Regenerative Endodontic Procedures Using Contemporary Endodontic Materials
Abstract
1. Introduction
2. Terminology
3. Clinical Step-By-Step for Endodontic Regenerative/Revitalization Procedures
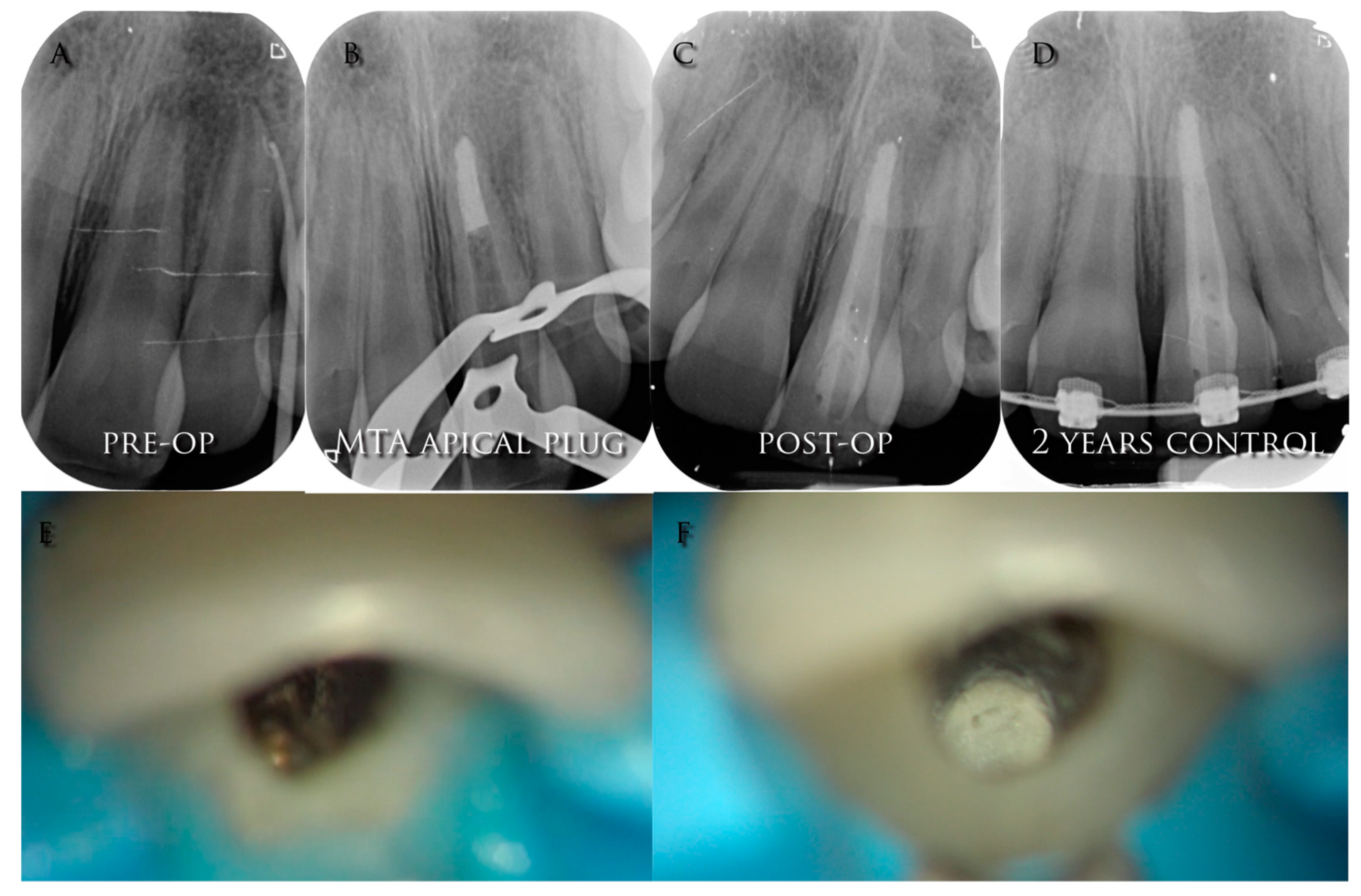
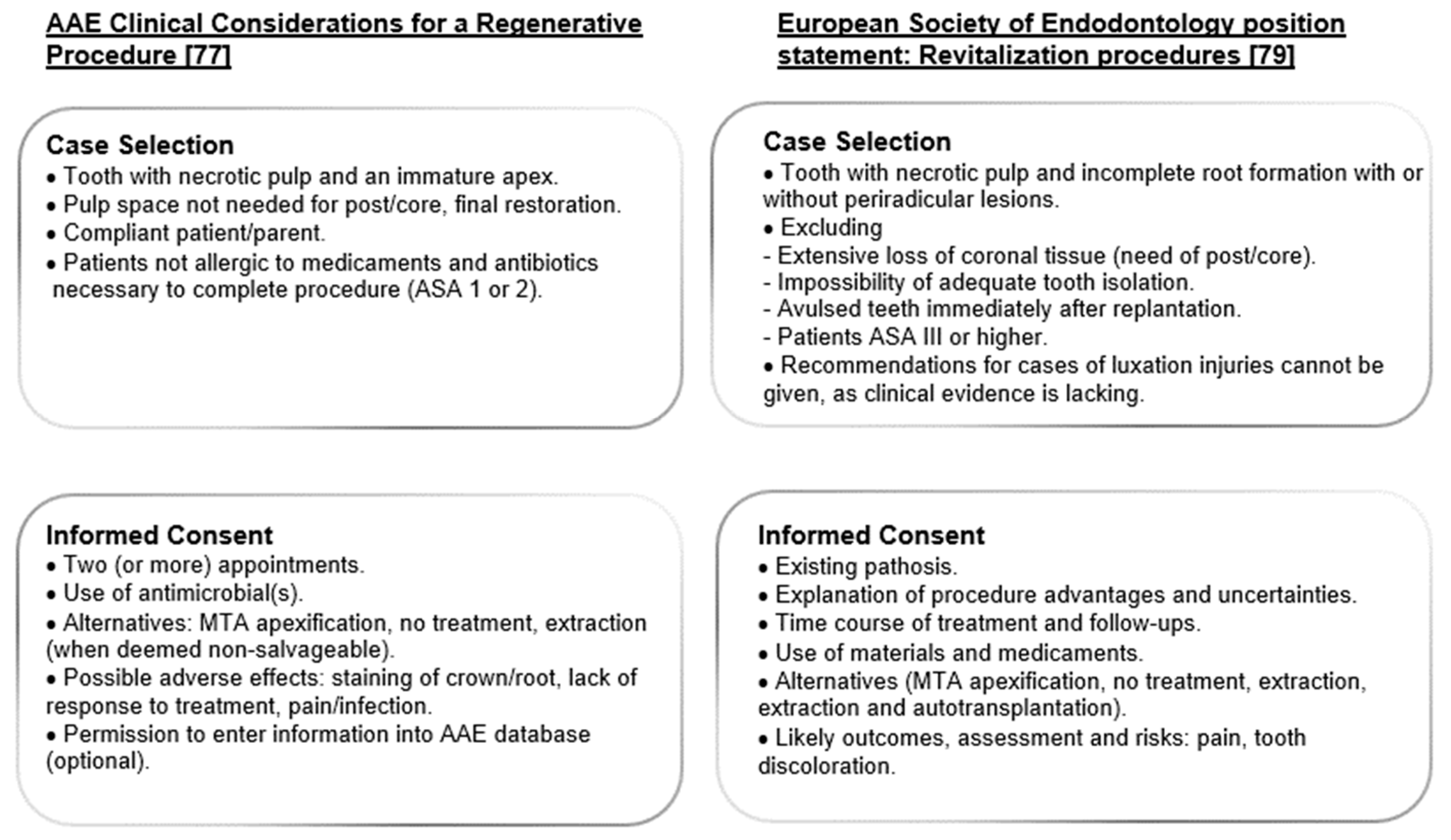
3.1. Case Selection
3.2. Informed Consent
3.3. First Appointment
3.3.1. Irrigation
3.3.2. Antimicrobial Medication
3.3.3. Coronal Seal
3.4. Second Appointment
3.4.1. Irrigation
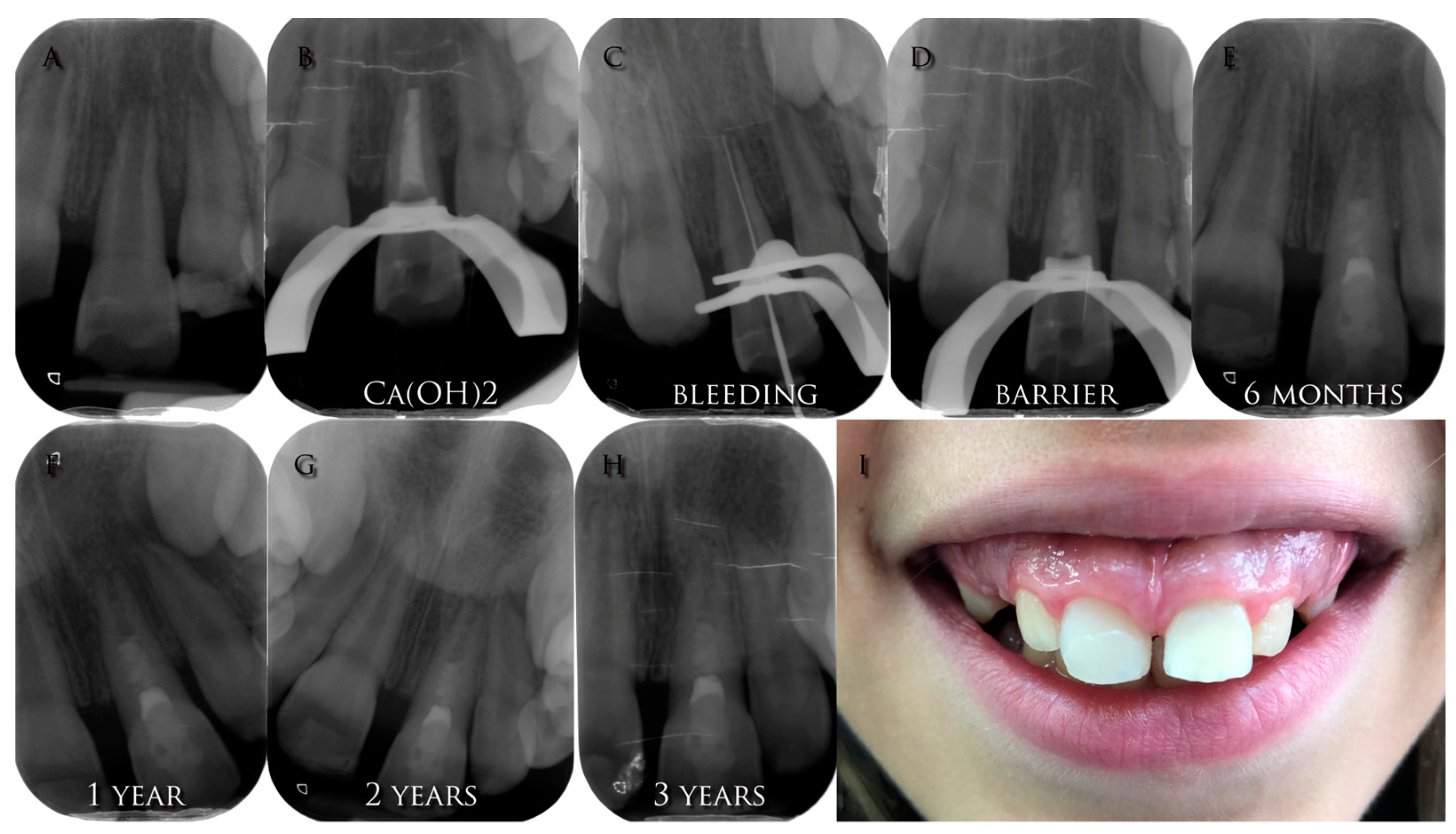
3.4.2. Promoting Blood Clot
3.4.3. Sealing
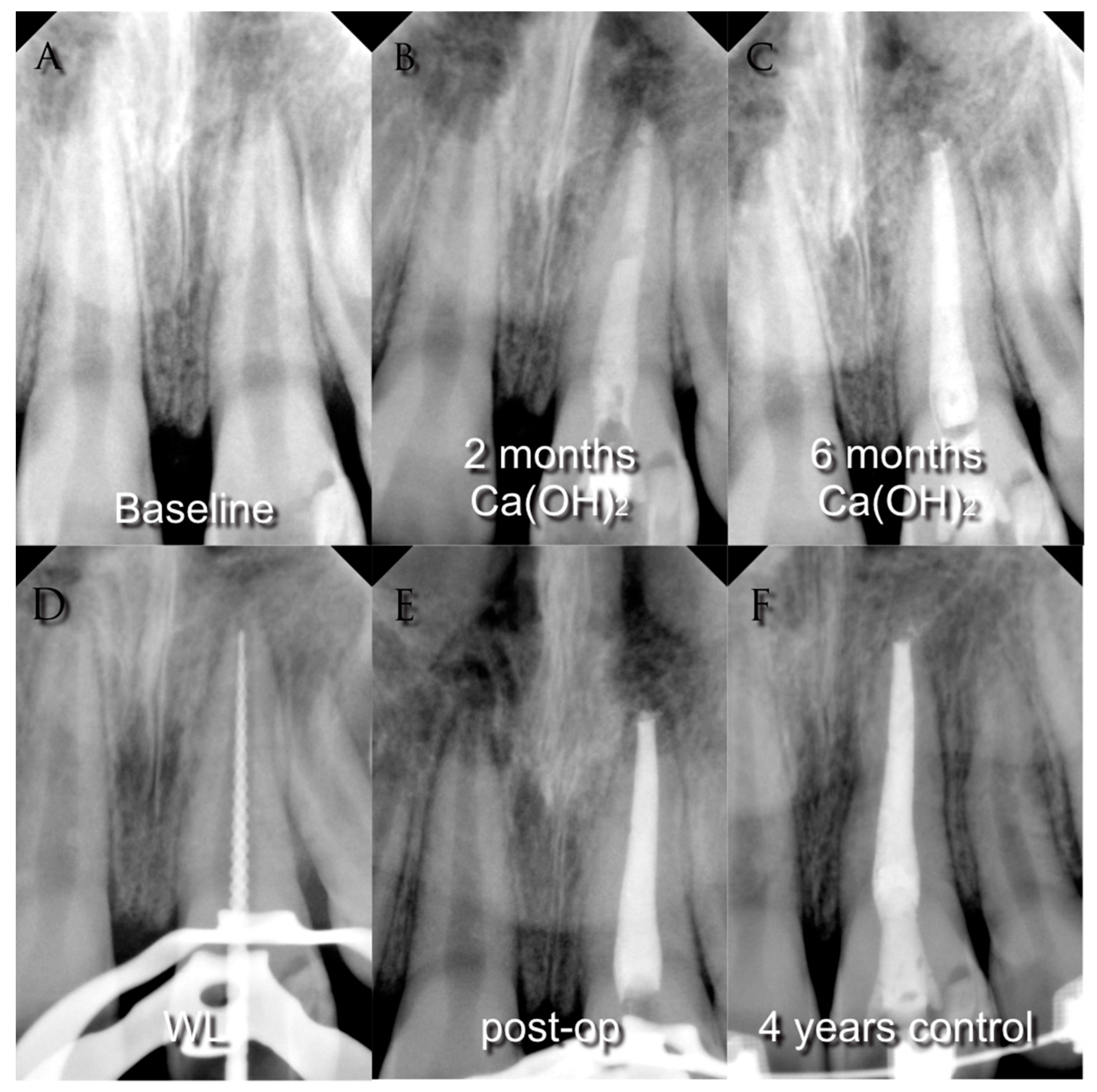
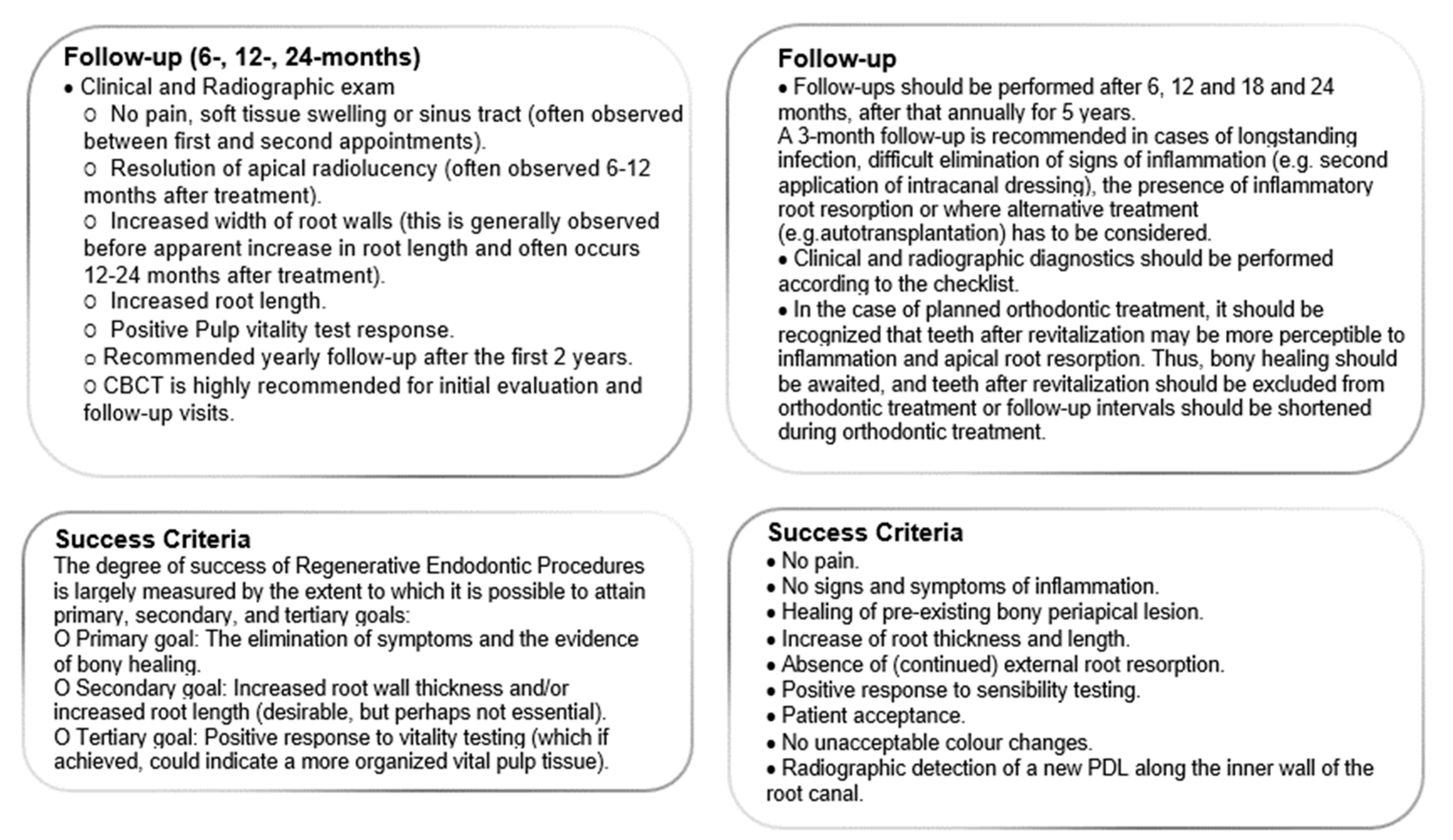
3.5. Follow Up
3.6. Success Criteria
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Frank, A.L. Therapy for the divergent pulpless tooth by continued apical formation. J. Am. Dent. Assoc. 1966, 72, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, H.J. Management of wide-open apex canals with calcium hydroxide. In Proceedings of the 21st Annual Meeting of the American Association of Endodontists, Washington, DC, USA, 17 April 1964. [Google Scholar]
- Chala, S.; Abouqal, R.; Rida, S. Apexification of immature teeth with calcium hydroxide or mineral trioxide aggregate: Systematic review and meta-analysis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2011, 112, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Sheehy, E.C.; Roberts, G.J. Use of calcium hydroxide for apical barrier formation and healing in non-vital immature permanent teeth: A review. Br. Dent. J. 1997, 11, 241–246. [Google Scholar] [CrossRef]
- Hermann, B. Ein weiterer Beitrag zur Frage der Pulpenbehandlung. Zahnärztl Rundsch 1928, 37, 1327–1376. [Google Scholar]
- Mohammadi, Z.; Dummer, P.M. Properties and applications of calcium hydroxide in endodontics and dental traumatology. Int. Endod. J. 2011, 44, 697–730. [Google Scholar] [CrossRef] [PubMed]
- Holland, R.; de Mello, W.; Nery, M.J.; Bernabe, P.F.; de Souza, V. Reaction of human periapical tissue to pulp extirpation and immediate root canal filling with calcium hydroxide. J. Endod. 1977, 3, 63–67. [Google Scholar] [CrossRef]
- Schroder, U.; Granath, L. Early reaction of intact human teeth to calcium hydroxide following experimental pulpotomy and its significance to the development of hard tissue barrier. Odontol. Revy 1971, 22, 379–395. [Google Scholar]
- Rafter, M. Apexification: A review. Dent. Traumatol. 2005, 21, 1–8. [Google Scholar] [CrossRef]
- Abbott, P.V. Apexification with calcium hydroxide—When should the dressing be changed? The case for regular dressing changes. Aust. Endod. J. 1998, 24, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Cvek, M. Prognosis of luxated non-vital maxillary incisors treated with calcium hydroxide and filled with gutta-percha. A retrospective clinical study. Dent. Traumatol. 1992, 8, 45–55. [Google Scholar] [CrossRef]
- Andreasen, J.O.; Farik, B.; Munksgaard, E.C. Long-term calcium hydroxide as a root canal dressing may increase risk of root fracture. Dent. Traumatol. 2002, 18, 134–137. [Google Scholar] [CrossRef]
- Zarei, M.; Afkhami, F.; Malek Poor, Z. Fracture resistance of human root dentin exposed to calcium hydroxide intervisit medication at various time periods: An in vitro study. Dent. Traumatol. 2013, 29, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Valera, M.C.; Albuquerque, M.T.; Yamasaki, M.C.; Vassallo, F.N.; da Silva, D.A.; Nagata, J.Y. Fracture resistance of weakened bovine teeth after long-term use of calcium hydroxide. Dent. Traumatol. 2015, 31, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Yassen, G.H.; Platt, J.A. The effect of nonsetting calcium hydroxide on root fracture and mechanical properties of radicular dentine: A systematic review. Int. Endod. J. 2013, 46, 112–118. [Google Scholar] [CrossRef]
- Stormer, K.; Jacobsen, I.; Attramadal, A. Hvor funkjonsdyktige blir rottfylte unge permanente incisiver? In Nordisk Forening for Pedodonti; Aarsmote: Bergen, Norway, 1988. [Google Scholar]
- Hawkins, J.J.; Torabinejad, M.; Li, Y.; Retamozo, B. Effect of three calcium hydroxide formulations on fracture resistance of dentin over time. Dent. Traumatol. 2015, 31, 380–384. [Google Scholar] [CrossRef]
