Investigation of Copper Alloying in a TNTZ-Cux Alloy
Abstract
1. Introduction
2. Materials and Methods
2.1. Computational Modelling of Alloys
2.2. Production of Alloys
2.3. Calorimetric Measurements of Phase Transformations
2.4. X-ray Diffraction
2.5. Microstructural Studies
2.6. Hardness Studies
3. Results
3.1. Phase Calculations
3.2. XRD and Microstructure
3.3. Chemical and Crystal Phase Analysis
3.4. Hardness
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mouritz, A. Titanium alloys for aerospace structures and engines. In Introduction to Aerospace Materials; Woodhead Publishing: Cambridge, UK, 2012; pp. 202–223. [Google Scholar] [CrossRef]
- Rehder, D. Vanadium. Its role for humans. Met. Ions Life Sci. 2013, 13, 139–169. [Google Scholar] [CrossRef] [PubMed]
- Klotz, K.; Weistenhöfer, W.; Neff, F.; Hartwig, A.; van Thriel, C.; Drexler, H. The Health Effects of Aluminum Exposure. Dtsch. Arztebl. Int. 2017, 114, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Gutierrez-Urrutia, I.; Emura, S.; Liu, T.; Hara, T.; Min, X.; Ping, D. Tsuchiya, Twinning behavior of orthorhombic-α” martensite in a Ti-7.5Mo alloy. Sci. Technol. Adv. Mater. 2019, 20, 401–411. [Google Scholar] [CrossRef]
- Sandu, A.V.; Baltatu, M.S.; Nabialek, M.; Savin, A.; Vizureanu, P. Characterization and Mechanical Proprieties of New TiMo Alloys Used for Medical Applications. Materials 2019, 12, 2973. [Google Scholar] [CrossRef] [PubMed]
- Ho, W.F.; Ju, C.P.; Lin, J.H.C. Structure and properties of cast binary Ti–Mo alloys. Biomaterials 1999, 20, 2115–2122. [Google Scholar] [CrossRef]
- Bai, W.; Xu, G.; Tan, M.; Yang, Z.; Zeng, L.; Wu, D.; Liu, L.; Zhang, L. Diffusivities and Atomic Mobilities in bcc Ti-Mo-Zr Alloys. Materials 2018, 11, 1909. [Google Scholar] [CrossRef] [PubMed]
- Ferrandini, P.L.; Cardoso, F.F.; Souza, S.A.; Afonso, C.R.; Caram, R. Aging response of the Ti–35Nb–7Zr–5Ta and Ti–35Nb–7Ta alloys. J. Alloy. Compd. 2007, 433, 207–210. [Google Scholar] [CrossRef]
- Afonso, C.R.M.; Ferrandini, P.L.; Ramirez, A.J.; Caram, R. High resolution transmission electron microscopy study of the hardening mechanism through phase separation in a β-Ti–35Nb–7Zr–5Ta alloy for implant applications. Acta Biomater. 2010, 6, 1625–1629. [Google Scholar] [CrossRef]
- Elmay, W.; Laheurte, P.; Eberhardt, A.; Bolle, B.; Gloriant, T.; Patoor, E.; Prima, F.; Laille, D.; Castany, P.; Wary, M. Stability and elastic properties of Ti-alloys for biomedical application designed with electronic parameters. EPJ Web Conf. 2010, 6, 29002. [Google Scholar] [CrossRef]
- Stenlund, P.; Omar, O.; Brohede, U.; Norgren, S.; Norlindh, B.; Johansson, A.; Lausmaa, J.; Thomsen, P.; Palmquist, A. Bone response to a novel Ti–Ta–Nb–Zr alloy. Acta Biomater. 2015, 20, 165–175. [Google Scholar] [CrossRef]
- Niinomi, M.; Liu, Y.; Nakai, M.; Liu, H.; Li, H. Biomedical titanium alloys with Young’s moduli close to that of cortical bone. Regen. Biomater. 2016, 3, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Heidenau, F.; Mittelmeier, W.; Detsch, R.; Haenle, M.; Stenzel, F.; Ziegler, G.; Gollwitzer, H. A novel antibacterial titania coating: Metal ion toxicity and in vitro surface colonization. J. Mater. Sci. Mater. Med. 2005, 16, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Arciola, C.R.; Campoccia, D.; Speziale, P.; Montanaro, L.; Costerton, J.W. Biofilm formation in Staphylococcus implant infections. A review of molecular mechanisms and implications for biofilm-resistant materials. Biomaterials 2012, 33, 5967–5982. [Google Scholar] [CrossRef] [PubMed]
