1. Introduction
Bioplastics have gained significant attention from the researches, manufacturers, customers, and investors worldwide over the past 25 years as the bio-alternatives for various fossil-resource based products. The pollution of the environment that is caused by plastics, the disappearance of landfill space, diminishes of petroleum resources, and the interest in controlling the toxic gases emissions are ranked among the reasons for this attention. The term “biopolymer” refers to polymers that are bio-based, biodegradable, or both; so, among the benefits of bioplastics from biopolymers that are based on renewable resources (or even on waste materials) is reduced carbon dioxide production during the bioplastics synthesis and their biodegradability [
1,
2,
3,
4].
Biobased material has some or all of its carbon produced from renewable resources like corn or sugar cane, or is produced in yeast, bacteria, or algae. The conditions for the determination of biobased content of polymeric materials are described in the European standard CEN/TS 16295:2012 [
4].
Under appropriate conditions of moisture, temperature, and oxygen availability, the biodegradation leads to the fragmentation or disintegration of plastics with no toxic or environmentally harmful residue [
5]. According to CEN/TR 15932:2010, biodegradable polymers are able to completely degrade to low molecular products (carbon dioxide, methane, water) and the biomass (mineralization and bioassimilation) through the chain scission in the backbone [
6]. Specific test conditions, environments, and time-scales for laboratory tests are defined in European standards (EN ISO 14851:2004, EN ISO 14852:2004, EN ISO 17556:2004, EN ISO 14855-1:2007/AC:2009, EN ISO 14855-2:2009) [
7,
8,
9,
10,
11]. Complete biodegradation is a combination of physical and enzymatic processes (where the chain scission is caused by human, animal, or fungi cell activity). The biodegradation progress is monitored by the measurement of oxygen uptake and carbon dioxide production. A special case of biodegradability is the compostability, where the degradation of a sample takes place in a soil being strongly influenced by composting conditions, such as the substrate properties, the microbial activity, the water content, and pH (EN ISO 13432:200/AC:2005, EN ISO 14995:2006) [
4,
12,
13].
Although high costs of biodegradable polymers and the lack of regulation and composting facilities still prevent the massive distribution of bioplastics, the hypothetical global bioplastics production capacity is expected to increase from around 2.05 million tons in 2017 to approximately 2.44 million tons in 2022 [
1,
14]. Generally, bioplastics are used in a growing number of markets, from packaging, catering products, consumer electronics, automotive, agriculture/horticulture, toys and textiles to medical applications, and a number of other segments. Well-investigated biopolymers, such as PLA and PHAs, are the main drivers of production capacity growth [
14].
PHA includes a group of microbial polyester polymers and copolymers (150 different PHA monomers have been reported yet). These bio-based biodegradable linear polyesters feature a wide array of physical and mechanical properties depending on their chemical composition. The occurrence of short-chain-length PHA (scl-PHA) with 3–5 carbon atoms per unit is the most common [
15,
16]. The PHAs production is estimated to triplicate the capacities in next five years [
14]. PHAs are stored as intracellular carbon and energy reserve granules in diverse Gram-negative and Gram-positive bacteria from a range of substrates, including sugars and fatty acids [
15].
Poly(lactic acid) (PLA), –[CH(CH
3)COO]
n– is very versatile aliphatic polyester that features excellent barrier properties and is available in the form of high-performance PLA grades that are a good replacement for PS (polystyrene), PP (polypropylene), and ABS (acrylonitrile butadiene styrene) in more demanding applications [
14,
17]. PLA is synthesized from renewable resources through lactic acid fermentation and it is highly transparent, colorless, glossy, and rigid thermoplastic material [
18,
19]. The properties of PLA are dependent on the ratio between D and L enantiomers. The disadvantages of this polymer are its low heat distortion temperature (softening above 60 °C) and also its inability to quickly biodegrade at ambient temperature [
18]. The tensile properties of PLA can vary widely depending on whether it is annealed or oriented, or on its degree of crystallinity [
19,
20].
The combination of these non-toxic, biodegradable, and biocompatible materials (PHAs, PLA) with three-dimensional (3D) printing technologies is useful, especially for the biomedical field (scaffolds, implants), art objects, precise replicas of archeological objects, and for prototyping and the manufacture of spare parts for automotive, airplanes, etc. [
17]. PLA, as biodegradable polymer, is already used in 3D printing industry. Biodegradable PLA/PHA polymeric blends are also applicable for 3D printing, even in industry at the time [
21]. Their printability is good.
Fused Deposition Modeling (FDM) is the most productive technology used in low-cost 3D printers. A plastic filament used in this method is pushed through heated extrusion nozzle, melted and deposited on the bed in a tailored shape. The software that processes a STL (stereolithography) or CAD (computer-aided design) file, mathematically slicing and orienting the model for the building process, is embodied in this method [
22]. The FDM method is applied in this work for the preparation of 3D printed dogbones (double-paddle shaped testing elements), which are further used for tensile tests. Usually, it is necessary to deal with the warping problem, which occurs especially during the printing of large parts, where the internal temperature of printed object tends to vary. Our materials are printed at high temperature (190 °C, the temperature of the extrusion nozzle) onto the printing bed (which has ambient temperature) and cooled with an integrated fan for quick solidification. This usually causes the shrinkage and it can lead to the deformation through the warping, which can lead to the detachment from printing bed and to the product destruction [
23]. The warping wasn´t relevant in the case of our 3D printed dogbones.
Although PLA and Poly(3-hydroxybutyrate (PHB) can be potentially used as biodegradable and biocompatible alternatives to conventional polymers by many industrial sectors, both PLA and PHB have a few disadvantages. PHB is tough, fragile, and brittle in nature and it has poor processing properties. Blending with amorphous polymer, such as poly(
d,
l-lactic acid), is proposed to reduce the crystallinity of PHB, so that the applications of PHB can be expanded [
3,
24]. In spite of blending, these neat PHB/PLA blends are still stiff and brittle materials with poor mechanical properties and thermal degradation close to its melting point, limiting its processability [
25]. Poor ductile properties can be well improved by the addition of plasticizer.
Plasticizers are an important class of low molecular weight non-volatile compounds that were homogeneously incorporated in a material (usually plastics or elastomer) that are widely used in polymer industries as additives. The primary role of such substances is lowering the glass transition temperature (
Tg), the melting temperature (
Tm), and improving the flexibility, the distensibility, and the processability of biopolymers [
26,
27]. Plasticizers reduce the tension of deformation, hardness, density, viscosity, and electrostatic charge of a polymer, and at the same time, they increase the polymer chain flexibility, resistance to fracture, and dielectric constant [
26,
28]. The additions of plasticizer thus influence the mechanical properties—reduce the toughness, the tensile strength, and increase the elongation at break. Other properties are also affected, such as the degree of crystallinity, optical clarity, electric conductivity, fire behavior, and the resistance to biological degradation, amongst other physical properties [
28]. Among the most significant plasticizers are various monomeric esters of phthalic, phosphoric, adipic, sebacic, citric, and ricinoleic acids, furthermore chlorinated paraffins and low-molecular weight polyesters [
26,
27].