- Torabinejad, M.; Hong, C.U.; Pitt Ford, T.R. Physical properties of a new root end filling material. J. Endod. 1995, 21, 349–353. [Google Scholar] [CrossRef]
- Torabinejad, M.; Chivian, N. Clinical applications of mineral trioxide aggregate. J. Endod. 1999, 25, 197–205. [Google Scholar] [CrossRef]
- Torabinejad, M.; Watson, T.F.; Pitt Ford, T.R. The sealing ability of a mineral trioxide aggregate as a retrograde root filling material. J. Endod. 1993, 19, 591–595. [Google Scholar] [CrossRef]
- Torabinejad, M.; Hong, C.U.; Pitt Ford, T.R.; Kettering, J.D. Cytotoxicity of four root end filling materials. J. Endod. 1995, 21, 489–492. [Google Scholar] [CrossRef]
- Tawil, P.Z.; Duggan, D.J.; Galicia, J.C. MTA: A Clinical Review. Compend. Contin. Educ. Dent. 2015, 36, 247–264. [Google Scholar]
- Parirokh, M.; Torabinejad, M.; Dummer, P.M.H. Mineral trioxide aggregate and other bioactive endodontic cements: An updated overview—Part I: Vital pulp therapy. Int. Endod. J. 2018, 51, 177–205. [Google Scholar] [CrossRef]
- Islam, I.; Chng, H.K.; Yap, A.U. Comparison of the physical and mechanical properties of MTA and Portland cement. J. Endod. 2006, 32, 193–197. [Google Scholar] [CrossRef]
- Quintana, R.M.; Jardine, A.P.; Grechi, T.R.; Grazziotin-Soares, R.; Ardenghi, D.M.; Scarparo, R.K.; Grecca, F.S.; Kopper, P.M.P. Bone tissue reaction, setting time, solubility, and pH of root repair materials. Clin. Oral Investig. 2018. [Google Scholar] [CrossRef]
- Hachmeister, D.R.; Schindler, W.G.; Walker, W.A., 3rd; Thomas, D.D. The sealing ability and retention characteristics of mineral trioxide aggregate in a model of apexification. J. Endod. 2002, 28, 386–390. [Google Scholar] [CrossRef]
- Witherspoon, D.E.; Ham, K. One-visit apexification: Technique for inducing root-end barrier formation in apical closures. Pract. Proced. Aesthet. Dent. 2001, 13, 455–460. [Google Scholar]
- Lin, J.C.; Lu, J.X.; Zeng, Q.; Zhao, W.; Li, W.Q.; Ling, J.Q. Comparison of mineral trioxide aggregate and calcium hydroxide for apexification of immature permanent teeth: A systematic review and meta-analysis. J. Formos Med. Assoc. 2016, 115, 523–530. [Google Scholar] [CrossRef]
- Damle, S.G.; Bhattal, H.; Loomba, A. Apexification of anterior teeth: A comparative evaluation of mineral trioxide aggregate and calcium hydroxide paste. J. Clin. Pediatr. Dent. 2012, 36, 263–268. [Google Scholar] [CrossRef]
- Torabinejad, M.; Parirokh, M.; Dummer, P.M.H. Mineral trioxide aggregate and other bioactive endodontic cements: An updated overview—part II: Other clinical applications and complications. Int. Endod. J. 2018, 51, 284–317. [Google Scholar] [CrossRef]
- Bonte, E.; Beslot, A.; Boukpessi, T.; Lasfargues, J.J. MTA versus Ca(OH)2 in apexification of non-vital immature permanent teeth: A randomized clinical trial comparison. Clin. Oral Investig. 2015, 19, 1381–1388. [Google Scholar] [CrossRef]
- Torabinejad, M.; Hong, C.U.; Pitt Ford, T.R.; Kettering, J.D. Antibacterial effects of some root end filling materials. J. Endod. 1995, 21, 403–406. [Google Scholar] [CrossRef]
- Fernandez-Yanez Sanchez, A.; Leco-Berrocal, M.I.; Martinez-Gonzalez, J.M. Metaanalysis of filler materials in periapical surgery. Med. Oral Patol. Oral Cir. Bucal 2008, 13, E180-5. [Google Scholar]
- Camilleri, J. Characterization of hydration products of mineral trioxide aggregate. Int. Endod. J. 2008, 41, 408–417. [Google Scholar] [CrossRef]
- Tanomaru-Filho, M.; Tanomaru, J.M.; Barros, D.B.; Watanabe, E.; Ito, I.Y. In vitro antimicrobial activity of endodontic sealers, MTA-based cements and Portland cement. J. Oral Sci. 2007, 49, 41–45. [Google Scholar] [CrossRef]
- Guven, G.; Cehreli, Z.C.; Ural, A.; Serdar, M.A.; Basak, F. Effect of mineral trioxide aggregate cements on transforming growth factor beta1 and bone morphogenetic protein production by human fibroblasts in vitro. J. Endod. 2007, 33, 447–450. [Google Scholar] [CrossRef]
- Tecles, O.; Laurent, P.; Aubut, V.; About, I. Human tooth culture: A study model for reparative dentinogenesis and direct pulp capping materials biocompatibility. J. Biomed. Mater. Res. B Appl. Biomater. 2008, 85, 180–187. [Google Scholar] [CrossRef]
