [Google Scholar] [CrossRef]
- Yang, Z.; Fischer, H.; Polder, R. Laboratory investigation of the influence of two types of modified hydrotalcites on chloride ingress into cement mortar. Cem. Concr. Compos. 2015, 58, 105–113. [Google Scholar] [CrossRef]
- Xiong, L.X.; Liu, C.J.; Yuan, X.W. Effects of calcined Mg-Al-CO3 layered double hydroxides on resistance of cement mortar to chloride erosion. Ind. J. Eng. and Mater. Sci. 2015, 22, 225–230. [Google Scholar]
- Li, H.; Guan, X.; Yang, L.; Liu, S.; Zhang, J.; Guo, Y. Effects of LiAl-layered double hydroxides on early hydration of calcium sulphoaluminate cement paste. J. Wuhan Univ. Technol.-Mater. Sci. Ed. 2017, 32, 1101–1107. [Google Scholar] [CrossRef]
- Wu, Y.; Duan, P.; Yan, C. Role of layered double hydroxides in setting, hydration degree, microstructure and compressive strength of cement paste. Appl. Clay Sci. 2018, 158, 123–131. [Google Scholar] [CrossRef]
- Chen, Y.; Yu, R.; Wang, X.; Chen, J.; Shui, Z. Evaluation and optimization of Ultra-High Performance Concrete (UHPC) subjected to harsh ocean environment: Towards an application of Layered Double Hydroxides (LDHs). Constr. Build. Mater. 2018, 177, 51–62. [Google Scholar] [CrossRef]
- Zou, D.; Wang, K.; Li, H.; Guan, X. Effect of LiAl-layered double hydroxides on hydration of calcium sulfoaluminate cement at low temperature. Constr. Build. Mater. 2019, 223, 910–917. [Google Scholar] [CrossRef]
- Du, H.; Gao, H.J.; Dai Pang, S. Improvement in concrete resistance against water and chloride ingress by adding graphene nanoplatelet. Cem. Concr. Res. 2016, 83, 114–123. [Google Scholar] [CrossRef]
- Du, H.; Dai Pang, S. Enhancement of barrier properties of cement mortar with graphene nanoplatelet. Cem. Concr. Res. 2015, 76, 10–19. [Google Scholar] [CrossRef]
- Land, G.; Stephan, D. Controlling cement hydration with nanoparticles. Cem. Concr. Compos. 2015, 57, 64–67. [Google Scholar] [CrossRef]
- Bräu, M.; Ma-Hock, L.; Hesse, C.; Nicoleau, L.; Strauss, V.; Treumann, S.; Wiench, K.; Landsiedel, R.; Wohlleben, W. Nanostructured calcium silicate hydrate seeds accelerate concrete hardening: A combined assessment of benefits and risks. Arch. Toxicol. 2012, 86, 1077–1087. [Google Scholar] [CrossRef]
- Myrdal, R. Accelerating Admixtures for Concrete. State of the Art—(Technical Report SINTEF); SINTFE: Trondheim, Norway, 2007. [Google Scholar]
- Guan, X.; Li, H.; Luo, S.; Liu, X.; Zhang, J. Influence of LiAl-layered double hydroxides with 3D micro-nano structures on the properties of calcium sulphoaluminate cement clinker. Cem. Concr. Compos. 2016, 70, 15–23. [Google Scholar] [CrossRef]
- Šiler, P.; Kolářová, I.; Másilko, J.; Novotnỳ, R.; Opravil, T. The Effect of Zinc on the Portland Cement Hydration. In Key Engineering Materials; Trans Tech Publications Ltd: Stafa-Zurich, Switzerland, 2018; Volume 761, pp. 131–134. [Google Scholar]
- Asavapisit, S.; Fowler, G.; Cheeseman, C. Solution chemistry during cement hydration in the presence of metal hydroxide wastes. Cem. Concr. Res. 1997, 27, 1249–1260. [Google Scholar] [CrossRef]
- Trezza, M.A. Hydration study of ordinary portland cement in the presence of zinc ions. Mater. Res. 2007, 10, 331–334. [Google Scholar] [CrossRef]
- Li, F.; Duan, X. Applications of layered double hydroxides. In Layered Double Hydroxides; Springer: Berlin/Heidelberg, Germany, 2006; pp. 193–223. [Google Scholar]
