1. Introduction
Corrosion is an electrochemical phenomenon which leads to severe component, system and structural deteriorations. Various operational conditions lead to corresponding corrosion failure mechanisms such as pitting, crevice, uniform, galvanic and microbial corrosion. Combined with static or dynamic stresses corrosion often leads to corrosion fatigue, fretting and stress corrosion cracking. These damages cause replacement, unscheduled maintenance, reduced capacity, idle time and insurance issues. Various techniques are used to control corrosion, including design modification, inhibitors, cathodic protection, thereby, increasing the cost of maintaining operations. Corrosion also has several economic, health, safety and technological implications [
1]. Many industries face huge financial losses due to corrosion-led failures. Researchers have been developing dielectric materials with enhanced corrosion resistance capabilities to both avoid and mitigate corrosion failures within structures and interacting systems. Some researchers have found that hybrid organic and/or inorganic nanocomposite materials are becoming desirable candidate to perform well in corrosive applications.
Few Layers Graphene (FLG) with 5–8 layers of graphene exhibit the promising properties of thermal, chemical and mechanical stability and are also a good charge carrier; this makes them fascinating in various fields of application [
2,
3,
4]. Graphene provides resistance against oxidation while its hydrophobicity resists hydrogen bonding with water. Due to its multiple promising properties, graphene is being extensively explored for various applications including interacting systems and as nano-additives in lubrication. Recently, several studies have reported that graphene coating can effectively isolate the surface from environmental degradation [
5,
6,
7]. Aneja et al. [
4] studied functionalized graphene coating to evaluate its barrier and protection properties applied to mild steel.
Water uptake of the coating, its capacitance and pore resistance was obtained by using EIS. Experiments were conducted at various frequencies for a certain time period. Graphene decreased the water uptake and the activation energy peak was increased for water driven diffusivity process constraining the ion movement through the coating. Moreover, the network of functionalized graphene was found to be structurally dense [
4]. Similarly, Olad et al. [
8] investigated a triple hybrid system of conducting polyaniline, zinc and epoxy to evaluate their corrosion resistant properties when applied to metallic substrates. During this research, 75-micron thick nanocomposite coatings on iron substrate and in a solution of 0.1 M HCl solution were studied. In addition to anti corrosive properties, epoxy and zinc enhanced the mechanical strength of these nanocomposite coatings. Epoxy enhanced the mechanical properties by providing physical hindrance to electrolyte molecules while zinc improved the electrochemical behavior of polyaniline coating.
SAN is copolymer of styrene and acrylonitrile. It helps against corrosion degradation facilitated by its hydrophobicity and provides physical barrier, based on higher diffusion resistance to the corrosive ion [
9]. Conducting polymer prevents corrosion with the help of generated electric field that blocks the electrons flow from the metal to external oxidizing agents [
10]. Polyaniline is one of the promising polymers for corrosion protection. It is a p-type semiconductor with desirable electrical and chemical properties. Its conductive type contributes significantly to the effective resistance to corrosion.
New trends in the processing of Polyaniline have increased its range of practical applications. It protects against electrochemical decay by forming a thin oxide layer due to the interaction of polymer and metal substrate. It reduces the redox current as well as shifts the corrosion potential towards noble direction as compared to bare metal [
11]. ZnO is a wide bandgap inorganic conductor. It belongs to II-VI semiconductor family. It is found in the form of a white colored powder. Its hydrophobicity and ability to shift corrosion potential to noble direction makes it a good candidate for corrosion protection [
12,
13,
14].
