Structural, Electronic, and Optical Properties of CsPb(Br1−xClx)3 Perovskite: First-Principles Study with PBE–GGA and mBJ–GGA Methods
Abstract
1. Introduction
2. Computational Method
3. Results
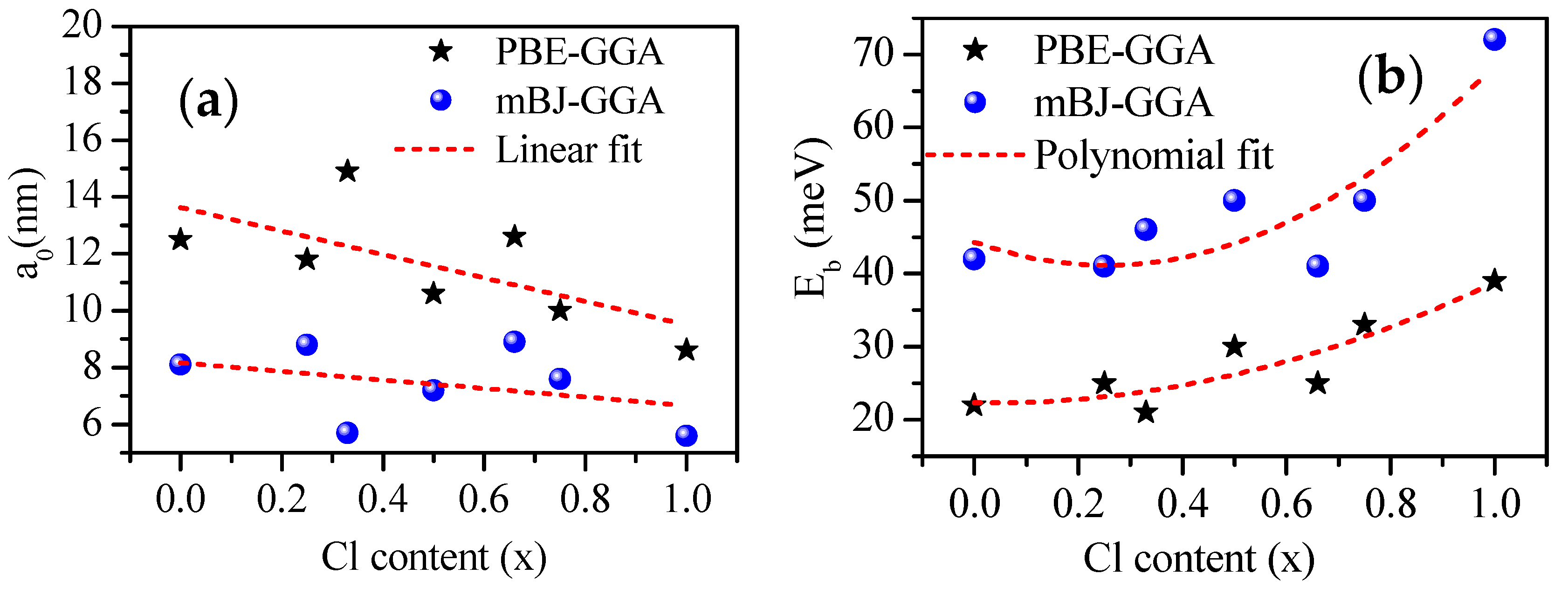
3.1. Structural Properties
3.2. Electronic Properties
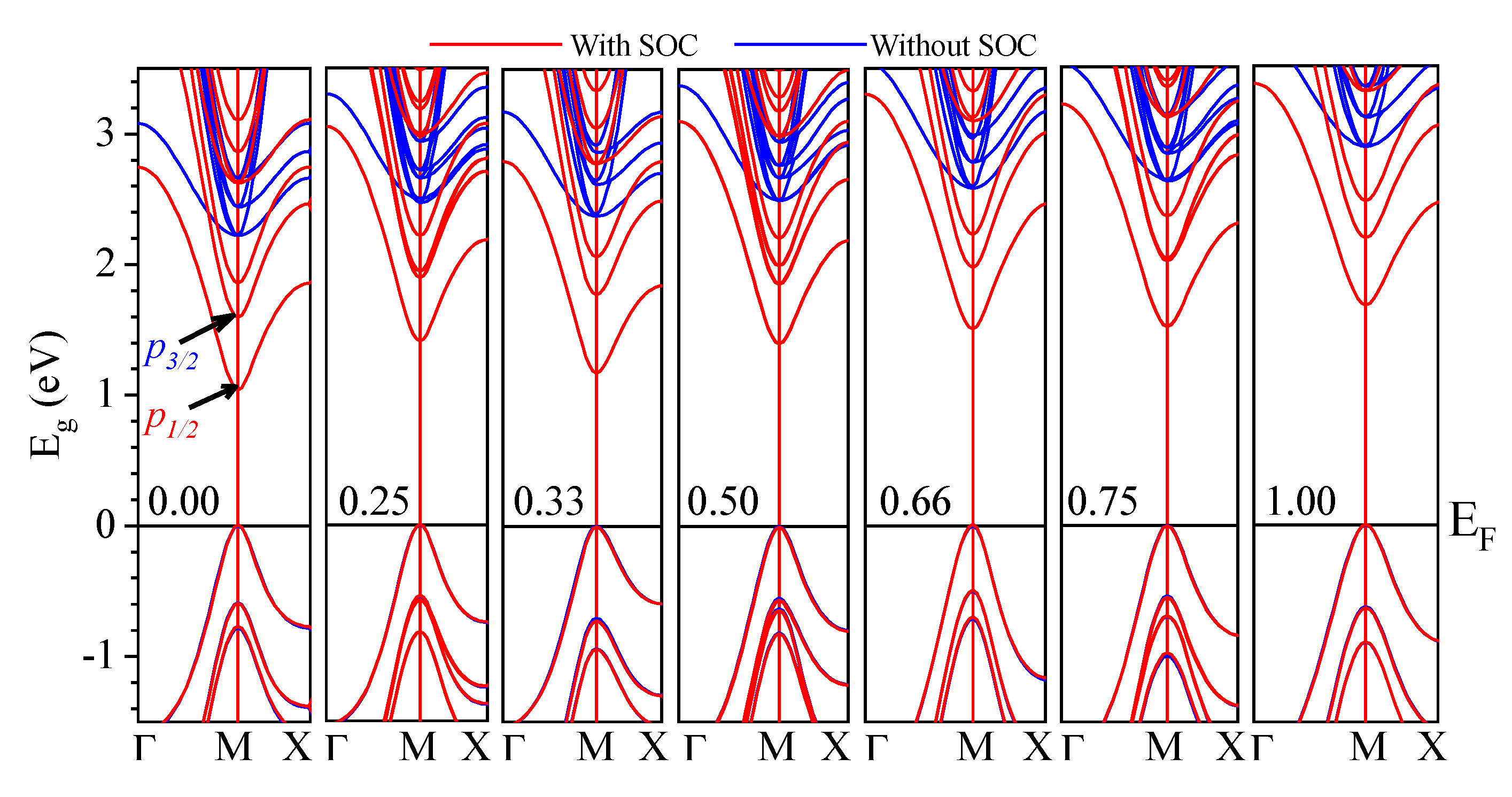
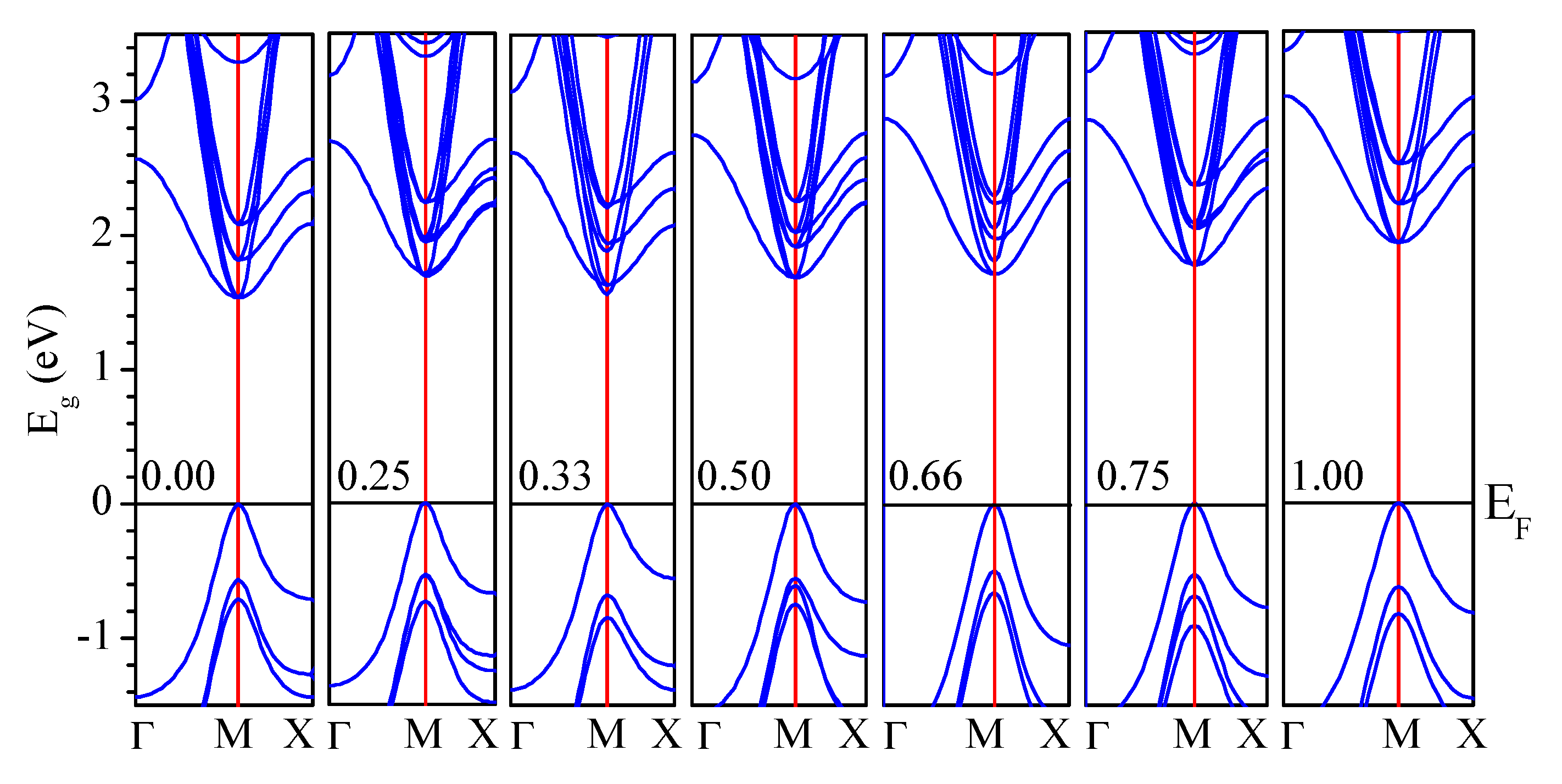
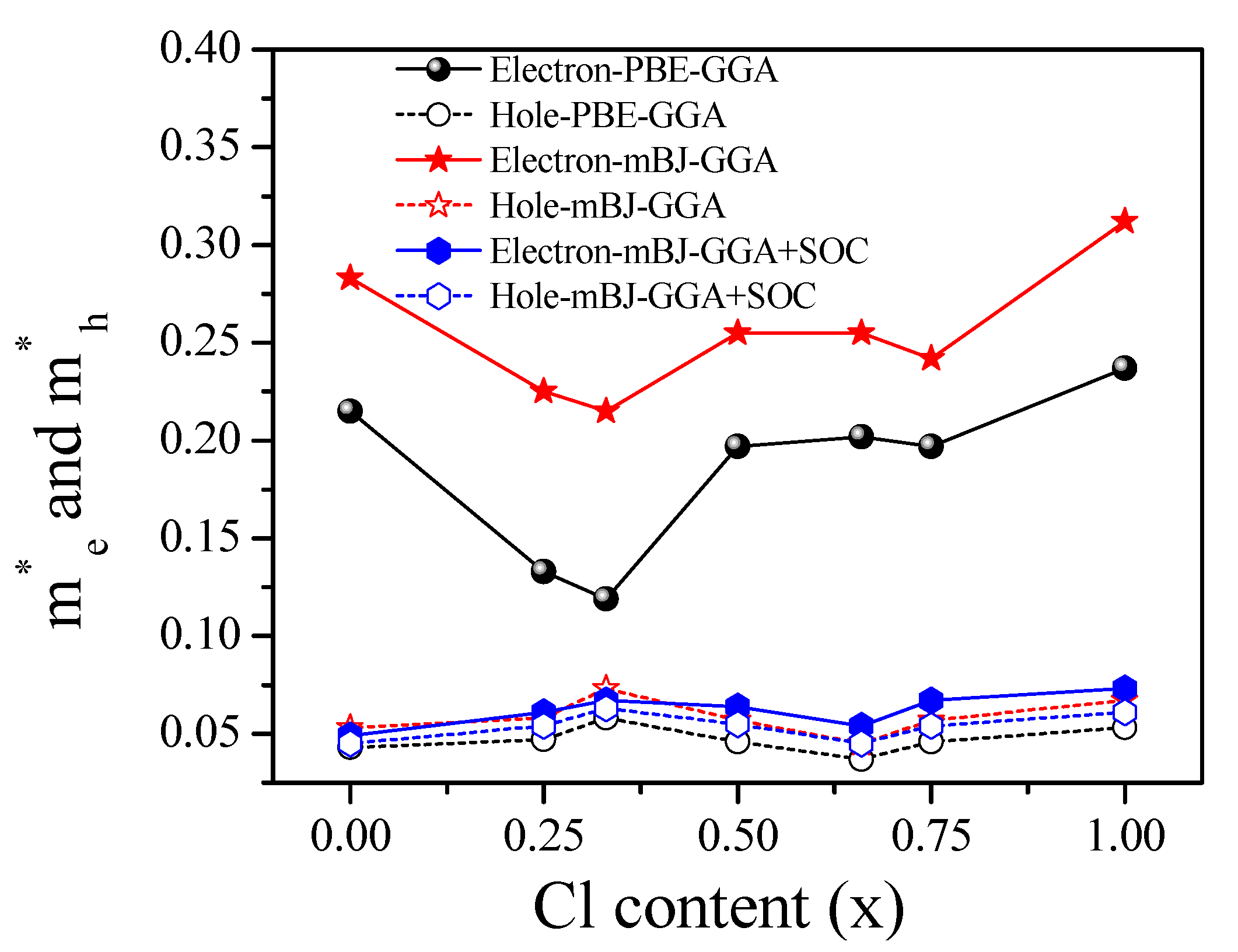
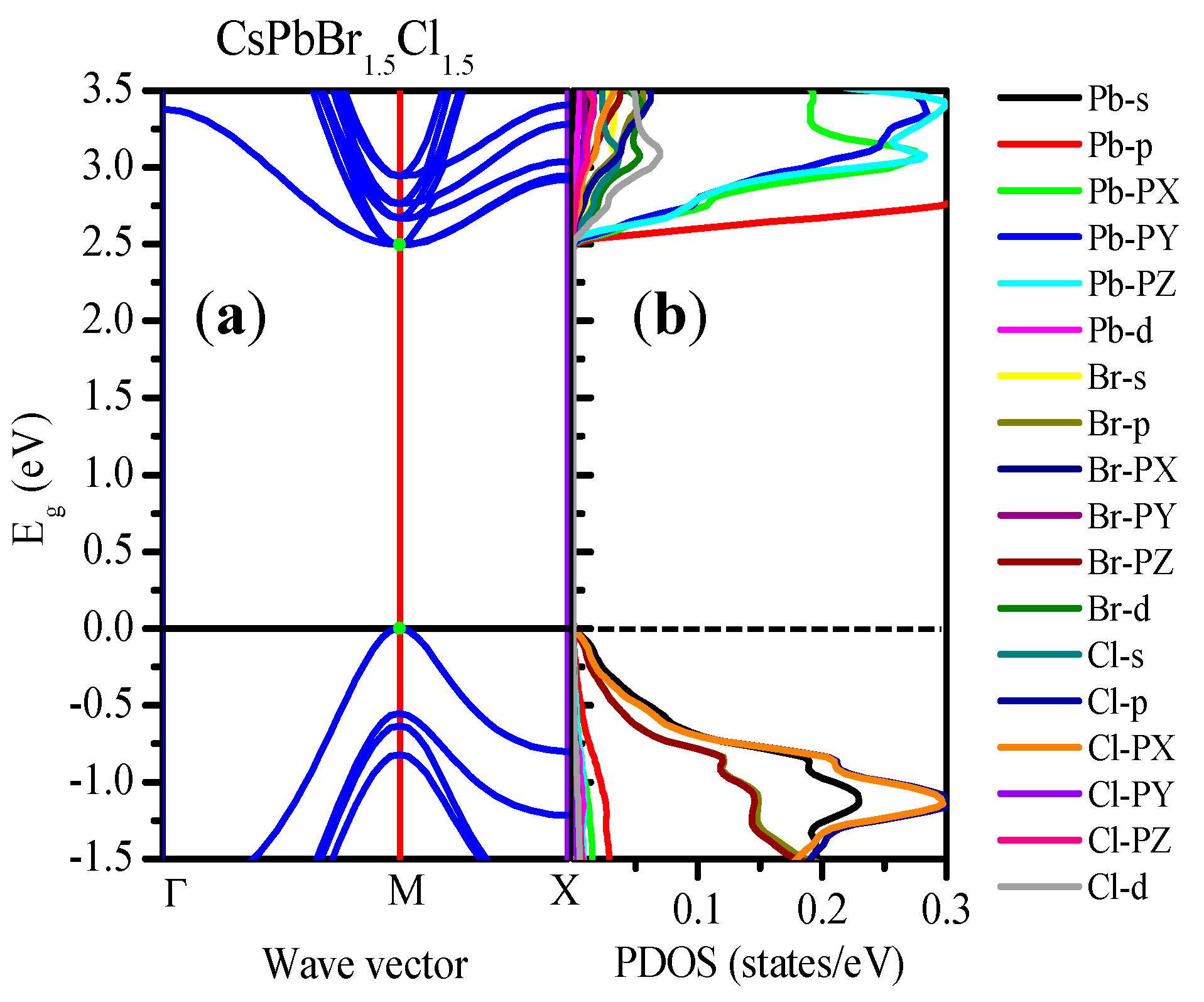
3.2.1. Electronic Band Structure
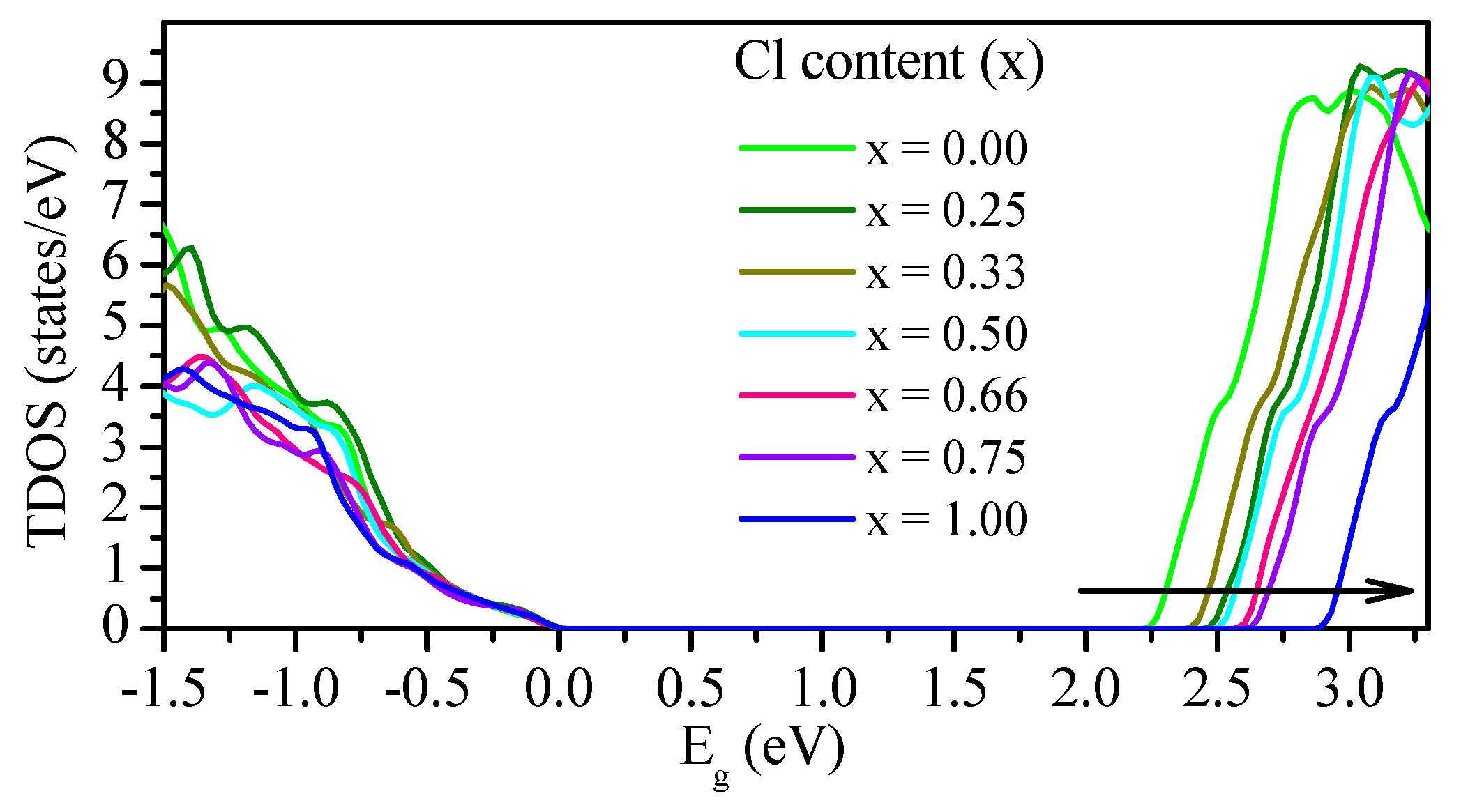
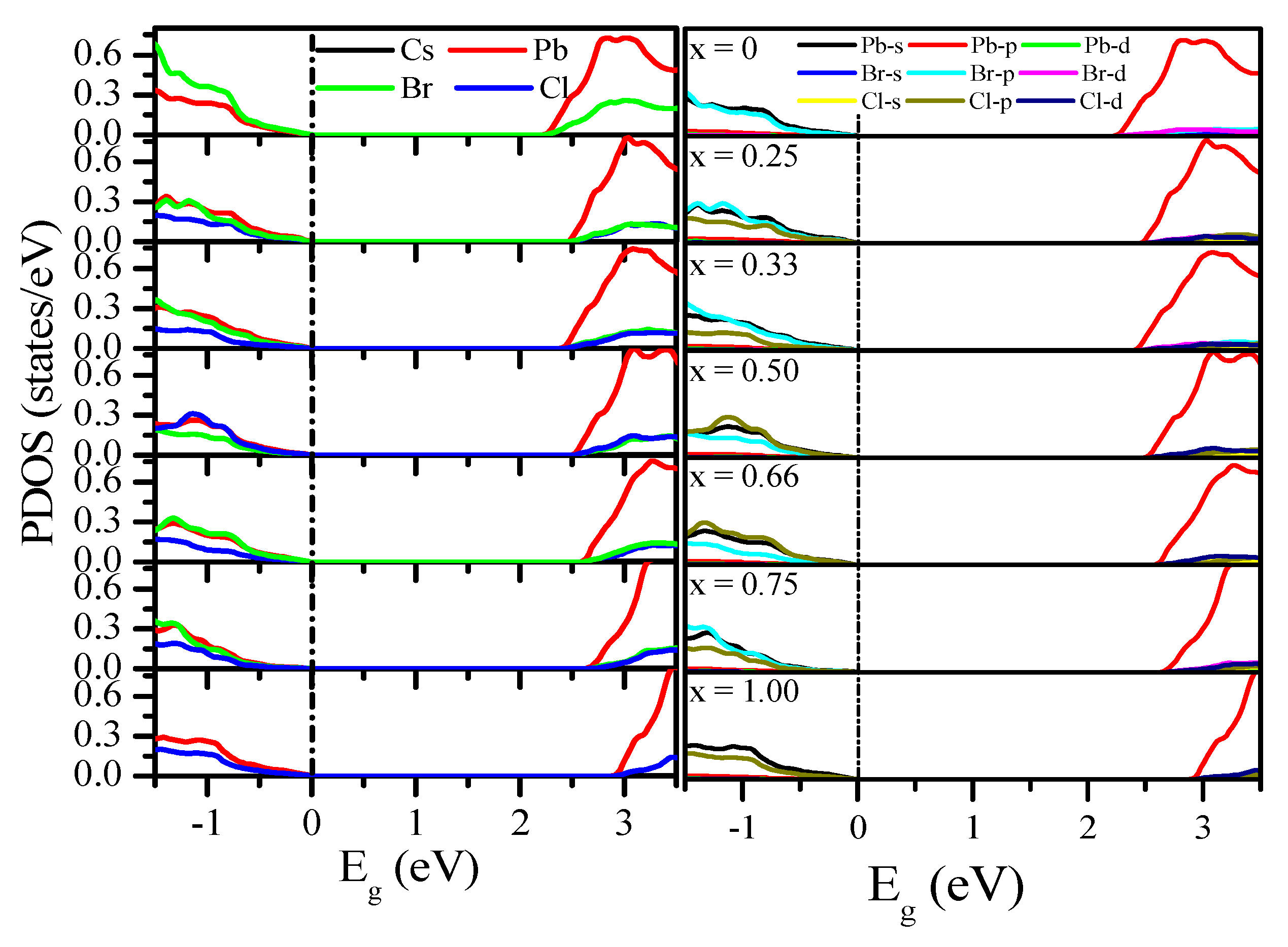
3.2.2. Density of States (DOS)
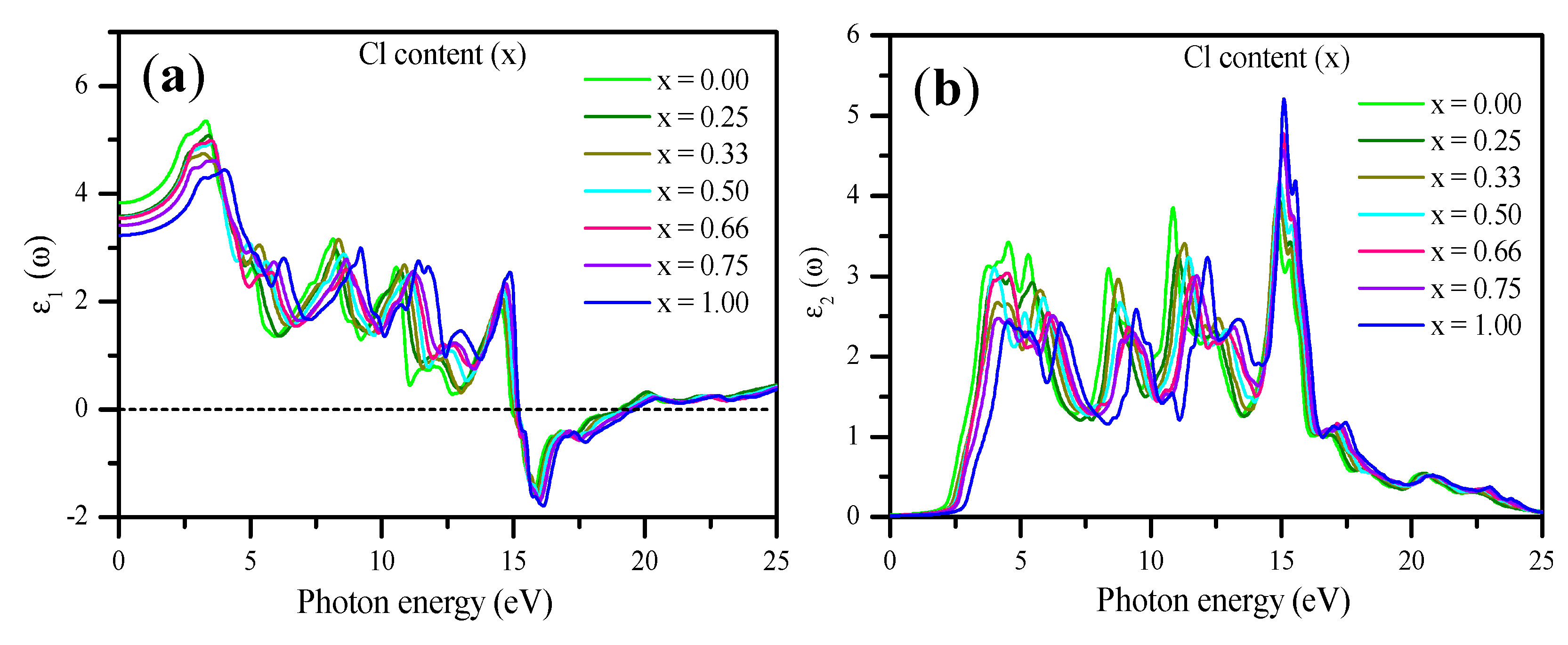
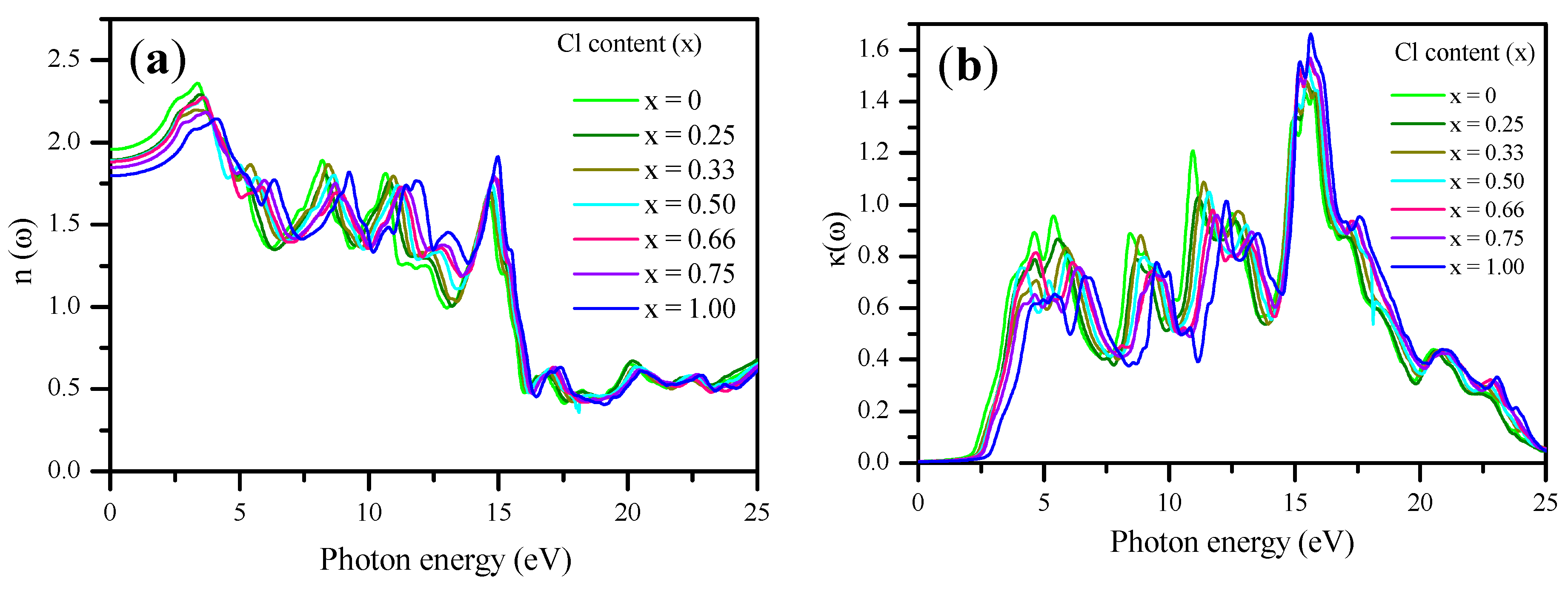
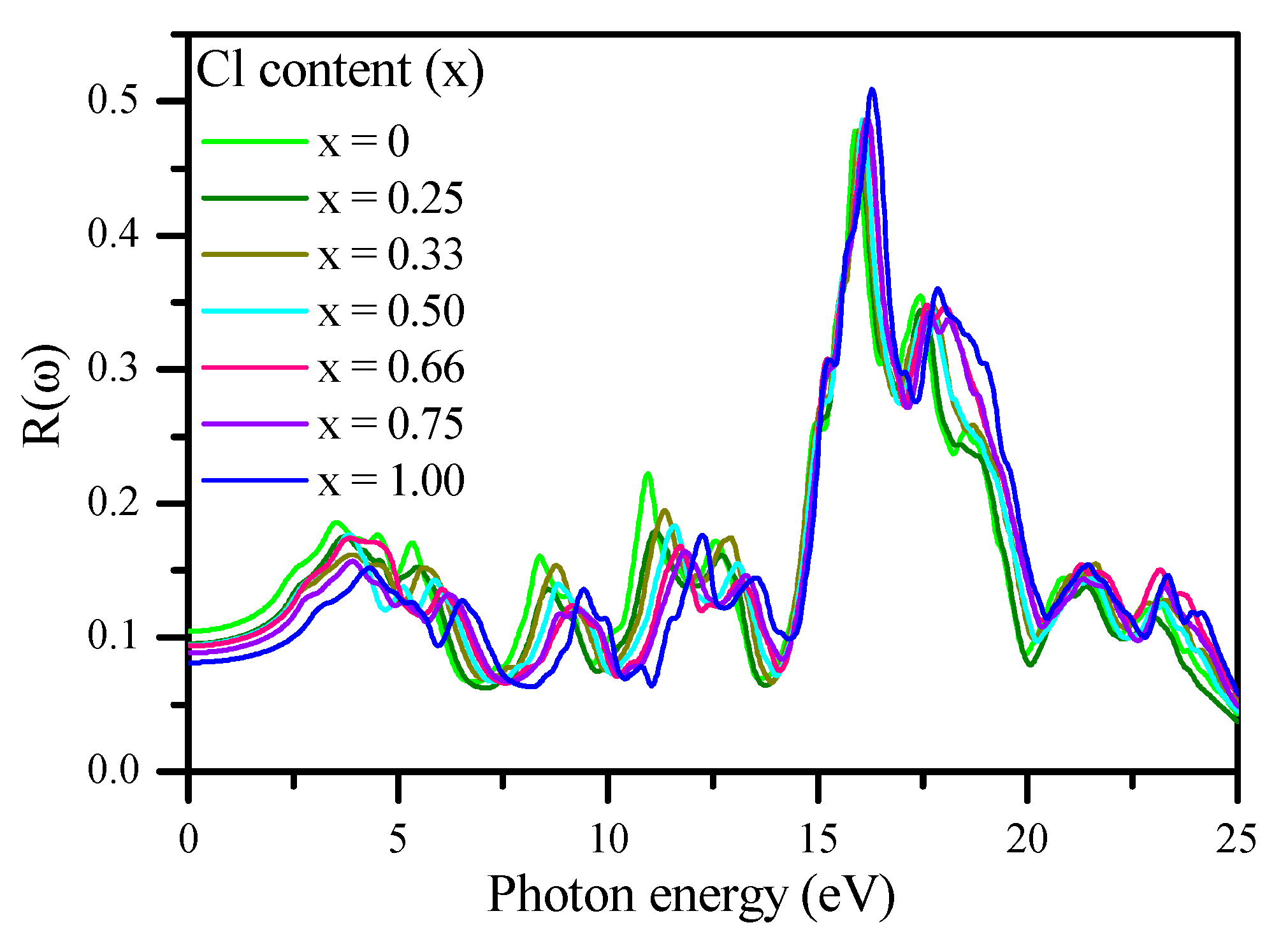
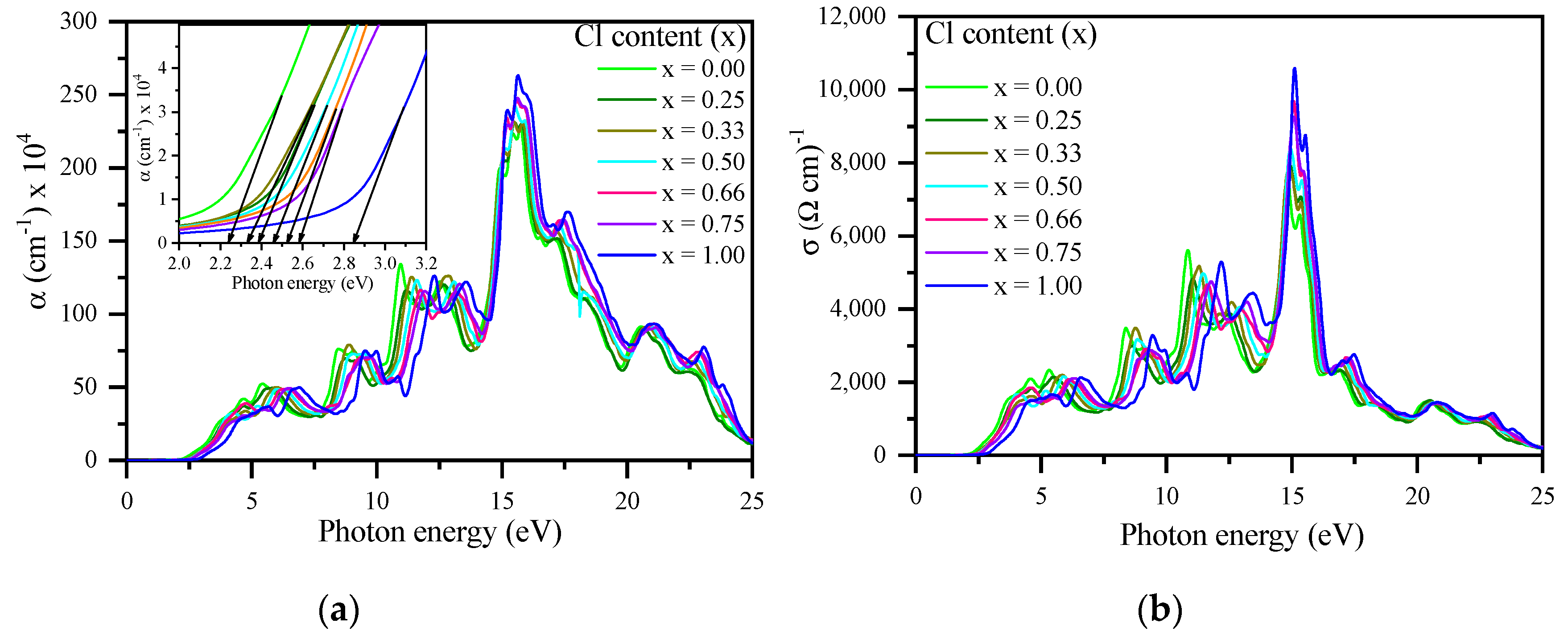
3.3. Optical Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Haddad, J.; Krogmeier, B.; Klingebiel, B.; Krückemeier, L.; Melhem, S.; Liu, Z.; Hüpkes, J.; Mathur, S.; Kirchartz, T. Analyzing Interface Recombination in Lead-Halide Perovskite Solar Cells with Organic and Inorganic Hole-Transport Layers. Adv. Mater. Interfaces 2020, 7, 2000366. [Google Scholar] [CrossRef]
- Zhao, Y.; Duan, J.; Wang, Y.; Yang, X.; Tang, Q. Precise stress control of inorganic perovskite films for carbon-based solar cells with an ultrahigh voltage of 1.622 V. Nano Energy 2020, 67, 104286. [Google Scholar] [CrossRef]
- Duan, J.; Wei, J.; Tang, Q.; Li, Q. Unveiling the interfacial charge extraction kinetics in inorganic perovskite solar cells with formamidinium lead halide (FAPbX3) nanocrystals. Sol. Energy 2020, 195, 644–650. [Google Scholar] [CrossRef]
- Ouedraogo, N.A.N.; Chen, Y.; Xiao, Y.Y.; Meng, Q.; Han, C.B.; Yan, H.; Zhang, Y. Stability of all-inorganic perovskite solar cells. Nano Energy 2020, 67, 104249. [Google Scholar] [CrossRef]
- Feng, J.; Zhu, X.; Yang, Z.; Zhang, X.; Niu, J.; Wang, Z.; Zuo, S.; Priya, S.; Liu, S. (Frank); Yang, D. Record Efficiency Stable Flexible Perovskite Solar Cell Using Effective Additive Assistant Strategy. Adv. Mater. 2018, 30, e1801418. [Google Scholar] [CrossRef]
- Wang, K.; Jin, Z.; Liang, L.; Bian, H.; Bai, D.; Wang, H.; Zhang, J.; Wang, Q.; Shengzhong, L. All-inorganic cesium lead iodide perovskite solar cells with stabilized efficiency beyond 15%. Nat. Commun. 2018, 9, 1–8. [Google Scholar] [CrossRef]
- Kim, H.S.; Lee, C.R.; Im, J.H.; Lee, K.B.; Moehl, T.; Marchioro, A.; Moon, S.J.; Humphry-Baker, R.; Yum, J.H.; Moser, J.E.; et al. Lead iodide perovskite sensitized all-solid-state submicron thin film mesoscopic solar cell with efficiency exceeding 9%. Sci. Rep. 2012, 2, 591. [Google Scholar] [CrossRef]
- Eperon, G.E.; Paternò, G.M.; Sutton, R.J.; Zampetti, A.; Haghighirad, A.A.; Cacialli, F.; Snaith, H.J. Inorganic caesium lead iodide perovskite solar cells. J. Mater. Chem. A 2015, 3, 19688–19695. [Google Scholar] [CrossRef]
- Lin, K.; Xing, J.; Quan, L.N.; de Arquer, F.P.G.; Gong, X.; Lu, J.; Xie, L.; Zhao, W.; Zhang, D.; Yan, C.; et al. Perovskite light-emitting diodes with external quantum efficiency exceeding 20 per cent. Nature 2018, 562, 245–248. [Google Scholar] [CrossRef]
- Matsushima, T.; Bencheikh, F.; Komino, T.; Leyden, M.R.; Sandanayaka, A.S.D.; Qin, C.; Adachi, C. High performance from extraordinarily thick organic light-emitting diodes. Nature 2019, 572, 502–506. [Google Scholar] [CrossRef]
- Zhang, Q.; Tavakoli, M.M.; Gu, L.; Zhang, D.; Tang, L.; Gao, Y.; Guo, J.; Lin, Y.; Leung, S.F.; Poddar, S.; et al. Efficient metal halide perovskite light-emitting diodes with significantly improved light extraction on nanophotonic substrates. Nat. Commun. 2019, 10, 727. [Google Scholar] [CrossRef]
- Veldhuis, S.A.; Boix, P.P.; Yantara, N.; Li, M.; Sum, T.C.; Mathews, N.; Mhaisalkar, S.G. Perovskite Materials for Light-Emitting Diodes and Lasers. Adv. Mater. 2016, 28, 6804–6834. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.K.; Moghaddam, R.S.; Lai, M.L.; Docampo, P.; Higler, R.; Deschler, F.; Price, M.; Sadhanala, A.; Pazos, L.M.; Credgington, D.; et al. Bright light-emitting diodes based on organometal halide perovskite. Nat. Nanotechnol. 2014, 9, 687–692. [Google Scholar] [CrossRef] [PubMed]
