Enhancing the Compatibility of Poly (1,4-butylene adipate) and Phenoxy Resin in Blends
Abstract
:1. Introduction
2. Experimental
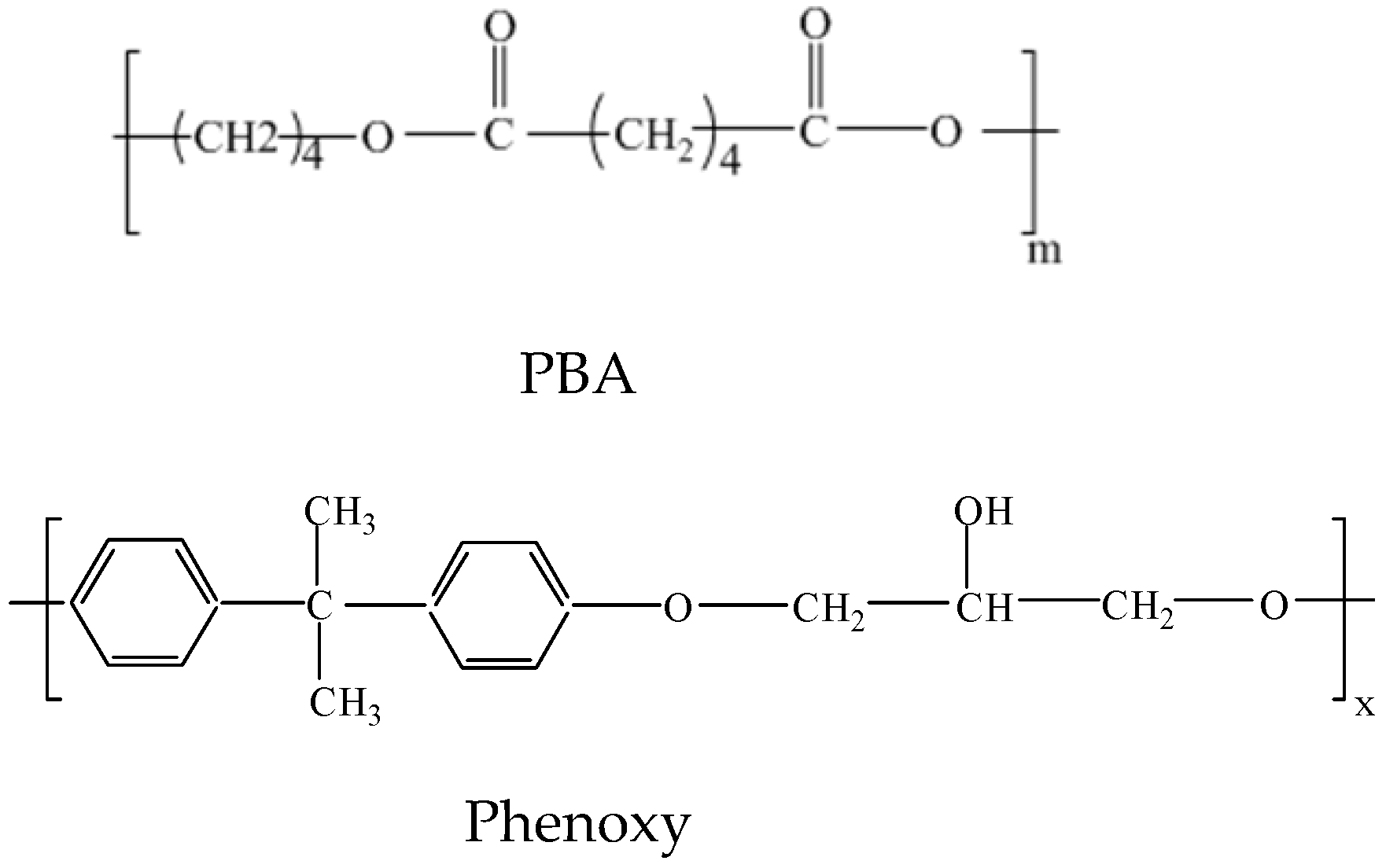
2.1. Materials
2.2. Preparation of Blends
2.3. Characterization
3. Results and Discussion
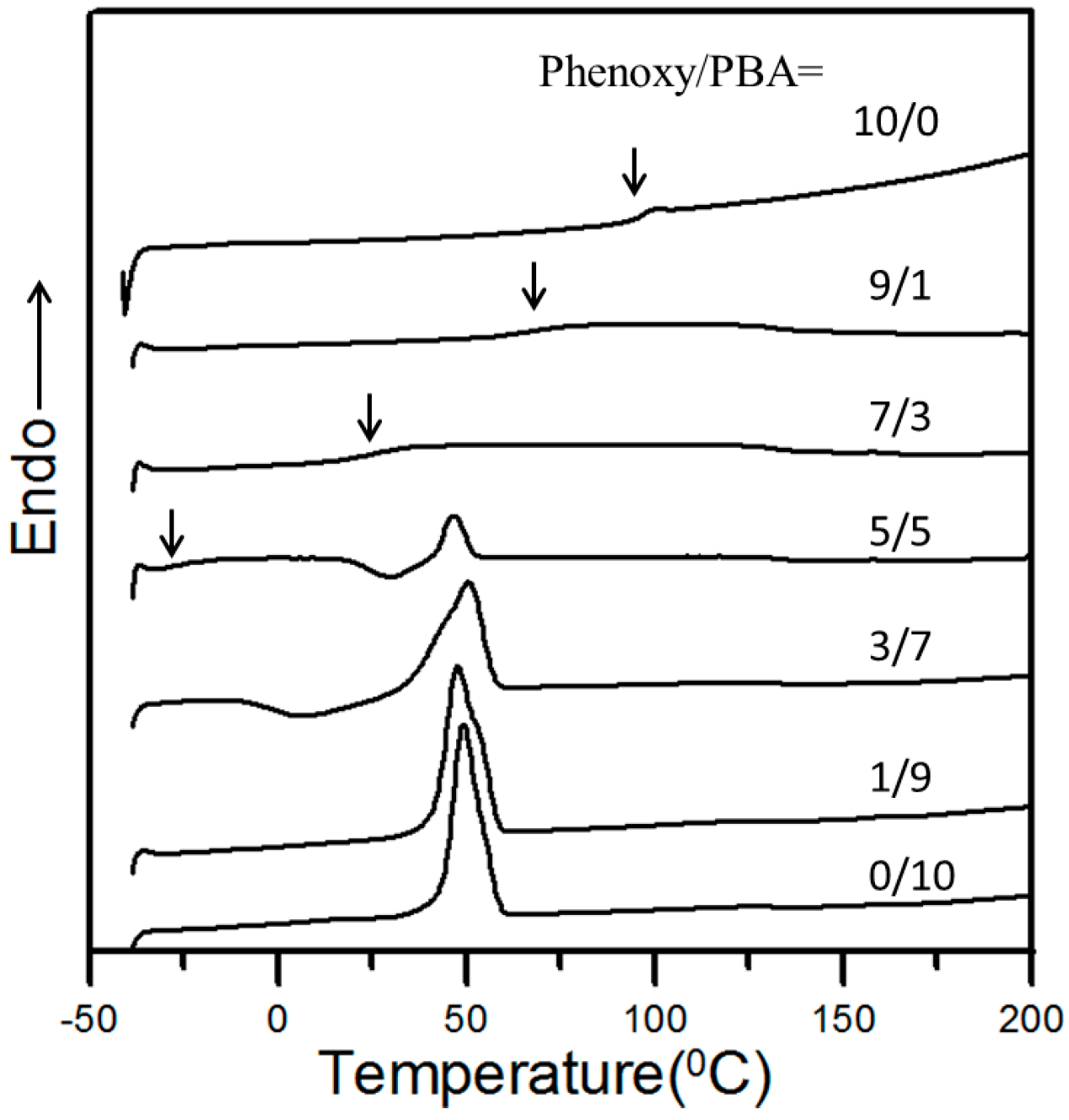
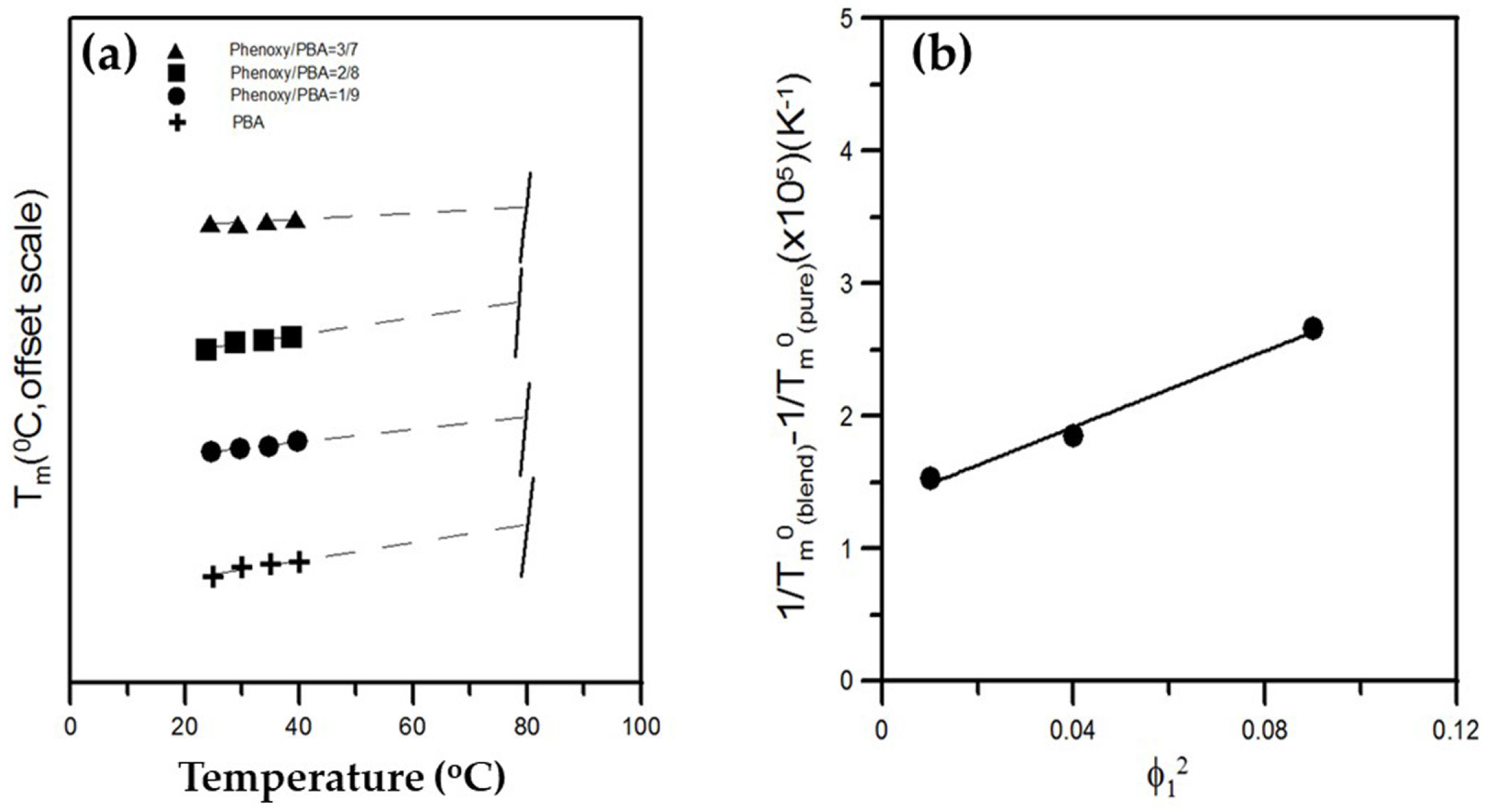
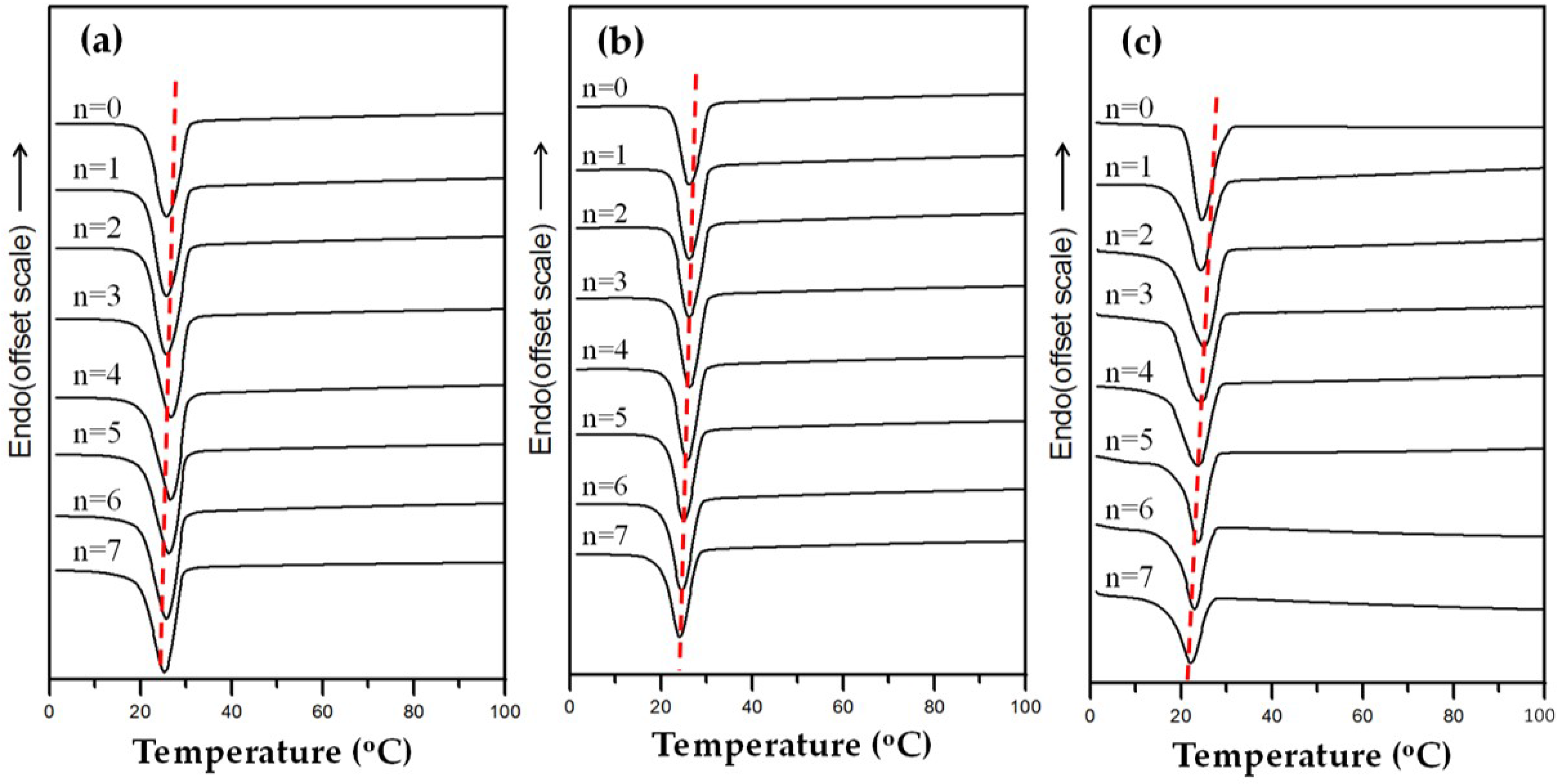
3.1. Thermal Analysis
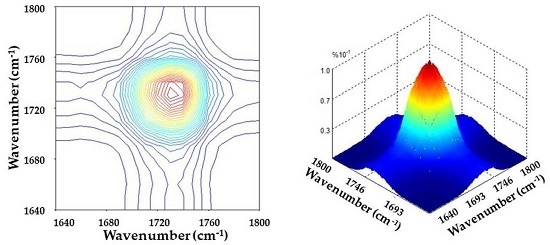
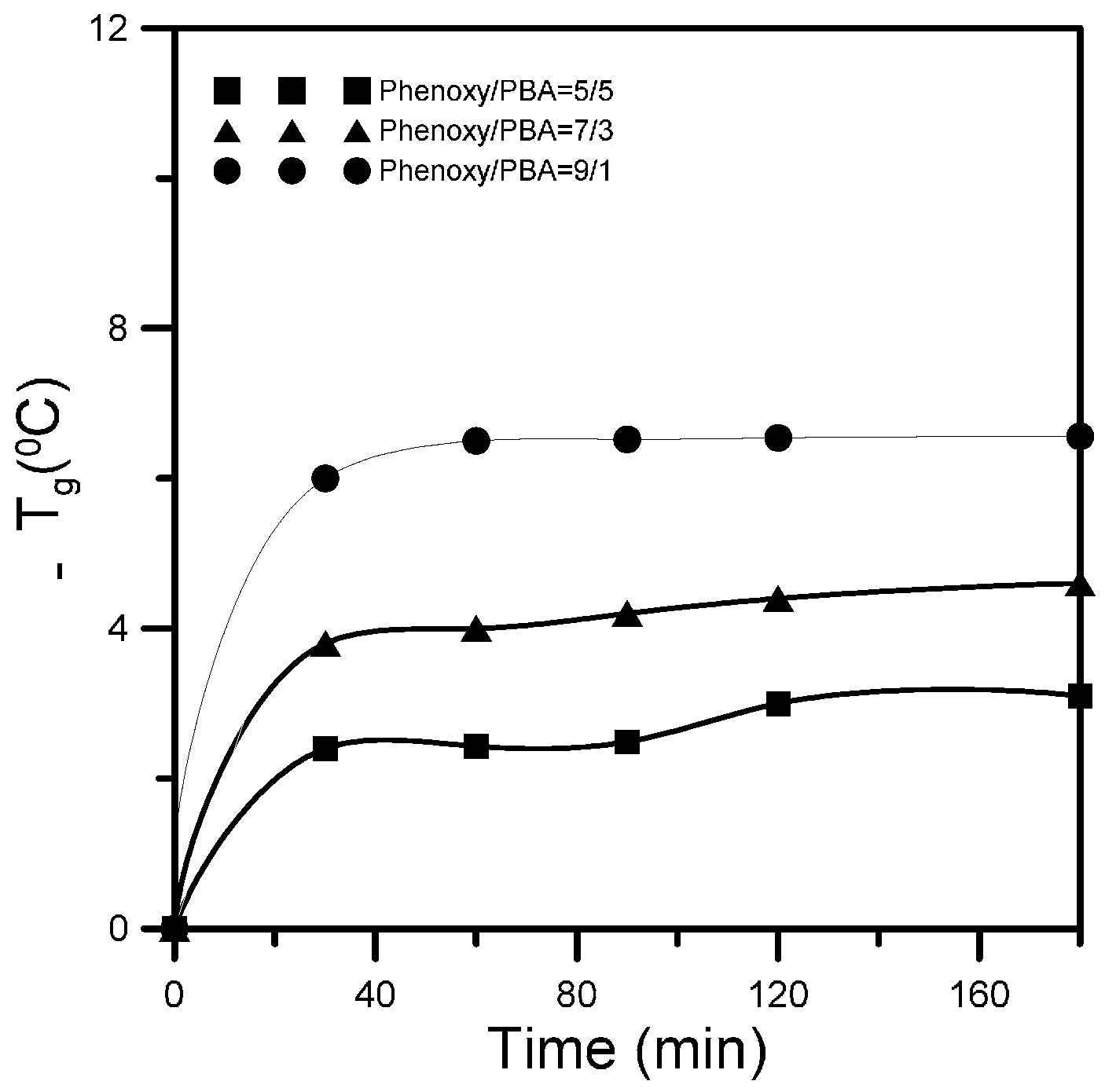
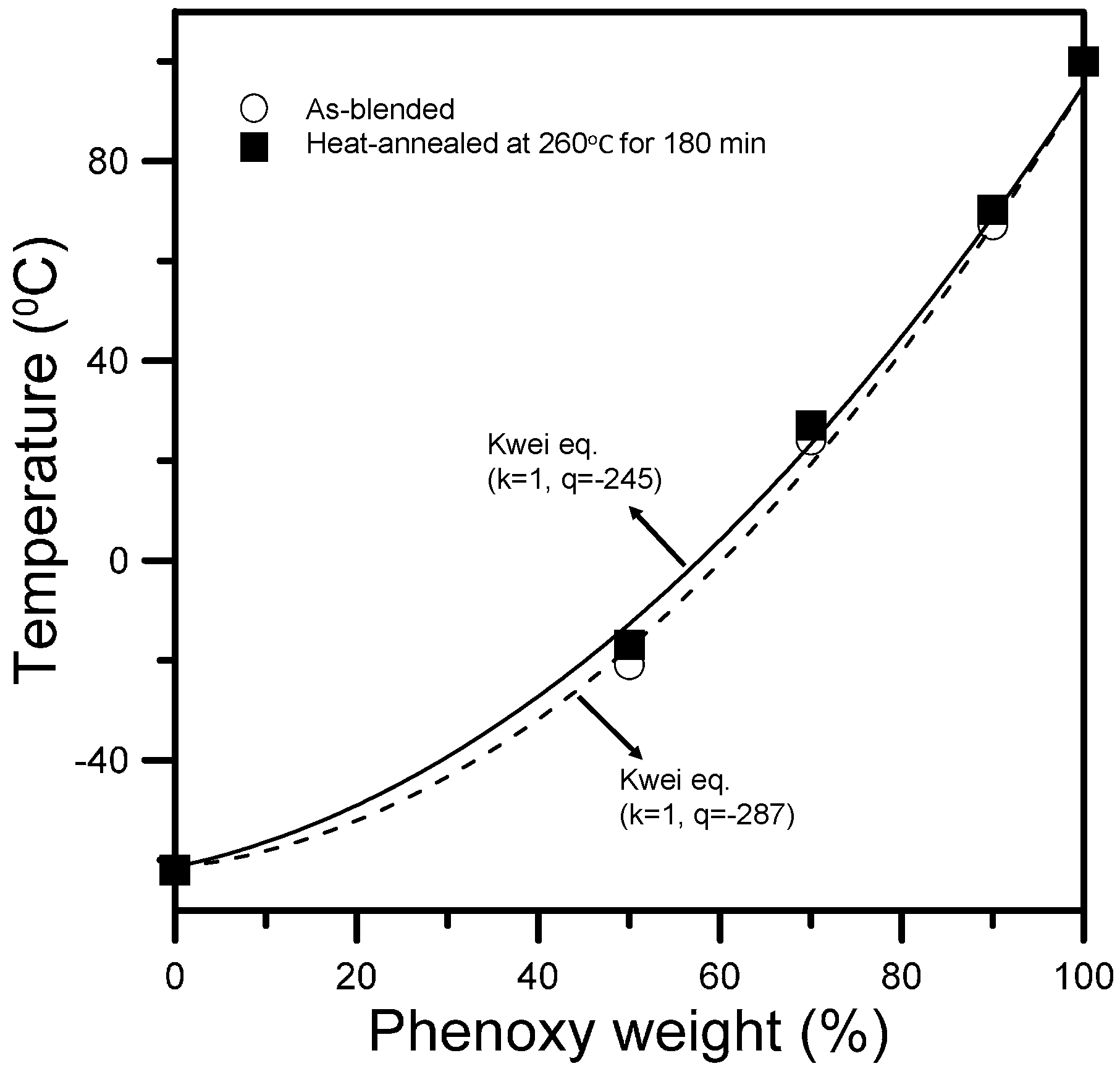
3.2. Specific Interactions upon Annealing
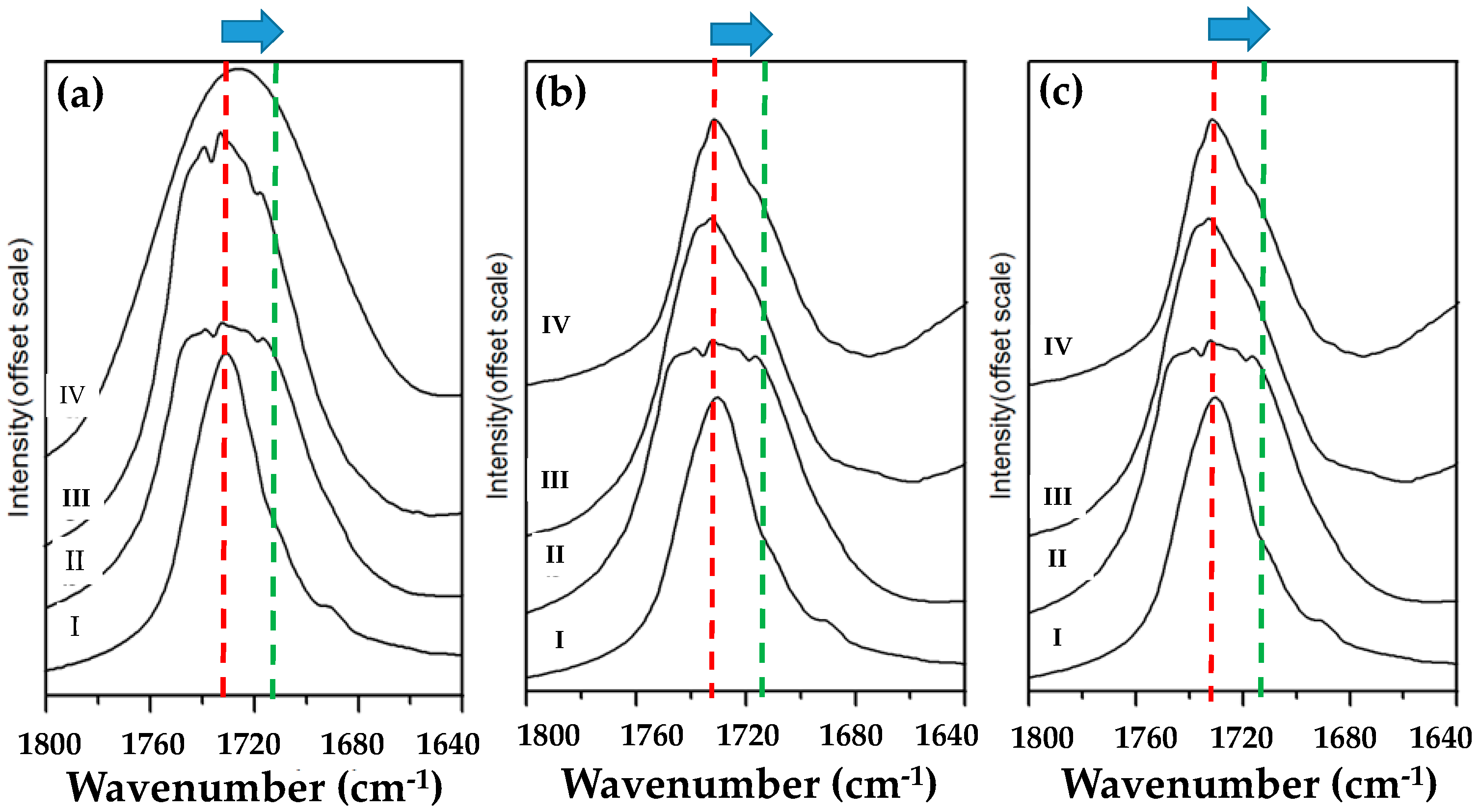
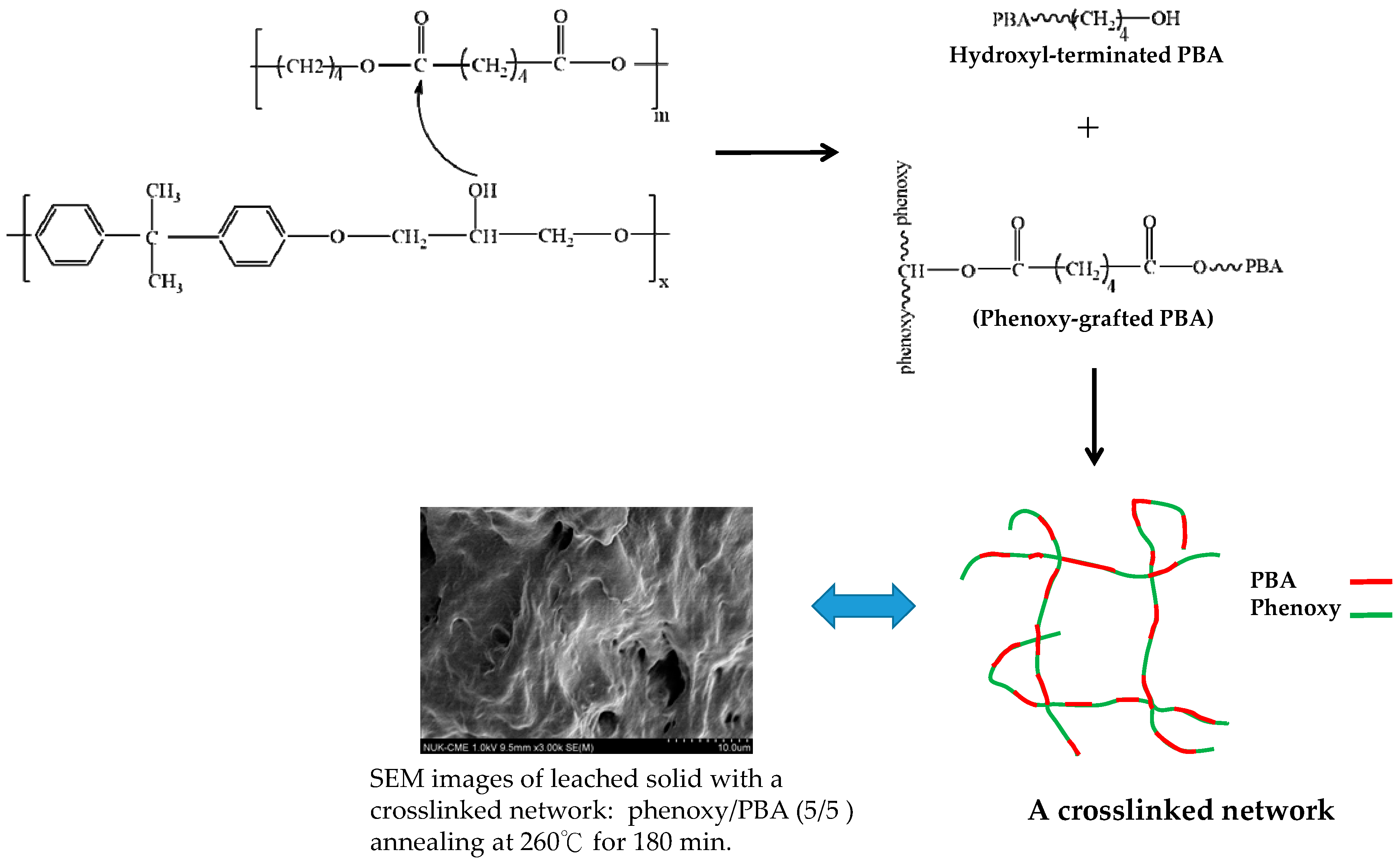
3.3. Chemical Interaction in PBA/Phenoxy Blends upon Annealing
3.4. Chemical Exchange Reactions in PBA/Phenoxy Blends
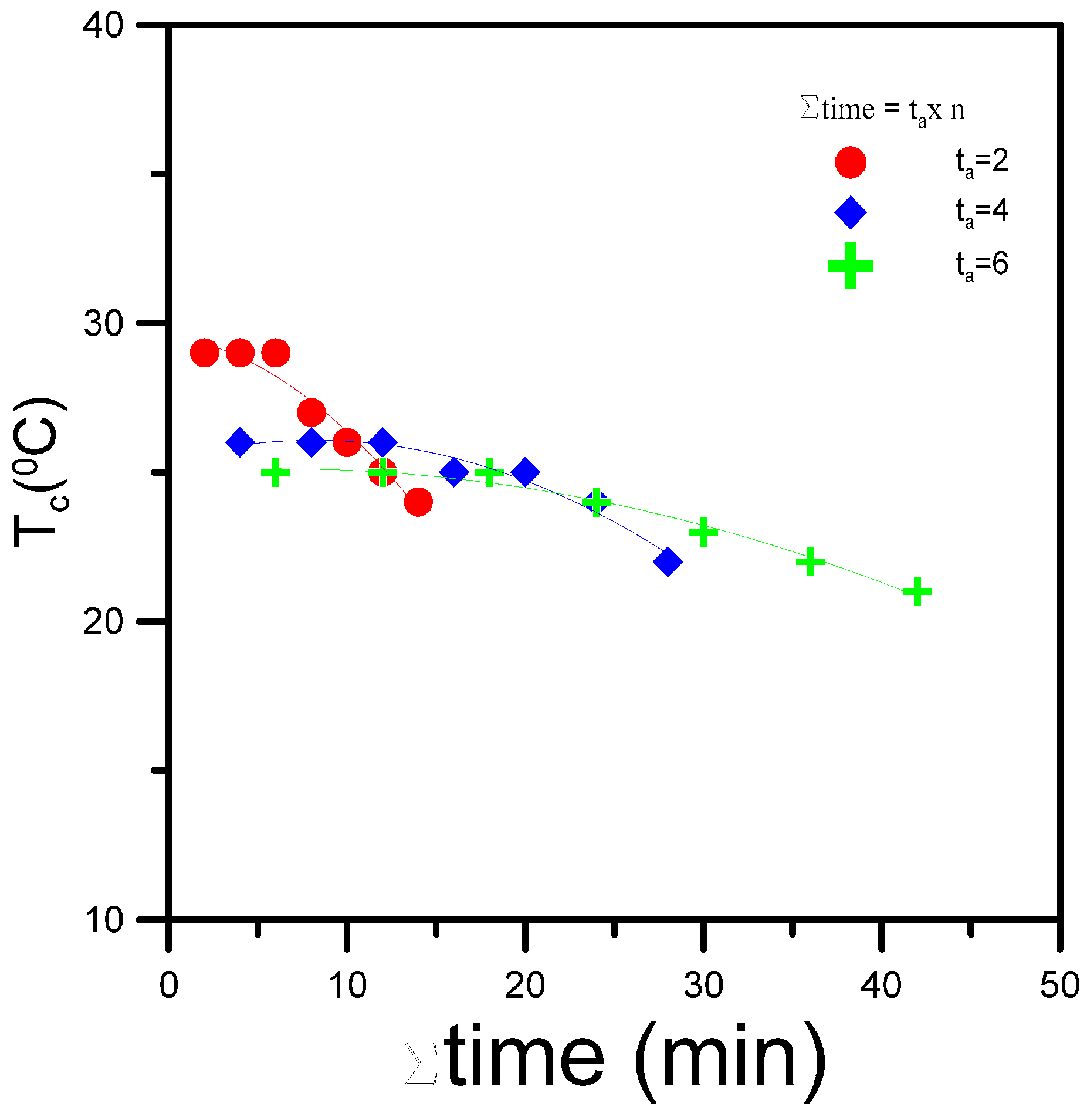
3.5. Effect of Heat-Annealed Samples on Crystallization Behavior
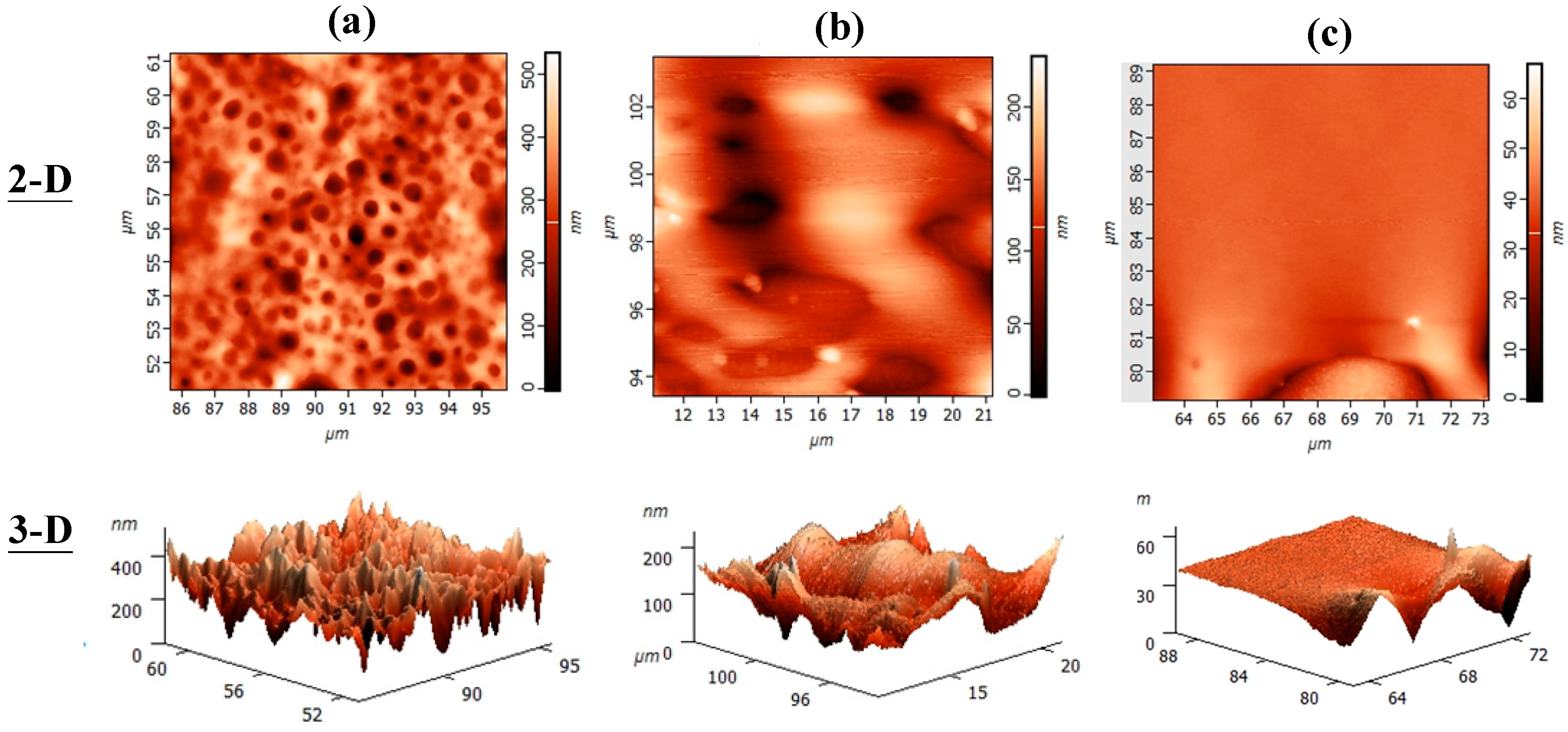
3.6. Surface Topography of Heat-Annealed Blends
