Polyacetylene: Myth and Reality
Abstract
:1. Introduction/Background History
2. Summary of This Review
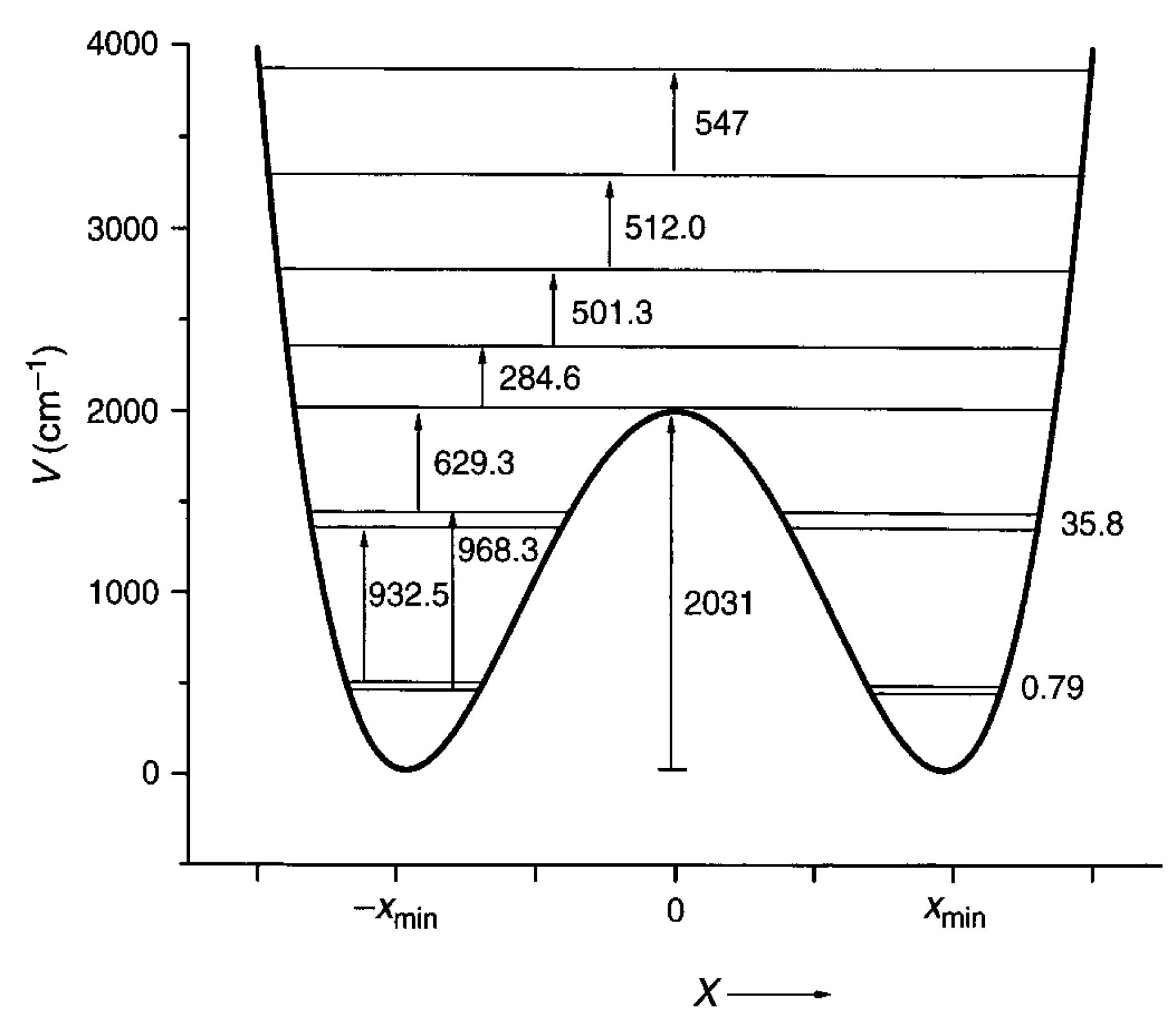
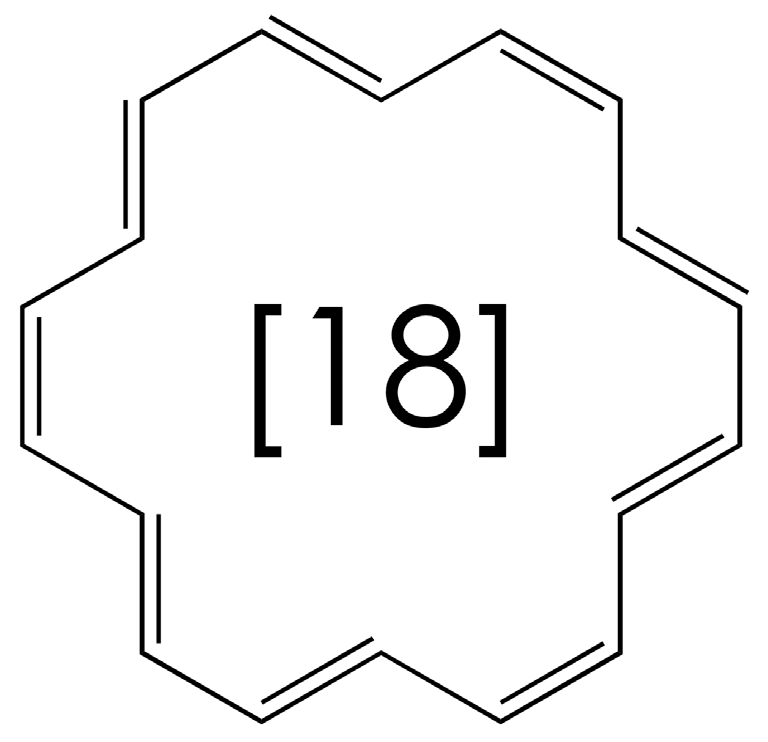
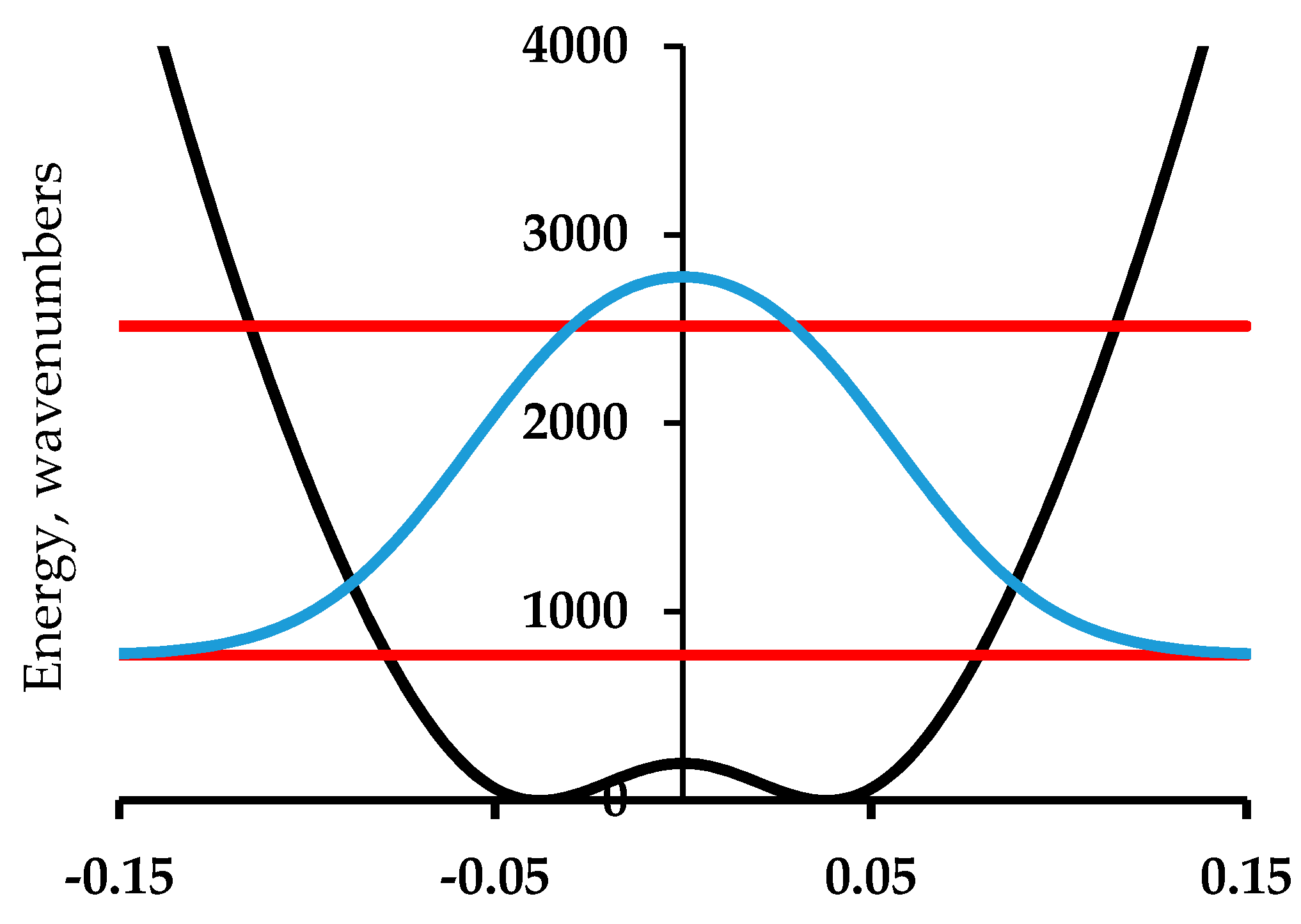
3. Double Minimum Potential Vibrational Energy Levels: Ammonia and [18]-Annulene
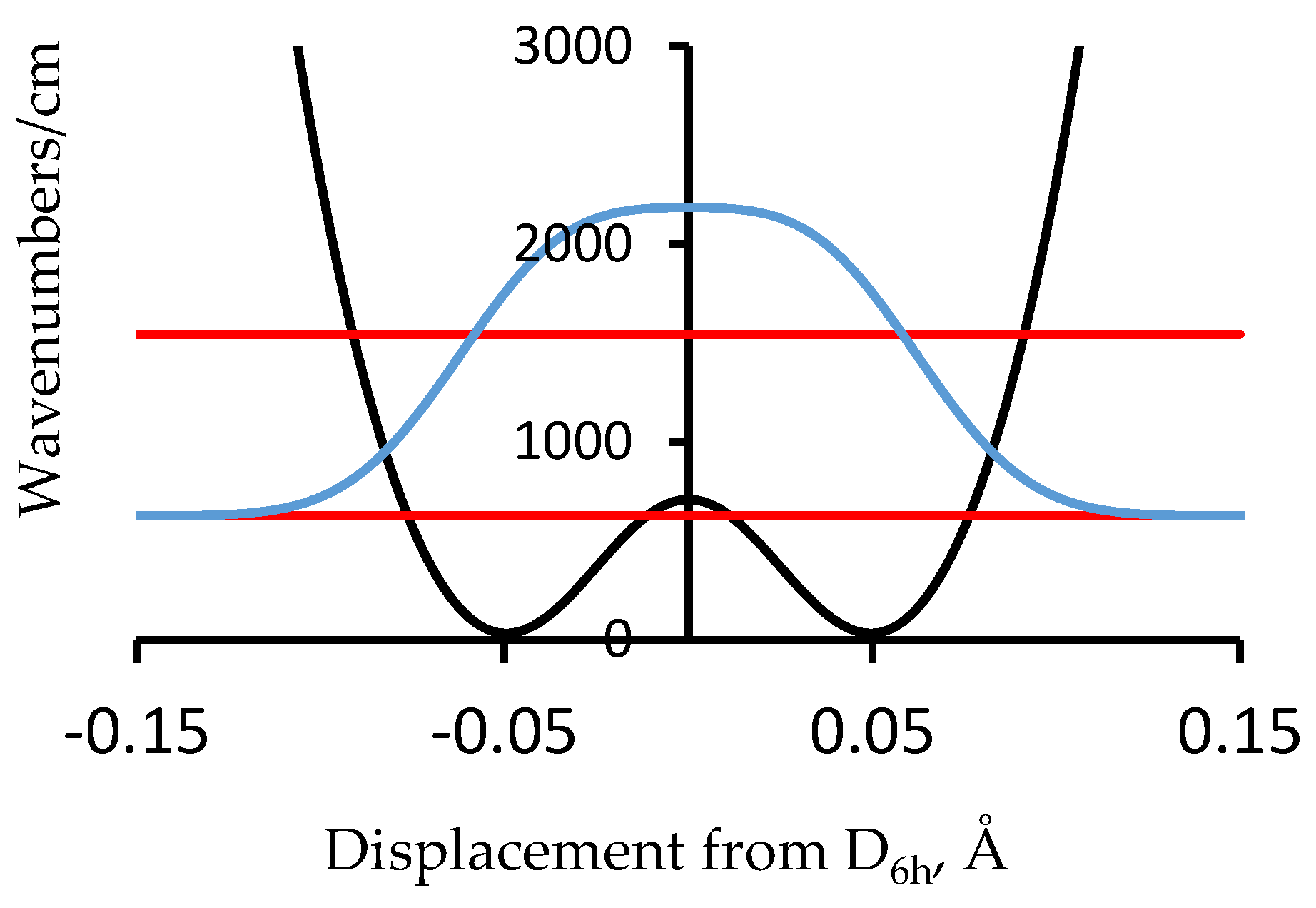
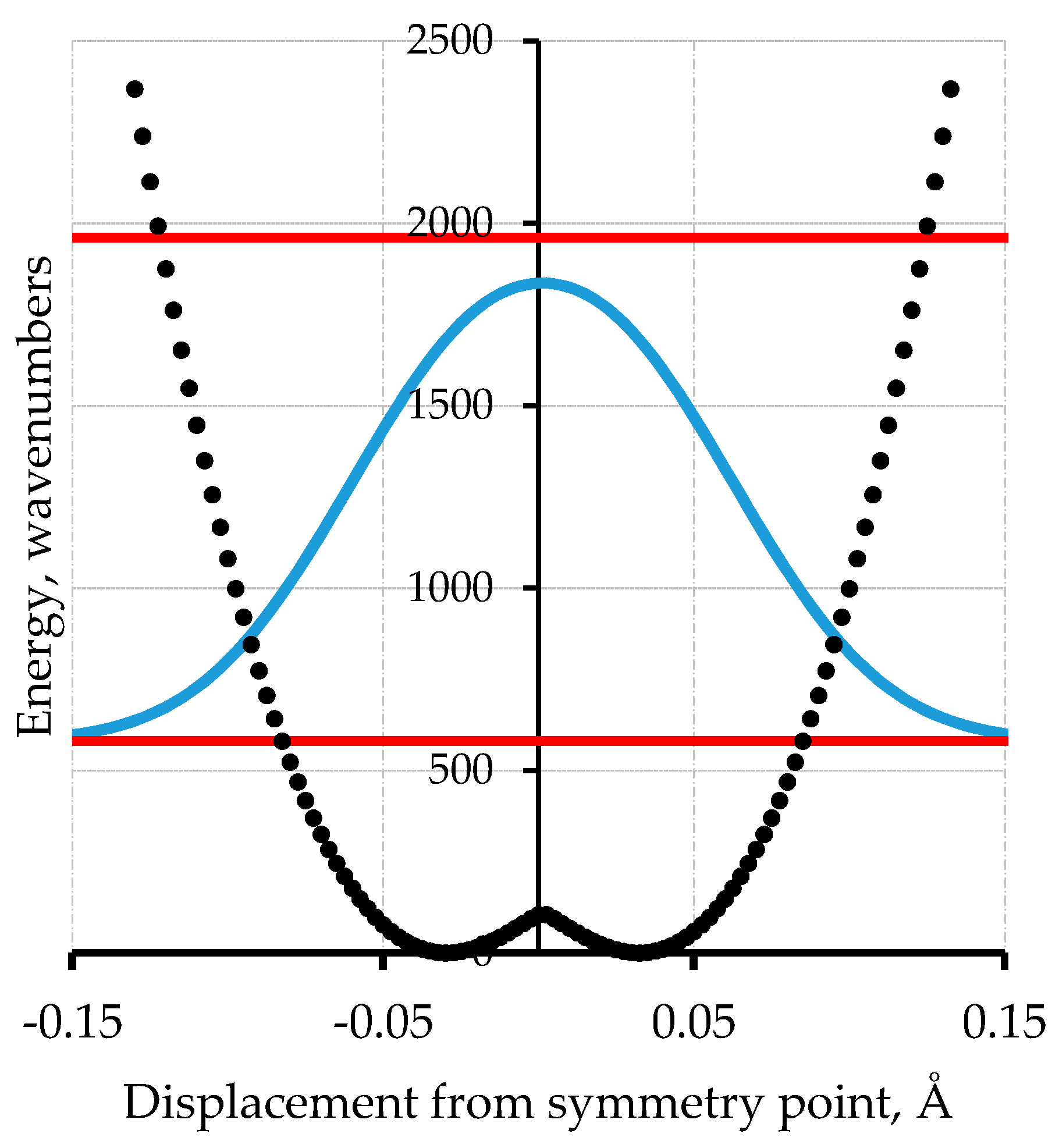
