Monocytic Cell-Induced Phase Transformation of Circulating Lipid-Based Liquid Crystalline Nanosystems
Abstract
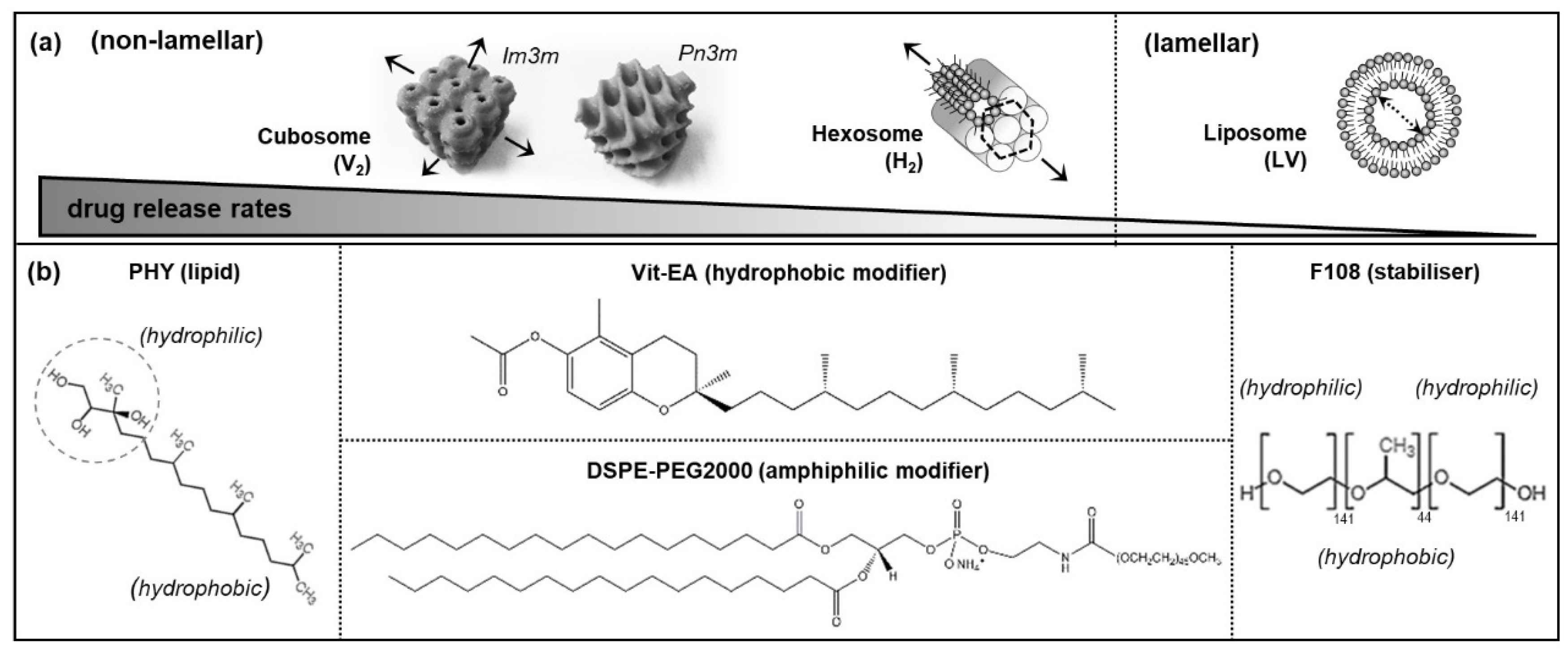
1. Introduction
2. Materials and Methods
2.1. Preparation of LLC Nanosystems
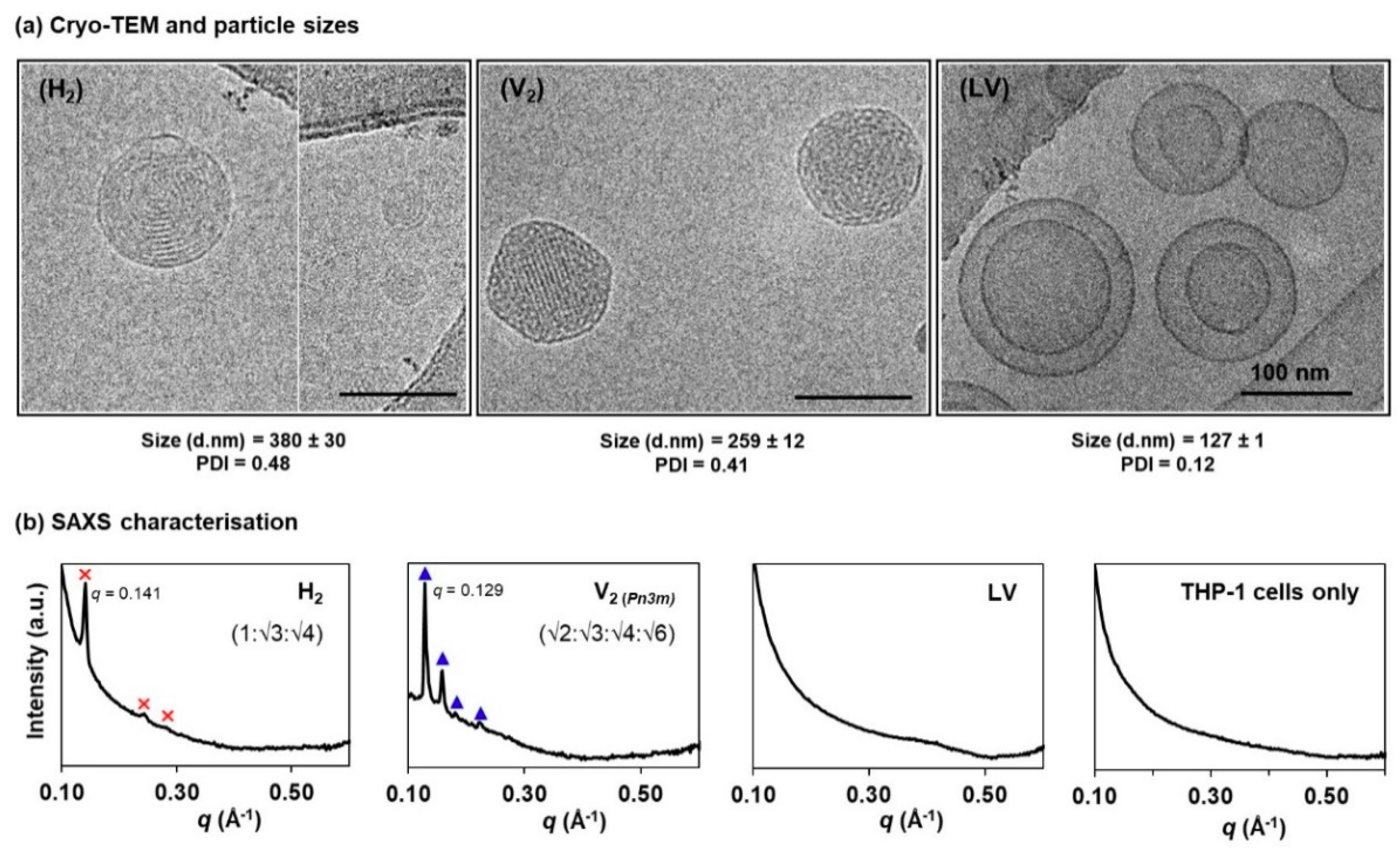
2.2. Particle Sizing and Cryogenic Transmission Electron Microscopy (Cryo-TEM) Analysis
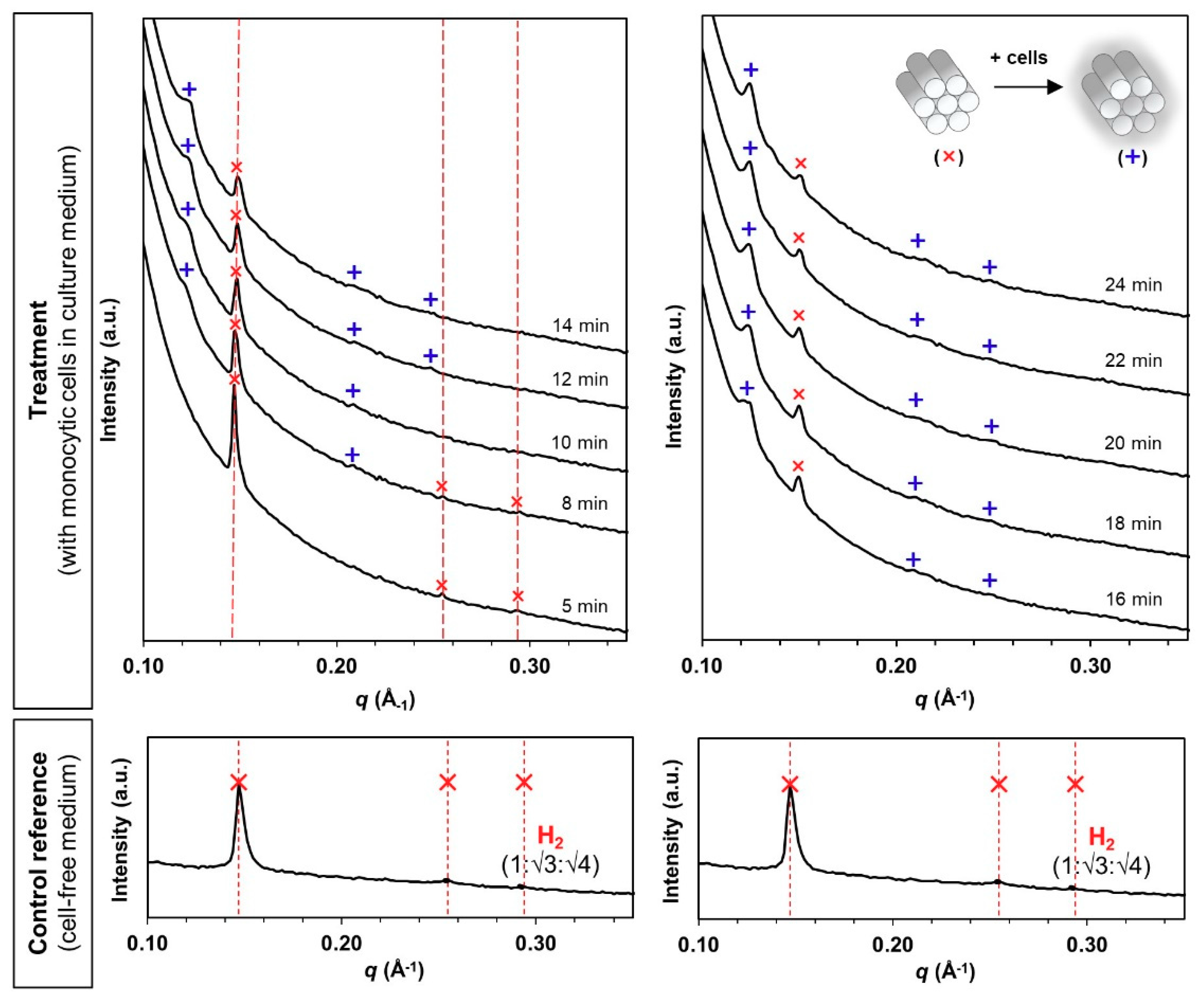
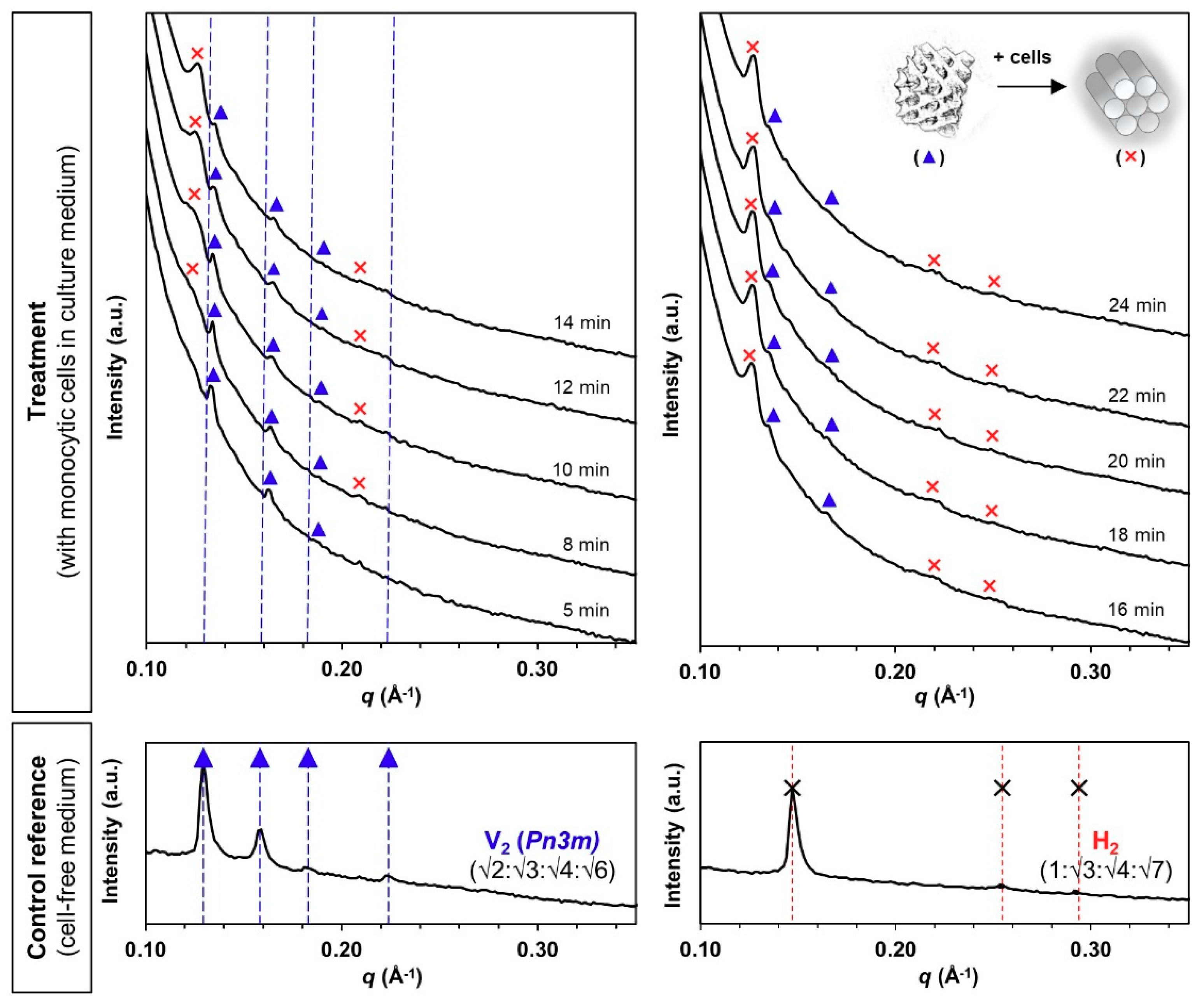
2.3. Biological Cell Flow-Through Setup for Small Angle X-Ray Scattering (Bio-SAXS) Analysis
3. Results and Discussion
