A Novel Bioactive Endodontic Sealer Containing Surface-Reaction-Type Prereacted Glass-Ionomer Filler Induces Osteoblast Differentiation
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Sealer Extracts
2.2. Cell Culture
2.3. Cell-Viability Assay
2.4. Nodule Formation
2.5. Reverse Transcription-Quantitative Polymerase Chain Reaction
2.6. Immunofluorescence
2.7. Western Blotting
2.8. Statistical Analysis
3. Results
3.1. Element/Ion Concentration of Sealer Extracts
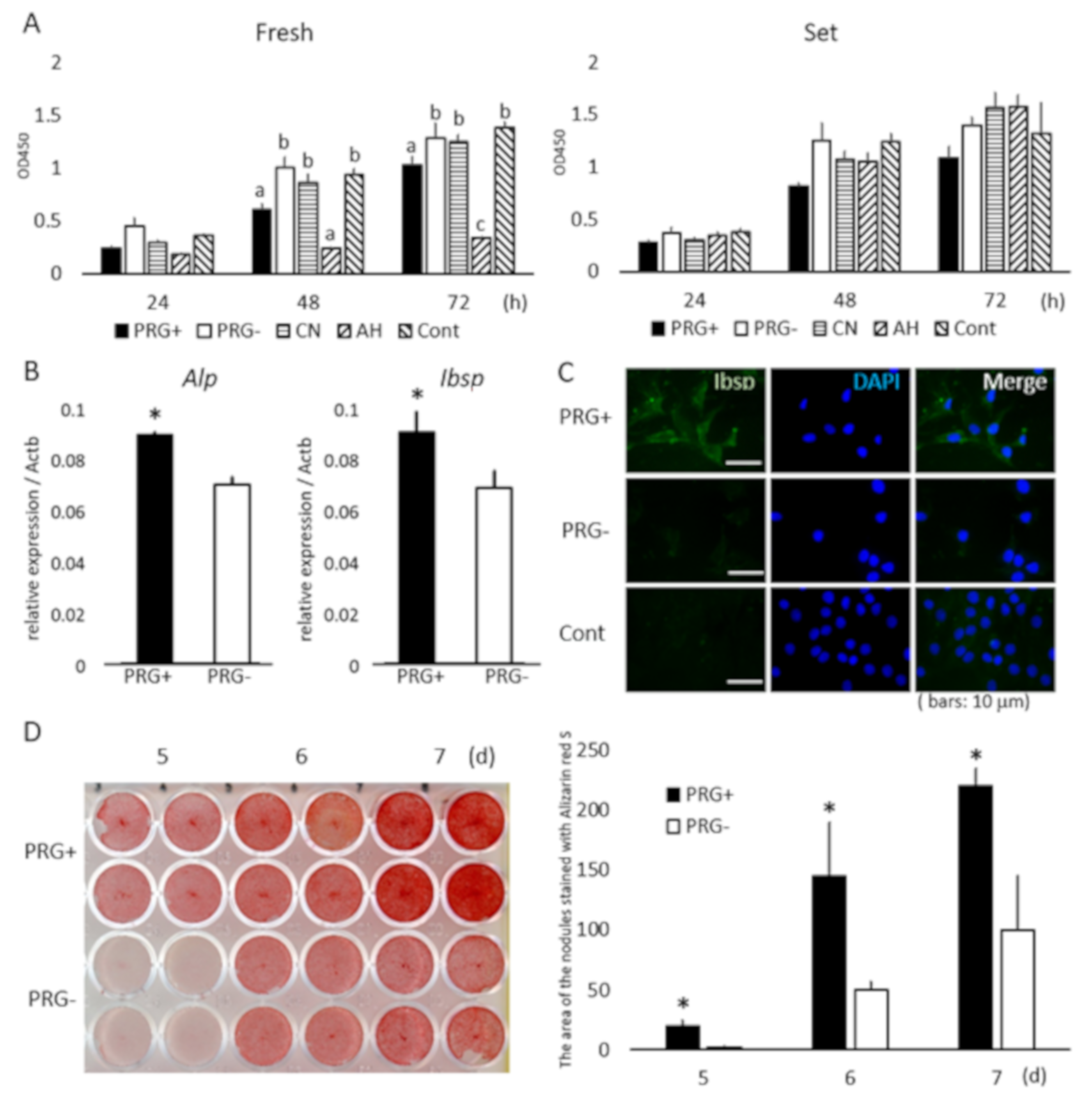
3.2. Cell Growth
3.3. Osteoblastic-Marker Expression and Mineralized-Module Formation
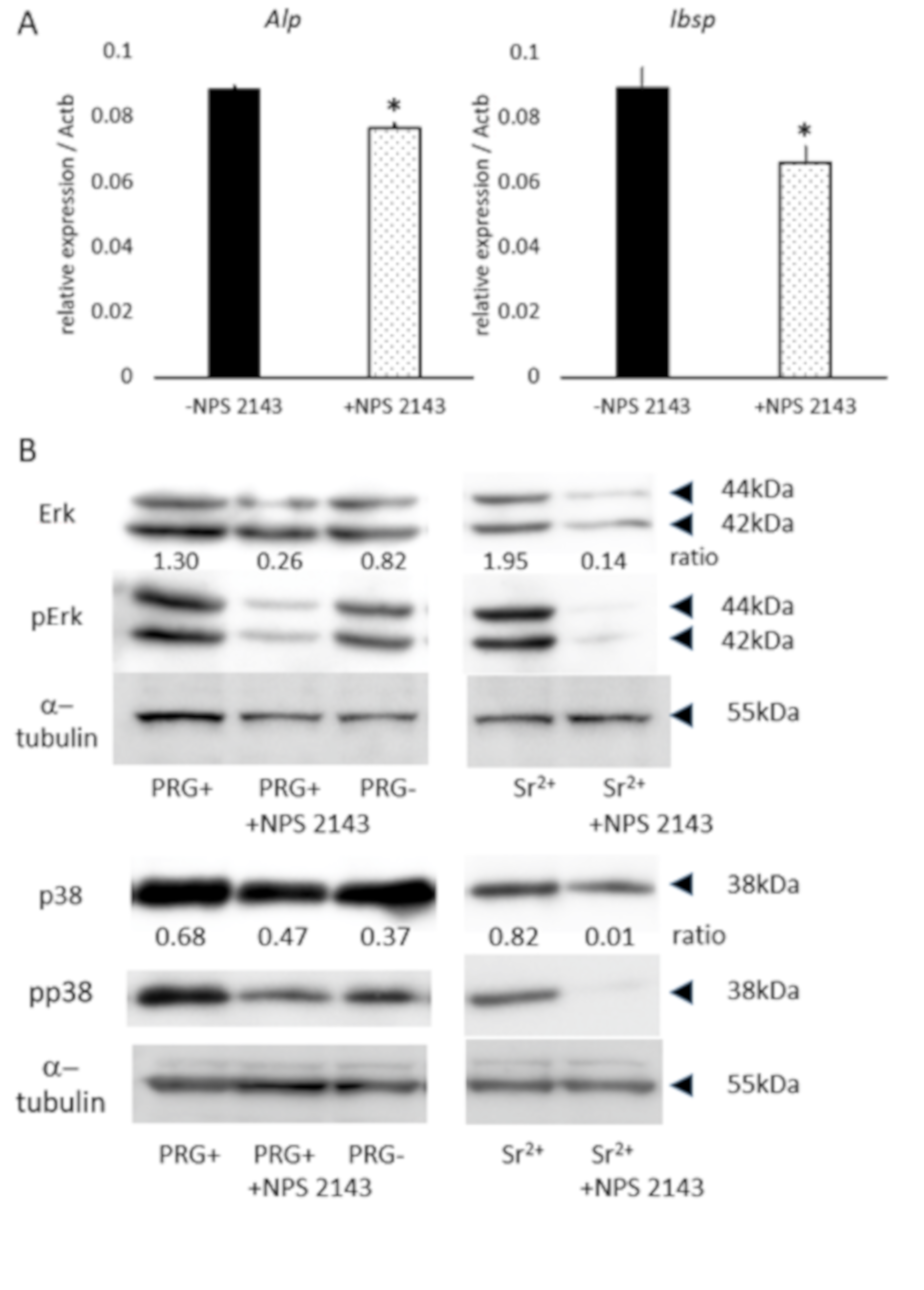
3.4. Effect of CaSR Antagonist on Osteoblastic-Marker Expression, and ERK and p38 Phosphorylation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Johnson, W.; Kulild, J.C.; Tay, F. Obturation of the Cleaned and Shaped Root Canal System. In Cohen’s Pathway of the Pulp, 11th ed.; Hargreaves, K.M., Berman, L.H., Eds.; Elsevier: St. Louis, MO, USA, 2016; pp. 280–322. [Google Scholar]
- Ørstavik, D. Endodontic filling materials. Endod. Top. 2014, 31, 53–67. [Google Scholar] [CrossRef]
- Donnermeyer, D.; Bürklein, S.; Dammaschke, T.; Schäfer, E. Endodontic sealers based on calcium silicates: A systematic review. Odontology 2019, 107, 421–436. [Google Scholar] [CrossRef]
- Koutroulis, A.; Kuehne, S.A.; Cooper, P.R.; Camilleri, J. The role of calcium ion release on biocompatibility and antimicrobial properties of hydraulic cements. Sci. Rep. 2019, 9, 19019. [Google Scholar] [CrossRef]
- Fujimoto, Y.; Iwasa, M.; Murayama, R.; Miyazaki, M.; Nagafuji, A.; Nakatsuka, T. Detection of ions released from S-PRG fillers and their modulation effect. Dent. Mater. J. 2010, 29, 392–397. [Google Scholar] [CrossRef]
- Kawasaki, K.; Kambara, M. Effects of ion-releasing tooth-coating material on demineralization of bovine tooth enamel. Int. J. Dent. 2014, 2014, 463149. [Google Scholar] [CrossRef]
