A Comparison of Electrochemical Performance of Carbon Aerogels with Adsorption Metal Ions for Super Capacitors
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of the Carbon Aerogels
2.1.2. Determination of the Metal Ions
2.1.3. Preparation of Metallic CA Electrodes
2.2. Carbon Aerogel Adsorption Kinetics
2.3. Characterization
3. Results and Discussion
3.1. Adsorption Kinetics of CA-X
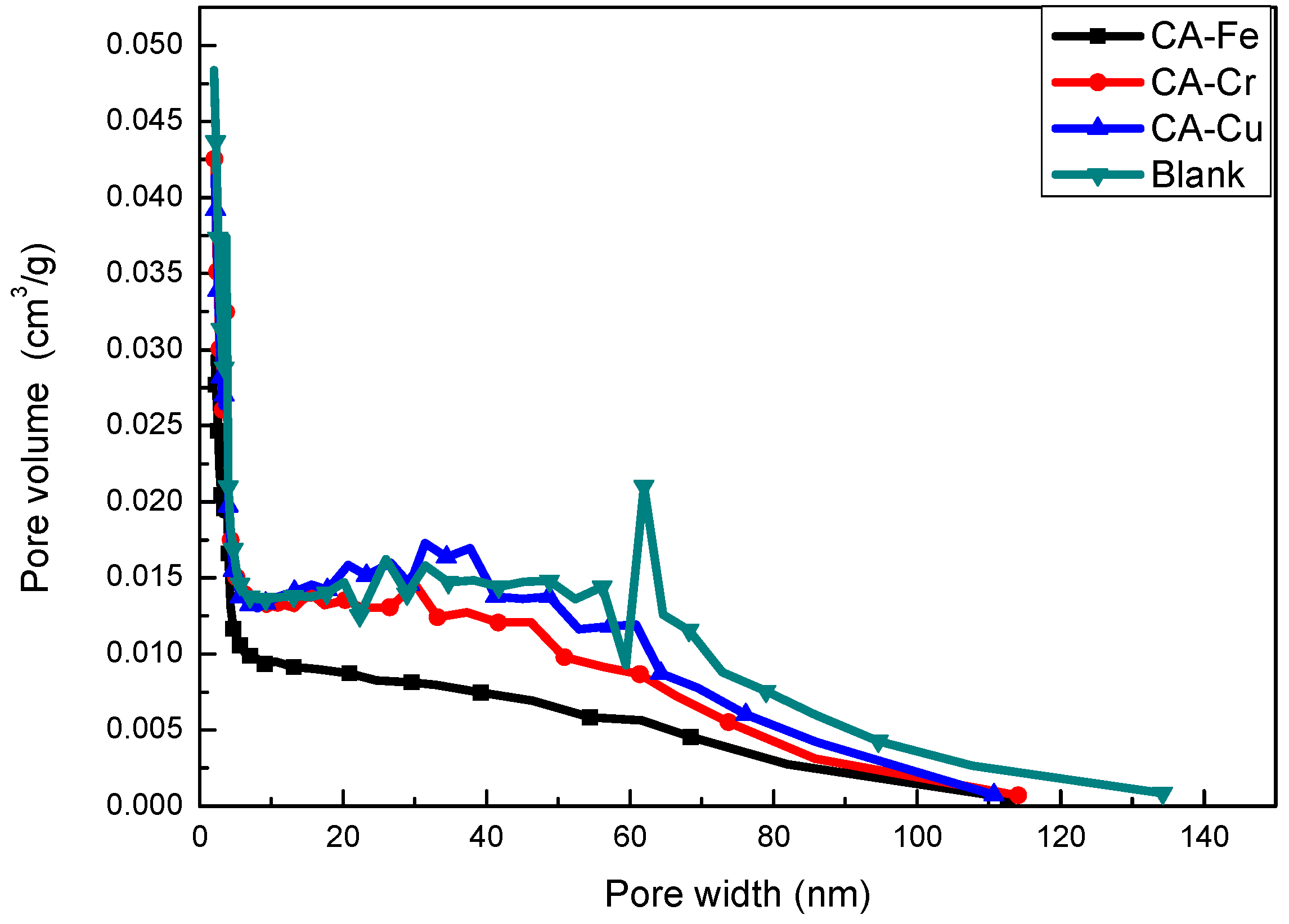
3.2. Pore structure of the CA-X
3.3. SEM and TEM of CA-X
3.4. XRD Patterns of CA-X
3.5. XPS Spectra of CA-X
3.6. Electrochemical Performances of CA-X
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Ge, F.; Li, M.M.; Ye, H.; Zhao, B.X. Effective removal of heavy metal ions Cd2+, Zn2+, Pb2+, Cu2+ from aqueous solution by polymer-modified magnetic nanoparticles. J. Hazard. Mater. 2003, 99, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Du, M.; Zhu, H.; Bao, S.; Yang, T.; Zou, M. Structure regulation of silica nanotubes and their adsorption behaviors for heavy metal ions: pH effect, kinetics, isotherms and mechanism. J. Hazard. Mater. 2015, 286, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Matlock, M.M.; Howerton, B.S.; Atwood, D.A. Chemical Precipitation of Lead from Lead Battery Recycling Plant Wastewater. Ind. Eng. Chem. Res. 2002, 41, 1579–1582. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Mohsen-Nia, M.; Montazeri, P.; Modarress, H. Removal of Cu2+ and Ni2+, from wastewater with a chelating agent and reverse osmosis processes. Desalination 2007, 217, 276–281. [Google Scholar] [CrossRef]
- Pekala, R.W.; Alviso, C.T.; Kong, F.M.; Hulsey, S.S. Aerogels derived from multifunctional organic monomers. J. Non-Cryst. Solids 1992, 145, 90–98. [Google Scholar] [CrossRef] [Green Version]
- Sadasivam, S.; Krishna, S.K.; Ponnusamy, K.; Nagarajan, G.S.; Kang, T.W.; Venkatesalu, S.C. Equilibrium and Thermodynamic Studies on the Adsorption of an Organophosphorous Pesticide onto “Waste” Jute Fiber Carbon. J. Chem. Eng. Data 2010, 55, 5658–5662. [Google Scholar] [CrossRef]
- Wu, M.; Jin, Y.; Zhao, G.; Li, M.; Li, D. Electrosorption-promoted Photodegradation of Opaque Wastewater on A Novel TiO2/Carbon Aerogel Electrode. Environ. Sci. Technol. 2010, 44, 1780–1785. [Google Scholar] [CrossRef] [PubMed]
- Kadirvelu, K.; Goel, J.; Rajagopal, C. Sorption of lead, mercury and cadmium ions in multi-component system using carbon aerogel as adsorbent. J. Hazard. Mater. 2008, 153, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Kabbashi, N.A.; Atieh, M.A.; Al-Mamun, A.; Mirghami, M.E.; Alam, M.D.Z.; Yahya, N. Kinetic adsorption of application of carbon nanotubes for Pb (II) removal from aqueous solution. J. Environ. Sci. 2009, 21, 539–544. [Google Scholar] [CrossRef]