- Martin, K.J.; Pinnavaia, T. Layered double hydroxides as supported anionic reagents. Halide-ion reactivity in zinc chromium hexahydroxide halide hydrates [Zn2Cr (OH) 6X. nH2O](X= Cl, I). J. Am. Chem. Soc. 1986, 108, 541–542. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Dong, C.; Wang, G.; Cheng, X.; Li, X. Zn–Al–NO2 layered double hydroxide as a controlled-release corrosion inhibitor for steel reinforcements. Mater. Lett. 2019, 236, 517–520. [Google Scholar] [CrossRef]
- Poznyak, S.; Tedim, J.; Rodrigues, L.; Salak, A.; Zheludkevich, M.; Dick, L.; Ferreira, M. Novel inorganic host layered double hydroxides intercalated with guest organic inhibitors for anticorrosion applications. ACS Appl. Mater. Interfaces 2009, 1, 2353–2362. [Google Scholar] [CrossRef]
- Cao, Y.; Dong, S.; Zheng, D.; Wang, J.; Zhang, X.; Du, R.; Song, G.; Lin, C. Multifunctional inhibition based on layered double hydroxides to comprehensively control corrosion of carbon steel in concrete. Corros. Sci. 2017, 126, 166–179. [Google Scholar] [CrossRef]
- Carlino, S. The intercalation of carboxylic acids into layered double hydroxides: A critical evaluation and review of the different methods. Solid State Ionics 1997, 98, 73–84. [Google Scholar] [CrossRef]
- DAvo, L. Anion-exchange in takovite: Applications to other hydroxide minerals. Bull. Miner. 1980, 103, 170–175. [Google Scholar]
- Meyn, M.; Beneke, K.; Lagaly, G. Anion-exchange reactions of layered double hydroxides. Inorg. Chem. 1990, 29, 5201–5207. [Google Scholar] [CrossRef]
- Costa, F.R.; Abdel-Goad, M.; Wagenknecht, U.; Heinrich, G. Nanocomposites based on polyethylene and Mg–Al layered double hydroxide. I. Synthesis and characterization. Polymer 2005, 46, 4447–4453. [Google Scholar] [CrossRef]
- Pöllmann, H.; Göske, J. Fixation of Chromate in Layered Double Hydroxides of the TCAH Type and Some Complex Application Mixtures. In Minerals as Advanced Materials II; Springer: Berlin/Heidelberg, Germany, 2011; pp. 103–114. [Google Scholar]
- Hibino, T. Anion Selectivity of Layered Double Hydroxides: Effects of Crystallinity and Charge Density. Eur. J. Inorg. Chem. 2018, 2018, 722–730. [Google Scholar] [CrossRef]
- Miyata, S. Anion-exchange properties of hydrotalcite-like compounds. Clays Clay Miner. 1983, 31, 305–311. [Google Scholar] [CrossRef]
- Costa, D.G.; Rocha, A.B.; Souza, W.F.; Chiaro, S.S.X.; Leitão, A.A. Comparative Structural, thermodynamic and electronic analyses of Zn–Al–An- hydrotalcite-like compounds (An−=Cl−, F−, Br−, OH−, CO or NO): An ab initio study. Appl. Clay Sci. 2012, 56, 16–22. [Google Scholar] [CrossRef]
- Miyata, S. The Syntheses of Hydrotalcite-Like Compounds and Their Structures and Physico-Chemical Properties—I: The Systems Mg2+-Al3+-NO, Mg2+-Al3+-Cl−, Mg2+-Al3+-ClO, Ni2+-Al3+-Cl− and Zn2+-Al3+-Cl−. Clays Clay Miner. 1975, 23, 369–375. [Google Scholar] [CrossRef]
- Miyata, S.; Okada, A. Synthesis of hydrotalcite-like compounds and their physico-chemical properties—The systems Mg 2+-Al 3+-SO 4 2- and Mg 2+-Al 3+-CrO 4 2-. Clays Clay Miner. 1977, 25, 14–18. [Google Scholar] [CrossRef]
- Duan, P.; Yan, C.; Zhou, W. Effects of calcined layered double hydroxides on carbonation of concrete containing fly ash. Constr. Build. Mater. 2018, 160, 725–732. [Google Scholar] [CrossRef]