- Okamoto, M.; Ali, M.; Komichi, S.; Watanabe, M.; Huang, H.; Ito, Y.; Miura, J.; Hirose, Y.; Mizuhira, M.; Takahashi, Y.; et al. Surface pre-reacted glass filler contributes to tertiary dentin formation through a mechanism different than that of hydraulic calcium-silicate cement. J. Clin. Med. 2019, 8, 1440. [Google Scholar] [CrossRef]
- Han, L.; Okiji, T. Evaluation of the ions release/incorporation of the prototype S-PRG filler-containing endodontic sealer. Dent. Mater. J. 2011, 30, 898–903. [Google Scholar] [CrossRef]
- Han, L.; Okamoto, A.; Fukushima, M.; Okiji, T. Evaluation of a new fluoride-releasing one-step adhesive. Dent. Mater. J. 2006, 25, 509–515. [Google Scholar] [CrossRef][Green Version]
- Ito, S.; Iijima, M.; Hashimoto, M.; Tsukamoto, N.; Mizoguchi, I.; Saito, T. Effects of surface pre-reacted glass-ionomer fillers on mineral induction by phosphoprotein. J. Dent. 2011, 39, 72–79. [Google Scholar] [CrossRef]
- Miki, S.; Kitagawa, H.; Kitagawa, R.; Kiba, W.; Hayashi, M.; Imazato, S. Antibacterial activity of resin composites containing surface pre-reacted glass-ionomer (S-PRG) filler. Dent. Mater. 2016, 32, 1095–1102. [Google Scholar] [CrossRef]
- Takahashi, Y.; Okamoto, M.; Komichi, S.; Imazato, S.; Nakatsuka, T.; Sakamoto, S.; Kimoto, K.; Hayashi, M. Application of a direct pulp capping cement containing S-PRG filler. Clin. Oral. Investig. 2019, 23, 1723–1731. [Google Scholar] [CrossRef]
- Nemoto, A.; Chosa, N.; Kyakumoto, S.; Yokota, S.; Kamo, M.; Noda, M.; Ishisaki, A. Water-soluble factors eluated from surface pre-reacted glass-ionomer filler promote osteoblastic differentiation of human mesenchymal stem cells. Mol. Med. Rep. 2018, 17, 3448–3454. [Google Scholar] [CrossRef]
- Bakhit, A.; Kawashima, N.; Hashimoto, K.; Noda, S.; Nara, K.; Kuramoto, M.; Tazawa, K.; Okiji, T. Strontium ranelate promotes odonto-/osteogenic differentiation/mineralization of dental papillae cells in vitro and mineralized tissue formation of the dental pulp in vivo. Sci. Rep. 2018, 8, 9224. [Google Scholar] [CrossRef]
- Thomsen, A.R.; Worm, J.; Jacobsen, S.E.; Stahlhut, M.; Latta, M.; Bräuner-Osborne, H. Strontium is a biased agonist of the calcium-sensing receptor in rat medullary thyroid carcinoma 6-23 cells. J. Pharmacol. Exp. Ther. 2012, 343, 638–649. [Google Scholar] [CrossRef]
