The Influence of Thrust Force on the Vitality of Bone Chips Harvested for Autologous Augmentation during Dental Implantation
Abstract
1. Introduction
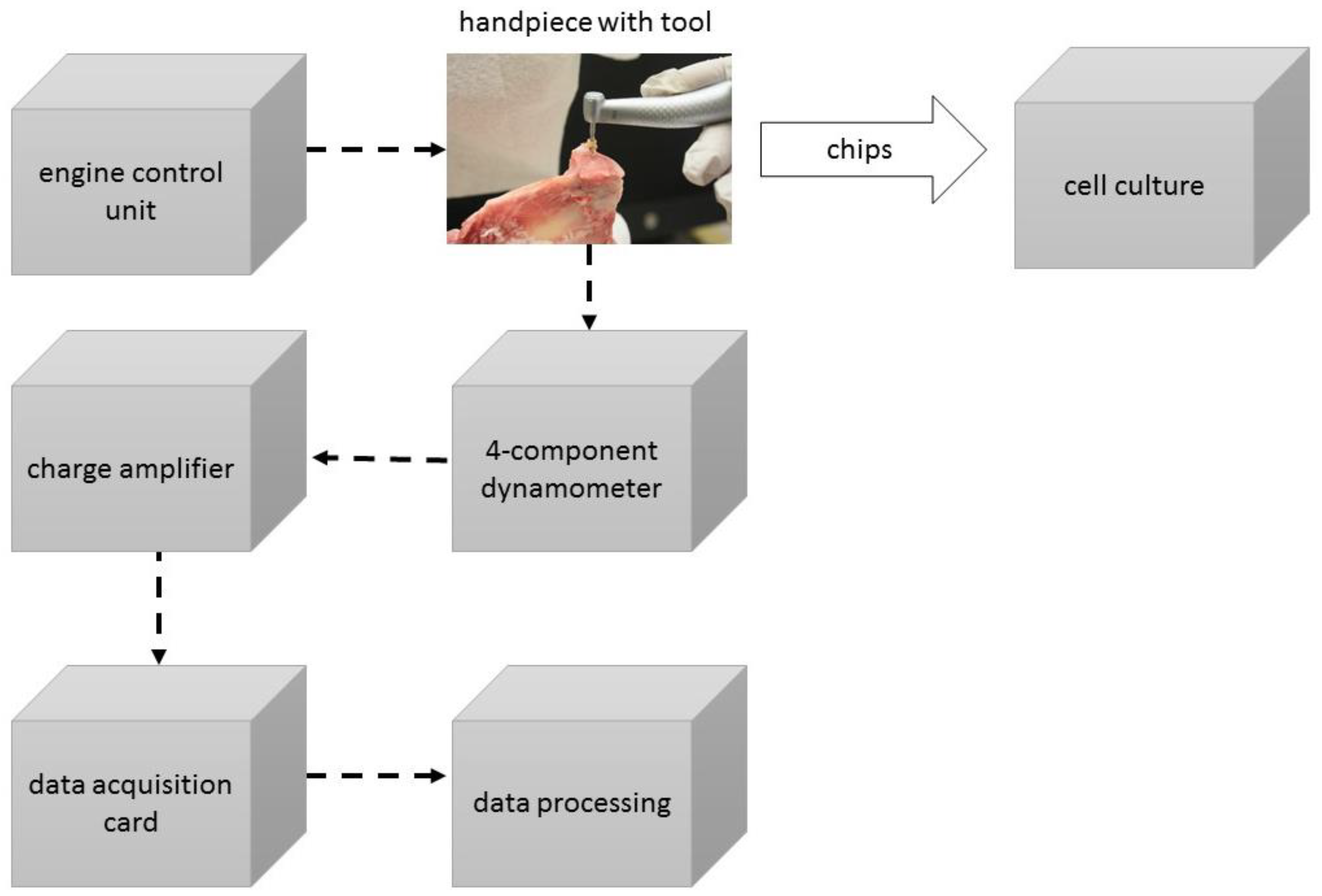
2. Materials and Methods
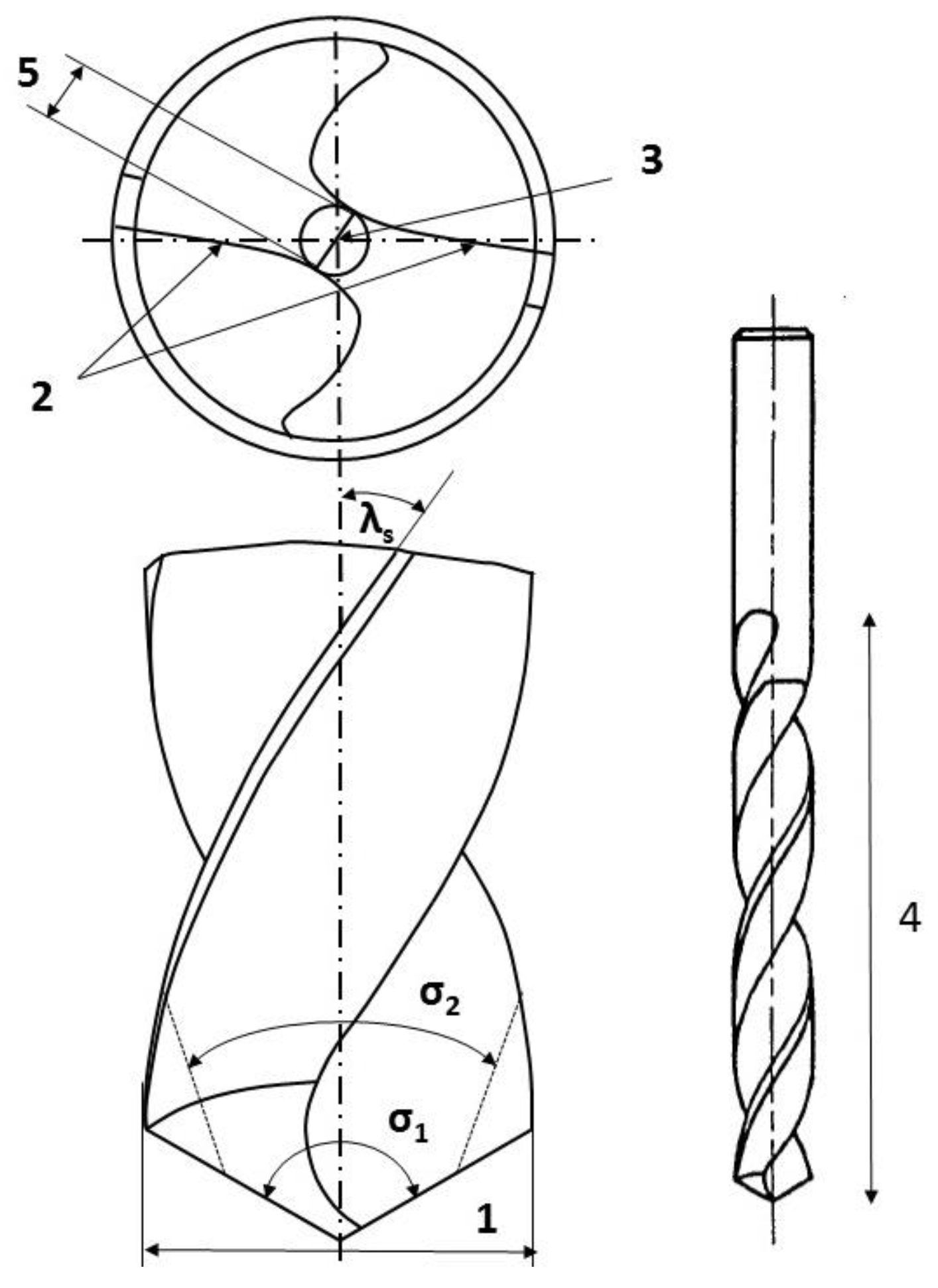
2.1. Implant Drills
2.2. Bone Material
2.3. Conducting the Extraction of Chips
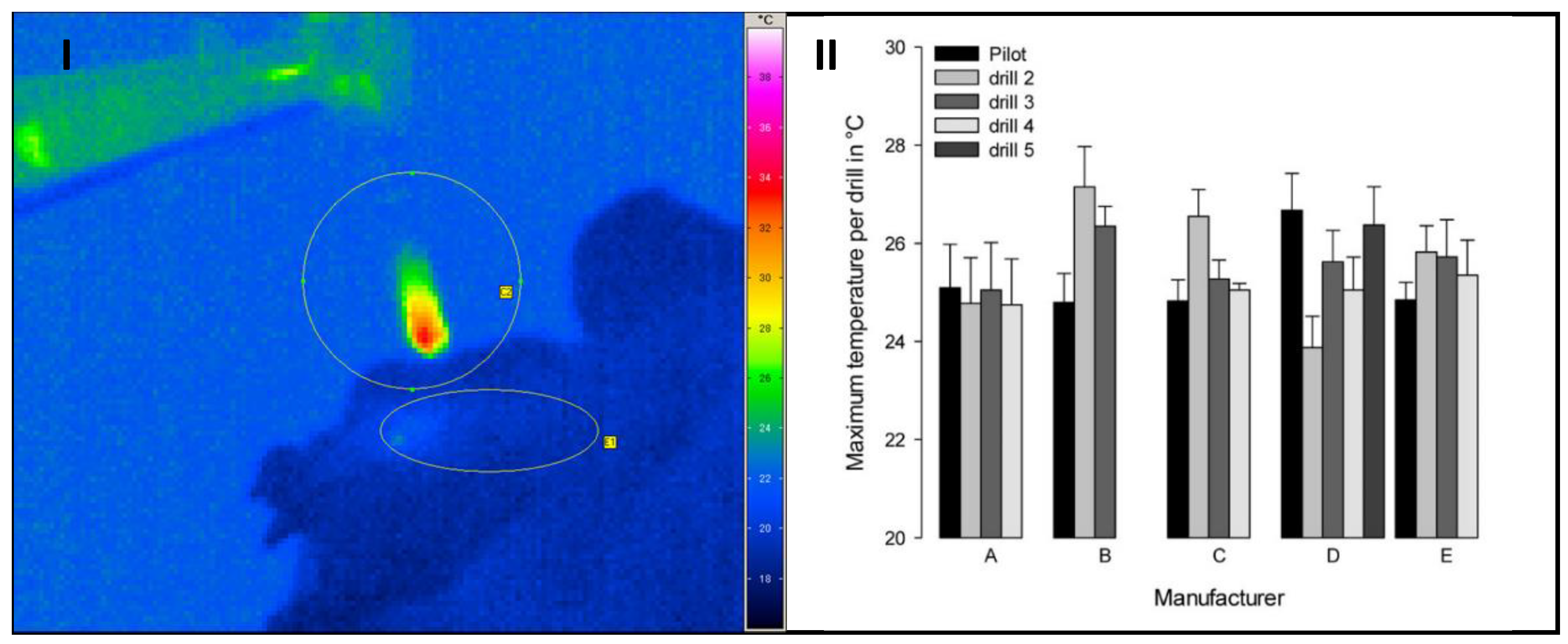
2.4. Temperature Measurement
2.5. Analysis of the Bone Chips
2.5.1. Determination of the Chip Mass
2.5.2. Determination of the Chip Sizes
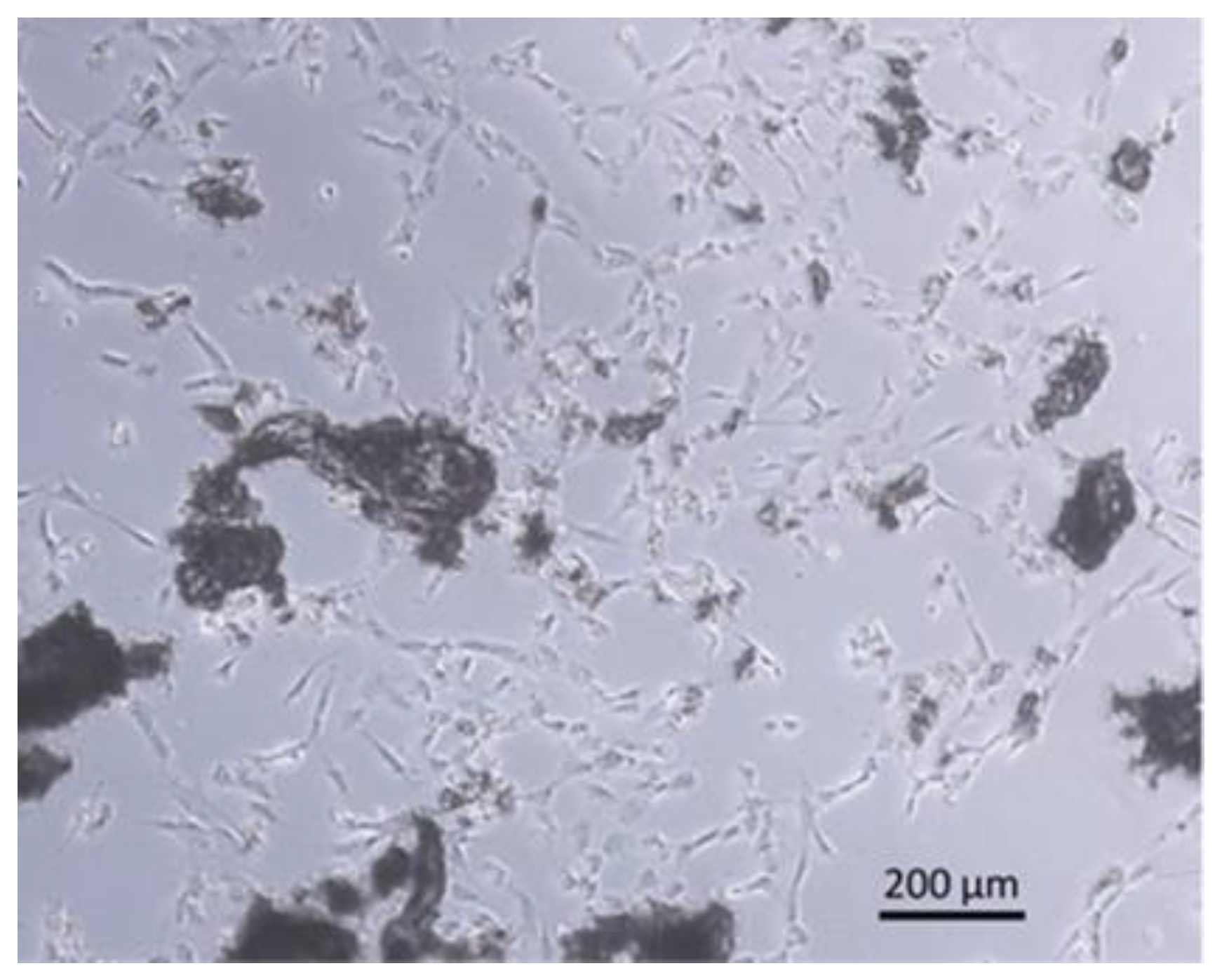
2.5.3. Cell Culture
2.5.4. Determination of the Number of Outgrown Cells via DNA Content
2.5.5. Data Expression and Statistics
3. Results
3.1. Drill Geometry
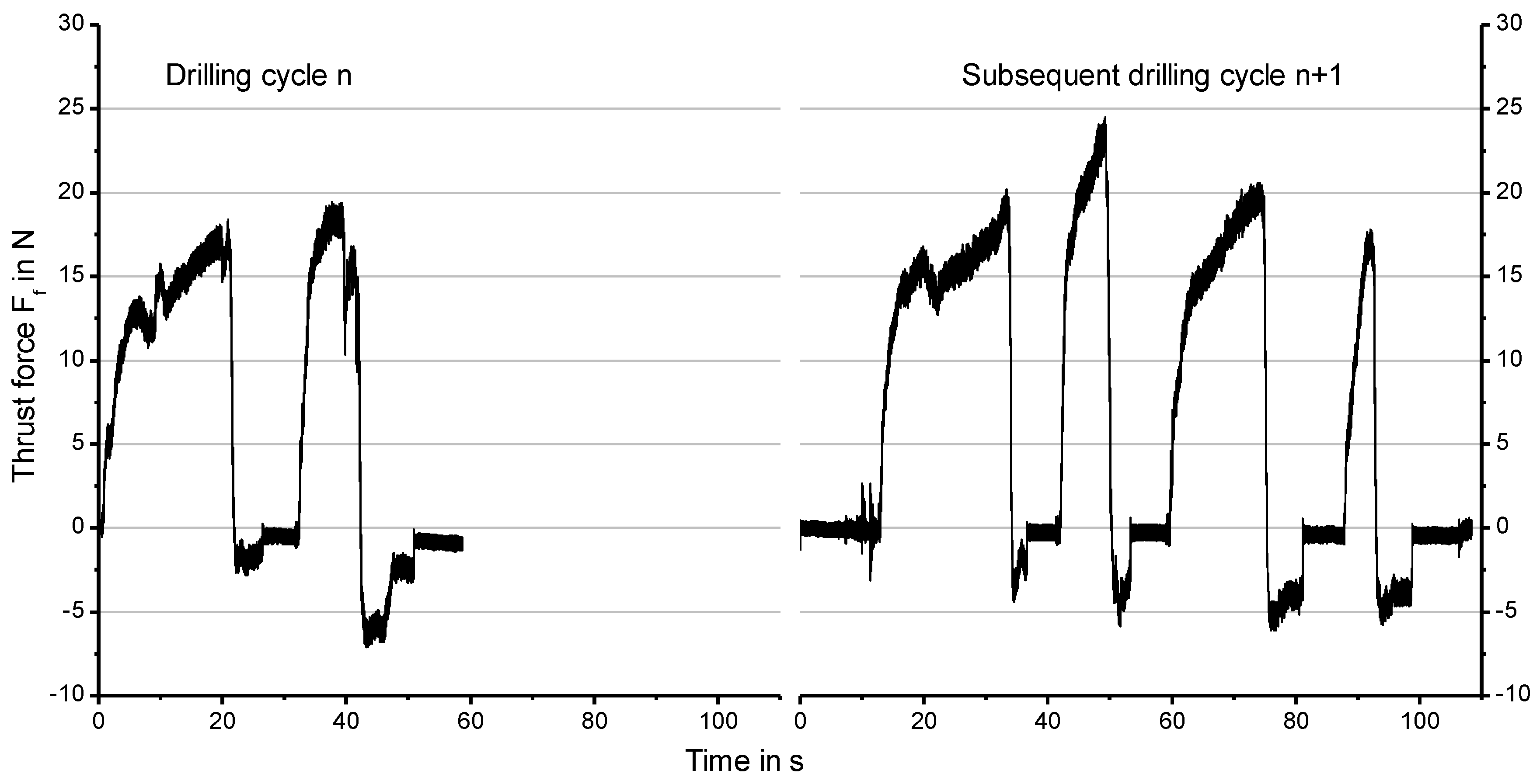
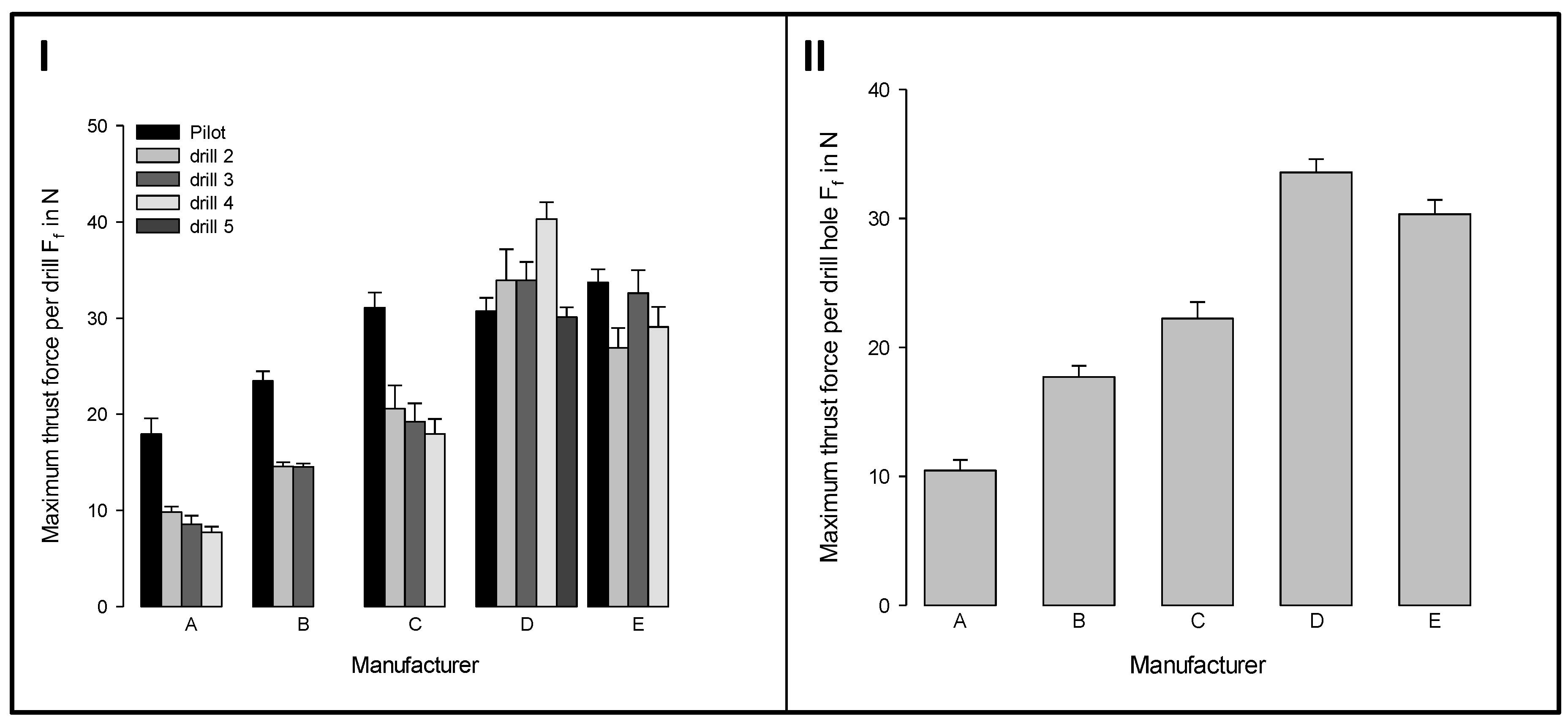
3.2. Influence of the Thrust Force
3.3. Influence of the Temperature
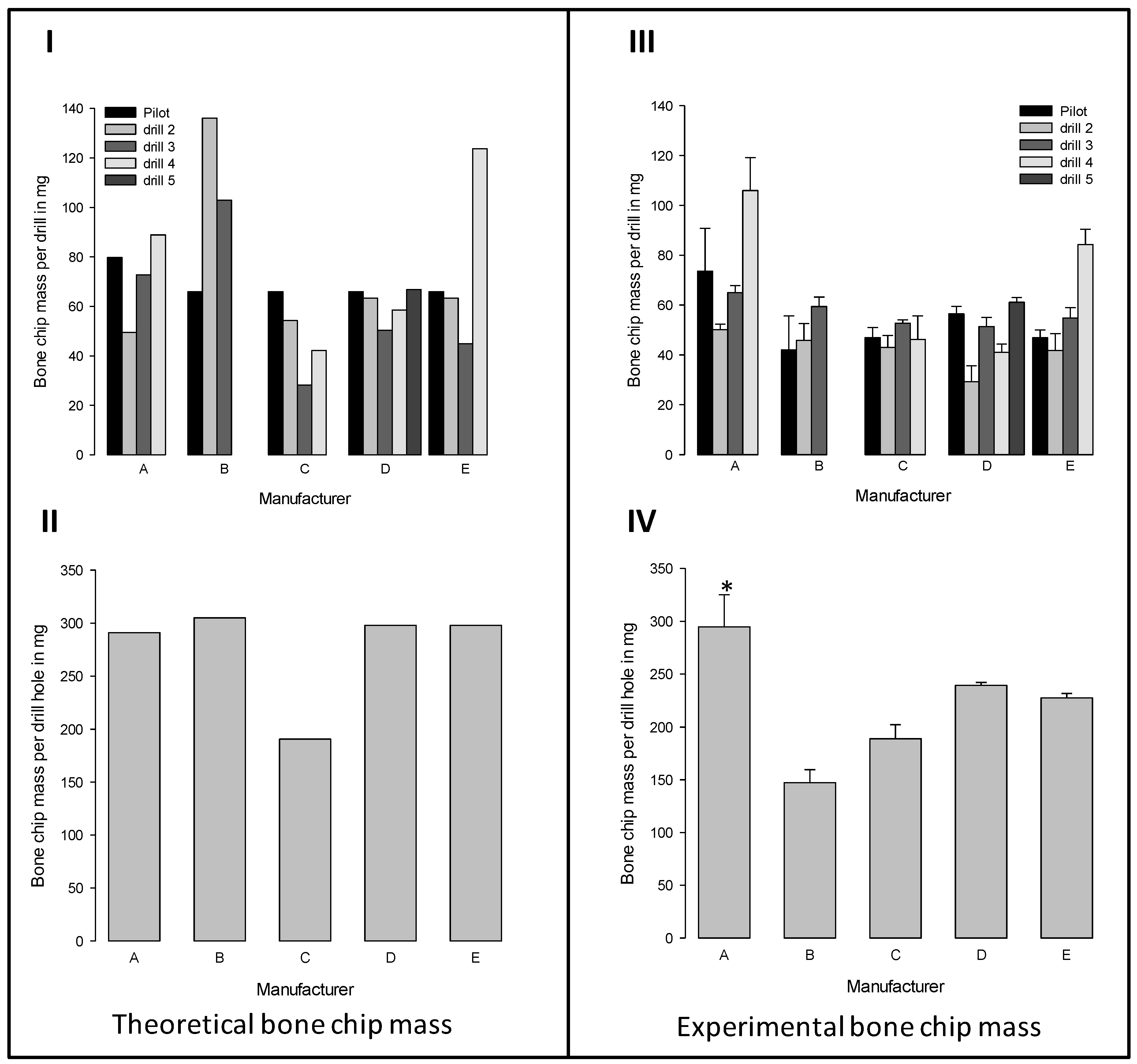
3.4. Theoretical and Experimental Bone Chip Mass
3.5. Bone Chip Size and Their Geometry
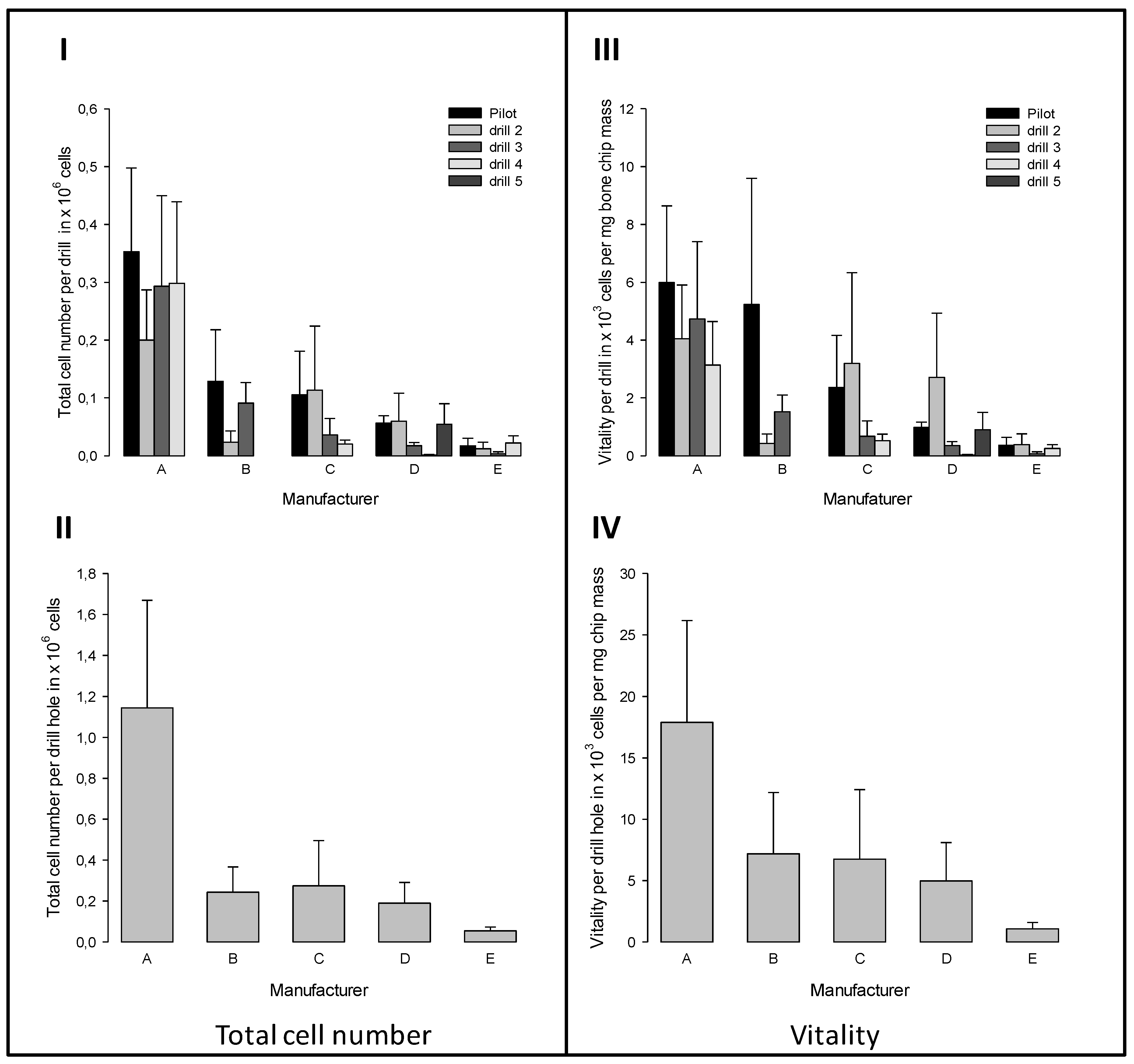
3.6. Vitality of the Bone Chips
4. Discussion
- The material of the drilling tools has a high thermal conductivity. In addition, the drills are removed relatively slowly from the drilling hole. This means that the withdrawal speed is low. The position of the drilling tools varies in the area of the focal point of the IR camera due to the manual drilling process. Only minor temperature changes are measured in the area of the tool cutting edge due to the interaction of these factors.
