Dynamics of Superparamagnetic Iron Oxide Nanoparticles with Various Polymeric Coatings
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Iron Oxide-Based Nanoparticles
2.3. Experimental
2.3.1. Dynamic Light Scattering (DLS)
2.3.2. Scanning Transmission Electron Microscopy (STEM)
2.3.3. Alternating Current Magnetic Susceptibility (ACMS)
2.3.4. Mössbauer Spectroscopy (MS)
2.3.5. X-Ray Diffraction (XRD)
3. Results and Discussion
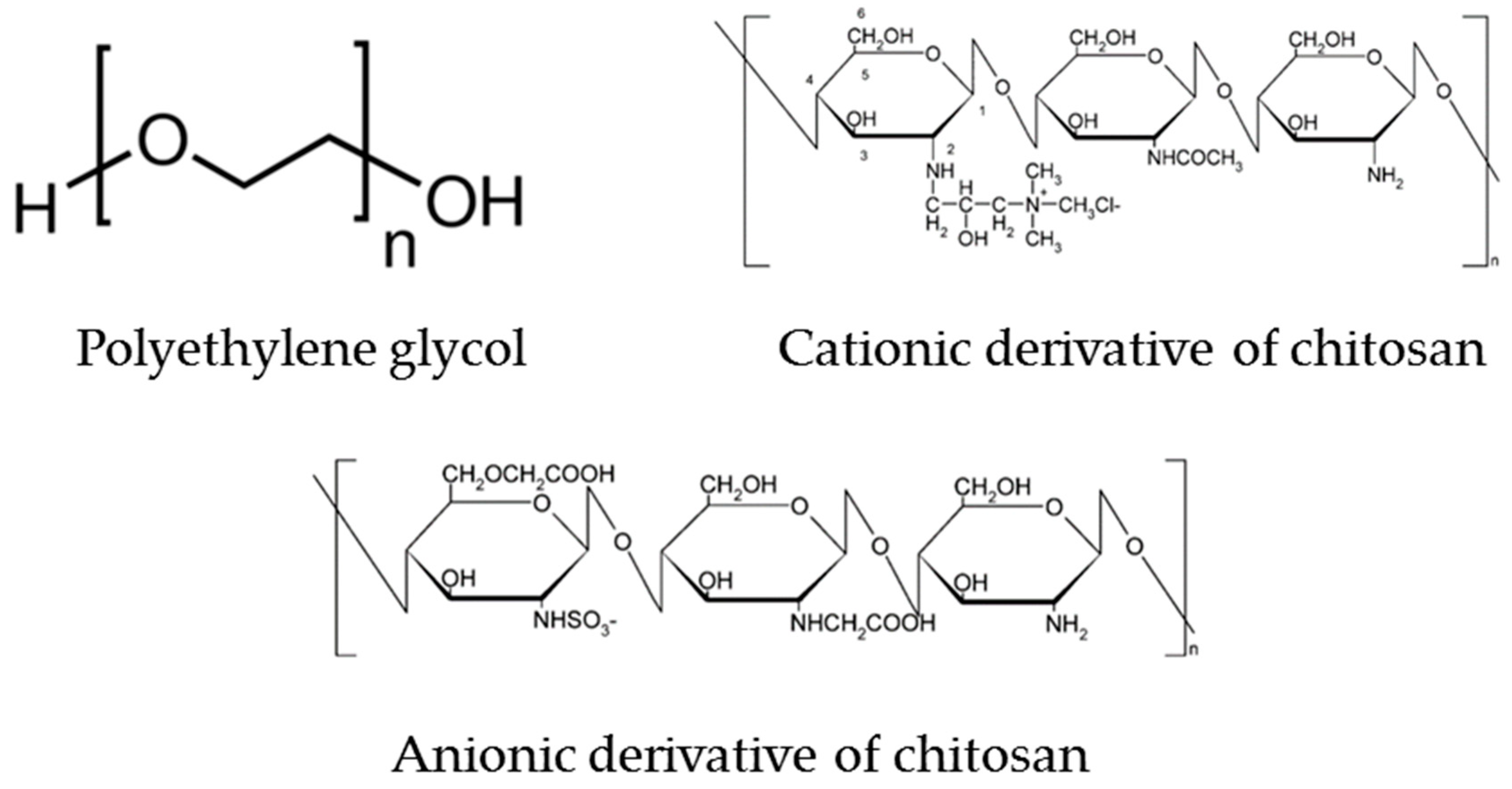
3.1. Nanoparticle Materials Studied
3.2. Physicochemical Properties of the Materials Studied
3.2.1. Results of DLS and Zeta Potential Measurements
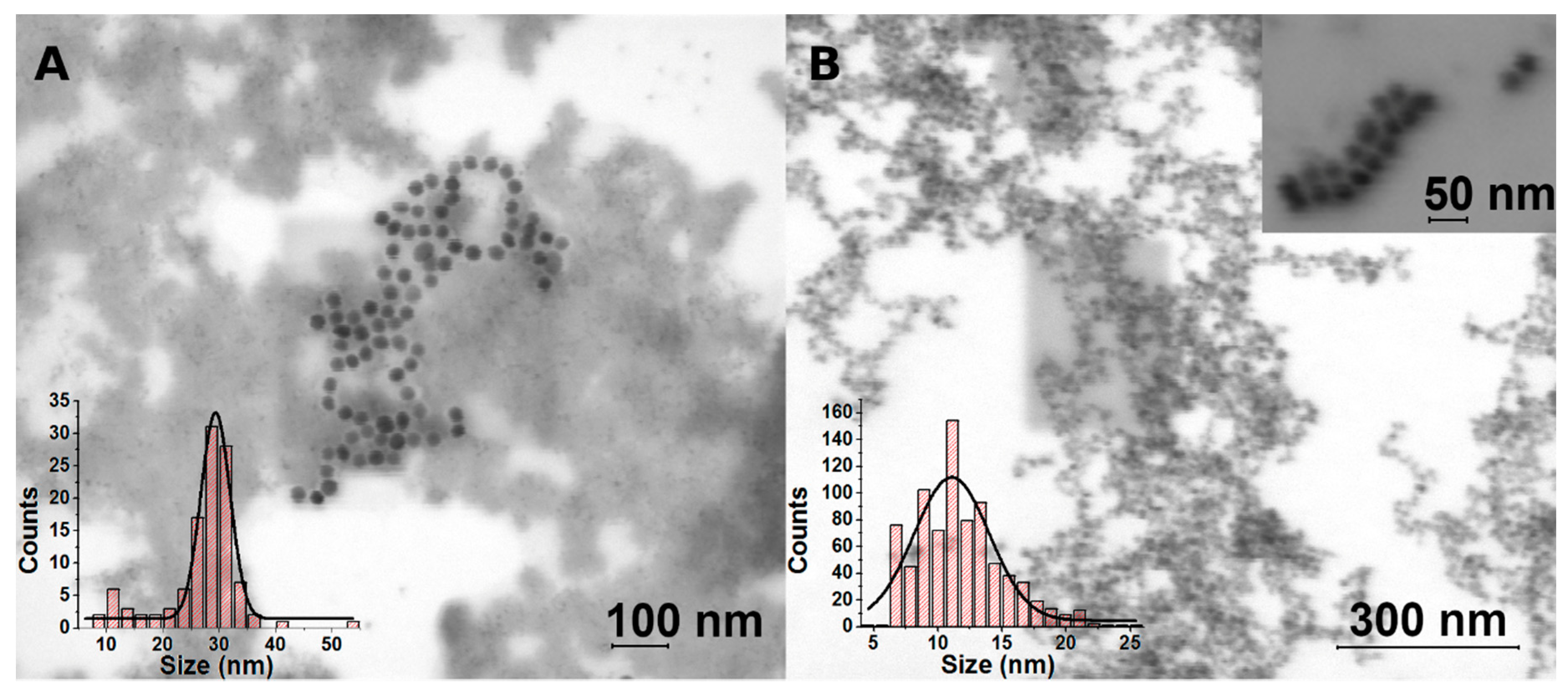
3.2.2. Results of STEM Study
3.3. Structural and Magnetic Characterization
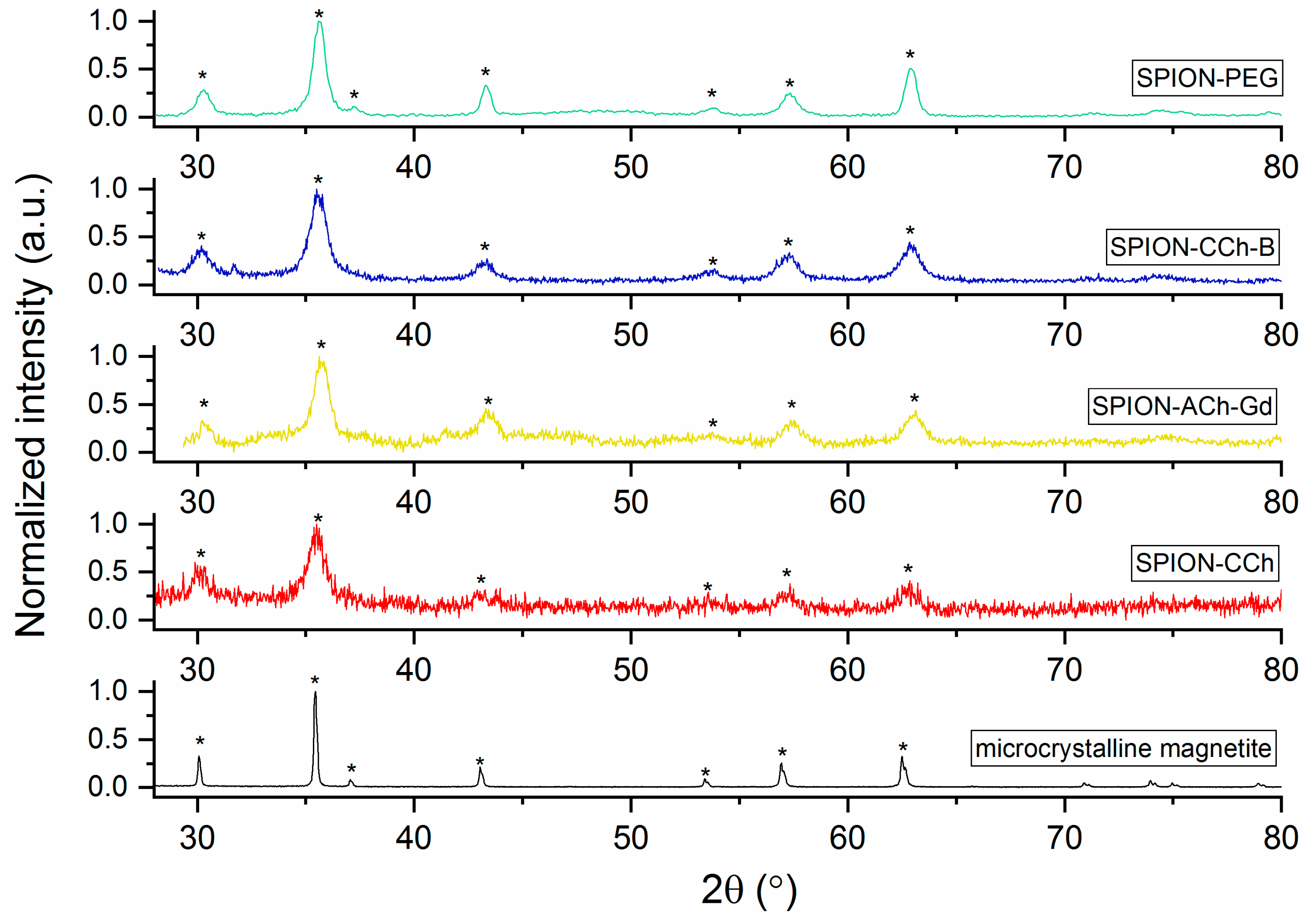
3.3.1. Results of XRD Study
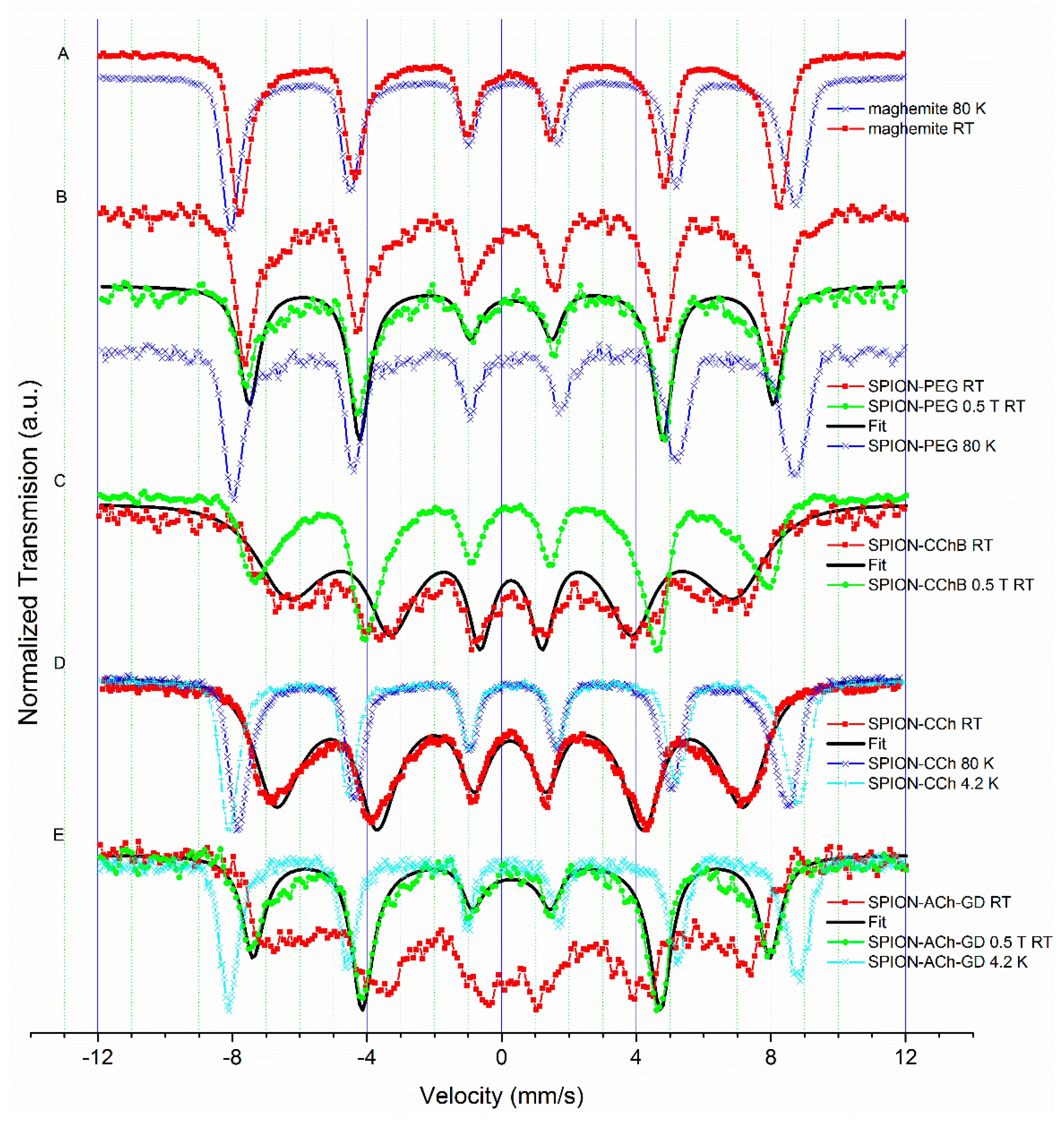
3.3.2. Results of Mössbauer Study
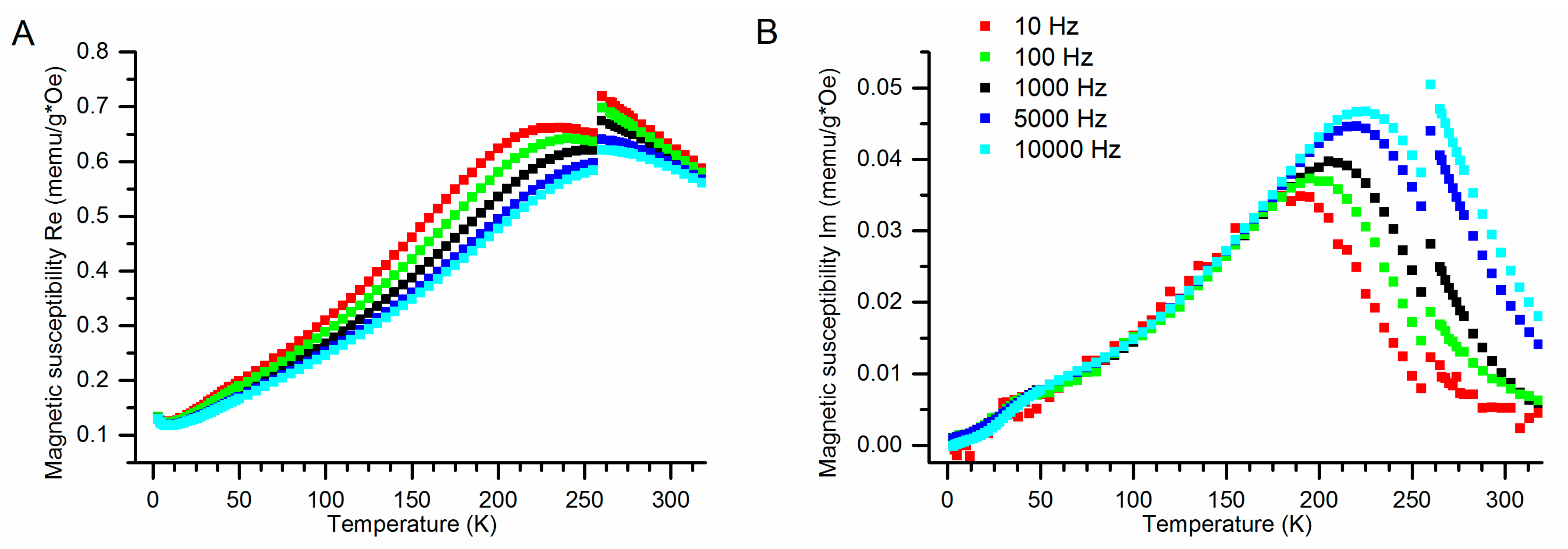
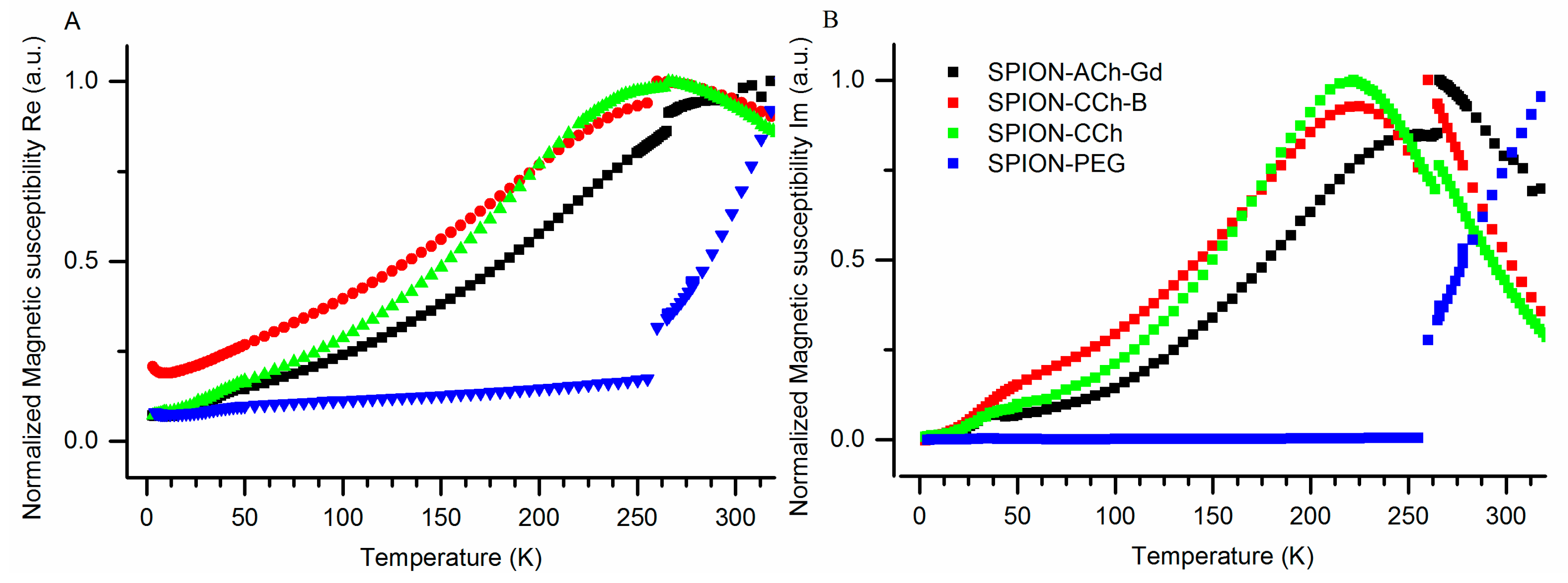
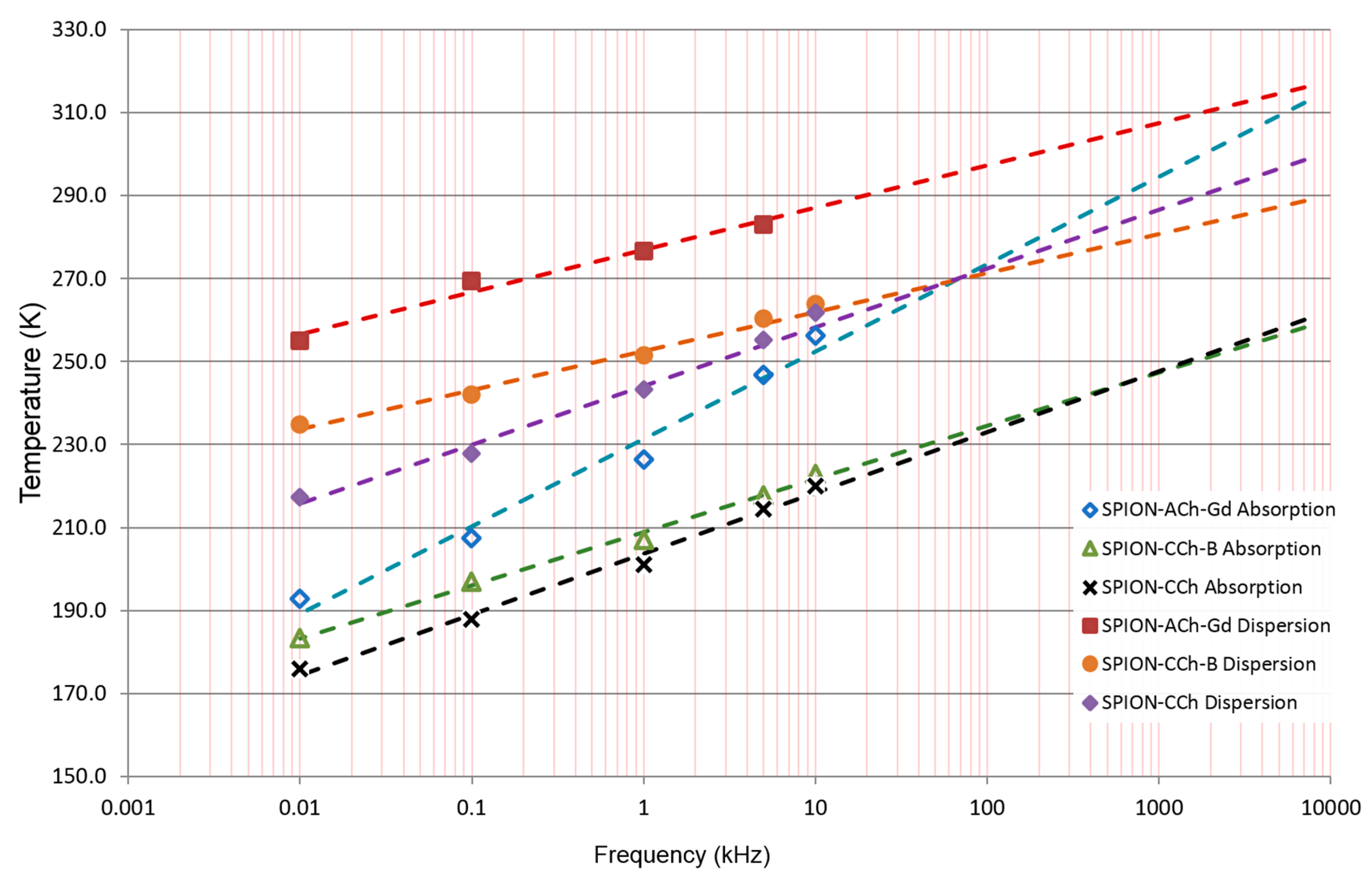
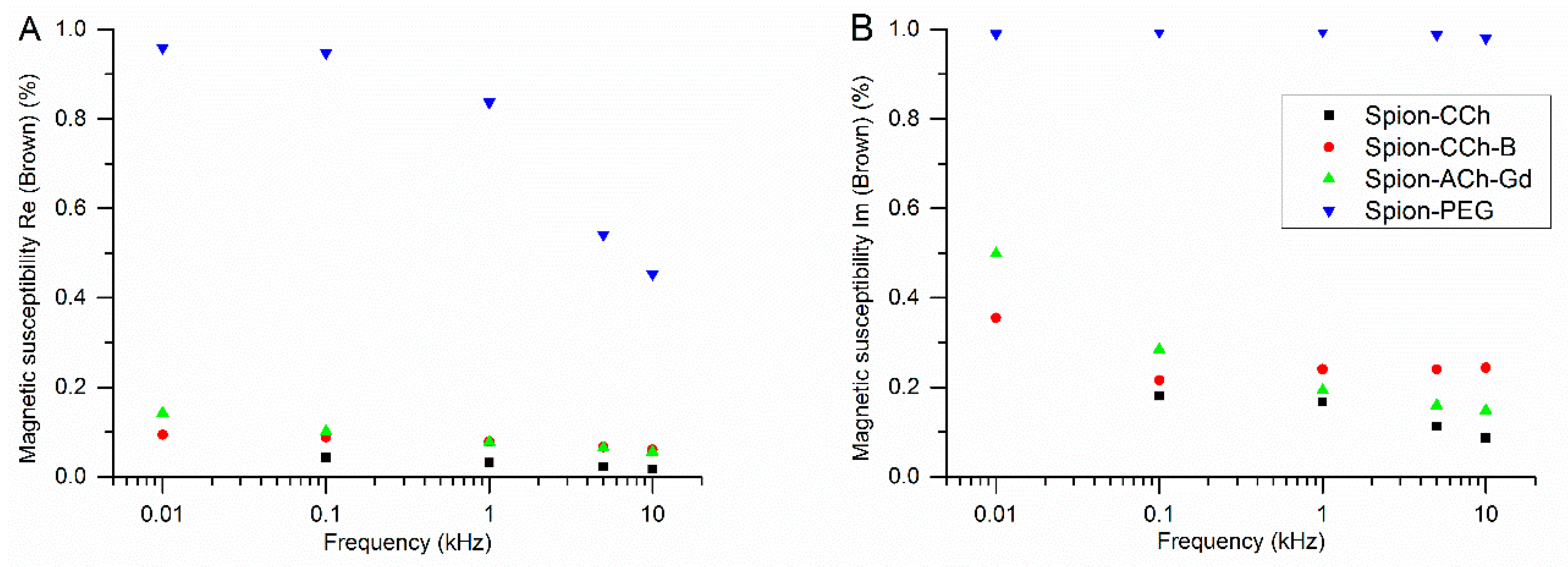
3.3.3. Results of ACMS Measurements
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ansari, S.A.M.K.; Ficiarà, E.; Ruffinatti, F.A.; Stura, I.; Argenziano, M.; Abollino, O.; D’Agata, F. Magnetic Iron Oxide Nanoparticles: Synthesis, Characterization and Functionalization for Biomedical Applications in the Central Nervous System. Materials 2019, 12, 465. [Google Scholar] [CrossRef]
- Kebede, A.; Singh, A.K.; Rai, P.K.; Giri, N.K.; Rai, A.K.; Watal, G.; Gholap, A.V. Controlled synthesis, characterization, and application of iron oxide nanoparticles for oral delivery of insulin. Lasers Med. Sci. 2013, 28, 579–587. [Google Scholar] [CrossRef]
- Kim, D.H.; Nikles, D.E.; Brazel, C.S. Synthesis and characterization of multifunctional chitosan-MnFe2O4 nanoparticles for magnetic hyperthermia and drug delivery. Materials 2010, 3, 4051–4065. [Google Scholar] [CrossRef]
- Chen, H.; Zhen, Z.; Todd, T.; Chu, P.K.; Xie, J. Nanoparticles for improving cancer diagnosis. Mater. Sci. Eng. Rep. 2013, 74, 35–69. [Google Scholar] [CrossRef]
- Siddiqi, K.S.; ur Rahman, A.; Husen, A. Biogenic fabrication of iron/iron oxide nanoparticles and their application. Nanoscale Res. Lett. 2016, 11, 498. [Google Scholar] [CrossRef]