Plasticizers can be divided into two principal groups: primary and secondary. Primary plasticizers lower the glass transition temperature
Tg and increase the elongation and softness of the polymer. Secondary plasticizers, when added to the polymer alone, do not bring about such changes and may also have limited compatibility with the polymer. However, when added to the polymer in the presence of a primary plasticizer, secondary plasticizers enhance the plasticizing performance of the primary plasticizer [
29]. In this current study, the primary plasticizers that externally plasticized the polymeric blends were used.
The principle disadvantage of external plasticizing is the migration of the plasticizer from the plasticized material to other contacting media. This process includes the diffusion of the plasticizer from bulk material towards the surface, the interface phenomena, and the sorption or evaporation into the surrounding medium [
28]. One of the consequences of the plasticizer decrement is the loss of the material elasticity and ductility. In addition, the plasticizer leakage from the material can be the cause of surrounded media contamination (medical application issues). Among the main factors that affect the plasticizer migration are the type and the concentration of the plasticizer, its molecular weight, branching, and polarity. Low-molecular weight plasticizers tend to leak away from the polymeric material easier. The more linear the plasticizer’s structure, the more extraction rate and migration occurred when compared to more branched plasticizers. The plasticizer migration is also affected by the type of polymer, its molecular weight, and its compatibility with the plasticizer; and, by the plasticization process and homogeneity of the product. The migration is also influenced by the conditions of the migration test (type of contact, time, temperature, polymer probe, specimen thickness). Higher temperatures simplify the plasticizer migration, while lower temperatures decelerate the migration [
27,
29,
30,
31,
32,
33,
34,
35].
In this current study, four commercial monomeric plasticizers that are based on esters of citric acid were used. The plasticizers miscibility with PHB/PLA blends in the weight ratio (wt %) of 85 (polymer)/15 (plasticizer) was confirmed using a kneading machine. The PHB/PLA/plasticizer blends in the weight ratio of 60/25/15 were then prepared by single screw and corotating meshing twin screw extruders in the form of filaments (for further 3D printing). The mechanical properties of plasticized blends were determined by tensile test of filaments as well as 3D printed dogbones. The diffusion of plasticizers from blends that were exposed at 110 °C in the drying oven was also investigated. The determination of the degree of disintegration of selected plasticized blend when exposed to a laboratory-scale composting environment was executed to roughly check their biodegradability. The Modulated Differential Scanning Calorimetry (MDSC) was performed to check the thermal properties of the plasticized blends and Scanning Electron Microscopy (SEM) was used to check their morphology. The goal was to create new types of eco-friendly PHB/PLA/plasticizers blends and to highly improve poor mechanical properties of neat PHB/PLA blends (with majority of PHB), by adding appropriate plasticizer.
These environmentally friendly PHB/PLA/plasticizer blends with the majority of PHB are biocompatible and suitable for 3D printing technology, and thus for biomedical applications and prototyping. Additionally, such type of eco-friendly blends, containing the PHB majority, can be well compostable. PHB can be prepared, even from waste cooking oil, so, unlike other types of biodegradable bioplastics, it does not require the agricultural feedstock, such as corn, potato, sugar cane, or sugar beet [
36,
37]. Quite many plasticizers were tested with PHB or PLA apart to improve their thermal and mechanical properties for various applications. Only some studies have focused on improving the thermal and mechanical properties of PHB/PLA/plasticizer blends, but always with the PLA majority.
Armentato et al. [
38] investigated binary and ternary films made of PLA, 15% PHB and OLA (oligomeric lactic acid), which were prepared under optimized extrusion conditions, followed by a filmature procedure. The mechanical, thermal, morphological, and functional properties were investigated as the function of OLA composition ratio (15, 20, and 30 wt % of OLA by weight).
Arrieta et al. [
25] blended PLA with PHB (75:25 wt % ratio) to increase the crystallinity. The plasticizers acetyl(tributyl citrate) (ATBC) and poly(ethylene glycol) (PEG) were added in the amount of 15 wt % when PLA/PHB blends achieved the melt state, to increase the processability and ductility. Each blend was then processed into biodegradable flexible film with improved properties for food packaging.
Abdelwahab et al. [
3] investigated a blend of PLA (75% by weight) and PHB (25%) with a polyester plasticizer (Lapol 108) at two different concentrations (5 and 7% by weight per 100 parts of the blends).
This paper describes the influence of four plasticizers on thermal, mechanical, and morphological properties of plasticized PHB/PLA blends, but with the PHB majority. This kind of work has not been likely published yet, and thus it is unique in this context.
2. Materials and Methods
2.1. Materials
Poly-3-hydroxybutyrate (P3HB Biomer
®,
ρ = 1.23 g∙cm
−3,
Mw = 410,000 g·mol
−1) was provided by Biomer Company (Krailling, Germany) [
39]. The polymer was in the form of white powder.
The PLA used in this study is poly(
d,
l-lactic acid), which is amorphous at
Tg around 60 °C. This amorphous PLA was chosen for its ability to reduce the overall crystallinity content in our PHB/PLA/plasticizers blends, which are further used for 3D printing. Amorphous polylactic acid (aPLA) granules (Ingeo™ 4060D,
Mw = 180 000 g·mol
−1,
Tg = 55–60 °C,
ρ = 1.24 g∙cm
−3) were supplied by NatureWorks LLC (Minnetonka, MIN, USA) Company [
40].