- The measured temperature is influenced, among other things, by the emission coefficient, which is effective in the cutting area. This, in turn, varies depending on the sample, since the cutting area is generally wetted with bone material, drill chips, tissue, or blood.
- The effect size is too small to statistically confirm the differences. In the opinion of some users/surgeons, water-cooling of the drill bit should be dispensed and, in return, the rotational speed should be lowered to obtain bone chips that are as vital as possible for augmentation.
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aghaloo, T.L.; Moy, P.K. Which hard tissue augmentation techniques are the most successful in furnishing bony support for implant placement? Int. J. Oral Maxillofac. Implant. 2007, 22, 49–70. [Google Scholar]
- Giesenhagen, B.; Martin, N.; Jung, O.; Barbeck, M. Bone Augmentation and Simultaneous Implant Placement with Allogenic Bone Rings and Analysis of Its Purification Success. Materials 2019, 12, 1291. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, A.; Wilde, F.; Heufelder, M.; Winter, K.; Schramm, A. Autogenous bone grafts in oral implantology-is it still a "gold standard"? A consecutive review of 279 patients with 456 clinical procedures. Int. J. Implant. Dent. 2017, 3, 23. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, Z.; Sima, C.; Glogauer, M. Bone Replacement Materials and Techniques Used for Achieving Vertical Alveolar Bone Augmentation. Materials 2015, 8, 2953–2993. [Google Scholar] [CrossRef]
- Haggerty, C.J.; Vogel, C.T.; Fisher, G.R. Simple Bone Augmentation for Alveolar Ridge Defects. Oral Maxillofac. Surg. Clin. N. Am. 2015, 27, 203–226. [Google Scholar] [CrossRef] [PubMed]
