Effects of Cr, W, and Mo on the High Temperature Oxidation of Ni-Based Superalloys
Abstract
1. Introduction
2. Experimental Procedures
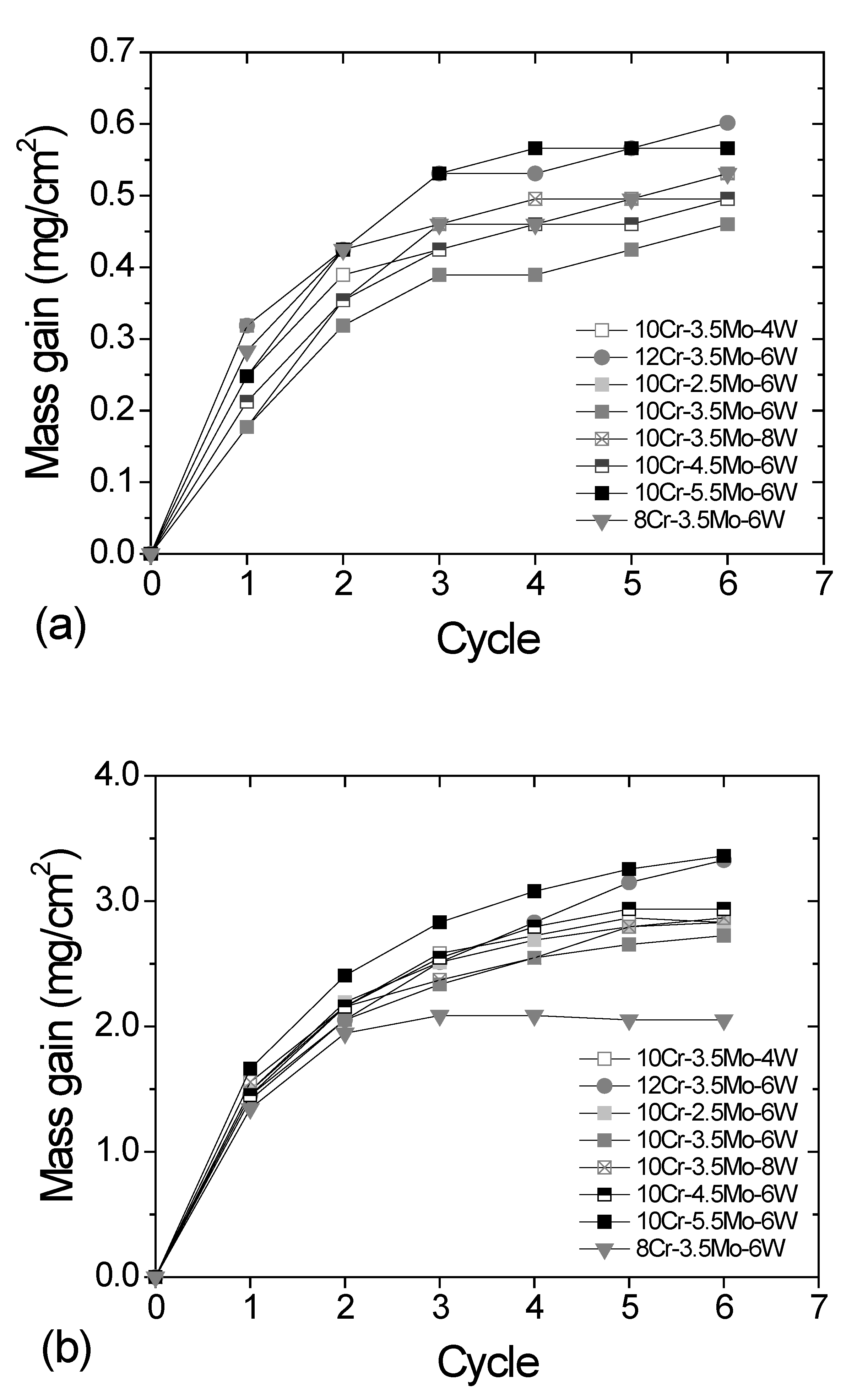
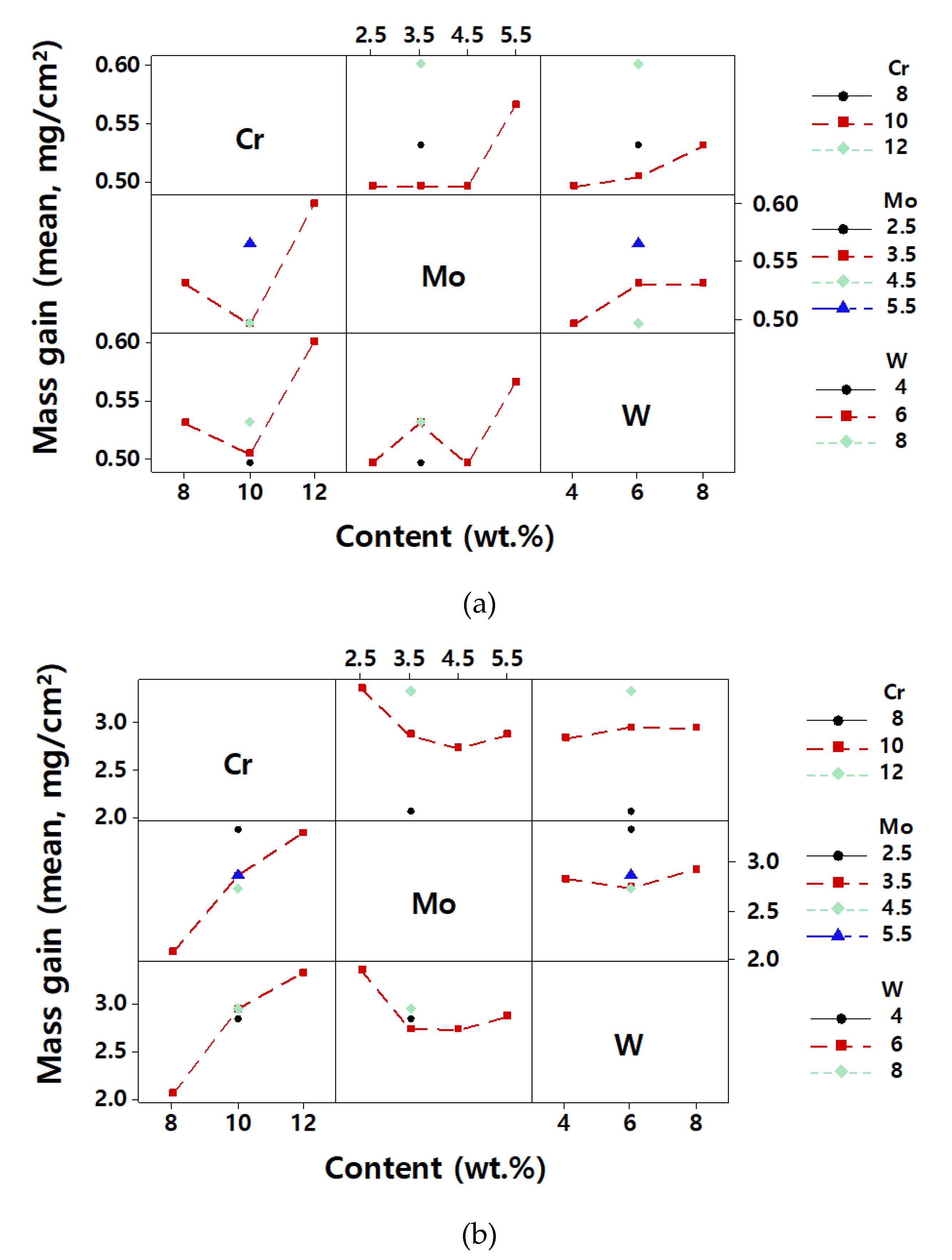
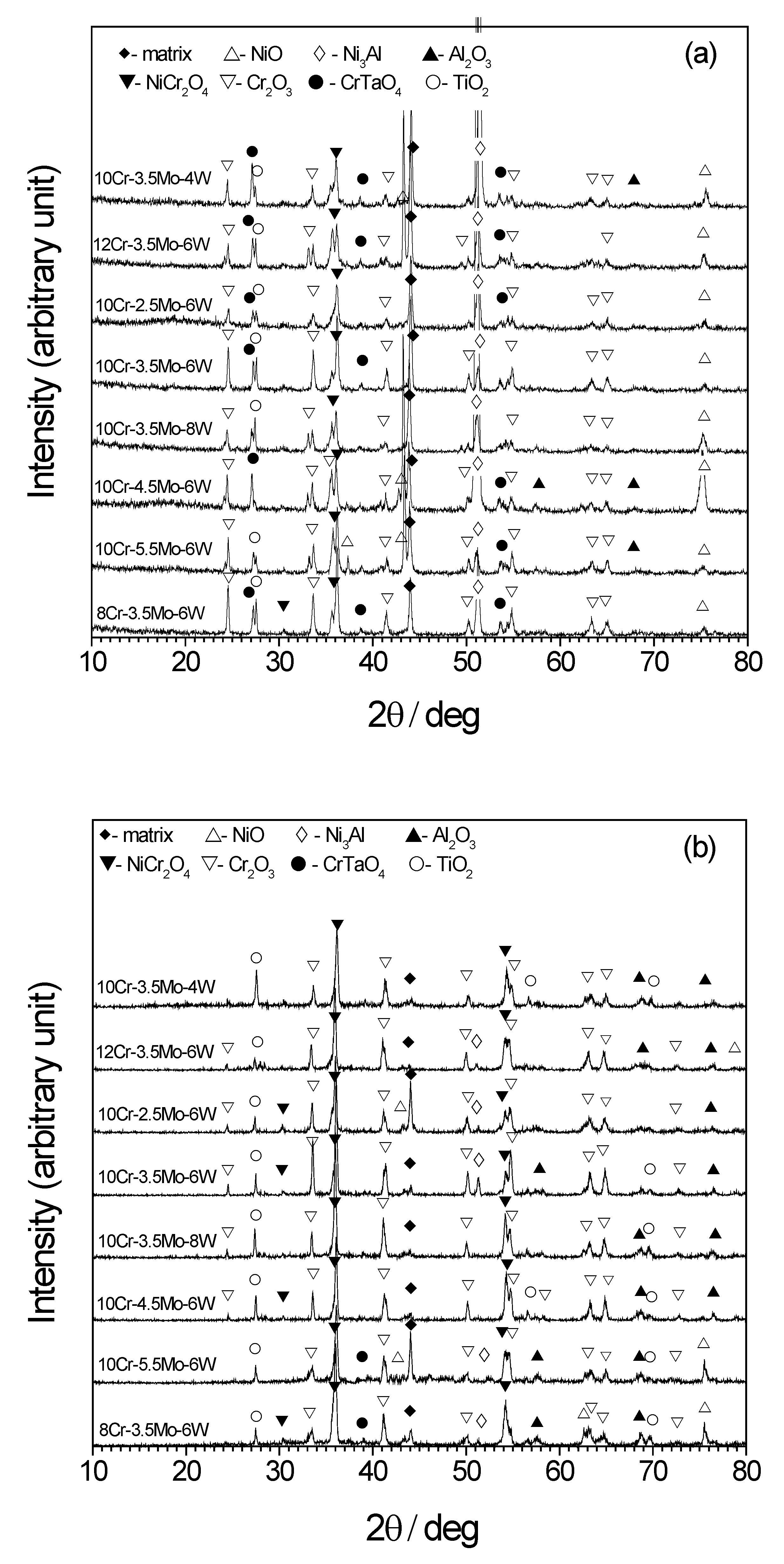
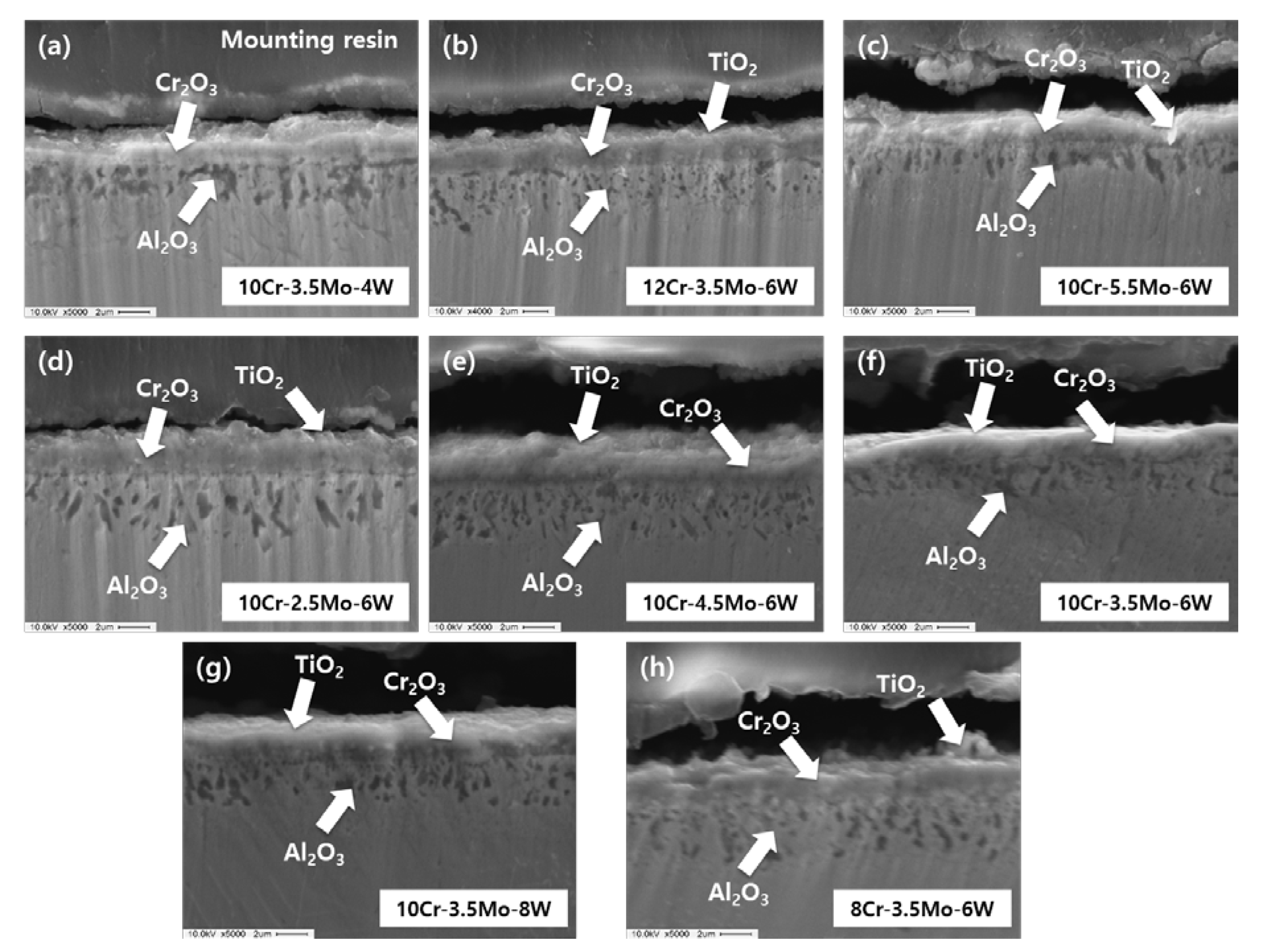
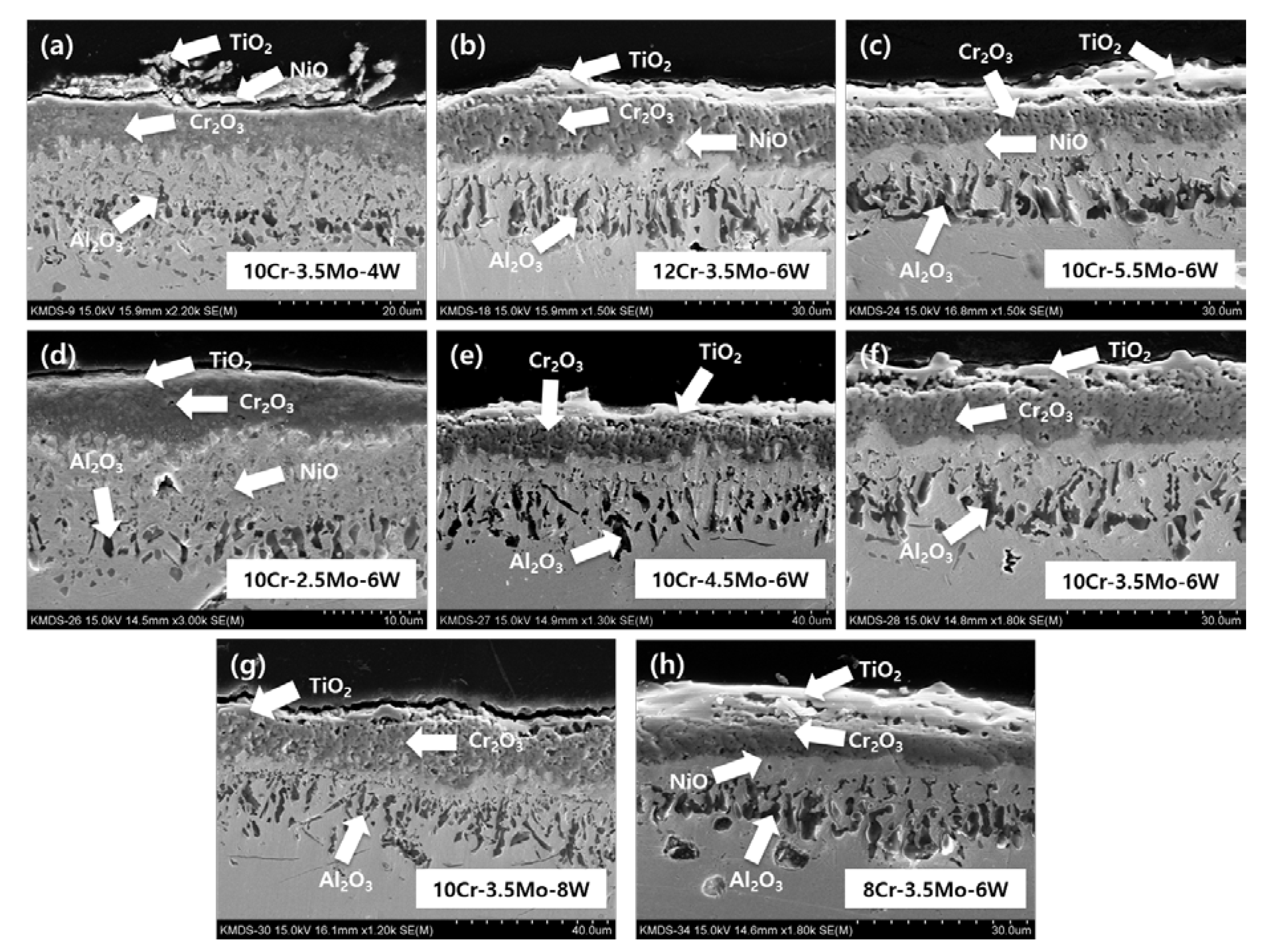
3. Results
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Donachie, M.J.; Donachie, S.J. Superalloys: A Technical Guide, 2nd ed.; ASM International: Almere, The Netherlands, 2002; Chapter 1. [Google Scholar]
- Guo, H.; Li, D.; Zheng, L.; Gong, S.; Xu, H. Effect of co-doping of two reactive elements on alumina scale growth of β-NiAl at 1200 °C. Corros. Sci. 2014, 88, 197–208. [Google Scholar] [CrossRef]
- Li, D.; Guo, H.; Wang, D.; Zhang, T.; Gong, S.; Xu, H. Cyclic oxidation of β-NiAl with various reactive element dopants at 1200 °C. Corros Sci. 2013, 66, 125–135. [Google Scholar] [CrossRef]
- Yan, K.; Guo, H.; Gong, S. High-temperature oxidation behavior of β-NiAl with various reactive element dopants in dry and humid atmospheres. Corros. Sci. 2014, 83, 335–342. [Google Scholar] [CrossRef]
