Synthesis of Heart/Dumbbell-Like CuO Functional Nanostructures for the Development of Uric Acid Biosensor
Abstract
1. Introduction
2. Experimental Section
2.1. Chemicals Used
2.2. Synthesis of CuO Nanostructures Using a Low-Temperature Aqueous Chemical Growth Method with Vitamin B12 on Gold Coated Glass Substrates
2.3. Material Characterization
2.4. The Immobilization of Uricase Enzymes on the Nanostructured CuO and Potentiometric Measurement
3. Results and Discussion
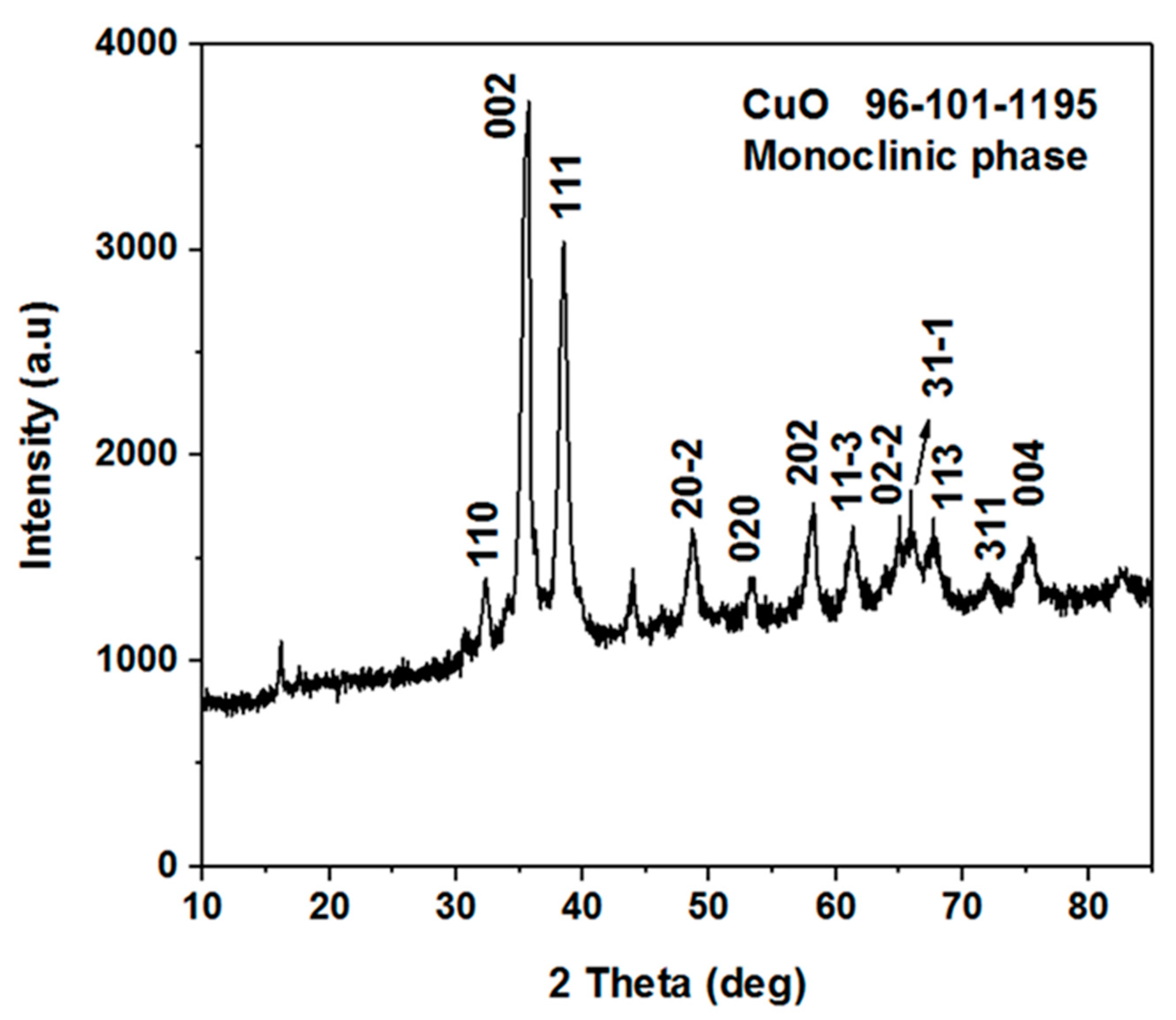
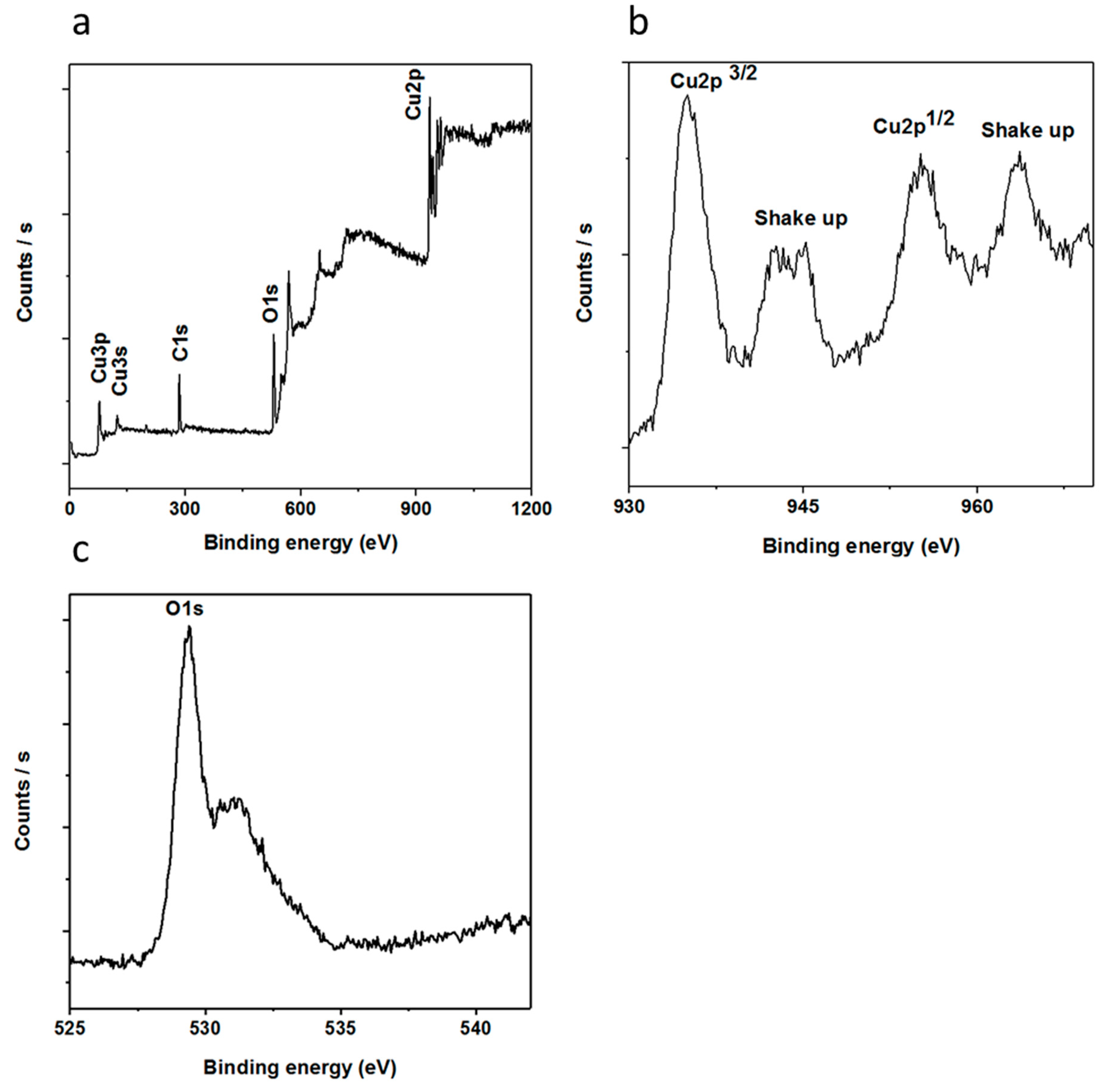
3.1. The Morphological, Structural, and Composition Studies of as Prepared CuO Nanostructures Using Vitamin B12
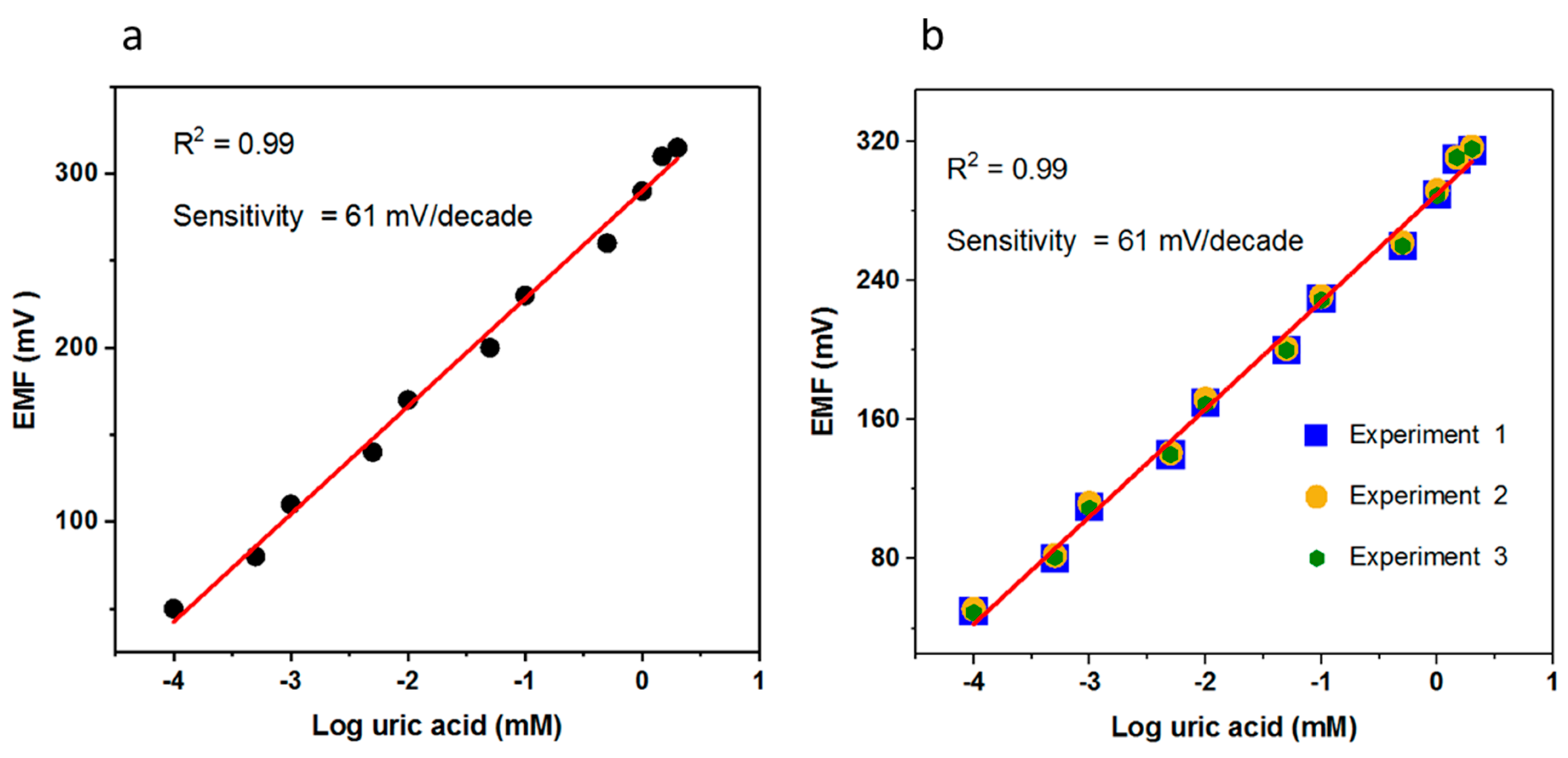
3.2. The Potentiometric Response of the Proposed Uric Acid Biosensor Based on Uricase Immobilized Heart/Dumbbell-Like CuO Nanostructures
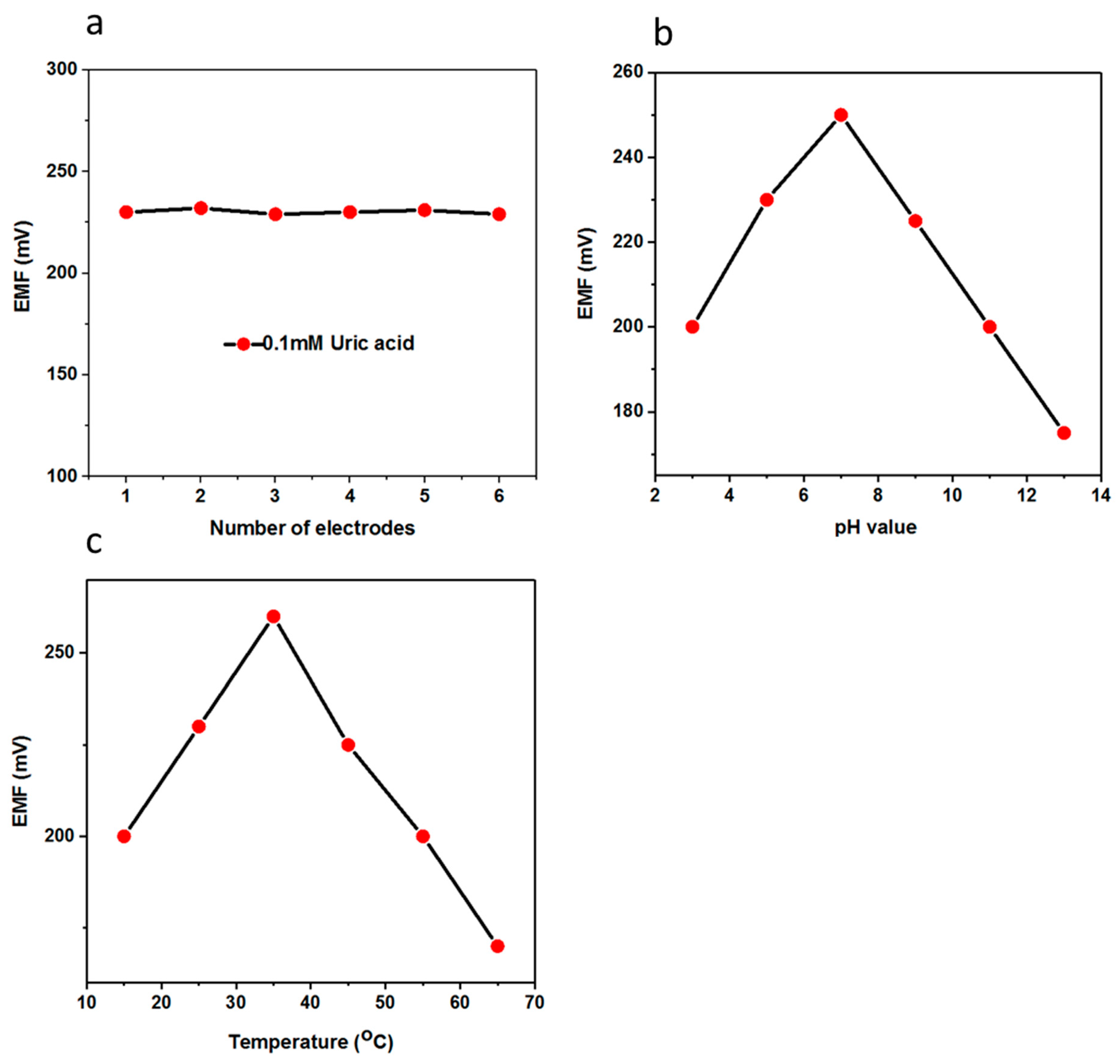
3.3. pH and Temperature Studies
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sang, X.; Zhang, J.; Wu, T.; Zhang, B.; Ma, X.; Peng, L.; Han, B.; Kang, X.; Liu, C.; Yang, G. Room-temperature synthesis of mesoporous CuO and its catalytic activity for cyclohexene oxidation. RSC Adv. 2015, 5, 67168–67174. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, K.; Xu, D.; Yang, G.; Huang, H.; Nie, F.; Liu, C.; Yang, C. CuO nanostructures: Synthesis, characterization, growth mechanisms, fundamental properties, and applications. Prog. Mater. Sci. 2014, 60, 208–337. [Google Scholar] [CrossRef]
- Rout, L.; Sen, T.K.; Punniyamurthy, T. Efficient CuO nanoparticle catalyzed C-S cross coupling of thiols with iodobenzene. Angew. Chem. 2007, 46, 5583. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Sithambaram, S.; Zhang, Y.; Chen, C.H.; Jin, L.; Joesten, R.; Suib, S.L. Novel urchin-like CuO synthesized by a facile reflux method with efficient olefin epoxidation catalytic performance. Chem. Mater. 2009, 21, 1253–1259. [Google Scholar] [CrossRef]
- Cao, H.; Jiang, H.F.; Zhou, X.S.; Qi, C.R.; Lin, Y.G.; Wu, J.Y.; Liang, Q.M. CuO/CNTs-catalyzed heterogeneous process: A convenient strategy to prepare furan derivatives from electron-deficient alkynes and α-hydroxy ketones. Green Chem. 2012, 14, 2710–2714. [Google Scholar] [CrossRef]