- Aloy-Prósper, A.; Maestre-Ferrin, L.; Peñarrocha-Oltra, D.; Peñarrocha-Diago, M. Bone regeneration using particulate grafts: an update. Medicina Oral Patología Oral y Cirugia Bucal 2011, 16, e210–e214. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lauer, G.; Dent, M.; Schilli, W.; Med, P. Collected Implant Cavity Borings Used as Peri-lmplant Osseous Augmentation Material. J. Periodontol. 1997, 9, 437–443. [Google Scholar]
- Stacchi, C.; Cusimano, P.; Berton, F.; Lauritano, F.; Cervino, G.; Cicciù, M.; Lombardi, T.; Di Lenarda, R. Bone Scrapers Versus Piezoelectric Surgery in the Lateral Antrostomy for Sinus Floor Elevation. J. Craniofacial Surg. 2017, 28, 1191–1196. [Google Scholar] [CrossRef]
- Smukler, H.; Capri, D.; Landi, L. Harvesting bone in the recipient sites for ridge augmentation. Int. J. Periodontics Restor. Dent. 2008, 28, 411–419. [Google Scholar]
- Pape, H.C.; Evans, A.; Kobbe, P. Autologous Bone Graft: Properties and Techniques. J. Orthop. Trauma 2010, 24, 36–40. [Google Scholar] [CrossRef]
- Möhlhenrich, S.; Modabber, A.; Steiner, T.; Mitchell, D.; Hölzle, F.; Mitchell, D. Heat generation and drill wear during dental implant site preparation: systematic review. Br. J. Oral Maxillofac. Surg. 2015, 53, 679–689. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, A.; Albrektsson, T.; Grane, B.; McQueen, D. Thermal injury to bone. Int. J. Oral Surg. 1982, 11, 115–121. [Google Scholar] [CrossRef]