- Casterou, G.; Collière, V.; Lecante, P.; Coppel, Y.; Eliat, P.A.; Gauffre, F.; Kahn, M.L. Improved Transversal Relaxivity for Highly Crystalline Nanoparticles of Pure γ-Fe2O3 Phase. Chem. Eur. J. 2015, 21, 18855–18861. [Google Scholar] [CrossRef]
- Veintemillas-Verdaguer, S.; del Puerto Morales, M.; Bomati-Miguel, O.; Bautista, C.; Zhao, X.; Bonville, P.; Ferreirós, J. Colloidal dispersions of maghemite nanoparticles produced by laser pyrolysis with application as NMR contrast agents. J. Phys. D Appl. Phys. 2004, 37, 2054. [Google Scholar] [CrossRef]
- Múzquiz-Ramos, E.M.; Guerrero-Chávez, V.; Macías-Martínez, B.I.; López-Badillo, C.M.; García-Cerda, L.A. Synthesis and characterization of maghemite nanoparticles for hyperthermia applications. Ceram. Int. 2015, 41, 397–402. [Google Scholar] [CrossRef]
- Hergt, R.; Hiergeist, R.; Hilger, I.; Kaiser, W.A.; Lapatnikov, Y.; Margel, S.; Richter, U. Maghemite nanoparticles with very high AC-losses for application in RF-magnetic hyperthermia. J. Magn. Magn. Mater. 2004, 270, 345–357. [Google Scholar] [CrossRef]
- Dulińska-Litewka, J.; Łazarczyk, A.; Hałubiec, P.; Szafrański, O.; Karnas, K.; Karewicz, A. Superparamagnetic Iron Oxide Nanoparticles—Current and Prospective Medical Applications. Materials 2019, 12, 617. [Google Scholar] [CrossRef]
- Sodipo, B.K.; Aziz, A.A. Recent advances in synthesis and surface modification of superparamagnetic iron oxide nanoparticles with silica. J. Magn. Magn. Mater. 2016, 416, 275–291. [Google Scholar] [CrossRef]
- Hufschmid, R.; Arami, H.; Ferguson, R.M.; Gonzales, M.; Teeman, E.; Brush, L.N.; Krishnan, K.M. Synthesis of phase-pure and monodisperse iron oxide nanoparticles by thermal decomposition. Nanoscale 2015, 7, 11142–11154. [Google Scholar] [CrossRef]
- Darmawan, A.; Smart, S.; Julbe, A.; Diniz da Costa, J.C. Iron oxide silica derived from sol-gel synthesis. Materials 2011, 4, 448–456. [Google Scholar] [CrossRef]
- Okoli, C.; Sanchez-Dominguez, M.; Boutonnet, M.; Järås, S.; Civera, C.; Solans, C.; Kuttuva, G.R. Comparison and functionalization study of microemulsion-prepared magnetic iron oxide nanoparticles. Langmuir 2012, 28, 8479–8485. [Google Scholar] [CrossRef]
- Wu, W.; He, Q.; Jiang, C. Magnetic iron oxide nanoparticles: Synthesis and surface functionalization strategies. Nanoscale Res. Lett. 2008, 3, 397. [Google Scholar] [CrossRef]