- Rout, S.K.; Guin, S.; Nath, J.; Patel, B.K. An “on-water” exploration of CuO nanoparticle catalysed synthesis of 2-aminobenzothiazoles. Green Chem. 2012, 4, 2491–2498. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, H.Y.; Jia, X.; Liu, B.; Yang, Y. Cheminform abstract: One-dimensional metal oxide nanostructures for heterogeneous catalysis. Nanoscale 2013, 5, 7175–7183. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Roy, M.; Naskar, M.K. A Facile Soft-chemical synthesis of cube-shaped mesoporous CuO with microcarpet-like interior. Cryst. Growth Des. 2014, 14, 2977–2984. [Google Scholar] [CrossRef]
- Song, M.K.; Park, S.; Alamgir, F.M.; Cho, J.; Liu, M. Nanostructured electrodes for lithium-ion and lithium-air batteries: The latest developments, challenges, and perspectives. Mater. Sci. Eng. R 2011, 72, 203–252. [Google Scholar] [CrossRef]
- Wang, B.; Wu, X.L.; Shu, C.Y.; Guo, Y.G.; Wang, C.R. Synthesis of CuO/graphene nanocomposite as a high-performance anode material for lithium-ion batteries. J. Mater. Chem. 2010, 20, 10661–10664. [Google Scholar] [CrossRef]
- Ko, S.; Lee, J.I.; Yang, H.S.; Park, S.; Jeong, U. Mesoporous CuO particles threaded with CNTs for high-performance lithium-ion battery anodes. Adv. Mater. 2012, 24, 4451–4456. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Liu, Y.; Wang, J.; Gao, M.; Peng, X.; Ye, Z. Self-assembly of mesoporous CuO nanosheets–CNT 3D-network composites for lithium-ion batteries. Nanoscale 2013, 5, 1785–1788. [Google Scholar] [CrossRef] [PubMed]
- Kargar, A.; Jing, Y.; Kim, S.J.; Riley, C.T.; Pan, X.; Wang, D. ZnO/CuO heterojunction branched nanowires for photoelectrochemical hydrogen generation. ACS Nano 2013, 7, 11112–11120. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Pu, X.; Liu, J.; Liang, J.; Liu, P.; Li, X.; Yu, X. CuO nanoleaves enhance the c-Si solar cell efficiency. J. Mater. Chem. A 2014, 2, 6796–6800. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, L.; Zhang, J. A review of electrode materials for electrochemical supercapacitors. Chem. Soc. Rev. 2012, 41, 797–828. [Google Scholar] [CrossRef] [PubMed]
- Faraji, S.; Ani, F.N. Microwave-assisted synthesis of metal oxide/hydroxide composite electrodes for high power supercapacitors—A review. J. Power Sources 2014, 263, 338–360. [Google Scholar] [CrossRef]
- Bedi, R.K.; Singh, I. Room-temperature ammonia sensor based on cationic surfactant-assisted nanocrystalline CuO. ACS Appl. Mater. Interfaces 2010, 2, 1361–1368. [Google Scholar] [CrossRef] [PubMed]