- Even, J.; Pedesseau, L.; Jancu, J.M.; Katan, C. DFT and k · p modelling of the phase transitions of lead and tin halide perovskites for photovoltaic cells. Phys. Status Solidi Rapid Res. Lett. 2014, 8, 31–35. [Google Scholar] [CrossRef]
- Droseros, N.; Longo, G.; Brauer, J.C.; Sessolo, M.; Bolink, H.J.; Banerji, N. Origin of the Enhanced Photoluminescence Quantum Yield in MAPbBr3 Perovskite with Reduced Crystal Size. ACS Energy Lett. 2018, 3, 1458–1466. [Google Scholar] [CrossRef]
- Protesescu, L.; Yakunin, S.; Bodnarchuk, M.I.; Krieg, F.; Caputo, R.; Hendon, C.H.; Yang, R.X.; Walsh, A.; Kovalenko, M.V. Nanocrystals of Cesium Lead Halide Perovskites (CsPbX3, X = Cl, Br, and I): Novel Optoelectronic Materials Showing Bright Emission with Wide Color Gamut. Nano Lett. 2015, 15, 3692–3696. [Google Scholar] [CrossRef]
- Nedelcu, G.; Protesescu, L.; Yakunin, S.; Bodnarchuk, M.I.; Grotevent, M.J.; Kovalenko, M.V. Fast Anion-Exchange in Highly Luminescent Nanocrystals of Cesium Lead Halide Perovskites (CsPbX3, X = Cl, Br, I). Nano Lett. 2015, 15, 5635–5640. [Google Scholar] [CrossRef]
- Liashenko, T.G.; Cherotchenko, E.D.; Pushkarev, A.P.; Pakštas, V.; Naujokaitis, A.; Khubezhov, S.A.; Polozkov, R.G.; Agapev, K.B.; Zakhidov, A.A.; Shelykh, I.A.; et al. Electronic structure of CsPbBr3-xClx perovskites: Synthesis, experimental characterization, and DFT simulations. Phys. Chem. Chem. Phys. 2019, 21, 18930–18938. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, X.; Jiang, Q.; Wang, P.; Yin, Z.; Zhang, X.; Tan, H.; Yang, Y.M.; Wei, M.; Sutherland, B.R.; et al. Ultra-bright and highly efficient inorganic based perovskite light-emitting diodes. Nat. Commun. 2017, 8, 15640. [Google Scholar] [CrossRef]
- Song, J.; Li, J.; Li, X.; Xu, L.; Dong, Y.; Zeng, H. Quantum Dot Light-Emitting Diodes Based on Inorganic Perovskite Cesium Lead Halides (CsPbX3). Adv. Mater. 2015, 27, 7162–7167. [Google Scholar] [CrossRef]
- Zhang, X.; Jin, Z.; Zhang, J.; Bai, D.; Bian, H.; Wang, K.; Sun, J.; Wang, Q.; Liu, S.F. All-Ambient Processed Binary CsPbBr3-CsPb2Br5 Perovskites with Synergistic Enhancement for High-Efficiency Cs-Pb-Br-Based Solar Cells. ACS Appl. Mater. Interfaces 2018, 10, 7145–7154. [Google Scholar] [CrossRef]
- Stoumpos, C.C.; Malliakas, C.D.; Peters, J.A.; Liu, Z.; Sebastian, M.; Im, J.; Chasapis, T.C.; Wibowo, A.C.; Chung, D.Y.; Freeman, A.J.; et al. Crystal growth of the perovskite semiconductor CsPbBr3: A new material for high-energy radiation detection. Cryst. Growth Des. 2013, 13, 2722–2727. [Google Scholar] [CrossRef]
- Ahmad, M.; Rehman, G.; Ali, L.; Shafiq, M.; Iqbal, R.; Ahmad, R.; Khan, T.; Jalali-Asadabadi, S.; Maqbool, M.; Ahmad, I. Structural, electronic and optical properties of CsPbX3 (X=Cl, Br, I) for energy storage and hybrid solar cell applications. J. Alloys Compd. 2017, 705, 828–839. [Google Scholar] [CrossRef]
- Yaffe, O.; Guo, Y.; Hull, T.; Stoumpos, C.C.; Tan, L.Z.; Egger, D.A.; Zheng, F.; Szpak, G.; Semonin, O.E.; Beecher, A.N.; et al. The nature of dynamic disorder in lead halide perovskite crystals (Conference Presentation). Phys. Chem. Interfaces Nanomater. XV 2016. [Google Scholar] [CrossRef]
- Rodova, M.; Brozek, J.; Knizek, K.; Nitsch, K. Phase transitions in ternary caesium lead bromide. J. Therm. Anal. Calorim. 2003, 71, 667–673. [Google Scholar] [CrossRef]
- Bechtel, J.S.; Van Der Ven, A. First-principles thermodynamics study of phase stability in inorganic halide perovskite solid solutions. Phys. Rev. Mater. 2018, 2, 045401. [Google Scholar] [CrossRef]
- Zhang, M.; Zheng, Z.; Fu, Q.; Chen, Z.; He, J.; Zhang, S.; Yan, L.; Hu, Y.; Luo, W. Growth and characterization of all-inorganic lead halide perovskite semiconductor CsPbBr3 single crystals. CrystEngComm 2017, 19, 6797–6803. [Google Scholar] [CrossRef]
