Biological Compatibility of a Polylactic Acid Composite Reinforced with Natural Chitosan Obtained from Shrimp Waste
Abstract
1. Introduction
2. Experimental Procedures
2.1. Materials and Methods
2.1.1. Extraction of Chitosan
2.1.2. Composites Preparation
2.2. Microstructural and Mechanical Characterization
2.3. Biocompatibility Analysis
2.3.1. In Vitro Degradation in PBS
2.3.2. Cell Culture
Cell Culture on Biomaterials
Actin Stain
Scanning Electron Microscopy
Cell Viability
Alizarin Red Stain
3. Results and Discussion
3.1. Distribution of Chitosan in PLA
3.2. Structural Characterization of the PLA/Chitosan Composites
3.3. Mechanical Properties of the PLA/Chitosan Composites
3.4. Biocompatibility of the PLA/Chitosan Composites
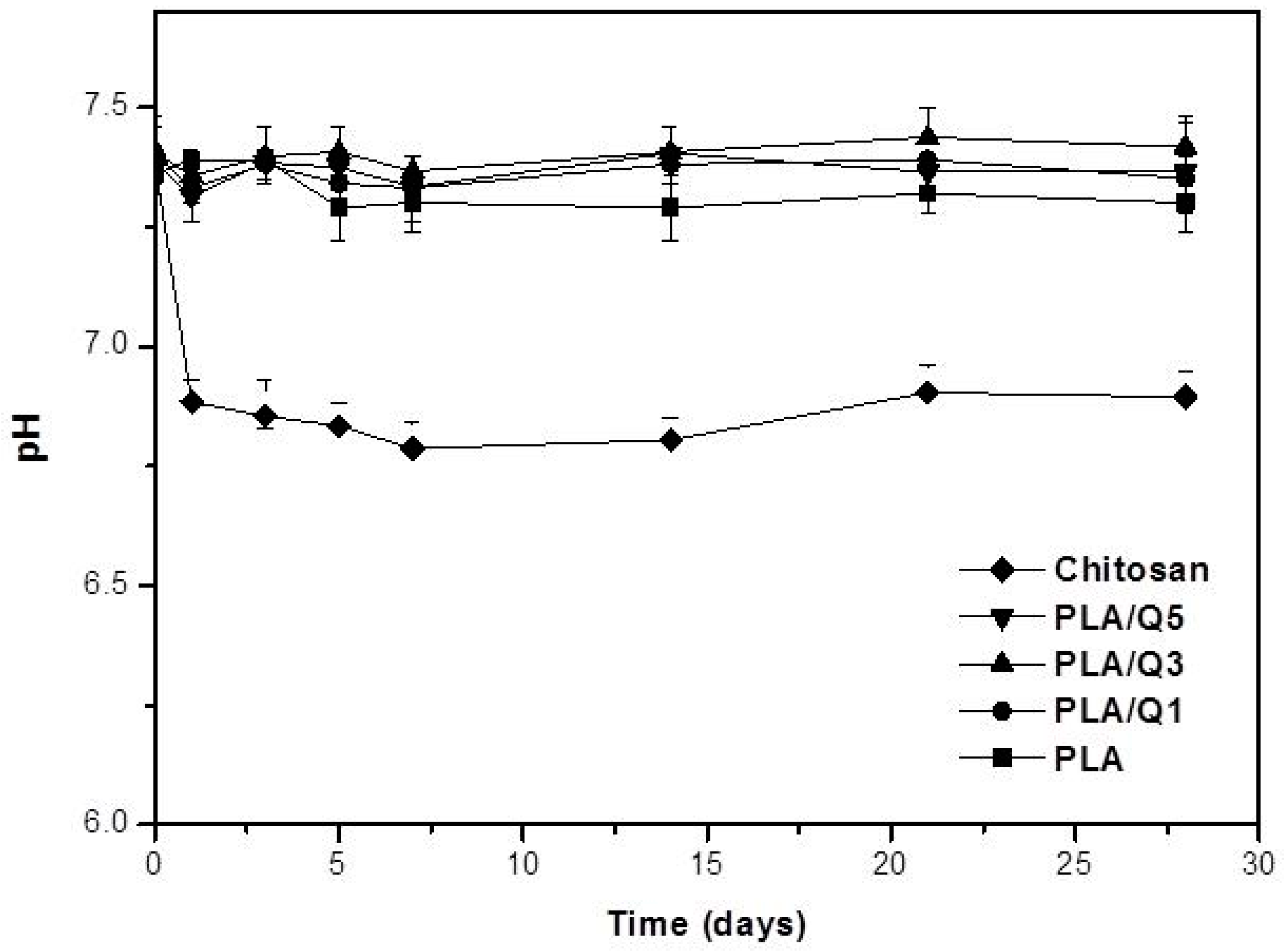
3.4.1. Hydrolytic Degradation of Composites in PBS
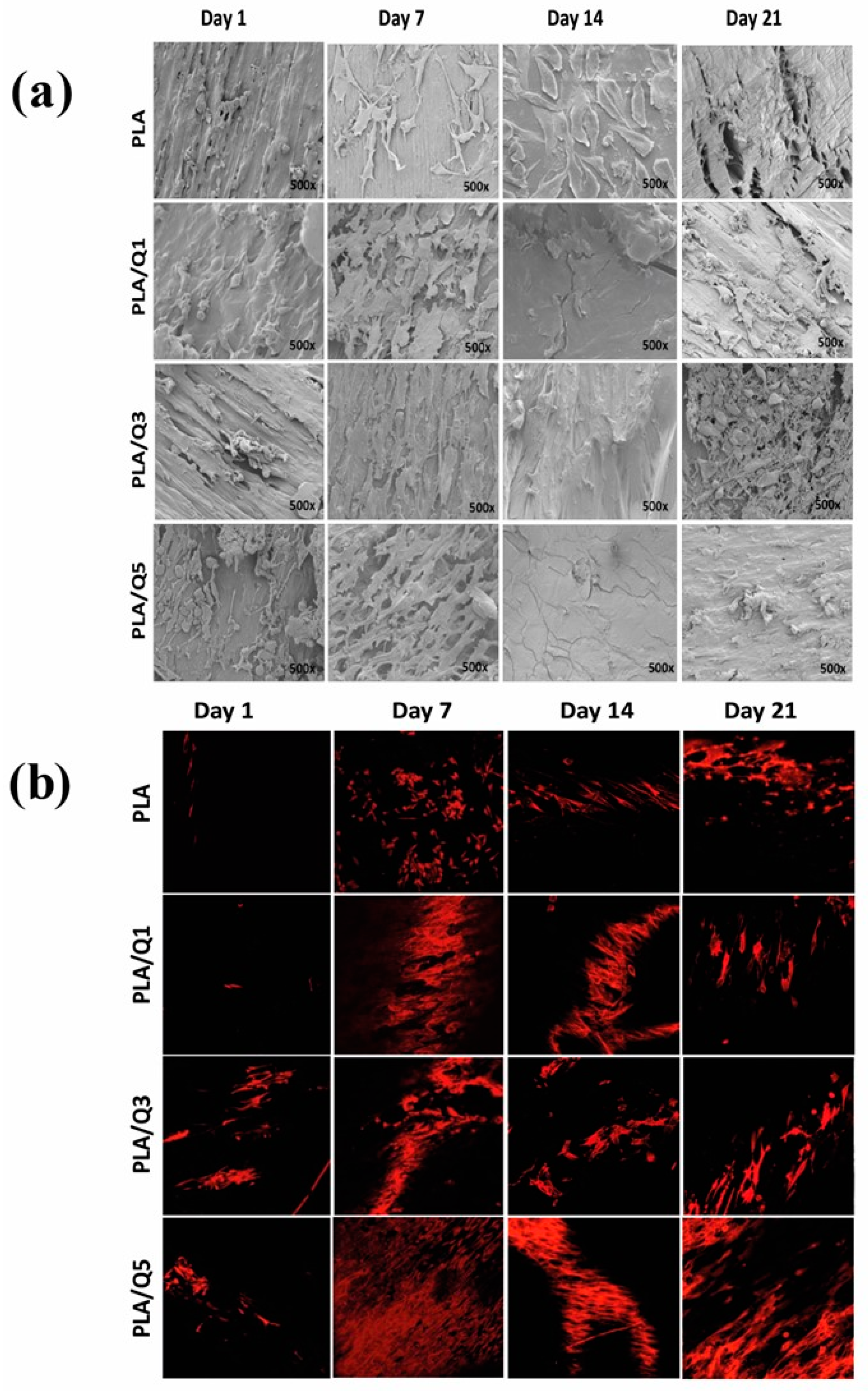
3.4.2. Adhesion of MG-63 Osteoblasts on the Composites PLA/Chitosan
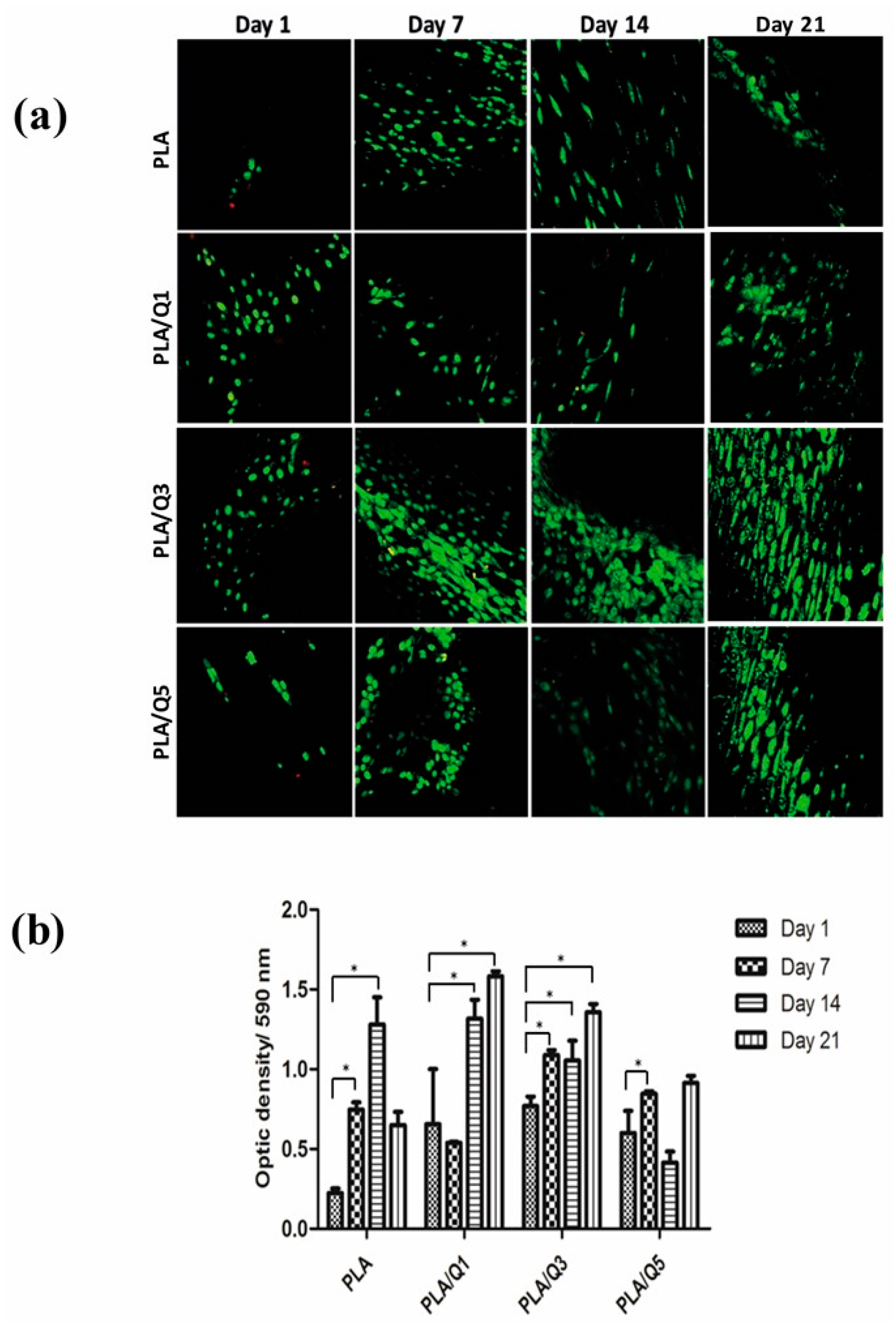
3.4.3. Viability and Proliferation of MG-63 Osteoblasts on the PLA/Chitosan Composites
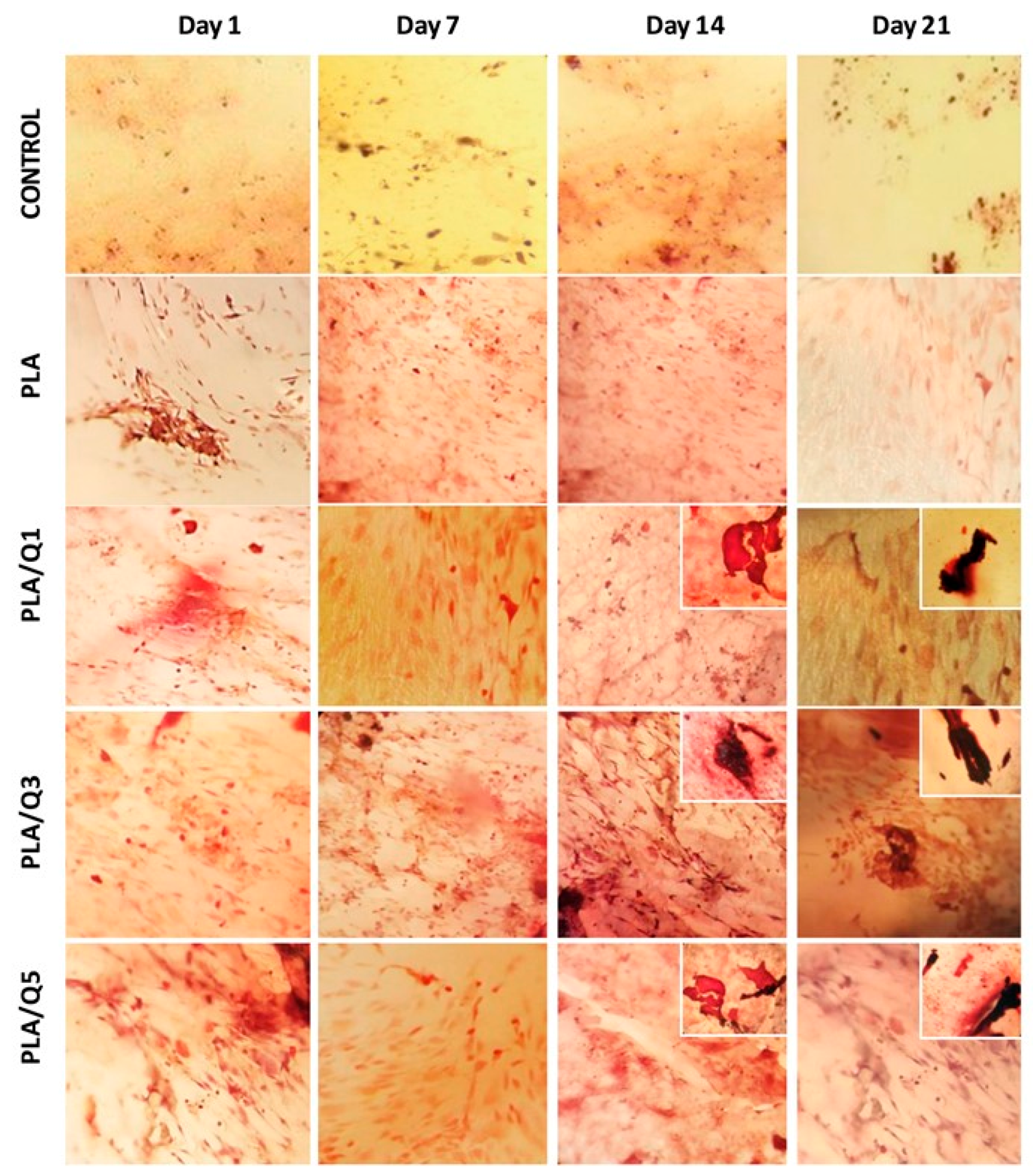
3.4.4. Qualitative Evaluation of Mineral Deposition on PLA/Chitosan Surfaces
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rodríguez-Vázquez, M.; Vega-Ruiz, B.; Ramos-Zúñiga, R.; Alexander Saldaña-Koppel, D.; Quiñones-Olvera, L.F. Chitosan and Its Potential Use as a Scaffold for Tissue Engineering in Regenerative Medicine. BioMed Res. Int. 2015, 2015, 821279. [Google Scholar] [CrossRef] [PubMed]
- Amini, A.; Laurencin, C.; Nukavarapu, S. Bone Tissue Engineering: Recent Advances and Challenges. Crit. Rev. Biomed. Eng. 2012, 40, 363–408. [Google Scholar] [CrossRef] [PubMed]
- Vacanti, J.; Langer, R. Tissue engineering: The design and fabrication of living replacement devices for surgical reconstruction and transplantation. Lancet 1999, 354, S32–S34. [Google Scholar] [CrossRef]
