Sinus Membrane Elevation with Heterologous Cortical Lamina: A Randomized Study of a New Surgical Technique for Maxillary Sinus Floor Augmentation without Bone Graft
Abstract
1. Introduction
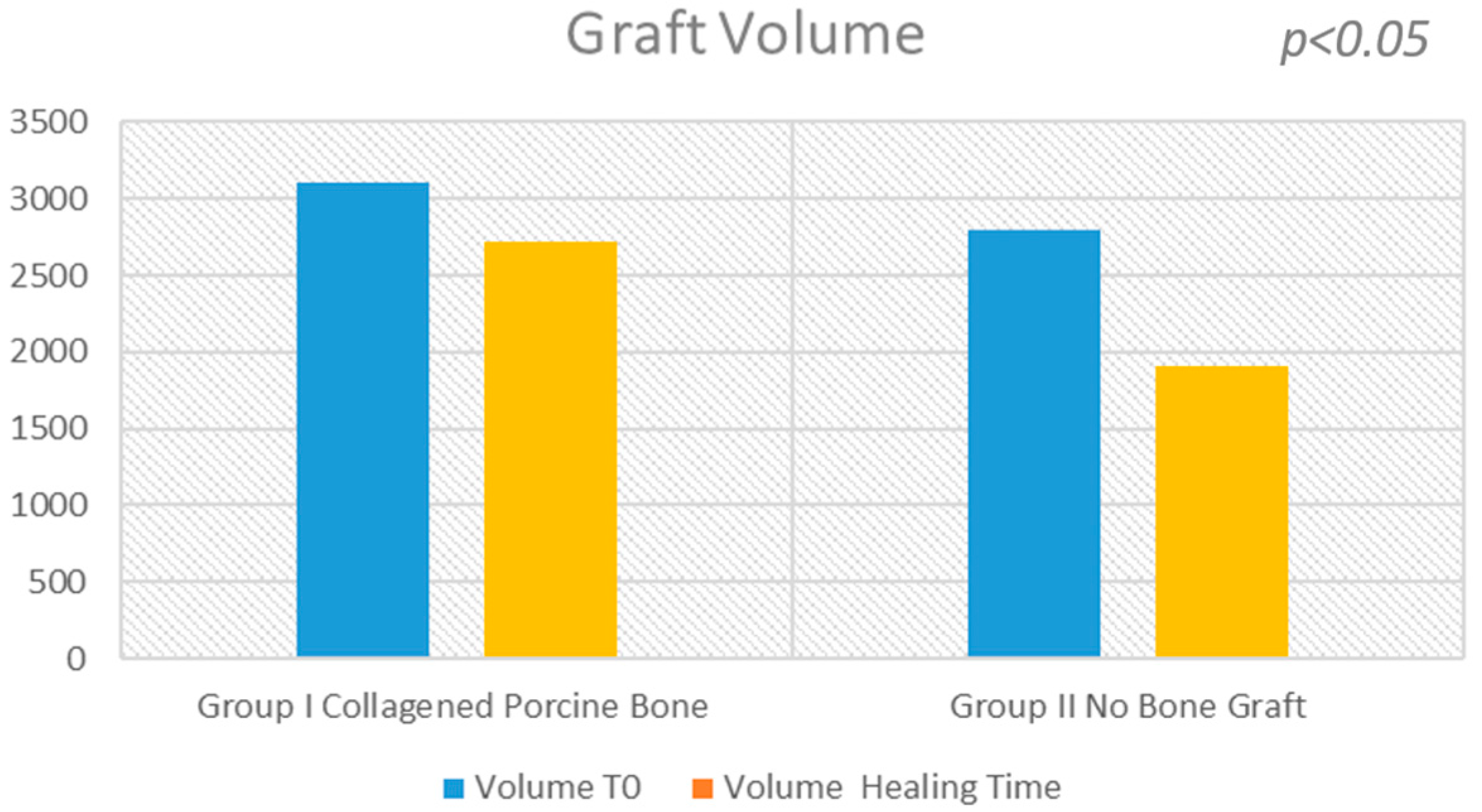
2. Results
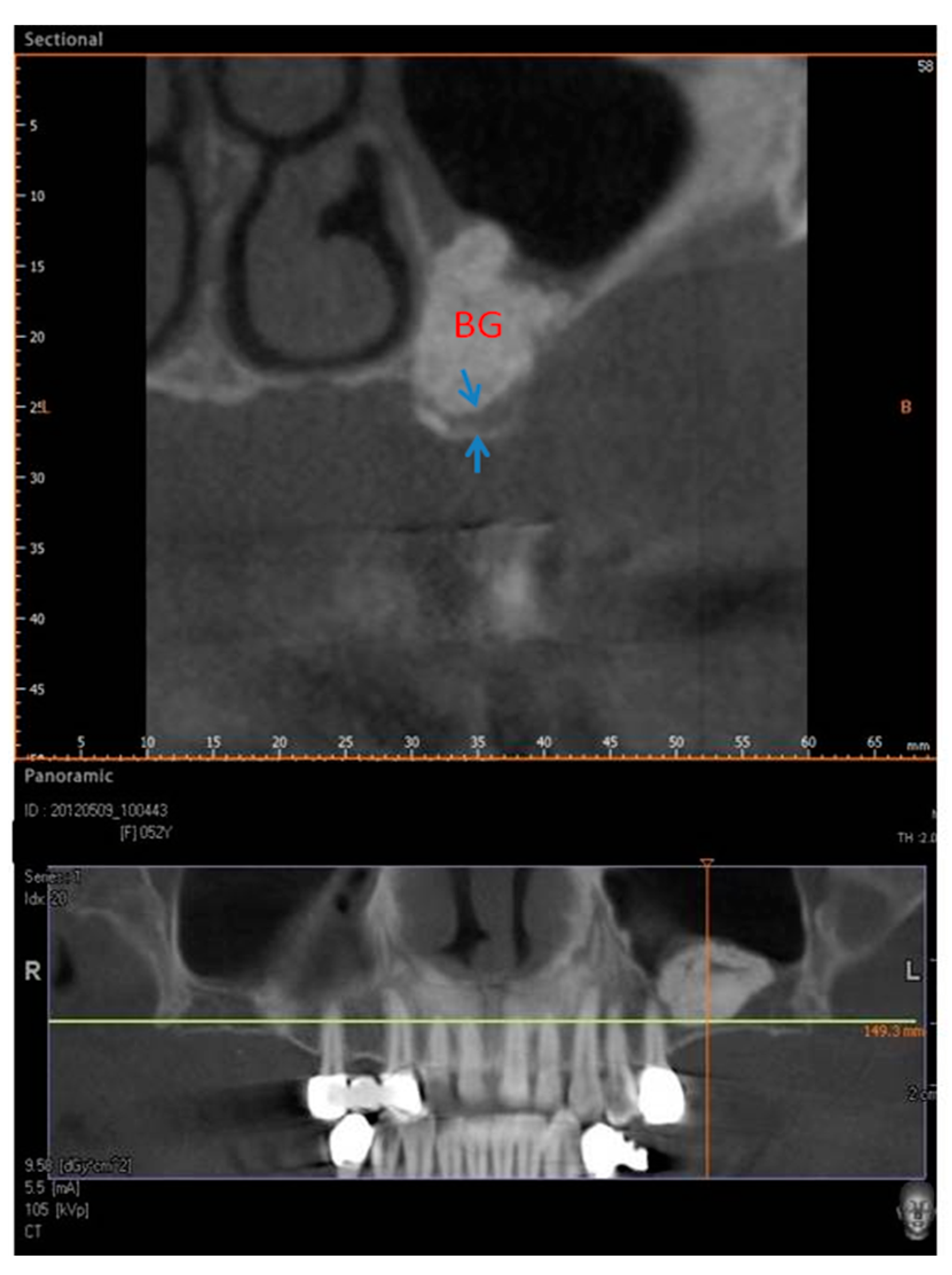
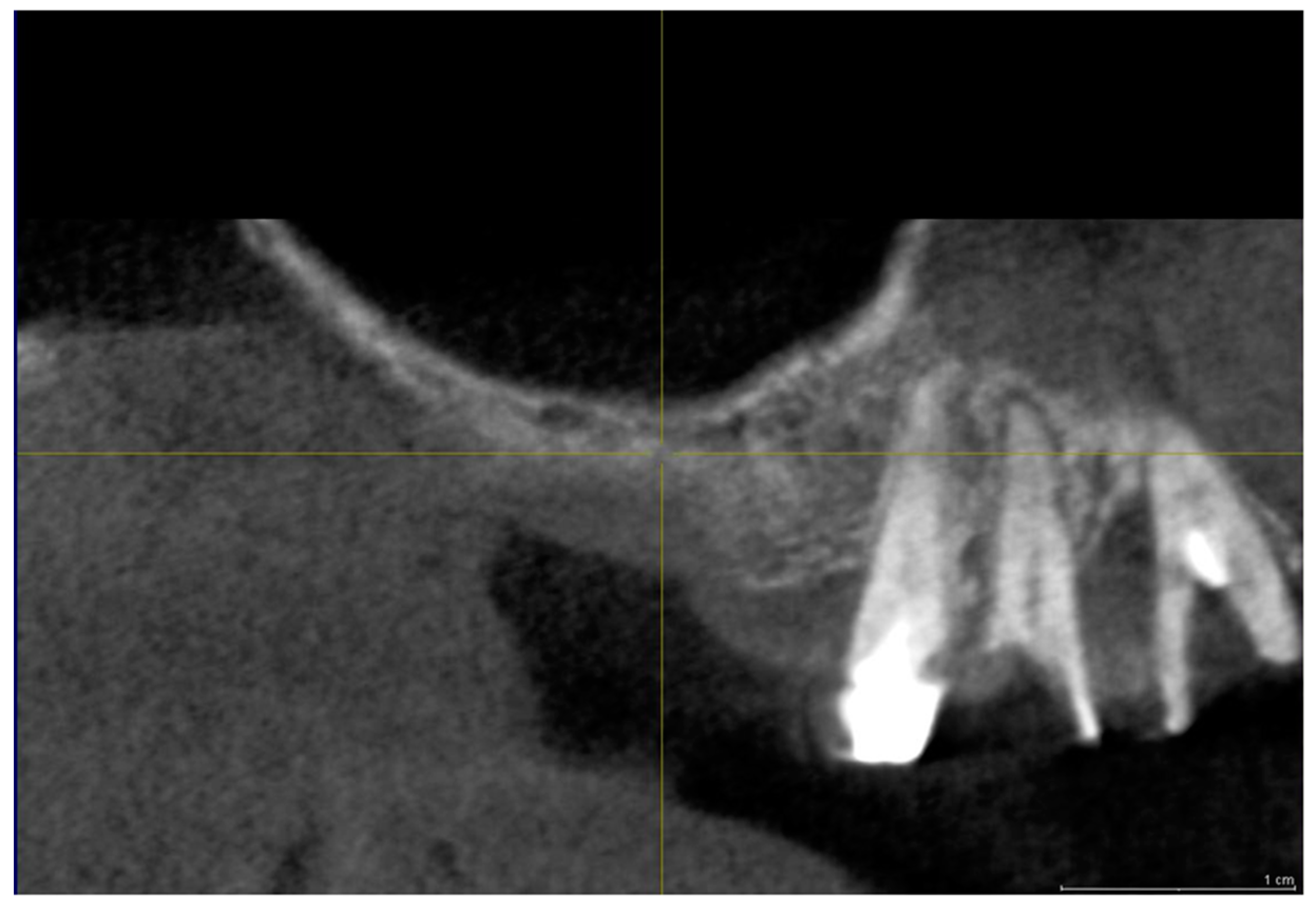
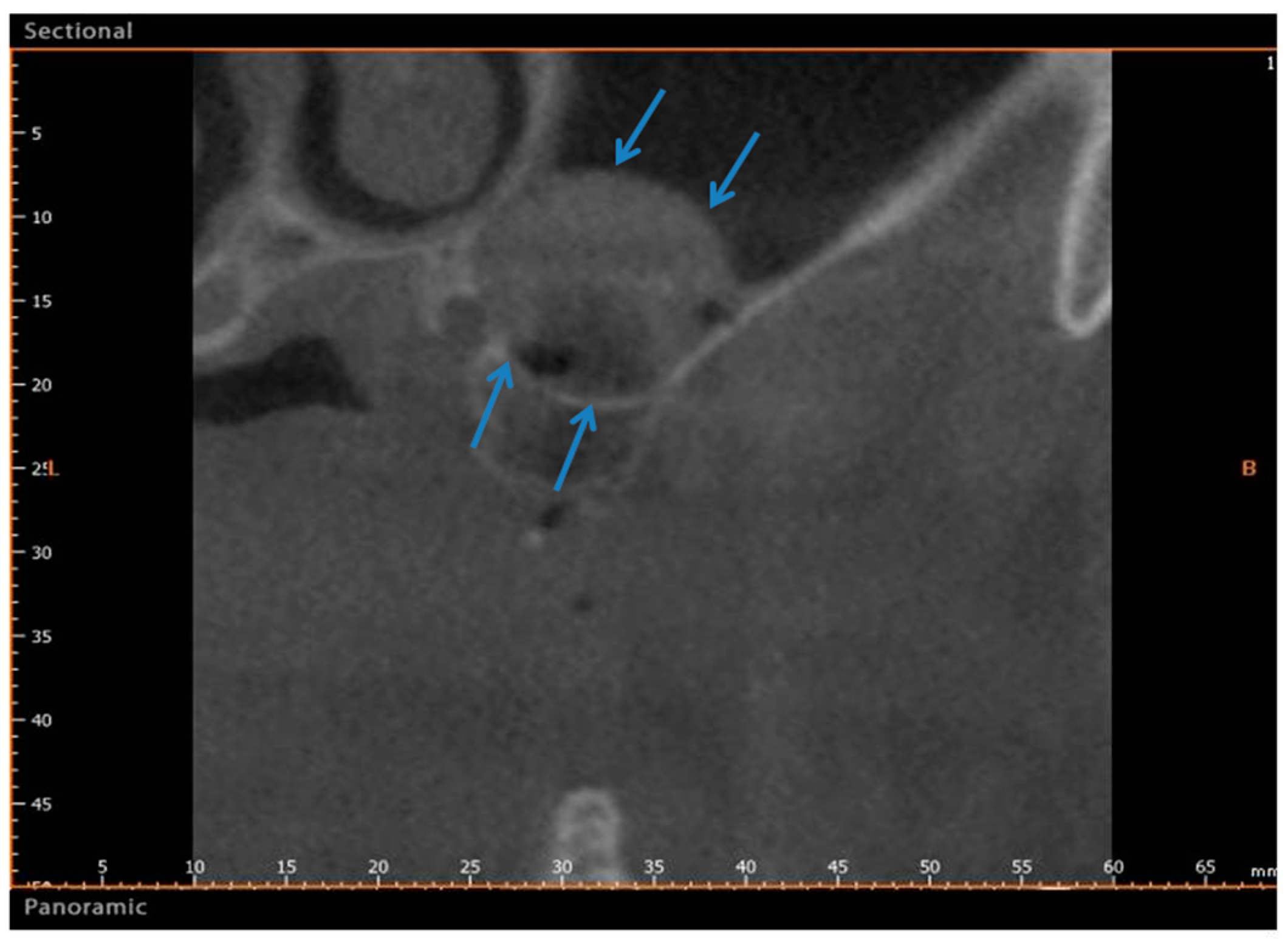
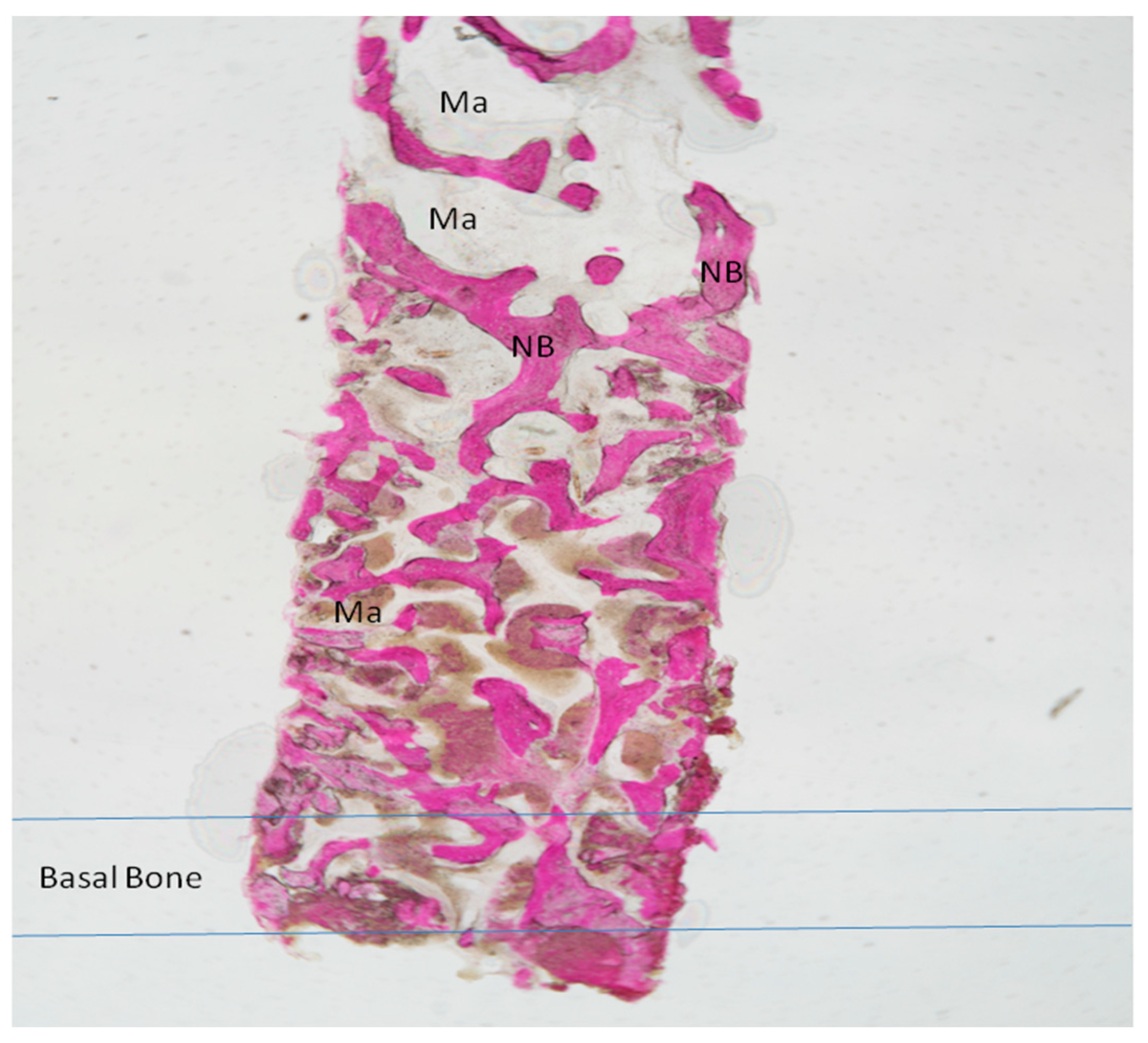
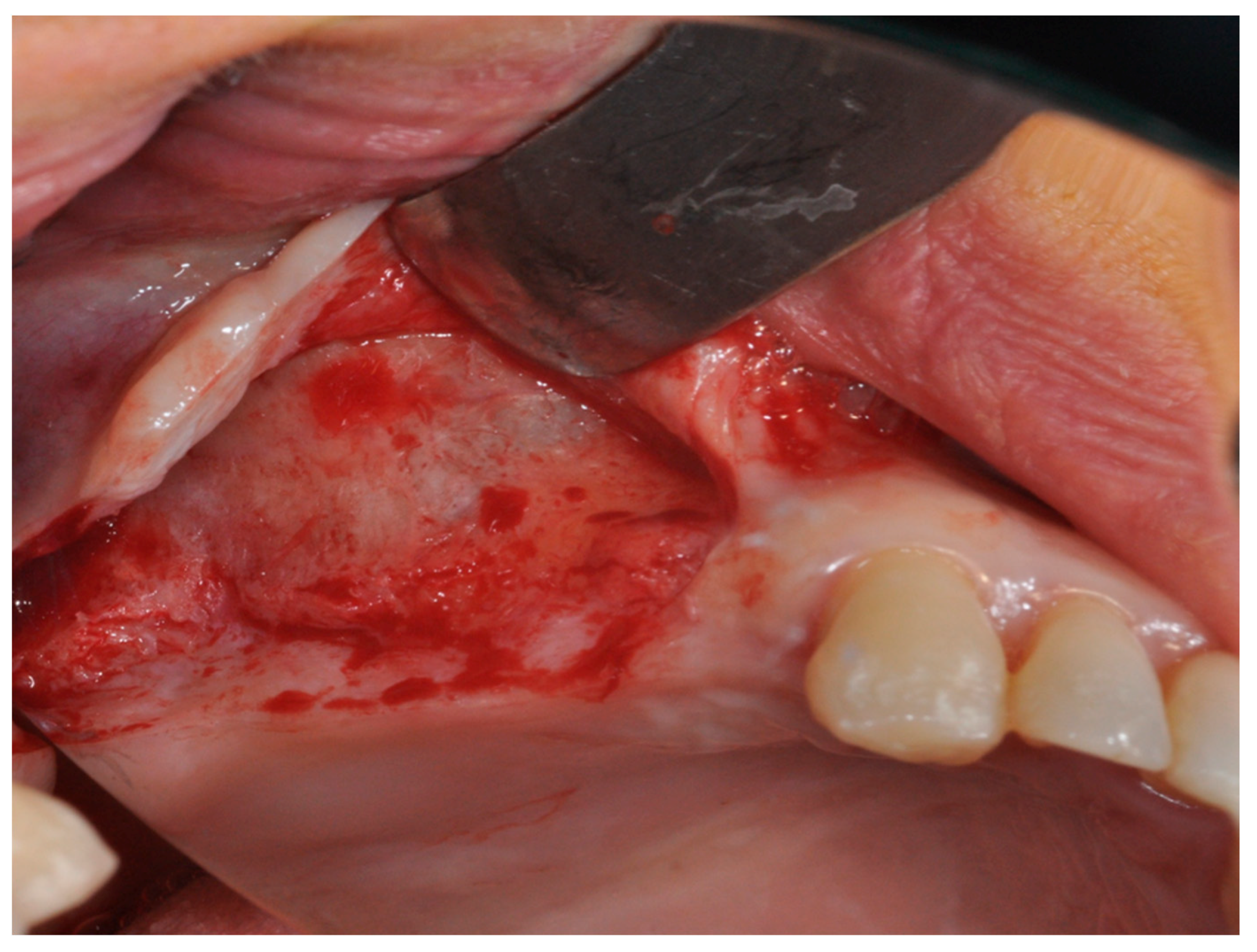
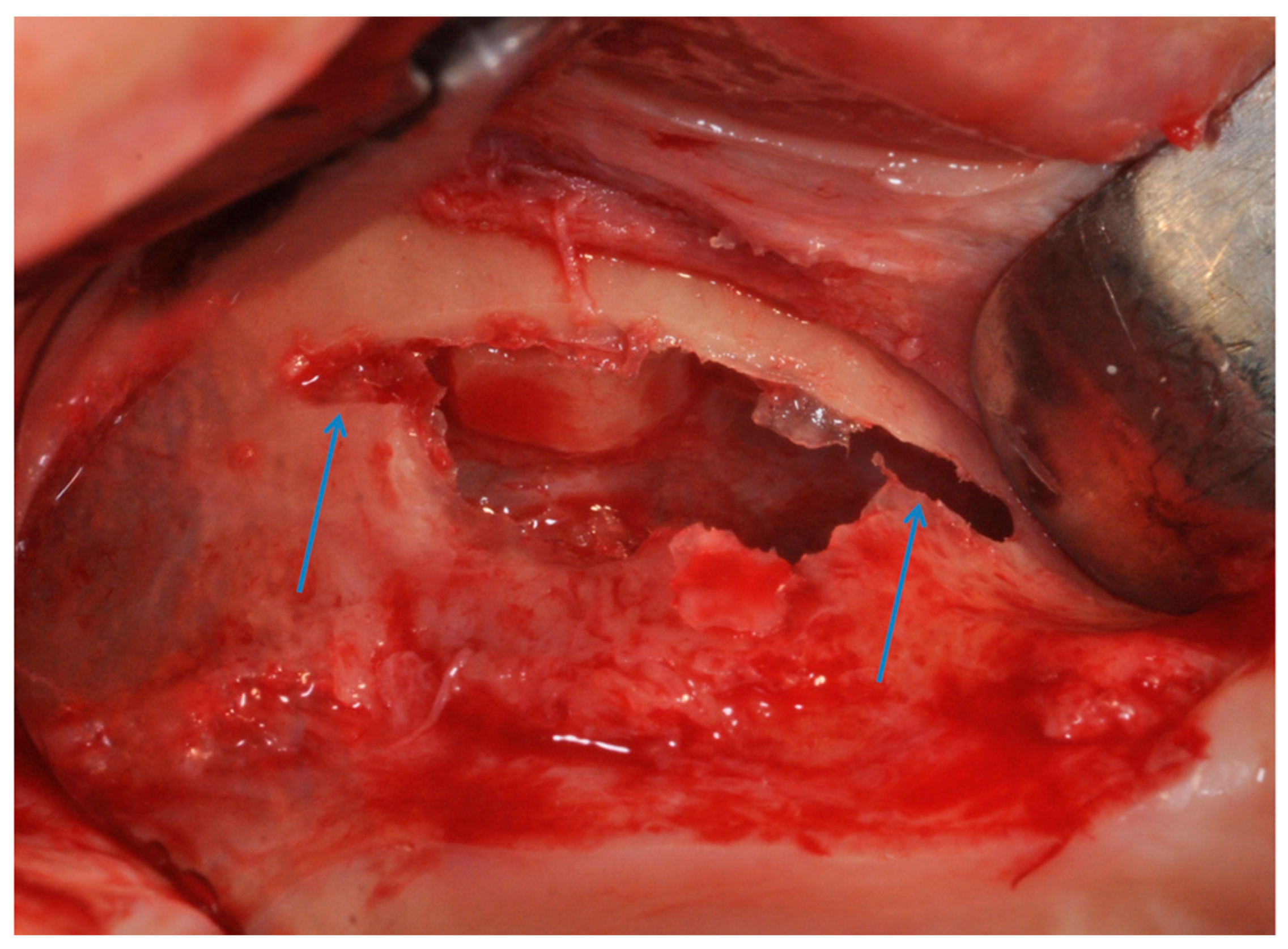
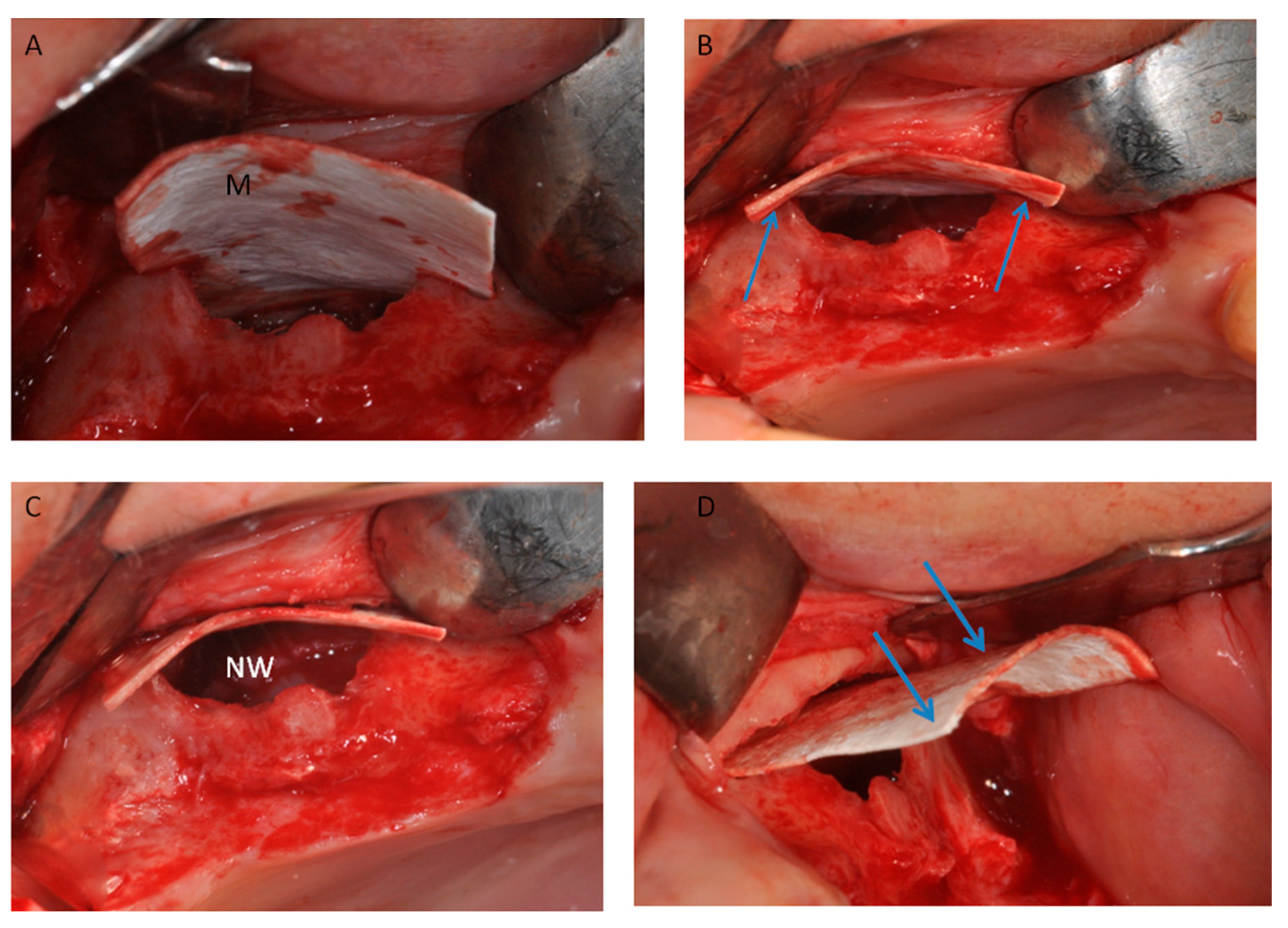
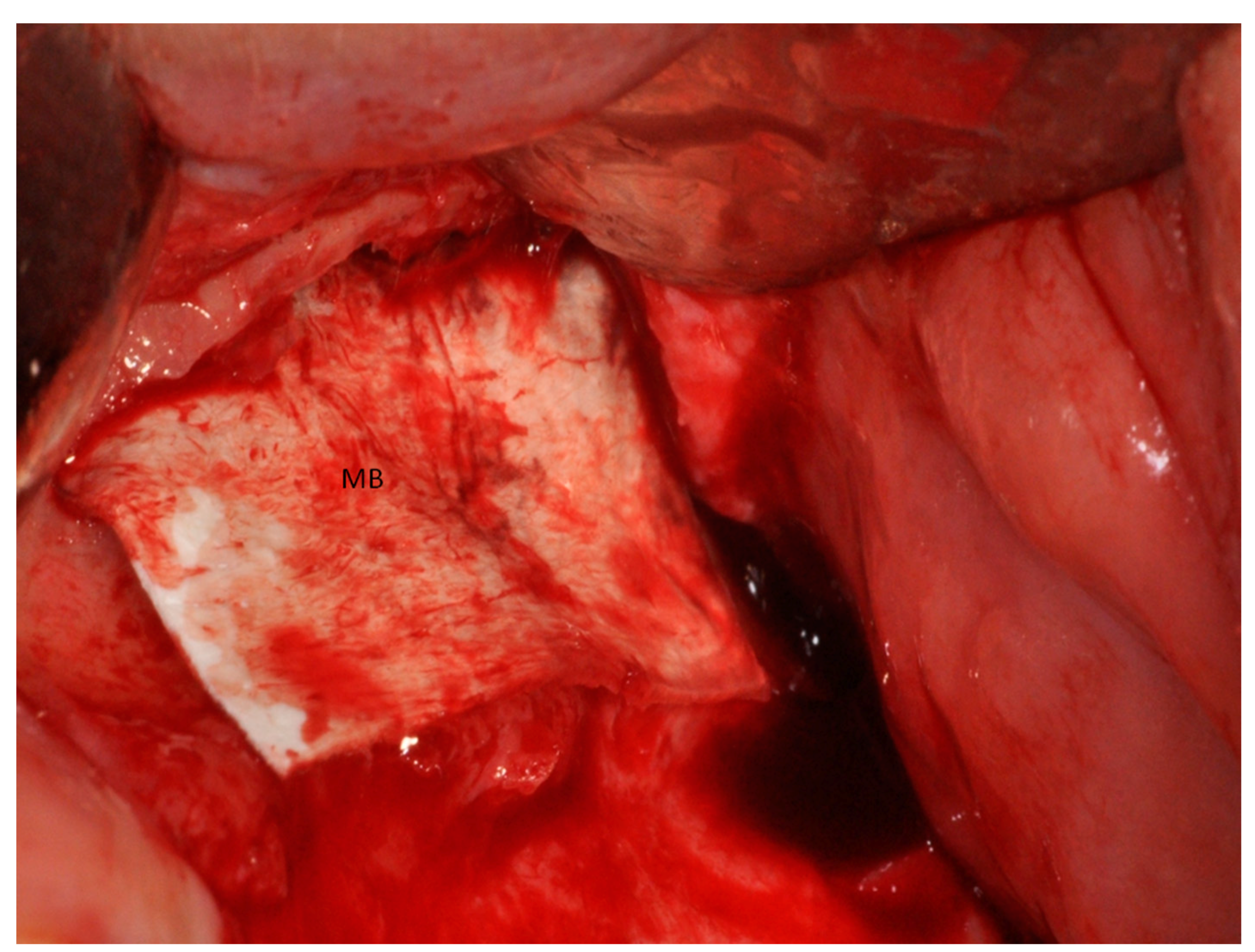
2.1. Group I
2.2. Group II
2.3. Statistical Analysis
3. Discussion
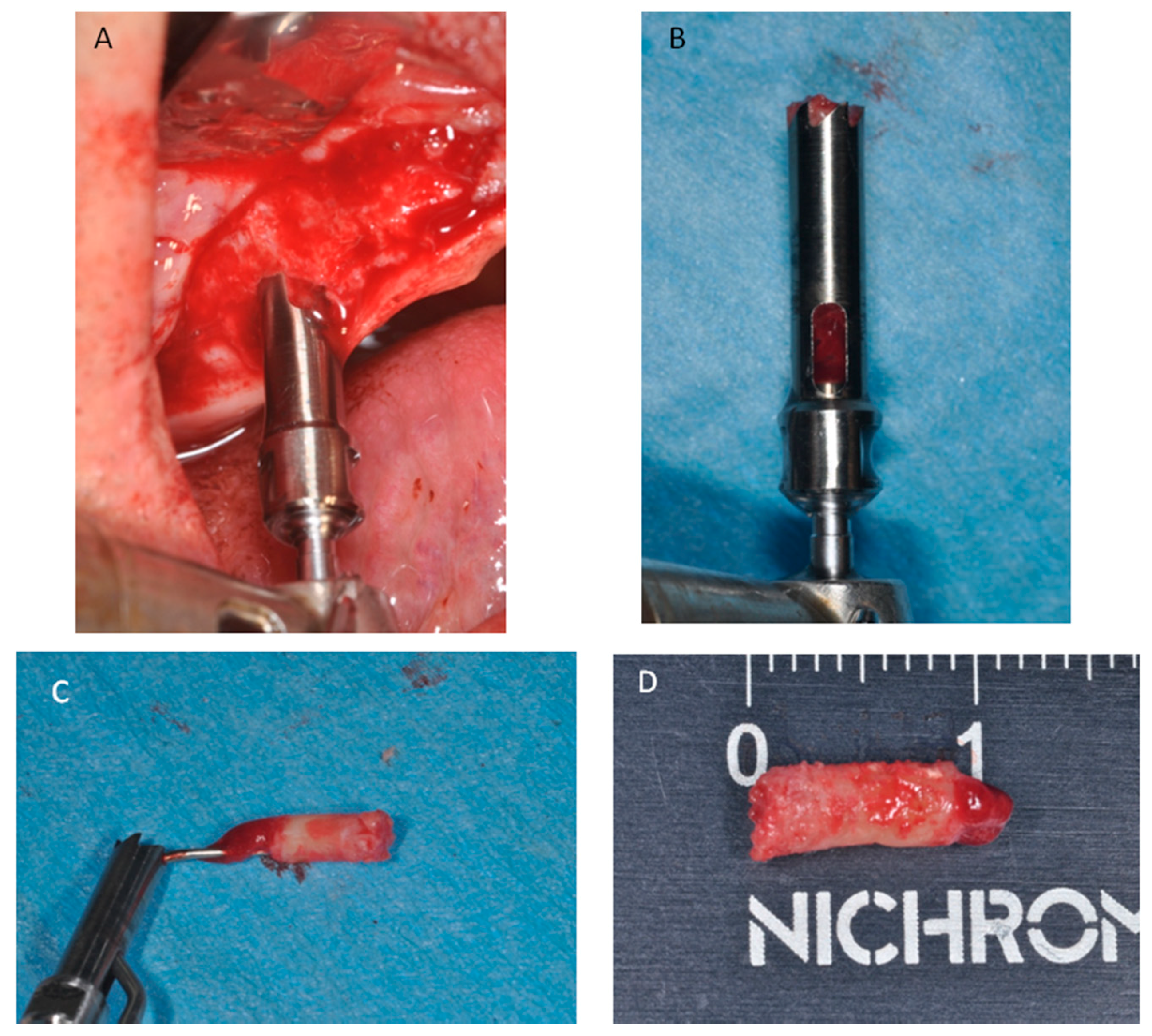
4. Materials and Methods
Processing of Specimens
5. Statistical Evaluation
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Scarano, A.; Perrotti, V.; Carinci, F.; Shibli, J.A. Removal of a migrated dental implant from the maxillary sinus after 7 years: A case report. Oral Maxillofac. Surg. 2011, 15, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Fürst, G.; Gruber, R.; Tangl, S.; Zechner, W.; Haas, R.; Mailath, G.; Sanroman, F.; Watzek, G. Sinus grafting with autogenous platelet-rich plasma and bovine hydroxyapatite. A histomorphometric study in minipigs. Clin. Oral Implants Res. 2003, 14, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Tatum, H. Maxillary and sinus implant reconstructions. Dent. Clin. North. Am. 1986, 30, 207–229. [Google Scholar] [PubMed]
- Boyne, P.J.; James, R.A. Grafting of the maxillary sinus floor with autogenous marrow and bone. J. Oral Surg. 1980, 38, 613–616. [Google Scholar] [PubMed]
- Scarano, A.; Piattelli, A.; Assenza, B.; Quaranta, A.; Perrotti, V.; Piattelli, M.; Iezzi, G. Porcine bone used in sinus augmentation procedures: A 5-year retrospective clinical evaluation. J. Oral Maxillofac. Surg. 2010, 68, 1869–1873. [Google Scholar] [CrossRef] [PubMed]