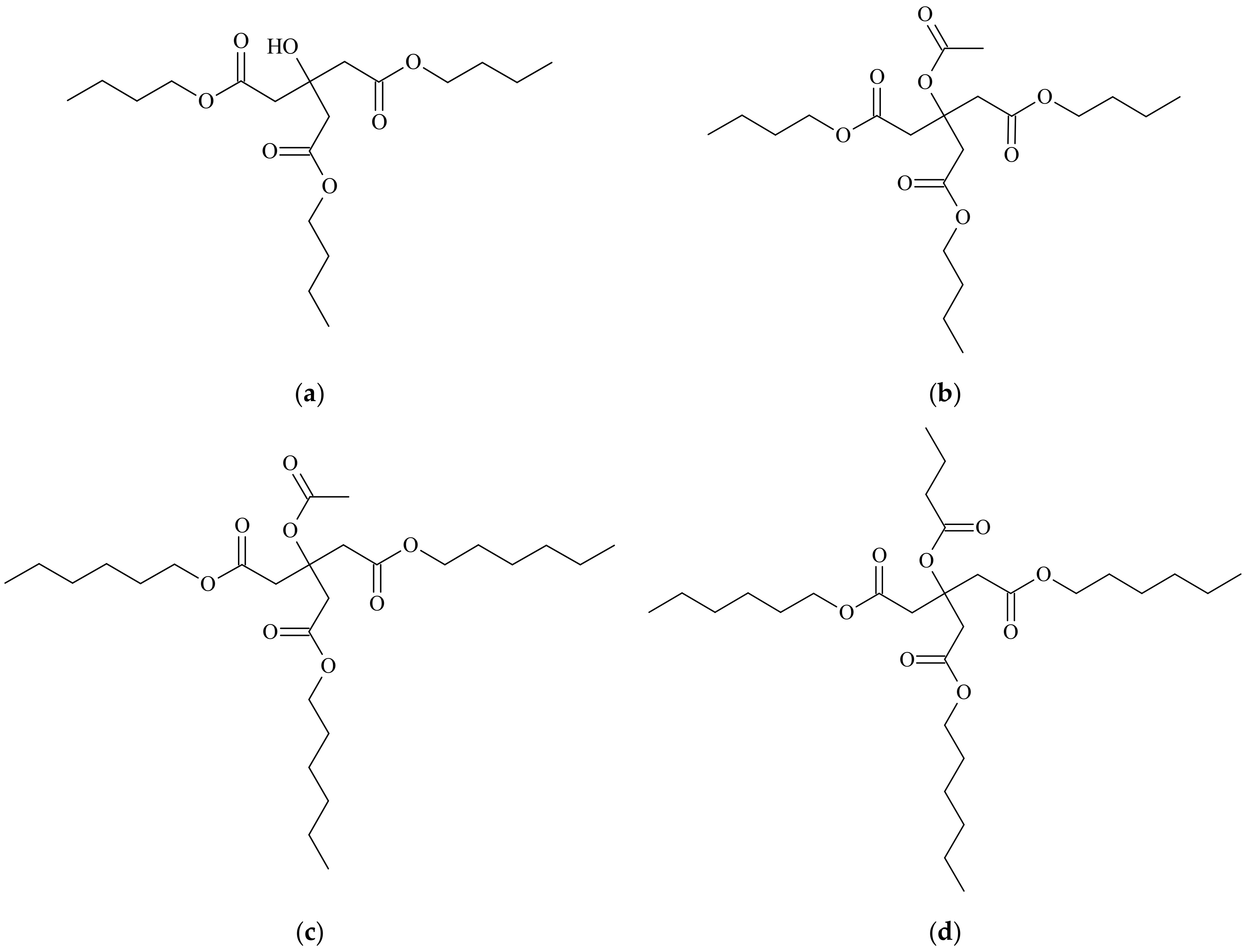
Four monomeric commercial plasticizers that are selected for this study are based on esters of citric acid, as provided by Vertellus Holdings LLC Company (Indianapolis, IN, USA), with trademark Citroflex
® [
41]. Their commercial and chemical names, molecular weights, and labels can be seen in
Table 1. Their structure can be seen in
Figure 1.
2.2. Plasticizers Miscibility with PLA/PHB Polymers
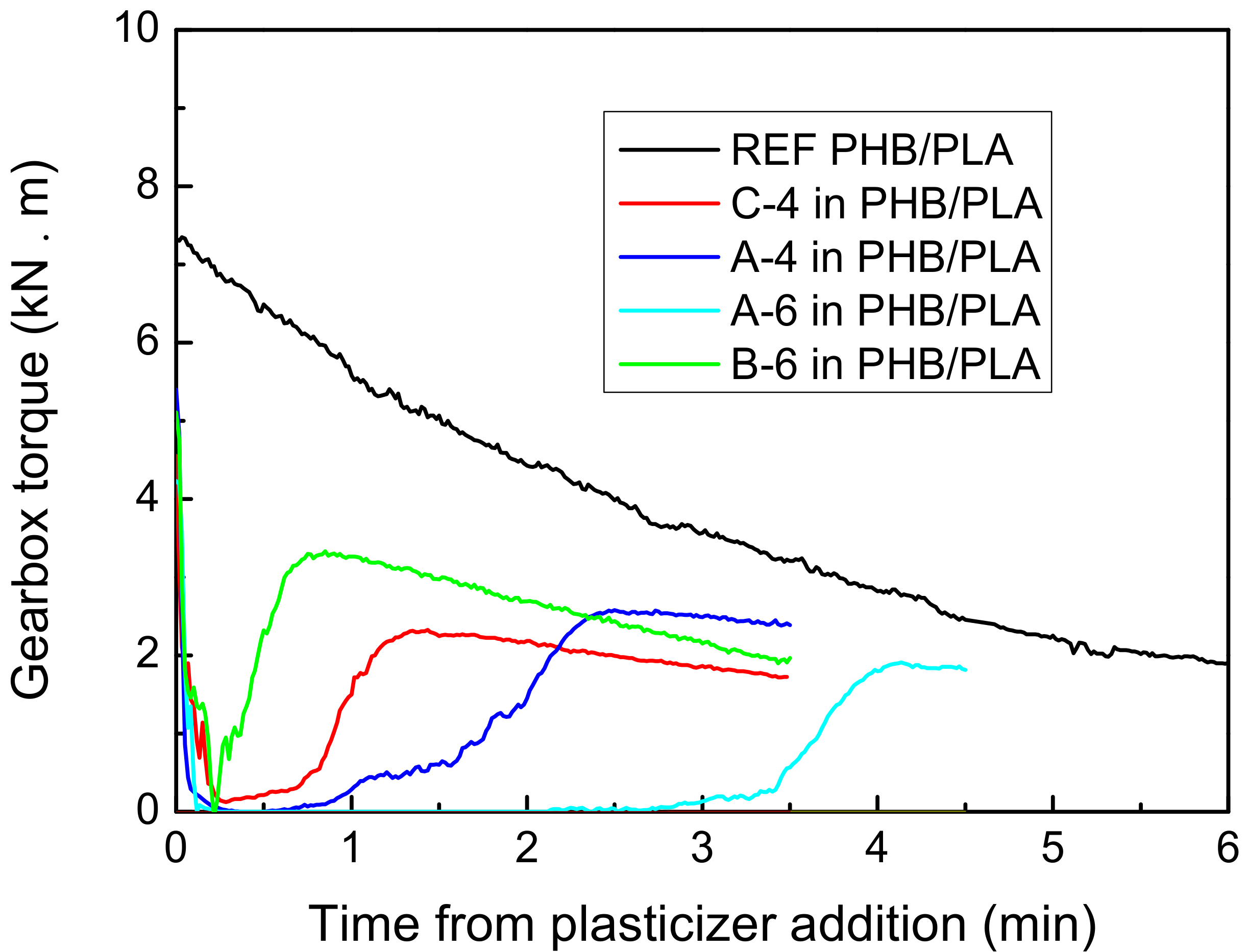
The miscibility of plasticizers with PHB/PLA polymer mixture was tested using the kneading machine PLASTI-CORDER Brabender. Citroflex® plasticizers were kneaded with PHB/PLA mixture in the weight ratio (wt %) of 60 PHB:25 PLA:15 plasticizer. Non-plasticized PHB/PLA was kneaded as a reference for the comparison (70 PHB:30 PLA). The total weight of each sample before the kneading process was 30 g. The process of kneading plasticizers with the PHB/PLA mixtures was performed at 178–180 °C (chamber temperature) at 45 rev./min, and the kneading process of reference was performed at 180 °C. The PHB/PLA mixture was dosed into the PLASTI-CORDER Brabender machine (Brabender GmbH & Co. KG, Duisburg, Germany) first and then kneaded for 1.5 min. Then, the plasticizer was added and the kneading process continued for following 3.5 min in the case of C-4, A-4 and B-6 plasticizers and for 4.5 min in the case of A-6 plasticizer. The total kneading time of PHB/PLA with plasticizers (sample dosing with kneading) was 6–7 min. The total kneading time of reference was 9 min. The gearbox torque dependence on the total plasticization time was measured and the miscibility was evaluated.
