Thermal Behavior and Structural Study of SiO2/Poly(ε-caprolactone) Hybrids Synthesized via Sol-Gel Method
Abstract
:1. Introduction
2. Results and Discussion
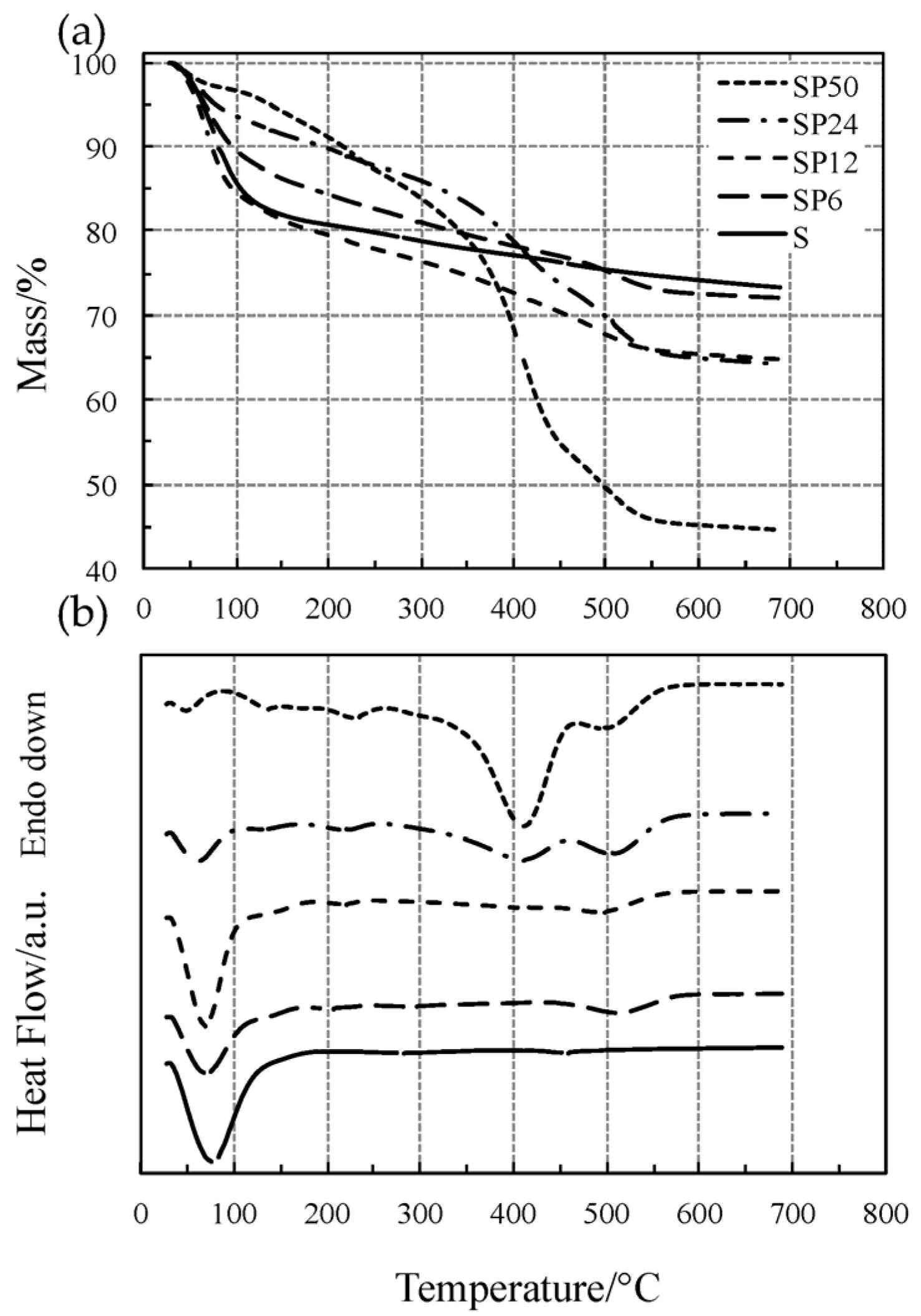
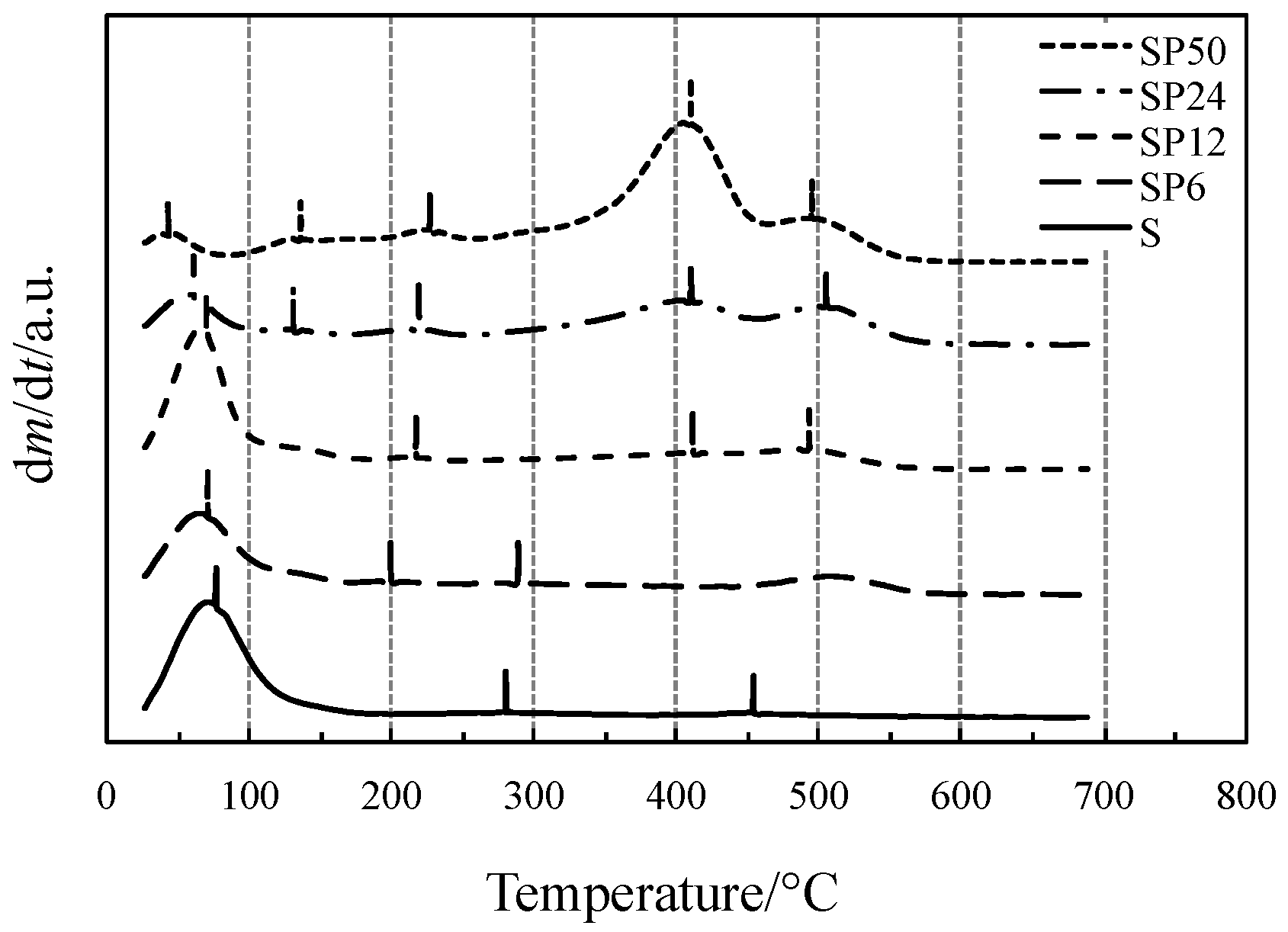
2.1. Thermal Behavior Study
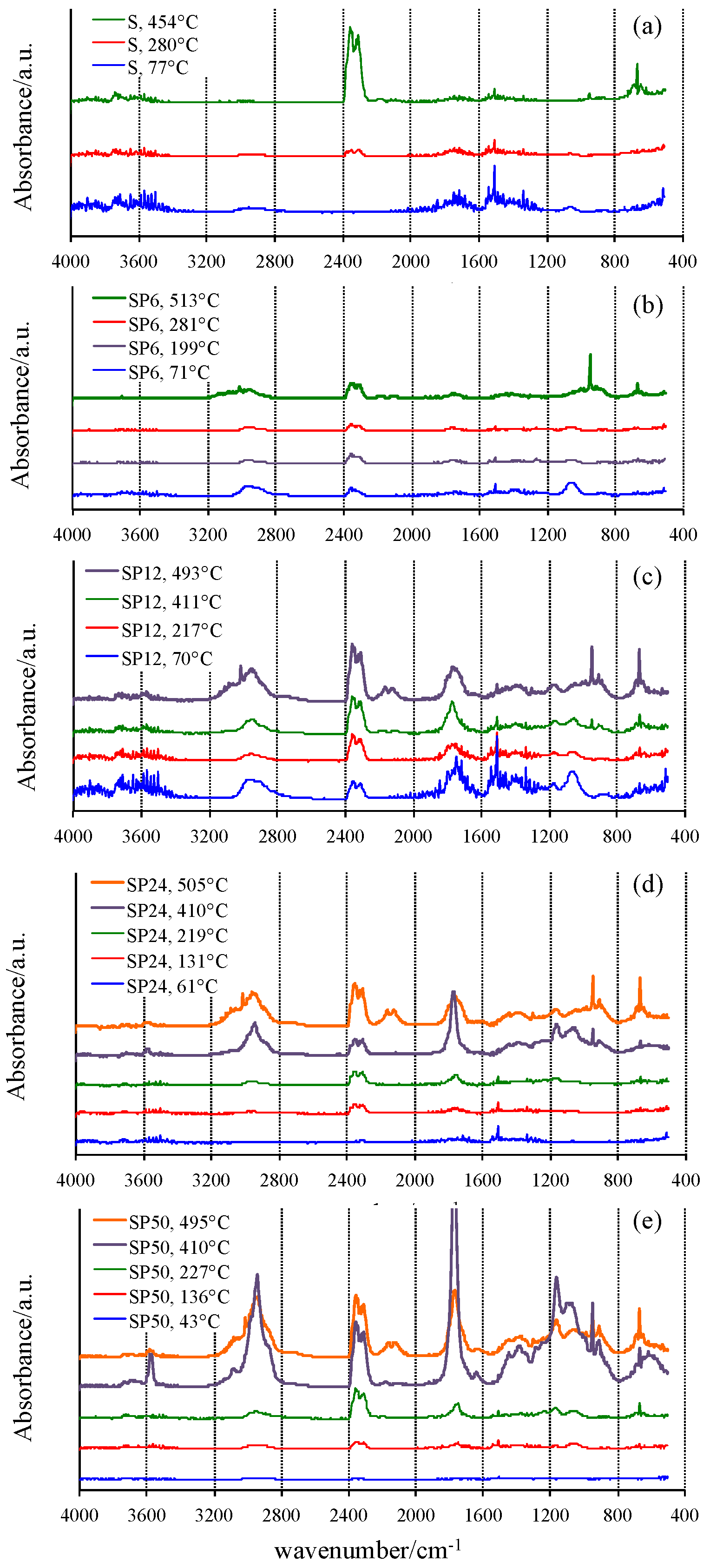
2.2. FTIR Evolution Gas Analysis to Provide a Mechanistic Interpretation of the Thermally Stimulated Processes
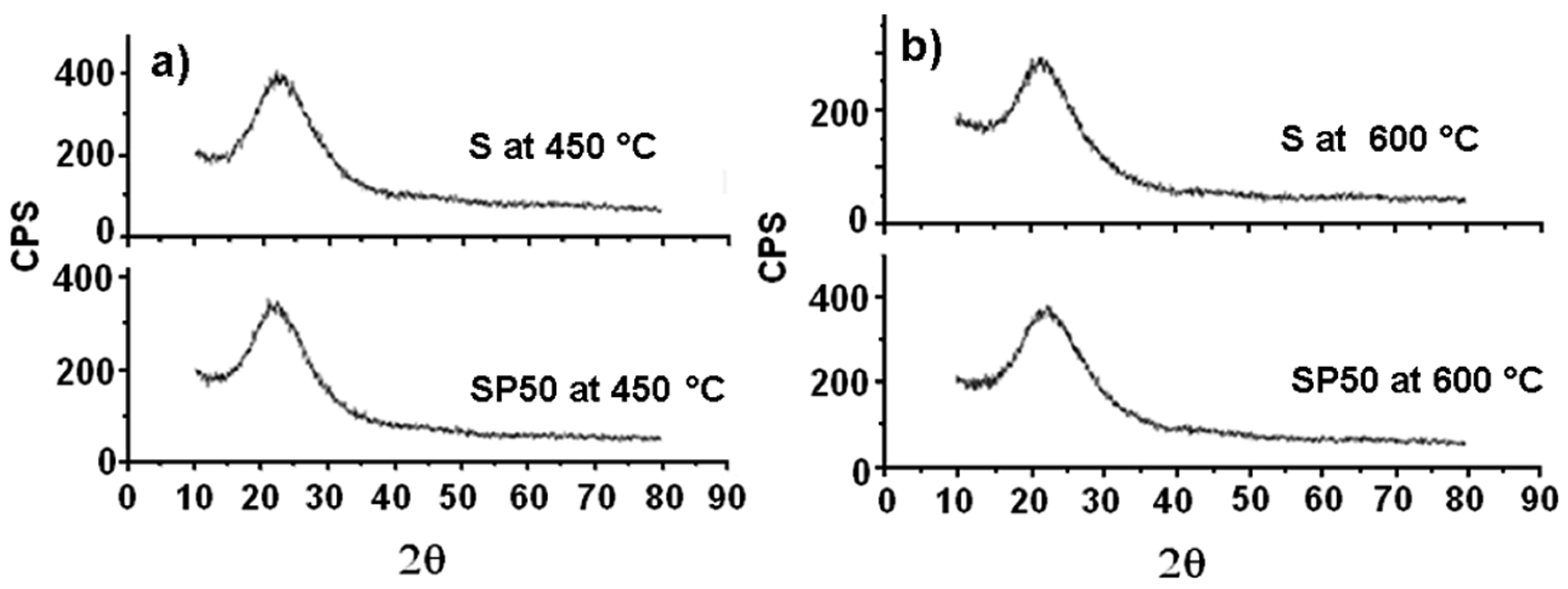
2.3. XRD Analysis to Provide a Mechanistic Interpretation of the Thermally Stimulated Processes
3. Materials and Methods
3.1. Synthesis of the Hybrid Materials
3.2. Instrumental Details to Study the Thermal Behavior of the Hybrid Materials
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Joshua, D.Y.; Damron, M.; Tang, G.; Zheng, H.; Chu, C.-J.; Osborne, J.H. Inorganic/organic hybrid coatings for aircraft aluminum alloy substrates. Prog. Org. Coat. 2001, 41, 226–232. [Google Scholar] [CrossRef]
- Catauro, M.; Bollino, F. Anti-inflammatory entrapment in polycaprolactone/silica hybrid material prepared by Sol-Gel route, characterization, bioactivity and in vitro release behavior. J. Appl. Biomater. Funct. Mater. 2013, 11, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Klukowska, A.; Posset, U.; Schottner, G.; Wis, M.L.; Salemi-Delvaux, C.; Malatesta, V. Photochromic hybrid Sol-Gel coatings: Preparation, properties, and applications. Mater. Sci. 2002, 20, 95–104. [Google Scholar]
- Samuneva, B.; Djambaski, P.; Kashchieva, E.; Chernev, G.; Kabaivanova, L.; Emanuilova, E.; Salvado, I.M.M.; Fernandes, M.H.V.; Wu, A. Sol-Gel synthesis and structure of silica hybrid biomaterials. J. Non-Cryst. Solids 2008, 354, 733–740. [Google Scholar] [CrossRef]
