1. Introduction
Stone is one of the most durable construction materials used in historic buildings. However, over time, the stone can be subject to various degradation processes leading to physical and chemical modifications [
1,
2,
3,
4,
5]. Although these effects may be limited to the surface and negligible to the structural stability of the affected buildings, they can represent a major problem in carved decorative elements of artistic value, where any detail should be preserved.
The challenge for conservators and material scientists involved in stone conservation has always been to find a way to stop or delay the effects of these degradation processes. Since the 19th century, craftsmen and restorers have been applying a variety of products to stone elements with the aim of regaining strength and cohesion. However, in many cases, these attempts relied on empirical evidence and were based on a hit-or-miss approach and on the use of locally available consolidants [
6]. A more scientifically-sound approach gradually followed over the last century, leading to the development of novel products specially tailored for stone consolidation. Although some good results have been obtained for indoor stone surfaces, the development of long-term solutions for stone exposed to outdoor weathering is still an open challenge, in particular on account of climatic changes effects [
7].
Deteriorated sedimentary stones typically show loss of intergranular cohesion due to the impoverishment of the natural interstitial binder. This usually affects an external stone layer and is the result of chemical processes or physical and thermal stresses that cause an abrupt variation of the chemical, physical and/or mechanical properties within the stone [
5,
7,
8]. To re-establish cohesion within the altered layer and continuity between this and the underlying sound material, a consolidation treatment is usually necessary. The products used in such interventions are liquids (solutions or colloidal suspensions) able to penetrate the stone pore network where a solid phase with binding properties is precipitated. Ideally, consolidants should be able to significantly improve some measurable properties of the stones (i.e., the abrasion resistance), have good adhesion with the substrate, be compatible with the substrate and allow future treatments [
6,
7,
8,
9,
10]. Currently, the most common products used for stone consolidation are lime-based, polymeric, and silicate-based consolidants [
6,
7].
Lime-based treatments, such as limewater and lime milk, have been traditionally used for the consolidation of calcareous substrates (e.g., limestone), and are considered highly compatible because of their chemical and structural affinity with the substrate, made up of calcium carbonate [
11]. However, it is generally acknowledged that they have very limited penetration and little consolidation effect [
6,
12,
13]. Furthermore, being water-based, their application may entail the mobilisation of salts already within the substrate that, upon re-crystallisation, can have detrimental effects on the treated stone [
13].
Polymeric consolidants, such as acrylic solutions (e.g., Paraloid
® B72) and emulsions (e.g., Primal
® AC 33) have been extensively used for stone consolidation [
14]. These products can strengthen the substrate when they solidify upon solvent evaporation and are appreciated for their good adhesive properties [
10]. However, many questions have been recently raised about their long-term stability, mostly affected by photo-oxidative degradation [
15,
16,
17,
18]. Furthermore, some of these products are reported to have low impregnation capacity and to form hard crusts, leading to severe detachment when used in highly porous stones and in combination with silicate-based consolidants [
6].
Alkoxysilanes, primarily methyltrimethoxysilane (MTMOS) and tetraethoxysilane (TEOS), have been the most widely used consolidants over the past twenty years. They impart strength to the stone by forming a silica gel inside the pore network upon polymerisation of the silanol units. Their popularity is due to several advantageous properties, such as good penetration, thermal and oxidative stability, long-term experience, as well as their commercial availability [
6,
9]. The main issue related to their use is the high sensitivity to environmental conditions: relative humidity and pH variations, and presence of soluble salts or clay minerals can affect the curing process, leading to surface whitening and micro-fissures formation [
6,
17]. Furthermore, silicate consolidants are often stated to hardly bond to calcareous substrates [
6,
19,
20] and are subject to shrinking upon cross-linking.
Over the last years, various innovative stone consolidants have been developed and tested with particular attention on their compatibility and environmental impact [
16].
The use of nanoparticles for the conservation of various artistic materials (e.g., frescoes and paper) has been promising [
16,
21,
22,
23]. Several advantages were identified for the use of calcium hydroxide nanoparticles over the traditional limewater treatment, such as a higher calcium concentration (up to 50 g/L, against 1.7 g/L of a saturated solution of Ca(OH)
2), and the use of an organic carrier allowing for less pronounced agglomeration, easier capillary absorption, quick evaporation and reduced diffusion of soluble salts [
11,
24]. Treatments based on alcoholic dispersions of calcium hydroxide nanoparticles have been successfully applied for the conservation of historical renders, porous limestone, marble and wall paintings [
25,
26,
27,
28,
29,
30].
