Al2O3-Cu Substrate with Co-Continuous Phases Made by Powder Sintering Process
Abstract
1. Introduction
2. Experiment
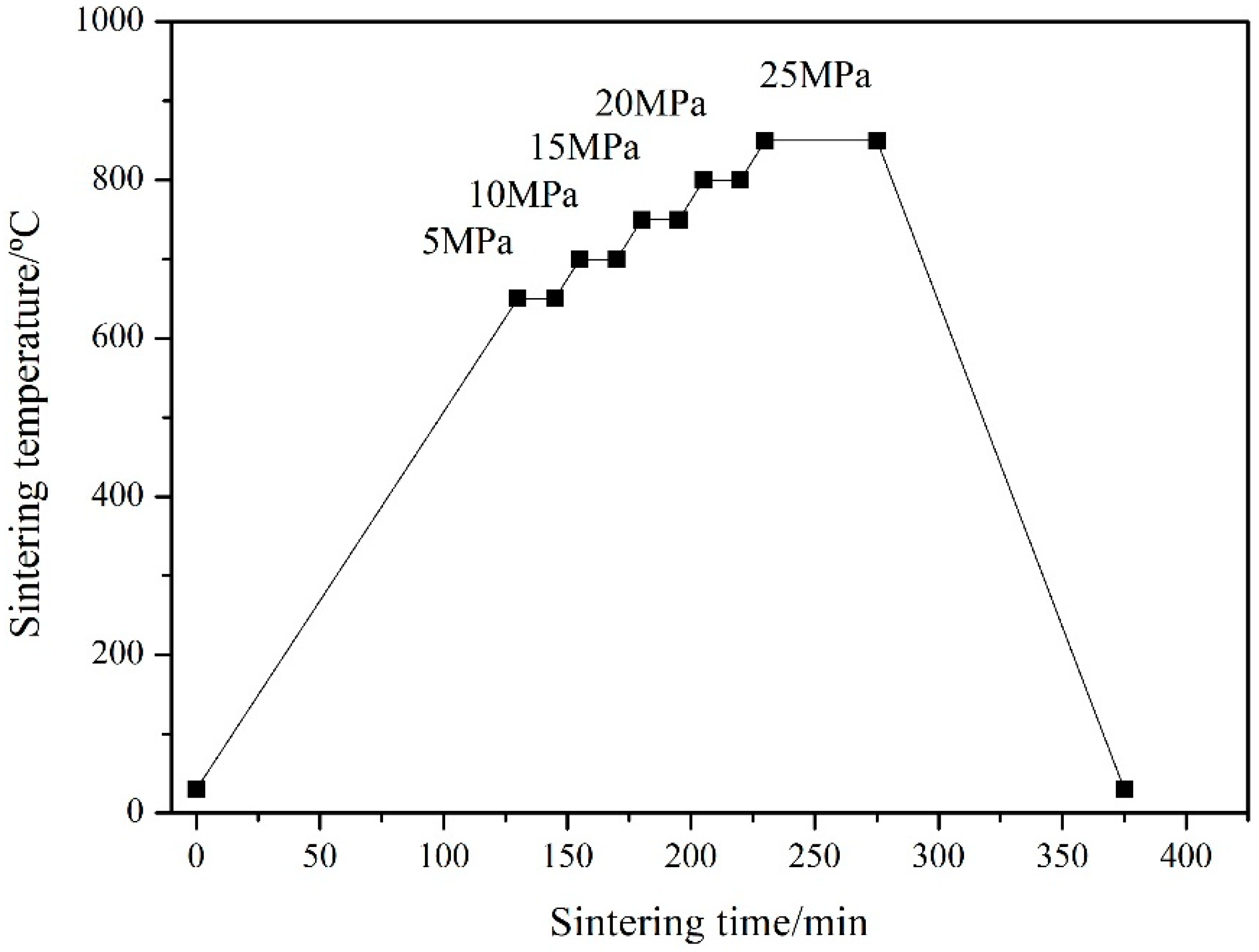
2.1. Materials and Fabrication Process