- Klevens, R.M.; Morrison, M.A.; Nadle, J.; Petit, S.; Gershman, K.; Ray, S.; Harrison, L.H.; Lynfield, R.; Dumyati, G.; Townes, J.M.; et al. Active Bacterial Core surveillance (ABCs) MRSA Investigators, Invasive methicillin-resistant Staphylococcus aureus infections in the United States. JAMA 2007, 298, 1763–1771. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, F.; Liu, C.; Wang, H.; Ren, B.; Yang, K.; Zhang, E. Effect of Cu content on the antibacterial activity of titanium–copper sintered alloys. Mater. Sci. Eng. C 2014, 35, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Ke, Z.; Yi, C.; Zhang, L.; He, Z.; Tan, J.; Jiang, Y. Characterization of a new Ti-13Nb-13Zr-10Cu alloy with enhanced antibacterial activity for biomedical applications. Mater. Lett. 2019, 253, 335–338. [Google Scholar] [CrossRef]
- Donthula, H.; Vishwanadh, B.; Alam, T.; Borkar, T.; Contieri, R.J.; Caram, R.; Banerjee, R.; Tewari, R.; Dey, G.K.; Banerjee, S. Morphological evolution of transformation products and eutectoid transformation(s) in a hyper-eutectoid Ti-12 at% Cu alloy. Acta Mater. 2019, 168, 63–75. [Google Scholar] [CrossRef]
- Contieri, R.J.; Lopes, E.S.N.; Caram, R.; Devaraj, A.; Nag, S.; Banerjee, R. Effects of cooling rate on the microstructure and solute partitioning in near eutectoid Ti–Cu alloys. Philos. Mag. 2014, 94, 2350–2371. [Google Scholar] [CrossRef]
- Fowler, L.; Janson, O.; Engqvist, H.; Norgren, S.; Öhman-Mägi, C. Antibacterial investigation of titanium-copper alloys using luminescent Staphylococcus epidermidis in a direct contact test. Mater. Sci. Eng. C 2019, 97, 707–714. [Google Scholar] [CrossRef]
- Banerjee, R.; Nag, S.; Stechschulte, J.; Fraser, H.L. Strengthening mechanisms in Ti–Nb–Zr–Ta and Ti–Mo–Zr–Fe orthopaedic alloys. Biomaterials 2004, 25, 3413–3419. [Google Scholar] [CrossRef]
- Lukas, H.L.; Fries, S.G.; Sundman, B. Computational Thermodynamics—The Calphad Method. 2007. Available online: https://app.knovel.com/hotlink/toc/id:kpCTTCM005/computational-thermodynamics/computational-thermodynamics (accessed on 28 July 2019).
- Kumar, K.C.H.; Ansara, I.; Wollants, P.; Delaey, L. Thermodynamic optimisation of the Cu-Ti system. Zeitschrift Für Metallkunde 1996, 87, 666–672. [Google Scholar]
- Canale, P.; Servant, C. Thermodynamic Assessment of the Cu–Ti System Taking into Account the New Stable Phase CuTi3. MEKU 2002, 93, 273–276. [Google Scholar] [CrossRef]
- Zhang, E.; Ren, J.; Li, S.; Yang, L.; Qin, G. Optimization of mechanical properties, biocorrosion properties and antibacterial properties of as-cast Ti–Cu alloys. Biomed. Mater. 2016, 11, 065001. [Google Scholar] [CrossRef] [PubMed]
- Porter, D.A.; Easterling, K.E. Thermodynamics and Phase Diagrams. Phase Transformations in Metals and Alloys; Van Nostrand Reinhold Co. Ltd.: Berkshire, UK, 1981; pp. 1–57. [Google Scholar]
- Voort, G.V. Materials Characterization & Testing: Microstructure of Titanium and its Alloys. Ind. Heat. 2006, 73, 77–80, 82–84. [Google Scholar]
- PDF-4+ 2019, ICDD. (n.d.). Available online: http://www.icdd.com/index.php/pdf-4/ (accessed on 10 May 2019).