4. Double Minimum Potential Vibrational Energy Levels: Polyacetylene
5. Review of Experimental Observations on Polyacetylene with Emphasis on Bond Alternation
5.1. X-ray Diffraction
5.2. Infrared Dichroism
5.3. NMR Spectroscopy
5.4. Resonance Raman Spectroscopy
5.5. A Cautionary Note on Doping
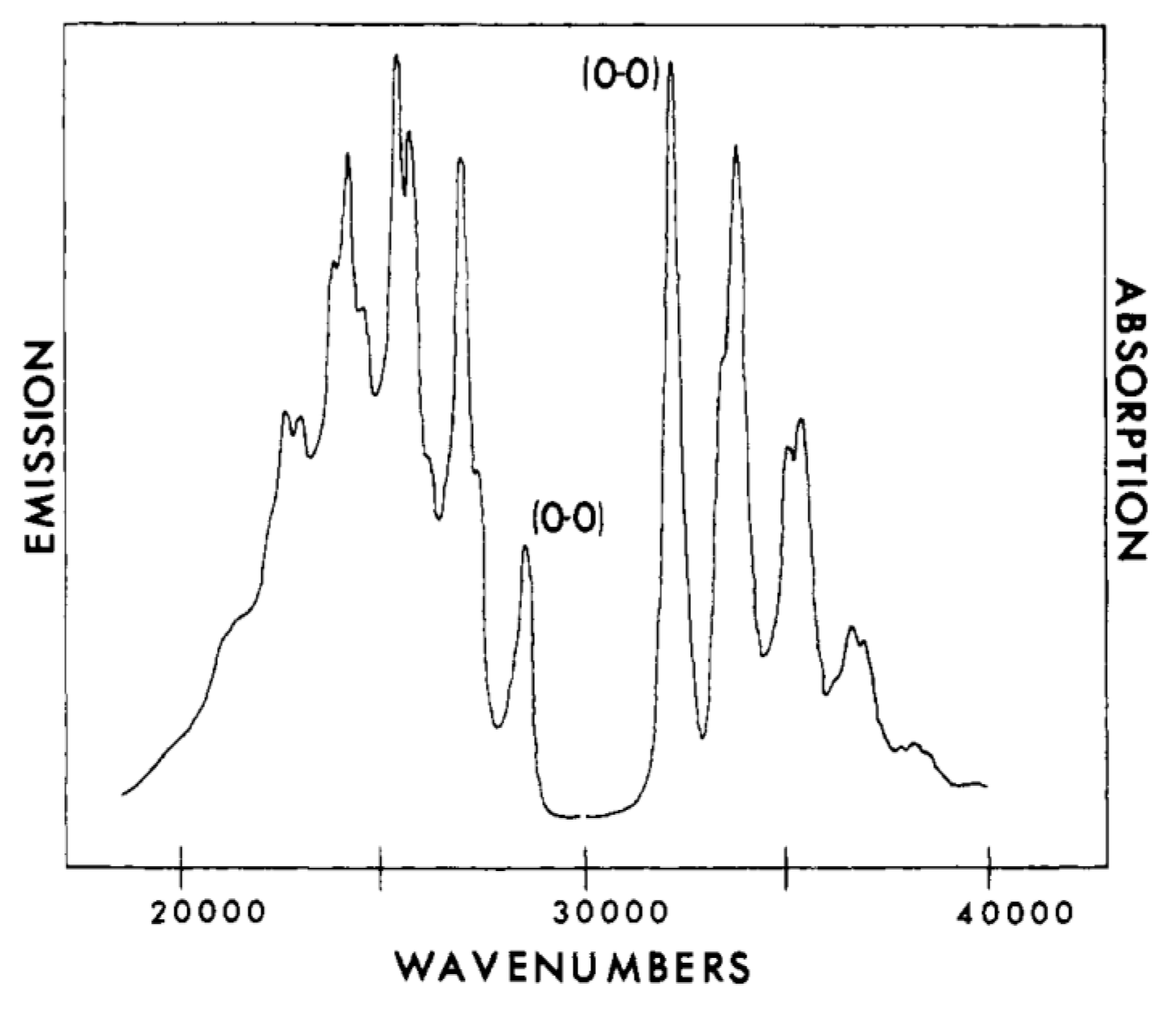
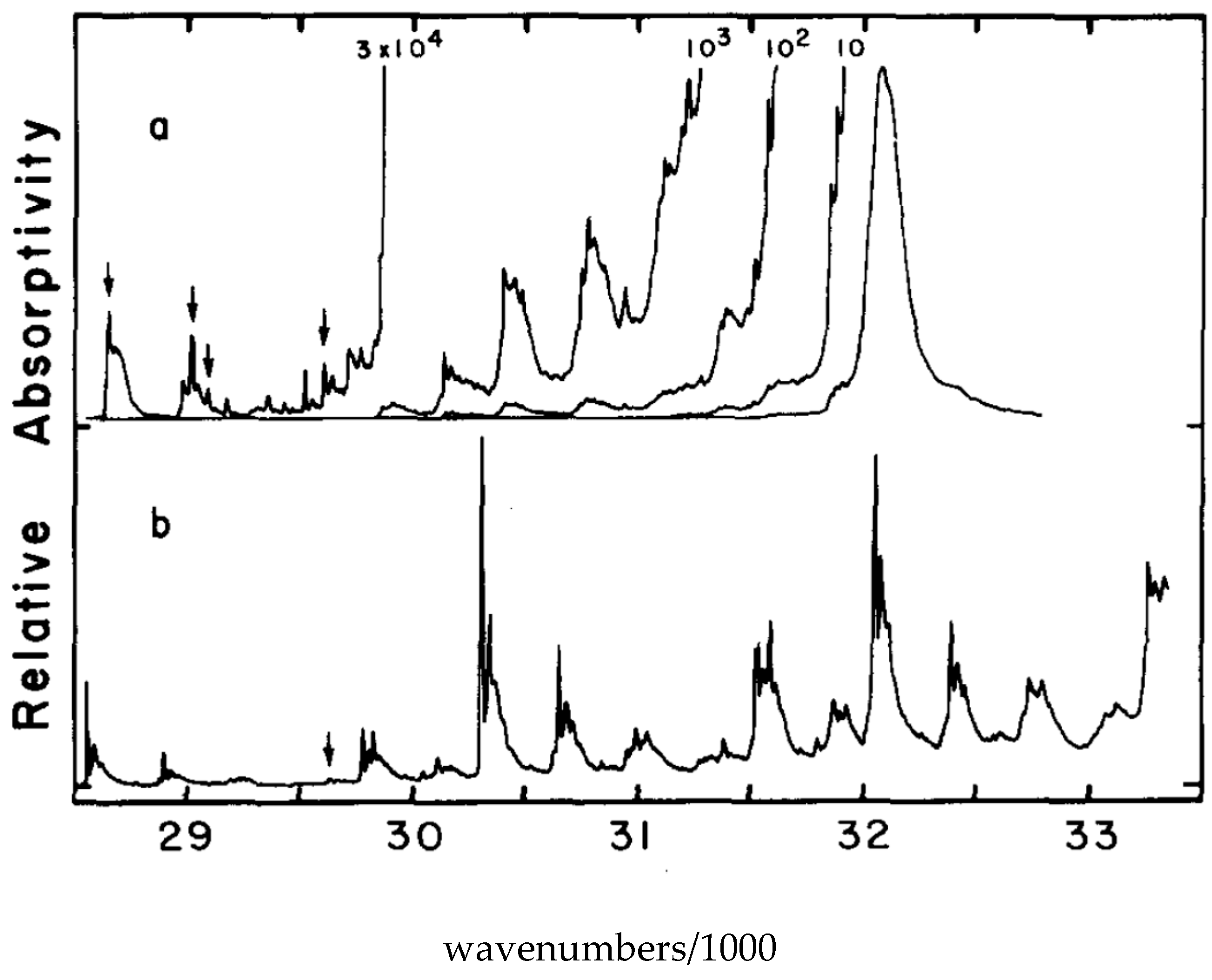
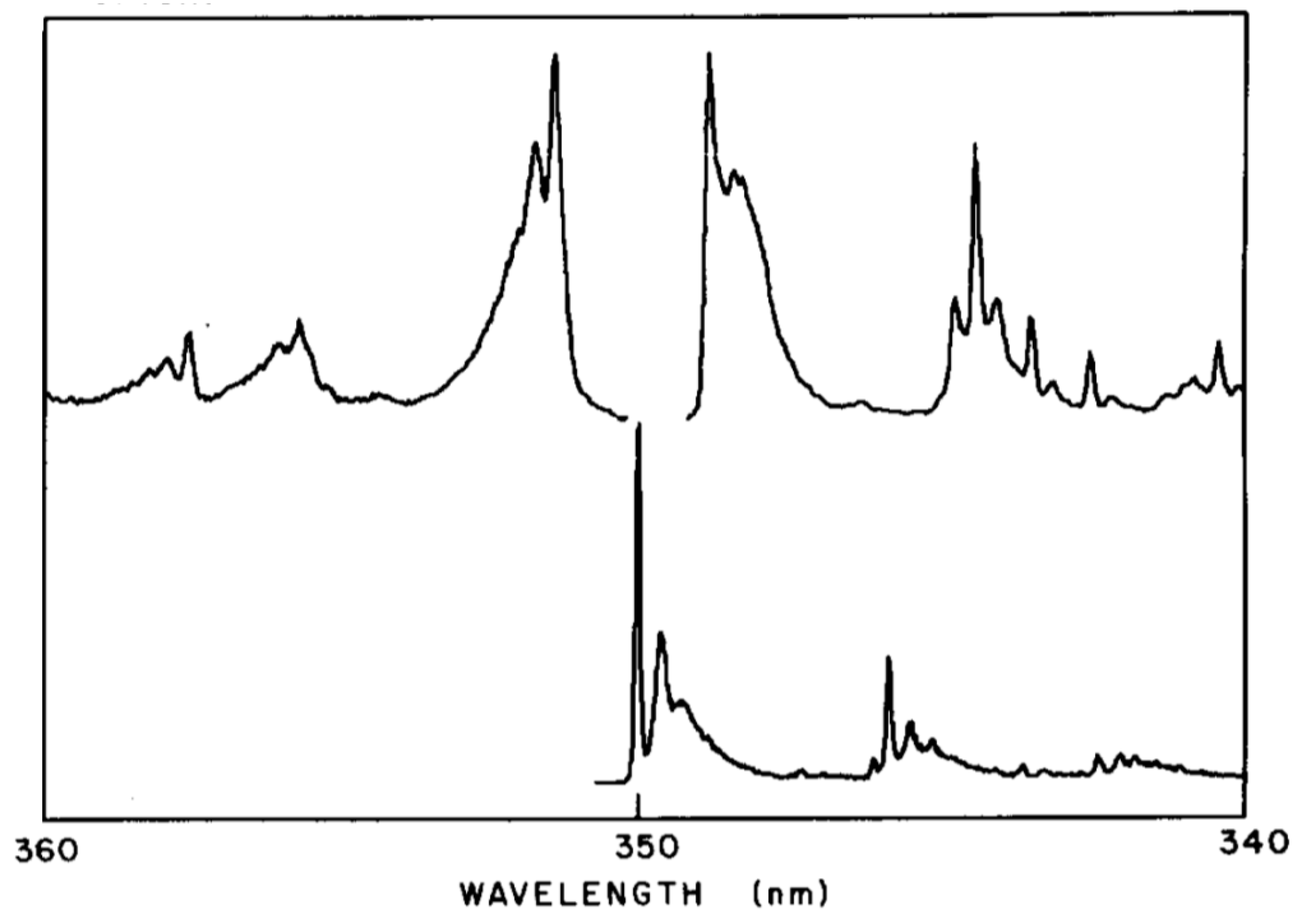
6. Electronic Spectroscopy of Finite Linear Conjugated Polyenes
7. Raman Vibrational Spectroscopy of Finite Polyenes
- synthesis of these compounds is limited by solubility to N =12, i.e., N > 12 are insoluble;
- there are two strong Raman features near 1100 cm−1 and in the 1600–1500 cm−1 region;
- the lower C–C mode is not very sensitive to chain length;
- the higher C=C mode moves to lower frequency as the chain elongates;
- when plotted vs. 1/N, this C=C mode extrapolates to a value of ca. 1440 cm−1;
- the integrated intensity of the C–C mode increases relative the C=C mode as N increases.
- there is a loss of mass corresponding quantitatively to the loss of iodine;
- loss of Raman intensity due to the decreasing effect of a one-electron excitation on a large chain.