- Sodipo, B.K.; Aziz, A.A. One minute synthesis of amino-silane functionalized superparamagnetic iron oxide nanoparticles by sonochemical method. Ultrason. Nanochem. 2018, 40, 837–840. [Google Scholar] [CrossRef]
- García-Jimeno, S.; Estelrich, J. Ferrofluid based on polyethylene glycol-coated iron oxide nanoparticles: Characterization and properties. Colloids Surf. A Physicochem. Eng. Asp. 2013, 420, 74–81. [Google Scholar] [CrossRef]
- Huang, J.; Bu, L.; Xie, J.; Chen, K.; Cheng, Z.; Li, X.; Chen, X. Effects of nanoparticle size on cellular uptake and liver MRI with polyvinylpyrrolidone-coated iron oxide nanoparticles. ACS Nano 2010, 4, 7151–7160. [Google Scholar] [CrossRef]
- Schleich, N.; Sibret, P.; Danhier, P.; Ucakar, B.; Laurent, S.; Muller, R.N.; Danhier, F. Dual anticancer drug/superparamagnetic iron oxide-loaded PLGA-based nanoparticles for cancer therapy and magnetic resonance imaging. Int. J. Pharm. 2013, 447, 94–101. [Google Scholar] [CrossRef]
- Tassa, C.; Shaw, S.Y.; Weissleder, R. Dextran-coated iron oxide nanoparticles: A versatile platform for targeted molecular imaging, molecular diagnostics, and therapy. Acc. Chem. Res. 2011, 44, 842–852. [Google Scholar] [CrossRef]
- Cole, A.J.; David, A.E.; Wang, J.; Galbán, C.J.; Hill, H.L.; Yang, V.C. Polyethylene glycol modified, cross-linked starch-coated iron oxide nanoparticles for enhanced magnetic tumor targeting. Biomaterials 2011, 32, 2183–2193. [Google Scholar] [CrossRef]
- Unsoy, G.; Yalcin, S.; Khodadust, R.; Gunduz, G.; Gunduz, U. Synthesis optimization and characterization of chitosan-coated iron oxide nanoparticles produced for biomedical applications. J. Nanopart Res. 2012, 14, 964. [Google Scholar] [CrossRef]
- Gaihre, B.; Khil, M.S.; Lee, D.R.; Kim, H.Y. Gelatin-coated magnetic iron oxide nanoparticles as carrier system: Drug loading and in vitro drug release study. Int. J. Pharm. 2009, 365, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.L.; Qi, X.R.; Maitani, Y.; Nagai, T. Preparation and characterization of superparamagnetic iron oxide nanoparticles stabilized by alginate. Int. J. Pharm. 2007, 333, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Bloemen, M.; Brullot, W.; Luong, T.T.; Geukens, N.; Gils, A.; Verbiest, T. Improved functionalization of oleic acid-coated iron oxide nanoparticles for biomedical applications. J. Nanopart Res. 2012, 14, 1100. [Google Scholar] [CrossRef] [PubMed]
