Surface Characterization and Conductivity of Two Types of Lithium-Based Glass Ceramics after Accelerating Ageing
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparations
2.2. Ageing Procedure
2.3. Surface Investigation Methods
2.4. Changes in Material Structure
2.5. Electrical Properties
3. Results and Discussion
3.1. SEM Study
3.2. XRPD Analysis
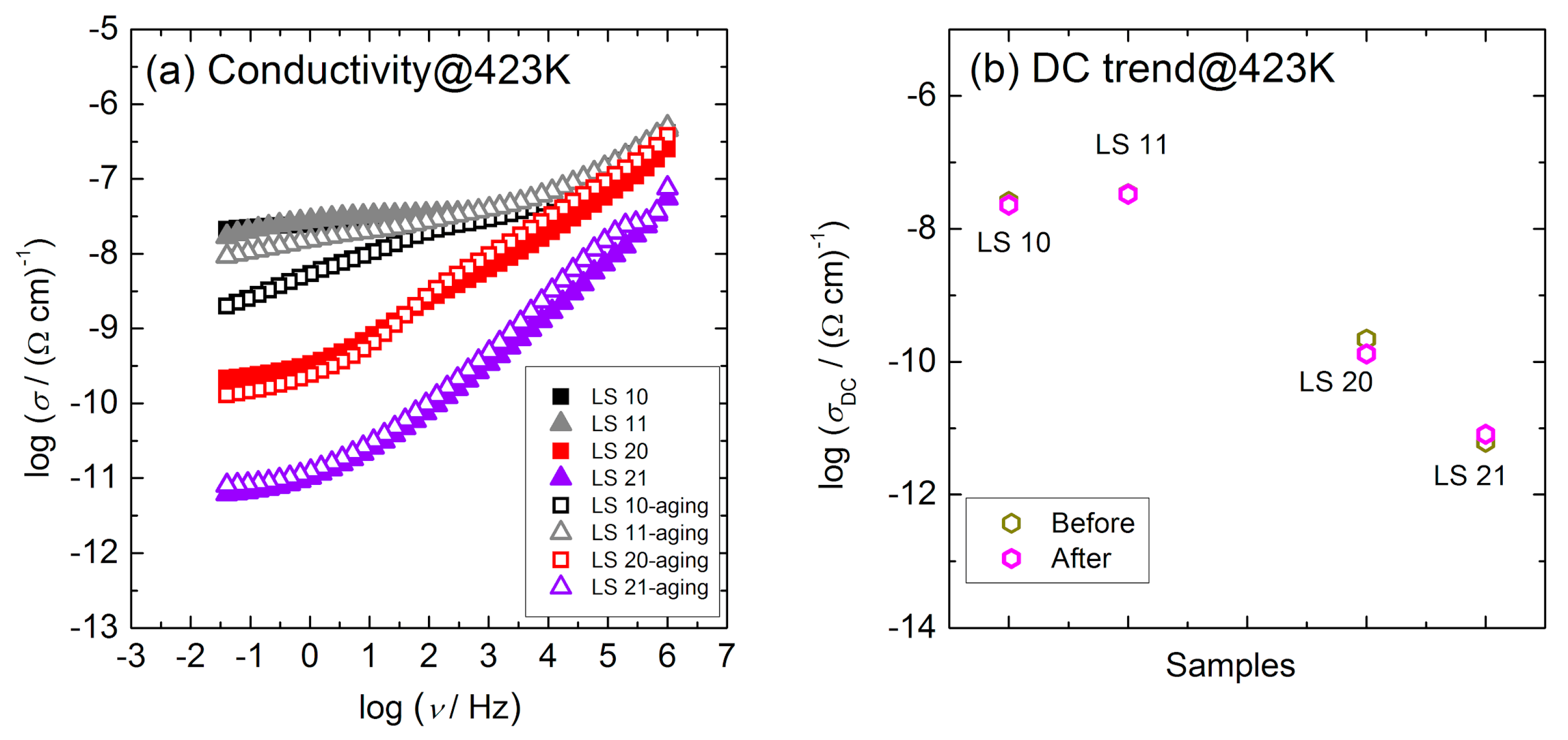
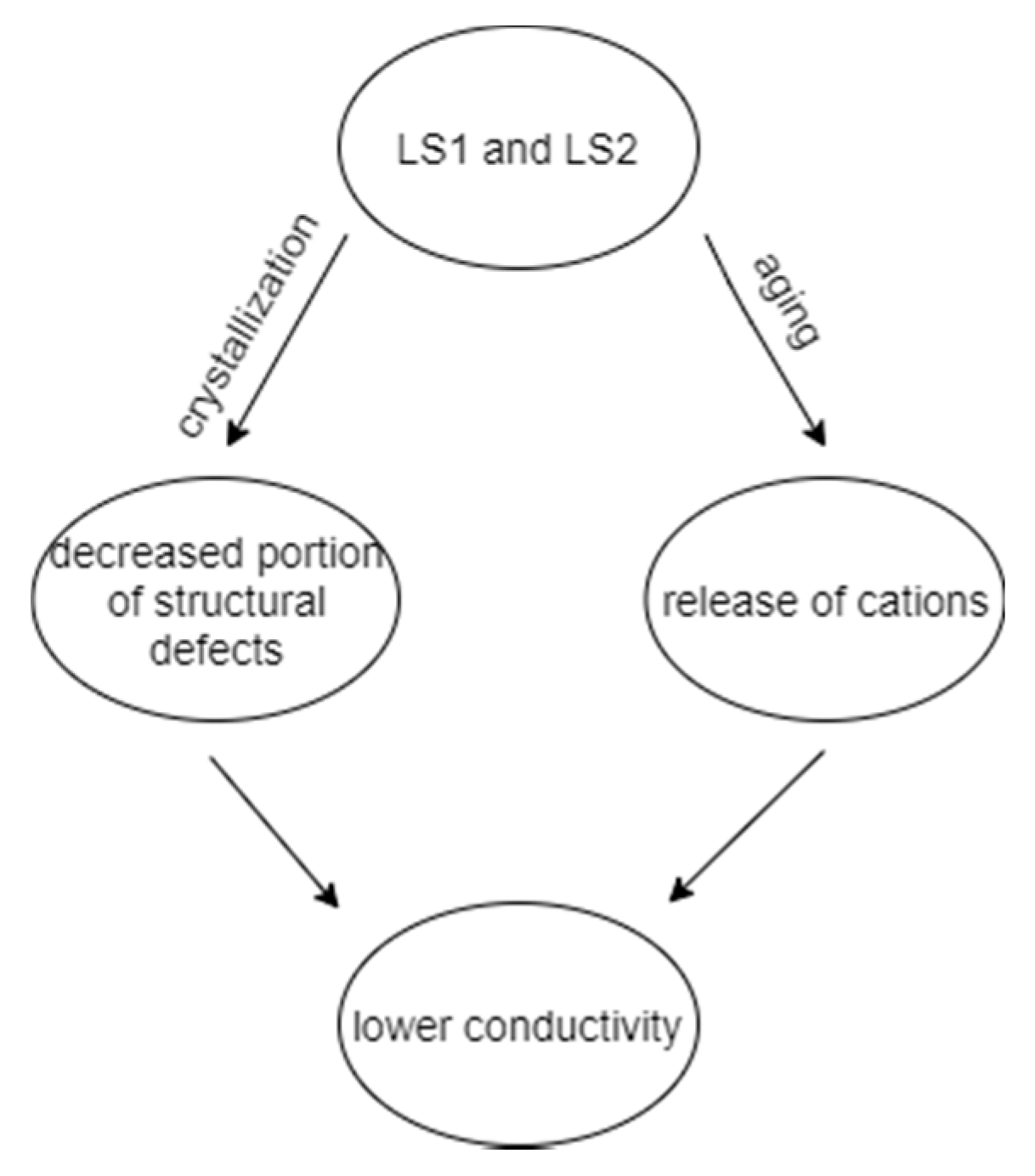
3.3. Electrical Conductivity
3.4. Limitation of This Study
3.5. The Future Tasks
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, P.-R.; Essig, M.E. Panorama of dental CAD/CAM restorative systems. Compend. Contin. Educ. Dent. 2008, 29, 482–484. [Google Scholar]
- Mörmann, W.H. CAD/CIM in Aesthetic Dentistry: CEREC 10 Year Anniversary Symposium; Quintessence Publishing (IL): Batavia, IL, USA, 1996. [Google Scholar]
- Mangano, F.G.; Admakin, O.; Bonacina, M.; Lerner, H.; Rutkunas, V.; Mangano, C. Trueness of 12 intraoral scanners in the full-arch implant impression: A comparative in vitro study. BMC Oral Health 2020, 20, 263. [Google Scholar] [CrossRef]
- Schlenz, M.A.; Schubert, V.; Schmidt, A.; Wöstmann, B.; Ruf, S.; Klaus, K. Digital versus Conventional Impression Taking Focusing on Interdental Areas: A Clinical Trial. Int. J. Environ. Res. Public Health 2020, 17, 4725. [Google Scholar] [CrossRef]
- Schmidt, A.; Klussmann, L.; Wöstmann, B.; Schlenz, M.A. Accuracy of Digital and Conventional Full-Arch Impressions in Patients: An Update. J. Clin. Med. Res. 2020, 9, 688. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Chen, J.K.; Deng, K.H.; Wang, Y.; Sun, Y.C.; Zhao, Y.J. Accuracy of three intraoral scans for primary impressions of edentulous jaws. Beijing Da Xue Xue Bao 2020, 52, 129–137. [Google Scholar] [PubMed]
- Oyar, P.; Durkan, R.; Deste, G. Effects of sintering time and hydrothermal aging on the mechanical properties of monolithic zirconia ceramic systems. J. Prosthet. Dent. 2020. [Google Scholar] [CrossRef]