- Freed, L.E.; Vunjak-Novakovic, G.; Biron, R.J.; Eagles, D.B.; Lesnoy, D.C.; Barlow, S.K.; Langer, R. Biodegradable polymer scaffolds for tissue engineering. Nat. Biotechnol. 1994, 12, 689. [Google Scholar] [CrossRef]
- Razak, S.I.A.; Sharif, N.F.A.; Rahman, W.A.W.A. Biodegradable Polymers and their Bone Applications: A Review. Int. J. Basic Appl. Sci. 2012, 12, 31–49. [Google Scholar]
- Tanase, C.E.; Spiridon, I. PLA/Chitosan/keratin composites for biomedical applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 40, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Rasal, R.M.; Janorkar, A.V.; Hirt, D.E. Poly(lactic acid) modifications. Prog. Polym. Sci. 2010, 35, 338–356. [Google Scholar] [CrossRef]
- Balakrishnan, H.; Hassan, A.; Wahit, M.U.; Yussuf, A.A.; Razak, S.B.A. Novel toughened polylactic acid nanocomposite: Mechanical, thermal and morphological properties. Mater. Des. 2010, 31, 3289–3298. [Google Scholar] [CrossRef]
- Carrasco, P.; Pagès, J.; Gámez-Pérez, O.O.; Santana, M.L. Processing of poly(lactic acid): Characterization of chemical structure, thermal stability and mechanical properties. Polym. Degrad. Stab. 2010, 95, 116–125. [Google Scholar] [CrossRef]
- Hamad, K.; Kaseem, M.; Yang, H.W.; Deri, F.; Ko, Y.G. Properties and medical applications of polylactic acid: A review. Express Polym. Lett. 2015, 9, 435–455. [Google Scholar] [CrossRef]
- Rezwan, K.; Chen, Q.Z.; Blaker, J.J.; Boccaccini, A.R. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials 2006, 27, 3413–3431. [Google Scholar] [CrossRef] [PubMed]
- Scaffaro, R.; Lopresti, F.; Botta, L.; Maio, A. Mechanical behavior of polylactic acid/polycaprolactone porous layered functional composites. Compos. Part B Eng. 2016, 98, 70–77. [Google Scholar] [CrossRef]