3.1. Physicochemical Characteristics of Self-Assembled LLC Nanosystems
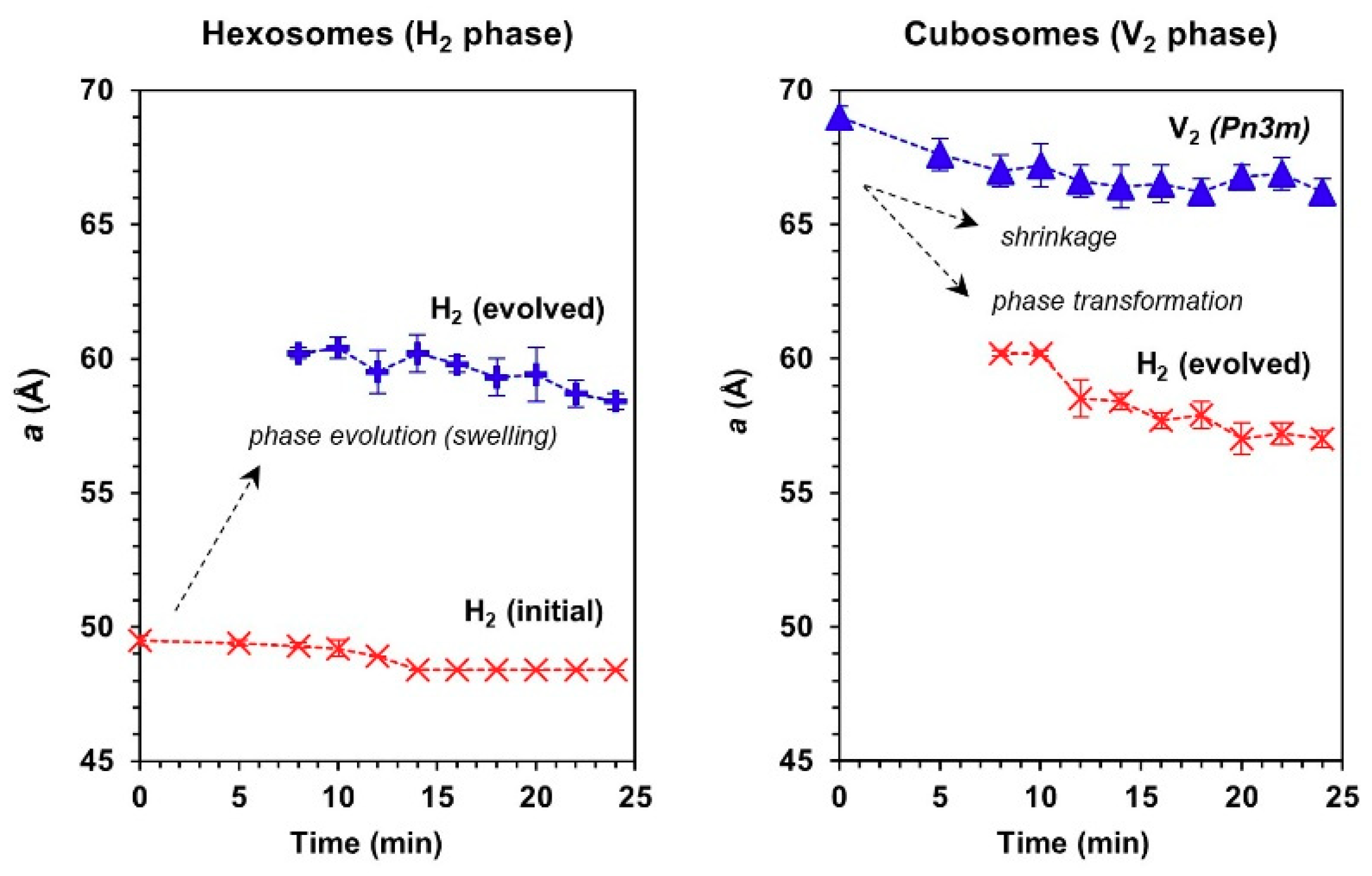
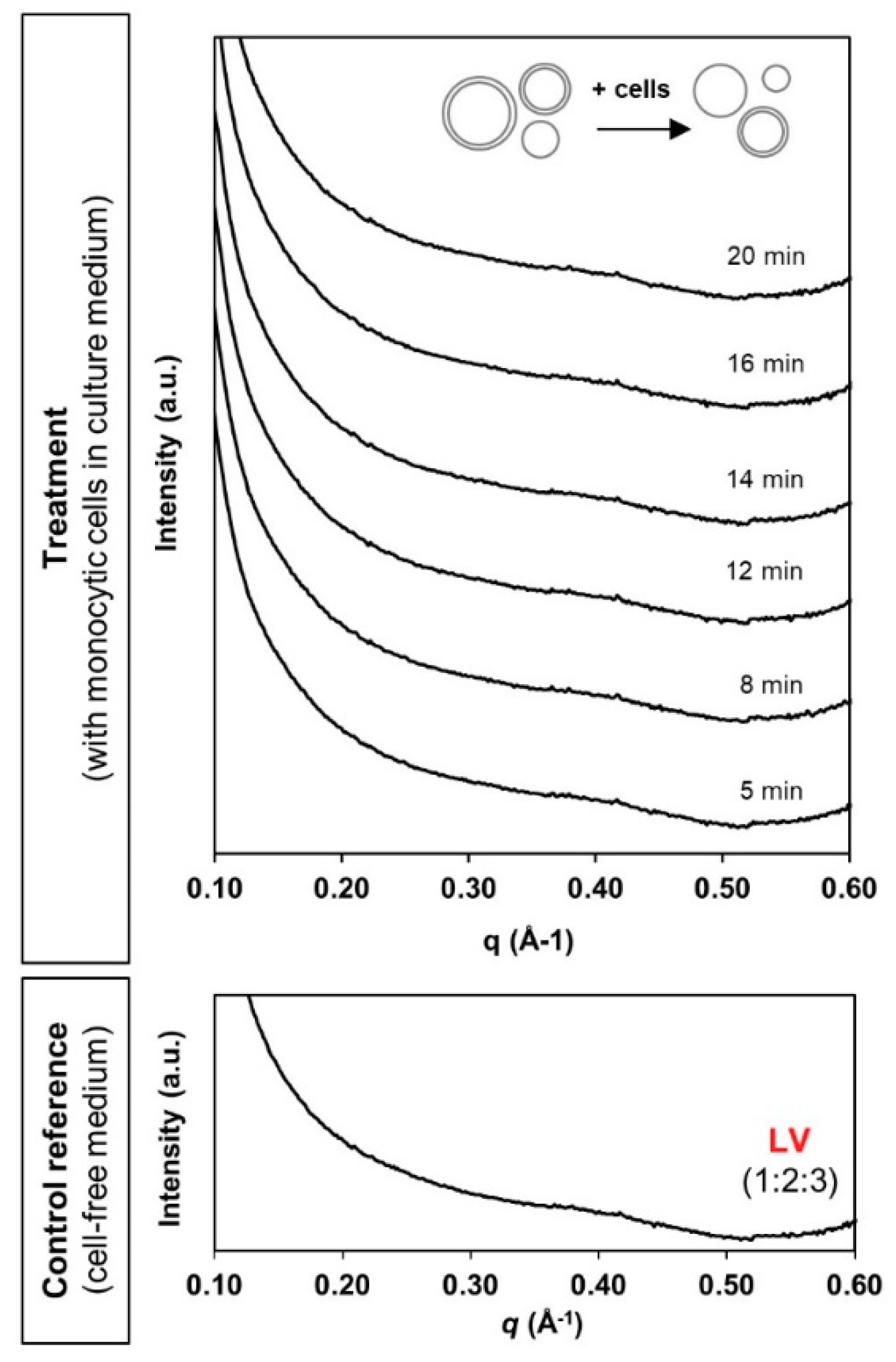
3.2. Phase Transformation of LLC Systems was Triggered by Monocytic Cells but not by Cell Culture Medium
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Latowski, D.; Åkerlund, H.-E.; Strzałka, K. Violaxanthin De-Epoxidase, the Xanthophyll Cycle Enzyme, Requires Lipid Inverted Hexagonal Structures for Its Activity†. Biochemistry 2004, 43, 4417–4420. [Google Scholar] [CrossRef] [PubMed]