- Taubert, A.; Stange, F.; Li, Z.; Junginger, M.; Gunter, C.; Neumann, M.; Friedrich, A. CuO nanoparticles from the strongly hydrated ionic liquid precursor (ILP) tetrabutylammonium hydroxide: Evaluation of the ethanol sensing activity. ACS Appl. Mater. Interfaces 2012, 4, 791–795. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Lee, J.H. Highly sensitive and selective gas sensors using p-type oxide semiconductors. Sens. Actuators B 2014, 192, 607–627. [Google Scholar] [CrossRef]
- Tian, W.; Zhi, C.; Zhai, T.; Wang, X.; Liao, M.; Li, S.; Chen, S.; Golberg, D.; Bando, Y. Ultrahigh quantum efficiency of CuO nanoparticle decorated In2Ge2O7 nanobelt deep-ultraviolet photodetectors. Nanoscale 2012, 4, 6318–6324. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Shi, W.; Zhu, J.; Kharistal, D.J.; Zhao, W.; Lalia, B.S.; Hng, H.H.; Yan, Q. High-power and high-energy-density flexible pseudocapacitor electrodes made from porous CuO nanobelts and single-walled carbon nanotubes. ACS Nano 2011, 5, 2013–2019. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Rioux, R.M.; Erdonmez, C.K.; Hughes, S.; Somorjai, G.A.; Alivisatos, A.P. Formation of hollow nanocrystals through the nanoscale Kirkendall effect. Science 2004, 304, 711–714. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Her, Y.S.; Matijevic, E. Preparation and growth mechanism of uniform colloidal copper oxide by the controlled double-iet precipitation. J. Coll. Int. Sci. 1997, 186, 193–202. [Google Scholar] [CrossRef]
- Jiang, X.C.; Herricks, T.; Xia, Y.N. CuO nanowires can be synthesized by heating copper substrate in air. Nano Lett. 2002, 2, 1333–1338. [Google Scholar] [CrossRef]
- Lu, C.H.; Qi, L.M.; Yang, J.H.; Zhang, D.Y.; Wu, N.Z.; Ma, J.M. Simple template-free solution route for the controlled synthesis of Cu(OH)2 and CuO nanostructures. J. Phys. Chem. B 2004, 108, 17825–17831. [Google Scholar] [CrossRef]
- Hou, H.W.; Xie, Y.; Li, Q. Large-scale synthesis of single-crystalline quasi-aligned submicrometer CuO ribbons. Cryst. Grow. Des. 2005, 5, 201–205. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhu, J.J.; Hong, J.M.; Bian, N.S.; Chen, H.Y. Microwave-induced polyol-process synthesis of copper and copper oxide nanocrystals with controllable morphology. Eur. J. Inorg. Chem. 2004, 2004, 4072–4080. [Google Scholar] [CrossRef]