In this research novel hybrid nanocomposite coatings of SAN/PANI/FLG and ZnO/GO were synthesized and characterized through various characterizing tools. An electrochemical framework of Gamry
® Instruments was used to evaluate the performances of coating systems against corrosion to protect ductile cast iron pipeline in seawater and crude oil produced water. Electrochemical Impedance Spectroscopy (EIS) technique employs a short perturbation AC signals during electrochemical investigation [
15,
16,
17,
18,
19,
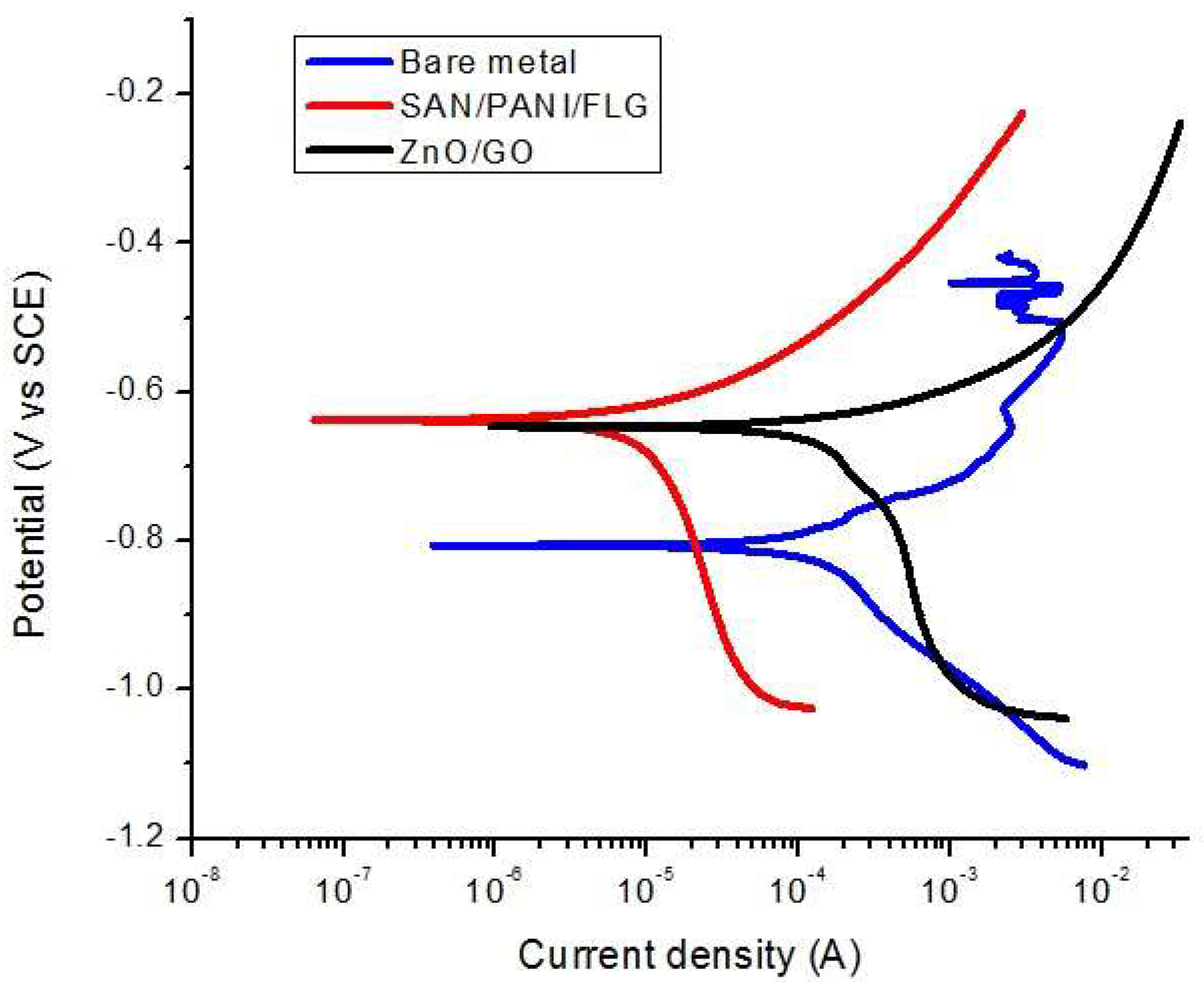
20]. Amplitude of 10 mV AC was applied to investigate the response of system under various frequencies. Corrosion resistance and protection are analyzed by plots Bode and Nyquist generated by EIS. Phase shift and impedance values are plotted against frequency in Bode plot while imaginary frequency is scanned versus real impedance in Nyquist plot. SAN/PANI/FLG performed better in protecting metal samples in aggressive environments against corrosion compared to ceramic ZnO/GO nanocomposite coating.
2. Materials and Methods
2.1. Sample Preparation
Ductile cast iron industrial pipeline samples were used as substrates. Samples were prepared in ~4 × 4 cm
2 squared sections. Samples were ground with abrasive paper of 120–320 grades in order to develop a texture for adhesion of nanocomposites coating.
Table 1 shows composition of the pipeline cast iron used for study.
2.2. Synthesis of Coating
2.2.1. SAN/PANI/FLG
Table 2 demonstrates materials employed for SAN/PANI/FLG coating.