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Su, C.C.; Shieh, C.K. Determination of interactions between aryl polyesters and poly (ether imide) via glass transition temperatures of separated phases in immiscible blends. Colloid Polym. Sci. 2005, 284, 66–73. [Google Scholar] [CrossRef]
- Su, C.C.; Wang, S.C.; Chen, W.J.; Lee, L.T. Phase compatibilization through chemical exchange reactions in blends of copolyesters with poly (hydroxyether of bisphenol-A) upon Annealing. Ind. Eng. Chem. Res. 2013, 52, 12587–12595. [Google Scholar] [CrossRef]
- Su, C.C.; Cheng, C.H. Miscibility windows in ternary polymer bles of a polyester, polymethacrylate and poly (styrene-co-acry). Polym. Int. 2003, 52, 1209–1216. [Google Scholar] [CrossRef]
- Lee, L.T.; Woo, E.M.; Chen, W.T.; Chang, L.; Yen, K.C. Phase behavior and interactions in blends of poly [(butylene adipate)-co-poly (butylene terephthalate)] copolyester with poly (4-vinyl phenol). Colloid Polym. Sci. 2010, 288, 439–448. [Google Scholar] [CrossRef]
- Paul, D.R.; Bucknall, C.B. Polymer Blends; John Wiley & Sons: New York, NY, USA, 2000; Volume 1, pp. 1–10. [Google Scholar]
- Su, C.C.; Shieh, C.K. Effect of ENPT Copolymers on Miscibility of Multiblend Blends of Poly (ethylene naphthalate)/Aryl Polyesters/Poly (ether imide). Polym. Eng. Sci. 2006, 46, 337–343. [Google Scholar] [CrossRef]
- Rana, D.; Mandal, B.M.; Bhattacharyya, S.N. Analogue calorimetric studies of blends of poly (vinyl ester)s and polyacrylates. Macromolecules 1996, 29, 1579–1583. [Google Scholar] [CrossRef]
- Rana, D.; Mandal, B.M.; Bhattacharyya, S.N. Analogue calorimetry of polymer blends: Poly (styrene-co-acrylonitrile) and poly (phenyl acrylate) or poly (vinyl benzoate). Polymer 1996, 37, 2439–2443. [Google Scholar] [CrossRef]
- Rana, D.; Mandal, B.M.; Bhattacharyya, S.N. Miscibility and phase diagrams of poly (phenyl acrylate) and poly (styrene-coacrylonitrile) blends. Polymer 1993, 34, 1454–1459. [Google Scholar] [CrossRef]
- Rana, D.; Kim, H.L.; Kwag, H.; Choe, S. Hybrid blends of similar ethylene 1-octene copolymers. Polymer 2000, 41, 7067–7082. [Google Scholar] [CrossRef]