2.3. Preparation of Plasticized PHB/PLA Blends
PHB and PLA polymers were firstly pre-treated by drying at 80 °C in the drying oven (Memmert, Schwabach, Germany) for 2 h to eliminate possible absorbed water. As the first step, the corotating meshing twin screw extruder with the screw auger parameters D = 16 mm and L/D = 40, from Labtech Engineering Company (Samutprakarn, Thailand), was used for the preparation of plasticized PHB/PLA blends. The total weight of each sample for extrusion was 350 g. The chamber temperature profile from hopper to nozzle was set to 160–175–175–75–175–175–175–170–165–160 °C and the rotational speed of screw was set to 130 rev./min. The PHB/PLA blends in the weight ratio of 70/30 (wt %) (reference) as well as the PHB/PLA/plasticizer blends in the weight ratio of 60/25/15 (wt %) were extruded in the form of filaments. Four Citroflex® plasticizers were used one by one. Unfortunately, the filament, which was pulled from the nozzle, had diverse non-defined thickness and diameter and so it was unsuitable for 3D printing. Therefore, the filaments were pulled through the water tank (water with ambient temperature) and then the filaments were pulled into the granulator to get pellets, which are later used for the second extrusion.
The PHB/PLA (reference) pellets and the PHB/PLA/plasticizer pellets that were prepared by twin screw extruder, were at first pre-treated by drying at 80 °C for 2 h in the drying oven. Then single screw extruder HAAKETM Rheomex OS (Haake Technik GmbH, Vreden, Germany) was used for the second extrusion in order to achieve the filaments with a defined diameter of 1.75 mm. The chamber temperature profile from hopper