- Novak, B.M. Hybrid nanocomposite materials—Between inorganic glasses and organic polymers. Adv. Mater. 1993, 5, 422–433. [Google Scholar] [CrossRef]
- Diaz, U.; Corma, A. Organic-inorganic hybrid materials: Multi-functional solids for multi-step reaction processes. Chem. Eur. J. 2018, 2, 1–16. [Google Scholar] [CrossRef] [PubMed]
- You, N.; Liu, T.-H.; Fan, H.-T.; Shen, H. An efficient mercapto-functionalized organic-inorganic hybrid sorbent for selective separation and preconcentration of antimony(iii) in water samples. RSC Adv. 2018, in press. [Google Scholar] [CrossRef]
- Sanchez, C.; Ribot, F. Design of hybrid organic-inorganic materials synthesized via Sol-Gel chemistry. New J. Chem. 1994, 18, 1007–1047. [Google Scholar]
- Wei, Y.; Jin, D.; Brennan, D.J.; Rivera, D.N.; Zhuang, Q.; DiNardo, N.J.; Qiu, K. Atomic force microscopy study of organic-inorganic hybrid materials. Chem. Mater. 1998, 10, 769–772. [Google Scholar] [CrossRef]
- Judeinstein, P.; Sanchez, C. Hybrid organic-inorganic materials: A land of multidisciplinarity. J. Mater. Chem. 1996, 6, 511–525. [Google Scholar] [CrossRef]
- Brinker, C.; Scherer, G. Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing; Academic Press: San Diego, CA, USA, 1989. [Google Scholar]
- Catauro, M.; Bollino, F.; Papale, F. Synthesis of SiO2 system via Sol-Gel process: Biocompatibility tests with a fibroblast strain and release kinetics. J. Biomed. Mater. Res. Part A 2014, 102, 1677–1680. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C.; Julian, B.; Belleville, P.; Popall, M. Applications of hybrid organic-inorganic nanocomposites. J. Mater. Chem. 2005, 15, 3559–3592. [Google Scholar] [CrossRef]