- Sarkar, N.K.; Caicedo, R.; Ritwik, P.; Moiseyeva, R.; Kawashima, I. Physicochemical basis of the biologic properties of mineral trioxide aggregate. J. Endod. 2005, 31, 97–100. [Google Scholar] [CrossRef]
- Bozeman, T.B.; Lemon, R.R.; Eleazer, P.D. Elemental analysis of crystal precipitate from gray and white MTA. J. Endod. 2006, 32, 425–428. [Google Scholar] [CrossRef]
- Reyes-Carmona, J.F.; Felippe, M.S.; Felippe, W.T. Biomineralization ability and interaction of mineral trioxide aggregate and white portland cement with dentin in a phosphate-containing fluid. J. Endod. 2009, 35, 731–736. [Google Scholar] [CrossRef]
- Moore, A.; Howley, M.F.; O’Connell, A.C. Treatment of open apex teeth using two types of white mineral trioxide aggregate after initial dressing with calcium hydroxide in children. Dent. Traumatol. 2011, 27, 166–173. [Google Scholar] [CrossRef]
- Vanka, A.; Ravi, K.S.; Shashikiran, N.D. Apexification with MTA using internal matrix: Report of 2 cases. J. Clin. Pediatr. Dent. 2010, 34, 197–200. [Google Scholar] [CrossRef]
- Sood, R.; Kumar Hans, M.; Shetty, S. Apical barrier technique with mineral trioxide aggregate using internal matrix: A case report. Compend. Contin. Educ. Dent. 2012, 33, e88–e90. [Google Scholar]
- Yadav, P.; Pruthi, P.J.; Naval, R.R.; Talwar, S.; Verma, M. Novel use of platelet-rich fibrin matrix and MTA as an apical barrier in the management of a failed revascularization case. Dent. Traumatol. 2015, 31, 328–331. [Google Scholar] [CrossRef]
- Giovarruscio, M.; Uccioli, U.; Malentacca, A.; Koller, G.; Foschi, F.; Mannocci, F. A technique for placement of apical MTA plugs using modified Thermafil carriers for the filling of canals with wide apices. Int. Endod. J. 2013, 46, 88–97. [Google Scholar] [CrossRef]
- Parirokh, M.; Torabinejad, M. Mineral trioxide aggregate: A comprehensive literature review—Part I: Chemical, physical, and antibacterial properties. J. Endod. 2010, 36, 16–27. [Google Scholar] [CrossRef]
- Parirokh, M.; Torabinejad, M. Mineral trioxide aggregate: A comprehensive literature review—Part III: Clinical applications, drawbacks, and mechanism of action. J. Endod. 2010, 36, 400–413. [Google Scholar] [CrossRef]
- Parirokh, M.; Torabinejad, M. Calcium Silicate-Based Cements. In Mineral Trioxide Aggregate, Properties and Clinical Applications, 1st ed.; Torabinejad, M., Ed.; Wiley Blackwell: Oxford, UK, 2014; pp. 284–320. [Google Scholar]
- Tuloglu, N.; Bayrak, S. Comparative evaluation of mineral trioxide aggregate and bioaggregate as apical barrier material in traumatized nonvital, immature teeth: A clinical pilot study. Niger. J. Clin. Pract. 2016, 19, 52–57. [Google Scholar] [CrossRef]
- Lertmalapong, P.; Jantarat, J.; Srisatjaluk, R.L.; Komoltri, C. Bacterial leakage and marginal adaptation of various bioceramics as apical plug in open apex model. J. Investig. Clin. Dent. 2018, 20, e12371. [Google Scholar] [CrossRef]
- Sharma, S.; Sharma, V.; Passi, D.; Srivastava, D.; Grover, S.; Dutta, S.R. Large Periapical or Cystic Lesions in Association with Roots Having Open Apices Managed Nonsurgically Using 1-step Apexification Based on Platelet-rich Fibrin Matrix and Biodentine Apical Barrier: A Case Series. J. Endod. 2018, 44, 179–185. [Google Scholar] [CrossRef]
- Bani, M.; Sungurtekin-Ekçi, E.; Odabaş, M.E. Efficacy of Biodentine as an Apical Plug in Nonvital Permanent Teeth with Open Apices: An In Vitro Study. Biomed. Res. Int. 2015, 2015, 359275. [Google Scholar] [CrossRef]
- Khetarpal, A.; Chaudhary, S.; Talwar, S.; Verma, M. Endodontic management of open apex using Biodentine as a novel apical matrix. Indian J. Dent. Res. 2014, 25, 513–516. [Google Scholar] [CrossRef]
- Katebzadeh, N.; Dalton, B.C.; Trope, M. Strengthening immature teeth during and after apexification. J. Endod. 1998, 24, 256–259. [Google Scholar] [CrossRef]
- Jeeruphan, T.; Jantarat, J.; Yanpiset, K.; Suwannapan, L.; Khewsawai, P.; Hargreaves, K.M. Mahidol study 1: Comparison of radiographic and survival outcomes of immature teeth treated with either regenerative endodontic or apexification methods: A retrospective study. J. Endod. 2012, 38, 1330–1336. [Google Scholar] [CrossRef]