- Dong, Z.; Peng, X.; Guan, Y.; Li, L.; Wang, F. Optimization of composition and structure of electrodeposited Ni–Cr composites or increasing the oxidation resistance. Corros. Sci. 2012, 62, 147–152. [Google Scholar] [CrossRef]
- Young, D.; Zurek, J.; Singheiser, L.; Quadakkers, W.; Young, D. Temperature dependence of oxide scale formation on high-Cr ferritic steels in Ar-H2-H2O. Corros. Sci. 2011, 53, 2131–2141. [Google Scholar] [CrossRef]
- Yang, Z.; Xia, G.-G.; Stevenson, J.W. Evaluation of Ni–Cr-base alloys for SOFC interconnect applications. J. Power Sources 2006, 160, 1104–1110. [Google Scholar] [CrossRef]
- Park, S.-J.; Seo, S.-M.; Yoo, Y.-S.; Jeong, H.-W.; Jang, H. Effects of Al and Ta on the high temperature oxidation of Ni-based superalloys. Corros. Sci. 2015, 90, 305–312. [Google Scholar] [CrossRef]
- Irving, G.; Stringer, J.; Whittle, D. The oxidation of Co-20% Cr base alloys containing Nb or Ta. Corros. Sci. 1975, 15, 337–344. [Google Scholar] [CrossRef]
- Guo, H.; Zhang, T.; Wang, S.; Gong, S. Effect of Dy on oxide scale adhesion of NiAl coatings at 1200°C. Corros. Sci. 2011, 53, 2228–2232. [Google Scholar] [CrossRef]