- Casarin, J.; Goncalves, A.C., Jr.; Segatelli, M.G.; Tarley, C.R.T. Poly(methacrylic acid)/SiO2/AL2O3 based organic-inorganic hybrid adsorbent for adsorption of imazethapyr herbicide from aqueous medium. React. Funct. Polym. 2017, 121, 101–109. [Google Scholar] [CrossRef]
- Niknahad, M.; Mannari, V. Corrosion protection of aluminum alloy substrate with nano-silica reinforced organic-inorganic hybrid coatings. J. Coat. Technol. Res. 2016, 13, 1035–1046. [Google Scholar] [CrossRef]
- Qu, K.; Pu, Q.; Shan, G. Preparation and characterization of organic-inorganic hybrid flexible silica aerogel. Huagong Xuebao Chin. Ed. 2014, 65, 346–351. [Google Scholar]
- Ren, Y.; Zhang, Y.; Gu, Y.; Zeng, Q. Flame retardant polyacrylonitrile fabrics prepared by organic-inorganic hybrid silica coating via Sol-Gel technique. Prog. Org. Coat. 2017, 112, 225–233. [Google Scholar] [CrossRef]
- Zhang, X.; Lin, W.; Zheng, J.; Sun, Y.; Xia, B.; Yan, L.; Jiang, B. Insight into the organic-inorganic hybrid and microstructure tailor mechanism of SoL-GeL ORMOSIL antireflective coatings. J. Phys. Chem. C 2018, 122, 596–603. [Google Scholar] [CrossRef]
- Catauro, M.; Bollino, F.; Dell’Era, A.; Ciprioti, S.V. Pure AL2O3·2SiO2 synthesized via a Sol-Gel technique as a raw material to replace metakaolin: Chemical and structural characterization and thermal behavior. Ceram. Int. 2016, 42, 16303–16309. [Google Scholar] [CrossRef]
- Catauro, M.; Bollino, F.; Papale, F.; Vecchio Ciprioti, S. Investigation on bioactivity, biocompatibility, thermal behavior and antibacterial properties of calcium silicate glass coatings containing ag. J. Non-Cryst. Solids 2015, 422, 16–22. [Google Scholar] [CrossRef]