- Wigler, R.; Kaufman, A.Y.; Lin, S.; Steinbock, N.; Hazan-Molina, H.; Torneck, C.D. Revascularization: A treatment for permanent teeth with necrotic pulp and incomplete root development. J. Endod. 2013, 39, 319–326. [Google Scholar] [CrossRef]
- Nosrat, A.; Homayounfar, N.; Oloomi, K. Drawbacks and unfavorable outcomes of regenerative endodontic treatments of necrotic immature teeth: A literature review and report of a case. J. Endod. 2012, 38, 1428–1434. [Google Scholar] [CrossRef]
- Silujjai, J.; Linsuwanont, P. Treatment Outcomes of Apexification or Revascularization in Nonvital Immature Permanent Teeth: A Retrospective Study. J. Endod. 2017, 43, 238–245. [Google Scholar] [CrossRef]
- Ostby, B.N. The role of the blood clot in endodontic therapy. An experimental histologic study. Acta Odontol. Scand. 1961, 19, 324–353. [Google Scholar] [CrossRef]
- Rule, D.C.; Winter, G.B. Root growth and apical repair subsequent to pulpal necrosis in children. Br. Dent. J. 1966, 120, 586–590. [Google Scholar]
- Nygaard-Ostby, B.; Hjortdal, O. Tissue formation in the root canal following pulp removal. Scand. J. Dent. Res. 1971, 79, 333–349. [Google Scholar] [CrossRef]
- Matusow, R.J. Acute pulpal-alveolar cellulitis syndrome V. Apical closure of immature teeth by infection control: Case report and a possible microbial-immunologic etiology, Part 1. Oral Surg. Oral Med. Oral Pathol. 1991, 71, 737–742. [Google Scholar] [CrossRef]
- Matusow, R.J. Acute pulpal-alveolar cellulitis syndrome V. Apical closure of immature teeth by infection control: The importance of an endodontic seal with therapeutic factors, Part 2. Oral Surg. Oral Med. Oral Pathol. 1991, 72, 96–100. [Google Scholar] [CrossRef]
- Saad, A.Y. Calcium hydroxide and apexogenesis. Oral Surg. Oral Med. Oral Pathol. 1988, 66, 499–501. [Google Scholar] [CrossRef]
- Skoglund, A.; Tronstad, L.; Wallenius, K. A microradiographic study of vascular changes in replanted and autotransplanted teeth in young dogs. Oral Surg. Oral Med. Oral Pathol. 1978, 1, 172–178. [Google Scholar]
- Andreasen, J.O.; Andreasen, F.M. Textbook and Color Atlas of Traumatic Injuries to the Teeth; Munksgaard: Copenhagen, Denmark, 1994. [Google Scholar]
- Nevins, A.; Wrobel, W.; Valachovic, R.; Finkelstein, F. Hard tissue induction into pulpless open-apex teeth using collagen-calcium phosphate gel. J. Endod. 1977, 3, 431–433. [Google Scholar] [CrossRef]
- Amit, V.; Jain, A.; Nayak, U.A.; Bhat, M. Maturogenesis by revascularization in an infected immature permanent tooth. J. Indian Soc. Pedod. Prev. Dent. 2014, 32, 172–175. [Google Scholar] [CrossRef]
- Weisleder, R.; Benitez, C.R. Maturogenesis: Is it a new concept? J. Endod. 2003, 29, 776–778. [Google Scholar] [CrossRef]
- Murray, P.E.; Garcia-Godoy, F.; Hargreaves, K.M. Regenerative endodontics: A review of current status and a call for action. J. Endod. 2007, 33, 377–390. [Google Scholar] [CrossRef]
- Wang, X.; Thibodeau, B.; Trope, M.; Lin, L.M.; Huang, G.T. Histologic characterization of regenerated tissues in canal space after the revitalization/revascularization procedure of immature dog teeth with apical periodontitis. J. Endod. 2010, 36, 56–63. [Google Scholar] [CrossRef]
- Trope, M. Regenerative potential of dental pulp. J. Endod. 2008, 34, S13–S17. [Google Scholar] [CrossRef]
- Andreasen, J.O.; Borum, M.K.; Jacobsen, H.L.; Andreasen, F.M. Replantation of 400 avulsed permanent incisors. 2. Factors related to pulpal healing. Endod. Dent. Traumatol. 1995, 11, 59–68. [Google Scholar] [CrossRef]
- Huang, G.T.; Lin, L.M. Letter to the editor: Comments on the use of the term ‘‘revascularization’’ to describe root regeneration. J. Endod. 2008, 34, 511–512. [Google Scholar] [CrossRef]
- Lenzi, R.; Trope, M. Revitalization procedures in two traumatized incisors with different biological outcomes. J. Endod. 2012, 38, 411–414. [Google Scholar] [CrossRef]
- Geisler, T.M. Clinical considerations for regenerative endodontic procedures. Dent. Clin. N. Am. 2012, 56, 603–626. [Google Scholar] [CrossRef]
- American Association of Endodontists. AAE Position Statement, Scope of Endodontics: Regenerative Endodontics. Available online: http://www.aae.org/uploadedfiles/clinical_resources/guidelines_and_position_statements/scopeofendo_regendo.pdf (accessed on 13 December 2018).