- Michailova, M.; Elsayed, A.; Fabel, G.; Edelhoff, D.; Zylla, I.-M.; Stawarczyk, B. Comparison between novel strength-gradient and color-gradient multilayered zirconia using conventional and high-speed sintering. J. Mech. Behav. Biomed. Mater. 2020, 111, 103977. [Google Scholar]
- Azuma, T.; Kumagai, T.; Sato, T.; Mashio, G.; Fujimoto, T.; Akiyama, S.; Yokohara, H.; Hoshino, T.; Miyake, T. Evaluation of machinable lithium disilicate glass ceramic block for CAD/CAM. Dent. Mater. 2017, 33, e8. [Google Scholar] [CrossRef]
- Miranda, G.L.P.; Boyes, V.; Silva, N.R.F.A.; Thompson, V.P. Crystallized e-max CAD response to several surface modifications. Dent. Mater. 2018, 34, e78–e79. [Google Scholar] [CrossRef]
- Ho, G.W.; Matinlinna, J.P. Insights on Ceramics as Dental Materials. Part I: Ceramic Material Types in Dentistry. Silicon 2011, 3, 109–115. [Google Scholar] [CrossRef]
- Mackert, J.R.; Butts, M.B.; Fairhurst, C.W. The effect of the leucite transformation on dental porcelain expansion. Dent. Mater. 1986, 2, 32–36. [Google Scholar] [CrossRef]
- Dündar, M.; Artunç, C.; Toksavul, S.; Ozmen, D.; Turgan, N. Determination of elemental composition of substance lost following wear of all-ceramic materials. Int. J. Prosthodont. 2003, 16, 261–264. [Google Scholar] [PubMed]
- Fischer, H.; Maier, H.R.; Marx, R. Improved reliability of leucite reinforced glass by ion exchange. Dent. Mater. 2000, 16, 120–128. [Google Scholar] [CrossRef]
- Cattell, M.J.; Chadwick, T.C.; Knowles, J.C.; Clarke, R.L.; Lynch, E. Flexural strength optimisation of a leucite reinforced glass ceramic. Dent. Mater. 2001, 17, 21–33. [Google Scholar] [CrossRef]
- Fu, L.; Engqvist, H.; Xia, W. Glass-Ceramics in Dentistry: A Review. Materials 2020, 13, 1049. [Google Scholar] [CrossRef]