- Catauro, M.; Dell’Era, A.; Vecchio Ciprioti, S. Synthesis, structural, spectroscopic and thermoanalytical study of Sol-Gel derived SiO2-CaO-P2O5 gel and ceramic materials. Thermochim. Acta 2016, 625, 20–27. [Google Scholar] [CrossRef]
- Vecchio Ciprioti, S.; Catauro, M. Synthesis, structural and thermal behavior study of four Ca-containing silicate gel-glasses: Activation energies of their dehydration and dehydroxylation processes. J. Therm. Anal. Calorim. 2016, 123, 2091–2101. [Google Scholar] [CrossRef]
- Russo, V.; Masiello, D.; Trifuoggi, M.; Di Serio, M.; Tesser, R. Design of an adsorption column for methylene blue abatement over silica: From batch to continuous modeling. Chem. Eng. J. 2016, 302, 287–295. [Google Scholar] [CrossRef]
- Catauro, M.; Renella, R.A.; Papale, F.; Vecchio Ciprioti, S. Investigation of bioactivity, biocompatibility and thermal behavior of Sol-Gel silica glass containing a high peg percentage. Mater. Sci. Eng. C 2016, 61, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Vecchio Ciprioti, S.; Catauro, M.; Bollino, F.; Tuffi, R. Thermal behavior and dehydration kinetic study of SiO2/PEG hybrid gel glasses. Polym. Eng. Sci. 2017, 57, 606–612. [Google Scholar] [CrossRef]
- Catauro, M.; Bollino, F.; Mozzati, M.C.; Ferrara, C.; Mustarelli, P. Structure and magnetic properties of SiO2/PCL novel Sol-Gel organic-inorganic hybrid materials. J. Solid State Chem. 2013, 203, 92–99. [Google Scholar] [CrossRef]