- Umezawa, A.; Maruyama, T.; Segawa, K.; Shadduck, R.K.; Waheed, A.; Hata, J. Multipotent marrow stromal cell line is able to induce hematopoiesis in vivo. J. Cell. Physiol. 1992, 151, 197–205. [Google Scholar] [CrossRef]
- Shindo, K.; Kawashima, N.; Sakamoto, K.; Yamaguchi, A.; Umezawa, A.; Takagi, M.; Katsube, K.; Suda, H. Osteogenic differentiation of the mesenchymal progenitor cells, Kusa is suppressed by Notch signaling. Exp. Cell Res. 2003, 290, 370–380. [Google Scholar] [CrossRef]
- Kishi, Y.; Fujihara, H.; Kawaguchi, K.; Yamada, H.; Nakayama, R.; Yamamoto, N.; Fujihara, Y.; Hamada, Y.; Satomura, K.; Masutani, M. PARP inhibitor PJ34 suppresses osteogenic differentiation in mouse mesenchymal stem cells by modulating BMP-2 signaling pathway. Int. J. Mol. Sci. 2015, 16, 24820–24838. [Google Scholar] [CrossRef]
- Jung, S.; Sielker, S.; Hanisch, M.R.; Libricht, V.; Schafer, E.; Dammaschke, T. Cytotoxic effects of four different root canal sealers on human osteoblasts. PLoS ONE 2018, 13, e0194467. [Google Scholar] [CrossRef]
- Chang, M.C.; Lin, L.D.; Chen, Y.J.; Tsai, Y.L.; Cheng, Y.A.; Kuo, C.S.; Chang, H.H.; Tai, T.F.; Lin, H.J.; Jeng, J.H. Comparative cytotoxicity of five root canal sealers on cultured human periodontal ligament fibroblasts. Int. Endod. J. 2010, 43, 251–257. [Google Scholar] [CrossRef]
- Schmid-Schwap, M.; Franz, A.; Konig, F.; Bristela, M.; Lucas, T.; Piehslinger, E.; Watts, D.C.; Schedle, A. Cytotoxicity of four categories of dental cements. Dent. Mater. 2009, 25, 360–368. [Google Scholar] [CrossRef]
- Brennan, S.C.; Thiem, U.; Roth, S.; Aggarwal, A.; Fetahu, I.; Tennakoon, S.; Gomes, A.R.; Brandi, M.L.; Bruggeman, F.; Mentaverri, R.; et al. Calcium sensing receptor signalling in physiology and cancer. Biochim. Biophys. Acta 2013, 1833, 1732–1744. [Google Scholar] [CrossRef]
- Liu, Y.J.; Su, W.T.; Chen, P.H. Magnesium and zinc borate enhance osteoblastic differentiation of stem cells from human exfoliated deciduous teeth in vitro. J. Biomater. Appl. 2018, 32, 765–774. [Google Scholar] [CrossRef]