- Ebrahiminezhad, A.; Ghasemi, Y.; Rasoul-Amini, S.; Barar, J.; Davaran, S. Impact of amino-acid coating on the synthesis and characteristics of iron-oxide nanoparticles (IONs). Bull Korean Chem. Soc. 2012, 33, 3957–3962. [Google Scholar] [CrossRef]
- Alwi, R.; Telenkov, S.; Mandelis, A.; Leshuk, T.; Gu, F.; Oladepo, S.; Michaelian, K. Silica-coated super paramagnetic iron oxide nanoparticles (SPION) as biocompatible contrast agent in biomedical photoacoustics. Biomed. Opt. Express 2012, 3, 2500–2509. [Google Scholar] [CrossRef] [PubMed]
- Lewandowska-Łańcucka, J.; Staszewska, M.; Szuwarzyński, M.; Kępczyński, M.; Romek, M.; Tokarz, W.; Nowakowska, M. Synthesis and characterization of the superparamagnetic iron oxide nanoparticles modified with cationic chitosan and coated with silica shell. J. Alloys Compd. 2014, 586, 45–51. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Serpooshan, V. Silver-coated engineered magnetic nanoparticles are promising for the success in the fight against antibacterial resistance threat. ACS Nano 2012, 6, 2656–2664. [Google Scholar] [CrossRef]
- Ali, A.; Hira Zafar, M.Z.; ul Haq, I.; Phull, A.R.; Ali, J.S.; Hussain, A. Synthesis, characterization, applications, and challenges of iron oxide nanoparticles. Nanotechnol. Sci. Appl. 2016, 9, 49. [Google Scholar] [CrossRef]
- Mahajan, S.; Koul, V.; Choudhary, V.; Shishodia, G.; Bharti, A.C. Preparation and in vitro evaluation of folate-receptor-targeted SPION–polymer micelle hybrids for MRI contrast enhancement in cancer imaging. Nanotechnology 2012, 24, 015603. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Yeap, S.P.; Che, H.X.; Low, S.C. Characterization of magnetic nanoparticle by dynamic light scattering. Nanoscale Res. Lett. 2013, 8, 381. [Google Scholar] [CrossRef] [PubMed]
- Smolensky, E.D.; Park, H.Y.E.; Zhou, Y.; Rolla, G.A.; Marjańska, M.; Botta, M.; Pierre, V.C. Scaling laws at the nanosize: The effect of particle size and shape on the magnetism and relaxivity of iron oxide nanoparticle contrast agents. J. Mater. Chem. B 2013, 1, 2818–2828. [Google Scholar] [CrossRef] [PubMed]