- Li, J.; Zheng, L.; Liu, H. A novel carbon aerogel prepared for adsorption of copper (II) ion in water. J. Porous Mater. 2017, 24, 1575–1580. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, A.; Peng, F.; Yu, H.; Yang, J. Mechanism study on adsorption of acidified multiwalled carbon nanotubes to Pb (II). J. Colloid Interface Sci. 2007, 316, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Snook, G.A.; Kao, P.; Best, A.S. Conducting-polymer-based supercapacitor devices and electrodes. J. Power Sources 2011, 196, 1–12. [Google Scholar] [CrossRef]
- Yu, Z.; Tetard, L.; Zhai, L.; Thomas, J. Supercapacitor electrode materials: Nanostructures from 0 to 3 dimensions. Energy Environ. Sci. 2015, 8, 702–730. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, M.; Yang, Y.; Ran, F. Hybrid Electrode Material of Vanadium Nitride and Carbon Fiber with Cigarette Butt/Metal Ions Wastes as the Precursor for Supercapacitors. Electrochim. Acta 2016, 222, 1914–1921. [Google Scholar] [CrossRef]
- Hao, P.; Ma, X.; Xie, J.; Lei, F.; Li, L.; Zhu, W.; Cheng, X.; Cui, G.; Tang, B. Removal of toxic metal ions using chitosan coated carbon nanotube composites for supercapacitors. Sci. China Chem. 2018, 61, 797–805. [Google Scholar] [CrossRef]
- Yu, D.; Wang, H.; Yang, J.; Niu, Z.; Lu, H.; Yang, Y.; Cheng, L.; Guo, L. Dye Wastewater Cleanup by Graphene Composite Paper for Tailorable Supercapacitors. ACS Appl. Mater. Interfaces 2017, 9, 21298–21306. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, N.; Rinaldi, A.; Hamid, S.B.A. Synthesis and Adsorption Performance of Carbon Materials for the Removal of Iron (III) from Aqueous Solution. ACS Appl. Mech. Mater. 2015, 699, 988–993. [Google Scholar] [CrossRef]
- Zhao, Y.; Kunieda, M.; Obi, N.; Watanabe, S. Development of electrolyte filtration system for ECM taking into account removal of chromium (VI) ions. Precis. Eng. 2017, 49, 211–219. [Google Scholar] [CrossRef]
- Pang, J.S.; Deng, A.H.; Mao, L.B.; Chen, J.; Peng, X.J.; Zhu, J. Adsorption of Heavy Metal Ions with Magnetic Carbon-Coated Iron Nanoparitcles. Adv. Mater. Res. 2012, 490, 3049–3053. [Google Scholar] [CrossRef]
- Yuan, X.; An, N.; Zhu, Z.; Sun, H.; Zheng, J.; Jia, M.; Lu, C.; Zhang, W.; Liu, N. Hierarchically porous nitrogen–doped carbon materials as efficient adsorbent for removal of heavy metal ions. J. Dairy Sci. 2018, 119, 320–329. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Chem. Phys. 2015, 40, 1361–1403. [Google Scholar] [CrossRef]
- Xu, Y.; Yan, M.; Wang, S.; Zhang, L.; Liu, H.; Liu, Z. Synthesis, characterization and electrochemical properties of carbon aerogels using different organic acids as polymerization catalysts. J. Porous Mater. 2017, 24, 1375–1381. [Google Scholar] [CrossRef]