- Chen, D.; Shen, G.; Tang, K.; Zheng, H.; Qian, Y. Low-temperature synthesis of metal tungstates nanocrystallites in ethylene glycol. Mater. Res. Bull. 2003, 38, 1783–1789. [Google Scholar] [CrossRef]
- Yao, W.T.; Yu, S.H.; Zhou, Y.; Jiang, J.; Wu, Q.S.; Zhang, L.; Jiang, J. Formation of uniform CuO nanorods by spontaneous aggregation: Selective synthesis of CuO, Cu2O, and Cu nanoparticles by a solid-liquid phase arc discharge process. J. Phys. Chem. B 2005, 109, 14011–14016. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.X.; Wen, X.G.; Yang, S.H.; Berta, Y.; Wang, Z.L. Single-crystalline scroll-type nanotube arrays of copper hydroxide synthesized at room temperature. Adv. Mater. 2003, 15, 822–825. [Google Scholar] [CrossRef]
- Zhang, W.X.; Wen, X.G.; Yang, S.H. Controlled reactions on a copper surface: Synthesis and characterization of nanostructured copper compound films. Inorg. Chem. 2003, 42, 5005–5014. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.P.; Sun, H.P.; Shao, X.Q.; Li, D.F.; Yu, H.D.; Han, M.Y. Three-dimensionally oriented aggregation of a few hundred nanoparticles into monocrystalline architectures. Adv. Mater. 2005, 17, 42–47. [Google Scholar] [CrossRef]
- Zhang, L.Z.; Yu, J.C.; Xu, A.W.; Li, Q.; Kwong, K.W.; Yu, S.H. Peanut-shaped nanoribbon bundle superstructures of malachite and copper oxide. J. Cryst. Grow. 2004, 266, 545–551. [Google Scholar] [CrossRef]
- Li, S.Z.; Zhang, H.; Ji, Y.J.; Yang, D.R. CuO nanodendrites synthesized by a novel hydrothermal route. Nanotechnology 2004, 15, 1428–1432. [Google Scholar] [CrossRef]
- Xu, J.S.; Xue, D.F. Fabrication of malachite with a hierarchical sphere-like architecture. J. Phys. Chem. B 2005, 109, 17157–17161. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zeng, H.C. Mesoscale organization of CuO nanoribbons: Formation of dandelions. J. Am. Chem. Soc. 2004, 126, 8124–8125. [Google Scholar] [CrossRef] [PubMed]
- Zaman, S.; Asif, M.H.; Zainelabdin, A.; Amin, G.; Nur, O.; Willander, M. CuO nanoflowers as an electrochemical pH sensor and the effect of pH on the growth. J. Electroana. Chem. 2011, 662, 421–425. [Google Scholar] [CrossRef]
- Akyilmaz, E.; Sezgintürk, M.K.; Dinckaya, E. A biosensor based on urate oxidase-peroxidase coupled enzyme system for uric acid determination in urine. Talanta 2003, 61, 73–79. [Google Scholar] [CrossRef]