- Aubin, J.E. Regulation of osteoblast formation and function. Rev. Endocr. Metab. Disord. 2001, 2, 81–94. [Google Scholar] [CrossRef]
- Golub, E.E.; Boesze-Battaglia, K. The role of alkaline phosphatase in mineralization. Curr. Opin. Orthop. 2007, 18, 444–448. [Google Scholar] [CrossRef]
- Gorski, J.P.; Wang, A.; Lovitch, D.; Law, D.; Powell, K.; Midura, R.J. Extracellular bone acidic glycoprotein-75 defines condensed mesenchyme regions to be mineralized and localizes with bone sialoprotein during intramembranous bone formation. J. Biol. Chem. 2004, 279, 25455–25463. [Google Scholar] [CrossRef]
- Bonnelye, E.; Chabadel, A.; Saltel, F.; Jurdic, P. Dual effect of strontium ranelate: Stimulation of osteoblast differentiation and inhibition of osteoclast formation and resorption in vitro. Bone 2008, 42, 129–138. [Google Scholar] [CrossRef]
- Yang, F.; Yang, D.; Tu, J.; Zheng, Q.; Cai, L.; Wang, L. Strontium enhances osteogenic differentiation of mesenchymal stem cells and in vivo bone formation by activating Wnt/catenin signaling. Stem Cells 2011, 29, 981–991. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.M.; MacLeod, R.J. Extracellular calcium sensing and extracellular calcium signaling. Physiol. Rev. 2001, 81, 239–297. [Google Scholar] [CrossRef]
- Dvorak-Ewell, M.M.; Chen, T.H.; Liang, N.; Garvey, C.; Liu, B.; Tu, C.; Chang, W.; Bikle, D.D.; Shoback, D.M. Osteoblast extracellular Ca2+ -sensing receptor regulates bone development, mineralization, and turnover. J. Bone Miner. Res. 2011, 26, 2935–2947. [Google Scholar] [CrossRef]
- Diepenhorst, N.A.; Leach, K.; Keller, A.N.; Rueda, P.; Cook, A.E.; Pierce, T.L.; Nowell, C.; Pastoureau, P.; Sabatini, M.; Summers, R.J.; et al. Divergent effects of strontium and calcium-sensing receptor positive allosteric modulators (calcimimetics) on human osteoclast activity. Br. J. Pharmacol. 2018, 175, 4095–4108. [Google Scholar] [CrossRef]