- Lu, X.; Tian, S.; Yu, X.; Wang, C. Oxidation behavior of a single-crystal Ni-base superalloy in air at 900 and 1050 °C. Rare Met. 2011, 30, 439–442. [Google Scholar] [CrossRef]
- Guo, H.; Wang, D.; Peng, H.; Gong, S.; Xu, H. Effect of Sm, Gd, Yb, Sc and Nd as reactive elements on oxidation behaviour of β-NiAl at 1200 °C. Corros. Sci. 2014, 78, 369–377. [Google Scholar] [CrossRef]
- Qin, L.; Pei, Y.; Li, S.; Zhao, X.; Gong, S.; Xu, H. Role of volatilization of molybdenum oxides during the cyclic oxidation of high-Mo containing Ni-based single crystal superalloys. Corros. Sci. 2017, 129, 192–204. [Google Scholar] [CrossRef]
- Goebel, J.A.; Pettit, F.S.; Goward, G.W. Mechanisms for the hot corrosion of nickel-base alloys. Met. Mater. Trans. A 1973, 4, 261–278. [Google Scholar] [CrossRef]
- Carrasco, J.G.; Adeva, P.; Aballe, M. The role of microstructure on oxidation of Ni-Cr-Al base alloys at 1023 and 1123 K in air. Oxid. Met. 1990, 33, 1–17. [Google Scholar] [CrossRef]