- Alderman, M.; Aiyer, K.J.V. Uric acid: Role in cardiovascular disease and effects of losartan. Curr. Med. Res. Opin. 2004, 20, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Arakawa, H.; Karube, I. Fabrication of a sensing module using micro machined biosensors. Biosens. Bioelectron. 2001, 16, 725–733. [Google Scholar] [CrossRef]
- Vchiyama, S.; Sakamoto, H. Immobilization of uricase to gas diffusion carbon felt by electropolymerization of aniline and its application as an enzyme reactor for uric acid sensor. Talanta 1997, 44, 1435–1439. [Google Scholar] [CrossRef]
- Yao, D.; Vlessidis, A.G.; Evmiridis, N.P. Microdialysis sampling and monitoring of uric acid in vivo by a chemiluminescence reaction and an enzyme on immobilized chitosan support membrane. Anal. Chim. Acta. 2003, 478, 23–30. [Google Scholar] [CrossRef]
- Hong, H.C.; Huang, H.J. Flow injection analysis of uric acid with an uricase and horseradish peroxidase-coupled sepharose column based luminol chemiluminescence system. Anal. Chim. Acta. 2003, 499, 41–46. [Google Scholar] [CrossRef]
- Deyhimi, F.; Ahangar, R.S. An initial-rate potentiometric method for the determination of uric acid using a fluoride ion-selective electrode. Talanta 2003, 61, 493–499. [Google Scholar] [CrossRef]
- Zhao, C.; Wan, L.; Wang, Q.; Liu, S.; Jiao, K. Highly sensitive and selective uric acid biosensor based on direct electron transfer of hemoglobin-encapsulated chitosan-modified glassy carbon electrode. Anal. Sci. 2009, 25, 1013–1017. [Google Scholar] [CrossRef] [PubMed]
- Bhambi, M.; Sum, G.; Malhotra, B.D.; Pundir, C.S. An amperometric uric acid biosensor based on immobilization of uricase onto polyaniline-multiwalled carbon nanotube composite film. Artif. Cells Blood Substit. Biotechnol. 2010, 38, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Kuwabata, S.; Nakaminami, T.; Ito, S.I.; Yoneyama, H. Preparation and properties of amperometric uric acid sensors. Sens. Act. B 1998, 52, 72–77. [Google Scholar] [CrossRef]
- Hoshi, T.; Saiki, H.; Anzai, J.I. Amperometric uric acid sensors based on polyelectrolyte multilayer films. Talanta 2003, 61, 363–368. [Google Scholar] [CrossRef]
- Harwood, G.W.J.; Pouton, C.W. Amperometric enzyme biosensors for the analysis of drugs and metabolites. Adv. Drug Del. Rev. 1996, 18, 163–191. [Google Scholar] [CrossRef]
- Cui, G.; Yoo, J.H.; Woo, B.W.; Kim, S.S.; Cha, G.S.; Nam, H. Disposable amperometric glucose sensor electrode with enzyme-immobilized nitrocellulose strip. Talanta 2001, 54, 1105–1111. [Google Scholar] [CrossRef]