- Lee, E.-J.; Teng, S.-H.; Jang, T.-S.; Wang, P.; Yook, S.-W.; Kim, H.-E.; Koh, Y.-H. Nanostructured poly(ε-caprolactone)–silica xerogel fibrous membrane for guided bone regeneration. Acta Biomater. 2010, 6, 3557–3565. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.-H.; Choi, J.-Y.; Kim, H.-M. Preparation of a bioactive and degradable poly(ε-caprolactone)/silica hybrid through a Sol−Gel method. Biomaterials 2002, 23, 4915–4921. [Google Scholar] [CrossRef]
- Tian, D.; Dubois, P.; Grandfils, C.; Jérôme, R.; Viville, P.; Lazzaroni, R.; Brédas, J.-L.; Leprince, P. A novel biodegradable and biocompatible ceramer prepared by the Sol−Gel process. Chem. Mater. 1997, 9, 871–874. [Google Scholar] [CrossRef]
- Catauro, M.; Bollino, F.; Papale, F. Surface modifications of titanium implants by coating with bioactive and biocompatible poly (ε-caprolactone)/SiO2 hybrids synthesized via Sol-Gel. Arab. J. Chem. 2014. [Google Scholar] [CrossRef]
- Bollino, F.; Armenia, E.; Tranquillo, E. Zirconia/hydroxyapatite composites synthesized via Sol-Gel: Influence of hydroxyapatite content and heating on their biological properties. Materials 2017, 10, 757. [Google Scholar] [CrossRef] [PubMed]
- Catauro, M.; Bollino, F.; Renella, R.A.; Papale, F. Sol-Gel synthesis of SiO2-CaO-P2O5 glasses: Influence of the heat treatment on their bioactivity and biocompatibility. Ceram. Int. 2015, 41, 12578–12588. [Google Scholar] [CrossRef]
- Chen, L.; Xu, L.-L. Research and preparing of SiO2-CaO-P2O5 bioactive material. Guisuanyan Tongbao 2009, 28, 686–691. [Google Scholar]
- Ma, J.; Chen, C.Z.; Wang, D.G.; Meng, X.G.; Shi, J.Z. Influence of the sintering temperature on the structural feature and bioactivity of Sol-Gel derived SiO2-CaO-P2O5 bioglass. Ceram. Int. 2010, 36, 1911–1916. [Google Scholar] [CrossRef]