- Hashizume, R.; Yoshinari, A.; Murata, Y.; Morinaga, M. Development of Ni-based Single Crystal Superalloys for Power-generation Gas-turbine Blades. Tetsu-to-Hagane 2004, 90, 518–525. [Google Scholar] [CrossRef]
- Hobbs, R.; Brewster, G.; Rae, C.; Tin, S. Evaluation of Ruthenium-Bearing Single Crystal Superalloys: A Design of Experiments. Superalloys 2008, 171–180. [Google Scholar]
- Ul-Hamid, A.; Mohammed, A.I.; Al-Jaroudi, S.S.; Tawancy, H.M.; Abbas, N.M. Evolution of oxide scale on a Ni-Mo-Cr alloy at 900 °C. Mater. Character. 2007, 58, 13–23. [Google Scholar] [CrossRef]
- Huang, X.; Li, J.R.; Hu, J.; Bai, G.; Fu, H. Evolution of oxidation in Ni-Cr-W alloy at 1100 °C. Rare Metal. Mat. Eng. 2010, 39, 1908–1911. [Google Scholar]
- El-Dahshan, M.E.; Whittle, D.P.; Stringer, J. The oxidation of nickel-tungsten alloys. Corros. Sci. 1976, 16, 83–90. [Google Scholar] [CrossRef]
- Espevik, S.; Rapp, R.A.; Daniel, P.L.; Hirth, J.P. Oxidation of Ni-Cr-W ternary alloys. Oxid. Met. 1980, 14, 85–108. [Google Scholar] [CrossRef]
- Kofstad, P. High Temperature Corrosion; Elsevier Applied Science: London, UK, 1988. [Google Scholar]