- Jouhet, J. Importance of the hexagonal lipid phase in biological membrane organization. Front. Plant Sci. 2013, 4, 4. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Almsherqi, Z.A. Evolution of cubic membranes as antioxidant defence system. Interface Focus 2015, 5, 20150012. [Google Scholar] [CrossRef] [PubMed]
- Almsherqi, Z.A.; Landh, T.; Kohlwein, S.D.; Deng, Y. Chapter 6 Cubic Membranes. Int. Rev. Cell Mol. Biol. 2009, 274, 275–342. [Google Scholar] [PubMed]
- Woltman, S.J.; Jay, G.D.; Crawford, G.P. Liquid-crystal materials find a new order in biomedical applications. Nat. Mater. 2007, 6, 929–938. [Google Scholar] [CrossRef] [PubMed]
- Lagerwall, J.P.F.; Scalia, G. A new era for liquid crystal research: Applications of liquid crystals in soft matter nano-, bio- and microtechnology. Curr. Appl. Phys. 2012, 12, 1387–1412. [Google Scholar] [CrossRef]
- van’t Hag, L.; Gras, S.; Conn, C.E.; Drummond, C.J. Lyotropic liquid crystal engineering moving beyond binary compositional space—ordered nanostructured amphiphile self-assembly materials by design. Chem. Soc. Rev. 2017, 46, 2705–2731. [Google Scholar]