- Chen, Y.; Shui, Z.; Chen, W.; Chen, G. Chloride binding of synthetic Ca–Al–NO3 LDHs in hardened cement paste. Constr. Build. Mater. 2015, 93, 1051–1058. [Google Scholar] [CrossRef]
- Shui, Z.; Yu, R.; Chen, Y.; Duan, P.; Ma, J.; Wang, X. Improvement of concrete carbonation resistance based on a structure modified Layered Double Hydroxides (LDHs): Experiments and mechanism analysis. Constr. Build. Mater. 2018, 176, 228–240. [Google Scholar] [CrossRef]
- Tatematsu, H.; Sasaki, T. Repair materials system for chloride-induced corrosion of reinforcing bars. Cem. Concr. Compos. 2003, 25, 123–129. [Google Scholar] [CrossRef]
- Zhonghe, S.; Juntao, M.; Wei, C.; Xiaoxing, C. Chloride binding capacity of cement paste containing layered double hydroxide (LDH). J. Test. Eval. 2012, 40, 796–800. [Google Scholar] [CrossRef]
- Glasser, F.; Kindness, A.; Stronach, S. Stability and solubility relationships in AFm phases-Its solid solutions and their role in chloride binding. Cem. Concr. Res. 1999, 29, 861–866. [Google Scholar] [CrossRef]
- Larsen, C. Chloride Binding in Concrete, Effect of Surrounding Environment and Concrete Composition. Ph.D. Thesis, The Norwegian University of Science and Technology, Trondheim, Norway, 1998. [Google Scholar]
- Hobbs, M.Y. Solubilities and Ion Exchange Properties of Solid Solutions between the Hydroxyl, Chlorine and Carbon Trioxide end Members of the Monocalcium Aluminate Hydrates. Ph.D. Thesis, University of Waterloo, Waterloo, ON, USA, 2001. [Google Scholar]
- Suryavanshi, A.; Swamy, R.N. Stability of Friedel’s salt in carbonated concrete structural elements. Cem. Concr. Res. 1996, 26, 729–741. [Google Scholar] [CrossRef]
- Jones, M.; Macphee, D.E.; Chudek, J.; Hunter, G.; Lannegrand, R.; Talero, R.; Scrimgeour, S. Studies using 27Al MAS NMR of AFm and AFt phases and the formation of Friedel’s salt. Cem. Concr. Res. 2003, 33, 177–182. [Google Scholar] [CrossRef]
- Struble, L. Synthesis and characterization of ettringite and related phases. In Proceedings of the 8th International Congress on yhe Chemistry of Cement, Rio de Janeiro, Brazil, 22–27 September 1986; Volume 1, pp. 449–482. [Google Scholar]
- Hirao, H.; Yamada, K.; Takahashi, H.; Zibara, H. Chloride binding of cement estimated by binding isotherms of hydrates. J. Adv. Concr. Technol. 2005, 3, 77–84. [Google Scholar] [CrossRef]
- Birnin-Yauri, U.; Glasser, F. Friedel’s salt, Ca2Al(OH)6 (Cl, OH)·2H2O: Its solid solutions and their role in chloride binding. Cem. Concr. Res. 1998, 28, 1713–1723. [Google Scholar] [CrossRef]
- Berke, N.S.; Rosenberg, A. Technical review of calcium nitrite corrosion inhibitor in concrete. Transp. Res. Rec. 1989, 1211, 18–27. [Google Scholar]
- Ann, K.Y.; Jung, H.; Kim, H.; Kim, S.; Moon, H.Y. Effect of calcium nitrite-based corrosion inhibitor in preventing corrosion of embedded steel in concrete. Cem. Concr. Res. 2006, 36, 530–535. [Google Scholar] [CrossRef]
- Yang, Z.; Polder, R.; Mol, J. Modified hydrotalcites as chloride scavengers and inhibitor release agents for improved corrosion protection of reinforced concrete. Heron 2017, 62, 61. [Google Scholar]
- Dry, C.; Corsaw, M. A time-release technique for corrosion prevention. Cem. Concr. Res. 1998, 28, 1133–1140. [Google Scholar] [CrossRef]
- Chen, L.J.; Chen, X.X.; Chen, W. Research on the Carbonation of Cement Paste Modified with Layered Double Hydroxides. In Applied Mechanics and Materials; Trans Tech Publications Ltd: Stafa-Zurich, Switzerland, 2012; Volume 174, pp. 706–710. [Google Scholar]