- Nannmark, U.; Sennerby, L. The bone tissue responses to prehydrated and collagenated cortico-cancellous porcine bone grafts: A study in rabbit maxillary defects. Clin. Implant Dent. Relat. Res. 2008, 10, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Mangano, C.; Scarano, A.; Perrotti, V.; Iezzi, G.; Piattelli, A. Maxillary sinus augmentation with a porous synthetic hydroxyapatite and bovine-derived hydroxyapatite: A comparative clinical and histologic study. Int. J. Oral Maxillofac. Implants 2007, 22, 980–986. [Google Scholar] [PubMed]
- Scarano, A.; Degidi, M.; Iezzi, G.; Pecora, G.; Piattelli, M.; Orsini, G.; Caputi, S.; Perrotti, V.; Mangano, C.; Piattelli, A. Maxillary sinus augmentation with different biomaterials: A comparative histologic and histomorphometric study in man. Implant Dent. 2006, 15, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Inchingolo, F.; Murmura, G.; Traini, T.; Piattelli, A.; Lorusso, F. Three-Dimensional Architecture and Mechanical Properties of Bovine Bone Mixed with Autologous Platelet Liquid, Blood, or Physiological Water: An In Vitro Study. Int. J. Mol. Sci. 2018, 19, 1230. [Google Scholar] [CrossRef] [PubMed]
- Froum, S.J.; Tarnow, D.P.; Wallace, S.S.; Rohrer, M.D.; Cho, S.C. Sinus floor elevation using anorganic bovine bone matrix (OsteoGraf/N) with and without autogenous bone: A clinical, histologic, radiographic, and histomorphometric analysis-Part 2 of an ongoing prospective study. Int. J. Periodontics Restor. Dent. 1998, 18, 528–543. [Google Scholar]