- Bleacha, N.C.; Nazhata, S.N.; Tannera, K.E.; Kellom, M.; Tormala, P. Effect of filler content on mechanical and dynamic mechanical properties of particulate biphasic calcium phosphate-polylactide composites. Biomaterials 2002, 7, 1579–1585. [Google Scholar] [CrossRef]
- Xiao, L.; Wang, B.; Yang, G.; Gauthier, M. Poly(Lactic Acid)-Based Biomaterials: Synthesis, Modification and Applications. In Biomedical Science, Engineering and Technology; IntechOpen: London, UK, 2012. [Google Scholar] [CrossRef]
- Danoux, C.B.; Barbieri, D.; Yuan, H.; Bruijn, J.D.; Blitterswijk, C.A.; Habibovic, P. In vitro and in vivo bioactivity assessment of a polylactic acid/hydroxyapatite composite for bone regeneration. Biomatter 2014, 4, 2159–2535. [Google Scholar] [CrossRef] [PubMed]
- Busscher, H.J.; Engels, E.; Dijkstra, R.J.B.; Van Der Mei, H.C. Influence of a chitosan on oral bacterial adhesion and growth in vitro. Eur. J. Oral Sci. 2008, 116, 493–495. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, H.; Wang, Z.; Wen, D. Porous conductive chitosan scaffolds for tissue engineering II, in vitro and in vivo degradation. J. Mater. Sci. Mater. Med. 2005, 16, 1017–1028. [Google Scholar]
- Rinaudo, M. Chitin and chitosan: Properties and application. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Kumar, M.N.R. A review of chitin and chitosan applications. React. Funct. Polym. 2000, 46, 1–27. [Google Scholar] [CrossRef]
- Percot, A.; Viton, C.; Domard, A. Optimization of chitin extraction from shrimp shells. Biomacromolecules 2003, 4, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Kandra, P.; Mohan, M.; Padma, J.H.K. Efficient use of shrimp waste: Present and future trends. Appl. Microbiol. Biotechnol. 