- Birks, N.; Meier, G.H.; Pettit, F.S. Introduction to the High. Temperature Oxidation of Metals, 2nd ed.; Cambridge University Press: New York, NY, USA, 2006. [Google Scholar]
- Bai, C.-Y. Effects of electrical discharge surface modification of superalloy Haynes 230 with aluminum and molybdenum on oxidation behavior. Corros. Sci. 2007, 49, 3889–3904. [Google Scholar] [CrossRef]
- Khalid, F.; Hussain, N.; Shahid, K. Microstructure and morphology of high temperature oxidation in superalloys. Mater. Sci. Eng. A 1999, 265, 87–94. [Google Scholar] [CrossRef]
- Hussain, N.; Shahid, K.A.; Khan, I.H.; Rahman, S. Oxidation of high-temperature alloys (superalloys) at elevated temperatures in air: I. Oxid. Met. 1994, 41, 251–269. [Google Scholar] [CrossRef]
- Yang, Q.; Xiong, W.; Li, S.; Dai, H.; Li, J. Characterization of oxide scales to evaluate high temperature oxidation behavior of Ti(C,N)-based cermets in static air. J. Alloy. Compd. 2010, 506, 461–467. [Google Scholar] [CrossRef]
- Park, S.-J.; Seo, S.-M.; Yoo, Y.-S.; Jeong, H.-W.; Jang, H. Statistical Study of the Effects of the Composition on the Oxidation Resistance of Ni-Based Superalloys. J. Nanomater. 2015, 2015, 1–11. [Google Scholar] [CrossRef]