- Lundgren, S.; Andersson, S.; Gualini, F.; Sennerby, L. Bone reformation with sinus membrane elevation: A new surgical technique for maxillary sinus floor augmentation. Clin. Implant Dent. Relat. Res. 2004, 6, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Felice, P.; Scarano, A.; Pistilli, R.; Checchi, L.; Piattelli, M.; Pellegrino, G.; Esposito, M. A comparison of two techniques to augment maxillary sinuses using the lateral window approach: Rigid synthetic resorbable barriers versus anorganic bovine bone. Five-month post-loading clinical and histological results of a pilot randomised controlled clinical trial. Eur. J. Oral Implantol. 2009, 2, 293–306. [Google Scholar] [PubMed]
- Borges, F.L.; Dias, R.O.; Piattelli, A.; Onuma, T.; Gouveia Cardoso, L.A.; Salomão, M.; Scarano, A.; Ayub, E.; Shibli, J.A. Simultaneous sinus membrane elevation and dental implant placement without bone graft: A 6-month follow-up study. J. Periodontol. 2011, 82, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Bayliss, P.E.; Bellavance, K.L.; Whitehead, G.G.; Abrams, J.M.; Aegerter, S.; Robbins, H.S.; Cowan, D.B.; Keating, M.T.; O’Reilly, T.; Wood, J.M.; et al. Chemical modulation of receptor signaling inhibits regenerative angiogenesis in adult zebrafish. Nat. Chem. Biol. 2006, 2, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Rabie, A.B.M. VEGF: An essential mediator of both angiogenesis and endochondral ossification. J. Dent. Res. 2007, 86, 937–950. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Ceccarelli, M.; Marchetti, M.; Piattelli, A.; Mortellaro, C. Soft Tissue Augmentation with Autologous Platelet Gel and β-TCP: A Histologic and Histometric Study in Mice. Biomed. Res. Int. 2016, 2016, 2078104. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Valbonetti, L.; Marchetti, M.; Lorusso, F.; Ceccarelli, M. Soft Tissue Augmentation of the Face with Autologous Platelet-Derived Growth Factors and Tricalcium Phosphate. Microtomography Evaluation of Mice. J. Craniofac. Surg. 2016, 27, 1212–1214. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Piattelli, A.; Iezzi, G.; Varvara, G. Spontaneous bone formation on the maxillary sinus floor in association with surgery to remove a migrated dental implant: A case report. Minerva Stomatol. 2014, 63, 351–359. [Google Scholar] [PubMed]