- Boyd, B.J.; Dong, Y.-D.; Rades, T. Nonlamellar liquid crystalline nanostructured particles: advances in materials and structure determination. J. Liposome Res. 2009, 19, 12–28. [Google Scholar] [CrossRef]
- Jewell, S.A. Liquid Crystals in Living Systems and Liquid Crystals in the Development of Life. In Handbook of Liquid Crystals; Wiley: Hoboken, NJ, USA, 2014; Volume 7, pp. 1–25. [Google Scholar]
- Israelachvili, J.N.; Mitchell, D.J.; Ninham, B. Theory of self-assembly of hydrocarbon amphiphiles into micelles and bilayers. J. Chem. Soc. Faraday Trans. 2 1976, 72, 1525–1568. [Google Scholar] [CrossRef]
- Tan, A.; Hong, L.; Du, J.D.; Boyd, B.J. Self-assembled nanostructured lipid systems: Is there a link between structure and cytotoxicity? Adv. Sci. 2019, 6, 1801223. [Google Scholar] [CrossRef]
- Bulbake, U.; Doppalapudi, S.; Kommineni, N.; Khan, W. Liposomal Formulations in Clinical Use: An Updated Review. Pharmaceutics 2017, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Camurus. Development Pipeline—Pharmaceuticals. Available online: https://www.camurus.com/pipeline/ (accessed on 15 January 2020).