- Park, S.J.; Lee, K.H.; Seo, S.M.; Jeong, H.W.; Yoo, Y.S.; Jang, H.J. Statistics of oxidation resistance of Ni-(0–15)Co-(8–15)Cr-(0–5)Mo-(0–10)W-(3–8)Al-(0–5)Ti-(0–10)Ta-0.1C-0.01B superalloys at 1000 °C by compositional variations. Rare Met. 2019, in press. [Google Scholar] [CrossRef]
- Kim, H.-S.; Park, S.-J.; Seo, S.-M.; Yoo, Y.-S.; Jeong, H.-W.; Jang, H. High temperature oxidation resistance of Ni-(5∼13)Co-(10∼16)Cr-(5∼9)W-5Al-(1∼1.5)Ti-(3∼6)Ta alloys. Met. Mater. Int. 2016, 22, 789–796. [Google Scholar] [CrossRef]
- Giggins, C.S.; Pettit, F.S. Oxidation of Ni-Cr-Al Alloys Between 1000 °C and 1200 °C. J. Electrochem. Soc. 1971, 118, 1782. [Google Scholar] [CrossRef]
- Hass, G. Über das Wachstum und die Struktur dünner Oxydschichten auf Aluminium. Z. Anorg. Allg. Chem. 1947, 254, 96–106. [Google Scholar] [CrossRef]
- Kubaschewski, O.; Hopkins, B.E. Oxidation of Metals and Alloys; Butterworths: London, UK, 1962. [Google Scholar]
- Hauffe, K. Oxidation of Metals; Plenum Press: New York, NY, USA, 1966. [Google Scholar]
- Li, M. High Temperature Corrosion of Metals; Metallurgical Industry Press: Beijing, China, 2001. [Google Scholar]