- American Dental Association. CDT 2011–2012: Current Dental Terminology: The ADA Practical Guide to Dental Procedure Codes; American Dental Association: Chicago, IL, USA, 2010. [Google Scholar]
- Galler, K.M.; Krastl, G.; Simon, S.; Van Gorp, G.; Meschi, N.; Vahedi, B.; Lambrechts, P. European Society of Endodontology position statement: Revitalization procedures. Int. Endod. J. 2016, 49, 717–723. [Google Scholar] [CrossRef]
- Hargreaves, K.M.; Giesler, T.; Henry, M.; Wang, Y. Regeneration potential of the young permanent tooth: What does the future hold? J. Endod. 2008, 34, S51–S56. [Google Scholar] [CrossRef]
- Kim, S.G.; Malek, M.; Sigurdsson, A.; Lin, L.M.; Kahler, B. Regenerative endodontics: A comprehensive review. Int. Endod. J. 2018, 51, 1367–1388. [Google Scholar] [CrossRef]
- Petrino, J.A. Revascularization of necrotic pulp of immature teeth with apical periodontitis. Northwest Dent. 2007, 86, 33–35. [Google Scholar]
- Nosrat, A.; Seifi, A.; Asgary, S. Regenerative endodontic treatment (revascularization) for necrotic immature permanent molars: A review and report of two cases with a new biomaterial. J. Endod. 2011, 37, 562–567. [Google Scholar] [CrossRef]
- Banchs, F.; Trope, M. Revascularization of immature permanent teeth with apical periodontitis: New treatment protocol? J. Endod. 2004, 30, 196–200. [Google Scholar] [CrossRef]
- Aksel, H.; Serper, A. Recent considerations in regenerative endodontic treatment approaches. J. Dent. Sci. 2014, 9, 207–213. [Google Scholar] [CrossRef]
- Dewan, R.G.; Kochhar, R.; Bhandari, P.P.; Tyagi, N. Regenerative endodontics in the light of recent research. Ind. J. Dent. Sci. 2013, 5, 132–135. [Google Scholar]
- Torabinejad, M.; Nosrat, A.; Verma, P.; Udochukwu, O. Regenerative endodontic treatment or mineral trioxide aggregate apical plug in teeth with necrotic pulps and open apices: A systematic review and meta-analysis. J. Endod. 2017, 43, 1806–1820. [Google Scholar] [CrossRef]
- Tong, H.J.; Rajan, S.; Bhujel, N.; Kang, J.; Duggal, M.; Nazzal, H. Regenerative endodontic therapy in the management of nonvital immature permanent teeth: A systematic review-outcome evaluation and meta-analysis. J. Endod. 2017, 43, 1453–1464. [Google Scholar] [CrossRef]
- American Association of Endodontists. Considerations for Regenerative Procedures. Available online: https://www.aae.org/specialty/wp-content/uploads/sites/2/2018/06/ConsiderationsForRegEndo_AsOfApril2018.pdf (accessed on 20 October 2018).
- Sonoyama, W.; Liu, Y.; Yamaza, T.; Tuan, R.S.; Wang, S.; Shi, S.; Huang, G.T. Characterization of the apical papilla and its residing stem cells from human immature permanent teeth: A pilot study. J. Endod. 2008, 34, 166–171. [Google Scholar] [CrossRef]
- Murray, P.E.; Stanley, H.R.; Matthews, J.B.; Sloan, A.J.; Smith, A.J. Age-related odontometric changes of human teeth. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2002, 93, 474–482. [Google Scholar] [CrossRef]
- Lovelace, T.W.; Henry, M.A.; Hargreaves, K.M.; Diogenes, A. Evaluation of the delivery of mesenchymal stem cells into the root canal space of necrotic immature teeth after clinical regenerative endodontic procedure. J. Endod. 2011, 37, 133–138. [Google Scholar] [CrossRef]
- Kling, M.; Cvek, M.; Mejàre, I. Rate and predictability of pulp revascularization in therapeutically reimplanted permanent incisors. Endod. Dent. Traumatol. 1986, 2, 83–89. [Google Scholar] [CrossRef]
- Trevino, E.G.; Henry, M.A.; Patwardhan, A. The effect of different irrigation solutions on the survival of stem cells of the apical papilla (SCAP) in a PRP scaffold in human root tips. J. Endod. 2009, 35, 428. [Google Scholar]
- Huang, G.T. A paradigm shift in endodontic management of immature teeth: Conservation of stem cells for regeneration. J. Dent. 2008, 36, 379–386. [Google Scholar] [CrossRef]
- Jung, I.Y.; Lee, S.J.; Hargreaves, K.M. Biologically based treatment of immature permanent teeth with pulpal necrosis: A case series. J. Endod. 2008, 34, 876–887. [Google Scholar] [CrossRef]
- Essner, M.D.; Javed, A.; Eleazer, P.D. Effect of sodium hypochlorite on human pulp cells: An in vitro study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2011, 112, 662–666. [Google Scholar] [CrossRef]
- Spangberg, L.; Engstrom, B.; Langeland, K. Biologic effects of dental materials. 3. Toxicity and antimicrobial effect of endodontic antiseptics in vitro. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1973, 36, 856–870. [Google Scholar] [CrossRef]
- Trevino, E.G.; Patwardhan, A.N.; Henry, M.A.; Perry, G.; Dybdal-Hargreaves, N.; Hargreaves, K.M.; Diogenes, A. Effect of irrigants on the survival of human stem cells of the apical papilla in a platelet-rich plasma scaffold in human root tips. J. Endod. 2011, 37, 1109–1115. [Google Scholar] [CrossRef]
- Diogenes, A.; Henry, M.A.; Teixeira, F.B.; Hargreaves, K.M. An update on clinical regenerative endodontics. Endod. Top. 2013, 28, 2–23. [Google Scholar] [CrossRef]
- Hülsmann, M.; Hahn, W. Complications during root canal irrigation—Literature review and case reports. Int. Endod. J. 2000, 33, 186–193. [Google Scholar] [CrossRef]
- Law, A.S. Considerations for regeneration procedures. J. Endod. 2013, 39 (Suppl. 3), S44–S56. [Google Scholar] [CrossRef]
- Mitchell, R.P.; Yang, S.E.; Baumgartner, J.C. Comparison of apical extrusion of NaOCl using the EndoVac or needle irrigation of root canals. J. Endod. 2010, 36, 338–341. [Google Scholar] [CrossRef]
- Ram, Z. Effectiveness of root canal irrigation. Oral Surg. Oral Med. Oral Pathol. 1977, 44, 306–312. [Google Scholar] [CrossRef]
- Ring, K.C.; Murray, P.E.; Namerow, K.N.; Kuttler, S.; Garcia-Godoy, F. The comparison of the effect of endodontic irrigation on cell adherence to root canal dentin. J. Endod. 2008, 34, 1474–1479. [Google Scholar] [CrossRef]
- Shin, S.Y.; Albert, J.S.; Mortman, R.E. One step pulp revascularization treatment of an immature permanent tooth with chronic apical abscess: A case report. Int. Endod. J. 2009, 42, 1118–1126. [Google Scholar] [CrossRef]
- Basrani, B.R.; Manek, S.; Mathers, D.; Fillery, E.; Sodhi, R.N. Determination of 4-chloroaniline and its derivatives formed in the interaction of sodium hypochlorite and chlorhexidine by using gas chromatography. J. Endod. 2010, 36, 312–314. [Google Scholar] [CrossRef]
- Krishnamurthy, S.; Sudhakaran, S. Evaluation and prevention of the precipitate formed on interaction between sodium hypochlorite and chlorhexidine. J. Endod. 2010, 36, 1154–1157. [Google Scholar] [CrossRef]
- Nagata, J.Y.; Soares, A.J.; Souza-Filho, F.J.; Zaia, A.A.; Ferraz, C.C.; Almeida, J.F.; Gomes, B.P. Microbial evaluation of traumatized teeth treated with triple antibiotic paste or calcium hydroxide with 2% chlorhexidine gel in pulp revascularization. J. Endod. 2014, 40, 778–783. [Google Scholar] [CrossRef]
- Widbiller, M.; Althumairy, R.I.; Diogenes, A. Direct and indirect effect of chlorhexidine on survival of stem cells from the apical papilla and its neutralization. J. Endod. 2019, 45, 156–160. [Google Scholar] [CrossRef]
- Cotti, E.; Mereu, M.; Lusso, D. Regenerative treatment of an immature, traumatized tooth with apical periodontitis: Report of a case. J. Endod. 2008, 34, 611–616. [Google Scholar] [CrossRef]
- Martin, D.E.; De Almeida, J.F.; Henry, M.A.; Khaing, Z.Z.; Schmidt, C.E.; Teixeira, F.B.; Diogenes, A. Concentration-dependent effect of sodium hypochlorite on stem cells of apical papilla survival and differentiation. J. Endod. 2014, 40, 51–55. [Google Scholar] [CrossRef]
- Hoshino, E.; Kurihara-Ando, N.; Sato, I.; Uematsu, H.; Sato, M.; Kota, K.; Iwaku, M. In-vitro antibacterial susceptibility of bacteria taken from infected root dentine to a mixture of ciprofloxacin, metronidazole and minocycline. Int. Endod. J. 1996, 29, 125–130. [Google Scholar] [CrossRef]
- Sato, I.; Ando-Kurihara, N.; Kota, K.; Iwaku, M.; Hoshino, E. Sterilization of infected root-canal dentine by topical application of a mixture of ciprofloxacin, metronidazole and minocycline in situ. Int. Endod. J. 1996, 29, 118–124. [Google Scholar] [CrossRef]
- Cheek, C.C.; Heymann, H.O. Dental and oral discolorations associated with minocycline and other tetracycline analogs. J. Esthet. Dent. 1999, 11, 43–48. [Google Scholar] [CrossRef]
- Kim, J.; Kim, Y.; Shin, S.; Park, J.; Jung, I. Tooth discoloration of immature permanent incisor associated with triple antibiotic therapy: A case report. J. Endod. 2010, 36, 1086–1091. [Google Scholar] [CrossRef]
- Reynolds, K.; Johnson, J.D.; Cohenca, N. Pulp revascularization of necrotic bilateral bicuspids using a modified novel technique to eliminate potential coronal discoloration: A case report. Int. Endod. J. 2009, 42, 84–92. [Google Scholar] [CrossRef]