- He, Y.; Matei, L.; Jung, H.J.; McCall, K.M.; Chen, M.; Stoumpos, C.C.; Liu, Z.; Peters, J.A.; Chung, D.Y.; Wessels, B.W.; et al. High spectral resolution of gamma-rays at room temperature by perovskite CsPbBr 3 single crystals. Nat. Commun. 2018, 9, 1–8. [Google Scholar] [CrossRef]
- Qiao, B.; Song, P.; Cao, J.; Zhao, S.; Shen, Z.; Di, G.; Liang, Z.; Xu, Z.; Song, D.; Xu, X. Water-resistant, monodispersed and stably luminescent CsPbBr3/CsPb2Br5 core-shell-like structure lead halide perovskite nanocrystals. Nanotechnology 2017, 28, 445602. [Google Scholar] [CrossRef]
- Bertolotti, F.; Protesescu, L.; Kovalenko, M.V.; Yakunin, S.; Cervellino, A.; Billinge, S.J.L.; Terban, M.W.; Pedersen, J.S.; Masciocchi, N.; Guagliardi, A. Coherent Nanotwins and Dynamic Disorder in Cesium Lead Halide Perovskite Nanocrystals. ACS Nano 2017, 11, 3819–3831. [Google Scholar] [CrossRef]
- Dos Reis, R.; Yang, H.; Ophus, C.; Ercius, P.; Bizarri, G.; Perrodin, D.; Shalapska, T.; Bourret, E.; Ciston, J.; Dahmen, U. Determination of the structural phase and octahedral rotation angle in halide perovskites. Appl. Phys. Lett. 2018, 112, 071901. [Google Scholar] [CrossRef]
- Cottingham, P.; Brutchey, R.L. On the crystal structure of colloidally prepared CsPbBr3 quantum dots. Chem. Commun. 2016, 52, 5246–5249. [Google Scholar] [CrossRef]
- Wolf, C.; Lee, T.W. Exciton and lattice dynamics in low-temperature processable CsPbBr3 thin-films. Mater. Today Energy 2018, 7, 199–207. [Google Scholar] [CrossRef]
- Zhang, M.; Zheng, Z.; Fu, Q.; Chen, Z.; He, J.; Zhang, S.; Chen, C.; Luo, W. Synthesis and single crystal growth of perovskite semiconductor CsPbBr3. J. Cryst. Growth 2018, 484, 37–42. [Google Scholar] [CrossRef]
- Goesten, M.G.; Hoffmann, R. Mirrors of Bonding in Metal Halide Perovskites. J. Am. Chem. Soc. 2018, 140, 12996–13010. [Google Scholar] [CrossRef]
- Møller, C.K. The Structure of Perovskite-Like Cæsium Plumbo Trihalides. Mater. Fys. Medd. Dan. Vid. Selsk 1959, 32, 1–27. [Google Scholar]
- Yang, R.X.; Skelton, J.M.; Da Silva, E.L.; Frost, J.M.; Walsh, A. Spontaneous octahedral tilting in the cubic inorganic cesium halide perovskites CsSnX3 and CsPbX3 (X = F, Cl, Br, I). J. Phys. Chem. Lett. 2017, 8, 4720–4726. [Google Scholar] [CrossRef] [PubMed]
- Kirschner, M.S.; Diroll, B.T.; Guo, P.; Harvey, S.M.; Helweh, W.; Flanders, N.C.; Brumberg, A.; Watkins, N.E.; Leonard, A.A.; Evans, A.M.; et al. Photoinduced, reversible phase transitions in all-inorganic perovskite nanocrystals. Nat. Commun. 2019, 10, 1–8. [Google Scholar] [CrossRef]
- Plesko, S.; Kind, R.; Roos, J. Structural Phase Transitions in CsPbCl3 and RbCdCl3. J. Phys. Soc. Jpn. 1978, 45, 553–557. [Google Scholar] [CrossRef]
- Yu, H.S.; Li, S.L.; Truhlar, D.G. Perspective: Kohn-Sham density functional theory descending a staircase. J. Chem. Phys. 2016, 145, 130901. [Google Scholar] [CrossRef]
- Li, B.; Zhang, Y.; Zhang, L.; Yin, L. PbCl2-tuned inorganic cubic CsPbBr3(Cl) perovskite solar cells with enhanced electron lifetime, diffusion length and photovoltaic performance. J. Power Sources 2017, 360, 11–20. [Google Scholar] [CrossRef]
- Kohn, W.; Sham, L.J. Self-Consistent Equations Including Exchange and Correlation Effects. Phys. Rev. 1965, 140, A1133. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 66, 3865–3868. [Google Scholar] [CrossRef]
- Ziesche, P.; Kurth, S.; Perdew, J.P. Density functionals from LDA to GGA. Comput. Mater. Sci. 1998, 11, 122–127. [Google Scholar] [CrossRef]
- Raju, N.P.; Thangavel, R. Theoretical investigation of spin–orbit coupling on structural, electronic and optical properties for CuAB2 (A = Sb, Bi; B = S, Se) compounds using Tran–Blaha-modified Becke–Johnson method: A first-principles approach. J. Alloys Compd. 2020, 2, 154621. [Google Scholar] [CrossRef]
- Tran, F.; Blaha, P. Implementation of screened hybrid functionals based on the Yukawa potential within the LAPW basis set. Phys. Rev. B 2011, 83, 235118. [Google Scholar] [CrossRef]