- Lundgren, S.; Cricchio, G.; Palma, V.C.; Salata, L.A.; Sennerby, L. Sinus membrane elevation and simultaneous insertion of dental implants: A new surgical technique in maxillary sinus floor augmentation. Periodontol. 2000 2008, 47, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.-S.; Chung, S.-W.; Nam, W.; Cho, I.-H.; Cha, I.-H.; Park, H.-S. Spontaneous bone formation on the maxillary sinus floor in association with an extraction socket. Int. J. Oral Maxillofac. Surg. 2007, 36, 656–657. [Google Scholar] [CrossRef] [PubMed]
- Lundgren, S.; Andersson, S.; Sennerby, L. Spontaneous bone formation in the maxillary sinus after removal of a cyst: Coincidence or consequence? Clin. Implant Dent. Relat. Res. 2003, 5, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Srouji, S.; Kizhner, T.; Ben David, D.; Riminucci, M.; Bianco, P.; Livne, E. The Schneiderian membrane contains osteoprogenitor cells: In vivo and in vitro study. Calcif. Tissue Int. 2009, 84, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Misch, C.E. Treatment Planning for the Edentulous Posterior Maxilla. In Conemporary Implant Dentistry; Misch, C.E., Ed.; Elsevier: St. Louis, MO, USA, 1999. [Google Scholar]
- Scala, A.; Botticelli, D.; Rangel, I.G.; De Oliveira, J.A.; Okamoto, R.; Lang, N.P. Early healing after elevation of the maxillary sinus floor applying a lateral access: A histological study in monkeys. Clin. Oral Implants Res. 2010, 21, 1320–1326. [Google Scholar] [CrossRef] [PubMed]