- Rana, D.; Kim, H.L.; Kwag, H.; Rhee, J.; Cho, K.; Woo, T.; Lee, B.H.; Choe, S. Blends of ethylene 1-Octene copolymer synthesized by Ziegler–Natta and metallocene catalysts. II. Rheology and morphological behaviors. J. Appl. Polym. Sci. 2000, 76, 1950–1964. [Google Scholar] [CrossRef]
- Rana, D.; Cho, K.; Woo, T.; Lee, B.H.; Choe, S. Blends of ethylene 1-Octene copolymer synthesized by Ziegler–Natta and metallocene catalysts. I. thermal and mechanical properties. J. Appl. Polym. Sci. 1999, 74, 1169–1177. [Google Scholar] [CrossRef]
- Rana, D.; Lee, C.H.; Cho, K.; Lee, B.H.; Choe, S. Thermal and mechanical properties for binary blends of metallocene polyethylene with conventional polyolefins. J. Appl. Polym. Sci. 1998, 69, 2441–2450. [Google Scholar] [CrossRef]
- Baker, W.E.; Scott, C.E.; Hu, G.H. Reactive Polymer Blending; Hanser: Munchen, Germany, 2001. [Google Scholar]
- Bank, M.; Leffingwell, J.; Thies, C. The Influence of Solvent upon the Compatibility of Polystyrene and Poly (vinyl methyl ether). Macromolecules 1971, 4, 43–46. [Google Scholar] [CrossRef]
- Coleman, M.M.; Moskala, E.J. FTi.r. Studies of polymer blends containing the poly (hydroxy ether of bisphenol A) and poly (ε-caprolactone). Polymer 1983, 24, 251–257. [Google Scholar] [CrossRef]
- Su, C.C.; Lin, J.H. Ringed spherulites in ternary polymer blends of poly (ε-caprolactone), poly (styrene-co-acrylonitrile) and polymethacrylate. Colloid Polym. Sci. 2004, 283, 182–193. [Google Scholar] [CrossRef]
- Su, C.C.; Chen, C.H.; Shih, N.L.; Li, Y.S. Chemical Exchange Reactions in Blends of the Biodegradable Polyester with poly (hydroxy ether of bisphenol-A). Adv. Mater. Res. 2014, 953, 1246–1249. [Google Scholar] [CrossRef]
- Kalogeras, I.M. Glass-transition phenomena in polymer blends. In Encyclopedia of Polymer Blends, Volume 3: Structure; Isayev, A.I., Ed.; Wiley-VCH: Weinheim, Germany, 2016; Chapter 1; pp. 1–134. ISBN 978-3-527-31931-2. [Google Scholar]