2.2. Measurements
3. Results and Discussion
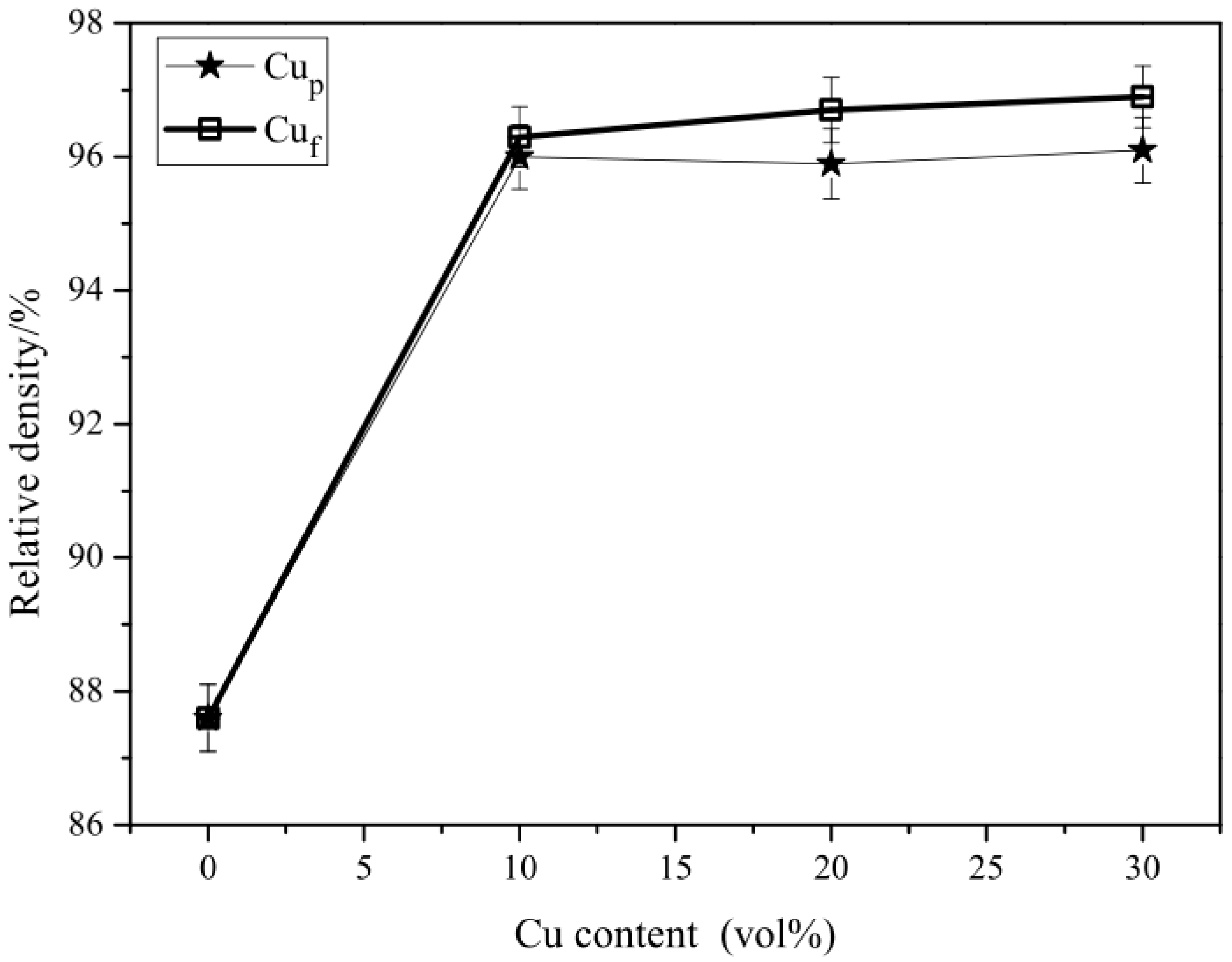
3.1. Relative Density of the Composites
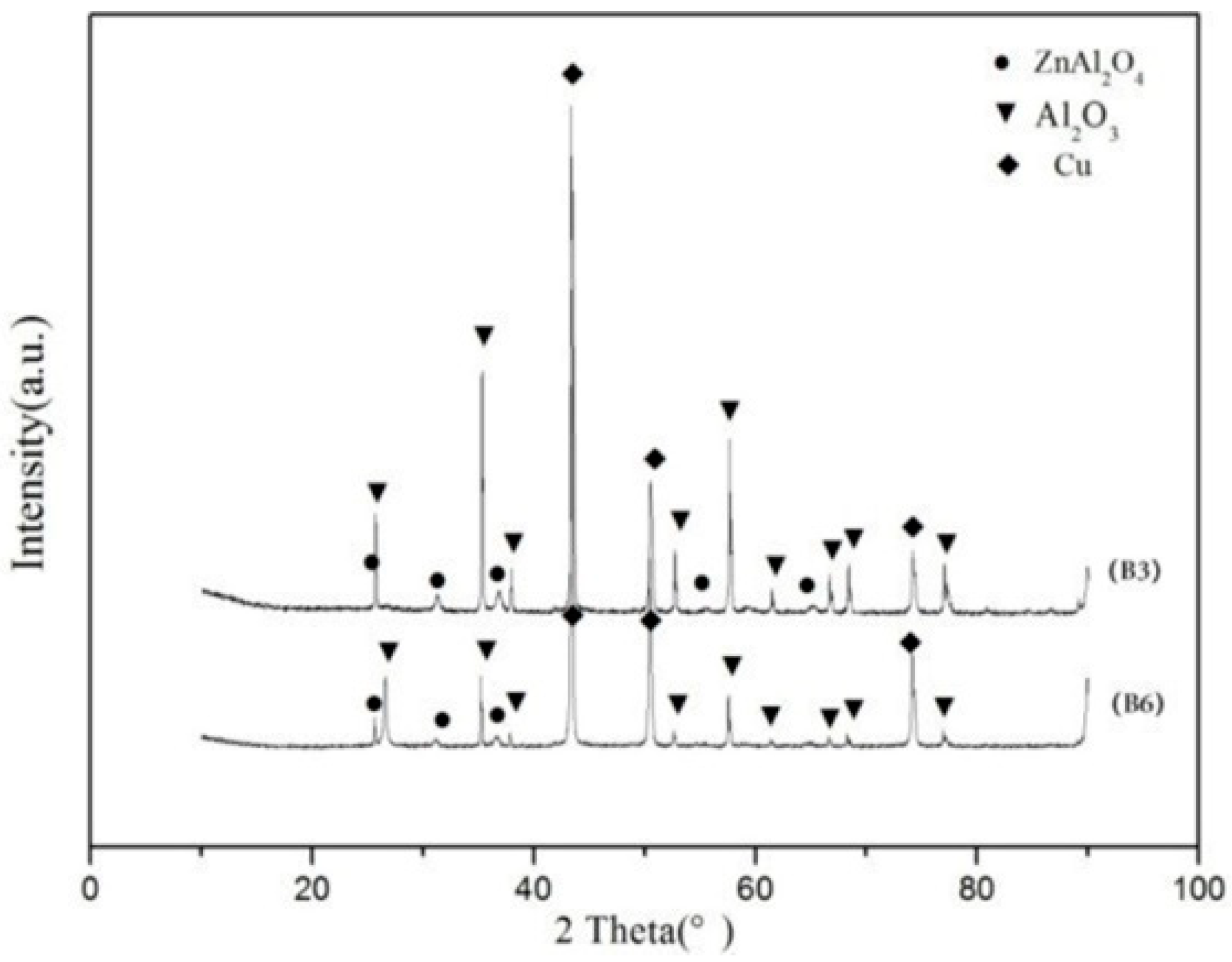
3.2. Microstructure of the Composites
- (1)
- At low temperature, the green body of Al2O3/glass/Cuf was formed, the Cu fiber deformed and the oxide layer on the surface of copper fiber was destroyed by glass and Al2O3 particles under outside pressure.