- Pellicer-Chover, H.; Peñarrocha-Oltra, D.; Aloy-Prosper, A.; Sanchis-Gonzalez, J.-C.; Peñarrocha-Diago, M.; Peñarrocha-Diago, M. Comparison of peri-implant bone loss between conventional drilling with irrigation versus low-speed drilling without irrigation. Med. Oral Patología Oral y Cirugia Bucal 2017, 22, e730–e736. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.; Lin, X.; Wang, S.-L.; Guo, L.-H.; Wang, X.-Y.; Li, J. Osteogenic potential of three different autogenous bone particles harvested during implant surgery. Oral Dis. 2017, 23, 1099–1108. [Google Scholar] [CrossRef] [PubMed]
- Manzano-Moreno, F.J.; Herrera-Briones, F.J.; Linares-Recatala, M.; Ocaña-Peinado, F.M.; Reyes-Botella, C.; Vallecillo-Capilla, M.F. Bacterial Contamination Levels of Autogenous Bone Particles Collected by 3 Different Techniques for Harvesting Intraoral Bone Grafts. J. Oral Maxillofac. Surg. 2015, 73, 424–429. [Google Scholar] [CrossRef]
- Oh, J.-H.; Fang, Y.; Jeong, S.-M.; Choi, B.-H. The effect of low-speed drilling without irrigation on heat generation: an experimental study. J. Korean Assoc. Oral Maxillofac. Surg. 2016, 42, 9. [Google Scholar] [CrossRef]
- Marenzi, G.; Sammartino, J.C.; Scherillo, F.; Rengo, C.; De Rosa, A.; Graziano, V.; Spagnuolo, G. Comparative Analysis of the Chemical Composition and Microstructure Conformation Between Different Dental Implant Bone Drills. Materials 2019, 12, 1866. [Google Scholar] [CrossRef]
- Lim, H.-C.; Ha, K.-I.; Hong, J.-Y.; Han, J.-Y.; Shin, S.-I.; Shin, S.-Y.; Herr, Y.; Chung, J.-H. Comparison of the Bone Harvesting Capacity of an Intraoral Bone Harvesting Device and Three Different Implant Drills. BioMed Res. Int. 2017, 2017, 1–6. [Google Scholar] [CrossRef]