- Staroverov, V.N.; Scuseria, G.E.; Tao, J.; Perdew, J.P. Tests of a ladder of density functionals for bulk solids and surfaces. Phys. Rev. B 2004, 69, 075102. [Google Scholar] [CrossRef]
- Kurth, S.; Perdew, J.P.; Blaha, P. Molecular and solid-state tests of density functional approximations: LSD, GGAs, and Meta-GGAs. Int. J. Quantum Chem. 1999, 75, 889–909. [Google Scholar] [CrossRef]
- Camargo-Martínez, J.A.; Baquero, R. The band gap problem: The accuracy of the wien2k code confronted. Rev. Mex. Fis. 2013, 59, 453–459. [Google Scholar]
- Tran, F.; Blaha, P. Importance of the Kinetic Energy Density for Band Gap Calculations in Solids with Density Functional Theory. J. Phys. Chem. A 2017, 121, 3318–3325. [Google Scholar] [CrossRef]
- Akkerman, Q.A.; Motti, S.G.; Srimath Kandada, A.R.; Mosconi, E.; D’Innocenzo, V.; Bertoni, G.; Marras, S.; Kamino, B.A.; Miranda, L.; De Angelis, F.; et al. Solution Synthesis Approach to Colloidal Cesium Lead Halide Perovskite Nanoplatelets with Monolayer-Level Thickness Control. J. Am. Chem. Soc. 2016, 138, 1010–1016. [Google Scholar] [CrossRef]
- Li, Y.; Duan, J.; Yuan, H.; Zhao, Y.; He, B.; Tang, Q. Lattice Modulation of Alkali Metal Cations Doped Cs1−xRxPbBr3 Halides for Inorganic Perovskite Solar Cells. Sol. RRL 2018, 2, 1800164. [Google Scholar] [CrossRef]
- Qaid, S.M.H.; Al-Asbahi, B.A.; Ghaithan, H.M.; AlSalhi, M.S.; Al dwayyan, A.S. Optical and structural properties of CsPbBr3 perovskite quantum dots/PFO polymer composite thin films. J. Colloid Interface Sci. 2020, 563, 426–434. [Google Scholar] [CrossRef]
- Heidrich, K.; Schäfer, W.; Schreiber, M.; Söchtig, J.; Trendel, G.; Treusch, J.; Grandke, T.; Stolz, H.J. Electronic structure, photoemission spectra, and vacuum-ultraviolet optical spectra of CsPbCl3 and CsPbBr3. Phys. Rev. B 1981, 24, 5642–5649. [Google Scholar] [CrossRef]
- Pandey, N.; Kumar, A.; Chakrabarti, S. Investigation of the structural, electronic, and optical properties of Mn-doped CsPbCl3: Theory and experiment. RSC Adv. 2019, 9, 29556–29565. [Google Scholar] [CrossRef]
- Yakunin, S.; Protesescu, L.; Krieg, F.; Bodnarchuk, M.I.; Nedelcu, G.; Humer, M.; De Luca, G.; Fiebig, M.; Heiss, W.; Kovalenko, M.V. Low-threshold amplified spontaneous emission and lasing from colloidal nanocrystals of caesium lead halide perovskites lead halide perovskites. Nat. Commun. 2015, 6, 8056. [Google Scholar] [CrossRef]
- Afsari, M.; Boochani, A.; Hantezadeh, M.; Elahi, S.M. Topological nature in cubic phase of perovskite CsPbI3: By DFT. Solid State Commun. 2017, 259, 10–15. [Google Scholar] [CrossRef]
- Zhang, Q.; Su, R.; Du, W.; Liu, X.; Zhao, L.; Ha, S.T.; Xiong, Q. Advances in Small Perovskite-Based Lasers. Small Methods 2017, 1, 1700163. [Google Scholar] [CrossRef]
- Even, J.; Pedesseau, L.; Jancu, J.M.; Katan, C. Importance of spin-orbit coupling in hybrid organic/inorganic perovskites for photovoltaic applications. J. Phys. Chem. Lett. 2013, 4, 2999–3005. [Google Scholar] [CrossRef]
- Wang, K.; Yang, Q.; Duan, J.; Zhang, C.; Zhao, F.; Yu, H.; Hu, B. Spin-Polarized Electronic Transport through Ferromagnet/Organic–Inorganic Hybrid Perovskite Spinterfaces at Room Temperature. Adv. Mater. Interfaces 2019, 1900718. [Google Scholar] [CrossRef]
- Blaha, P.; Schwarz, K.; Trickey, S.B. Full-potential, linearized augmented plane wave programs for crystalline systems. Comput. Phys. Commun 1990, 59, 399–415. [Google Scholar] [CrossRef]
- Madsen, G.K.H.; Blaha, P.; Schwarz, K.; Sjöstedt, E.; Nordström, L. Efficient linearization of the augmented plane-wave method. Phys. Rev. B Condens. Matter Mater. Phys. 2001, 64, 195134. [Google Scholar] [CrossRef]
- Hohenberg, P.; Kohn, W. Inhomogeneous Electron Gas. Phys. Rev. 1964, 136, B864. [Google Scholar] [CrossRef]