- Scala, A.; Botticelli, D.; Faeda, R.S.; Garcia Rangel, I., Jr.; Américo de Oliveira, J.; Lang, N.P. Lack of influence of the Schneiderian membrane in forming new bone apical to implants simultaneously installed with sinus floor elevation: An experimental study in monkeys. Clin. Oral Implants Res. 2012, 23, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Caneva, M.; Lang, N.P.; Garcia Rangel, I.J.; Ferreira, S.; Caneva, M.; De Santis, E.; Botticelli, D. Sinus mucosa elevation using Bio-Oss® or Gingistat® collagen sponge: An experimental study in rabbits. Clin. Oral Implants Res. 2017, 28, e21–e30. [Google Scholar] [CrossRef] [PubMed]
- Omori, Y.; Ricardo Silva, E.; Botticelli, D.; Apaza Alccayhuaman, K.A.; Lang, N.P.; Xavier, S.P. Reposition of the bone plate over the antrostomy in maxillary sinus augmentation: A histomorphometric study in rabbits. Clin. Oral Implants Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Favero, V.; Lang, N.P.; Canullo, L.; Urbizo Velez, J.; Bengazi, F.; Botticelli, D. Sinus floor elevation outcomes following perforation of the S chneiderian membrane. An experimental study in sheep. Clin. Oral Implants Res. 2016, 27, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Scala, A.; Lang, N.P.; Velez, J.U.; Favero, R.; Bengazi, F.; Botticelli, D. Effects of a collagen membrane positioned between augmentation material and the sinus mucosa in the elevation of the maxillary sinus floor. An experimental study in sheep. Clin. Oral Implants Res. 2016, 27, 1454–1461. [Google Scholar] [CrossRef] [PubMed]
- Iida, T.; Martins Neto, E.C.; Botticelli, D.; Alccayhuaman, K.A.A.; Lang, N.P.; Xavier, S.P. Influence of a collagen membrane positioned subjacent the sinus mucosa following the elevation of the maxillary sinus. A histomorphometric study in rabbits. Clin. Oral Implants Res. 2017, 28, 1567–1576. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-C.; Kaigler, D.; Rice, K.G.; Krebsbach, P.H.; Mooney, D.J. Combined angiogenic and osteogenic factor delivery enhances bone marrow stromal cell-driven bone regeneration. J. Bone Miner. Res. 2005, 20, 848–857. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Lorusso, F.; Arcangelo, M.; D’Arcangelo, C.; Celletti, R. Lateral Sinus Floor Elevation Performed with Trapezoidal and Modified Triangular Flap Designs: A Randomized Pilot Study of Post-Operative Pain Using Thermal Infrared Imaging. Int. J. Environ. Res. Publ. Health 2018, 15, 1277. [Google Scholar] [CrossRef] [PubMed]
- Hatano, N.; Shimizu, Y.; Ooya, K. A clinical long-term radiographic evaluation of graft height changes after maxillary sinus floor augmentation with a 2:1 autogenous bone/xenograft mixture and simultaneous placement of dental implants. Clin. Oral Implants Res. 2004, 15, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Brånemark, P.I.; Adell, R.; Albrektsson, T.; Lekholm, U.; Lindström, J.; Rockler, B. An experimental and clinical study of osseointegrated implants penetrating the nasal cavity and maxillary sinus. J. Oral Maxillofac. Surg. 1984, 42, 497–505. [Google Scholar] [CrossRef]
- Shibli, J.A.; Faveri, M.; Ferrari, D.S.; Melo, L.; Garcia, R.V.; d’Avila, S.; Figueiredo, L.C.; Feres, M. Prevalence of maxillary sinus septa in 1024 subjects with edentulous upper jaws: A retrospective study. J. Oral Implantol. 2007, 33, 293–296. [Google Scholar] [CrossRef]
- Avila-Ortiz, G.; Wang, H.-L.; Galindo-Moreno, P.; Misch, C.E.; Rudek, I.; Neiva, R. Influence of lateral window dimensions on vital bone formation following maxillary sinus augmentation. Int. J. Oral Maxillofac. Implants 2012, 27, 1230–1238. [Google Scholar] [PubMed]