- Liao, C.W.; Chou, J.C.; Sun, T.P.; Hsiung, S.K.; Hsieh, J.H. Preliminary investigations on a new disposable potentiometric biosensor for uric acid. IEEE Trans. Biomed. Eng. 2006, 53, 1401–1408. [Google Scholar] [CrossRef] [PubMed]
- Usman Ali, S.M.; Nur, O.; Willander, M.; Danielsson, B. Glucose detection with a commercial MOSFET using a ZnO nanowires extended gate. IEEE Trans. Nanotechnol. 2009, 8, 678–683. [Google Scholar] [CrossRef]
- Usman Ali, S.M.; Nur, O.; Willander, M.; Danielsson, B. A fast and sensitive potentiometric glucose microsensor based on glucose oxidase coated ZnO nanowires grown on a thin silver wire. Sens. Act. B 2010, 145, 869–874. [Google Scholar] [CrossRef]
- Asif, M.H.; Usman Ali, S.M.; Nur, O.; Willander, M.; Brännmark, C.; Strålfors, P.; Englund, U.H.; Elinder, F.; Danielsson, B. Functionalized ZnO-nanorod-based selective electrochemical sensor for intracellular glucose. Biosens. Bioelectron. 2010, 25, 2205–2211. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Nagendra, V.; Baig, R.B.; Varma, R.S.; Nadagouda, M.N. Expeditious synthesis of noble metal nanoparticles using vitamin B12 under microwave irradiation. Appl. Sci. 2015, 5, 415–426. [Google Scholar] [CrossRef]
- Durando, M.; Morrish, R.; Muscat, A.J. Kinetics and mechanism for the reaction of hexafluoroacetyl acetone with CuO in supercritical carbon dioxide. J. Am. Chem. Soc. 2008, 130, 16659–16668. [Google Scholar] [CrossRef] [PubMed]
- Dar, M.A.; Nam, S.H.; Kim, Y.S.; Kim, W.B. Synthesis, characterization, and electrochemical properties of self-assembled leaf-like CuO nanostructures. J. Solid State Electrochem. 2010, 14, 1719–1726. [Google Scholar] [CrossRef]
- Dar, M.A.; Ahsanulhaq, Q.; Kim, Y.S.; Sohn, J.M.; Kim, W.B.; Shin, H.S. Versatile synthesis of rectangular shaped nanobat-like CuO nanostructures by hydrothermal method; structural properties and growth mechanism. Appl. Surface Sci. 2009, 255, 6279–6284. [Google Scholar] [CrossRef]
- Ibupoto, Z.H.; Shah, S.M.U.A.; Khun, K.M.; Willander, M. Electrochemical L-lactic acid sensor based on immobilized ZnO nanorods with lactate oxidase. Sensors 2012, 12, 2456–2466. [Google Scholar] [CrossRef] [PubMed]
- Usman Ali, S.M.; Alvi, N.H.; Ibupoto, Z.; Nur, O.; Willander, M.; Danielsson, B. Selective potentiometric determination of uric acid with uricase immobilized on ZnO nanowires. Sens. Act. B 2011, 152, 241–247. [Google Scholar] [CrossRef]