Preparation of Polyaniline
Chemical oxidation polymerization technique was used to prepare conductive Polyaniline (PANI). 6 g of aniline monomer was mixed in 100 mL deionized water using a vessel fixed with a stirrer and equipped with ice bath for low temperature arrangement. Conductivity of the solution was improved by adding 1 M solution of HCl to obtain aniline hydrochloride; a continuous stirring was ensured during addition. As a surfactant, 1 M solution of Sodium Dodecyl Sulfate (SDS) was added. To start polymerization, 1.2 g APS (Ammonium per sulfate) was dissolved as initiator in a 20 g of deionized water. Gradual addition of the above solution was facilitated using a stirring mechanism while the low temperature (0 °C) was maintained with the help of an ice bath; the mixing continued for one hour. The stirring continued for one a half hour at room temperature. Methanol was used to facilitate precipitation followed by washing using deionized water for multiple times. Final drying was performed in an oven at 50 °C. A green colored PANI powder was the final product. Initially, methanol precipitation was performed. The polymer was then washed by using distilled water, this process of conditioning with distilled was repeated several times. After washing the precipitate, it was dried in an oven at 50 °C. A green colored PANI powder was produced as the final product from the above process.
Preparation of SAN/PANI/FLG Nano Composite
Thin film of nanocomposite coating was prepared through solution casting method. SAN polymer was added to 1,2-dichloroethane and then continuously stirred for 40 min in order to completely dissolve the constituents of the solution. Then PANI was slowly added; and the solution color turned dark green. Solution was probe sonicated and stirred overnight for proper dispersion of PANI. Once the SAN and PANI was completely dissolved, then an appropriate quantity of FLG was added. The solution was then subjected to probe sonication for 45 min to disperse the PANI and FLG. The acquired solution was kept on overnight stirring after which the nanocomposite was ready to coat on the substrate. Mechanical properties of the SAN/PANI/FLG thin film are shown in
Table 3.
2.2.2. ZnO/GO Coating
Ceramic coatings were developed on the metallic substrate in two steps: synthesis of (a) ZnO nanoparticles sol gel; (b) ZnO-GO composite sol.
(a) Synthesis of ZnO Nanoparticle Sol Gel
Zinc Acetate Dehydrate (ZnAcDH) was used as a precursor in the synthesis. Isopropanol alcohol was used as the solvent. Ethanol amine was used as stabilizer in a specific amount for the preparation of 0.4 M ZnO sol. It was then stirred until opaque solution turned into transparent one i.e., ZnAcDH was completely dissolved. The resultant solution was left for 24 h for aging. The ingredients are given in the
Table 4.
(b) Synthesis of ZnO/GO Composite Sol
A solution containing 0.5 mg/1 mL of graphene oxide in 2-methoxyethanol was formulated and prepared. For proper dispersion of graphene oxide, sonication was performed for 45 min in ultrasonic water bath. Both graphene oxide and ZnO solutions were mixed together. The obtained ZnO nanoparticles had 13 nm to 22 nm size range.
2.3. Dip Coating
Dip coating is a time efficient and most commercially adapted technique for coating purposes. Dip coating process involves five steps: (i) Immersion; (ii) Dwell time; (iii) Deposition; (iv) Drainage; and (v) Evaporation. Samples were dipped in the solution for 3 min. Dwell time was kept 0.5 min to achieve desired thickness. When the solution was properly deposited on the metal samples, the coated samples were then placed in an oven to drive off the solvent from the solution resulting in a meaningful deposition of thin films. Coating thicknesses were kept in a range from 5 to 7 μm, by controlling the process parameter e.g., dwell time etc. using the Landau–Levich equation. Coating thickness was measured by surface profilometry and further manually confirmed by micrometer screw gauge and Landau–Levich equation.
2.4. Electrochemical Testing
Gamry® framework (Series G-750, Gamry Instruments, Inc., Warminster, PA, USA) equipped with DC corrosion testing as (Gamry Instruments, Inc.) EIS, was employed for all electrochemical measurements. EIS and Tafel scans were conducted by using three-electrode cell. The reference and counter electrodes in the cell were a Saturated Calomel Electrode (SCE) and a graphite rod respectfully. Metal samples of 4 cm × 4 cm dimensions were used. The surfaces were masked using epoxy leaving an exposed area of 3 cm × 3 cm. Two types of corrosive environments were used as electrolytes for investigation: (i) simulated seawater (pH slightly basic (7.9) and conductivity between ≥50 mS/cm); and (ii) a sample of produced water obtained from a local petroleum sector (pH 7.6) and bulk conductivity ~150 µS/cm).