- Tarantash, M.; Nosrati, H.; Kheiri Manjili, H.; Baradar Khoshfetrat, A. Preparation, characterization and in vitro anticancer activity of paclitaxel conjugated magnetic nanoparticles. Drug Dev. Ind. Pharm. 2018, 44, 1895–1903. [Google Scholar] [CrossRef] [PubMed]
- Ghazanfari, M.R.; Kashefi, M.; Shams, S.F.; Jaafari, M.R. Perspective of Fe3O4 Nanoparticles Role in Biomedical Applications. Biochem. Res. Int. 2016, 2016, 7840161. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.X.J.; Hussain, S.M.; Krestin, G.P. Superparamagnetic iron oxide contrast agents: Physicochemical characteristics and applications in MR imaging. Eur Radiol. 2001, 11, 2319–2331. [Google Scholar] [CrossRef] [PubMed]
- Kluchova, K.; Zboril, R.; Tucek, J.; Pecova, M.; Zajoncova, L.; Safarik, I.; Bartonkova, H. Superparamagnetic maghemite nanoparticles from solid-state synthesis–Their functionalization towards peroral MRI contrast agent and magnetic carrier for trypsin immobilization. Biomaterials 2009, 30, 2855–2863. [Google Scholar] [CrossRef]
- Kaczyńska, A.; Guzdek, K.; Derszniak, K.; Karewicz, A.; Lewandowska-Łańcucka, J.; Mateuszuk, Ł.; Skórka, T.; Banasik, T.; Jasiński, K.; Kapusta, C.; et al. Novel nanostructural contrast for magnetic resonance imaging of endothelial inflammation: Targeting SPIONs to vascular endothelium. RSC Adv. 2016, 6, 72586–72595. [Google Scholar] [CrossRef]
- Patel, D.; Moon, J.Y.; Chang, Y.; Kim, T.J.; Lee, G.H. Poly (d,l-lactide-co-glycolide) coated superparamagnetic iron oxide nanoparticles: Synthesis, characterization and in vivo study as MRI contrast agent. Colloids Surf. A Physicochem. Eng. Asp. 2008, 313, 91–94. [Google Scholar] [CrossRef]
- Lachowicz, D.; Kaczyńska, A.; Wirecka, R.; Kmita, A.; Szczerba, W.; Bodzoń-Kułakowska, A.; Sikora, M.; Karewicz, A.; Zapotoczny, S. A Hybrid System for Magnetic Hyperthermia and Drug Delivery: SPION Functionalized by Curcumin Conjugate. Materials 2018, 11, 2388. [Google Scholar] [CrossRef]
- Kandasamy, G.; Sudame, A.; Luthra, T.; Saini, K.; Maity, D. Functionalized hydrophilic superparamagnetic iron oxide nanoparticles for magnetic fluid hyperthermia application in liver cancer treatment. ACS Omega 2018, 3, 3991–4005. [Google Scholar] [CrossRef]