- Cao, Y.; Zheng, D.; Dong, S.; Zhang, F.; Lin, J.; Wang, C.; Lin, C. A Composite corrosion inhibitor of MgAl layered double hydroxides co-intercalated with hydroxide and organic anions for carbon steel in simulated carbonated concrete pore solutions. J. Electrochem. Soc. 2019, 166, C3106–C3113. [Google Scholar] [CrossRef]
- Shui, Z.H.; Ma, J.T.; Chen, W.; Gao, X. The effect of layered double hydroxides on the concrete resistance of chloride-ion penetration. In Key Engineering Materials; Trans Tech Publications Ltd: Stafa-Zurich, Switzerland, 2012; Volume 509, pp. 99–105. [Google Scholar]
- Yang, Z.; Fischer, H.; Polder, R. Synthesis and characterization of modified hydrotalcites and their ion exchange characteristics in chloride-rich simulated concrete pore solution. Cem. Concr. Compos. 2014, 47, 87–93. [Google Scholar] [CrossRef]
- Yang, Z.; Fischer, H.; Cerezo, J.; Mol, J.; Polder, R. Aminobenzoate modified MgAl hydrotalcites as a novel smart additive of reinforced concrete for anticorrosion applications. Constr. Build. Mater. 2013, 47, 1436–1443. [Google Scholar] [CrossRef]
- Matschei, T.; Lothenbach, B.; Glasser, F.P. Thermodynamic properties of Portland cement hydrates in the system CaO–Al2O3–SiO2–CaSO4–CaCO3–H2O. Cem. Concr. Res. 2007, 37, 1379–1410. [Google Scholar] [CrossRef]
- Xu, J.; Song, Y.; Zhao, Y.; Jiang, L.; Mei, Y.; Chen, P. Chloride removal and corrosion inhibitions of nitrate, nitrite-intercalated MgAl layered double hydroxides on steel in saturated calcium hydroxide solution. Appl. Clay Sci. 2018, 163, 129–136. [Google Scholar] [CrossRef]
- Chung, C.W.; Jung, H.Y.; Kwon, J.H.; Jang, B.K.; Kim, J.H. Use of calcium aluminum–layered double hydroxide to control chloride ion penetration of cement-based materials. J. Struct. Integr. Maint. 2019, 4, 37–42. [Google Scholar] [CrossRef]





| LDH | Cement | Dosage * | Concrete Properties | Age | Year | Ref. | |
|---|---|---|---|---|---|---|---|
| Type | Type | CS | FS | ||||
| CaAl LDH | CEM 42.5 | 1–5% | +6% (2%LDH) | - | 28d | 2009 | Xu et al. [98] |
| MgAl CO | [99] | 1–2% | inconclusive | - | 28, 48d | 2013 | Duan et al. [99] |
| MgAl LDH | [82] | 8.5% | ∼−2% | - | 28d | 2014 | Yoon et al. [82] |
| MgAl pAB | CEM I 42.5N | 5–10% | −17.2% (10%LDH) | −21.38% (10%LDH) | 28d | 2015 | Yang et al. [100] |
| MgAl NO | CEM I 42.5N | 5–10% | −14.2% (10%LDH) | −19.1% (10%LDH) | 28d | 2015 | Yang et al. [100] |
| MgAl LDH | [101] | 1% | −25% (28d) | - | 28–178d | 2015 | Xiong et al. [101] |
| LiAl LDH | [102] | 1–3% | +25% | - | 7d | 2017 | Haiyan et al. [102] |
| CaAl NO | CEM I 52.5R | 0.5–2 Vol. | +17% (1% LDH) | +55% (1%LDH) | 28d | 2018 | Qu et al. [97] |
| MgAl CO | CEM I 52.5 | 1–3% | ∼+3.5% (3% LDH) | - | 28d | 2018 | Wu et al. [103] |
| MgAl LDH | [104] | 1–2% | +8.2% (1% LDH) | - | 56d | 2018 | Chen et al. [104] |
| LiAl LDH | [105] | 0.5–1.5% | +46.2% (1%LDH) | - | 28d | 2019 | Zou et al. [105] |
| LDH | Experiments Conducted in | Preparation Method | Dosage | Application | Year / Ref. | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| SS | PS | CP | M | C | SS / PS | CP / M / C | ||||
| CaAl NO | - | - | ✓ | ✓ | - | - | - | 0.2 wt.% | Cl− uptake & Corr. inhibition | 2003/ [133] |
| MgAl NO | ✓ | - | ✓ | - | - | - | 1 g:10 mL | 1 wt.% | Cl− uptake | 2012 / [134] |