- Blaha, P.; Schwarz, K.; Madsen, G.K.H.; Kvasnicka, D.; Luitz, J. WIEN2k, An Augmented Plane Wave + Local Orbitals Program for Calculating Crystal Properties; Technische Universität Wien: Vienna, Austria, 2001; ISBN 3-9501031-1-2. [Google Scholar]
- Wu, Z.; Cohen, R.E. More accurate generalized gradient approximation for solids. Phys. Rev. B Condens. Matter Mater. Phys. 2006, 73, 235116. [Google Scholar] [CrossRef]
- Tran, F.; Blaha, P. Accurate band gaps of semiconductors and insulators with a semilocal exchange-correlation potential. Phys. Rev. Lett. 2009, 102, 226401. [Google Scholar] [CrossRef]
- Städele, M.; Majewski, J.A.; Vogl, P.; Görling, A. Exact Kohn-Sham Exchange Potential in Semiconductors. Phys. Rev. Lett. 1997, 79, 2089–2092. [Google Scholar] [CrossRef]
- G.F.S. The volume changes of five gases under high pressures. J. Franklin Inst. 1924, 197, 98. [Google Scholar] [CrossRef]
- Beal, R.E.; Slotcavage, D.J.; Leijtens, T.; Bowring, A.R.; Belisle, R.A.; Nguyen, W.H.; Burkhard, G.F.; Hoke, E.T.; McGehee, M.D. Cesium Lead Halide Perovskites with Improved Stability for Tandem Solar Cells. J. Phys. Chem. Lett. 2016, 7, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Lim, A.R.; Jeong, S.Y. Twin structure by 133Cs NMR in ferroelastic CsPbCl3 crystal. Solid State Commun. 1999, 110, 131–136. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Murtaza, G.; Ahmad, I. First principle study of the structural and optoelectronic properties of cubic perovskites CsPbM3 (M=Cl, Br, I). Phys. B Condens. Matter 2011, 406, 3222–3229. [Google Scholar] [CrossRef]
- Mahmood, Q.; Hassan, M.; Rashid, M.; Haq, B.U.; Laref, A. The systematic study of mechanical, thermoelectric and optical properties of lead based halides by first principle approach. Phys. B Condens. Matter 2019, 571, 87–92. [Google Scholar] [CrossRef]
- Jaroenjittichai, A.P.; Laosiritaworn, Y. Band alignment of cesium-based halide perovskites. Ceram. Int. 2018, 44, S161–S163. [Google Scholar] [CrossRef]
- Chang, Y.H.; Park, C.H. First-Principles Study of the Structural and the Electronic Properties of the Lead-Halide-Based Inorganic-Organic Perovskites (CH3NH3)PbX3 and CsPbX3 (X = Cl, Br, I). J. Korean Phys. Soc. 2004, 44, 889–893. [Google Scholar]
- Ghaithan, H.M.; AlAhmed, Z.A.; Lyras, A.; Qaid, S.M.H.; Aldwayyan, A.S. Computational Investigation of the Folded and Unfolded Band Structure and Structural and Optical Properties of CsPb(I1−xBrx)3 Perovskites. Crystals 2020, 10, 342. [Google Scholar] [CrossRef]
- Ghaithan, H.M.; Alahmed, Z.A.; Qaid, S.M.H.; Aldwayyan, A.S. First principle-based calculations of the optoelectronic features of 2 × 2 × 2 CsPb(I1-xBrx)3 perovskite. Superlattices Microstruct. 2020, 140, 106474. [Google Scholar] [CrossRef]
- Chen, X.; Han, D.; Su, Y.; Zeng, Q.; Liu, L.; Shen, D. Structural and Electronic Properties of Inorganic Mixed Halide Perovskites. Phys. Status Solidi Rapid Res. Lett. 2018, 12, 1800193. [Google Scholar] [CrossRef]
- Lang, L.; Yang, J.H.; Liu, H.R.; Xiang, H.J.; Gong, X.G. First-principles study on the electronic and optical properties of cubic ABX3 halide perovskites. Phys. Lett. A 2014, 378, 290–293. [Google Scholar] [CrossRef]
- Wanwieng, N.; Laosiritaworn, Y. Enthalpy of formation of CsSn(Clx(Br, I)1-x)3 and CsPb(Clx(Br, I)1-x )3. Siam Phys. Congr. 2018, 1144, 012137. [Google Scholar] [CrossRef]
- Ghebouli, M.A.; Ghebouli, B.; Fatmi, M. First-principles calculations on structural, elastic, electronic, optical and thermal properties of CsPbCl3 perovskite. Phys. B Condens. Matter 2011, 406, 1837–1843. [Google Scholar] [CrossRef]
- Ilyas, B.M.; Elias, B.H. A theoretical study of perovskite CsXCl3 (X=Pb, Cd) within first principles calculations. Phys. B Condens. Matter 2017, 510, 60–73. [Google Scholar] [CrossRef]
- Møller, C.K. Crystal structure and photoconductivity of cæsium plumbohalides. Nature 1958, 182, 1436. [Google Scholar] [CrossRef]
- Su, Y.; Chen, X.; Ji, W.; Zeng, Q.; Ren, Z.; Su, Z.; Liu, L. Highly Controllable and Efficient Synthesis of Mixed-Halide CsPbX3 (X = Cl, Br, I) Perovskite QDs toward the Tunability of Entire Visible Light. ACS Appl. Mater. Interfaces 2017, 9, 33020–33028. [Google Scholar] [CrossRef]