2012, 93, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Cui, H. Biodegradability and Biocompatibility Study of Poly(Chitosan-g-lactic Acid) Scaffolds. Molecules 2012, 17, 3243–3258. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.; Feng, Q.; Wang, M.; Guo, X.; Zheng, Q. In vitro degradation and release behavior of porous poly(lactic acid) scaffolds containing chitosan microspheres as a carrier for BMP-2-derived synthetic peptide. Polym. Degrad. Stab. 2009, 94, 176–182. [Google Scholar] [CrossRef]
- Wan, Y.; Wu, H.; Yu, A.; Wen, D. Biodegradable Polylactide/Chitosan Blend Membranes. Biomacromolecules 2006, 7, 1362–1372. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, J.; Fortunati, E.; Vargas, M.; Chiralt, A.; Kenny, J.M. Effects of chitosan on the physicochemical and antimicrobial properties of PLA films. J. Food Eng. 2013, 119, 236–243. [Google Scholar] [CrossRef]
- Almeida, C.R.; Serra, T.; Oliveira, M.I.; Planell, J.A.; Barbosa, M.A.; Navarro, M. Impact of 3-D printed PLA- and chitosan-based scaffolds on human monocyte/macrophage responses: Unraveling the effect of 3-D structures on inflammation. Acta Biomater. 2014, 10, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ma, P.X. Polymeric Scaffolds for Bone Tissue Engineering. Ann. Biomed. Eng. 2004, 32, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Leong, K.F.; Du, Z.; Chua, C.K. The Design of Scaffolds for Use in Tissue Engineering. Part II. Rapid Prototyping Techniques. Tissue Eng. 2002, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Murariu, M.; Dubois, P. PLA composites: From production to properties. Adv. Drug Deliv. Rev. 2016, 107, 17–46. [Google Scholar] [CrossRef] [PubMed]
- Standard Test Method for Tensile Properties of Plastics-1, ASTM Standard D-638-10; ASTM International: West Conshohocken, PA, USA, 2010.