- Voicu, G.; Popa, A.M.; Badanoiu, A.I.; Iordache, F. Influence of thermal treatment conditions on the properties of dental silicate cements. Molecules 2016, 21, 233. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Münstedt, H. Polyamide/silver antimicrobials: Effect of crystallinity on the silver ion release. Polym. Int. 2005, 54, 1180–1186. [Google Scholar] [CrossRef]
- Mavropoulos, E.; Costa, A.M.; Costa, L.T.; Achete, C.A.; Mello, A.; Granjeiro, J.M.; Rossi, A.M. Adsorption and bioactivity studies of albumin onto hydroxyapatite surface. Colloids Surf. B Biointerfaces 2011, 83, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Radev, L. Influence of thermal treatment on the structure and in vitro bioactivity of Sol-Gel prepared CaO-SiO2-P2O5 glass-ceramics. Process. Appl. Ceram. 2014, 8, 155–166. [Google Scholar] [CrossRef]
- Papadopoulos, C.; Kantiranis, N.; Vecchio, S.; Lalia-Kantouri, M. Lanthanide complexes of 3-methoxy-salicylaldehyde. J. Therm. Anal. Calorim. 2010, 99, 931–938. [Google Scholar] [CrossRef]
- Persenaire, O.; Alexandre, M.; Degée, P.; Dubois, P. Mechanisms and kinetics of thermal degradation of poly(ε-caprolactone). Biomacromolecules 2001, 2, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Domán, A.; Madarász, J.; László, K. In situ evolved gas analysis assisted thermogravimetric (TG-FTIR and TG/DTA–MS) studies on non-activated copper benzene-1,3,5-tricarboxylate. Thermochim. Acta 2017, 647, 62–69. [Google Scholar] [CrossRef]
- Vogel, C.; Siesler, H.W. Thermal degradation of poly(ε-caprolactone), poly(l-lactic acid) and their blends with poly(3-hydroxy-butyrate) studied by TGA/FT-IR spectroscopy. Macromol. Symposia 2008, 265, 183–194. [Google Scholar] [CrossRef]