to nozzle was set to 185–175–170–150 °C and the rotational speed of screw was set to 25 rev./min. The filament that extruded from the nozzle was pulled out into the draw-off device through the calibration mechanism located in the water tank. Water was tempered by the thermostat at 60 °C in order to finish the polymer crystallization. Prepared PHB/PLA/plasticizer filaments with defined diameter were then used for further characterizations and for 3D printing.
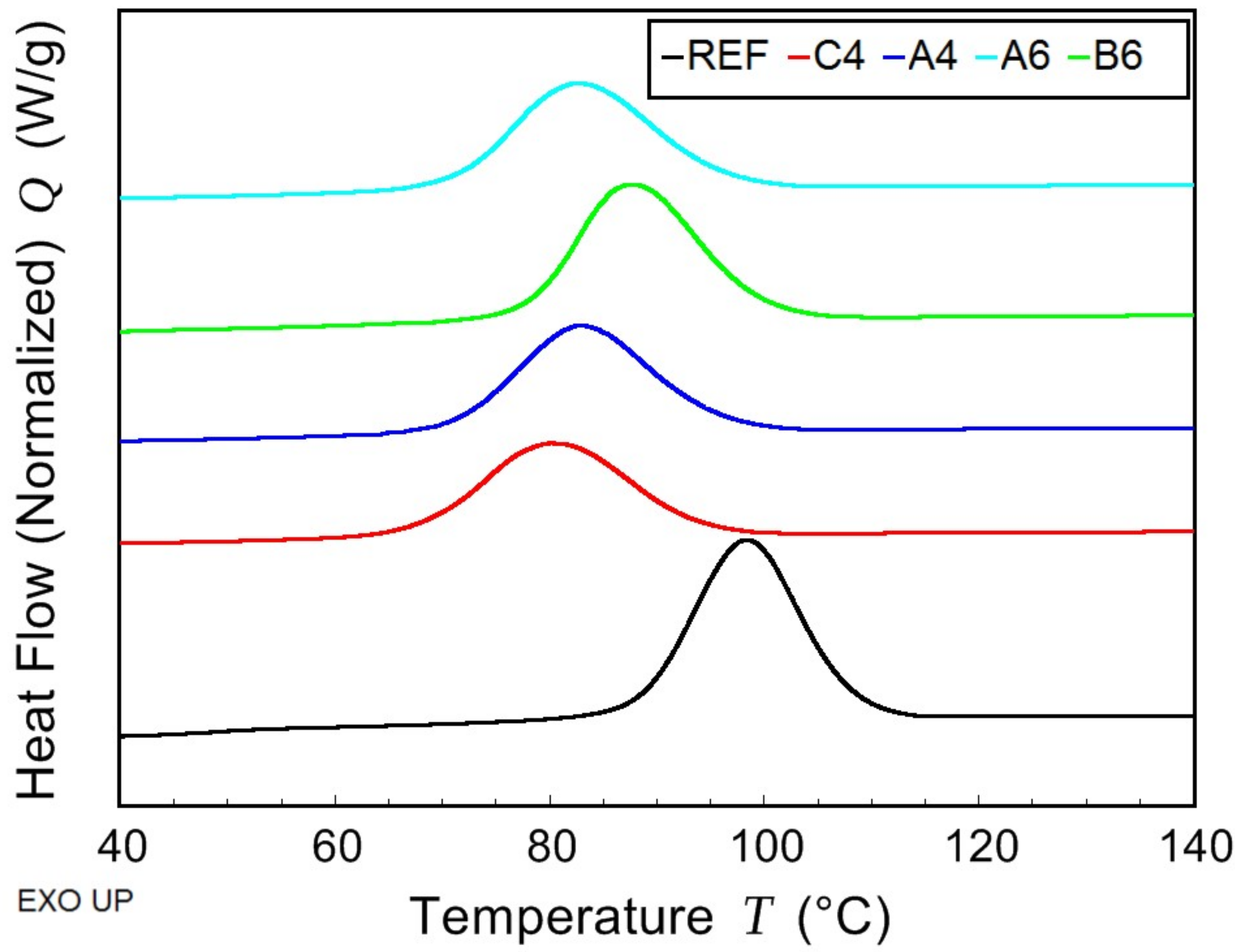
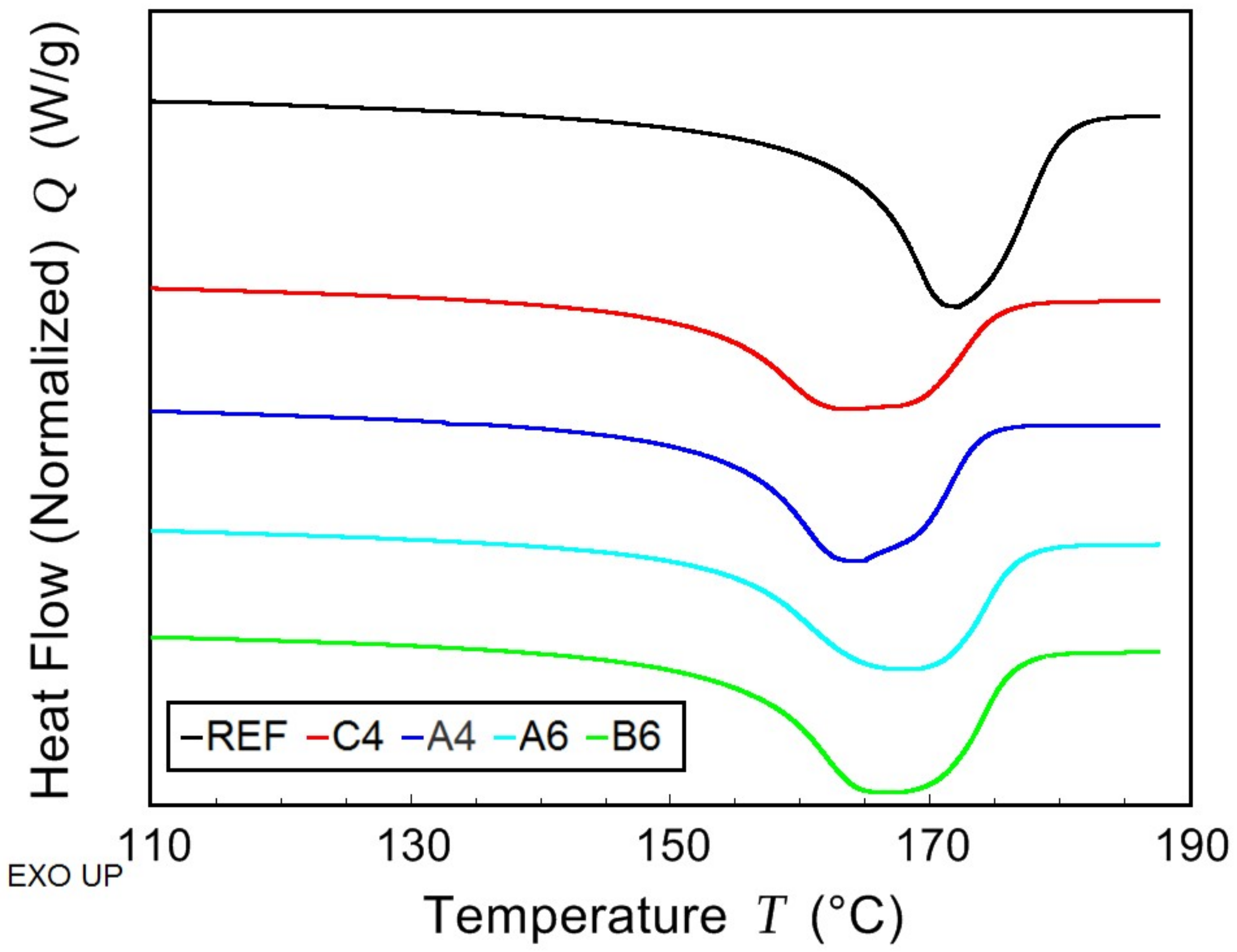
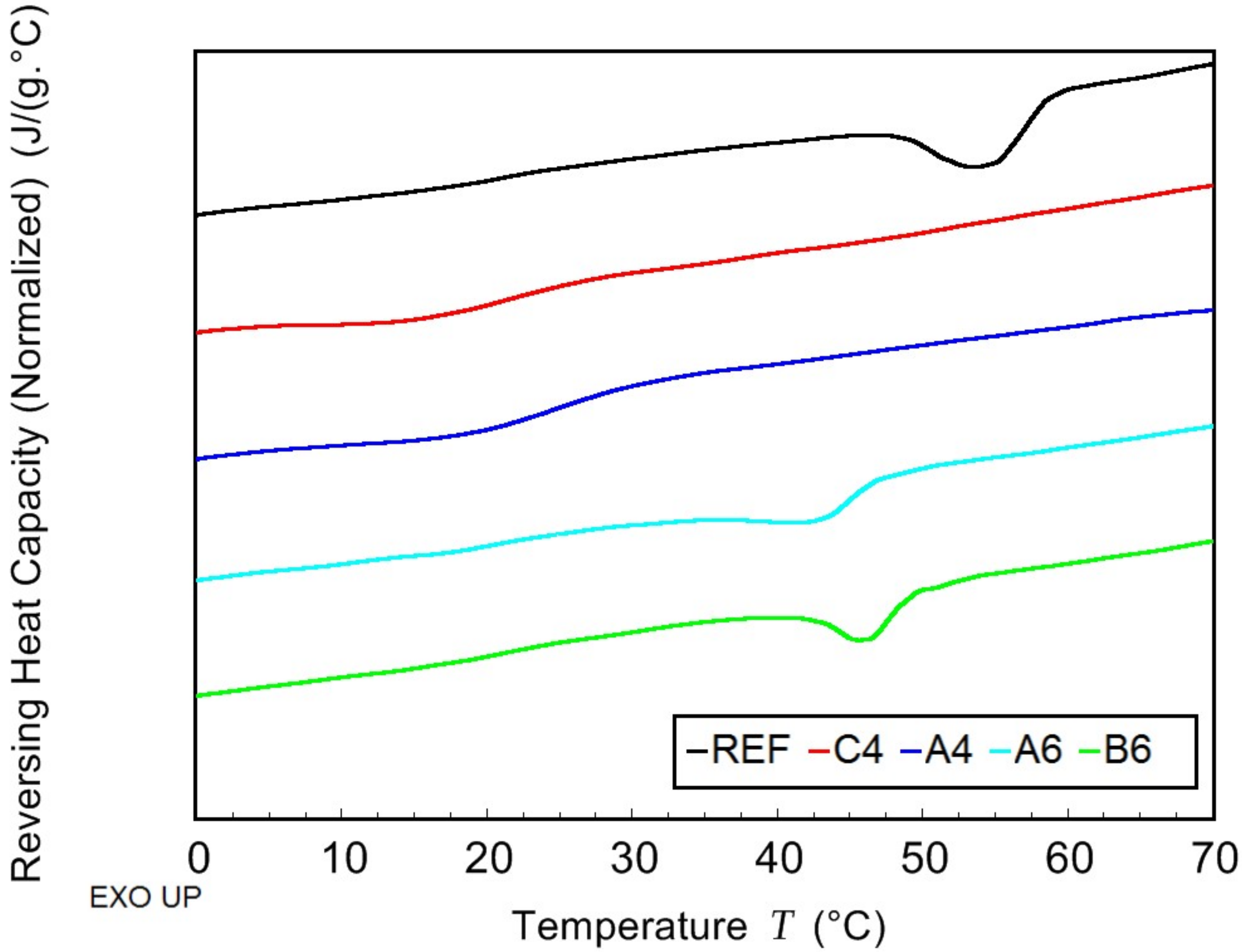
2.4. Modulated Differential Scanning Calorimetry—Thermal Characterization
The thermal properties of prepared samples were determined using the combination of modulated and conventional differential scanning calorimetry (MDSC and DSC, respectively). The measurements were performed on TA Instruments model DSC 2500 (TA Instruments, New Castle, DE, USA) under nitrogen atmosphere. The samples (approx. 13 mg) were sealed in hermetic aluminium pans. Firstly, they were cooled down to −50 °C and then kept in isothermal mode for 5 min. Afterwards, MDSC scan from −50 to 80 °C was carried out at average heating rate of 3 °C/min with the period of 60 s and modulation amplitude of 1.25 °C. This ensured that measured properties corresponded to as prepared samples without further thermal stress. Subsequently, two DSC scans (heating rate 10 °C/min), the first from 80 to 190 °C and the second from −30 to 190 °C, were performed. The glass transition temperature
Tg was determined from the MDSC curve of reversing heat capacity. The melting temperature
Tm and the crystallization temperature