- Li, R.W.K.; Chow, T.W.; Matinlinna, J.P. Ceramic dental biomaterials and CAD/CAM technology: State of the art. J. Prosthodont. Res. 2014, 58, 208–216. [Google Scholar] [CrossRef]
- Kukiattrakoon, B.; Hengtrakool, C.; Kedjarune-Leggat, U. Effect of acidic agents on surface roughness of dental ceramics. Dent. Res. J. 2011, 8, 6–15. [Google Scholar]
- Kang, S.-H.; Chang, J.; Son, H.-H. Flexural strength and microstructure of two lithium disilicate glass ceramics for CAD/CAM restoration in the dental clinic. Restor. Dent. Endod. 2013, 38, 134–140. [Google Scholar] [CrossRef]
- Aboushelib, M.N.; Sleem, D. Microtensile bond strength of lithium disilicate ceramics to resin adhesives. J. Adhes. Dent. 2014, 16, 547–552. [Google Scholar]
- Zhao, T.; Qin, Y.; Zhang, P.; Wang, B.; Yang, J.-F. High-performance, reaction sintered lithium disilicate glass–ceramics. Ceram. Int. 2014, 40, 12449–12457. [Google Scholar] [CrossRef]
- Abdulkader, K.F.; Elnaggar, G.A.E.; Kheiralla, L.S. Shear bond strength of cemented zirconia-reinforced lithium silicate ceramics (Celtra Duo) with two surface treatments (in vitro study). J. Adhes. Sci. Technol. 2020, 1–17. [Google Scholar] [CrossRef]
- Shakal, M.; Oraby, H. Translucency evaluation of zirconia lithium silicate and lithium disilicate glass ceramic with different thicknesses pre and post cementation. Egyptian Dent. J. 2018, 64, 1623–1630. [Google Scholar] [CrossRef][Green Version]
- Chen, X.-P.; Xiang, Z.-X.; Song, X.-F.; Yin, L. Machinability: Zirconia-reinforced lithium silicate glass ceramic versus lithium disilicate glass ceramic. J. Mech. Behav. Biomed. Mater. 2020, 101, 103435. [Google Scholar] [CrossRef]
- Rinke, S.; Pfitzenreuter, T.; Leha, A.; Roediger, M.; Ziebolz, D. Clinical evaluation of chairside-fabricated partial crowns composed of zirconia-reinforced lithium silicate ceramics: 3-year results of a prospective practice-based study. J. Esthet. Restor. Dent. 2020, 32, 226–235. [Google Scholar] [CrossRef]