- Spreadborough, J.; Christian, J.W. The Measurement of the Lattice Expansions and Debye Temperatures of Titanium and Silver by X-ray Methods. Proc. Phys. Soc. 1959, 74, 609–615. [Google Scholar] [CrossRef]
- Mueller, M.H.; Knott, H.W. The crystal structures of Ti2Cu, Ti2Ni, Ti4Ni2O, and Ti4Cu2O. Trans. Metall. Soc. AIME 1963, 227, 674–678. [Google Scholar]
- Bingert, J.; Mason, T.; Kaschner, G.; Maudlin, P.; Gray, G. Anisotropic plasticity modeling incorporating EBSD. In Electron Backscatter Diffraction in Materials Sciences; Plenun Publishing: New York, NY, USA, 2000; pp. 213–229. [Google Scholar]
- Porter, D.A.; Easterling, K.E. Particle coarsening. In Phase Transformations in Metals and Alloys; Van Nostrand Reinhold Co. Ltd.: Berkshire, UK, 1981; pp. 314–315. [Google Scholar]
- Devaraj, A.; Nag, S.; Muddle, B.C.; Banerjee, R. Competing Martensitic, Bainitic, and Pearlitic Transformations in a Hypoeutectoid Ti-5Cu Alloy. Metall. Mat. Trans. A 2011, 42, 1139–1143. [Google Scholar] [CrossRef]
- Dobromyslov, A.V.; Dolgikh, G.V.; Dutkevich, Y.; Trenogina, T.L. Phase and structural transformations in Ti-Ta alloys. Phys. Met. Metallogr. 2009, 107, 502–510. [Google Scholar] [CrossRef]
- Hennig, R.G.; Lenosky, T.J.; Trinkle, D.R.; Rudin, S.P.; Wilkins, J.W. Classical potential describes martensitic phase transformations between the α, β, and ω titanium phases. Phys. Rev. B 2008, 78, 054121. [Google Scholar] [CrossRef]
- Tobe, H.; Kim, H.Y.; Inamura, T.; Hosoda, H.; Miyazaki, S. Origin of {332} twinning in metastable β-Ti alloys. Acta Mater. 2014, 64, 345–355. [Google Scholar] [CrossRef]