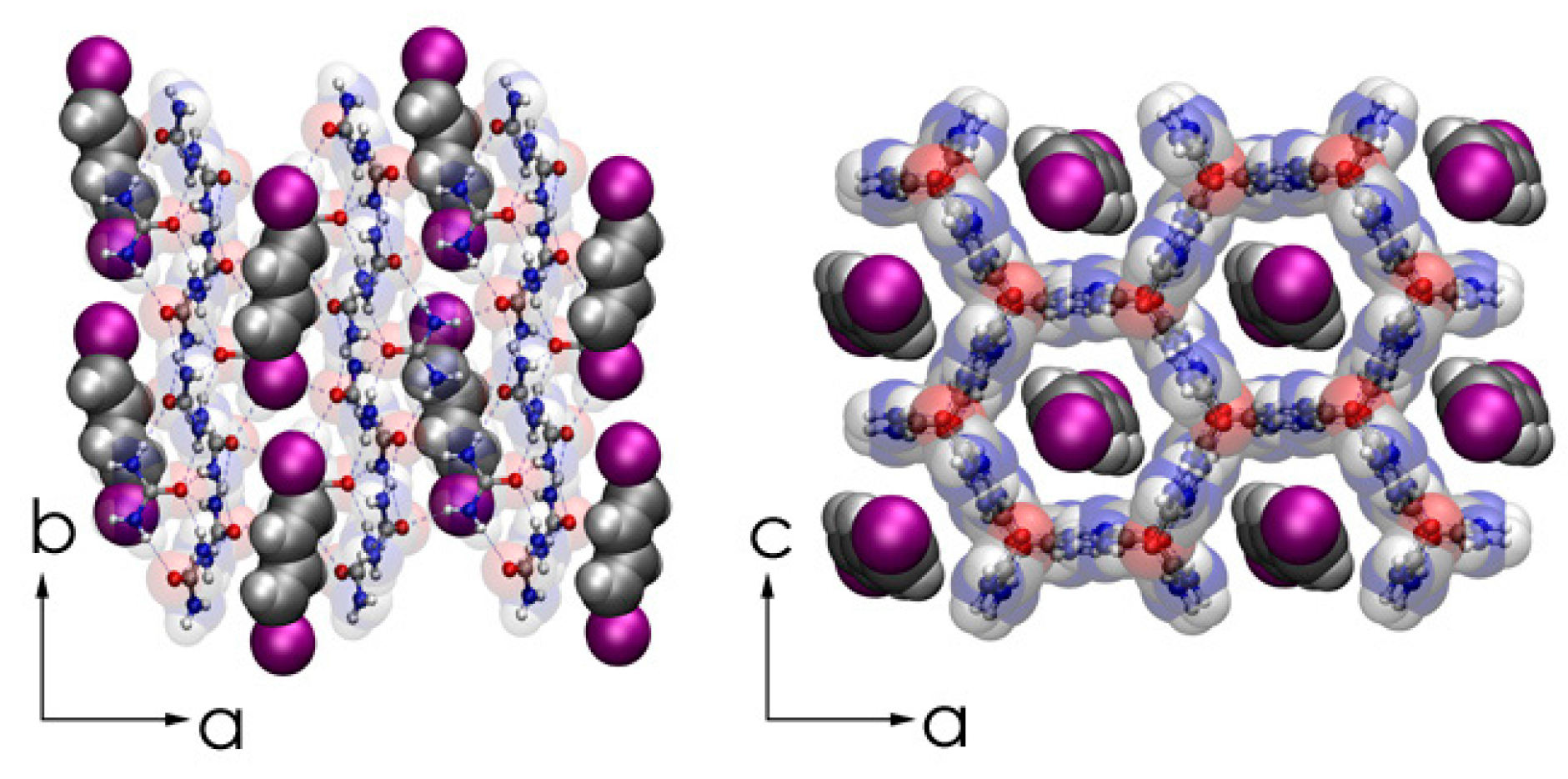
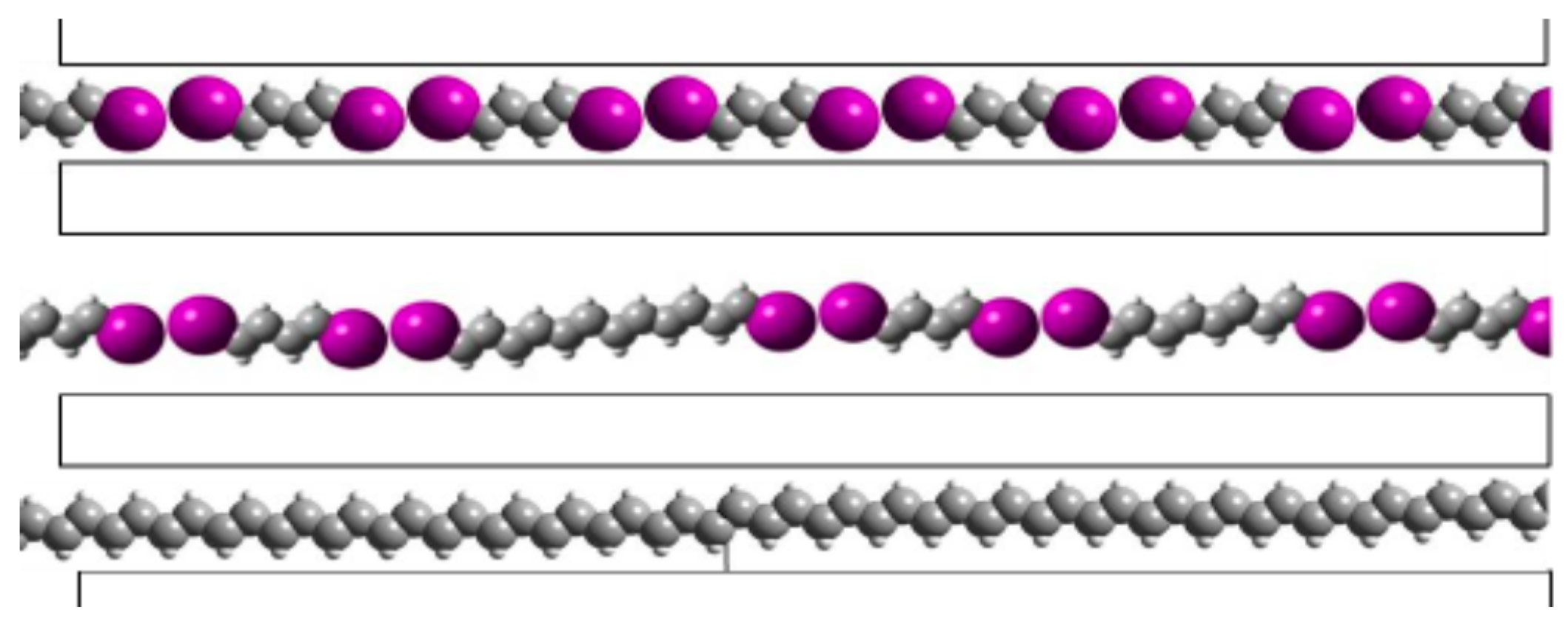
8. In Situ Synthesis of Oriented Insulated Polyacetylene
9. Summary of Lessons from the Literature on Polyacetylene
Acknowledgments
Conflicts of Interest
References
- Chiang, C.K.; Fincher, C.R.; Park, Y.W.; Heeger, A.J.; Shirakawa, H.; Louis, E.J.; Gau, S.C.; MacDiarmid, A.G. Electrical conductivity in doped polyacetylene. Phys. Rev. Lett. 1977, 39, 1098–1101. [Google Scholar] [CrossRef]
- Swager, T.M. 50th Anniversary Perspective: Conducting/Semiconducting Conjugated Polymers; A Personal Perspective on the Macromolecules; ACS Publications: Washington, DC, USA, 2017; Volume 50, pp. 4867–4886. [Google Scholar] [CrossRef]
- Ovchinnikov, A.A.; Ukrainskii, I.I.; Kventsel, G.F. Theory of one-dimensional Mott semiconductors and the electronic structure of long molecules with conjugated bonds. Uspekhi Fiziceskih Nauk 1972, 108, 81–111. [Google Scholar] [CrossRef]
- Suhai, S. Bond alternation in infinite polyene: Peierls distortion reduced by electron correlation. Chem. Phys. Lett. 1983, 96, 619–625. [Google Scholar] [CrossRef]
- Guo, H.; Paldus, J. Estimates of the Structure and Dimerization Energy of Polyacetylene from Ab Initio Calculations on Finite Polyenes. Int. J. Quant. Chem. 1997, 63, 345–360. [Google Scholar] [CrossRef]
- Lippincott, E.R.; White, C.E.; Siblia, J.P. Vibrational spectra and geometrical configuration of 1,3,5-hexatriene. J. Am. Chem. Soc. 1958, 80, 2926–2930. [Google Scholar] [CrossRef]
- Lippincott, E.R.; Kenney, T.E. Vibrational spectra and geometric configuration of cis-1,3,5-hexatriene. J. Am. Chem. Soc. 1962, 84, 3641–3648, (includes updated data for trans form.). [Google Scholar] [CrossRef]
- Guo, H.; Karplus, M. Ab initio studies of polyenes. I. 1,3-Butadiene. J. Chem. Phys. 1991, 94, 3679–3699. [Google Scholar] [CrossRef]
- Laane, J. Eigenvalues of the Potential Function V = z4 ± Bz2 and the Effect of Sixth Power Terms. Appl. Spectrosc. 1970, 24, 73–80. [Google Scholar] [CrossRef]
- Bernath, P.F. Spectra of Atoms and Molecules; Oxford Univ. Press: New York, NY, USA, 1994; pp. 273–274. [Google Scholar]
- Dennison, D.M.; Uhlenbeck, G.E. The two-minima problem and the ammonia molecule. Phys. Rev. 1932, 41, 313–321. [Google Scholar] [CrossRef]
- Dennison, D.M. The infrared spectra of polyatomic molecules. II. Rev. Mod. Phys. 1940, 12, 175–214. [Google Scholar] [CrossRef]
- Aquino, N.; Campoy, G.; Yee-Madeira, H. The inversion potential for NH3 using a DFT approach. Chem. Phys. Lett. 1998, 296, 111–116. [Google Scholar] [CrossRef]
- Marston, C.C.; Balin-Kurti, G.G. The Fourier grid Hamiltonian method for bound state eigenvalues and eigenfunctions. J. Chem. Phys 1989, 91, 3571–3576. [Google Scholar] [CrossRef]
- Johnson III, R.D. Fourier Grid Hamiltonian (FGH) 1D Program. Available online: https://www.nist.gov/chemical-informatics-research-group/products-and-services/fourier-grid-hamiltonian-fgh-1d-program (accessed on 26 December 2017).
- Wannere, C.S.; Sattelmeyer, K.W.; Schaefer III, H.F.; Schleyer, P.V.R. Aromaticity: The alternating C–C bond length structures of [14]-, [18]-, and [22]annulene. Angew. Chem. Int. Ed. 2004, 43, 4200–4206. [Google Scholar] [CrossRef] [PubMed]
- Hudson, B.S.; Allis, D.G. The Structure of [18]-annulene: Computed Raman Spectra, Zero-point Level and Proton NMR Chemical Shifts. J. Mol. Struct. 2012, 1023, 212–215. [Google Scholar] [CrossRef]
- Kwan, E.E.; Liu, R.Y. Enhancing NMR Prediction for Organic Compounds Using Molecular Dynamics. J. Chem. Theory Comp. 2015, 11, 5083–5089. [Google Scholar] [CrossRef] [PubMed]
- Hudson, B.S.; Allis, D.G. Bond alternation in infinite periodic polyacetylene: Dynamical treatment of the anharmonic potential. J. Mol. Struct. 2013, 1032, 78–82. [Google Scholar] [CrossRef]
- Born, M.; Kármán, T.V. Vibrations in Space-gratings (Molecular Frequencies). Phys. Z. 1912, 13, 297–309. [Google Scholar]
- Wilson, E.B., Jr.; Decius, J.C.; Cross, P.C. Molecular Vibrations: The Theory of Infrared and Raman Vibrational Spectra (Dover Books on Chemistry); Courier Corporation: North Chelmsford, MA, USA, 1980. [Google Scholar]
- Fincher, C.R., Jr.; Chen, C.E.; Heeger, A.J.; MacDiarmid, A.G.; Hastings, J.B. Structural determination of the symmetry-breaking parameter in trans-polyacetylene (CH)x. Phys. Rev. Lett. 1982, 48, 100–104. [Google Scholar] [CrossRef]
- Chien, J.C.W. Polyacetylene: Chemistry, Physics, and Materials Science; Academic Press: New York, NY, USA, 1984; pp. 112–117. ISBN 0-12-172460-3. [Google Scholar]