- Kon, K.; Shiota, M.; Ozeki, M.; Kasugai, S. The effect of graft bone particle size on bone augmentation in a rabbit cranial vertical augmentation model: a microcomputed tomography study. Int. J. Oral Maxillofac. Implant. 2014, 29, 402–406. [Google Scholar] [CrossRef]
- Pradel, W.; Tenbieg, P.; Lauer, G. Influence of harvesting technique and donor site location on in vitro growth of osteoblastlike cells from facial bone. Int. J. Oral Maxillofac. Implants 2005, 20, 860–866. [Google Scholar]
- Miron, R.J.; Hedbom, E.; Saulacic, N.; Zhang, Y.; Sculean, A.; Bosshardt, D.D.; Buser, D. Osteogenic Potential of Autogenous Bone Grafts Harvested with Four Different Surgical Techniques. J. Dent. Res. 2011, 90, 1428–1433. [Google Scholar] [CrossRef] [PubMed]
- Teicher, U.; Ben Achour, A.; Nestler, A.; Brosius, A.; Lauer, G. Process based analysis of manually controlled drilling processes for bone. AIP Conf. Proc. 2018, 1960, 070025. [Google Scholar]
- Giesen, E.; Ding, M.; Dalstra, M.; Van Eijden, T. Mechanical properties of cancellous bone in the human mandibular condyle are anisotropic. J. Biomech. 2001, 34, 799–803. [Google Scholar] [CrossRef]
- Park, S.-Y.; Shin, S.-Y.; Yang, S.-M.; Kye, S.-B. Effect of implant drill design on the particle size of the bone collected during osteotomy. Int. J. Oral Maxillofac. Surg. 2010, 39, 1007–1011. [Google Scholar] [CrossRef] [PubMed]