- Chen, J.P.; Wu, S.; Chong, K.H. Surface modification of a granular activated carbon by citric acid for enhancement of copper adsorption. Carbon 2003, 41, 1979–1986. [Google Scholar] [CrossRef]
- Chen, J.P.; Wu, S. Acid/Base-treated activated carbons: Characterization of functional groups and metal adsorptive properties. Langmuir 2004, 20, 2233–2242. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Li, Z.; Xiao, D.; Xiong, P.; Ye, N. Sulfonated multi-walled carbon nanotubes for the removal of copper (II) from aqueous solutions. J. Ind. Eng. Chem. 2014, 20, 1765–1771. [Google Scholar] [CrossRef]
- Li, Y.; Liu, F.; Xia, B.; Du, Q.; Zhang, P.; Wang, D.; Wang, Z.; Xia, Y. Removal of copper from aqueous solution by carbon nanotube/calcium alginate composites. J. Hazard. Mater. 2010, 177, 876–880. [Google Scholar] [CrossRef] [PubMed]
- Ademiluyi, F.T.; Ujile, A.A. Kinetics of Batch Adsorption of Iron II Ions from Aqueous Solution using Activated Carbon from Nigerian Bamboo. Int. J. Eng. Technol. 2013, 3, 623–631. [Google Scholar]
- Huang, R.; He, L.; Zhang, T.; Li, D.; Tang, P.; Feng, Y. Novel Carbon Paper@Magnesium Silicate Composite Porous Films: Design, Fabrication, and Adsorption Behavior for Heavy Metal Ions in Aqueous Solution. ACS Appl. Mater. Interfaces 2018, 10, 22776–22785. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Luo, C.; Sun, M.; Qiu, H. Synthesis of graphene oxide decorated with magnetic cyclodextrin for fast chromium removal. J. Mater. Chem. 2012, 22, 24577–24583. [Google Scholar] [CrossRef]
- Mishra, S.; Verma, N. Carbon bead-supported hollow carbon nanofibers synthesized via templating method for the removal of hexavalent chromium. J. Ind. Eng. Chem. 2016, 36, 346–354. [Google Scholar] [CrossRef]
- Burwell, R.L. Manual of Symbols and Terminology for Physico-Chemical Quantities and Units—Appendix II, 1st ed.; Pergamon Press: Oxford, UK, 1981; pp. 285–286. [Google Scholar]
- Chen, J.; Li, Y.; Ma, Y.; Qin, Y.; Chang, L. Formation of bamboo-shaped carbon filaments and dependence of their morphology on catalyst composition and reaction conditions. Carbon 2001, 39, 1467–1475. [Google Scholar] [CrossRef]
- Haas, K.H. Hybrid Inorganic–Organic Polymers Based on Organically Modified Si-Alkoxides. Adv. Eng. Mater. 2010, 2, 571–582. [Google Scholar] [CrossRef]
- Zhu, M.; Diao, G. Synthesis of Porous Fe3O4 Nanospheres and Its Application for the Catalytic Degradation of Xylenol Orange. J. Phys. Chem. C 2011, 115, 313–318. [Google Scholar] [CrossRef]
- Hu, J.; Li, H.; Huang, X.; Chen, L. Improve the electrochemical performances of Cr2O3, anode for lithium ion batteries. Solid State Ion. 2006, 177, 2791–2799. [Google Scholar] [CrossRef]