- Avila, G.; Wang, H.-L.; Galindo-Moreno, P.; Misch, C.E.; Bagramian, R.A.; Rudek, I.; Benavides, E.; Moreno-Riestra, I.; Braun, T.; Neiva, R. The influence of the bucco-palatal distance on sinus augmentation outcomes. J. Periodontol. 2010, 81, 1041–1050. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Lorusso, F.; Staiti, G.; Sinjari, B.; Tampieri, A.; Mortellaro, C. Sinus Augmentation with Biomimetic Nanostructured Matrix: Tomographic, Radiological, Histological and Histomorphometrical Results after 6 Months in Humans. Front. Physiol. 2017, 8, 565. [Google Scholar] [CrossRef] [PubMed]
- Wallace, S.S.; Froum, S.J. Effect of maxillary sinus augmentation on the survival of endosseous dental implants. A systematic review. Ann. Periodontol. 2003, 8, 328–343. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Cholakis, A.K.; Piattelli, A. Histologic Evaluation of Sinus Grafting Materials After Peri-implantitis-Induced Failure: A Case Series. Int. J. Oral Maxillofac. Implants 2017, 32, e69–e75. [Google Scholar] [CrossRef] [PubMed]
- Bianco, P.; Robey, P.G. Stem cells in tissue engineering. Nature 2001, 414, 118–121. [Google Scholar] [CrossRef] [PubMed]
- Bianco, P.; Robey, P.G. Skeletal stem cells. In Handbook of Adult and Fetal Stem Cells; Lanza, R., Ed.; Academic Press: San Diego, CA, USA, 2004. [Google Scholar]
- Trubiani, O.; Zalzal, S.F.; Paganelli, R.; Marchisio, M.; Giancola, R.; Pizzicannella, J.; Bühring, H.-J.; Piattelli, M.; Caputi, S.; Nanci, A. Expression profile of the embryonic markers nanog, OCT-4, SSEA-1, SSEA-4, and frizzled-9 receptor in human periodontal ligament mesenchymal stem cells. J. Cell. Physiol. 2010, 225, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Gruber, R.; Kandler, B.; Fuerst, G.; Fischer, M.B.; Watzek, G. Porcine sinus mucosa holds cells that respond to bone morphogenetic protein (BMP)-6 and BMP-7 with increased osteogenic differentiation in vitro. Clin. Oral Implants Res. 2004, 15, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Piattelli, A.; Scarano, A.; Quaranta, M. High-precision, cost-effective cutting system for producing thin sections of oral tissues containing dental implants. Biomaterials 1997, 18, 577–579. [Google Scholar] [CrossRef]