- Hindam, H.M.; Smeltzer, W.W. Growth and Microstructure of α-Al O3 on Ni-Al Alloys: Internal Precipitation and Transition to External Scale. J. Electrochem. Soc. 1980, 127, 1622–1630. [Google Scholar] [CrossRef]
- Wang, J.; Gupta, A.; Klein, T.M. Plasma enhanced chemical vapor deposition of Cr2O3 thin films using chromium hexacarbonyl (Cr(CO)6) precursor. Thin Solid Films 2008, 516, 7366–7372. [Google Scholar] [CrossRef]
- Zengwu, T.; Rui, H.; Jinshan, L. Isothermal Oxidation Behavior of Ni-20Cr-18W Superalloy at 1100 °C. Rare Metal. Mat. Eng. 2012, 41, 2081–2085. [Google Scholar] [CrossRef]

| Sample no. | Co | Cr | Mo | W | Al | Ti | Ta | C | B | Ni |
|---|---|---|---|---|---|---|---|---|---|---|
| 8Cr–3.5Mo–6W | 9.5 | 8 | 3.5 | 6 | 3 | 5 | 3 | 0.1 | 0.01 | Bal. |
| 10Cr–2.5Mo–6W | 9.5 | 10 | 2.5 | 6 | 3 | 5 | 3 | 0.1 | 0.01 | Bal. |
| 10Cr–3.5Mo–4W | 9.5 | 10 | 3.5 | 4 | 3 | 5 | 3 | 0.1 | 0.01 | Bal. |
| 10Cr–3.5Mo–6W | 9.5 | 10 | 3.5 | 6 | 3 | 5 | 3 | 0.1 | 0.01 | Bal. |
| 10Cr–3.5Mo–8W | 9.5 | 10 | 3.5 | 8 | 3 | 5 | 3 | 0.1 | 0.01 | Bal. |