- Zhu, Q.; Fischer, J.E.; Zuzok, R.; Roth, S. Crystal structure of polyacetylene revisited: An X-ray study. Solid State Commun. 1992, 83, 179–183. [Google Scholar] [CrossRef]
- Zicovich-Wilson, C.M.; Kirtman, B.; Civalleri, B.; Ramirez-Solis, A. Periodic density functional theory calculations for 3-dimensional polyacetylene with empirical dispersion terms. Phys. Chem. Chem. Phys. 2010, 1, 3289–3293. [Google Scholar] [CrossRef] [PubMed]
- Castiglioni, C.; Zerbi, G.; Gussoni, M. Peierls distortion in trans-polyacetylene—Evidence from infrared intensities. Solid State Commun. 1985, 56, 863–866. [Google Scholar] [CrossRef]
- Maricq, M.M.; Waugh, J.S.; MacDiarmid, A.G.; Shirakawa, H.; Heeger, A.J. Carbon-13 nuclear magnetic resonance of cis- and trans-polyacetylenes. J. Am. Chem. Soc. 1978, 100, 7729–7730. [Google Scholar] [CrossRef]
- Yannoni, C.S.; Clarke, T.C. Molecular Geometry of cis- and trans-Polyacetylene by Nutation NMR Spectroscopy. Phys. Rev. Lett. 1983, 51, 1191–1193. [Google Scholar] [CrossRef]
- Scott, J.C.; Clarke, T.C. Nuclear magnetic relaxation in polyacetylene. J. Phys. Colloq. 1983, 44, 365–368. [Google Scholar] [CrossRef]
- Izmaylov, A.F.; Scuseria, G.E. Efficient evaluation of analytic vibrational frequencies in Hartree-Fock and density functional theory for periodic nonconducting systems. J. Chem. Phys. 2007, 127, 144106/1–144106/9. [Google Scholar] [CrossRef] [PubMed]
- Lefrant, S.; Lichtmann, L.S.; Temkin, H.; Fitchen, D.B.; Miller, D.C.; Whitewell II, G.E.; Burlitch, J.M. Raman scattering in (polyacetylene) and (polyacetylene) treated with bromine and iodine. Solid State Commun. 1979, 29, 191–196. [Google Scholar] [CrossRef]
- Brivio, P.; Mulazzi, E. Absorption and resonant Raman scattering from trans-polyacetylene. Chem. Phys. Lett. 1983, 95, 555–560. [Google Scholar] [CrossRef]
- Mulazzi, E.; Brivio, P.; Falques, E.; Lefrant, S. Experimental and theoretical Raman results in transpolyacetylene. Solid State Commun. 1983, 46, 851–855. [Google Scholar] [CrossRef]
- Brivio, P.; Mulazzi, E. Theoretical analysis of absorption and resonant Raman scattering spectra of trans-polyacetylene ((CH)x). Phys. Rev. B 1984, 30, 876–882. [Google Scholar] [CrossRef]
- Schen, M.A.; Chien, J.C.W.; Perrin, E.; Lefrant, S.; Mulazzi, E. Resonant Raman scattering of controlled molecular weight polyacetylene. J. Chem. Phys. 1988, 89, 7615–7620. [Google Scholar] [CrossRef]
- Masetti, G.; Campani, E.; Gorini, G.; Piseri, L.; Tubino, R.; Piaggio, R.P.; Dellepiane, G. Resonance Raman-spectra of highly oriented trans-polyacetylene. Solid State Commun. 1985, 55, 737–742. [Google Scholar] [CrossRef]
- Kuzmany, H. Resonance Raman-Scattering from Neutral and Doped Polyacetylene. Phys. Status Soldi 1980, 97, 521–531. [Google Scholar] [CrossRef]
- Schuegerl, F.B.; Kuzmany, H. Optical modes of trans-polyacetylene. J. Chem. Phys. 1981, 74, 953–958. [Google Scholar] [CrossRef]
- Kuzmany, H.; Imhoff, E.A.; Fitchen, D.B.; Sarhangi, A. Franck-Condon approach for optical absorption and resonance Raman scattering in trans-polyacetylene. Phys. Rev. B 1982, 26, 7109–7112. [Google Scholar] [CrossRef]
- Kuzmany, H. The particle in the box model for resonance Raman scattering in polyacetylene. Pure Appl. Chem. 1985, 57, 235–246. [Google Scholar] [CrossRef]
- Kuzmany, H.; Knoll, P. Application of the particle in the box model for resonance Raman scattering to recent experimental results of polyacetylene. Mol. Cryst. Liq. Cryst. 1985, 117, 385–392. [Google Scholar] [CrossRef]
- Kuzmany, H.; Knoll, P. The Dispersion effect of Resonance Raman Lines in Trans-Polyacetylene, Springer Series in Solid-State Sciences. Electron. Prop. Polym. Relat. Compd. 1985, 63, 114–121. [Google Scholar]
- Eckhardt, H.; Steinhauser, S.W.; Chance, R.R.; Schott, M.; Silbey, R. Anti-Stokes Raman-scattering in trans polyacetylene. Solid State Commun. 1985, 55, 1075–1079. [Google Scholar] [CrossRef]
- Martin, R.M.; Falicov, L.M. Resonance Raman Scattering in Light Scattering in Solids; Cardona, M., Guntherodt, G., Eds.; Springer: New York, NY, USA, 1975; pp. 79–145. ISBN 03034216. [Google Scholar]
- Jin, B.; Silbey, R. Theory of resonance Raman scattering for finite and infinite polyenes. J. Chem. Phys. 1995, 102, 4251–4260. [Google Scholar] [CrossRef]
- Heller, E.J.; Yang, Y.; Kocia, L. Raman Scattering in Carbon Nanosystems: Solving Polyacetylene. ACS Cent. Sci. 2015, 1, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Thakur, M. A Class of Conducting Polymers Having Nonconjugated Backbones. Macromolecules 1988, 21, 661–664. [Google Scholar] [CrossRef]