- Wang, C.; Fu, X.Q.; Xue, X.Y.; Wang, Y.G.; Wang, T.H. Surface accumulation conduction controlled sensing characteristic of p-type CuO nanorods induced by oxygen adsorption. Nanotechnology 2007, 18, 145506. [Google Scholar] [CrossRef]
- El-Sheikh, S.M.; Rabah, M.A. Selective recovery of chromium ions from waste tannery solution for preparation of chromium oxide nanoparticles. Int. J. Environ. Sci. Technol. 2015, 12, 1–10. [Google Scholar] [CrossRef]
- Li, L.; Yan, Z.F.; Lu, G.Q.; Zhu, Z.H. Synthesis and structure characterization of chromium oxide prepared by solid thermal decomposition reaction. J. Phys. Chem. B 2006, 110, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Sun, Z.; Chen, M.; Cao, H.; Li, K.; Cai, Y.; Wang, F. Highly porous composite based on tungsten carbide and N-doped carbon aerogels for electrocatalyzing oxygen reduction reaction in acidic and alkaline media. Electrochim. Acta 2017, 236, 154–160. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, L.; Yang, S.; Li, D.; Xie, Z.; Wang, B.; Xia, Y.; Quan, F. Facile strategy to produce N-doped carbon aerogels derived from seaweed for lithium-ion battery anode. J. Alloy. Compd. 2017, 701, 256–261. [Google Scholar] [CrossRef]
- Fletcher, A.J.; Uygur, Y.; Thomas, K.M. Role of Surface Functional Groups in the Adsorption Kinetics of Water Vapor on Microporous Activated Carbons. J. Phys. Chem. C 2007, 111, 8349–8359. [Google Scholar] [CrossRef]
- Nightingale, E.R., Jr. Phenomenological Theory of Ion Solvation. Effective Radii of Hydrated Ions. J. Phys. Chem. 1959, 63, 1381–1387. [Google Scholar] [CrossRef]
- Tansel, B.; Sager, J.; Rector, T.; Garland, J.; Strayer, R.F.; Levine, L.; Roberts, M.; Hummerick, M.; Bauer, J. Significance of hydrated radius and hydration shells on ionic permeability during nanofiltration in dead end and cross flow modes. Sep. Purif. Technol. 2006, 51, 40–47. [Google Scholar] [CrossRef]
- Li, J.; Wang, N.; Deng, J.; Qian, W.; Chu, W. Flexible Metal-Templated Fabrication of Mesoporous Onion-Like Carbon and Fe2O3@N-Doped Carbon Foam for Electrochemical Energy Storage. J. Mater. Chem. A 2018, 6, 13012–13020. [Google Scholar] [CrossRef]
- Lee, Y.J.; Park, H.W.; Hong, U.G.; Song, I.K. Mn-doped activated carbon aerogel as electrode material for pseudo-capacitive supercapacitor: Effect of activation agent. Curr. Appl. Phys. 2012, 12, 1074–1080. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, J.; Tian, Z.; Ye, Y.; Cai, Y.; Liang, C.; Terabe, K. A general strategy toward transition metal carbide/carbon core/shell nanospheres and their application for supercapacitor electrode. Carbon 2016, 100, 590–599. [Google Scholar] [CrossRef]