Tc were determined from the conventional DSC scans. The crystallinity
Xc was calculated from the measured data using the following equation:
where Δ
Hm, and
(J/g) are the enthalpy of second melting peak and the enthalpy of fusion of 100% crystalline polymer (146 J/g for PHB), respectively.
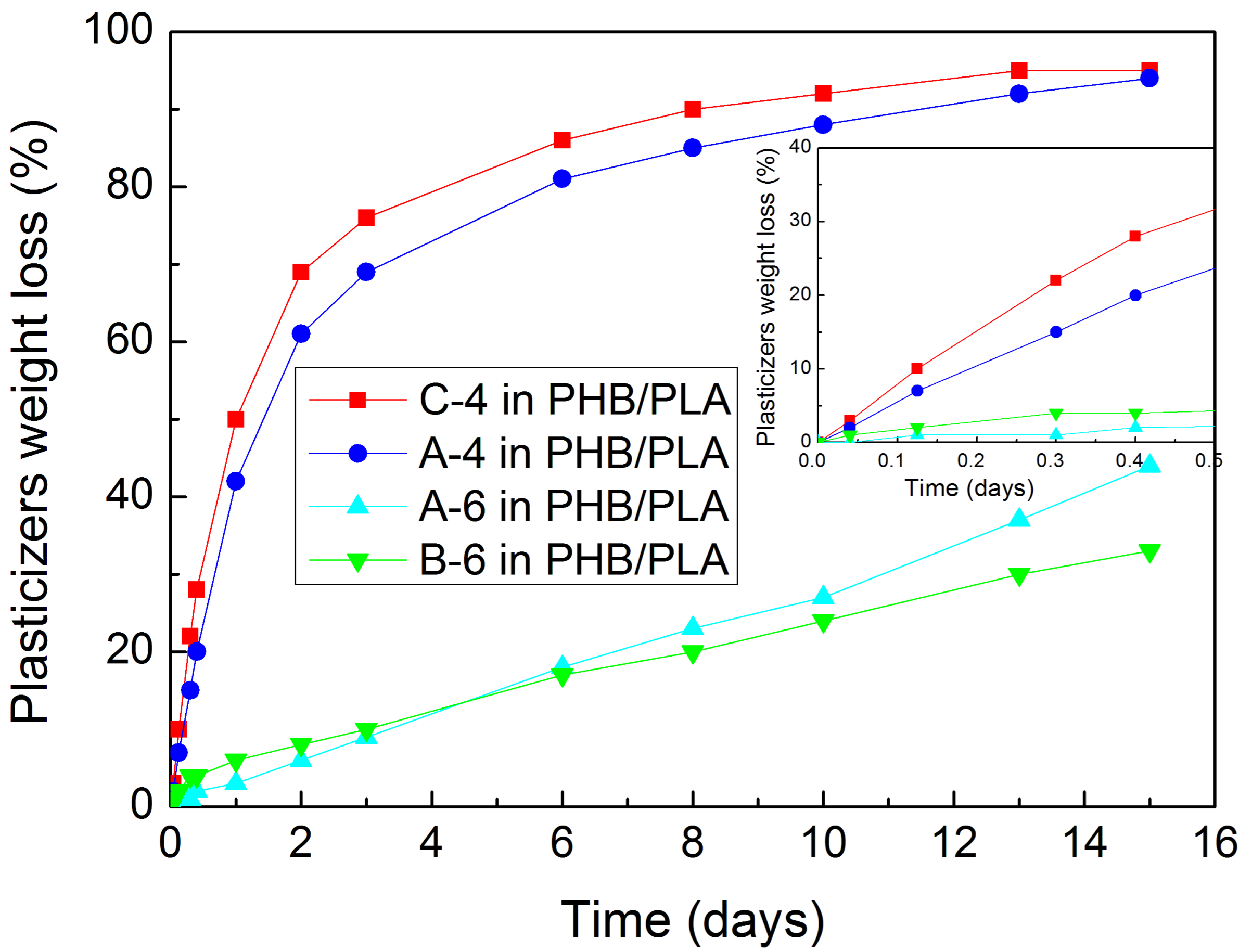
2.5. Migration Tests—Plasticizers Diffusion from PHB/PLA Blends
First experiments with the plasticizers diffusion from our blends were performed by the Thermogravimetric Analysis (TA Q500, TA Instruments, New Castle, Delaware USA). However, it was not possible to determine the diffusion speed exactly, as some of the plasticizers showed too long evaporation from the surface. Therefore another test was conducted. 5 g of extruded PHB/PLA/plasticizer pellets and 5 g of extruded PHB/PLA pellets (reference) were compression moulded on a laboratory hotpress to obtain planar samples of 0.70–0.72 mm thickness. The heating temperature of press desk was set to 190 °C. The laboratory press with the sample was closed during 0.5 min and the presswork itself lasted for 1 min. The samples of 3 × 3 cm square shape proportions were then cut from these moulded pressed planar samples, wiped by the napkin paper moistened in ethanol, laid down on the PET foil, and put into the drying oven heated to 110 °C with 50% air circulation. The samples were taken out from the drying oven after 1, 3, 7, 10 h, and 1, 2, 3, 6, 8, 10, 13, 15 days, and then they were wiped by the napkin paper moistened in ethanol and measured while using analytical balance. The dependencies of plasticizer’s weight loss from PHB/PLA planar samples on the time of exposition to 110 °C were evaluated.