- Kon, K.; Shiota, M.; Ozeki, M.; Yamashita, Y.; Kasugai, S. Bone augmentation ability of autogenous bone graft particles with different sizes: a histological and micro-computed tomography study. Clin. Oral Implant. Res. 2009, 20, 1240–1246. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.C.; Kim, Y.-T.; Kim, B.-S.; Kim, C.-S.; Park, J.-C.; Choi, S.-H.; Cho, K.-S.; Im, G.-I.; Park, J.; Choi, S.; et al. Acquisition of human alveolar bone-derived stromal cells using minimally irrigated implant osteotomy: in vitro and in vivo evaluations. J. Clin. Periodontol. 2012, 39, 495–505. [Google Scholar]
- Pallesen, L.; Schou, S.; Aaboe, M.; Hjørting-Hansen, E.; Nattestad, A.; Melsen, F. Influence of particle size of autogenous bone grafts on the early stages of bone regeneration: a histologic and stereologic study in rabbit calvarium. Int. J. Oral Maxillofac. Implant. 2002, 17, 498–506. [Google Scholar]
- Springer, I.N.; Terheyden, H.; Geiß, S.; Härle, F.; Hedderich, J.; Açil, Y. Particulated bone grafts - effectiveness of bone cell supply. Clin. Oral Implant. Res. 2004, 15, 205–212. [Google Scholar] [CrossRef]
- Sui, J.; Sugita, N. Experimental Study of Thrust Force and Torque for Drilling Cortical Bone. Ann. Biomed. Eng. 2019, 47, 802–812. [Google Scholar] [CrossRef]
- Ben Achour, A.; Meißner, H.; Teicher, U.; Haim, D.; Range, U.; Brosius, A.; Leonhardt, H.; Lauer, G. Biomechanical Evaluation of Mandibular Condyle Fracture Osteosynthesis Using the Rhombic Three-Dimensional Condylar Fracture Plate. J. Oral Maxillofac. Surg. 2019, 77, 1868.e1–1868.e15. [Google Scholar] [CrossRef]


| Attribute | Device 1 | Device 2 |
|---|---|---|
| Manufacturer | Mitutoyo GmbH (Japan) | Schneider Messtechnik GmbH (Bad Kreuznach, Germany) |