- Ke, Q.; Liao, Y.; Yao, S.; Song, L.; Xiong, X. A three-dimensional TiO2/graphene porous composite with nano-carbon deposition for supercapacitor. J. Mater. Sci. 2016, 51, 2008–2016. [Google Scholar] [CrossRef]
- Dong, B.; He, B.L.; Xu, C.L.; Li, H.L. Preparation and electrochemical characterization of polyaniline/multi-walled carbon nanotubes composites for supercapacitor. Mater. Sci. Eng. B 2007, 143, 7–13. [Google Scholar] [CrossRef]
- Wu, D. Kinetic performance of Li4Ti5O12, anode material synthesized by the solid-state method. Ionics 2012, 18, 559–564. [Google Scholar] [CrossRef]













| Entry | Adsorption Capacity (mg·g−1) |
|---|---|
| CA-Fe | 133 |
| CA-Cr | 139 |
| CA-Cu | 424 |
| Adsorbents | Metal Ions | Adsorption Capacities (mg·g−1) | Reference |
|---|---|---|---|
| Active carbon | Fe | 166.7 | [28] |
| Carbon paper@Magnesium silicate | Cu | 113.5 | [29] |
| carbon nanotubes | Cu | 123.7 | [16] |
| Magnetic b-cyclodextrin/graphene oxide | Cr | 120.19 | [30] |
| Carbon bead-supported hollow carbon nanofibers | Cr | 51 | [31] |
| Carbon aerogel | Cu/Cr/Fe | 424/139/133 | This work |
| Entry | SBET (m2·g−1) | Smicro (m2·g−1) | Daverage (nm) | Vtotal (cm3·g−1) |
|---|---|---|---|---|
| CA-Fe | 666 | 413 | 8.00 | 1.33 |
| CA-Cr | 627 | 374 | 7.50 | 1.18 |
| CA-Cu | 450 | 274 | 7.16 | 0.81 |
| Blank | 695 | 463 | 8.11 | 1.52 |
| Entry | Specific Capacitance (F·g−1) | ||
|---|---|---|---|
| 0.5 A·g−1 | 1.0 A·g−1 | 1.5 A·g−1 | |
| CA-Fe | 239 | 215 | 196 |
| CA-Cr | 256 | 238 | 213 |
| CA-Cu | 275 | 255 | 234 |
| CA-Cu-0 | 250 | 224 | 186 |
| Blank | 129 | 125 | 122 |
| Carbon Species | Metal Species | Capacitance (F·g−1) | Current Density (A·g−1) | Reference |
|---|---|---|---|---|
| Carbon fiber | V | 104.05 | 0.5 | [15] |
| Onion-like carbon | Fe | 251.2 | 0.5 | [45] |
| Active carbon aerogels | Mn | 152 | 1.0 | [46] |
| Carbon | Ta | 223 | 1.0 | [47] |
| Graphene | Zr-MOFs | 302 | 0.15 | [48] |
| Carbon aerogel | Cu | 255 | 1.0 | This work |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, X.; Xu, Y.; Wang, S.; Zhao, J.; Ren, B.; Zhang, L.; Liu, Z. A Comparison of Electrochemical Performance of Carbon Aerogels with Adsorption Metal Ions for Super Capacitors. Materials 2018, 11, 2271. https://doi.org/10.3390/ma11112271
Dong X, Xu Y, Wang S, Zhao J, Ren B, Zhang L, Liu Z. A Comparison of Electrochemical Performance of Carbon Aerogels with Adsorption Metal Ions for Super Capacitors. Materials. 2018; 11(11):2271. https://doi.org/10.3390/ma11112271
Chicago/Turabian StyleDong, Xiaoxi, Yuelong Xu, Shasha Wang, Junping Zhao, Bin Ren, Lihui Zhang, and Zhenfa Liu. 2018. "A Comparison of Electrochemical Performance of Carbon Aerogels with Adsorption Metal Ions for Super Capacitors" Materials 11, no. 11: 2271. https://doi.org/10.3390/ma11112271