| MgAl CO | ✓ | - | ✓ | - | - | - | 1 g:10 mL | 1 wt.% | Cl− uptake | 2012 / [134] |
| hydrotalcite | ✓ | - | - | - | - | calcination | 8–16 g/80 mL | - | Cl− uptake | 2012 / [93] |
| MgAl NO | - | - | - | - | ✓ | - | - | 0.5–3 wt.% | Cl− uptake | 2012 / [149] |
| MgAl LDH | ✓ | - | ✓ | - | - | calcination | - | 8.5 wt.% | Cl− uptake | 2014/ [82] |
| MgAl CO | - | - | ✓ | - | ✓ | - | - | 1–2 wt.% | Cl−/ CO uptake & Pore ref. | 2013 /[99] |
| CaAl-pAB | - | ✓ | - | - | - | Ion-exchange | 0.5 g/10 mL | - | Cl− uptake | 2014 / [150] |
| CaAl NO | - | ✓ | - | - | - | co-precipitation | 0.5 g/10 mL | - | Cl− uptake | 2014 / [150] |
| CaAl NO | ✓ | ✓ | ✓ | - | - | co-precipitation | 1 g/100 mL | 8.5 wt.% | Cl− uptake | 2015 / [131] |
| MgAl-pAB | - | - | - | ✓ | - | calcination-rehydration [151] | - | 5–10 wt.% | Cl−uptake | 2015 / [100] |
| MgAl-NO | - | - | - | ✓ | - | calcination-rehydration [151] | - | 5–10 wt.% | Cl−uptake | 2015 / [100] |
| MgAl LDH | - | - | - | ✓ | - | calcination | - | 1 wt.% | Cl− uptake & bond stress | 2015 / [101] |
| Strätlingite | - | ✓ | - | - | - | [152] | 0.4 g/40 g | - | Cl−/ CO uptake | 2017 / [83] |
| MgAl LDH | - | ✓ | - | - | - | calcination | 0.4 g/40 g | - | Cl−/ CO uptake | 2017 / [83] |
| MgAl NO | - | ✓ | ✓ | ✓ | - | calcination-rehydration | 0.5 g/20 mL | 0–10%(M), 20 wt.%(CP) | Cl−-uptake & Corr. inhibition | 2017/ [145] |
| MgAl-pAB | - | ✓ | ✓ | ✓ | - | calcination-rehydration | 0.5 g/20 mL | 0–10%(M), 20 wt.%(CP) | Cl−-uptake & Corr. inhibition | 2017/ [145] |
| CaAl NO | - | - | - | ✓ | - | co-precipitation | - | 0.5–2 vol.% | Cl− uptake | 2018 / [97] |
| MgAl CO | - | - | - | - | ✓ | calcination, other [130] | - | 2 wt.% | CO uptake | 2018 / [130] |
| MgAl LDH | - | - | - | - | ✓ | calcination, other [132] | - | 2–4 wt.% | CO uptake | 2018/ [132] |
| MgAl NO | - | ✓ | - | - | - | Ion-exchange | 1 g/10 mL, 1–2 wt.% | - | Cl-uptake & Corr. inhibition | 2018 / [153] |
| MgAl NO | - | ✓ | - | - | - | co-precipitation | 1 g/10 mL, 1–2 wt.% | - | Cl− uptake | 2018 / [153] |
| MgAl LDH | - | - | ✓ | - | - | calcination | - | 1–2 wt.% | Cl− uptake | 2018/ [104] |
| CaAl LDH | ✓ | - | - | - | ✓ | co-precipitation | 50 g/L | 10 wt.% | Cl− uptake | 2019/ [154] |
| ZnAl NO/NO | ✓ | ✓ | ✓ | - | - | co-precipitation | 1 g/50 mL | 2–5 wt.% | Cl− uptake & Corr. inhibition | 2019 / [24,25] |
| MgAl CO | - | - | - | - | ✓ | calcination | - | 2 wt.% | CO uptake | 2019 / [61] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mir, Z.M.; Bastos, A.; Höche, D.; Zheludkevich, M.L. Recent Advances on the Application of Layered Double Hydroxides in Concrete—A Review. Materials 2020, 13, 1426. https://doi.org/10.3390/ma13061426
Mir ZM, Bastos A, Höche D, Zheludkevich ML. Recent Advances on the Application of Layered Double Hydroxides in Concrete—A Review. Materials. 2020; 13(6):1426. https://doi.org/10.3390/ma13061426
Chicago/Turabian StyleMir, Zahid M., Alexandre Bastos, Daniel Höche, and Mikhail L. Zheludkevich. 2020. "Recent Advances on the Application of Layered Double Hydroxides in Concrete—A Review" Materials 13, no. 6: 1426. https://doi.org/10.3390/ma13061426
APA StyleMir, Z. M., Bastos, A., Höche, D., & Zheludkevich, M. L. (2020). Recent Advances on the Application of Layered Double Hydroxides in Concrete—A Review. Materials, 13(6), 1426. https://doi.org/10.3390/ma13061426