- Thibodeau, B.; Teixeira, F.; Yamauchi, M.; Caplan, D.J.; Trope, M. Pulp revascularization of immature dog teeth with apical periodontitis. J. Endod. 2007, 33, 680–689. [Google Scholar] [CrossRef]
- Iwaya, S.I.; Ikawa, M.; Kubota, M. Revascularization of an immature permanent tooth with apical periodontitis and sinus tract. Dent. Traumatol. 2001, 17, 185–187. [Google Scholar] [CrossRef]
- Sabrah, A.H.; Yassen, G.H.; Gregory, R.L. Effectiveness of antibiotic medicaments against biofilm formation of Enterococcus faecalis and Porphyromonas gingivalis. J. Endod. 2013, 39, 1385–1389. [Google Scholar] [CrossRef]
- Gomes-Filho, J.E.; Duarte, P.C.; de Oliveira, C.B.; Watanabe, S.; Lodi, C.S.; Cintra, L.T.; Bernabé, P.F. Tissue reaction to a triantibiotic paste used for endodontic tissue self-regeneration of nonvital immature permanent teeth. J. Endod. 2012, 38, 91–94. [Google Scholar] [CrossRef]
- Ruparel, N.B.; Teixeira, F.B.; Ferraz, C.C.; Diogenes, A. Direct effect of intracanal medicaments on survival of stem cells of the apical papilla. J. Endod. 2012, 38, 1372–1375. [Google Scholar] [CrossRef]
- Jadhav, G.; Shah, N.; Logani, A. Revascularization with and without platelet-rich plasma in nonvital, immature, anterior teeth: A pilot clinical study. J. Endod. 2012, 38, 1581–1587. [Google Scholar] [CrossRef]
- Chueh, L.H.; Huang, G.T. Immature teeth with periradicular periodontitis or abscess undergoing apexogenesis: A paradigm shift. J. Endod. 2006, 32, 1205–1213. [Google Scholar] [CrossRef]
- Bose, R.; Nummikoski, P.; Hargreaves, K. A retrospective evaluation of radiographic outcomes in immature teeth with necrotic root canal systems treated with regenerative endodontic procedures. J. Endod. 2009, 35, 1343–1349. [Google Scholar] [CrossRef]
- Windley, W.; Teixeira, F.; Levin, L.; Sigurdsson, A.; Trope, M. Disinfection of immature teeth with a triple antibiotic paste. J. Endod. 2005, 31, 439–443. [Google Scholar] [CrossRef]
- Nosrat, A.; Li, K.L.; Vir, K.; Hicks, M.L.; Fouad, A.F. Is pulp regeneration necessary for root maturation? J. Endod. 2013, 39, 1291–1295. [Google Scholar] [CrossRef]
- Lenherr, P.; Allgayer, N.; Weiger, R.; Filippi, A.; Attin, T.; Krastl, G. Tooth discoloration induced by endodontic materials: A laboratory study. Int. Endod. J. 2012, 45, 942–949. [Google Scholar] [CrossRef]
- Berkhoff, J.A.; Chen, P.B.; Teixeira, F.B.; Diogenes, A. Evaluation of triple antibiotic paste removal by different irrigation procedures. J. Endod. 2014, 40, 1172–1177. [Google Scholar] [CrossRef]
- Shah, N.; Logani, A.; Bhaskar, U.; Aggarwal, V. Efficacy of revascularization to induce apexification/apexogenesis in infected, nonvital, immature teeth: A pilot clinical study. J. Endod. 2008, 34, 919–925. [Google Scholar] [CrossRef]
- Law, A.S. Considerations for regeneration procedures. Pediatr. Dent. 2013, 35, 141–152. [Google Scholar] [CrossRef]
- Neha, K.; Kansal, R.; Garg, P.; Joshi, R.; Garg, D.; Grover, H.S. Management of immature teeth by dentin-pulp regeneration: A recent approach. Med. Oral Patol Oral Cir. Bucal 2011, 16, e997–e1004. [Google Scholar] [CrossRef] [PubMed]
- Thibodeau, B.; Trope, M. Pulp revascularization of a necrotic infected immature permanent tooth: Case report and review of the literature. Pediatr. Dent. 2007, 29, 47–50. [Google Scholar]
- Thomson, A.; Kahler, B. Regenerative endodontics--biologically-based treatment for immature permanent teeth: A case report and review of the literature. Aust. Dent. J. 2010, 55, 446–452. [Google Scholar] [CrossRef]
- Kottoor, J.; Velmurugan, N. Revascularization for a necrotic immature permanent lateral incisor: A case report and literature review. Int. J. Paediatr. Dent. 2013, 23, 310–316. [Google Scholar] [CrossRef]
- Petrino, J.A.; Boda, K.K.; Shambarger, S.; Bowles, W.R.; McClanahan, S.B. Challenges in regenerative endodontics: A case series. J. Endod. 2010, 36, 536–541. [Google Scholar] [CrossRef]
- Chanda, P.M.; Hegde, K.S.; Bhat, S.S.; Sargod, S.S.; Mantha, S.; Chattopadhyay, S. Tissue engineering in endodontics: Root canal revascularization. J. Clin. Pediatr. Dent. 2014, 38, 291–297. [Google Scholar]
- Bègue-Kirn, C.; Smith, A.J.; Ruch, J.V.; Wozney, J.M.; Purchio, A.; Hartmann, D.; Lesot, H. Effects of dentin proteins, transforming growth factor beta 1 (TGF beta 1) and bone morphogenetic protein 2 (BMP2) on the differentiation of odontoblast in vitro. Int. J. Dev. Biol. 1992, 36, 491–503. [Google Scholar]
- Zhao, S.; Sloan, A.J.; Murray, P.E.; Lumley, P.J.; Smith, A.J. Ultrastructural localisation of TGFbeta exposure in dentine by chemical treatment. Histochem. J. 2000, 32, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Galler, K.M.; D’Souza, R.N.; Federlin, M.; Cavender, A.C.; Hartgerink, J.D.; Hecker, S.; Schmalz, G. Dentin conditioning codetermines cell fate in regenerative endodontics. J. Endod. 2011, 37, 1536–1541. [Google Scholar] [CrossRef]
- Iwaya, S.; Ikawa, M.; Kubota, M. Revascularization of an immature permanent tooth with periradicular abscess after luxation. Dent. Traumatol. 2011, 27, 55–58. [Google Scholar] [CrossRef] [PubMed]
- El-Sharkawy, H.; Kantarci, A.; Deady, J.; Hasturk, H.; Liu, H.; Alshahat, M.; Van Dyke, T.E. Platelet-rich plasma: Growth factors and pro- and anti-inflammatory properties. J. Periodontol. 2007, 78, 661–669. [Google Scholar] [CrossRef]
- Ding, R.Y.; Cheung, G.S.; Chen, J.; Yin, X.Z.; Wang, Q.Q.; Zhang, C.F. Pulp revascularization of immature teeth with apical periodontitis: A clinical study. J. Endod. 2009, 35, 745–749. [Google Scholar] [CrossRef] [PubMed]
- Lovelace, T.W.; Henry, M.A.; Hargreaves, K.M. Evaluation of clinically delivered SCAP cells in regenerative endodontic procedures. J. Endod. 2010, 36, 554. [Google Scholar]
- Torabinejad, M.; Rastegar, A.F.; Kettering, J.D.; Pitt Ford, T.R. Bacterial leakage of mineral trioxide aggregate as a root-end filling material. J. Endod. 1995, 21, 109–112. [Google Scholar] [CrossRef]
- Watts, J.D.; Holt, D.M.; Beeson, T.J.; Kirkpatrick, T.C.; Rutledge, R.E. Effects of pH and mixing agents on the temporal setting of tooth-colored and gray mineral trioxide aggregate. J. Endod. 2007, 33, 970–973. [Google Scholar] [CrossRef]
- Belobrov, I.; Parashos, P. Treatment of tooth discoloration after the use of white mineral trioxide aggregate. J. Endod. 2011, 37, 1017–1020. [Google Scholar] [CrossRef]
- Wongwatanasanti, N.; Jantarat, J.; Sritanaudomchai, H.; Hargreaves, K. Effect of bioceramic materials on proliferation and odontoblast differentiation of human stem cells from the apical papilla. J. Endod. 2018, 44, 1270–1275. [Google Scholar] [CrossRef]
- Aldakak, M.M.; Capar, I.D.; Rekab, M.S.; Abboud, S. Single-visit pulp revascularization of a nonvital immature permanent tooth using biodentine. Iran Endod. J. 2016, 11, 246–249. [Google Scholar]
- Zanini, M.; Sautier, J.M.; Berdal, A.; Simon, S. Biodentine induces immortalized murine pulp cell differentiation into odontoblastlike cells and stimulates biomineralization. J. Endod. 2012, 38, 1220–1226. [Google Scholar] [CrossRef]
- Dawood, A.E.; Parashos, P.; Wong, R.H.K.; Reynolds, E.C.; Manton, D.J. Calcium silicate-based cements: Composition, properties, and clinical applications. J. Investig. Clin. Dent. 2017, 8. [Google Scholar] [CrossRef]
- Llaquet, M.; Mercadé, M.; Plotino, G. Regenerative endodontic procedures: A review of the literature and a case report of an immature central incisor. G. Ital. Endod. 2017, 31, 65–72. [Google Scholar] [CrossRef]
- Camilleri, J.; Sorrentino, F.; Damidot, D. Investigation of the hydration and bioactivity of radiopacified tricalcium silicate cement, Biodentine and MTA Angelus. Dent. Mater. 2013, 29, 580–593. [Google Scholar] [CrossRef]
- Grech, L.; Mallia, B.; Camilleri, J. Characterization of set intermediate restorative material, Biodentine, Bioaggregate and a prototype calcium silicate cement for use as root-end filling materials. Int. Endod. J. 2013, 46, 632–641. [Google Scholar] [CrossRef]
- Valles, M.; Mercade, M.; Duran-Sindreu, F.; Bourdelande, J.L.; Roig, M. Influence of light and oxygen on the color stability of five calcium silicate-based materials. J. Endod. 2013, 39, 525–528. [Google Scholar] [CrossRef]
- Rajasekharan, S.; Martens, L.C.; Cauwels, R.G.; Verbeeck, R.M. Biodentine™ material characteristics and clinical applications: A review of the literature. Eur. Arch. Paediatr. Dent. 2014, 15, 147–158. [Google Scholar] [CrossRef]
- EndoSequence Root Repair Material [Product Page on the Internet]. Issuu Inc., USA. Available online: https://issuu.com/brasseler/docs/brasseler-usa-dental-catalog-11/120 (accessed on 13 December 2018).