A further innovative approach is the use of ammonium phosphates solutions, introduced by some recent pioneering studies for consolidation of carbonate stones and for some types of calcium-bearing silicate stones, with several advantages such as high compatibility with carbonate stones and the possibility to use concentrated solutions [
10,
31,
32]. Ammonium phosphates are a family of salts extensively used in several industrial fields [
33,
34]. Among them, di-ammonium phosphate (DAP, formula (NH
4)
2HPO
4) is the reagent most commonly used in stone consolidation. When in contact with a calcareous substrate, DAP reacts with the calcium ions in solution according to Reaction (1):
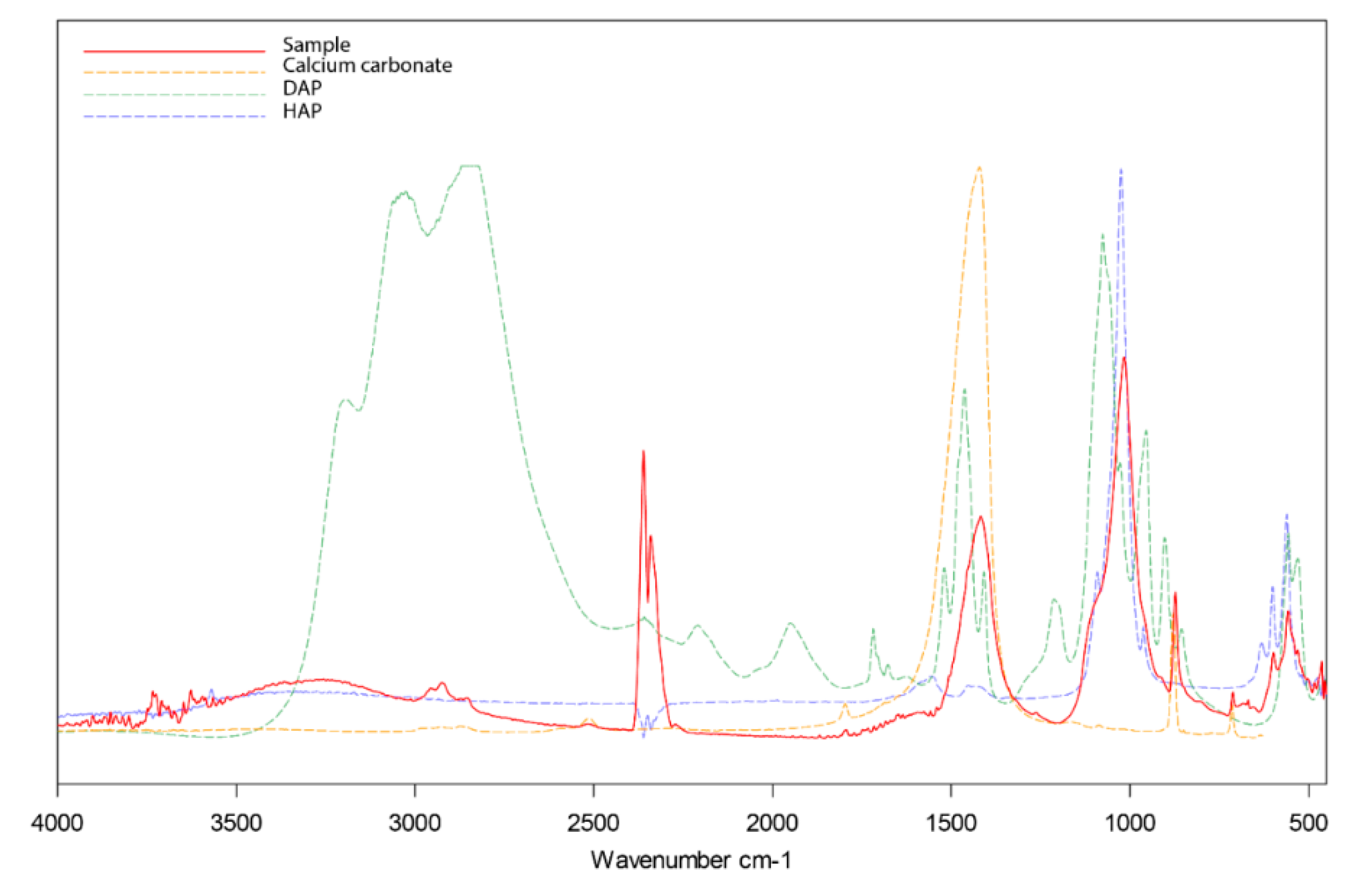
The reaction product is hydroxyapatite (HAP), a mineral widely studied in biology and medicine [
35]. HAP shows advantageous properties as a consolidant, such as its high stability and mineralogical compatibility with calcite [
10,
36]. Alongside HAP, other minerals form as a result of Reaction (1), i.e., metastable Ca-P phases and precursors, as well as amorphous calcium phosphate, depending on the surrounding conditions. Although the relationship between the chemical-mineralogical nature of such Ca-P phases and their respective consolidation action is not fully understood yet, most of them are less soluble than calcite and, therefore, are likely to contribute to the overall consolidation effect [
32].