- (2)
- ZnO-SiO2-CaO glass melted when the sintering temperature exceeded 600 °C.
- (3)
- Melted glass infiltrated along the Cu fiber surface and inner gas escaped due to the spatial structure of fibers. The local surface of copper fibers corroded by the combined alternative influences of relative high pressure and erosion of the molten glass when the sintering temperature approached 850 °C.
- (4)
- The ceramic green body gradually finished densification during the hot-pressing process.
- (5)
- The deformed Cu fibers welded at the overlapping node during cooling process.
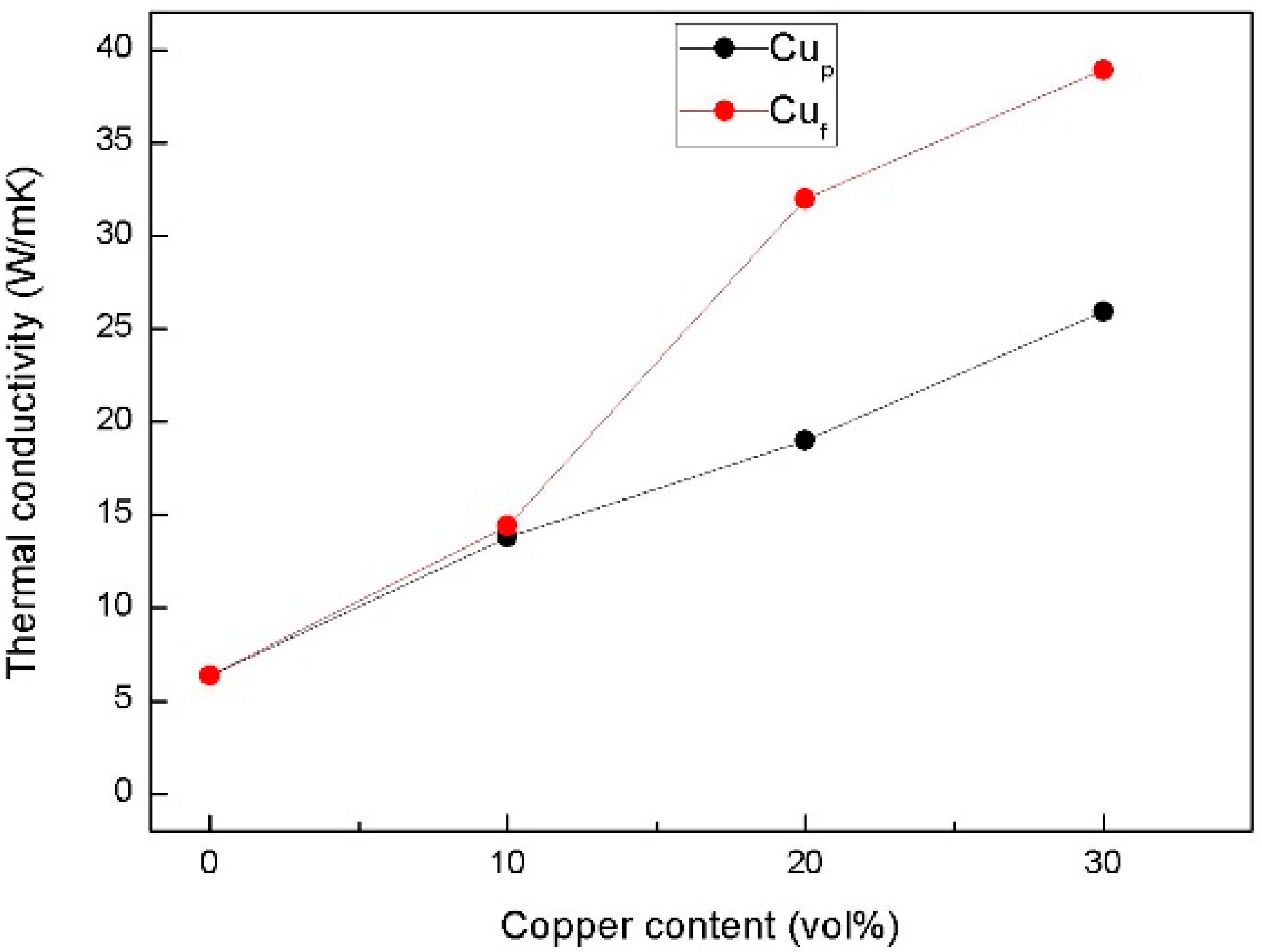
3.3. Thermal Performance
- (1)
- At the beginning of hot-pressing, copper fibers deformed and connected physically, forming a 3D network like that in the Al2O3/30glass/30Cf substrate. The oxide layer on the surface of copper fibers was destroyed by glass and Al2O3 particles.