- Zhou, H.M.; Shen, Y.; Zheng, W.; Li, L.; Zheng, Y.F.; Haapasalo, M. Physical properties of 5 root canal sealers. J. Endod. 2013, 39, 1281–1286. [Google Scholar] [CrossRef]
- Moinzadeh, A.T.; Aznar Portoles, C.; Schembri Wismayer, P.; Camilleri, J. Bioactivity potential of endosequence bc rrm putty. J. Endod. 2016, 42, 615–621. [Google Scholar] [CrossRef]
- Nasseh, A. The rise of bioceramics. Endod. Pract. 2009, 8, 21–26. [Google Scholar]
- Shokouhinejad, N.; Nekoofar, M.H.; Ashoftehyazdi, K.; Zahraee, S.; Khoshkhounejad, M. Marginal adaptation of new bioceramic materials and mineral trioxide aggregate: A scanning electron microscopy study. Iran. Endod. J. 2014, 9, 144–148. [Google Scholar]
- Nair, U.; Ghattas, S.; Saber, M.; Natera, M.; Walker, C.; Pileggi, R. A comparative evaluation of the sealing ability of 2 root-end filling materials: An in vitro leakage study using Enterococcus faecalis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2011, 112, e74–e77. [Google Scholar] [CrossRef]
- Ma, J.; Shen, Y.; Stojicic, S.; Haapasalo, M. Biocompatibility of two novel root repair materials. J. Endod. 2011, 37, 793–798. [Google Scholar] [CrossRef]
- Willershausen, I.; Wolf, T.; Kasaj, A.; Weyer, V.; Willershausen, B.; Marroquin, B.B. Influence of a bioceramic root end material and mineral trioxide aggregates on fibroblasts and osteoblasts. Arch. Oral Biol. 2013, 58, 1232–1237. [Google Scholar] [CrossRef]
- Bukhari, S.; Kohli, M.R.; Setzer, F.; Karabucak, B. Outcome of revascularization procedure: A retrospective case series. J. Endod. 2016, 42, 1752–1759. [Google Scholar] [CrossRef]
- Roghanizadeh, L.; Fazlyab, M. Revascularization and apical plug in an immature molar. Iran Endod. J. 2018, 13, 139–142. [Google Scholar]
- Asgary, S.; Eghbal, M.; Parirokh, M.; Ghoddusi, J.; Kheirieh, S.; Brink, F. Comparison of mineral trioxide aggregate’s composition with Portland cements and a new endodontic cement. J. Endod. 2009, 35, 243–250. [Google Scholar] [CrossRef]
- Asgary, S.; Eghbal, M.; Parirokh, M.; Ghoddusi, J. Effect of two storage solutions on surface topography of two root-end fillings. Aust. Endod. J. 2009, 35, 147–152. [Google Scholar] [CrossRef]
- Palma, P.J.; Marques, J.A.; Falacho, R.I.; Vinagre, A.; Santos, J.M.; Ramos, J.C. Does delayed restoration improve shear bond strength of different restorative protocols to calcium silicate-based cements? Materials (Basel) 2018, 11, 2216. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, C.; Huang, G.T.; Cheung, G.S.; Dissanayaka, W.L.; Zhu, W. Transplantation of dental pulp stem cells and platelet-rich plasma for pulp regeneration. J. Endod. 2012, 38, 1604–1609. [Google Scholar] [CrossRef]
- Torabinejad, M.; Faras, H.; Corr, R.; Wright, K.R.; Shabahang, S. Histologic examinations of teeth treated with 2 scaffolds: A pilot animal investigation. J. Endod. 2014, 40, 515–520. [Google Scholar] [CrossRef]
- Torabinejad, M.; Milan, M.; Shabahang, S.; Wright, K.R.; Faras, H. Histologic examination of teeth with necrotic pulps and periapical lesions treated with 2 scaffolds: An animal investigation. J. Endod. 2015, 41, 846–852. [Google Scholar] [CrossRef]
- Rodriguez-Benitez, S.; Stambolsky, C.; Gutierrez-Perez, J.L.; Torres-Lagares, D.; Segura-Egea, J.J. Pulp revascularization of immature dog teeth with apical periodontitis using triantibiotic paste and platelet-rich plasma: A radiographic study. J. Endod. 2015, 41, 1299–1304. [Google Scholar] [CrossRef]
- Moradi, S.; Talati, A.; Forghani, M.; Jafarian, A.H.; Naseri, M.; Shojaeian, S. Immunohistological evaluation of revascularized immature permanent necrotic teeth treated by platelet-rich plasma: An animal investigation. Cell J. 2016, 18, 389–396. [Google Scholar]
- Altaii, M.; Cathro, P.; Broberg, M.; Richards, L. Endodontic regeneration and tooth revitalisation in immature infected sheep teeth. Int. Endod. J. 2017, 50, 480–491. [Google Scholar] [CrossRef]
- Dianat, O.; Mashhadi Abas, F.; Paymanpour, P.; Eghbal, M.J.; Haddadpour, S.; Bahrololumi, N. Endodontic repair in immature dogs’ teeth with apical periodontitis: Blood clot vs. plasma rich in growth factors scaffold. Dent. Traumatol. 2017, 33, 84–90. [Google Scholar] [CrossRef]
- Torabinejad, M.; Turman, M. Revitalization of tooth with necrotic pulp and open apex by using platelet-rich plasma: A case report. J. Endod. 2011, 37, 265–268. [Google Scholar] [CrossRef]
- Chen, M.Y.; Chen, K.L.; Chen, C.A.; Tayebaty, F.; Rosenberg, P.A.; Lin, L.M. Responses of immature permanent teeth with infected necrotic pulp tissue and apical periodontitis/abscess to revascularization procedures. Int. Endod. J. 2012, 45, 294–305. [Google Scholar] [CrossRef]
- Martin, G.; Ricucci, D.; Gibbs, J.L.; Lin, L.M. Histological findings of revascularized/revitalized immature permanent molar with apical periodontitis using platelet-rich plasma. J. Endod. 2013, 39, 138–144. [Google Scholar] [CrossRef]
- Santiago, C.N.; Pinto, S.S.; Sassone, L.M.; Hirata, R., Jr.; Fidel, S.R. Revascularization technique for the treatment of external inflammatory root resorption: A report of 3 cases. J. Endod. 2015, 41, 1560–1564. [Google Scholar] [CrossRef]
- Subash, D.; Shoba, K.; Aman, S.; Bharkavi, S.K. Revitalization of an immature permanent mandibular molar with a necrotic pulp using platelet-rich fibrin: A case report. J. Clin. Diagn. Res. 2016, 10, ZD21–ZD23. [Google Scholar] [CrossRef]
- Kontakiotis, E.G.; Filippatos, C.G.; Tzanetakis, G.N.; Agrafioti, A. Regenerative endodontic therapy: A data analysis of clinical protocols. J. Endod. 2015, 41, 146–154. [Google Scholar] [CrossRef]
- Chueh, L.H.; Ho, Y.C.; Kuo, T.C.; Lai, W.H.; Chen, Y.H.; Chiang, C.P. Regenerative endodontic treatment for necrotic immature permanent teeth. J. Endod. 2009, 35, 160–164. [Google Scholar] [CrossRef]
- Trope, M. Treatment of the immature tooth with a non-vital pulp and apical periodontitis. Dent. Clin. N. Am. 2010, 54, 313–324. [Google Scholar] [CrossRef]
- Thibodeau, B. Case report: Pulp revascularization of a necrotic, infected, immature, permanent tooth. Pediatr. Dent. 2009, 31, 145–148. [Google Scholar]
- Cehreli, Z.C.; Isbitiren, B.; Sara, S.; Erbas, G. Regenerative endodontic treatment (revascularization) of immature necrotic molars medicated with calcium hydroxide: A case series. J. Endod. 2011, 37, 1327–1330. [Google Scholar] [CrossRef]
- Ritter, A.L.; Ritter, A.V.; Murrah, V.; Sigurdsson, A.; Trope, M. Pulp revascularization of replanted immature dog teeth after treatment with minocycline and doxycycline assessed by laser Doppler flowmetry, radiography and histology. Dent. Traumatol. 2004, 20, 75–84. [Google Scholar] [CrossRef]
- Sonoyama, W.; Liu, Y.; Fang, D.; Yamaza, T.; Seo, B.M.; Zhang, C.; Liu, H.; Gronthos, S.; Wang, C.Y.; Wang, S.; et al. Mesenchymal stem cell-mediated functional tooth regeneration in swine. PLoS ONE 2006, 20, e79. [Google Scholar] [CrossRef]
- Smith, A.J.; Smith, J.G.; Shelton, R.M.; Cooper, P.R. Harnessing the natural regenerative potential of the dental pulp. Dent. Clin. N. Am. 2012, 56, 589–601. [Google Scholar] [CrossRef]
- Narang, I.; Mittal, N.; Mishra, N. A comparative evaluation of the blood clot, platelet-rich plasma, and platelet-rich fibrin in regeneration of necrotic immature permanent teeth: A clinical study. Contemp. Clin. Dent. 2015, 6, 63–68. [Google Scholar] [CrossRef]
- Huang, G.T.; Sonoyama, W.; Liu, Y.; Liu, H.; Wang, S.; Shi, S. The hidden treasure in apical papilla: The potential role in pulp/dentin regeneration and bioroot engineering. J. Endod. 2008, 34, 645–651. [Google Scholar] [CrossRef]
- Palma, P.J.; Ramos, J.C.; Martins, J.B.; Diogenes, A.; Figueiredo, M.H.; Ferreira, P.; Viegas, C.; Santos, J.M. Histologic evaluation of regenerative endodontic procedures with the use of chitosan scaffolds in immature dog teeth with apical periodontitis. J. Endod. 2017, 43, 1279–1287. [Google Scholar] [CrossRef]
- Tobias Duarte, P.C.; Gomes-Filho, J.E.; Ervolino, E.; Marçal Mazza Sundefeld, M.L.; Tadahirowayama, M.; Lodi, C.S.; Dezan-Júnior, E.; Angelo Cintra, L.T. Histopathological condition of the remaining tissues after endodontic infection of rat immature teeth. J. Endod. 2014, 40, 538–542. [Google Scholar] [CrossRef]
- Smith, A.J.; Duncan, H.F.; Diogenes, A.; Simon, S.; Cooper, P.R. Exploiting the Bioactive Properties of the Dentin-Pulp Complex in Regenerative Endodontics. J. Endod. 2016, 42, 47–56. [Google Scholar] [CrossRef]

| Name | Manufacturer | Composition | Setting Time |
|---|---|---|---|
| ProRoot Mineral trioxide aggregate (Grey) | Dentsply Tulsa Dental Specialties, Johnson City, TN, USA | Tricalcium silicate, dicalcium silicate, bismuth oxide, tricalcium aluminate, calcium sulphate dihydrate (gypsum) and calcium aluminoferrite Liquid: distilled water | Initial setting time has been reported from 70 to 74 min, whilst the final setting time is 210–320 min |