- Angelov, B.; Garamus, V.M.; Drechsler, M.; Angelova, A. Structural analysis of nanoparticulate carriers for encapsulation of macromolecular drugs. J. Mol. Liq. 2017, 235, 83–89. [Google Scholar] [CrossRef]
- Angelova, A.; Angelov, B.; Garamus, V.M.; Couvreur, P.; Lesieur, S. Small-Angle X-ray Scattering Investigations of Biomolecular Confinement, Loading, and Release from Liquid-Crystalline Nanochannel Assemblies. J. Phys. Chem. Lett. 2012, 3, 445–457. [Google Scholar] [CrossRef] [PubMed]
- Kirby, N.; Mudie, S.T.; Hawley, A.M.; Cookson, D.; Mertens, H.D.T.; Cowieson, N.; Samardzic-Boban, V. A low-background-intensity focusing small-angle X-ray scattering undulator beamline. J. Appl. Crystallogr. 2013, 46, 1670–1680. [Google Scholar] [CrossRef]
- Dong, Y.-D.; Dong, A.W.; Larson, I.; Rappolt, M.; Amenitsch, H.; Hanley, T.; Boyd, B.J. Impurities in Commercial Phytantriol Significantly Alter Its Lyotropic Liquid-Crystalline Phase Behavior. Langmuir 2008, 24, 6998–7003. [Google Scholar] [CrossRef]
- Dong, Y.-D.; Larson, I.; Hanley, T.; Boyd, B.J. Bulk and Dispersed Aqueous Phase Behavior of Phytantriol: Effect of Vitamin E Acetate and F127 Polymer on Liquid Crystal Nanostructure. Langmuir 2006, 22, 9512–9518. [Google Scholar] [CrossRef]
- Muir, B.; Zhen, G.; Gunatillake, P.; Hartley, P. Salt Induced Lamellar to Bicontinuous Cubic Phase Transitions in Cationic Nanoparticles. J. Phys. Chem. B 2012, 116, 3551–3556. [Google Scholar] [CrossRef]
- Lenné, T.; Garvey, C.J.; Koster, K.L.; Bryant, G. Effects of sugars on lipid bilayers during dehydration—SAXS/WAXS measurements and quantitative model. J. Phys. Chem. B 2009, 113, 2486–2491. [Google Scholar] [CrossRef]
- Wang, Z.; Zheng, L.; Inoue, T. Effect of sucrose on the structure of a cubic phase formed from a monoolein/water mixture. J. Colloid Interface Sci. 2005, 288, 638–641. [Google Scholar] [CrossRef]
- Mezzenga, R.; Grigorov, M.; Zhang, Z.; Servais, C.; Sagalowicz, L.; Romoscanu, A.I.; Khanna, V.; Meyer, C. Polysaccharide-Induced Order-to-Order Transitions in Lyotropic Liquid Crystals. Langmuir 2005, 21, 6165–6169. [Google Scholar] [CrossRef]
- Du, J.; Liu, Q.; Salentinig, S.; Nguyen, T.-H.; Boyd, B.J. A novel approach to enhance the mucoadhesion of lipid drug nanocarriers for improved drug delivery to the buccal mucosa. Int. J. Pharm. 2014, 471, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Tajik-Ahmadabad, B.; Mechler, A.; Muir, B.; McLean, K.M.; Hinton, T.; Separovic, F.; Polyzos, A. A QCM-D and SAXS Study of the Interaction of Functionalised Lyotropic Liquid Crystalline Lipid Nanoparticles with siRNA. ChemBioChem 2017, 18, 921–930. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Fan, W.; Wu, N.; Zhu, J.; Miao, Y.; Miao, X.; Li, F.; Zhang, X.; Gan, Y. Lipid-Based Liquid Crystalline Nanoparticles Facilitate Cytosolic Delivery of siRNA via Structural Transformation. Nano Lett. 2018, 18, 2411–2419. [Google Scholar] [CrossRef]
- Tan, A.; Lam, Y.Y.; Pacot, O.; Hawley, A.; Boyd, B.J. Probing cell–nanoparticle (cubosome) interactions at the endothelial interface: do tissue dimension and flow matter? Biomater. Sci. 2019, 7, 3460–3470. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.; Hawley, A.M.; Hinton, T.; Mudie, S.T.; Giakoumatos, E.C.; Waddington, L.J.; Kirby, N.; Drummond, C.J.; Mulet, X.; Muir, B. Nanostructure and cytotoxicity of self-assembled monoolein–capric acid lyotropic liquid crystalline nanoparticles. RSC Adv. 2015, 5, 26785–26795. [Google Scholar] [CrossRef]
- Wibroe, P.P.; Azmi, I.D.M.; Nilsson, C.; Yaghmur, A.; Moghimi, S.M. Citrem modulates internal nanostructure of glyceryl monooleate dispersions and bypasses complement activation: Towards development of safe tunable intravenous lipid nanocarriers. Nanomed. Nanotechnol. Boil. Med. 2015, 11, 1909–1914. [Google Scholar] [CrossRef] [PubMed]
- Azmi, I.D.M.; Wu, L.; Wibroe, P.P.; Nilsson, C.; Østergaard, J.; Stürup, S.; Gammelgaard, B.; Urtti, A.; Moghimi, S.M.; Yaghmur, A. Modulatory Effect of Human Plasma on the Internal Nanostructure and Size Characteristics of Liquid-Crystalline Nanocarriers. Langmuir 2015, 31, 5042–5049. [Google Scholar] [CrossRef]