- Usman Ali, S.M.; Ibupoto, Z.H.; Chey, C.O.; Nur, O.; Willander, M. Functionalized ZnO nanotube arrays for the selective determination of uric acid with immobilized uricase. Chem. Sens. 2011, 19, 1–8. [Google Scholar] [CrossRef]
- Usman Ali, S.M.; Ibupoto, Z.H.; Kashif, M.; Hashim, U.; Willander, M. A potentiometric indirect uric acid sensor based on ZnO nanoflakes and immobilized uricase. Sensors 2012, 12, 2787–2797. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, X.; Ai, S.; Sun, Z.; Wan, Q.; Zhu, Z.; Xian, Y.; Jin, L.; Yamamoto, K. immobilization of uricase on ZnO nanorods for a reagent less uric acid biosensor. Anal. Chim. Acta 2004, 519, 155–160. [Google Scholar] [CrossRef]
- Wang, Y.T.; Yu, L.; Zhu, Z.Q.; Zhang, J.; Zhu, J.Z. Novel uric acid sensor based on enzyme electrode modified by ZnO nanoparticles and multiwall carbon nanotubes. Anal. Lett. 2009, 42, 775–789. [Google Scholar] [CrossRef]
- Harish, S.; Mathiyarasu, J.; Phani, K.L.N.; Yegnaraman, V. PEDOT/palladium composite material: Synthesis, characterization and application to simultaneous determination of dopamine and uric acid. J. Appl. Electrochem. 2008, 38, 1583–1588. [Google Scholar] [CrossRef]
- Xu, T.Q.; Zhang, Q.L.; Zheng, J.N.; Lv, Z.Y.; Wei, J.; Wang, A.J.; Feng, J.J. Simultaneous determination of dopamine and uric acid in the presence of ascorbic acid using Pt nanoparticles supported on reduced graphene oxide. Electrochim. Acta 2014, 115, 109–115. [Google Scholar] [CrossRef]
- Liu, X.; Xie, L.; Li, H. Electrochemical biosensor based on reduced graphene oxide and Au nanoparticles entrapped in chitosan/silica sol-gel hybrid membranes for determination of dopamine and uric acid. J. Electroanal. Chem. 2012, 682, 158–163. [Google Scholar] [CrossRef]
- Li, Y.; Ran, G.; Yi, W.J.; Luo, H.Q.; Li, N.B. A glassy carbon electrode modified with graphene and poly(acridine red) for sensing uric acid. Microchim. Acta 2012, 178, 115–121. [Google Scholar] [CrossRef]

| No of Weeks | Linear Range Uric Acid (mM) | Sensitivity (mV/Decade) | Limit of Detection Uric Acid (mM) |
|---|---|---|---|
| 1 | 0.001–10 | 61.88 | 0.0005 |
| 2 | 0.002–10 | 61 | 0.0005 |
| 3 | 0.001–10 | 61.58 | 0.0004 |
| 4 | 0.0025–10 | 60 | 0.0003 |