Some recent studies have been carried out to investigate the effectiveness and suitability of DAP treatments on weathered stones. Results thus far are promising and include a significant increase in dynamic elastic modulus and tensile strength, unaffected pore size distribution and moisture transfer properties, re-treatability of the consolidated surfaces, as well as full effectiveness after 48 h from the treatment [
10,
20,
31,
37,
38,
39].
This study aimed to evaluate the effectiveness and compatibility of the combinations of an aqueous solution of DAP and two Ca-based nanomaterials, namely an ethanol-based nanosuspension of Ca(OH)
2 and a novel aqueous nanosuspension of calcite. The advantages brought by the use of these products are manifold: (i) the low environmental impact of nanosuspensions and phosphate solutions with respect to other common formulations based on solvents harmful to the health and environment ([
16] and references therein]); (ii) calcium-based nanomaterials are aimed at boosting HAP formation and allow DAP application to calcium-poor substrates, as it had been previously tested using limewater poultice as additional calcium source [
37]; (iii) the reduced particle size should promote penetration and inter-molecular interaction with the substrate [
40]; and (iv) the high concentration allows for reducing the number of necessary applications, and therefore introducing a lessened amount of water into the stone, unlike limewater treatment.
4. Conclusions
The aim of this study was the evaluation of the effectiveness and compatibility of consolidation treatments based on various combinations of di-ammonium phosphate (DAP) solution and two calcium-based nanosuspensions, a nanolime (NL) and a nanocalcite (NC), applied to Schlaitdorfer sandstone and Auer limestone.
The treatments were able to improve the compactness of the stones they were applied to. However, the stone textural and chemical characteristics played a predominant role in the performance of the consolidants, as the substrate’s effect was greater than the effect of the treatments themselves. Overall, they produced a greater impact on the Schlaitdorfer sandstone than on the Auer limestone in terms of UPV and WAC. As regards physical compatibility, the treatments could be assessed with a low risk of incompatibility on the limestone, as water sorption had not dramatically reduced after application of the consolidants. On the other hand, a substantial reduction in water sorption was observed on the sandstone after the consolidants’ application, meaning a medium risk of incompatibility. Nanoconsolidants and DAP should thus be applied with caution on similar lithotypes, especially if high moisture is an issue of the treated object.
As regards the application of pure nanoconsolidants, their main issue concerned the limited penetration into the stone which reached a depth of about 0.5 mm. Such penetration can be evaluated in relation to the depth of the layer affected by the deterioration process, which typically varies from a few centimetres in porous stones to less than a millimetre in marbles. This suggests that nanomaterials could be successfully employed for the consolidation of fine-grained, compact stones affected by decohesion processes limited to the very surface. A further potential use could be the pre-consolidation of stones in an advanced deterioration state, requiring an immediate intervention to secure some initial surface cohesion. In such a case, the tested nanomaterials would be advantageous as they do not alter surface properties, allowing their retreatment. Moreover, in the case of NC, the consolidation effect is accomplished in short time after application (drying time, i.e., usually less than 48 h), whereas other common consolidants require several weeks to complete curing. The NC could also be assessed with low risk of aesthetical incompatibility on both stones, whereas the NL should be used with caution on the Schlaitdorf sandstone as slight yellowing might occur after treatment.