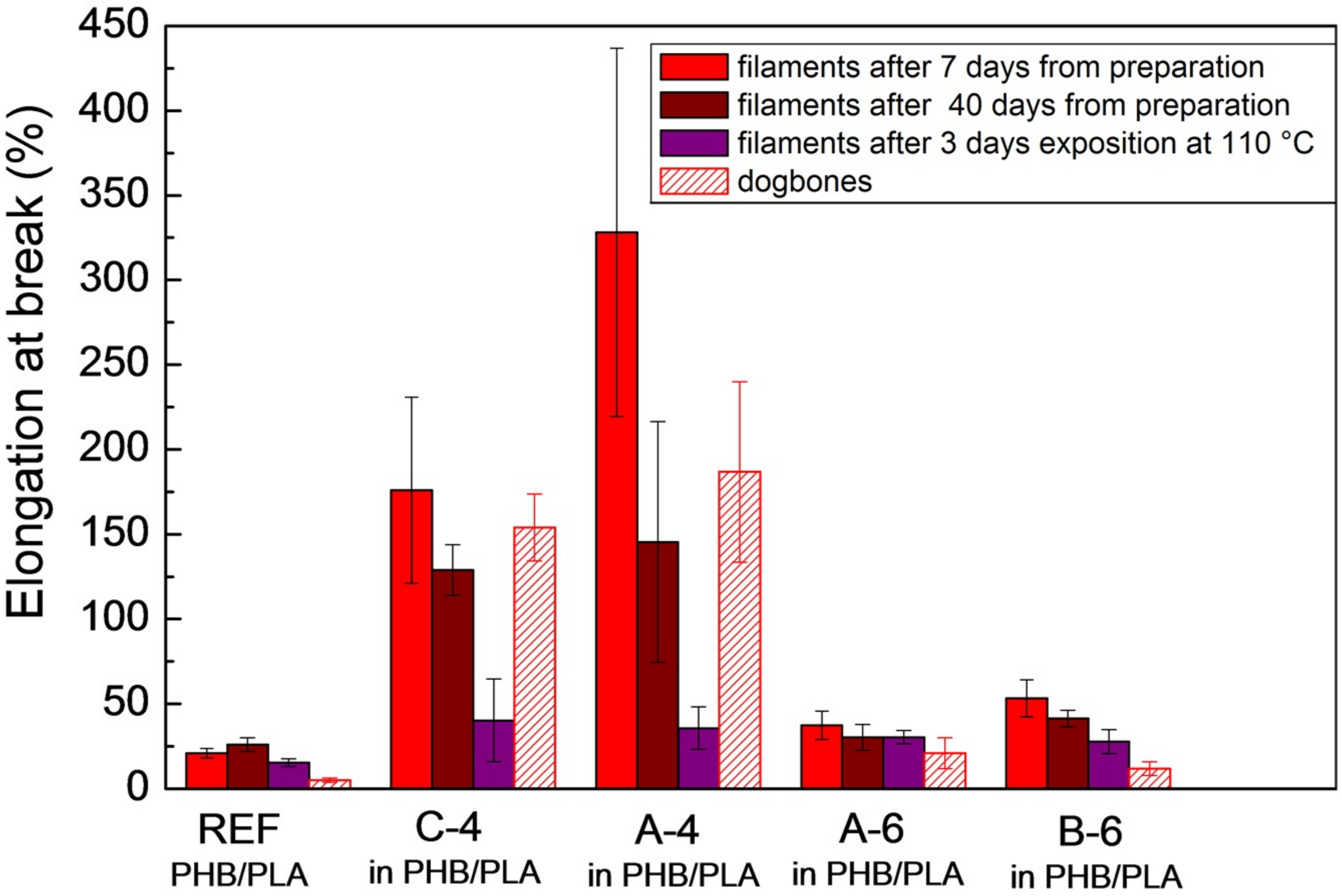
2.6. Tensile Tests—Mechanical Characterization
The elongation at break, the Young´s modulus, and the tensile strength were determined under ambient conditions, using the universal measurement device Zwick Z 010 (ZwickRoell GmbH & Co., Ulm, Germany). The tensile tests of non-plasticized extruded PHB/PLA samples (references), as well as plasticized PHB/PLA samples, were carried out using the load indicator with the maximal tensile force of 500 N and with pneumatic grips. The deformation rate was initially set to 5 mm/min to determine the elastic modulus, and then enhanced to 50 mm/min. The measurements were carried out without using the extensometer. The filaments with defined diameter were tested after three days of exposition to 110 °C in the drying oven and after 7 and 40 days after their preparation by extrusion. Double-paddle testing samples (dogbones, ~(5 × 2 × 75) mm, tensile test measurements approach CSN EN ISO 527-1) were 3D printed from mentioned extruded filaments (PHB/PLA reference sample and all PHB/PLA/plasticizer samples), and they were measured using Zwick Z 010 after seven days following the 3D printing [
42]. The reported values were the average of at least seven measurements. The data points were the mean value of each measurement, with the error bars in each graph representing the standard deviation.
2.7. 3D Printing
The dogbone samples for tensile tests were printed on PRUSA i3 MK2 3D printer (Prusa Research s.r.o., Praha, Czech Republic) by the FDM technology from PHB/PLA/plasticizer filaments with defined diameter, as mentioned above. AutoCAD (ver. 2018, Autodesk Inc., San Rafael, CA, USA) and Slic3r (software version 1.3.0, free software, developed by Alessandro Ranellucci) program were used for 3D virtual modeling and mathematical slicing. The dogbones were printed at 190 °C (195 °C during the first layers printing for better adhesion to the printing bed), at ambient conditions, without additional air cooling and bed heating. 3D printing was executed approximately six months after the filaments preparation.