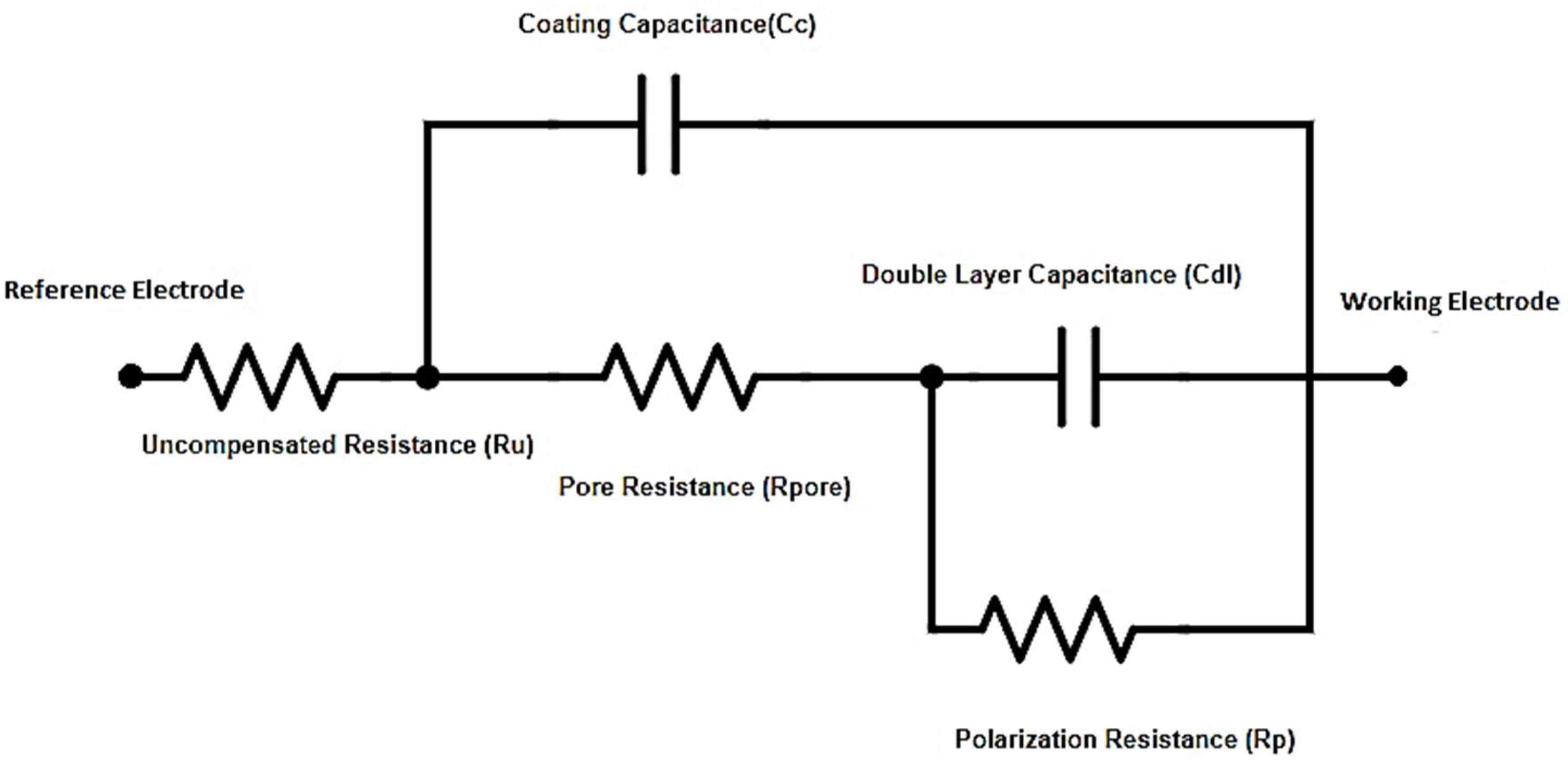
2.5. Equivalent Circuit Modeling
Equivalent circuit model of the cell system with input parameters is shown in
Figure 1. It was used for fitting the curves to obtain pore resistance and coating capacitance values of two nanocomposites systems using Reap2cpe model. The determined values are shown in the
Table 5.