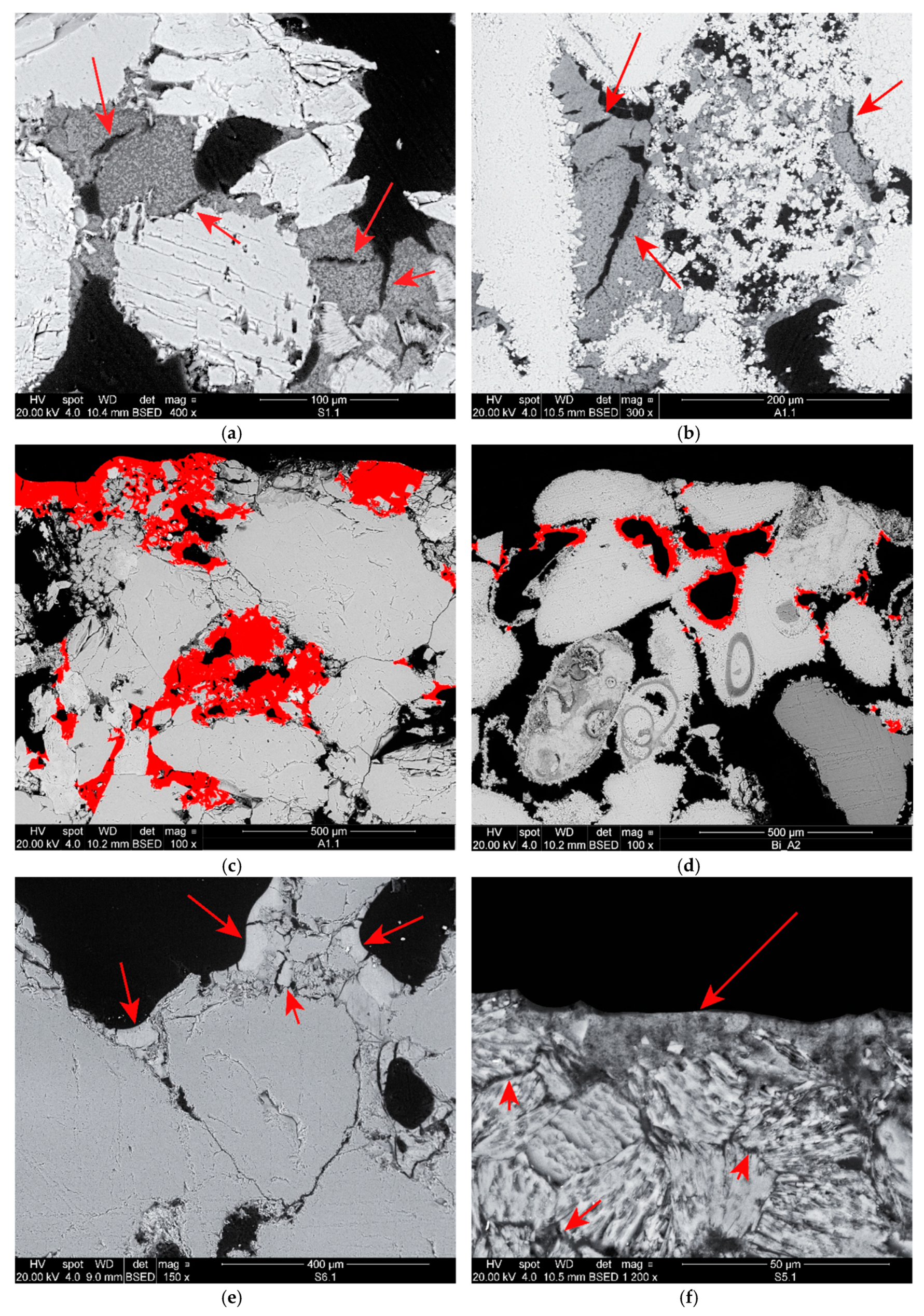
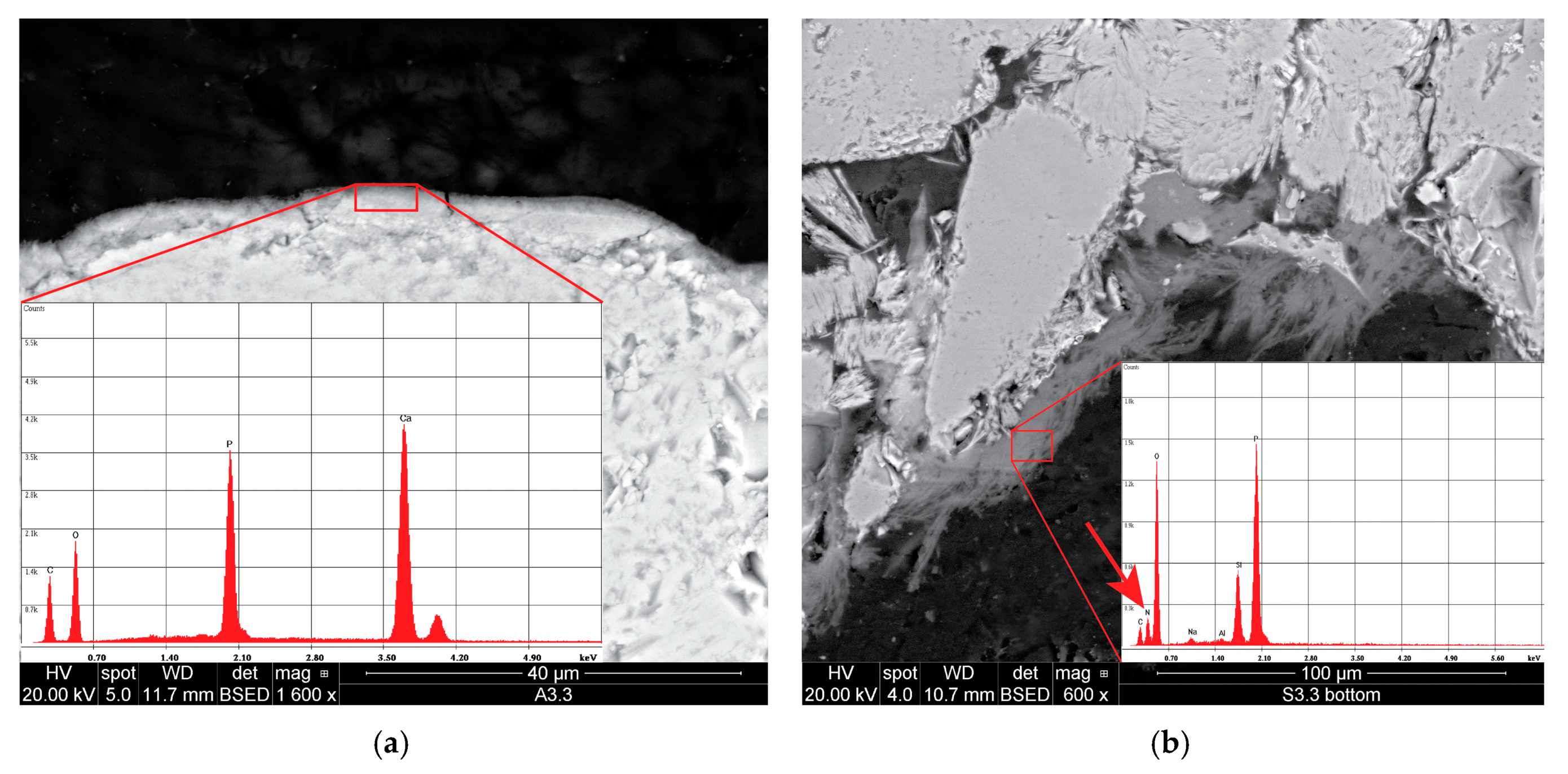
The application of pure DAP showed to be effective on the limestone, where most of the salt was able to react and convert into stable hydroxyapatite, well adhered to the grains’ surface. On the sandstone, residues of unconverted DAP were observed, hence a further source of Ca2+ ions is necessary, e.g., a subsequent application of a calcium-based consolidant. However, the presence of possible unreacted DAP residues and the detailed characterisation of the reaction products should be further clarified to avoid the introduction of detrimental soluble salts into the stone upon treatment.