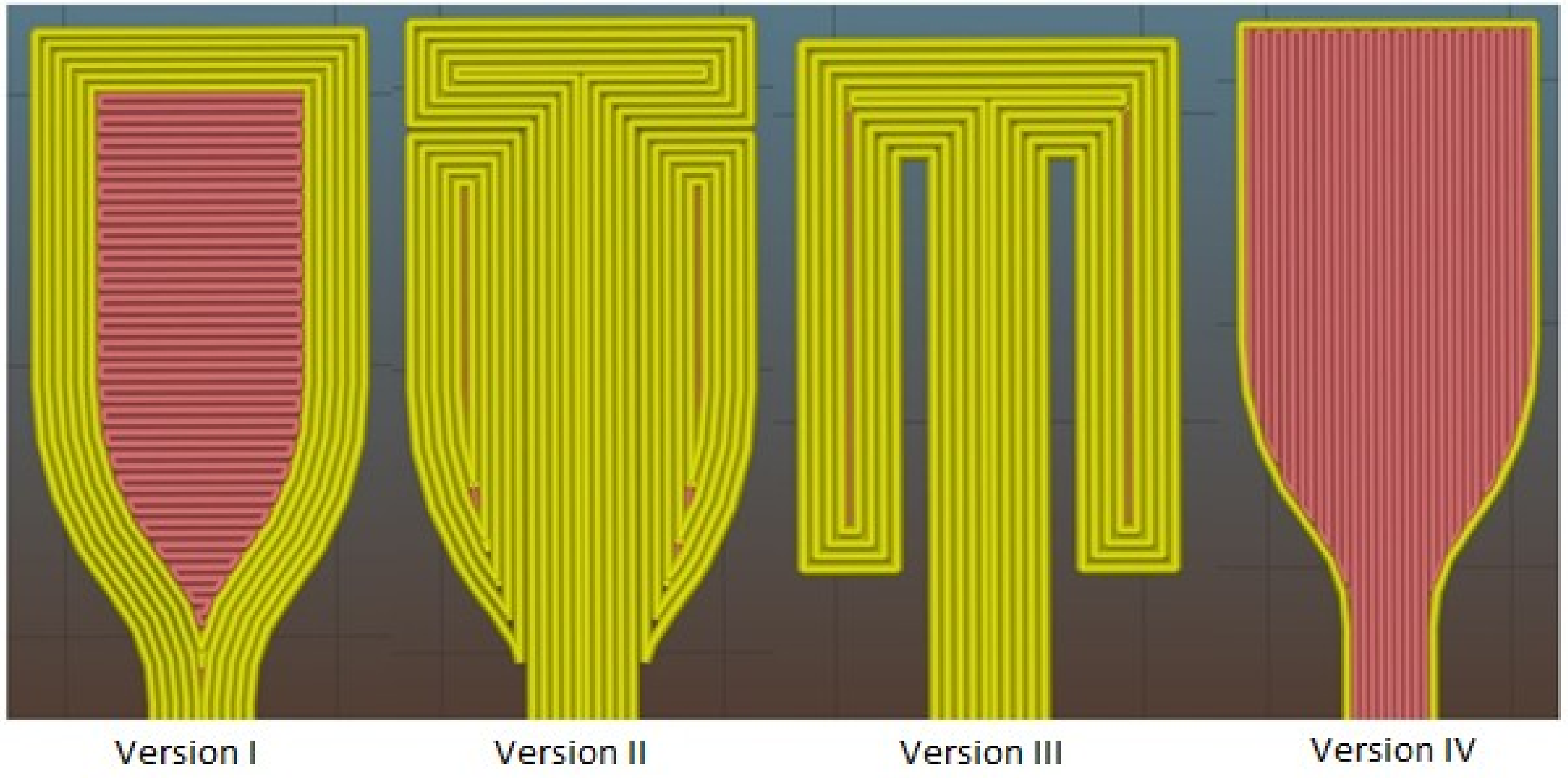
The evolution of 3D printed dogbone models that were prepared for further tensile tests can be seen in
Figure 2.
The Version I was created by simple import of fabricated model to Slic3r program and uploading to the printer. But, during the tensile test with dogbones of Version I, the delamination of perimeters occurred (illustrated by yellow color), and so the measured samples did not provide relevant results. To solve this problem, Version II was designed, where the perimeters of dogbone neck were elongated up to the upper part. The paddleboards were created from two parts. Unfortunately, during the tensile test, the defect at the point of paddleboard and neck connection occurred, because the printer created an inclusion in the paddleboard corner by releasing a large amount of melting. The inclusion acted as a disruption initiator and cracked untimely. The aim of Version III was to eliminate this problem, but the pressure of grips was not strong enough to hold the sample. The deformation then occurred in upper cross-sectional part and disagreed with real dogbone strength. The final Version (that could not be illustrated in
Figure 2) was created by the combination of 10 laminas of Version I and IV, which alternate regularly (five from each Version). The delamination of the neck in its extending part was eliminated by the insertion of vertical infill (illustrated by red color) and the vertical fracture of samples was eliminated by the combination with horizontal infill.
2.8. Scanning Electron Microscopy—Morphology Characterization
The Scanning Electron Microscopy (SEM) micrographs of dogbone areas that were fractured after tensile tests were obtained with the EVO® LS 10 (Zeiss Company, Oberkochen, Germany), operated at 10 kV. The samples were coated with a gold-palladium layer in vacuum conditions to increase their electrical conductivity. The micrographs were registered at 100× magnification. This method was used for the following samples (in wt %): 70PHB/30PLA (reference) and 60PHB/25PLA/15plasticizer samples (where all four Citroflex® plasticizers A-4, C-4, A-6, B-6 were used).
2.9. Determination of the Degree of Disintegration
The determination of the disintegration of plastic materials under simulated composting conditions in a laboratory-scale test was executed according to IS/ISO 20200 [
43]. Unfortunately, the method is not applicable to the determination of real biodegradability of plastic materials under garden or industrial composting conditions. The solid matrix used consists of synthetic solid waste inoculated with matured compost that was taken from a commercial garden composting plant. The pieces of plastic test material are composted with this prepared solid matrix. The degree of disintegration is determined after a composting cycle, by sieving the final matrix through a 2 mm sieve in order to recover the non-disintegrated residues. The reduction in mass of the test sample is considered as disintegrated material and is used to calculate the degree of disintegration [
43]. The composition of synthetic waste used in this method was: sawdust, rabbit-feed, ripe compost, corn-starch, saccharose, cornseed oil, and urea. The box with a lid made of polypropylene, having the dimensions of (30 × 20 × 10) cm, was used as a composting reactor. Two defined holes were made in the middle of two 20 cm sides to provide the gas exchange between inner atmosphere and outer environment. As samples, PHB/PLA/A-4 (60/25/15 in wt %) were used in the form of thin films with the dimensions approximately (6.5 × 10 × 0.08) cm. Two identical films were located in each box. The solid matrix was prepared manually by mixing different components that are mentioned above. Each box was filled by solid matrix up to the middle. Beforehand weighed and measured samples were laid on this matrix and covered with further matrix. Prepared content was then equally washed down by distilled water to overall 55% of humidity. Each reactor was closed, weighed, and placed in the air-circulation oven maintained at constant temperature of 58 °C for a minimum period of 45 days and maximum of 90 days, so that the samples were disintegrated under thermophilic conditions. The reactors were then weighed, water was added to restore the initial mass, or to restore the mass to 80% or 70% of the initial mass, the waste was stirred, and all of these actions were executed in particular days according to IS/ISO 20200. Also, other parameters, like visual appearance, odor, and color, were observed and the photos were taken.