- Yi, Z.; Fang, Z. Theoretical studies on the structural, electronic and optical properties of orthorhombic perovskites CH3NH3PbX3(X = I, Br, Cl). J. Phys. Chem. Solids 2017, 110, 145–151. [Google Scholar] [CrossRef]
- Umari, P.; Mosconi, E.; De Angelis, F. Relativistic GW calculations on CH3NH3PbI3 and CH3NH3SnI3 Perovskites for Solar Cell Applications. Sci. Rep. 2014, 4, 4467. [Google Scholar] [CrossRef]
- Ghaithan, H.M.; Alahmed, Z.A.; Qaid, S.M.H.; Hezam, M.; Aldwayyan, A.S. Density Functional Study of Cubic, Tetragonal, and Orthorhombic CsPbBr3 Perovskite. ACS Omega 2020, 5, 7468–7480. [Google Scholar] [CrossRef]
- Sun, P.P.; Li, Q.S.; Yang, L.N.; Li, Z.S. Theoretical insights into a potential lead-free hybrid perovskite: Substituting Pb2+ with Ge2+. Nanoscale 2016, 8, 1503–1512. [Google Scholar] [CrossRef]
- Yin, W.J.; Yan, Y.; Wei, S.H. Anomalous alloy properties in mixed halide perovskites. J. Phys. Chem. Lett. 2014, 5, 3625–3631. [Google Scholar] [CrossRef]
- Bellaiche, L.; Wei, S.H.; Zunger, A. Localization and percolation in semiconductor alloys: GaAsN vs GaAsP. Phys. Rev. B Condens. Matter Mater. Phys. 1996, 54, 17568–17576. [Google Scholar] [CrossRef]
- Wei, S.H.; Zunger, A. Giant and composition-dependent optical bowing coefficient in GaAsN alloys. Phys. Rev. Lett. 1996, 76, 664–667. [Google Scholar] [CrossRef]
- Jishi, R.A.; Ta, O.B.; Sharif, A.A. Modeling of lead halide perovskites for photovoltaic applications. J. Phys. Chem. C 2014, 118, 28344–28349. [Google Scholar] [CrossRef]
- Mao, X.; Sun, L.; Wu, T.; Chu, T.; Deng, W.; Han, K. First-Principles Screening of All-Inorganic Lead-Free ABX3 Perovskites. J. Phys. Chem. C 2018, 122, 7670–7675. [Google Scholar] [CrossRef]
- Paul, T.; Chatterjee, B.K.; Maiti, S.; Sarkar, S.; Besra, N.; Das, B.K.; Panigrahi, K.J.; Thakur, S.; Ghorai, U.K.; Chattopadhyay, K.K. Tunable cathodoluminescence over the entire visible window from all-inorganic perovskite CsPbX3 1D architecture. J. Mater. Chem. C 2018, 6, 3322–3333. [Google Scholar] [CrossRef]
- Diroll, B.T.; Zhou, H.; Schaller, R.D. Low-Temperature Absorption, Photoluminescence, and Lifetime of CsPbX3 (X = Cl, Br, I) Nanocrystals. Adv. Funct. Mater. 2018, 28, 1800945. [Google Scholar] [CrossRef]
- Castelli, I.E.; García-Lastra, J.M.; Thygesen, K.S.; Jacobsen, K.W. Bandgap calculations and trends of organometal halide perovskites. APL Mater. 2014, 2, 081514. [Google Scholar] [CrossRef]
- Feng, J.; Xiao, B. Effective masses and electronic and optical properties of nontoxic MASnX3 (X = Cl, Br, and I) perovskite structures as solar cell absorber: A theoretical study using HSE06. J. Phys. Chem. C 2014, 118, 19655–19660. [Google Scholar] [CrossRef]
- Feng, J.; Xiao, B. Crystal Structures, Optical Properties, and E ff ective Mass Tensors of CH3NH3PbX3 (X = I and Br) Phases Predicted from HSE06. J. Phys. Chem. Lett. 2014, 5, 1278–1282. [Google Scholar]
- Jong, U.G.; Yu, C.J.; Ri, J.S.; Kim, N.H.; Ri, G.C. Influence of halide composition on the structural, electronic, and optical properties of mixed CH3NH3Pb(I1-xBrx)3 perovskites calculated using the virtual crystal approximation method. Phys. Rev. B 2016, 94, 125139. [Google Scholar] [CrossRef]
- Qian, J.; Xu, B.; Tian, W. A comprehensive theoretical study of halide perovskites ABX3. Org. Electron. Phys. Mater. Appl. 2016, 37, 61–73. [Google Scholar] [CrossRef]
- Yuan, Y.; Xu, R.; Xu, H.T.; Hong, F.; Xu, F.; Wang, L.J. Nature of the band gap of halide perovskites ABX3(A = CH3NH3, Cs; B = Sn, Pb; X = Cl, Br, I): First-principles calculations. Chinese Phys. B 2015, 24, 116302. [Google Scholar] [CrossRef]
- Fang, Z.; Yi, Z. First principles study on mixed orthorhombic perovskite CH3NH3Pb(I1-xBrx)3. Chem. Phys. Lett. 2017, 687, 19–22. [Google Scholar] [CrossRef]