- Su, C.C.; Woo, E. M. Chemical interactions in blends of bisphenol A polycarbonate with tetraglycidyl-4,4′-diaminodiphenylmethane epoxy. Macromolecules 1995, 28, 6779–6786. [Google Scholar] [CrossRef]
- Seyfia, J.; Jafari, S.H.; Khonakdarb, H.A.; Sahac, P.; Goodarzia, V. Investigating the role of transreactions on degradation behavior of phenoxy/poly (trimethylene terephthalate)/clay nanocomposites using thermal analysis techniques. Thermochim. Acta 2010, 511, 59–66. [Google Scholar] [CrossRef]
- Farmahini-Farahani, M.; Jafari, S.H.; Khonakdar, H.A.; Bo¨hme, F.; Komber, H.; Yavari1, A. Tarameshlou1 M. Investigation of exchange reactions and rheological response of reactive blends of poly (trimethylene terephthalate) and phenoxy resin. Polym. Int. 2008, 57, 612–617. [Google Scholar] [CrossRef]
- Yang, F.; Qiu, Z.A. Comparative study of crystallization kinetics of a miscible biodegradable poly (butylene succinate-co-butylene adipate) blend with poly (hydroxyl ether biphenyl A) and its neat component. Thermochim. Acta 2011, 523, 200–206. [Google Scholar] [CrossRef]