| Spiked Concentration of Uric Acid (Mm) | Uric Acid Conc. as Quantified by Proposed Biosensor | % Uric Acid Biosensor Recovery |
|---|---|---|
| 0 | 1.5 | - |
| 1.8 | 2 | 111.11 |
| 3.5 | 3.7 | 105.71 |
| 2.5 | 2.4 | 96 |
| 4.1 | 4.2 | 102.43 |
| Interfering Species (B) | Log Kpot uric acid, B |
|---|---|
| Ascorbic acid | 2.5 |
| Urea | 1.9 |
| Glucose | 2.25 |
| Electrode Material | Technique | Linear Range mM | Sensitivity mV/Decade | References |
|---|---|---|---|---|
| ZnO nanowires | Potentiometry | 0.001–1 | 29 | [60] |
| ZnO nanotubes | Potentiometry | 0.05–1.5 | 68 | [61] |
| ZnO nanoflakes | Potentiometry | 0.0005–1.5 | 66 | [62] |
| ZnO nanorods | Amperometry | 0.005–1 | ---------- | [63] |
| ZnO nanoparticles | Amperometry | 0.00–1 | ---------- | [64] |
| PEDOT/Palladium | Differential pulse voltammetry | 0.007–0.011 | ---------- | [65] |
| PrGO | Differential pulse voltammetry | 0.3 | ---------- | [66] |
| Graphene-poly(acridine red)/GCE | Differential pulse voltammetry | 0.008–0.15 | ---------- | [67] |
| RGO–AuNPs–CSHMs | Differential pulse voltammetry | 0.001–0.3 | ---------- | [68] |
| CuO nanostrucutures | Potentiometery | 0.001–10 | 61 | This work |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibupoto, Z.H.; Tahira, A.; Raza, H.; Ali, G.; Khand, A.A.; Jilani, N.S.; Mallah, A.B.; Yu, C.; Willander, M. Synthesis of Heart/Dumbbell-Like CuO Functional Nanostructures for the Development of Uric Acid Biosensor. Materials 2018, 11, 1378. https://doi.org/10.3390/ma11081378
Ibupoto ZH, Tahira A, Raza H, Ali G, Khand AA, Jilani NS, Mallah AB, Yu C, Willander M. Synthesis of Heart/Dumbbell-Like CuO Functional Nanostructures for the Development of Uric Acid Biosensor. Materials. 2018; 11(8):1378. https://doi.org/10.3390/ma11081378
Chicago/Turabian StyleIbupoto, Zafar Hussain, Aneela Tahira, Hamid Raza, Gulzar Ali, Aftab Ahmed Khand, Nabila Shah Jilani, Arfana Begum Mallah, Cong Yu, and Magnus Willander. 2018. "Synthesis of Heart/Dumbbell-Like CuO Functional Nanostructures for the Development of Uric Acid Biosensor" Materials 11, no. 8: 1378. https://doi.org/10.3390/ma11081378
APA StyleIbupoto, Z. H., Tahira, A., Raza, H., Ali, G., Khand, A. A., Jilani, N. S., Mallah, A. B., Yu, C., & Willander, M. (2018). Synthesis of Heart/Dumbbell-Like CuO Functional Nanostructures for the Development of Uric Acid Biosensor. Materials, 11(8), 1378. https://doi.org/10.3390/ma11081378