2.6. Surface Morphology
The surface morphology of the samples was studied before and after the corrosion test to observe the damage and deterioration caused by the corrosive environments. This morphological study was conducted by using Scanning Electron Microscope (JOEL JSM-6490A, JEOL USA, INC., Peabody, MA, USA).
4. Conclusions
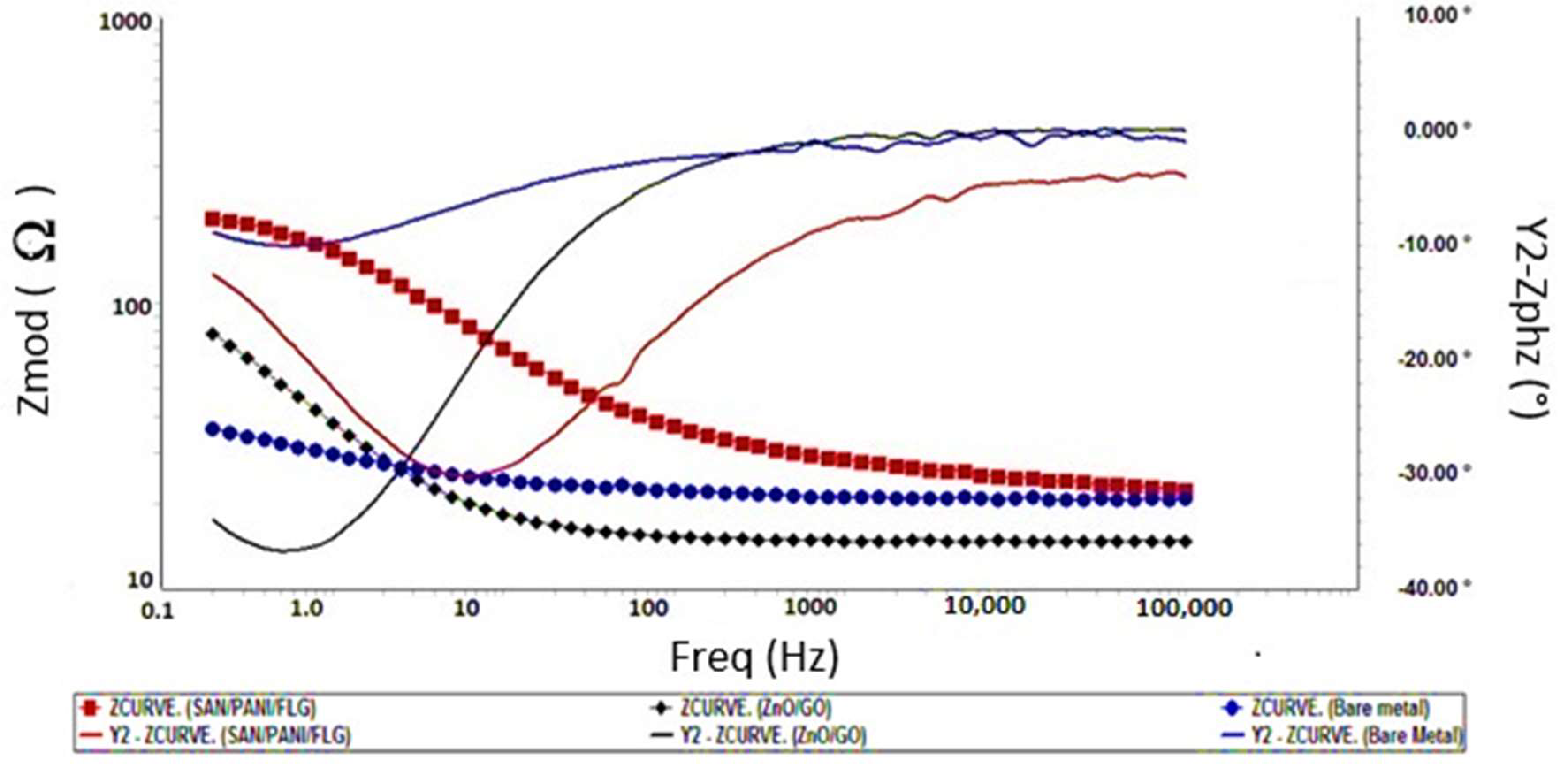
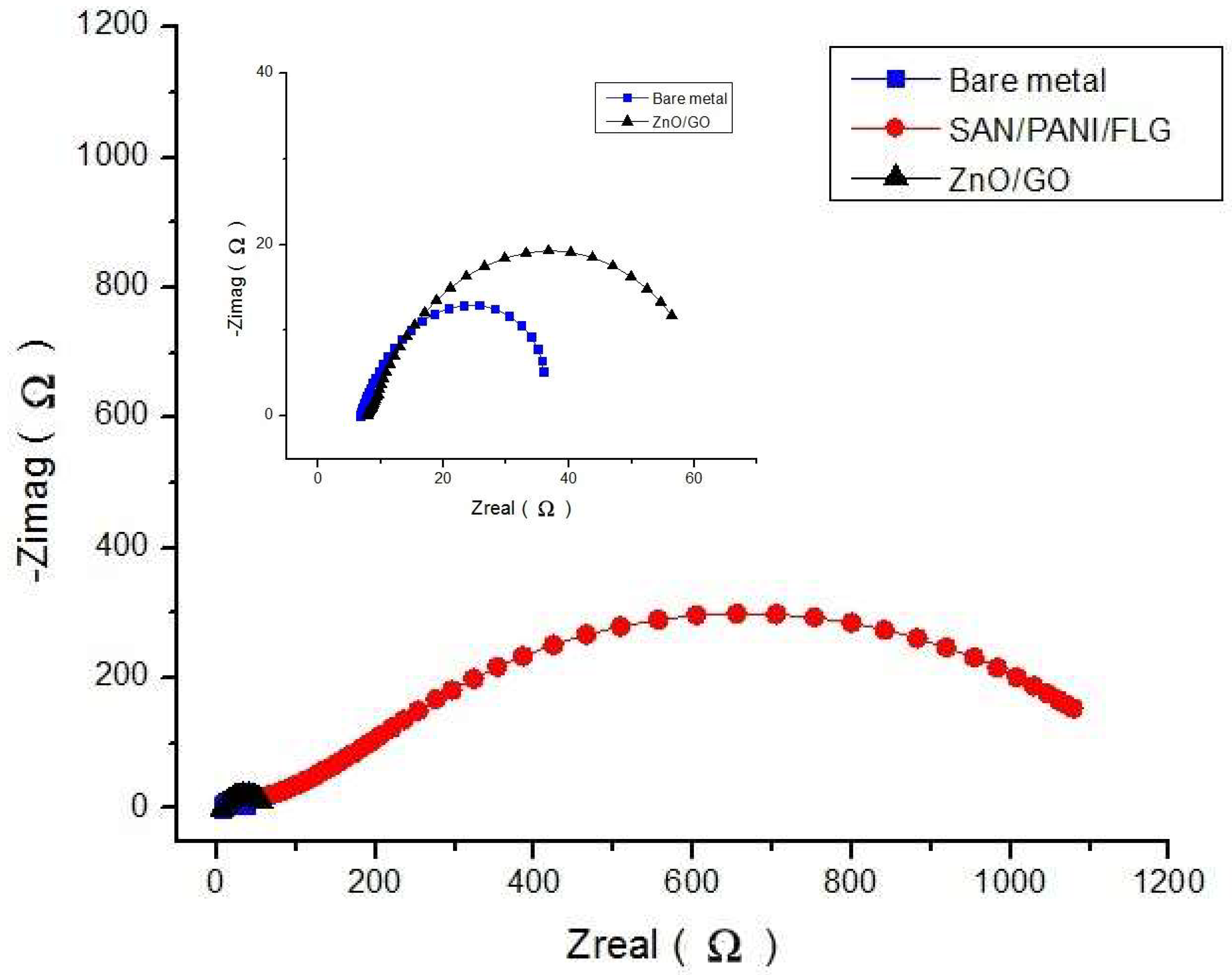
It was concluded that the corrosion protection of polymeric based SAN/PANI/FLG coating was found reasonably higher than ceramic based ZnO/GO coating in seawater as well as in produced water. SAN/PANI/FLG coating reduced the corrosion of bare metal up to 90% in seawater environment with the highest reported impedance value of 250 Ω, whereas, ZnO/GO suppressed the corrosion up to 75% having the impedance value of 100 Ω.