- Shang, Q.; Pramanick, S.; Hudson, B. Chemical nature of conduction in iodine-doped trans-1,4-poly(buta-1,3-diene) and some of its derivatives: The presence of I3− and the effect of double-bond configuration. Macromolecules 1990, 23, 1886–1889. [Google Scholar] [CrossRef]
- Birks, J.B.; Dyson, D.J. The relations between the fluorescence and absorption properties of organic molecules. Proc. R. Soc. Lond. Ser. A 1963, 275, 135–148. [Google Scholar] [CrossRef]
- Birks, J.B.; Birch, D.J.S. Fluorescence of diphenyl- and retinolpolyenes. Chem. Phys. Lett. 1975, 31, 608–610. [Google Scholar] [CrossRef]
- Hausser, K.W.; Kuhn, R.; Smakula, A.; Kreuchen, K.H. Absorption of light and double bonds. I. Problem and methods. Z. Phys. Chem. 1935, B29, 363–370. [Google Scholar]
- Hausser, K.W.; Kuhn, R.; Smakula, A.; Hoffer, M. Absorption of light and double bonds. II. Polyene aldehydes and polyene carboxylic acids. Z. Phys. Chem. 1935, B29, 371–377. [Google Scholar]
- Hausser, K.W.; Kuhn, R.; Smakula, A.; Deutsch, A. Absorption of light and double bonds. III. Investigation in the furane series. Z. Phys. Chem. 1935, B29, 378–383. [Google Scholar]
- Hausser, K.W.; Kuhn, R.; Smakula, A. Absorption of light and double bonds. IV. Diphenylpolyenes. Z. Phys. Chem. 1935, B29, 384–389. [Google Scholar]
- Hausser, K.W.; Kuhn, R. Seitz, Absorption of light and double bonds. V. The absorption of compounds with conjugate double bonds of carbon at low temperature. Z. Phys. Chem. 1935, B29, 391–416. [Google Scholar]
- Hausser, K.W.; Kuhn, R.; Kuhn, E. Absorption of light and double bonds. VI. The fluorescence of diphenylpolyenes. Z. Phys. Chem. 1935, B29, 417–454. [Google Scholar]
- Mulliken, R.S. Intensities of electronic transitions in molecular spectra. VII. Conjugated polyenes and carotenoids. J. Chem. Phys. 1939, 7, 364–373. [Google Scholar] [CrossRef]
- D’Amico, K.L.; Manos, C.; Christensen, R.L. Electronic energy levels in a homologous series of unsubstituted linear polyenes. J. Am. Chem. Soc. 1980, 102, 1777–1782. [Google Scholar] [CrossRef]
- Granville, M.F.; Holtom, G.R.; Kohler, B.E.; Christensen, R.L.; D’Amico, K.L. Experimental confirmation of the dipole forbidden character of the lowest excited singlet state in 1,3,5,7-octatetraene. J. Chem. Phys. 1979, 70, 593–594. [Google Scholar] [CrossRef]
- Granville, M.F.; Holtom, G.R.; Kohler, B.E. High-resolution one and two photon excitation spectra of trans, trans-1,3,5,7-octatetraene. J. Chem. Phys. 1980, 72, 4671–4675. [Google Scholar] [CrossRef]
- Hudson, B.S.; Kohler, B.E. Polyene spectroscopy. Lowest energy excited singlet state of diphenyloctatetraene and other linear polyenes. J. Chem. Phys. 1973, 59, 4984–5002. [Google Scholar] [CrossRef]
- Adamson, G.; Gradl, G.; Kohler, B.E. Photochemical hole burning for 1,3,5,7-octatetraene in n-hexane. J. Chem. Phys. 1989, 90, 3038–3042. [Google Scholar] [CrossRef]
- Kohler, B.E. Electronic Properties of Linear Polyenes; Conjugated Polymers; Brédas, J.L., Silbey, R.J., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1991; pp. 405–434. [Google Scholar]
- Kohler, B.E. Octatetraene photoisomerization. Chem. Rev. 1993, 93, 41–54. [Google Scholar] [CrossRef]
- Hudson, B.S.; Kohler, B.E. Low-lying weak transition in the polyene α,ω-diphenyl-octatetraene. Chem. Phys. Lett. 1972, 14, 299–304. [Google Scholar] [CrossRef]
- Hudson, B.; Kohler, B. Linear polyene electronic structure and spectroscopy. Annu. Rev. Phys. Chem. 1974, 25, 437–460. [Google Scholar] [CrossRef]
- Hudson, B.S.; Kohler, B.E.; Schulten, K. Linear polyene electronic structure and potential surfaces. Excit. States 1982, 6, 1–95. [Google Scholar]
- Hudson, B.; Kohler, B. Electronic structure and spectra of finite linear polyenes. Synth. Met. 1984, 9, 241–253. [Google Scholar] [CrossRef]
- Schulten, K.; Karplus, M. Origin of a low-lying forbidden transition in polyenes and related molecules. Chem. Phys. Lett. 1972, 14, 305–309. [Google Scholar] [CrossRef]
- Schulten, K.; Ohmine, I.; Karplus, M. Correlation effects in the spectra of polyenes. J. Chem. Phys. 1976, 64, 4422. [Google Scholar] [CrossRef]
- Tavan, P.; Schulten, K. The low-lying electronic excitations in long polyenes: A PPP-MRD-CI study. J. Chem. Phys. 1986, 85, 6602–6609. [Google Scholar] [CrossRef]
- Tavan, P.; Schulten, K. Electronic excitations in finite and infinite polyenes. Phys. Rev. B 1987, 36, 4337–4358. [Google Scholar] [CrossRef]