- Correlo, V.M.; Boesel, L.F.; Bhattacharya, M.; Mano, J.F.; Neves, N.M.; Reis, R.L. Properties of melt processed chitosan and aliphatic polyester blends. Mater. Sci. Eng. 2005, 403, 57–68. [Google Scholar] [CrossRef]
- Entsar, S.; Khaled, S.A.; Elsabee, M.Z. Extraction and characterization of chitin and chitosan from local sources. Bioresour. Technol. 2008, 99, 1359–1367. [Google Scholar]
- Liu, X.; Khor, S.; Petinaki, E.; Yu, L.; Simon, G.P.; Dean, K.M.; Bateman, S. Effects of hydrophilic fillers on the thermal degradation of poly(lactic acid). Thermochimica 2010, 509, 147–151. [Google Scholar] [CrossRef]
- Duarte, M.L.; Ferreira, M.C.; Marvão, M.R.; Rocha, J. An optimized method to determine the degree of acetylation of chitin and chitosan by FTIR spectroscopy. Int. J. Biol. Macromol. 2002, 31, 1–8. [Google Scholar] [CrossRef]
- Ahmed, J.; Mulla, M.; Arfat, Y.A. Mechanical, thermal, structural and barrier properties of crab shell chitosan/graphene oxide composite films. Food Hydrocoll. 2017, 71, 141–148. [Google Scholar] [CrossRef]
- Mi, H.Y.; Salick, M.R.; Jing, X.; Jacques, B.R.; Crone, W.C.; Peng, X.F.; Turng, L.S. Characterization of thermoplastic polyurethane/polylactic acid (TPU/PLA) tissue engineering scaffolds fabricated by microcellular injection molding. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 4767–4776. [Google Scholar] [CrossRef] [PubMed]