- Xing, H.; Sun, J. Mechanical twinning and omega transition by 〈111〉 {112} shear in a metastable β titanium alloy. Appl. Phys. Lett. 2008, 93, 031908. [Google Scholar] [CrossRef]
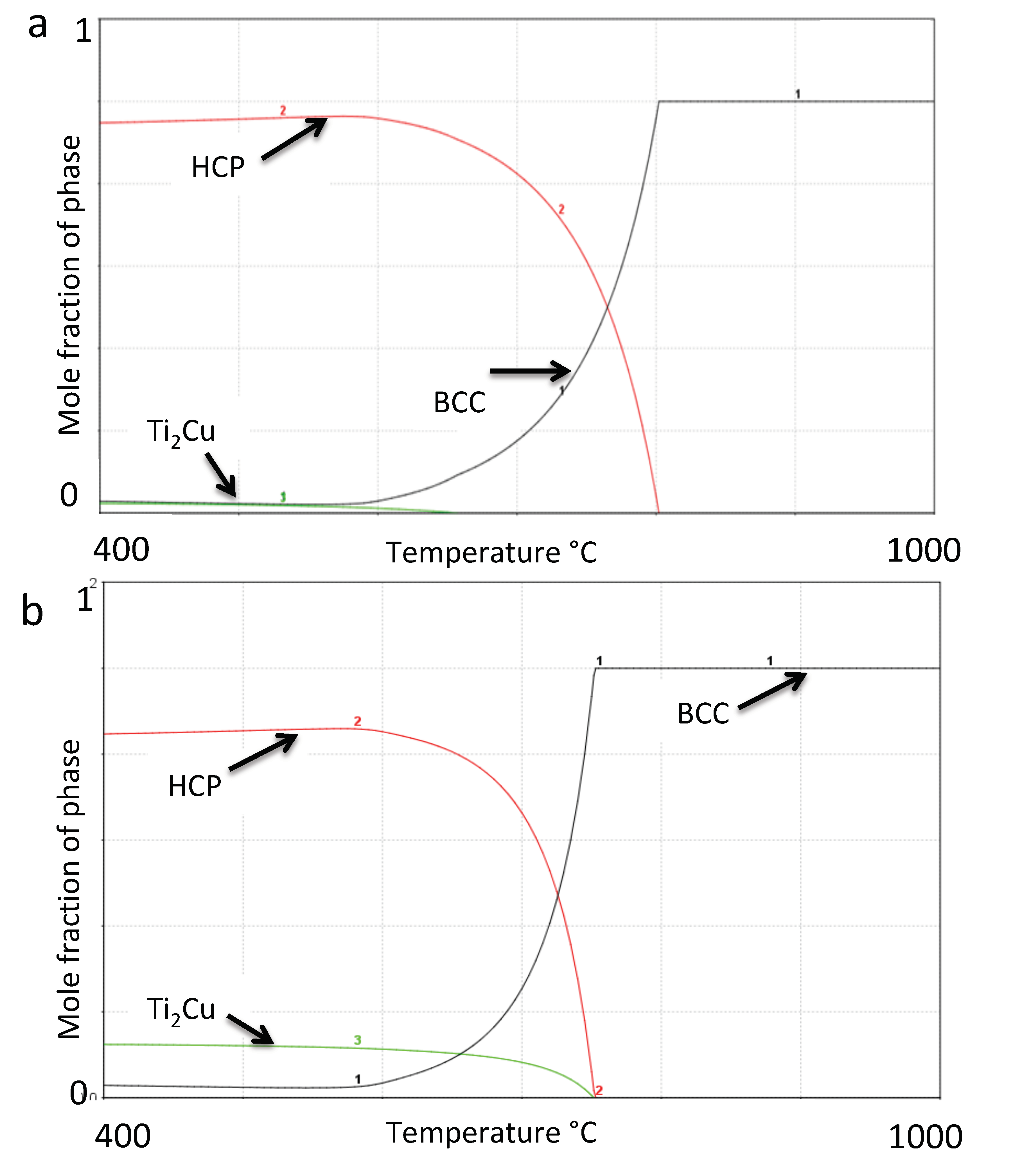
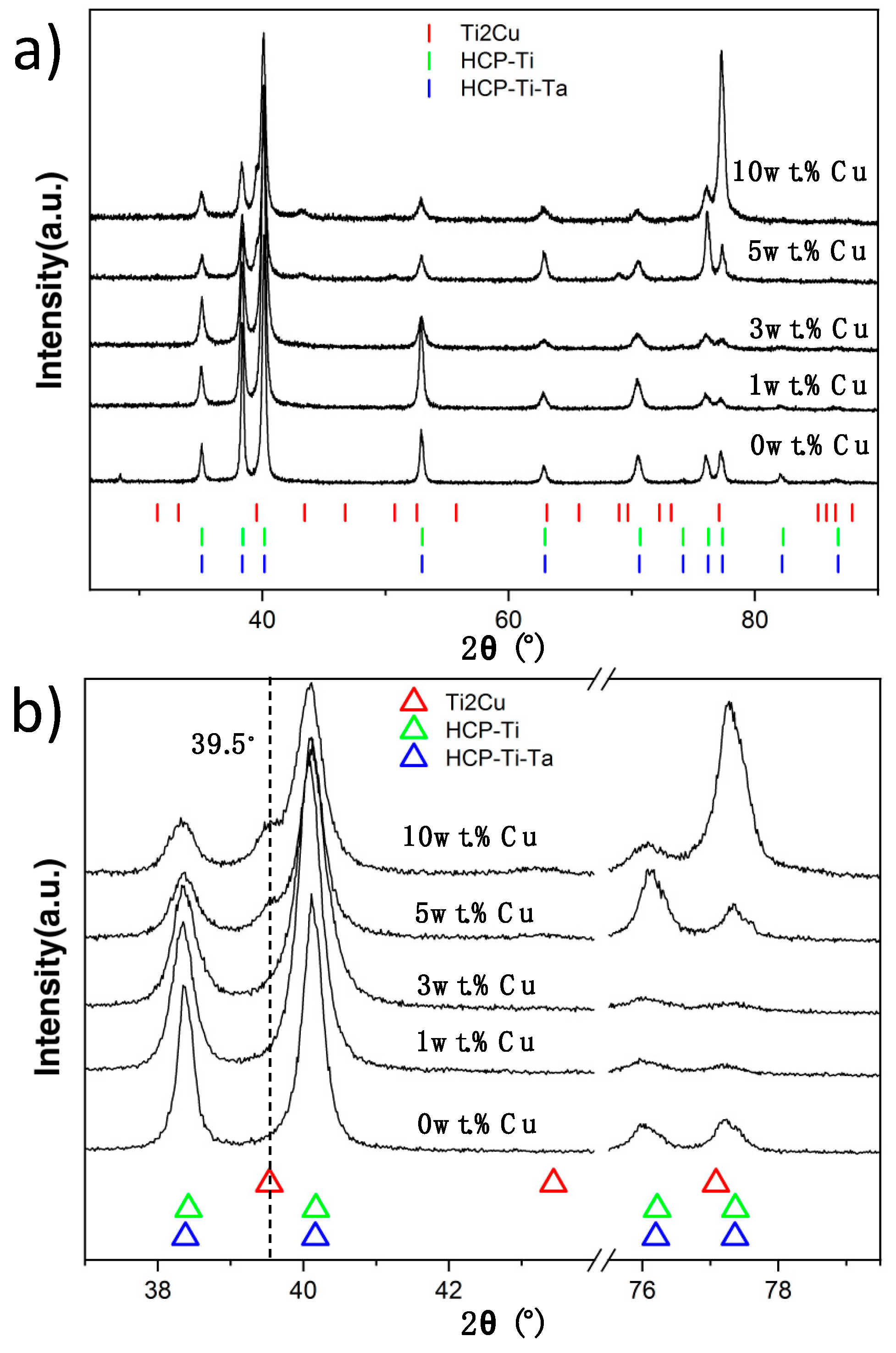


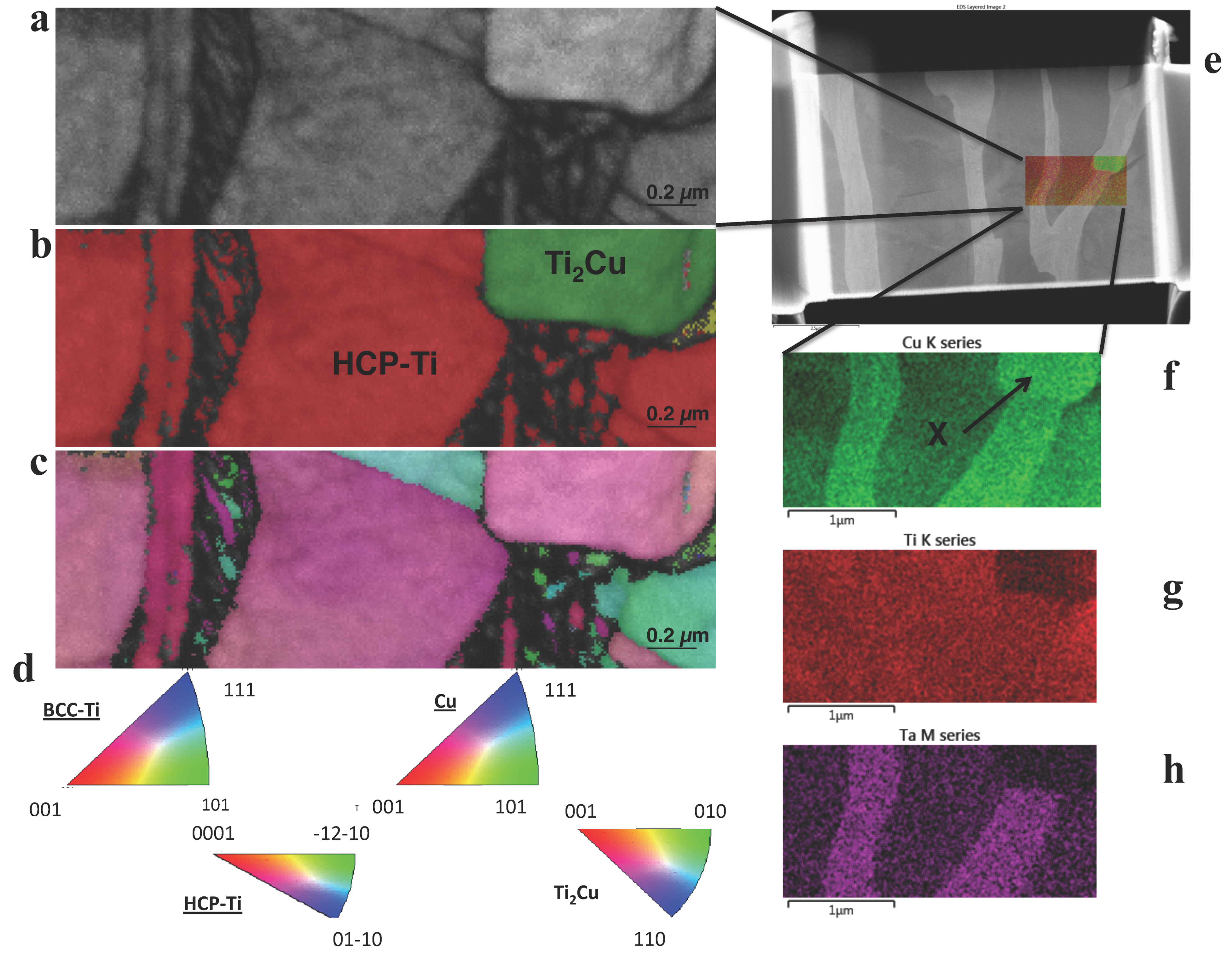

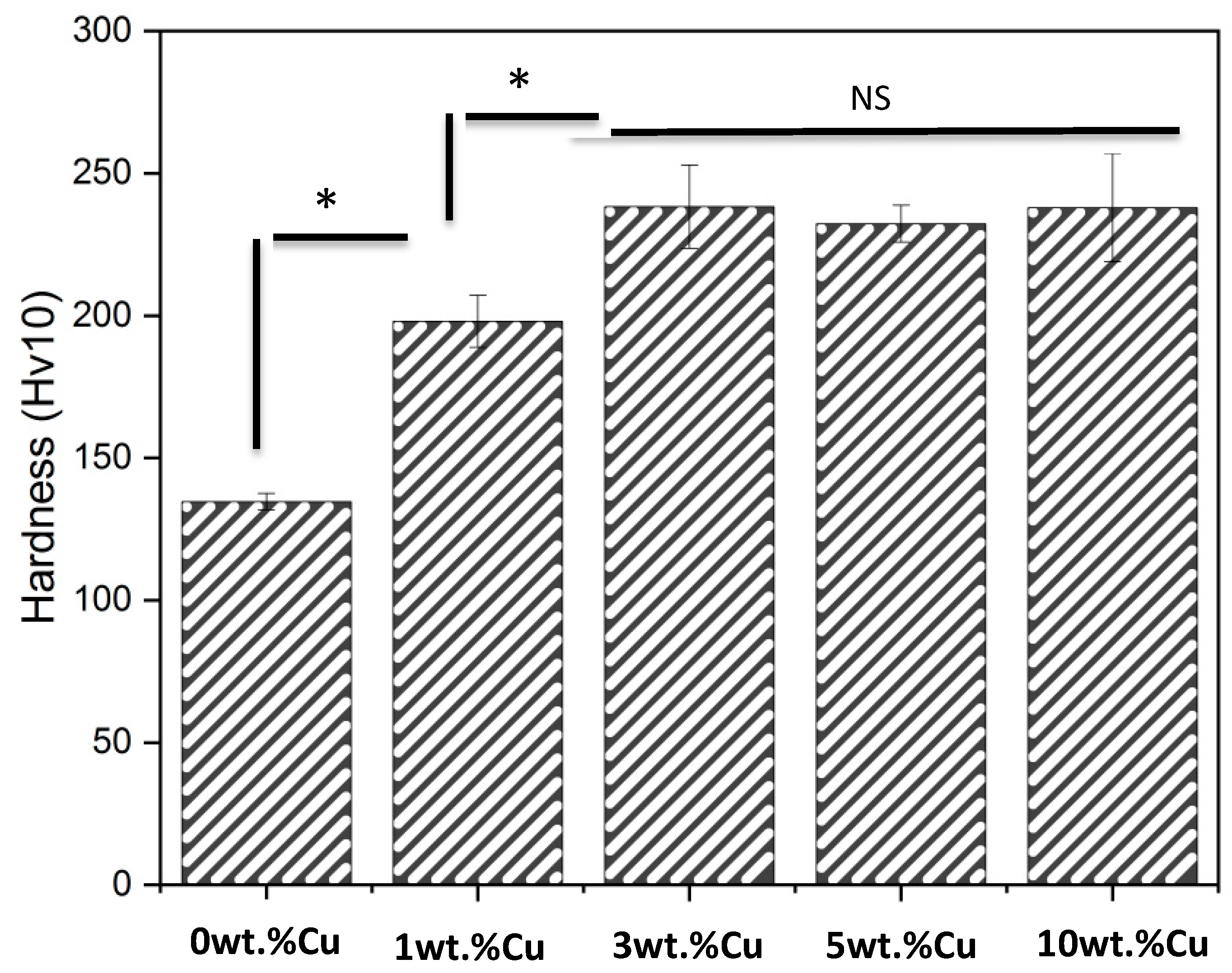
| Cu Addition [wt.%] | Calc. β-transus [Celsius] | Calc. Ti2Cu-transus [Celsius] |
|---|---|---|
| 0 | 814 | N/A |
| 1 | 802 | 655 |
| 3 | 777 | 728 |
| 5 | 753 | 752 |
| 10 | 746 | 859 |
| Designation | Cu | Ti | Nb | Zr | Ta |
|---|---|---|---|---|---|
| 0 Cu | 0 | 86.6 | 1.7 | 1.6 | 10.1 |
| 1 Cu | 1 | 85.74 | 1.68 | 1.58 | 10.0 |
| 3 Cu | 3 | 84 | 1.65 | 1.55 | 9.8 |