- Angeli, C.; Pastore, M. The lowest singlet states of octatetraene revisited. J. Chem. Phys. 2011, 134, 184302. [Google Scholar] [CrossRef] [PubMed]
- Torii, H.; Tasumi, M. Changes in the electronic structures of trans-polyenes in the 11Ag and 21Ag states induced by molecular vibrations. Chem. Phys. Lett. 1996, 260, 195–200. [Google Scholar] [CrossRef]
- Christensen, R.L.; Enriquez, M.M.; Wagner, N.L.; Peacock-Villada, A.Y.; Scriban, C.; Schrock, R.R.; Polivka, T.; Frank, H.A.; Birge, R.R. Energetics and Dynamics of the Low-Lying Electronic States of Constrained Polyenes: Implications for Infinite Polyenes. J. Phys. Chem. A 2013, 117, 1449–1465. [Google Scholar] [CrossRef] [PubMed]
- Orlandi, G.; Zerbetto, F. Vibronic coupling in polyenes: The frequency increase of the active C = C ag stretching mode in the absorption spectra. Chem. Phys. 1986, 108, 187–195. [Google Scholar] [CrossRef]
- Zerbetto, F.; Zgierski, M.Z.; Orlandi, G. Correlation between the frequency of the Franck-Condon active carbon:carbon ag stretch vibration and the excitation energy of the 1Bu electronic state in polyenes. Chem. Phys. Lett. 1987, 141, 138–142. [Google Scholar] [CrossRef]
- Buma, W.J.; Zerbetto, F. The large 11Ag−-21Ag− C=C and C–C stretch vibronic interaction in all-trans polyenes. Chem. Phys. Lett. 1998, 289, 118–124. [Google Scholar] [CrossRef]
- Buma, W.J.; Zerbetto, F. Modeling the Spectroscopy of the Lowest Excited Singlet State of cis,trans-1,3,5,7-Octatetraene: The Role of Symmetry Breaking and Vibronic Interactions. J. Phys. Chem. A 1999, 103, 2220–2226. [Google Scholar] [CrossRef]
- Fuss, W.; Haas, Y.; Zilberg, S. Twin states and conical intersections in linear polyenes. Chem. Phys. 2000, 259, 273–295. [Google Scholar] [CrossRef]
- Schaffer, H.E.; Chance, R.R.; Silbey, R.J.; Knoll, K.; Schrock, R.R. Conjugation length dependence of Raman scattering in a series of linear polyenes: Implications for polyacetylene. J. Chem. Phys. 1991, 94, 4161–4170. [Google Scholar] [CrossRef]
- Hollingsworth, M.D.; Harris, K.D.M. Urea, Thiourea, and Selenourea. In Comprehensive Supramolecular Chemistry; Solid State Supramolecular Chemistry: Crystal Engineering; Atwood, J.L., Davies, J.E.D., MacNicol, D.D., Vogtle, F., Eds.; Elsevier: Oxford, UK, 1996; Volume 6, pp. 177–237. [Google Scholar]
- Harris, K.D.M. Fundamental and applied aspects of urea and thiourea inclusion compounds. Supramol. Chem. 2007, 19, 47–53. [Google Scholar] [CrossRef]
- Harris, K.D.M.; Palmer, B.A.; Edwards-Gau, G.R. Reactions in Solid-State Inclusion Compounds. In Supramolecular Chemistry: From Molecules to Nanomaterials; Gale, P., Steed, J., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2012; Volume 4, pp. 1589–1612. ISBN 978-0-470-74640-0. [Google Scholar]
- Lashua, A.F.; Smith, T.M.; Hu, H.; Wei, L.; Allis, D.G.; Sponsler, M.B.; Hudson, B.S. Commensurate urea inclusion crystals with the guest (E,E)-1,4-diiodo-1,3-butadiene. Cryst. Growth Des. 2013, 13, 3852–3855. [Google Scholar] [CrossRef]
- Dincă, S.A.; Allis, D.G.; Lashua, A.F.; Sponsler, M.B.; Hudson, B.S. Insulated polyacetylene chains in an inclusion compound by photopolymerization. MRS Online Proc. Libr. Arch. 2015, 1799, 1–6. [Google Scholar] [CrossRef]
- Schuehler, D.E.; Williams, J.E.; Sponsler, M.B. Polymerization of acetylene with a ruthenium olefin metathesis catalyst. Macromolecules 2004, 37, 6255–6257. [Google Scholar] [CrossRef]
- Marti-Rujas, J.; Desmedt, A.; Harris, K.D.M.; Guillaume, F. Kinetics of molecular transport in a nanoporous crystal studied by confocal raman microspectrometry: Single-file diffusion in a densely filled tunnel. J. Phys. Chem. B 2007, 111, 12339–12344. [Google Scholar] [CrossRef] [PubMed]
- Marti-Rujas, J.; Desmedt, A.; Harris, K.D.M.; Guillaume, F. Bidirectional transport of guest molecules through the nanoporous tunnel structure of a solid inclusion compound. J. Phys. Chem. C 2009, 113, 736–743. [Google Scholar] [CrossRef]

© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hudson, B.S. Polyacetylene: Myth and Reality. Materials 2018, 11, 242. https://doi.org/10.3390/ma11020242
Hudson BS. Polyacetylene: Myth and Reality. Materials. 2018; 11(2):242. https://doi.org/10.3390/ma11020242
Chicago/Turabian StyleHudson, Bruce S. 2018. "Polyacetylene: Myth and Reality" Materials 11, no. 2: 242. https://doi.org/10.3390/ma11020242
APA StyleHudson, B. S. (2018). Polyacetylene: Myth and Reality. Materials, 11(2), 242. https://doi.org/10.3390/ma11020242