| Model | PJ 300 | ST 300 |
| Resolution | 1 µm | 0.5 µm |
| Magnification | 10×, 20×, 50×, 100× | 10×, 20×, 50×, 100× |
| Set | Drill Shape | Tool Diameter in mm | Number of Teeth z | Point Angle σ in ° | Helix Angle λs in ° | Flute Length in mm | Shape of the Drill Tip |
| Set A | twist | 2.2 | 2 | 122 | 25 | 14 |  |
| twist | 2.8 | 3 | 122 | 25 | 14 |  | |
| twist | 3.5 | 3 | 122 | 25 | 14 |  | |
| twist | 4.2 | 3 | 130 | 25 | 14 |  | |
| Set | Drill Shape | Tool Diameter in mm | Number of Teeth z | Point Angle σ in ° | Helix Angle λs in ° | Flute Length in mm | Shape of the Drill Tip |
| Set B | twist | 2 | 2 | 90 | 20 | 16 |  |
| tapered | 3.5 | 4 | 120 | 0 (straight) | 10 |  | |
| tapered | 4.3 | 4 | 120 | 0 (straight) | 11 |  | |
| Set | Drill Shape | Tool Diameter in mm | Number of Teeth z | Point Angle σ in ° | Helix Angle λs in ° | Flute Length in mm | Shape of the Drill Tip |
| Set C | twist | 2.0 | 2 | 118 | 15 | 14 |  |
| conical twist | 2.7 (3.0) | 2 | 125, 40 (segmented) | 15 | 14 |  | |
| conical twist | 3.0 (3.4) | 2 | 125, 40 (segmented) | 15 | 14 |  | |
| conical twist | 3.4 (3.8) | 2 | 125, 40 (segmented) | 15 | 14 |  | |
| Set | Drill Shape | Tool Diameter in mm | Number of Teeth z | Point Angle σ in ° | Helix Angle λs in ° | Flute Length in mm | Shape of the Drill Tip |
| Set D | conical tapered | 1.7–2.8 | 3 | 115, 9 | 0 (straight) | 14 |  |
| twist | 2.0 | 2 | 118 | 20 | 14 |  | |
| tapered | 3.3 | 4 | 140, 15 | 0 (straight) | 11 |  | |
| tapered | 3.8 | 4 | 140, 6 | 0 (straight) | 16 |  | |
| tapered | 4.3 | 4 | 140, 6 | 0 (straight) | 16 |  | |