| 5 Cu | 5 | 82.26 | 1.62 | 1.52 | 9.6 |
| 10 Cu | 10 | 77.94 | 1.53 | 1.44 | 9.09 |
| Steps | 1–Grind | 2–Rough Polish | 3–Final Polish |
|---|---|---|---|
| Surface | SiC–120 P | MD–Dur cloth | MD–Floc cloth |
| Abrasive | - | 6 µm diamond suspension | OP-S Si-Colloids and H2O2 (5:1) solution |
| Lubricant | water | DP lubricant red | - |
| Speed (rpm) | 250 contra | 200 contra | 150 contra |
| Duration (min) | until planar | 30 min | 30 min |
| Calculated Phases [mol%] | |||
|---|---|---|---|
| Cu Addition [wt.%] | α | β | Ti2Cu |
| 0 | 76 | 24 | 0 |
| 1 | 37 | 63 | 0 |
| 3 | 35 | 65 | 0 |
| 5 | 14 | 85 | 2 |
| 10 | 0 | 85 | 15 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fowler, L.; Janse Van Vuuren, A.; Goosen, W.; Engqvist, H.; Öhman-Mägi, C.; Norgren, S. Investigation of Copper Alloying in a TNTZ-Cux Alloy. Materials 2019, 12, 3691. https://doi.org/10.3390/ma12223691
Fowler L, Janse Van Vuuren A, Goosen W, Engqvist H, Öhman-Mägi C, Norgren S. Investigation of Copper Alloying in a TNTZ-Cux Alloy. Materials. 2019; 12(22):3691. https://doi.org/10.3390/ma12223691
Chicago/Turabian StyleFowler, Lee, Arno Janse Van Vuuren, William Goosen, Håkan Engqvist, Caroline Öhman-Mägi, and Susanne Norgren. 2019. "Investigation of Copper Alloying in a TNTZ-Cux Alloy" Materials 12, no. 22: 3691. https://doi.org/10.3390/ma12223691
APA StyleFowler, L., Janse Van Vuuren, A., Goosen, W., Engqvist, H., Öhman-Mägi, C., & Norgren, S. (2019). Investigation of Copper Alloying in a TNTZ-Cux Alloy. Materials, 12(22), 3691. https://doi.org/10.3390/ma12223691