| Tooth-coloured ProRoot Mineral trioxide aggregate (White) | Dentsply Tulsa Dental Specialties, Johnson City, TN, USA | Tricalcium silicate, dicalcium silicate, bismuth oxide, tricalcium aluminate, calcium sulphate dihydrate or gypsum Liquid: distilled water | 4 h |
| Angelus MTA (Grey and White) | Angelus, Londrina, Brazil | Tricalcium silicate, dicalcium silicate, bismuth oxide, tricalcium aluminate, calcium oxide, aluminium oxide, silicon dioxide Liquid: distilled water | The initial setting time of White Angelus MTA has been reported to be about 8.5 ± 2.4 min; however, other studies reported 130–230 min as the setting time for Angelus MTA |
| PD MTA White | Produits Dentaires SA, Vevey, Switzerland | SiO2, K2O, Al2O3, Na2O, Fe2O3, SO3, CaO, Bi2O3, MgO. Insoluble residues of CaO, KSO4, NaSO4 and crystalline silica. To mix with distilled water. | The material starts setting after approximately 10 min and the final setting time is 15 min. It is not necessary to wait for the final setting to continue the treatment procedure. |
| Endocem MTA | Maruchi, Wonju, Korea | CaO, Al2O3, SiO2, MgO, Fe2O3, SO3, TiO2, H2O/CO2, bismuth oxide | 4.5–15 min |
| MicroMega MTA | MicroMega, Besancon, France | Tricalcium silicate, dicalcium silicate, tricalcium aluminate, bismuth oxide, calcium sulphate dehydrate and magnesium oxide | The manufacturer has claimed that the MicroMega MTA setting time is 20 min; however, there are reports that announced MM MTA has a setting time of 120–150 min |
| MTA Bio | Angelus; Londrina, or Angelus Solucoes Odontologicas, PR, Brazil) | Portland cement and bismuth oxide | The initial setting time of MTA Bio is 11 min. The final setting time of the material is 23.22 min |
| MTA Plus (White) | Avalon Biomed Inc., Bradenton, FL, USA | Tricalcium silicate, 2CaOSiO2, Bi2O3, 3CaOAl2O3 and CaSO4 | MTA Plus setting time is 128 ± 8 min. In contact with moisture the material needs longer time to set |
| MTA Plus (Grey) | Avalon Biomed Inc., Bradenton, FL, USA | Tricalcium silicate, dicalcium silicate, bismuth oxide, tricalcium aluminium oxide, calcium sulphate and Ca2(Al,Fe)2O5 | Initial Setting Time at 37 °C: ~15 min when thickly mixed with gel; otherwise longer for sealer (~3 h.) |
| OrthoMTA | BioMTA, Seoul, Korea | Tricalcium silicate, dicalcium silicate, tricalcium aluminate, tetracalcium aluminoferrite, free calcium oxide and bismuth oxide | 324.0 ± 2.1 min |
| RetroMTA | BioMTA, Seoul, Korea | Calcium carbonate, silicon oxide, aluminium oxide and hydraulic calcium zirconia complex; Liquid: water | Initial setting time of 150–180 s and final setting time of 360 min |
| Aureoseal MTA | Giovanni Ogna and Figli, Muggio, Milano, Italy | The powder consists of Portland cement, bismuth oxide, setting-time controllers, plastifying agents and radiopaque substances. The liquid is distilled water | No setting time has been reported for the material |
| CPM MTA | EGEO SRL, Buenos Aires, Argentina | MTA, calcium chloride, calcium carbonate, sodium citrate, propylene glycol alginate and propylene glycol | The initial setting time of End-CPM is 6–15 min, whilst the material’s final setting time is 22–27 min |
| Name | Manufacturer | Composition | Setting Time |
|---|---|---|---|
| BioAggregate | Innovative BioCeramix, Vancouver, BC, Canada | Tricalcium silicate, dicalcium silicate, calcium phosphate monobasic, amorphous silicon oxide and tantalum pentoxides Liquid: deionized water | Based on the manufacturer data sheet, BioAggregate has a setting time of 240 min |
| Biodentine | Septodont, Saint-Maur-desFosses Cedex, France | Tricalcium silicate, dicalcium silicate, calcium carbonate, zirconium oxide, calcium oxide, iron oxide Liquid: calcium chloride, a hydrosoluble polymer and water | The setting time of Biodentine has been reported as 6.5–45 min |
| Calcium-enriched mixture (CEM) cement | BioniqueDent, Tehran, Iran | Calcium oxide, silicon dioxide, Al2O3, MgO, SO3, P2O5, Na2O, Cl and H&C Liquid: water-based solution | 50 min |
| EndoBinder | Binderware, Sao Carlos, Brazil | Al2O3 and CaO | 60 min |
| Endocem Zr | Maruchi, Wonju, Korea | Calcium oxide, silicon dioxide, aluminium oxide, magnesium oxide, ferrous oxide, zirconium oxide | - |
| EndoSequence, RRM, RRP | Brasseler, Savannah, GA, USA | Zirconium oxide, calcium silicates, tantalum oxide, calcium phosphate monobasic and filling and thickening agents | The setting time of EndoSequence putty is 61.1 ± 2.5 min and the final setting time is 208 ± 10 min |
| NeoMTA Plus | Avalon Biomed Inc., Bradenton, FL, USA | Tricalcium silicate, dicalcium silicate, tantalite, calcium sulphate and silica | NeoMTA Plus has had a 50- to 60-min setting time when prepared with putty consistency; otherwise, when used as a root canal sealer with loose consistency, it may take 5 h to set |
| Quick-Set | Avalon Biomed Inc., Bradenton, FL, USA, patent pending | Monocalcium aluminate powder that contains bismuth oxide (as a radiopacifier) and hydroxyapatite | 12 min |
| iRoot FS (Fast setting), iRoot BP (Injectable) and iRoot BP Plus (Putty) | Innovative BioCeramix Inc., Vancouver, Canada | iRoot FS: calcium silicates, zirconium oxide, tantalum oxide and calcium phosphate monobasic iRoot BP (BioCeramix Inc.) and EndoSequence BC sealer (Brasseler USA) have had the same formula including zirconium oxide, calcium silicates, tantalum oxide, calcium phosphate monobasic, and filler and thickening agents | iRoot FS showed setting after 1 h, iRoot BP and iRoot BP Plus became solid after 5–7 days |
| Tech Biosealer Capping, Tech Biosealer Root End, Tech Biosealer Apex | Isasan, Como, Italy | Mixture of white CEM, calcium sulphate, calcium chloride, bismuth oxide, montmorillonite | The final setting time of various types of Tech Biosealer differ from each other. Tech Biosealer Capping has a final setting time of 55 min. |
| Name | Manufacturer | Composition | Setting Time |
|---|---|---|---|
| BioRoot RCS (Root canal sealer) | Septodont, Saint-Maur-desFosses Cedex, France | Tricalcium silicate, zirconium oxide (opacifier) and excipients in its powder form, and calcium chloride and excipients as an aqueous liquid | Less than 4 h |
| Endosequence BC (Bioceramic) Sealer | Brasseler, Savannah, GA, USA | Zirconium oxide, calcium silicates, calcium phosphate monobasic, calcium hydroxide, filler and thickening agents. | Setting time is 4 h measured according to ISO 6876:2001. However, in very dry root canals, the setting time can be more than 10 h. |
| iRoot SP (Sealer) | Innovative BioCeramix Inc., Vancouver, Canada | iRoot SP:zirconium oxide, calcium silicates, calcium phosphate, calcium hydroxide, filler and thickening agents | 4 h |
| Tech Biosealer Endo | Isasan, Como, Italy | Mixture of white CEM, calcium sulphate, calcium chloride, bismuth oxide, montmorillonite | Tech Biosealer Endo has a final setting time of 77 min |
| EndoSeal MTA | Maruchi, Wonju, Korea | Calcium silicates, calcium aluminates, calcium aluminoferrite, calcium sulphates, radiopacifier and a thickening agent | 12.31 min |
| MTA Fillapex | Angelus Industria de Produtos Odontologicos S/A, Londrina, Brazil | A MTA root canal sealer with nanoparticles of silica | The material’s setting time is 19.3 min. In dry conditions, the material fails to set |
| TheraCal LC (Light cured) | Bisco Inc., Schaumburg, IL, USA | CaO, Sr glass, fumed silica, barium sulphate, barium zirconate, Portland cement type III and resin containing Bis-GMA (bisphenol A-glycidyl methacrylate) and PEGDMA (polyethylene glycol-dimethacrylate) | The setting time has been reported to be 0.3 min because of the use of light cure technology |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Staffoli, S.; Plotino, G.; Nunez Torrijos, B.G.; Grande, N.M.; Bossù, M.; Gambarini, G.; Polimeni, A. Regenerative Endodontic Procedures Using Contemporary Endodontic Materials. Materials 2019, 12, 908. https://doi.org/10.3390/ma12060908
Staffoli S, Plotino G, Nunez Torrijos BG, Grande NM, Bossù M, Gambarini G, Polimeni A. Regenerative Endodontic Procedures Using Contemporary Endodontic Materials. Materials. 2019; 12(6):908. https://doi.org/10.3390/ma12060908
Chicago/Turabian StyleStaffoli, Simone, Gianluca Plotino, Barbara G. Nunez Torrijos, Nicola M. Grande, Maurizio Bossù, Gianluca Gambarini, and Antonella Polimeni. 2019. "Regenerative Endodontic Procedures Using Contemporary Endodontic Materials" Materials 12, no. 6: 908. https://doi.org/10.3390/ma12060908
APA StyleStaffoli, S., Plotino, G., Nunez Torrijos, B. G., Grande, N. M., Bossù, M., Gambarini, G., & Polimeni, A. (2019). Regenerative Endodontic Procedures Using Contemporary Endodontic Materials. Materials, 12(6), 908. https://doi.org/10.3390/ma12060908