- Schweitzer, F.; Spintzyk, S.; Geis-Gerstorfer, J.; Huettig, F. Influence of minimal extended firing on dimensional, optical, and mechanical properties of crystalized zirconia-reinforced lithium silicate glass ceramic. J. Mech. Behav. Biomed. Mater. 2020, 104, 103644. [Google Scholar] [CrossRef]
- Diniz, V.; Condé Oliveira Prado, P.H.; Meireles Rodrigues, J.V.; Monteiro, J.B.; Zucuni, C.; Valandro, L.F.; Melo, R.M. Ceramic firing protocols and thermocycling: Effects on the load-bearing capacity under fatigue of a bonded zirconia lithium silicate glass-ceramic. J. Mech. Behav. Biomed. Mater. 2020, 110, 103963. [Google Scholar] [CrossRef] [PubMed]
- Jakovac, M.; Živko-Babić, J.; Ćurković, L.; Aurer, A. Measurement of ion elution from dental ceramics. J. Eur. Ceram. Soc. 2006, 26, 1695–1700. [Google Scholar] [CrossRef]
- Lakhloufi, S.; Labjar, H.; El Hamdouni, Y.; Bouhouche, I.; Labjar, N.; Dahrouch, A.; Serghini-Idriissi, M.; El Yamani, A.; El Hajjaji, S. Electrochemical Behavior and Surface Characterization of Two Dental Materials: Glass Ceramic and Zirconia in Artificial Saliva. J. Fail. Anal. Prev. 2020, 20, 1771–1783. [Google Scholar] [CrossRef]
- Chevalier, J.; Gremillard, L.; Deville, S. Low-Temperature Degradation of Zirconia and Implications for Biomedical Implants. Annu. Rev. Mater. Res. 2007, 37, 1–32. [Google Scholar] [CrossRef]
- Swain, M.V. Impact of oral fluids on dental ceramics: What is the clinical relevance? Dent. Mater. 2014, 30, 33–42. [Google Scholar] [CrossRef]
- Porojan, L.; Vasiliu, R.-D.; Bîrdeanu, M.-I.; Porojan, S.-D. Surface Characterization and Optical Properties of Reinforced Dental Glass-Ceramics Related to Artificial Aging. Molecules 2020, 25, 3407. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.acdrc.com/wp-content/uploads/2015/03/IPS-e-max-CAD-Chairside.pdf (accessed on 29 October 2020).