- Rahman, N.M.; Adnaan, M.; Adhikary, D.; Islam, M.; Alam, M.K. First-principles calculation of the optoelectronic properties of doped methylammonium lead halide perovskites: A DFT-based study. Comput. Mater. Sci. 2018, 150, 439–447. [Google Scholar] [CrossRef]
- Kang, Y.; Han, S. Intrinsic Carrier Mobility of Cesium Lead Halide Perovskites. Phys. Rev. Appl. 2018, 10, 044013. [Google Scholar] [CrossRef]
- Tomanová, K.; Čuba, V.; Brik, M.G.; Mihóková, E.; Martinez Turtos, R.; Lecoq, P.; Auffray, E.; Nikl, M. On the structure, synthesis, and characterization of ultrafast blue-emitting CsPbBr3 nanoplatelets. APL Mater. 2019, 7, 011104. [Google Scholar] [CrossRef]
- Wu, K.; Liang, G.; Shang, Q.; Ren, Y.; Kong, D.; Lian, T. Ultrafast interfacial electron and hole transfer from CsPbBr3 perovskite quantum dots. J. Am. Chem. Soc. 2015, 137, 12792–12795. [Google Scholar] [CrossRef]
- Du, W.; Zhang, S.; Shi, J.; Chen, J.; Wu, Z.; Mi, Y.; Liu, Z.; Li, Y.; Sui, X.; Wang, R.; et al. Strong Exciton-Photon Coupling and Lasing Behavior in All-Inorganic CsPbBr3 Micro/Nanowire Fabry-Pérot Cavity. ACS Photonics 2018, 5, 2051–2059. [Google Scholar] [CrossRef]
- Jin, H.; Im, J.; Freeman, A.J. Topological insulator phase in halide perovskite structures. Phys. Rev. B Condens. Matter Mater. Phys. 2012, 86, 121102. [Google Scholar] [CrossRef]
- Sutton, R.J.; Filip, M.R.; Haghighirad, A.A.; Sakai, N.; Wenger, B.; Giustino, F.; Snaith, H.J. Cubic or Orthorhombic? Revealing the Crystal Structure of Metastable Black-Phase CsPbI3 by Theory and Experiment. ACS Energy Lett. 2018, 3, 1787–1794. [Google Scholar] [CrossRef]
- Reshak, A.H.; Alahmed, Z.A.; Bila, J. Phase transition in BaThO3 from Pbnm to Ibmm turn the fundamental energy band gap from indirect to direct. J. Alloys Compd. 2019, 771, 607–613. [Google Scholar] [CrossRef]
- Callister, W.D., Jr.; Rethwisch, D.G. Fundamentals of Materials Science and Engineering, 5th ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2018; ISBN 978-1-119-17550-6. [Google Scholar]
- Pitriana, P.; Wungu, T.D.K.; Herman; Hidayat, R. The characteristics of band structures and crystal binding in all-inorganic perovskite APbBr3 studied by the first principle calculations using the Density Functional Theory (DFT) method. Results Phys. 2019, 15, 102592. [Google Scholar] [CrossRef]
- Ambrosch-Draxl, C.; Sofo, J.O. Linear optical properties of solids within the full-potential linearized augmented planewave method. Comput. Phys. Commun. 2006, 175, 1–14. [Google Scholar] [CrossRef]
- Dar, S.A.; Sharma, R.; Srivastava, V.; Sakalle, U.K. Investigation on the electronic structure, optical, elastic, mechanical, thermodynamic and thermoelectric properties of wide band gap semiconductor double perovskite Ba2InTaO6. RSC Adv. 2019, 9, 9522–9532. [Google Scholar] [CrossRef]
- Adachi, S. Properties of Semiconductor Alloys: Group-IV, III-V and II-VI Semiconductors; John Wiley & Sons: Hoboken, NJ, USA, 2009; ISBN 9780470744383. [Google Scholar]
- Benchehima, M.; Abid, H.; Sadoun, A.; Chabane Chaouche, A. Optoelectronic properties of aluminum bismuth antimony ternary alloys for optical telecommunication applications: First principles calculation. Comput. Mater. Sci. 2018, 155, 224–234. [Google Scholar] [CrossRef]
- Amin, B.; Ahmad, I.; Maqbool, M.; Goumri-Said, S.; Ahmad, R. Ab initio study of the bandgap engineering of Al1-xGaxN for optoelectronic applications. J. Appl. Phys. 2011, 109, 023109. [Google Scholar] [CrossRef]
- Kushwaha, A.K.; Laref, A.; Laref, S. First-Principles Investigation of Structural, Electronic, Optical, and Magnetic Properties of Ternary Mixed Compound CsTexS1−x. J. Electron. Mater. 2019, 48, 3479–3489. [Google Scholar] [CrossRef]
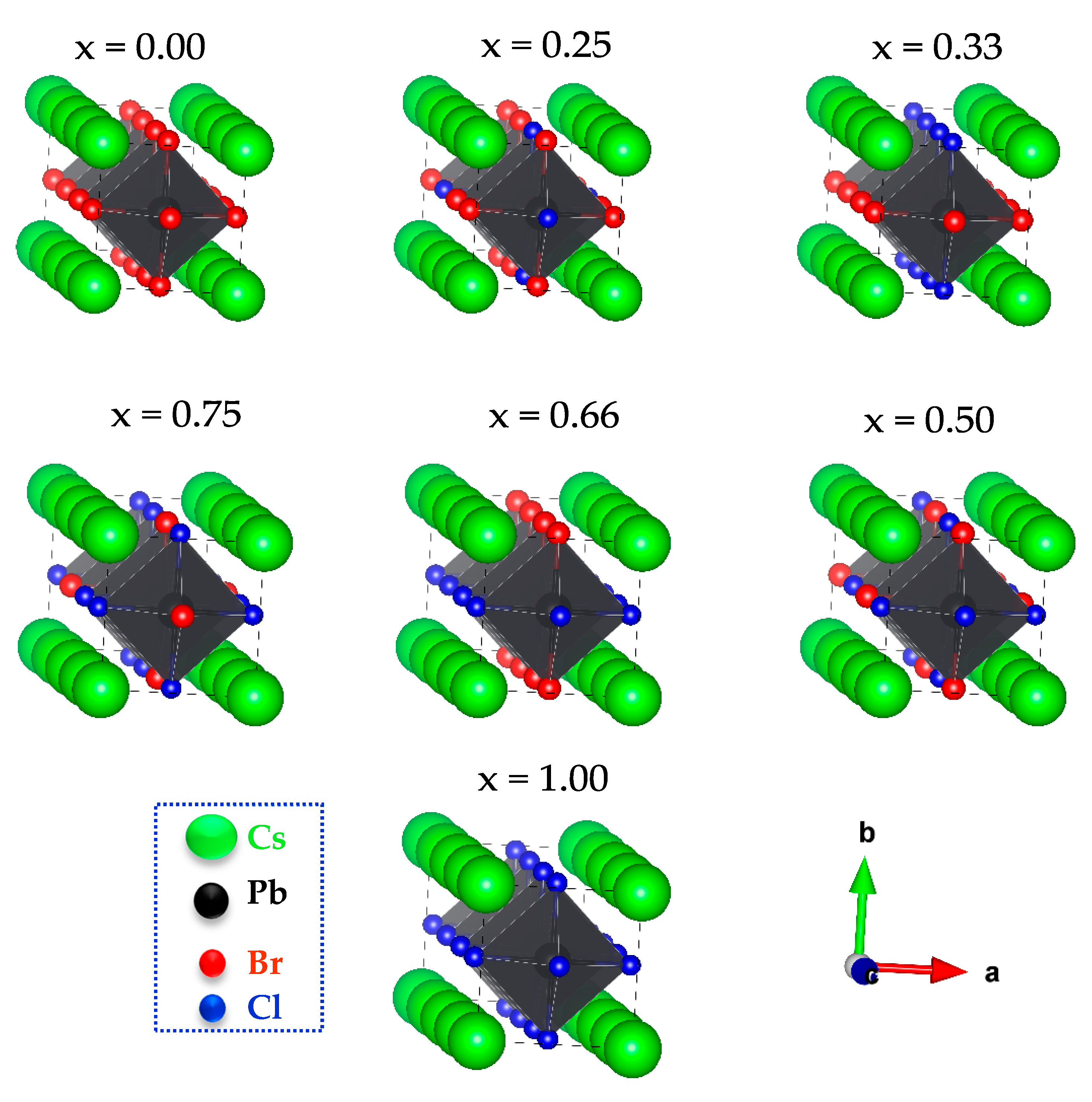
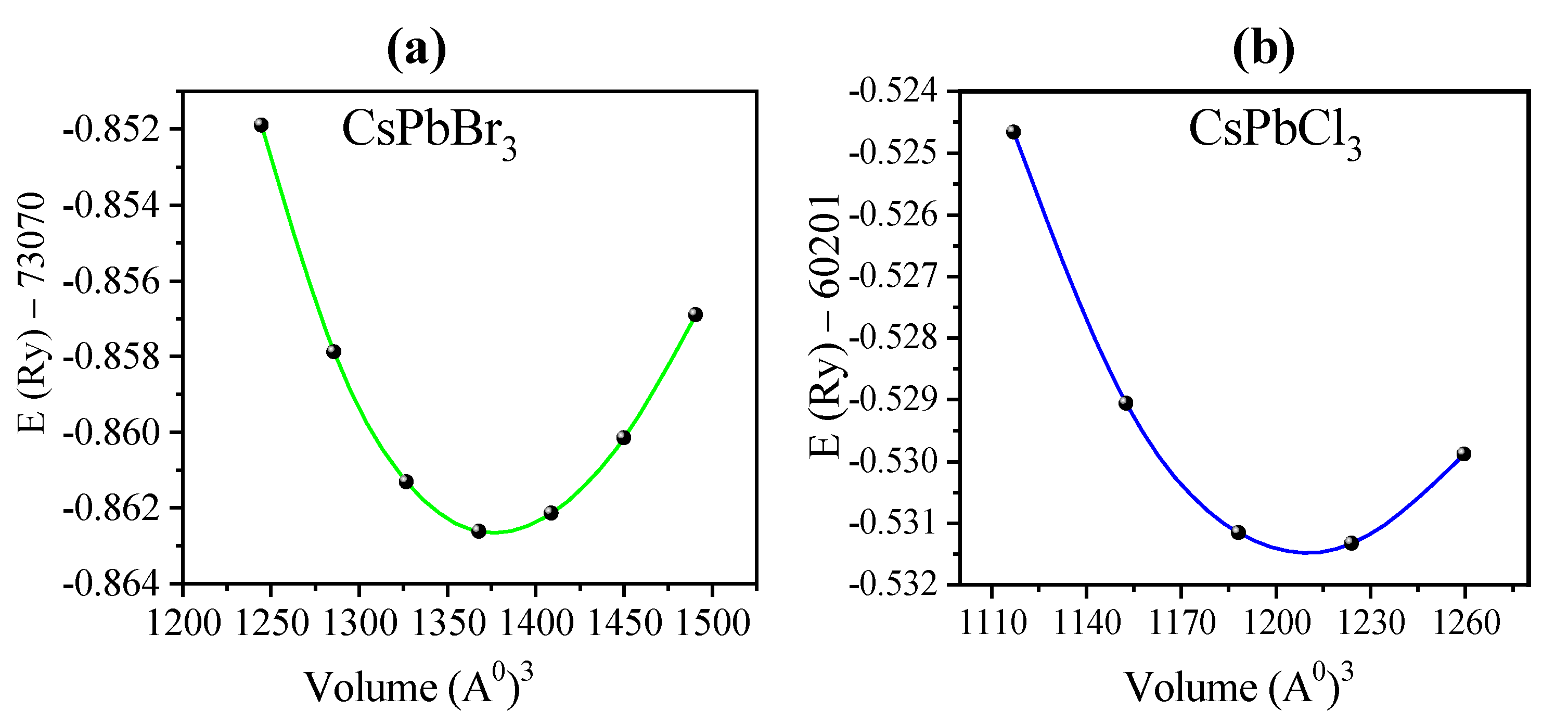
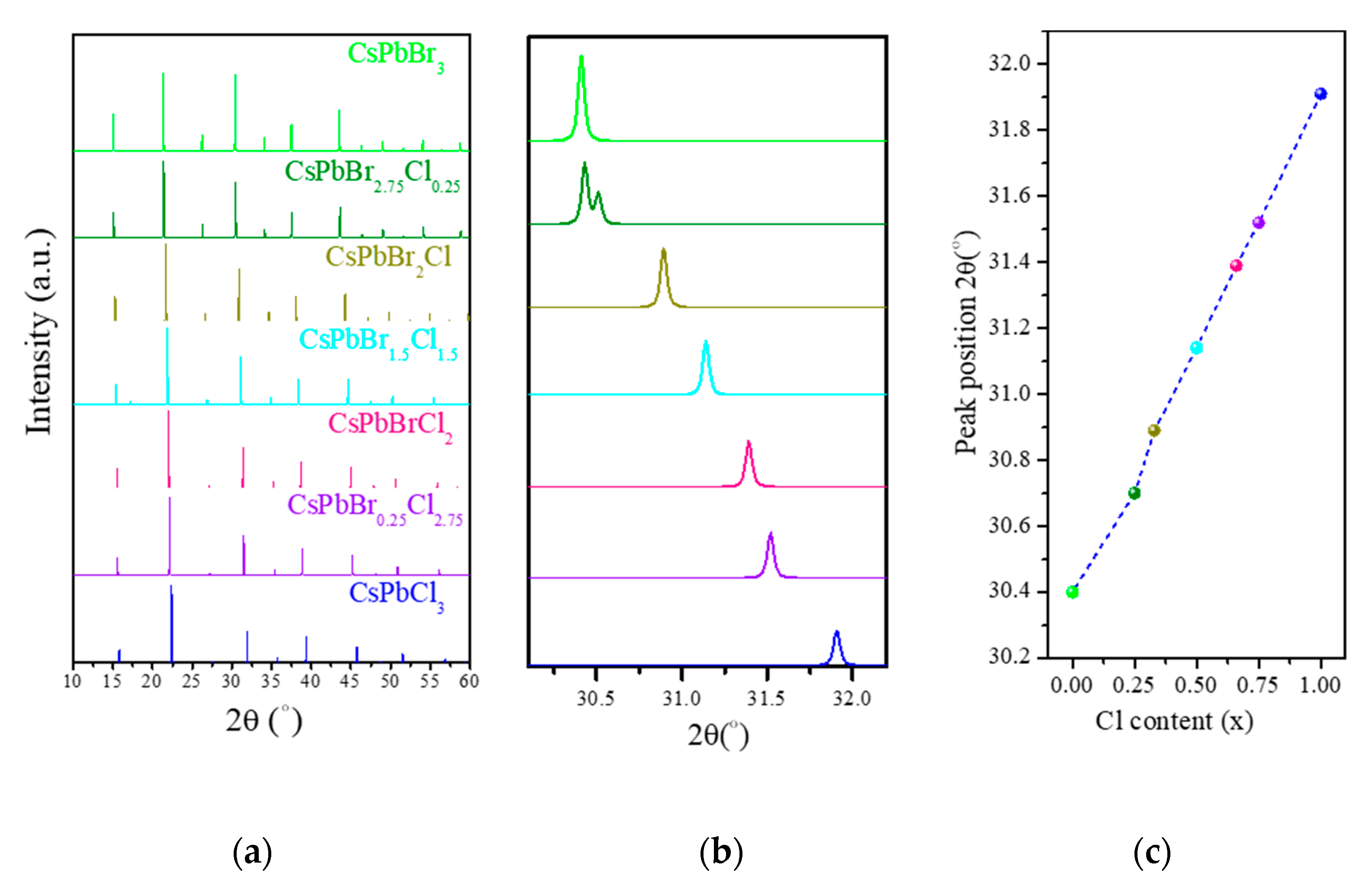
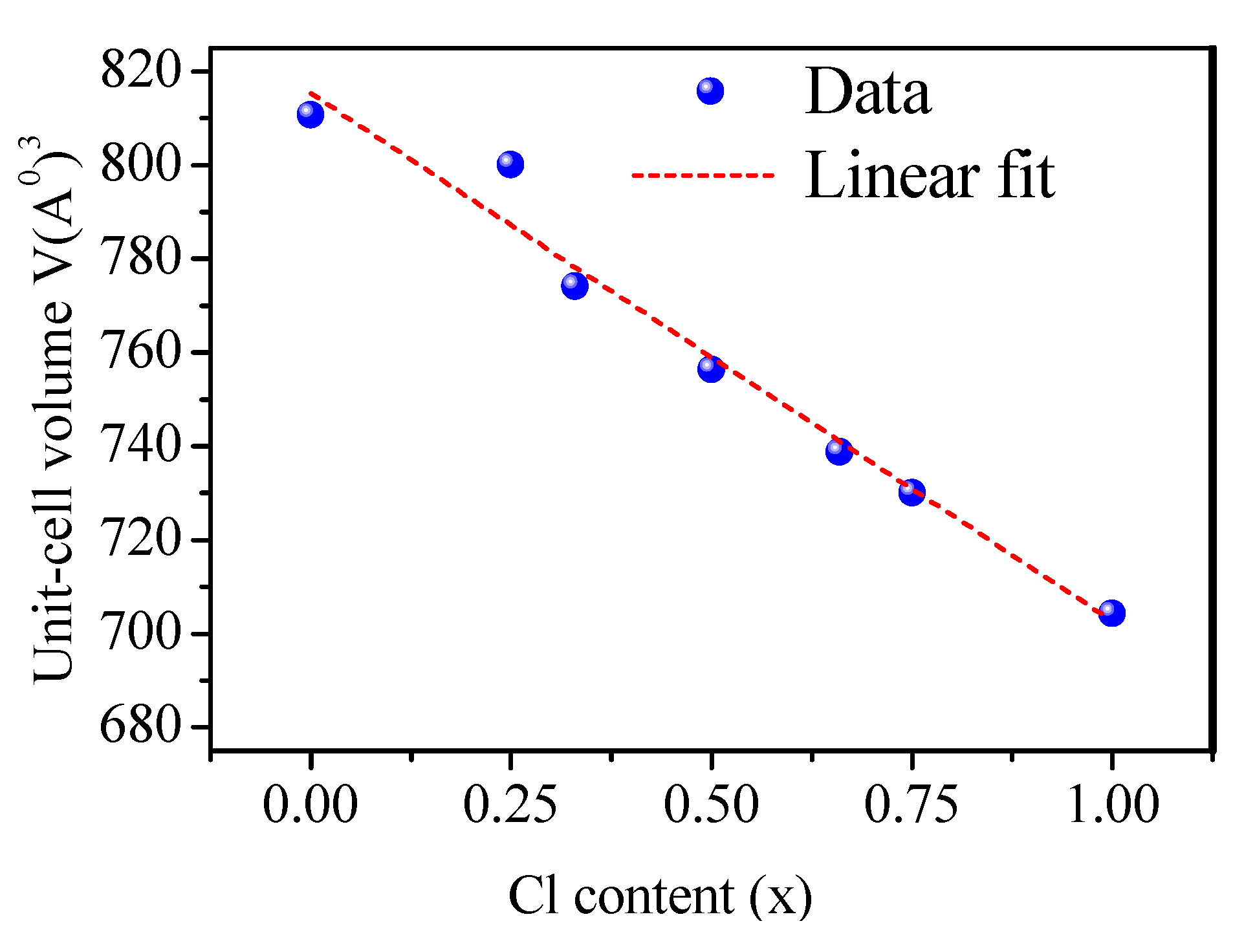
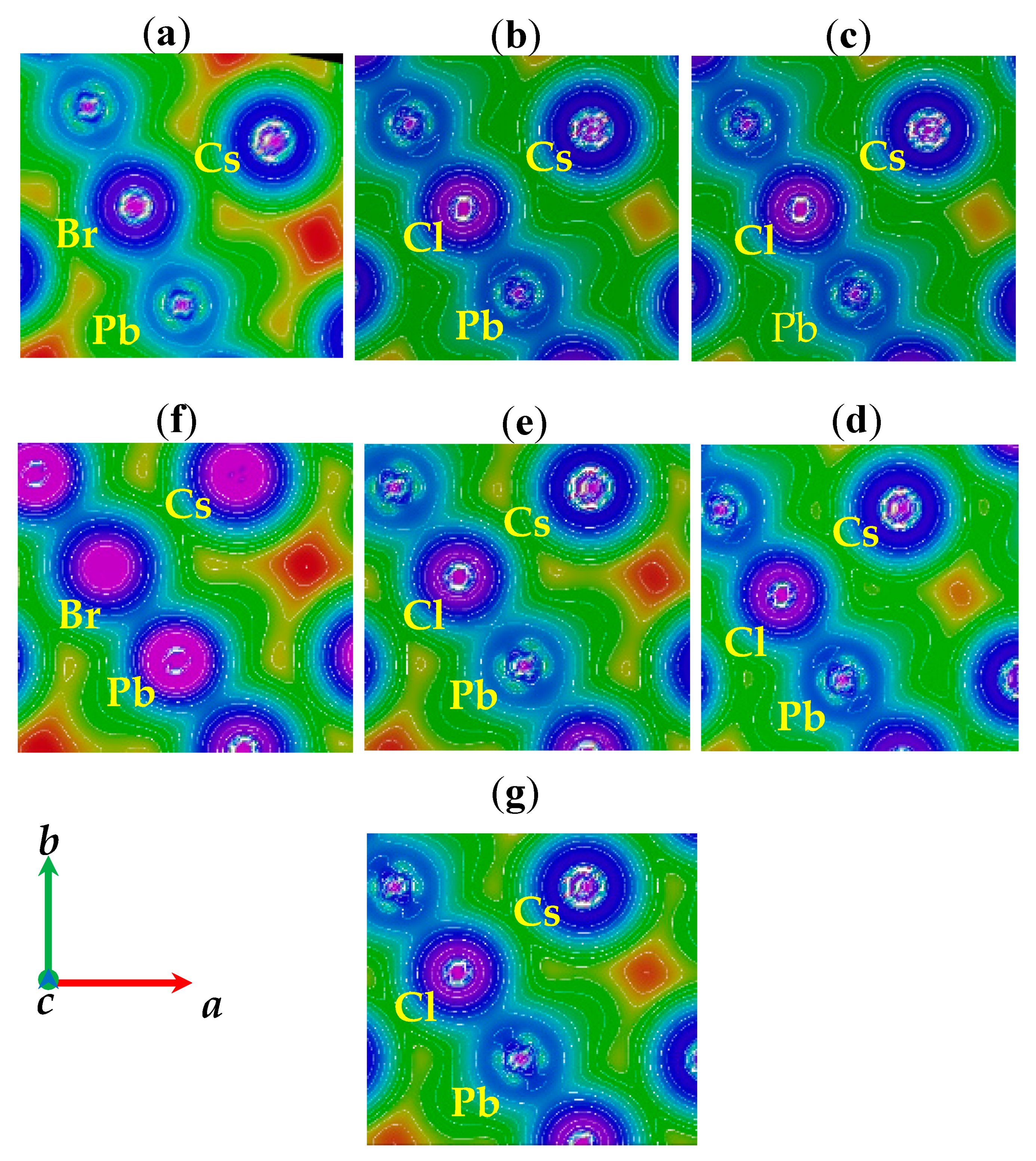
| CsPb(Br1−xClx)3 | Present Work | Other Calculations (Exp.) | |||||