- Bulwan, M.; Zapotoczny, S.; Nowakowska, M. Robust “one-component” chitosan-based ultrathin films fabricated using layer-by-layer technique. Soft Matter 2009, 5, 4726–4732. [Google Scholar] [CrossRef]
- Szpak, A.; Kania, G.; Skórka, T.; Tokarz, W.; Zapotoczny, S.; Nowakowska, M. Stable aqueous dispersion of superparamagnetic iron oxide nanoparticles protected by charged chitosan derivatives. J. Nanopart Res. 2013, 15, 1372. [Google Scholar] [CrossRef] [PubMed]
- Szpak, A.; Fiejdasz, S.; Prendota, W.; Strączek, T.; Kapusta, C.; Szmyd, J.; Nowakowska, M.; Zapotoczny, S. T1–T2 dual-modal MRI contrast agents based on superparamagnetic iron oxide nanoparticles with surface attached gadolinium complexes. J. Nanopart Res. 2014, 16, 2678. [Google Scholar] [CrossRef] [PubMed]
- Rancourt, D.G. Analytical methods for Mössbauer spectral analysis of complex materials. In Mössbauer Spectroscopy Applied to Magnetism and Materials Science; Long, G.J., Grandjean, F., Eds.; Springer: Boston, MA, USA, 1996; pp. 105–124. [Google Scholar] [CrossRef]
- Mørup, S. Mössbauer effect in small particles. Hyperfine Interact 1990, 60, 959–973. [Google Scholar] [CrossRef]
- Cornell, R.M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses, 2nd ed.; John Wiley & Sons: Weinheim, Germany, 2003. [Google Scholar] [CrossRef]
- da Costa, G.M.; Blanco-Andujar, C.; De Grave, E.; Pankhurst, Q.A. Magnetic nanoparticles for in vivo use: A critical assessment of their composition. J. Phys. Chem. B 2014, 118, 11738–11746. [Google Scholar] [CrossRef] [PubMed]
- Cullity, B.D. Element of X-Ray Diffraction, 2nd ed.; Addison-Wesley Publishing Company Inc.: Boston, MA, USA, 1978; p. 102. [Google Scholar]
- Dahal, J.N.; Neupane, D.; Mishra, S.R. Exchange-Coupling Behavior in SrFe12O19/La0.7Sr0.3MnO3 Nanocomposites. Ceramics 2019, 2, 100–111. [Google Scholar] [CrossRef]
- Figueroa, A.I.; Bartolomé, J.; García, L.M.; Bartolomé, F.; Arauzo, A.; Millán, A.; Palacio, F. Magnetic anisotropy of maghemite nanoparticles probed by RF transverse susceptibility. Phys. Procedia 2015, 75, 1050–1057. [Google Scholar] [CrossRef]
- Komorida, Y.; Mito, M.; Deguchi, H.; Takagi, S.; Millán, A.; Silva, N.J.O.; Palacio, F. Surface and core magnetic anisotropy in maghemite nanoparticles determined by pressure experiments. Appl. Phys. Lett. 2009, 94, 202503. [Google Scholar] [CrossRef]