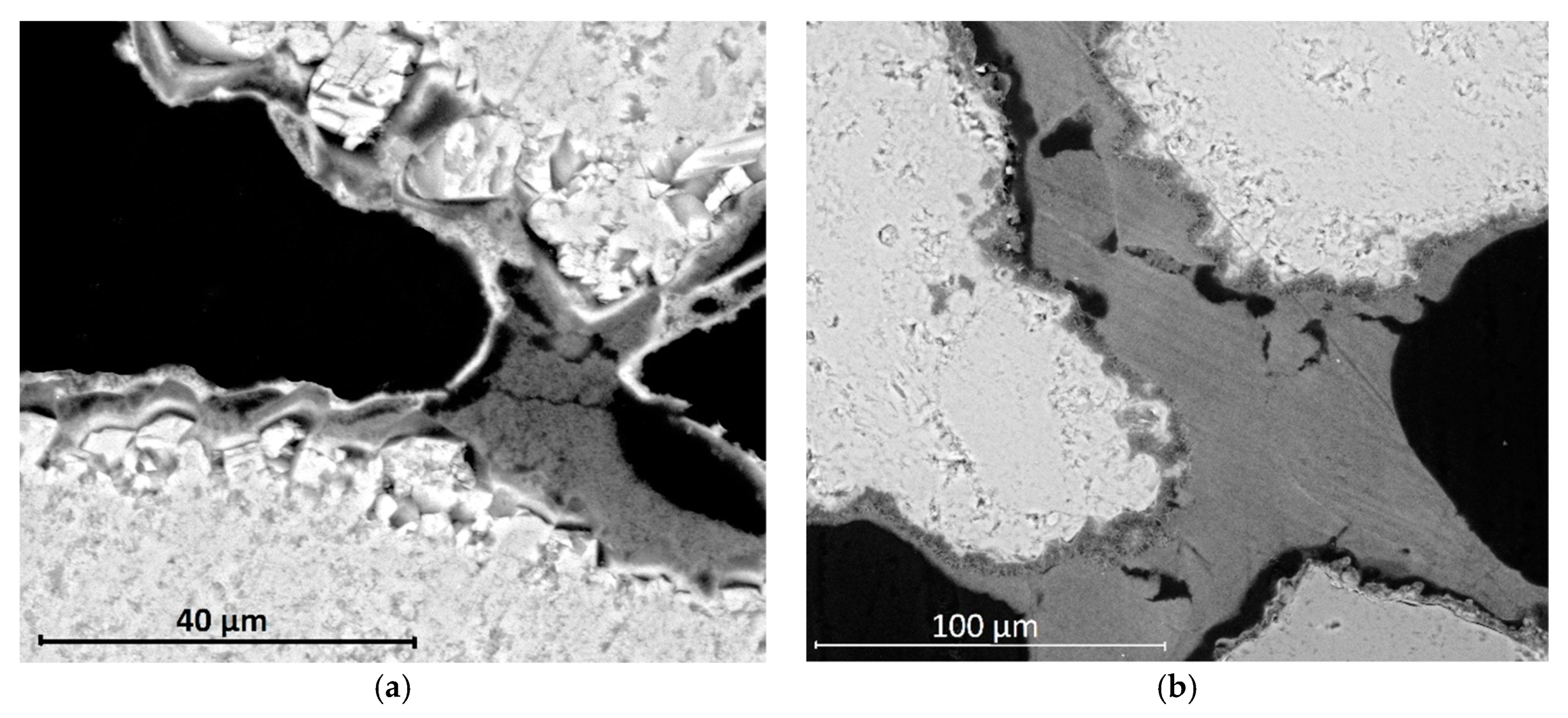
The combined application of the nanoconsolidants and DAP showed to produce two different morphologies, one produced by hydroxyapatite precipitation in the form of a thin, compact layer on the grains’ surface, and the other produced by the deposition of the nanoparticles in the form of toroidal bridges linking the grains. The combined treatments showed a greater effectiveness than those based on neat products, as a result of a more effective pore filling and a higher penetration depth of about 1 cm. Moreover, DAP is recommended to be applied first and then the nanoconsolidant, as a stronger adhesion to the substrate was obtained than through the reverse order of application.
A limitation of the combined treatments highlighted by this study was the high risk of aesthetical compatibility as a result of yellowing of the surface after applications. The treatments should thus only be applied to stones that naturally show high colour variability, where colour variations brought by the treatments become less significant. It should also be taken into account that effects of long-term exposure likely attenuate such risk and this is one of the reasons a monitoring schedule after any consolidation intervention is of uttermost importance.
Finally, although it should be stressed that field conditions are usually hardly controllable and that unexpected responses to treatments are likely to be found, preliminary evaluation is a crucial step in the selection of a consolidation treatment. In such context, laboratory-based trials carried out in controlled and reproducible conditions allow for an initial evaluation of the consolidants performance.