- (2)
- The surfaces of copper fibers tend to react with molten glass phase.
- (3)
- Copper fibers achieved metallurgical bonding with each other and exhibited a molten-like vermiform shape.
- (4)
- A continuous metallurgy 3D network of copper fiber was established, similar to that of die-casting or reactive metal penetration processes.
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wang, J.; Zhao, X.J.; Cai, Y.X.; Zhang, C.; Bao, W.W. Experimental study on the thermal management of high-power LED headlight cooling device integrated with thermoelectric cooler package. Energy Convers. Manag. 2015, 101, 532–540. [Google Scholar] [CrossRef]
- Ye, H.; Mihailovic, M.; Wong, C.K.Y.; Zeijl, H.W.V.; Gielen, A.W.J.; Zhang, G.Q.; Sarro, P.M. Two-phase cooling of light emitting diode for higher light output and increased efficiency. Appl. Therm. Eng. 2013, 52, 353–359. [Google Scholar] [CrossRef]
- Narendran, N.; Gu, Y. Life of LED-based white light sources. J. Disp. Technol. 2005, 1, 167–171. [Google Scholar] [CrossRef]
- Zhang, L.Y.; Zhang, Y.F.; Chen, J.Q.; Bai, S.L. Fluid flow and heat transfer characteristics of liquid cooling microchannels in LTCC multilayered packaging substrate. Int. J. Heat Mass Transf. 2015, 84, 339–345. [Google Scholar] [CrossRef]
- Wu, H.H.; Lin, K.H.; Lin, S.T. A study on the heat dissipation of high power multi-chip COB LEDs. Microelectron. J. 2012, 43, 280–287. [Google Scholar] [CrossRef]
- Couturier, R.; Ducret, D.; Merle, P.; Disson, J.P.; Joubert, P. Elaboration and characterization of a metal matrix composite: Al/AlN. J. Eur. Ceram. Soc. 1997, 17, 1861–1866. [Google Scholar] [CrossRef]
- Kidalov, S.V.; Shakhov, F.M. Thermal conductivity of diamond composites. Materials 2009, 2, 2467–2495. [Google Scholar] [CrossRef]
- Yung, K.C.; Liem, H.; Choy, H.S. Heat dissipation performance of a high-brightness LED package assembly using high-thermal conductivity filler. Appl. Opt. 2013, 52, 8484–8493. [Google Scholar] [CrossRef] [PubMed]
- Raza, K.; Khalid, F.A. Optimization of sintering parameters for diamond-copper composites in conventional sintering and their thermal conductivity. J. Alloys Compd. 2014, 615, 111–118. [Google Scholar] [CrossRef]
- Mizuuchi, K.; Inoue, K.; Agari, Y.; Nagaoka, T.; Sugioka, M.; Tanaka, M.; Takeuchi, T.; Tani, J.; Kawahara, M.; Makino, Y.; et al. Processing and thermal properties of Al/AlN composites in continuous solid-liquid co-existent state by spark plasma sintering. Compos. Part B Eng. 2012, 43, 1557–1563. [Google Scholar] [CrossRef]