- Murad, E.; Johnston, J.H. Iron Oxides and Oxyhydroxides. In Mössbauer Spectroscopy Applied to Inorganic Chemistry; Long, G.J., Ed.; Plenum Press: New York, NY, USA, 1989. [Google Scholar] [CrossRef]
- Brown, R. XXVII. A brief account of microscopical observations made in the months of June, July and August 1827, on the particles contained in the pollen of plants; and on the general existence of active molecules in organic and inorganic bodies. Philos. Mag. 1828, 4, 161–173. [Google Scholar] [CrossRef]
- Debye, P. Polar molecules, Pp. 172. New York: Chemical Catalog Co., Inc. J. Chem. Technol. Biotechnol. 1929, 48, 1036–1037. [Google Scholar] [CrossRef]
- Néel, L. Influence des fluctuations thermiques sur l’aimantation de grains ferromagnétiques très fins. C. R. Hebd. Seances Acad. Sci. 1949, 228, 664–666. (In French) [Google Scholar]
- Balanda, M. AC susceptibility studies of phase transitions and magnetic relaxation: Conventional, molecular and low-dimensional magnets. Acta Phys. Pol. A 2013, 124, 964–976. [Google Scholar] [CrossRef]
- Baker, I.; Zeng, Q.; Li, W.; Sullivan, C.R. Heat deposition in iron oxide and iron nanoparticles for localized hyperthermia. J. Appl. Phys. 2006, 99, H106–H108. [Google Scholar] [CrossRef]
- Goya, G.F.; Fernandez-Pacheco, R.; Arruebo, M.; Cassinelli, N.; Ibarra, M.R. Brownian rotational relaxation and power absorption in magnetite nanoparticles. J. Magn. Magn. Mater. 2007, 316, 132–135. [Google Scholar] [CrossRef]
- Torres, T.E.; Lima, E.; Calatayud, M.P.; Sanz, B.; Ibarra, A.; Fernández-Pacheco, R.; Mayoral, A.; Marquina, C.; Ibarra, M.R.; Goya, G.F. The relevance of Brownian relaxation as power absorption mechanism in Magnetic Hyperthermia. Sci. Rep. 2019, 9, 3992. [Google Scholar] [CrossRef] [PubMed]
| Sample Name | Mean Diameter (by Number) d [nm] | Zeta Potential ξ [mV] |
|---|---|---|
| SPION-CCh | 121 | +32 ± 6 |
| SPION-CCh-B | 195 | −12 ± 5 |
| SPION-ACh-Gd | 93 | −34 ± 7 |
| SPION-PEG | 49 | −9 ± 4 |
| Fitted Parameters | SPION -PEG RT | SPION -PEG RT; 0.5 T | SPION-CCH RT | SPION-ACh-Gd RT | SPION-Ach-Gd RT; 0.5 T | SPION-CCh-B RT | SPION -CCh-B RT; 0.5 T |
|---|---|---|---|---|---|---|---|
| IS * (mm/s) | 0.38(2) | 0.39(1) | 0.36(1) | 0.39(2) | 0.4 | 0.39(1) | 0.38(1) |
| BHF (T) | 48.3(1) | 48.3 | 46.2(2) | 44.5(4) | 47.7 | 46(6) | 46.5(7) |