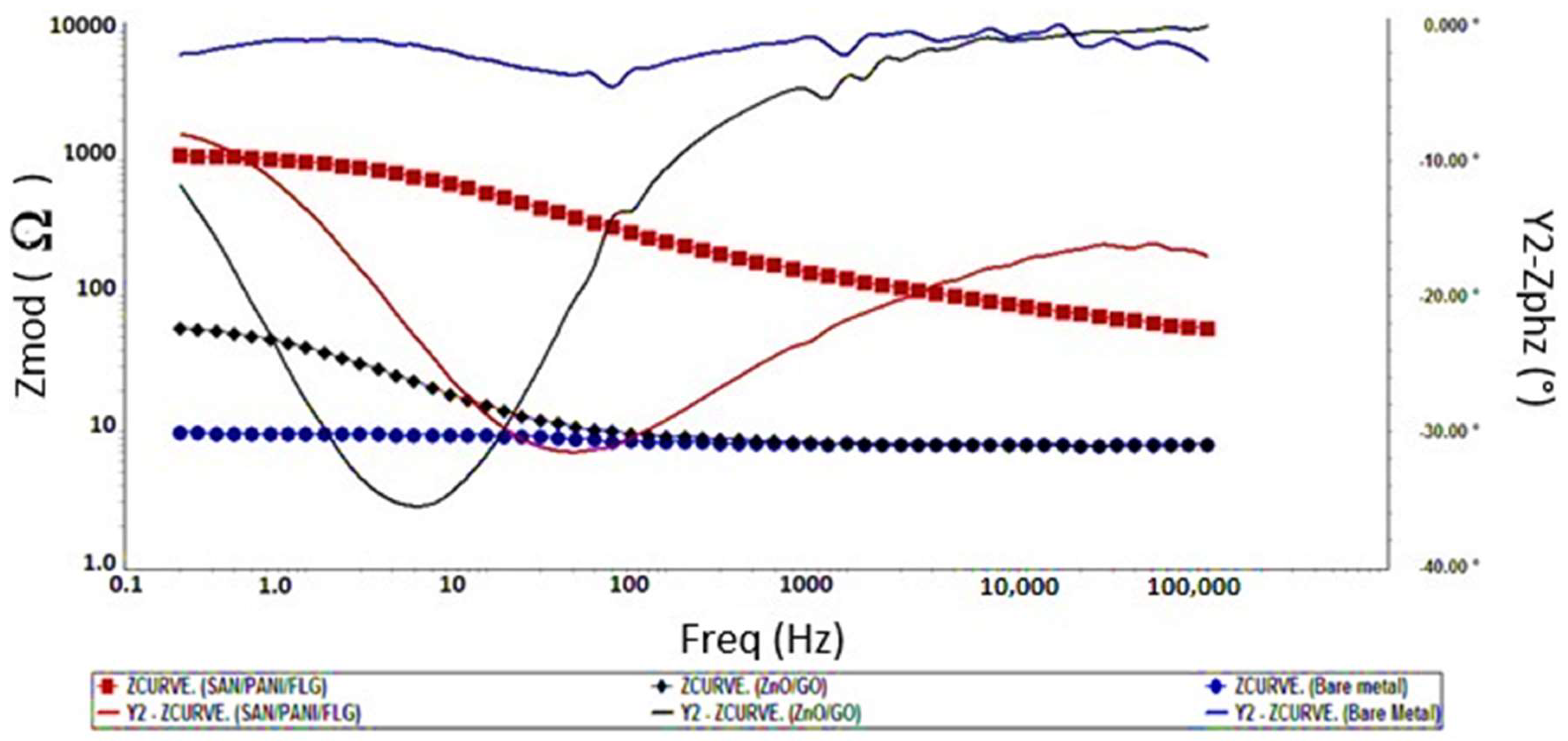
In crude oil produced water, SAN/PANI/FLG coating proved to be significantly better corrosion resistant compared to ZnO/GO coating. SAN/PANI/FLG reduced the corrosion up to 95% with impedance value of 1000 Ω while ZnO/GO suppressed the corrosion up to 10% with the impedance value of 90 Ω.
Both polymeric and ceramic based nanocomposites coatings demonstrated better protection against sea and produced water. However, the variation in protection levels of both polymeric and ceramic based nanocomposites coatings between the two environments can be seen. SAN/PANI/FLG coating performed better in terms of reducing corrosion of bare metal as compared to ZnO/GO coating. This was due to better resistance against pore formation and strong barrier properties against the electrolyte of SAN/PANI/FLG coating.