- Shoufeng, Y.; Leong, K.F.; Du, Z.; Chua, C.K. The design of scaffolds for use in tissue engineering. Part I. Traditional Factors. Tissue Eng. 2001, 6, 679–689. [Google Scholar]
- Di Martino, A.; Sittinger, M.; Risbud, M.V. Chitosan: A versatile biopolymer for orthopaedic tissue-engineering. Biomaterials 2015, 26, 5983–5990. [Google Scholar] [CrossRef] [PubMed]
- Bhumkar, D.R.; Pokharkar, V.B. Studies on effect of pH on cross-linking of chitosan with sodium tripolyphosphate: A technical note. AAPS PharmSciTech 2006, 7, E138–E143. [Google Scholar] [CrossRef] [PubMed]
- Cai, K.; Yao, K.; Cui, Y.L.; Lin, S.; Yang, Z.; Li, X.; Xie, H.; Qing, T.; Luo, J. Surface modification of poly(d,l-lactic acid) with chitosan and its effects on the culture of osteoblasts in vitro. J. Biomed. Mater. Res. 2002, 60, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.H.; Liao, M.H.; Lin, Y.L.; Lai, C.H.; Lin, P.I.; Chen, R.M. Improving effects of chitosan nanofiber scaffolds on osteoblast proliferation and maturation. Int. J. Nanomed. 2014, 9, 4293–4304. [Google Scholar]
- Wan, Y.; Xiao, Y.C.; Zhang, S.; Wang, S.; Wu, Q. Fibrous poly(chitosan-g-dl-lactic acid) scaffolds prepared via electro-wet-spinning. Acta Biomater. 2008, 4, 876–886. [Google Scholar] [CrossRef] [PubMed]