| G (mm/s) | 0.35 | 0.35 | 0.54(1) | 0.78(4) | 0.32 | 0.35 | 0.35 |
| F (MHz) | 1.2(7) | 1.2(4) | 106(3) | 43(4) | 3.8(5) | 72(6) | 4.1(4) |
| ρ | 0.44(4) | 0.98(8) | 0.91(2) | 0.88(5) | 0.82(4) | 0.78(1) | 0.8 |
| χ2 | 0.65 | 1.17 | 2.19 | 0.62 | 1.19 | 1.33 | 6.81 |
| Sample | Relative Intensity (%) | IS (mm/s) | BHF (T) | QS (mm/s) |
|---|---|---|---|---|
| SPION-CCh at 4.2 K | 61(1) | 0.48(1) | 52.3(3) | 0.02(1) |
| 39(1) | 0.41(1) | 50.3(4) | −0.01(1) | |
| SPION-CCh at 80 K | 68(1) | 0.45(1) | 50.9(2) | −0.02(1) |
| 32(1) | 0.39(1) | 48.4(5) | −0.02(1) | |
| SPION-ACh-Gd at 4.2 K | 64(2) | 0.45(1) | 52.3(4) | 0.01(1) |
| 36(2) | 0.40(1) | 50.3(7) | −0.01(1) | |
| SPION-PEG at 80 K | 58(3) | 0.52(1) | 52.3(7) | −0.02(1) |
| 42(3) | 0.46(1) | 50.2(9) | −0.02(1) | |
| Maghemite at 80 K | 61(1) | 0.46(1) | 52.5(3) | −0.01(1) |
| 39(1) | 0.40(1) | 50.6(5) | −0.02(7) |
| Sample | SPION-ACh-Gd | SPION-CCh-B | SPION-CCh |
|---|---|---|---|
| Fmax312 dispersion (MHz) | 2.7 | 967 | 30 |
| Fmax312 absorption (MHz) | 6.6 | 108 000 | 208 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strączek, T.; Fiejdasz, S.; Rybicki, D.; Goc, K.; Przewoźnik, J.; Mazur, W.; Nowakowska, M.; Zapotoczny, S.; Rumian, S.; Kapusta, C. Dynamics of Superparamagnetic Iron Oxide Nanoparticles with Various Polymeric Coatings. Materials 2019, 12, 1793. https://doi.org/10.3390/ma12111793
Strączek T, Fiejdasz S, Rybicki D, Goc K, Przewoźnik J, Mazur W, Nowakowska M, Zapotoczny S, Rumian S, Kapusta C. Dynamics of Superparamagnetic Iron Oxide Nanoparticles with Various Polymeric Coatings. Materials. 2019; 12(11):1793. https://doi.org/10.3390/ma12111793
Chicago/Turabian StyleStrączek, Tomasz, Sylwia Fiejdasz, Damian Rybicki, Kamil Goc, Janusz Przewoźnik, Weronika Mazur, Maria Nowakowska, Szczepan Zapotoczny, Stanisław Rumian, and Czesław Kapusta. 2019. "Dynamics of Superparamagnetic Iron Oxide Nanoparticles with Various Polymeric Coatings" Materials 12, no. 11: 1793. https://doi.org/10.3390/ma12111793
APA StyleStrączek, T., Fiejdasz, S., Rybicki, D., Goc, K., Przewoźnik, J., Mazur, W., Nowakowska, M., Zapotoczny, S., Rumian, S., & Kapusta, C. (2019). Dynamics of Superparamagnetic Iron Oxide Nanoparticles with Various Polymeric Coatings. Materials, 12(11), 1793. https://doi.org/10.3390/ma12111793