- Dempster, D.W.; Compston, J.E.; Drezner, M.K.; Glorieux, F.H.; Kanis, J.A.; Malluche, H.; Meunier, P.J.; Ott, S.M.; Recker, R.R.; Parfitt, A.M. Standardized nomenclature, symbols, and units for bone histomorphometry: A 2012 update of the report of the ASBMR Histomorphometry Nomenclature Committee. J. Bone Miner. Res. 2013, 28, 2–17. [Google Scholar] [CrossRef] [PubMed]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scarano, A.; De Oliveira, P.S.; Traini, T.; Lorusso, F. Sinus Membrane Elevation with Heterologous Cortical Lamina: A Randomized Study of a New Surgical Technique for Maxillary Sinus Floor Augmentation without Bone Graft. Materials 2018, 11, 1457. https://doi.org/10.3390/ma11081457
Scarano A, De Oliveira PS, Traini T, Lorusso F. Sinus Membrane Elevation with Heterologous Cortical Lamina: A Randomized Study of a New Surgical Technique for Maxillary Sinus Floor Augmentation without Bone Graft. Materials. 2018; 11(8):1457. https://doi.org/10.3390/ma11081457
Chicago/Turabian StyleScarano, Antonio, Pablo Santos De Oliveira, Tonino Traini, and Felice Lorusso. 2018. "Sinus Membrane Elevation with Heterologous Cortical Lamina: A Randomized Study of a New Surgical Technique for Maxillary Sinus Floor Augmentation without Bone Graft" Materials 11, no. 8: 1457. https://doi.org/10.3390/ma11081457
APA StyleScarano, A., De Oliveira, P. S., Traini, T., & Lorusso, F. (2018). Sinus Membrane Elevation with Heterologous Cortical Lamina: A Randomized Study of a New Surgical Technique for Maxillary Sinus Floor Augmentation without Bone Graft. Materials, 11(8), 1457. https://doi.org/10.3390/ma11081457