- Takaoka, S.; Yamaguchi, T.; Yano, S.; Yamauchi, M.; Sugimoto, T. The Calcium-sensing Receptor (CaR) is involved in strontium ranelate-induced osteoblast differentiation and mineralization. Horm. Metab. Res. 2010, 42, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Kifor, O.; MacLeod, R.J.; Diaz, R.; Bai, M.; Yamaguchi, T.; Yao, T.; Kifor, I.; Brown, E.M. Regulation of MAP kinase by calcium-sensing receptor in bovine parathyroid and CaR-transfected HEK293 cells. Am. J. Physiol. Renal Physiol. 2001, 280, F291–F302. [Google Scholar] [CrossRef]
- McNeil, S.E.; Hobson, S.A.; Nipper, V.; Rodland, K.D. Functional calcium-sensing receptors in rat fibroblasts are required for activation of SRC kinase and mitogen-activated protein kinase in response to extracellular calcium. J. Biol. Chem. 1998, 273, 1114–1120. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, T.; Chan, Y.Y.; Kawanami, A.; Balmes, G.; Landreth, G.E.; Murakami, S. Extracellular signal-regulated kinase 1 (ERK1) and ERK2 play essential roles in osteoblast differentiation and in supporting osteoclastogenesis. Mol. Cell. Biol. 2009, 29, 5843–5857. [Google Scholar] [CrossRef] [PubMed]
- Maiti, A.; Hait, N.C.; Beckman, M.J. Extracellular calcium-sensing receptor activation induces vitamin D receptor levels in proximal kidney HK-2G cells by a mechanism that requires phosphorylation of p38alpha MAPK. J. Biol. Chem. 2008, 283, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Chan, E.; Wang, S.X.; Li, B. Activation of p38 mitogen-activated protein kinase is required for osteoblast differentiation. Endocrinology 2003, 144, 2068–2074. [Google Scholar] [CrossRef]
| Materials | Main Composition | Elements/Ion Concentration (ppm) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| B | Al | Si | Na | Zn | Sr | F- | |||
| Prototype endodontic sealer containing S-PRG filler (Shofu; PRG+) | Powder: zinc oxide-based inorganic compound filler, S-PRG filler *, additive Liquid: poly carboxylic acid sodium salt, water, other | Fresh | 236 | 28 | 42 | 240 | 61 | 27 | 236 |
| Set | 198 | 0 | 2 | 240 | 46 | 47 | 76 | ||