- Chmielewski, M.; Weglewski, W. Comparison of experimental and modelling results of thermal properties in Cu-AlN composite materials. Bull. Pol. Acad. Sci. Tech. 2013, 61, 507–514. [Google Scholar] [CrossRef]
- Chen, Z.; Tan, Z.; Ji, G.; Fan, G.; Schryvers, D.; Ouyang, Q.; Li, Z. Effect of interface evolution on thermal conductivity of vacuum hot pressed SiC/Al composites. Adv. Eng. Mater. 2015, 17, 1076–1084. [Google Scholar] [CrossRef]
- Wang, D.; Zheng, Z.; Lv, J.; Xu, G.; Zhou, S.; Tang, W.; Wu, Y. Enhanced thermal conductive 3D-SiC/Al-Si-Mg interpenetrating composites fabricated by pressureless infiltration. Ceram. Int. 2017, 43, 1755–1761. [Google Scholar] [CrossRef]
- Travitzky, N. Processing of ceramic-metal composites. Adv. Appl. Ceram. 2012, 111, 286–300. [Google Scholar] [CrossRef]
- Manfredi, D.; Pavese, M.; Biamino, S.; Antonini, A.; Fino, P.; Badini, C. Microstructure and mechanical properties of co-continuous metal/ceramic composites obtained from reactive metal penetration of commercial aluminium alloys into cordierite. Compos. Part A Appl. Sci. Manuf. 2010, 41, 639–645. [Google Scholar] [CrossRef]
- Wang, S.X.; Liu, G.S.; Ouyang, X.Q.; Wang, Y.D.; Zhang, D. Fabrication of Al2O3/glass/Cf composite substrate with high thermal conductivity. J. Mater. Eng. Perform. 2016, 25, 642–647. [Google Scholar] [CrossRef]
- Sevik, H.; Kurnaz, S.C. Properties of alumina particulate reinforced aluminum alloy produced by pressure die casting. Mater. Des. 2006, 27, 676–683. [Google Scholar] [CrossRef]






| ZnO | SiO2 | CaCO3 | Al2O3 | TiO2 | MgO | Na2CO3 |
|---|---|---|---|---|---|---|
| 20 | 45 | 18 | 6 | 2 | 8 | 1 |
| No. | Al2O3 | Cuf(p) | Glass |
|---|---|---|---|
| F-0 | 70 | 0 | 30 |
| F-1 | 60 | 10 | 30 |
| F-2 | 50 | 20 | 30 |
| F-3 | 40 | 30 | 30 |
| Substrate | CTE (10−6/K) |
|---|---|
| F-0 | 5.41 |
| F-1 | 7.6 |
| F-2 | 8.01 |
| F-3 | 8.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Lan, H.; Wang, W.; Liu, G.; Zhang, D. Al2O3-Cu Substrate with Co-Continuous Phases Made by Powder Sintering Process. Materials 2018, 11, 1477. https://doi.org/10.3390/ma11081477
Wang S, Lan H, Wang W, Liu G, Zhang D. Al2O3-Cu Substrate with Co-Continuous Phases Made by Powder Sintering Process. Materials. 2018; 11(8):1477. https://doi.org/10.3390/ma11081477
Chicago/Turabian StyleWang, Shuangxi, Haifeng Lan, Wenjun Wang, Gaoshan Liu, and Dan Zhang. 2018. "Al2O3-Cu Substrate with Co-Continuous Phases Made by Powder Sintering Process" Materials 11, no. 8: 1477. https://doi.org/10.3390/ma11081477
APA StyleWang, S., Lan, H., Wang, W., Liu, G., & Zhang, D. (2018). Al2O3-Cu Substrate with Co-Continuous Phases Made by Powder Sintering Process. Materials, 11(8), 1477. https://doi.org/10.3390/ma11081477