| Set | Drill Shape | Tool Diameter in mm | Number of Teeth z | Point Angle σ in ° | Helix Angle λs in ° | Flute Length in mm | Shape of the Drill Tip |
| Set E | twist | 2 | 2 | 103 | 25 | 15 |  |
| stepped, twist | 2.8 | 3 | 118 | 25 | 15 |  | |
| twist | 3.25 | 3 | 140, 90 (segmented) | 20 | 16 |  | |
| twist | 4.25 | 3 | 140, 90 (segmented) | 20 | 16 |  |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ben Achour, A.; Petto, C.; Meißner, H.; Hipp, D.; Nestler, A.; Lauer, G.; Teicher, U. The Influence of Thrust Force on the Vitality of Bone Chips Harvested for Autologous Augmentation during Dental Implantation. Materials 2019, 12, 3695. https://doi.org/10.3390/ma12223695
Ben Achour A, Petto C, Meißner H, Hipp D, Nestler A, Lauer G, Teicher U. The Influence of Thrust Force on the Vitality of Bone Chips Harvested for Autologous Augmentation during Dental Implantation. Materials. 2019; 12(22):3695. https://doi.org/10.3390/ma12223695
Chicago/Turabian StyleBen Achour, Anas, Carola Petto, Heike Meißner, Dominik Hipp, Andreas Nestler, Günter Lauer, and Uwe Teicher. 2019. "The Influence of Thrust Force on the Vitality of Bone Chips Harvested for Autologous Augmentation during Dental Implantation" Materials 12, no. 22: 3695. https://doi.org/10.3390/ma12223695
APA StyleBen Achour, A., Petto, C., Meißner, H., Hipp, D., Nestler, A., Lauer, G., & Teicher, U. (2019). The Influence of Thrust Force on the Vitality of Bone Chips Harvested for Autologous Augmentation during Dental Implantation. Materials, 12(22), 3695. https://doi.org/10.3390/ma12223695