- Wurm, M.; Moest, T.; Bergauer, B.; Rietzel, D.; Wilhelm, N.F.; Cifuentes, S.; Wilmowsky, C. In-vitro evaluation of Polylactic acid (PLA) manufactured by fused deposition modeling. J. Biol. Eng. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, G.; Liu, L.; Zhang, D. The mechanism of a chitosan-collagen composite film used as biomaterial support for MC3T3-E1 cell differentiation. Sci. Rep. 2016, 6, 39322. [Google Scholar] [CrossRef] [PubMed]
- Lai, G.J.; Shalumon, K.T.; Chen, S.H.; Chen, J.P. Composite chitosan/silk fibroin nanofibers for modulation of osteogenic differentiation and proliferation of human mesenchymal stem cells. Carbohydr. Polym. 2014, 111, 288–297. [Google Scholar] [CrossRef] [PubMed]
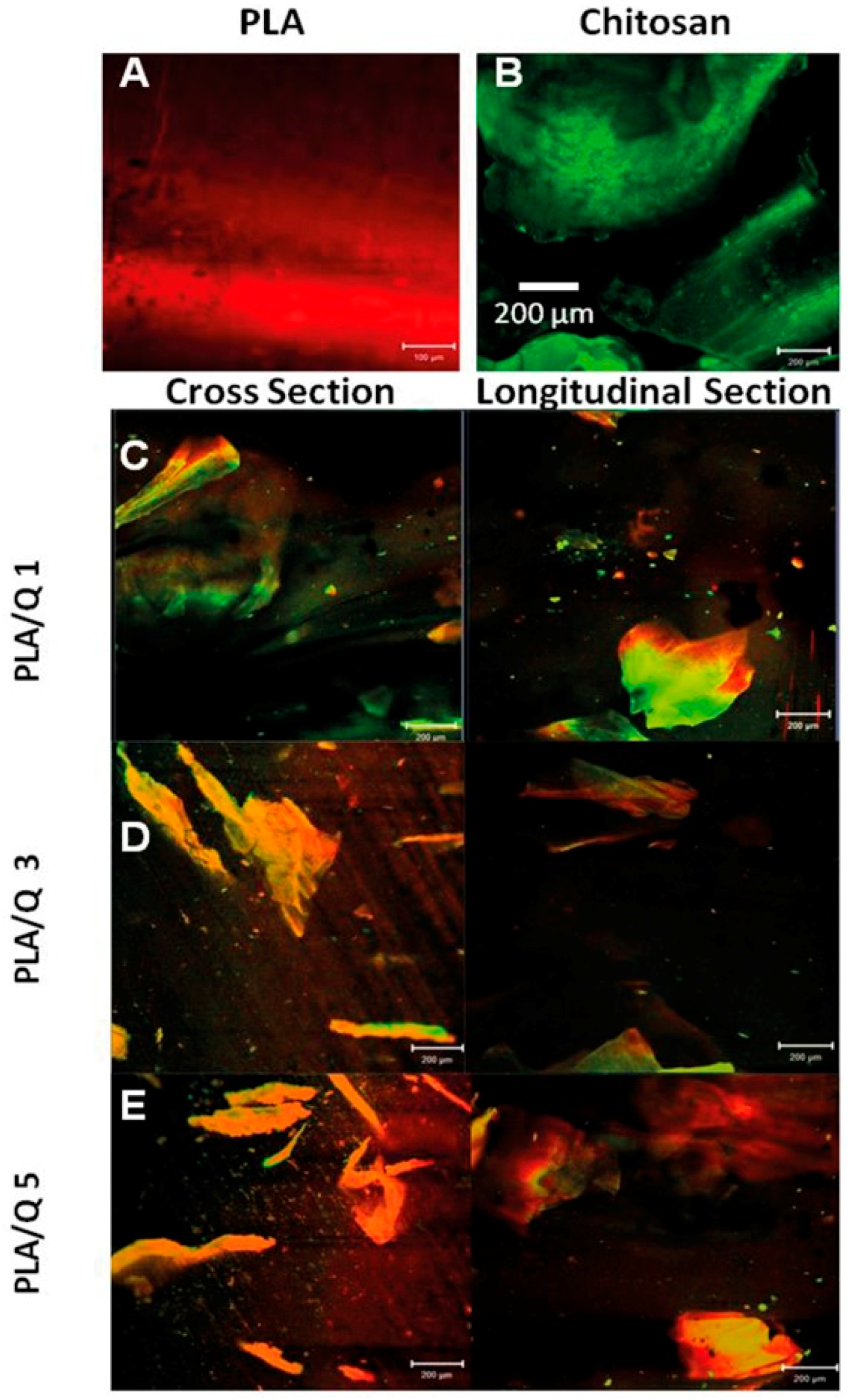
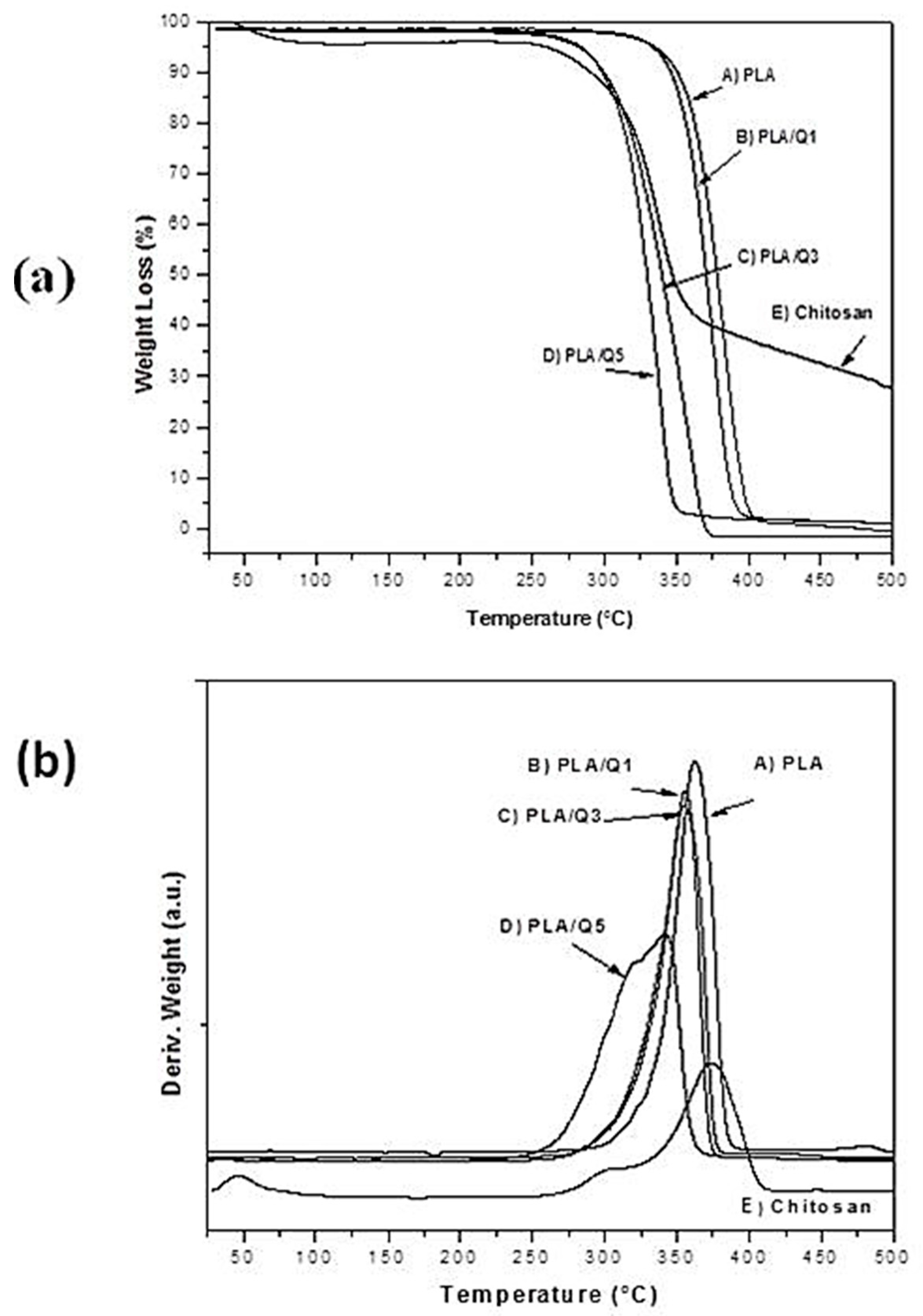
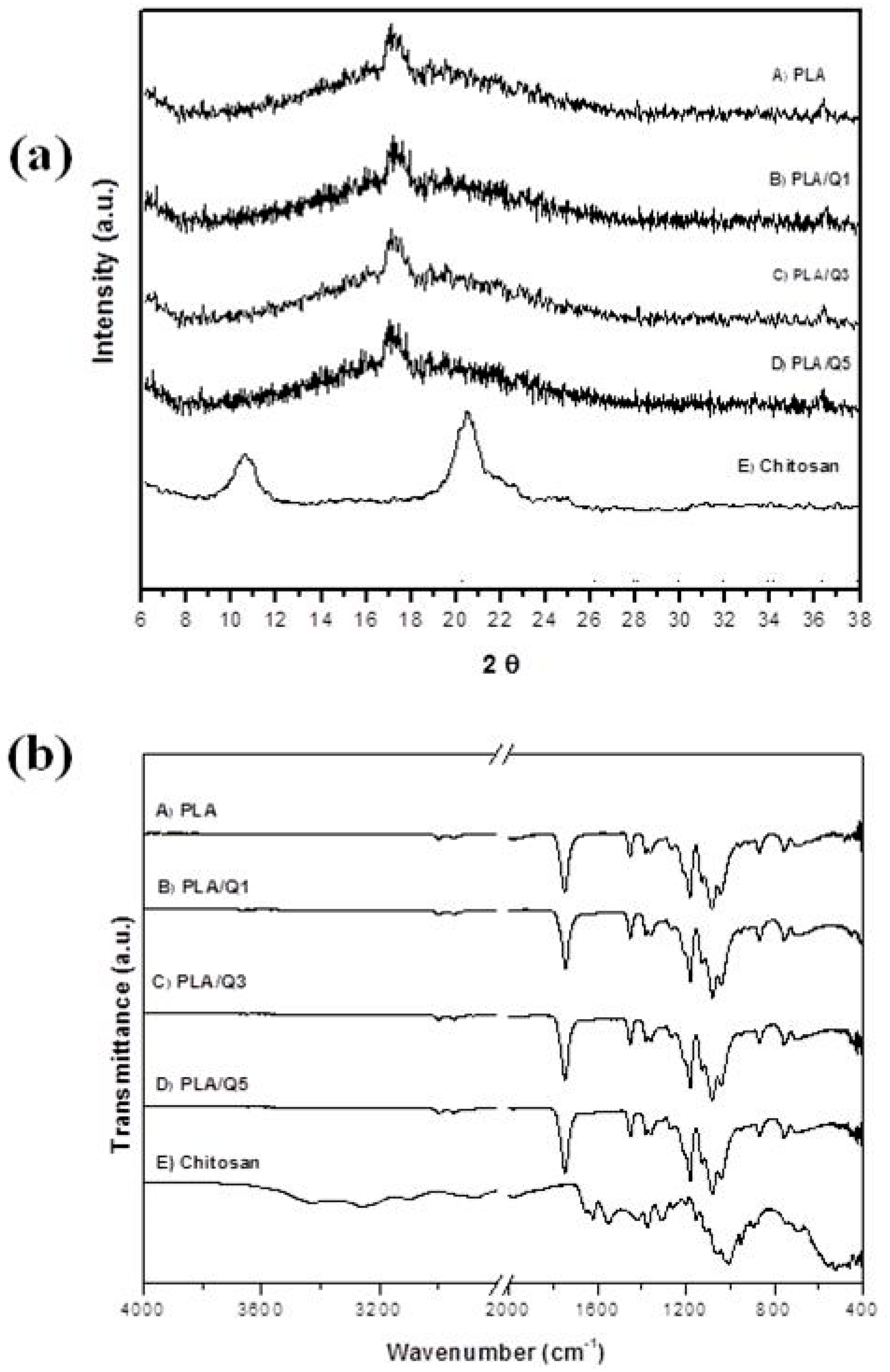
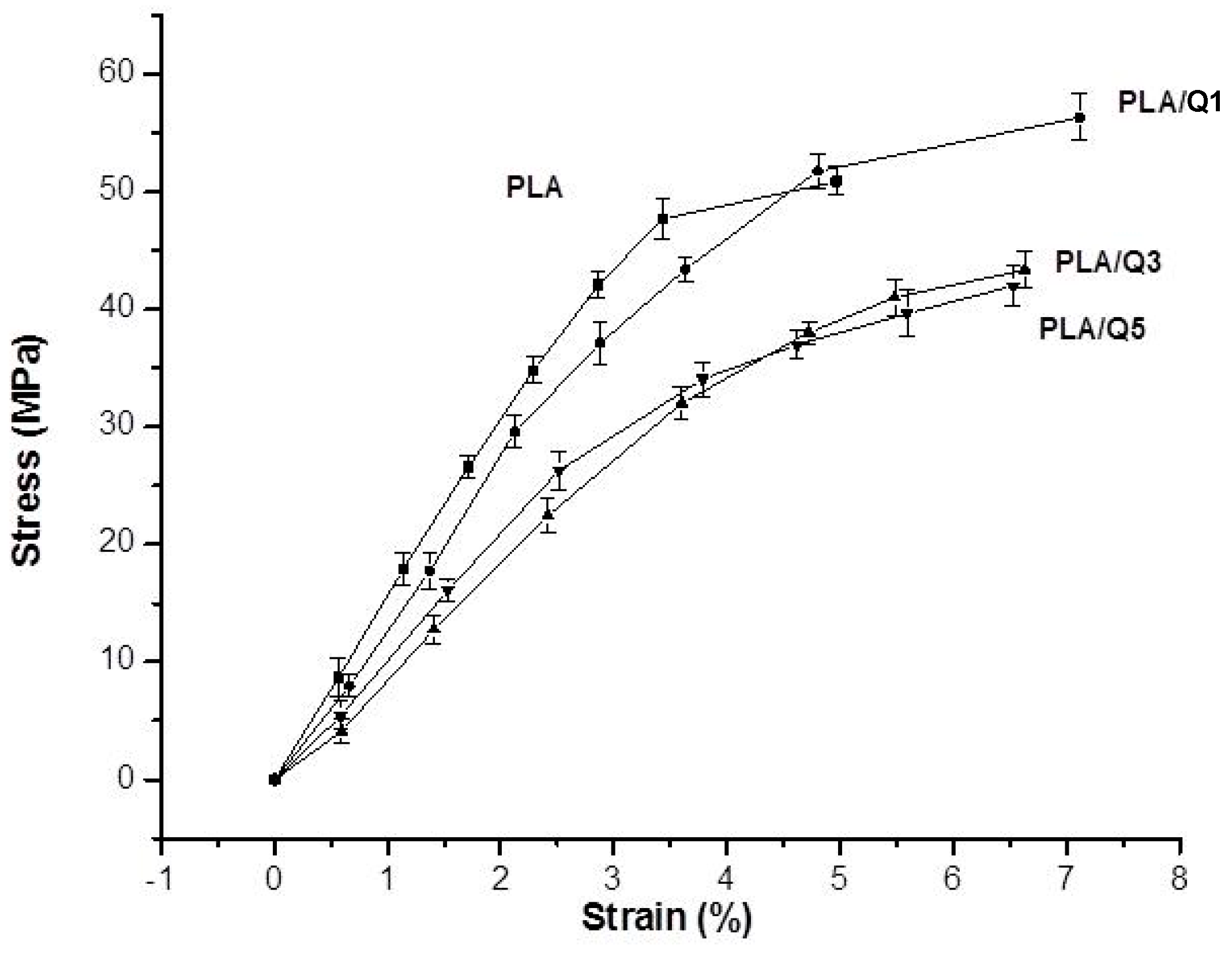
| Sample | Amount of Chitosan (wt %) | Tensile Strenght (MPa) | Young’s Modulus (MPa) | Elongation at Break (%) |
|---|---|---|---|---|
| PLA | – | 49.36 ± 4.44 | 1252 ± 62 | 4.97 ± 0.76 |
| PLA/Q 1 | 1 | 56.34 ± 2.64 | 1110 ± 66 | 7.12 ± 0.63 |
| PLA/Q 3 | 3 | 43.38 ± 3.29 | 983 ± 55 | 6.63 ± 0.54 |