|---|---|---|---|---|---|---|---|
| a (Å) | V (Å)3 | B (GPa) | B′ | a (Å) | B (GPa) | B′ | |
| CsPbBr3 | a = 5.874 | 810.703 | 20.7379 | 4.881 | 5.84 (WC–GGA) [72] 5.86 (TB–mBJ) * [73] 5.74 (LDA) [74,75] 6.005 (PBE–GGA) [23] 5.87 (PBEsol) [23] 5.87 (PBE–GGA) [76] 5.77 (LDA) [23] 6.0039 (PBE–GGA) [77] (5.874) [36] (5.85) [69] | 23.5 [72] | 5.0 [72] |
| CsPbBr2.75Cl0.25 | a = 5.801 c = 5.855 | 807.008 | a = 6.005 c = 5.859 (PBE–GGA) [78] | ||||
| CsPbBr2Cl | a = 5.784 c = 5.748 | 774.139 | a = 5.708 c = 6.012 (PBE–GGA) [78] | ||||
| CsPbBr1.5Cl1.5 | a = 5.739 c = 5.7395 | 756.278 | a = 5.718 c = 5.874 (PBE–GGA) [78] | - | - | ||
| CsPbBrCl2 | a = 5.695 c = 5.6947 | 738.692 | a = 5.725 c = 6.012 (PBE–GGA) [78] | ||||
| CsPbBr0.25Cl2.75 | a = 5.672 c = 5.6722 | 730.005 | a = 5.728 c = 5.879 (PBE–GGA) [78] | ||||
| CsPbCl3 | a = 5.605 | 704.347 | 24.2106 | 5.0142 | 5.56 (WC–GGA) [72] 5.61 (TB–mBJ) [73] 5.73 (PBE–GGA) [79] 5.49 (LDA) [75] 5.743 (PBE–GGA) [80] 5.726 (PBE–GGA) [78] 5.728 (PBE–GGA) [81] 5.618 (PBE–GGA)[82] 5.740 (LDA) [74] 5.603 (PBE–GGA) [55] 5.605 [70,83] 5.61 [55] 5.6228 [30] | 25.8 [72] 22.59 [81] 25.447[82] 26.33 [73] | 5.0 [72] 4.33 [81] 4.4 [82] |
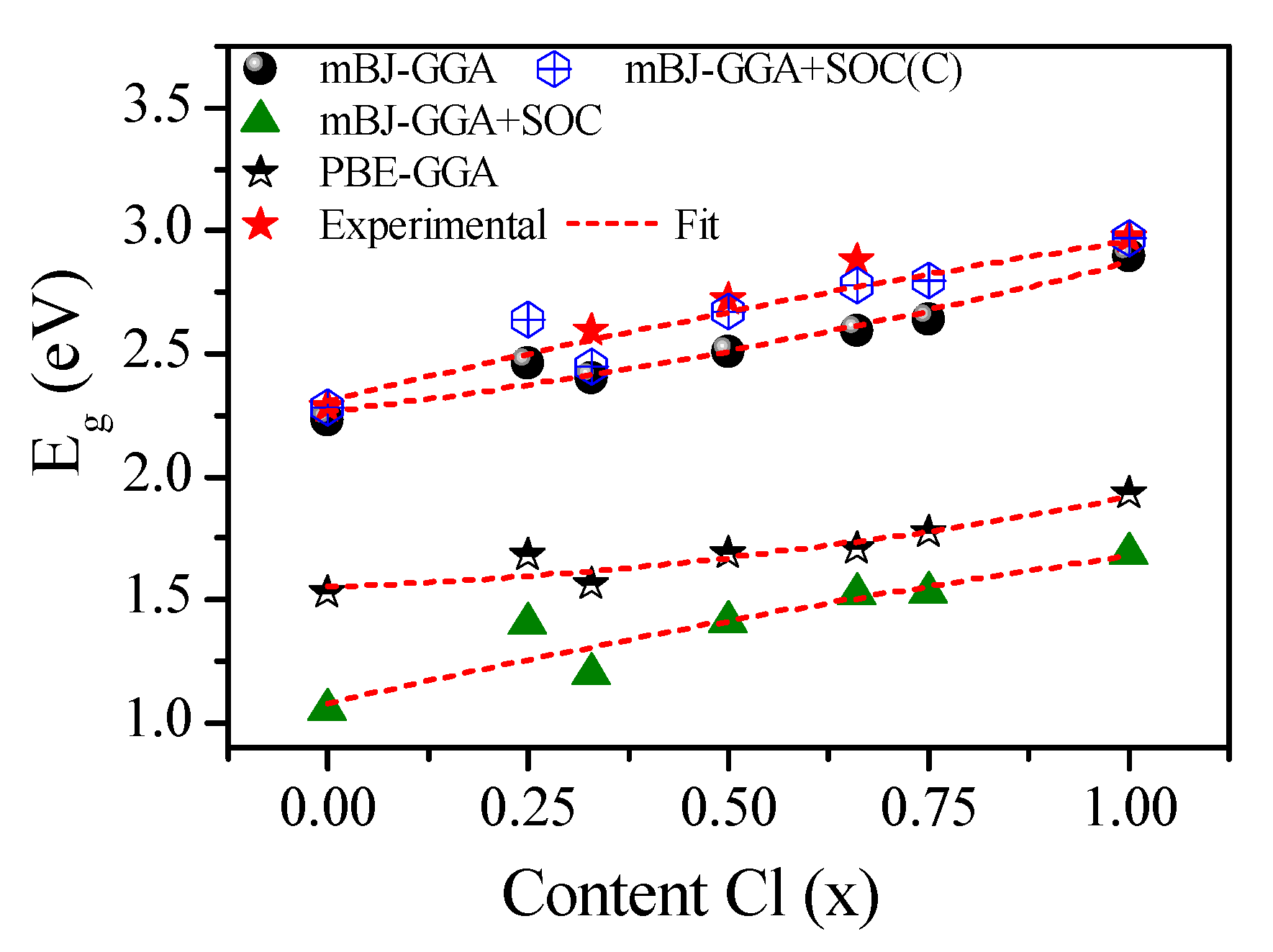
| CsPb(Br1−xClx)3 | Eg (eV) | ||||
|---|---|---|---|---|---|
| This Work | Other (Exp.) | ||||
| PBE–GGA | mBJ–GGA | mBJ–GGA + SOC | mBJ–GGA + SOC (C) | ||
| CsPbBr3 | 1.53 | 2.23 | 1.05 | 2.28 | 2.34 (GW) [74] 1.61 (PBE–GGA) [23] 2.36 (nTmBj) [23] 2.228 (KTB–mBJ) * [92] 2.08 (GLLB-SC) ** [93] 2.10 (QE) *** [35] 2.50 (mBJ–GGA) [77] (2.36) [51,94] (2.32) [52] (2.282) [53] (2.35) [95] |
| CsPbBr2.75Cl0.25 | 1.68 | 2.46 | 1.40 | 2.64 | 1.809 (PBE–GGA) [78] |
| CsPbBr2Cl | 1.56 | 2.40 | 1.20 | 2.45 | 1.827 (PBE–GGA) [78] (2.59) [94] |
| CsPbBr1.5Cl1.5 | 1.69 | 2.51 | 1.41 | 2.67 | 1.859 (PBE–GGA) [78] (2.72) [94] |
| CsPbBrCl2 | 1.71 | 2.59 | 1.52 | 2.78 | 1.881(PBE–GGA) [78] (2.88) [94] |
| CsPbBr0.25Cl2.75 | 1.77 | 2.64 | 1.53 | 2.80 | 2.05(PBE–GGA) [78] |
| CsPbCl3 | 1.93 | 2.90 | 1.69 | 2.97 | 2.20 (PBE–GGA) [78,96] 2.829 (KTB–mBJ) [92] 2.92 (HSE) **** [79] 3.406 (PBE–GGA)[82] 2.88 (GW) [74] 2.74 (TB–mBJ) [73] 2.168 (PBE–GGA) [23] 3.10 (nTmBj) [23] (3.00) [54] (2.97) [55] (3.04) [95](2.98) [94] |
| CsPb(Br1−xClx)3 | mBJ–GGA (others) | ||
|---|---|---|---|
| ε1 (0) | n (0) | R (0)% | |
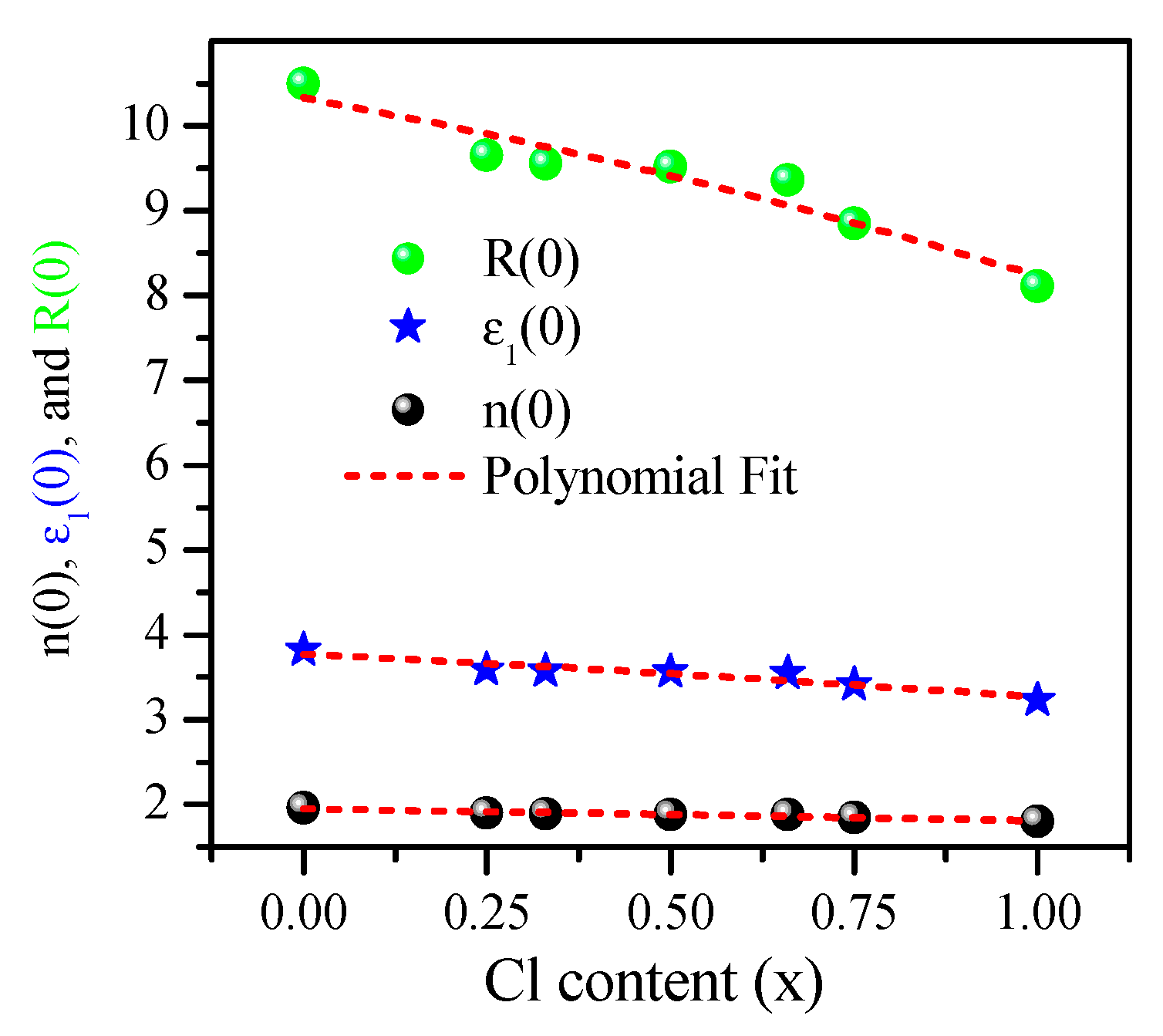
| CsPbBr3 | 3.82 (4.30) [104] (4.60) [23] (4.63) [72] | 1.96 Exp. (1.85–2.3) [56] (2.152) [72] | 10.50 (13.4) [72] |
| CsPbBr2.75Cl0.25 | 3.59 | 1.897 | 9.65 |
| CsPbBr2Cl | 3.57 | 1.890 | 9.55 |
| CsPbBr1.5Cl1.5 | 3.56 | 1.882 | 9.52 |
| CsPbBrCl2 | 3.55 | 1.880 | 9.36 |
| CsPbBr0.25Cl2.75 | 3.41 | 1.848 | 8.85 |
| CsPbCl3 | 3.23 (3.69) [104] (3.00) [81] (4.10) [23] (4.43) [72] | 1.798 (1.739) [81] (2.105) [72] | 8.11 (12.7) [72] (10) [82] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghaithan, H.M.; Alahmed, Z.A.; Qaid, S.M.H.; Aldwayyan, A.S. Structural, Electronic, and Optical Properties of CsPb(Br1−xClx)3 Perovskite: First-Principles Study with PBE–GGA and mBJ–GGA Methods. Materials 2020, 13, 4944. https://doi.org/10.3390/ma13214944
Ghaithan HM, Alahmed ZA, Qaid SMH, Aldwayyan AS. Structural, Electronic, and Optical Properties of CsPb(Br1−xClx)3 Perovskite: First-Principles Study with PBE–GGA and mBJ–GGA Methods. Materials. 2020; 13(21):4944. https://doi.org/10.3390/ma13214944
Chicago/Turabian StyleGhaithan, Hamid M., Zeyad. A. Alahmed, Saif M. H. Qaid, and Abdullah S. Aldwayyan. 2020. "Structural, Electronic, and Optical Properties of CsPb(Br1−xClx)3 Perovskite: First-Principles Study with PBE–GGA and mBJ–GGA Methods" Materials 13, no. 21: 4944. https://doi.org/10.3390/ma13214944
APA StyleGhaithan, H. M., Alahmed, Z. A., Qaid, S. M. H., & Aldwayyan, A. S. (2020). Structural, Electronic, and Optical Properties of CsPb(Br1−xClx)3 Perovskite: First-Principles Study with PBE–GGA and mBJ–GGA Methods. Materials, 13(21), 4944. https://doi.org/10.3390/ma13214944