- de Rijk, W.G.; de Rijk, W.G.; Jennings, K.A.; Menis, D.L. A comparison of chemical durability test solutions for dental porcelains. Biomed. Eng. IV 1985, 152–155. [Google Scholar] [CrossRef]
- Skoko, Z.; Popovic, J.; Dekanic, K.; Kolbas, V.; Popovic, S. XBroad: Program for extracting basic microstructure information from X-ray diffraction patterns in few clicks. J. Appl. Crystallogr. 2012, 45, 594–597. [Google Scholar] [CrossRef]
- Skoko, Ž.; Popović, J.; Dekanić, K. Insight into microstructural development by XBroad program: Case of in-situ formation of the Al–Zn solid solution. J. Phys. Chem. Solids 2015, 80, 34–38. [Google Scholar] [CrossRef]
- Pavić, L.; Nikolić, J.; Graça, M.P.F.; Costa, B.F.O.; Valente, M.A.; Skoko, Ž.; Šantić, A.; Moguš-Milanković, A. Effect of controlled crystallization on polaronic transport in phosphate-based glass-ceramics. Int. J. Appl. Glass Sci. 2020, 11, 97–111. [Google Scholar] [CrossRef]
- Pavić, L.; Šantić, A.; Nikolić, J.; Mošner, P.; Koudelka, L.; Pajić, D.; Moguš-Milanković, A. Nature of mixed electrical transport in Ag2O–ZnO–P2O5 glasses containing WO3 and MoO3. Electrochim. Acta 2018, 276, 434–445. [Google Scholar] [CrossRef]
- Šantić, A.; Banhatti, R.D.; Pavić, L.; Ertap, H.; Yüksek, M.; Karabulut, M.; Moguš-Milanković, A. Polaronic transport in iron phosphate glasses containing HfO and CeO. Phys. Chem. Chem. Phys. 2017, 19, 3999–4009. [Google Scholar] [CrossRef] [PubMed]
- Thoms, E.; Sippel, P.; Reuter, D.; Weiß, M.; Loidl, A.; Krohns, S. Dielectric study on mixtures of ionic liquids. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.R.; Gileadi, E. Physical Interpretation of the Warburg Impedance. Corrosion 1995, 51, 664–671. [Google Scholar] [CrossRef]
- Žic, M. Solving CNLS problems by using Levenberg-Marquardt algorithm: A new approach to avoid off-limits values during a fit. J. Electroanal. Chem. 2017, 799, 242–248. [Google Scholar] [CrossRef]
- Žic, M.; Pereverzyev, S. Optimizing noisy CNLS problems by using Nelder-Mead algorithm: A new method to compute simplex step efficiency. J. Electroanal. Chem. 2019, 851, 113439. [Google Scholar] [CrossRef]
- Žic, M.; Subotić, V.; Pereverzyev, S.; Fajfar, I. Solving CNLS problems using Levenberg-Marquardt algorithm: A new fitting strategy combining limits and a symbolic Jacobian matrix. J. Electroanal. Chem. 2020, 866, 114171. [Google Scholar] [CrossRef]
- Žic, M.; Pereverzyev, S., Jr.; Subotić, V.; Pereverzyev, S. Adaptive multi-parameter regularization approach to construct the distribution function of relaxation times. GEM 2020, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Bazant, M.Z. Electrochemical Impedance Imaging via the Distribution of Diffusion Times. Phys. Rev. Lett. 2018, 120, 116001. [Google Scholar] [CrossRef]
- Pereverzev, S.V.; Solodky, S.G.; Vasylyk, V.B.; Žic, M. Regularized Collocation in Distribution of Diffusion Times Applied to Electrochemical Impedance Spectroscopy. Comput. Methods Appl. Math. 2020, 20, 517–530. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jakovac, M.; Klaser, T.; Radatović, B.; Skoko, Ž.; Pavić, L.; Žic, M. Surface Characterization and Conductivity of Two Types of Lithium-Based Glass Ceramics after Accelerating Ageing. Materials 2020, 13, 5632. https://doi.org/10.3390/ma13245632
Jakovac M, Klaser T, Radatović B, Skoko Ž, Pavić L, Žic M. Surface Characterization and Conductivity of Two Types of Lithium-Based Glass Ceramics after Accelerating Ageing. Materials. 2020; 13(24):5632. https://doi.org/10.3390/ma13245632
Chicago/Turabian StyleJakovac, Marko, Teodoro Klaser, Borna Radatović, Željko Skoko, Luka Pavić, and Mark Žic. 2020. "Surface Characterization and Conductivity of Two Types of Lithium-Based Glass Ceramics after Accelerating Ageing" Materials 13, no. 24: 5632. https://doi.org/10.3390/ma13245632
APA StyleJakovac, M., Klaser, T., Radatović, B., Skoko, Ž., Pavić, L., & Žic, M. (2020). Surface Characterization and Conductivity of Two Types of Lithium-Based Glass Ceramics after Accelerating Ageing. Materials, 13(24), 5632. https://doi.org/10.3390/ma13245632