| PLA/Q 5 | 5 | 42.19 ± 2.55 | 1051 ± 34 | 6.52 ± 0.86 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres-Hernández, Y.G.; Ortega-Díaz, G.M.; Téllez-Jurado, L.; Castrejón-Jiménez, N.S.; Altamirano-Torres, A.; García-Pérez, B.E.; Balmori-Ramírez, H. Biological Compatibility of a Polylactic Acid Composite Reinforced with Natural Chitosan Obtained from Shrimp Waste. Materials 2018, 11, 1465. https://doi.org/10.3390/ma11081465
Torres-Hernández YG, Ortega-Díaz GM, Téllez-Jurado L, Castrejón-Jiménez NS, Altamirano-Torres A, García-Pérez BE, Balmori-Ramírez H. Biological Compatibility of a Polylactic Acid Composite Reinforced with Natural Chitosan Obtained from Shrimp Waste. Materials. 2018; 11(8):1465. https://doi.org/10.3390/ma11081465
Chicago/Turabian StyleTorres-Hernández, Yaret Gabriela, Gloria Michel Ortega-Díaz, Lucía Téllez-Jurado, Nayeli Shantal Castrejón-Jiménez, Alejandro Altamirano-Torres, Blanca Estela García-Pérez, and Heberto Balmori-Ramírez. 2018. "Biological Compatibility of a Polylactic Acid Composite Reinforced with Natural Chitosan Obtained from Shrimp Waste" Materials 11, no. 8: 1465. https://doi.org/10.3390/ma11081465
APA StyleTorres-Hernández, Y. G., Ortega-Díaz, G. M., Téllez-Jurado, L., Castrejón-Jiménez, N. S., Altamirano-Torres, A., García-Pérez, B. E., & Balmori-Ramírez, H. (2018). Biological Compatibility of a Polylactic Acid Composite Reinforced with Natural Chitosan Obtained from Shrimp Waste. Materials, 11(8), 1465. https://doi.org/10.3390/ma11081465