- Sarikaya, M.; Turan, M.D.; Aydogmus, R.; Yucel, A.; Kizilkaya, N.; Depci, T. Extraction of meso-pores amorphous SiO2 from van pumice. Curr. Phys. Chem. 2017, 7, 301–304. [Google Scholar] [CrossRef]
- Zhang, G.; Xu, Y.; Xu, D.; Wang, D.; Xue, Y.; Su, W. Pressure-induced crystallization of amorphous SiO2 with silicon–hydroxy group and the quick synthesis of coesite under lower temperature. High Press. Res. 2008, 28, 641–650. [Google Scholar] [CrossRef]
- Keen, D.A.; Dove, M.T. Local structures of amorphous and crystalline phases of silica, SiO2, by neutron total scattering. J. Phys. Condens. Matter 1999, 11, 9263. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vecchio Ciprioti, S.; Tuffi, R.; Dell’Era, A.; Dal Poggetto, F.; Bollino, F. Thermal Behavior and Structural Study of SiO2/Poly(ε-caprolactone) Hybrids Synthesized via Sol-Gel Method. Materials 2018, 11, 275. https://doi.org/10.3390/ma11020275
Vecchio Ciprioti S, Tuffi R, Dell’Era A, Dal Poggetto F, Bollino F. Thermal Behavior and Structural Study of SiO2/Poly(ε-caprolactone) Hybrids Synthesized via Sol-Gel Method. Materials. 2018; 11(2):275. https://doi.org/10.3390/ma11020275
Chicago/Turabian StyleVecchio Ciprioti, Stefano, Riccardo Tuffi, Alessandro Dell’Era, Francesco Dal Poggetto, and Flavia Bollino. 2018. "Thermal Behavior and Structural Study of SiO2/Poly(ε-caprolactone) Hybrids Synthesized via Sol-Gel Method" Materials 11, no. 2: 275. https://doi.org/10.3390/ma11020275
APA StyleVecchio Ciprioti, S., Tuffi, R., Dell’Era, A., Dal Poggetto, F., & Bollino, F. (2018). Thermal Behavior and Structural Study of SiO2/Poly(ε-caprolactone) Hybrids Synthesized via Sol-Gel Method. Materials, 11(2), 275. https://doi.org/10.3390/ma11020275