| 10Cr–4.5Mo–6W | 9.5 | 10 | 4.5 | 6 | 3 | 5 | 3 | 0.1 | 0.01 | Bal. |
| 10Cr–5.5Mo–6W | 9.5 | 10 | 5.5 | 6 | 3 | 5 | 3 | 0.1 | 0.01 | Bal. |
| 12Cr–3.5Mo–6W | 9.5 | 12 | 3.5 | 6 | 3 | 5 | 3 | 0.1 | 0.01 | Bal. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.-J.; Seo, S.-M.; Yoo, Y.-S.; Jeong, H.-W.; Jang, H. Effects of Cr, W, and Mo on the High Temperature Oxidation of Ni-Based Superalloys. Materials 2019, 12, 2934. https://doi.org/10.3390/ma12182934
Park S-J, Seo S-M, Yoo Y-S, Jeong H-W, Jang H. Effects of Cr, W, and Mo on the High Temperature Oxidation of Ni-Based Superalloys. Materials. 2019; 12(18):2934. https://doi.org/10.3390/ma12182934
Chicago/Turabian StylePark, Si-Jun, Seong-Moon Seo, Young-Soo Yoo, Hi-Won Jeong, and HeeJin Jang. 2019. "Effects of Cr, W, and Mo on the High Temperature Oxidation of Ni-Based Superalloys" Materials 12, no. 18: 2934. https://doi.org/10.3390/ma12182934
APA StylePark, S.-J., Seo, S.-M., Yoo, Y.-S., Jeong, H.-W., & Jang, H. (2019). Effects of Cr, W, and Mo on the High Temperature Oxidation of Ni-Based Superalloys. Materials, 12(18), 2934. https://doi.org/10.3390/ma12182934