| Experimental endodontic sealer containing S-PRG-free silica filler (Shofu; PRG-) | Powder: zinc oxide-based inorganic compound filler, silica-filler, additive Liquid: poly carboxylic acid sodium salt, water, other | Fresh | 0 | 0 | 53 | 480 | 1024 | 28 | 199 |
| Set | 0 | 0 | 4 | 400 | 57 | 0 | 154 | ||
| Canals N (Showa Yakuhin Kako; CN) | Powder: zinc oxide, barium sulfate, bismuth carbonate oxide, rosin Liquid: fatty acids, propylene glycol | Fresh | 0 | 0 | 7 | 120 | 29 | 0 | 0 |
| Set | 0 | 0 | 10 | 100 | 16 | 0 | 0 | ||
| AH Plus (Dentsply Sirona; AH) | Paste A: bisphenol-A epoxy resin, bisphenol-F epoxy resin, calcium tungstate, zirconium oxide, iron oxide pigments, Aerosil Paste B: N-dibenziyl-5-oxanonane, aminoadamantane, tricyclodecane-diamine, calcium tungstate, zirconium oxide, Aerosil, silicone oil | Fresh | 0 | 0 | 28 | 320 | 0 | 0 | 0 |
| Set | 0 | 0 | 42 | 660 | 0 | 0 | 0 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kawashima, N.; Hashimoto, K.; Kuramoto, M.; Bakhit, A.; Wakabayashi, Y.; Okiji, T. A Novel Bioactive Endodontic Sealer Containing Surface-Reaction-Type Prereacted Glass-Ionomer Filler Induces Osteoblast Differentiation. Materials 2020, 13, 4477. https://doi.org/10.3390/ma13204477
Kawashima N, Hashimoto K, Kuramoto M, Bakhit A, Wakabayashi Y, Okiji T. A Novel Bioactive Endodontic Sealer Containing Surface-Reaction-Type Prereacted Glass-Ionomer Filler Induces Osteoblast Differentiation. Materials. 2020; 13(20):4477. https://doi.org/10.3390/ma13204477
Chicago/Turabian StyleKawashima, Nobuyuki, Kentaro Hashimoto, Masashi Kuramoto, Alamuddin Bakhit, Yasumiko Wakabayashi, and Takashi Okiji. 2020. "A Novel Bioactive Endodontic Sealer Containing Surface-Reaction-Type Prereacted Glass-Ionomer Filler Induces Osteoblast Differentiation" Materials 13, no. 20: 4477. https://doi.org/10.3390/ma13204477
APA StyleKawashima, N., Hashimoto, K., Kuramoto, M., Bakhit, A., Wakabayashi, Y., & Okiji, T. (2020). A Novel Bioactive Endodontic Sealer Containing Surface-Reaction-Type Prereacted Glass-Ionomer Filler Induces Osteoblast Differentiation. Materials, 13(20), 4477. https://doi.org/10.3390/ma13204477