- Fong, C.; Weerawardena, A.; Sagnella, S.M.; Mulet, X.; Waddington, L.; Krodkiewska, I.; Drummond, C.J. Monodisperse nonionic phytanyl ethylene oxide surfactants: High throughput lyotropic liquid crystalline phase determination and the formation of liposomes, hexosomes and cubosomes. Soft Matter 2010, 6, 4727. [Google Scholar] [CrossRef]
- Schmutz, M.; Durand, D.; Debin, A.; Palvadeau, Y.; Etienne, A.; Thierry, A.R. DNA packing in stable lipid complexes designed for gene transfer imitates DNA compaction in bacteriophage. Proc. Natl. Acad. Sci. USA 1999, 96, 12293–12298. [Google Scholar] [CrossRef]
- Boyd, B.J.; Rizwan, S.B.; Dong, Y.-D.; Hook, S.; Rades, T. Self-Assembled Geometric Liquid-Crystalline Nanoparticles Imaged in Three Dimensions: Hexosomes Are Not Necessarily Flat Hexagonal Prisms. Langmuir 2007, 23, 12461–12464. [Google Scholar] [CrossRef]
- Hinton, T.; Grusche, F.; Acharya, D.; Shukla, R.; Bansal, V.; Waddington, L.J.; Monaghan, P.; Muir, B. Bicontinuous cubic phase nanoparticle lipid chemistry affects toxicity in cultured cells. Toxicol. Res. 2014, 3, 11–22. [Google Scholar] [CrossRef]
- Shen, H.-H.; Crowston, J.G.; Huber, F.; Saubern, S.; McLean, K.M.; Hartley, P. The influence of dipalmitoyl phosphatidylserine on phase behaviour of and cellular response to lyotropic liquid crystalline dispersions. Biomaterials 2010, 31, 9473–9481. [Google Scholar] [CrossRef]
- Zhai, J.; Suryadinata, R.; Luan, B.; Tran, N.; Ratcliffe, J.; Hinton, T.; Hao, X.; Drummond, C.J. Amphiphilic brush polymers produced using the RAFT polymerisation method stabilise and reduce the cell cytotoxicity of lipid lyotropic liquid crystalline nanoparticles. Faraday Discuss. 2016, 191, 545–563. [Google Scholar] [CrossRef]
- Li, T.; Cipolla, D.; Rades, T.; Boyd, B.J. Drug nanocrystallisation within liposomes. J. Control. Release 2018, 288, 96–110. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, C.V.; Wächter, W.; Salto, G.I.; Engelskirchen, S.; Ahualli, S. Monoolein: A magic lipid? Phys. Chem. Chem. Phys. 2011, 13, 3004–3021. [Google Scholar] [CrossRef] [PubMed]
- Tilley, A.; Dong, Y.-D.; Amenitsch, H.; Rappolt, M.; Boyd, B.J. Transfer of lipid and phase reorganisation in self-assembled liquid crystal nanostructured particles based on phytantriol. Phys. Chem. Chem. Phys. 2011, 13, 3026. [Google Scholar] [CrossRef]
- Salonen, A.; Moitzi, C.; Salentinig, S.; Glatter, O. Material Transfer in Cubosome−Emulsion Mixtures: Effect of Alkane Chain Length. Langmuir 2010, 26, 10670–10676. [Google Scholar] [CrossRef]
- Kang, B.; Okwieka, P.; Schöttler, S.; Winzen, S.; Langhanki, J.; Mohr, K.; Opatz, T.; Mailänder, V.; Landfester, K.; Wurm, F.R.; et al. Carbohydrate-Based Nanocarriers Exhibiting Specific Cell Targeting with Minimum Influence from the Protein Corona. Angew. Chem. Int. Ed. 2015, 54, 7436–7440. [Google Scholar] [CrossRef]
- Suk, J.S.; Xu, Q.; Kim, N.; Hanes, J.; Ensign, L. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv. Drug Deliv. Rev. 2016, 99, 28–51. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, A.; Lam, Y.Y.; Sun, X.; Boyd, B. Monocytic Cell-Induced Phase Transformation of Circulating Lipid-Based Liquid Crystalline Nanosystems. Materials 2020, 13, 1013. https://doi.org/10.3390/ma13041013
Tan A, Lam YY, Sun X, Boyd B. Monocytic Cell-Induced Phase Transformation of Circulating Lipid-Based Liquid Crystalline Nanosystems. Materials. 2020; 13(4):1013. https://doi.org/10.3390/ma13041013
Chicago/Turabian StyleTan, Angel, Yuen Yi Lam, Xiaohan Sun, and Ben Boyd. 2020. "Monocytic Cell-Induced Phase Transformation of Circulating Lipid-Based Liquid Crystalline Nanosystems" Materials 13, no. 4: 1013. https://doi.org/10.3390/ma13041013
APA StyleTan, A., Lam, Y. Y., Sun, X., & Boyd, B. (2020). Monocytic Cell-Induced Phase Transformation of Circulating Lipid-Based Liquid Crystalline Nanosystems. Materials, 13(4), 1013. https://doi.org/10.3390/ma13041013