- Woo, E.M.; Mao, Y.J.; Wu, P.; Su, C.C. Ternary miscibility in blends of three polymers with balanced binary interactions. Polym. Eng. Sci. 2003, 43, 543–557. [Google Scholar] [CrossRef]
- Harris, J.E.; Goh, S.H.; Paul, D.R.; Barlow, J.W. Miscible binary blends containing the polyhydroxy ether of bisphenol-A and various aliphatic polyesters. J. Appl. Polym. Sci. 1982, 27, 839–855. [Google Scholar] [CrossRef]
- Nurkhamidah, S.; Eamor, M.W.; Huang, I.H.; Su, C.C. Phase behavior and crystal morphology in poly (ethylene succinate) biodegradably modified with tannin. Colloid Polym. Sci. 2011, 289, 1563–1578. [Google Scholar] [CrossRef]
- Stuart, W. Chemistry of the Carbonyl Group, 99th ed.; John Wiley & Sons, Ltd.: New York, NY, USA, 1991; Volume 1, pp. 89–121. [Google Scholar]
- Vollhardt, K.; Peter, C. The Carbonyl Group-Organic Chemistry Structure and Function, 5th ed.; Freeman: New York, NY, USA, 2007; Volume 1, pp. 14–35. [Google Scholar]
- Vollhardt, P.; Schore, N. Organic Chemistry: Structure and Function, 6th ed.; Freeman: New York, NY, USA, 2013; pp. 886–898. [Google Scholar]
- Robeson, L.M.; Hale, W.F.; Merriam, C.N. Misciblity of the poly (hydroxy ether) of bisphenol A with water-soluble polyethers. Macromolecules 1981, 14, 1644–1650. [Google Scholar] [CrossRef]
- Chiou, J.S.; Paul, D.R. Phase behavior of PMMA-phenoxy blends. J. Appl. Polym. Sci. 1991, 42, 279–280. [Google Scholar] [CrossRef]
- Zheng, S.Q.; Guo, Y.M. Examination of miscibility at molecular level of poly (hydroxy ether of bisphenol A)/poly (N-vinylpyrrolidone) blends by cross-polarization/magic angle spinning 13C nuclear magnetic resonance spectroscopy. J. Polym. Sci. Part B Polym. Phys. Ed. 1998, 36, 2291–2300. [Google Scholar] [CrossRef]
- Dai, J.; Goh, S.H.; Lee, S.Y.; Siow, K.S. Complexation between poly (hydroxyether of bisphenol A) and three tertiary amide polymers. Polymer 1996, 37, 3259–3264. [Google Scholar] [CrossRef]
- Yuan, Y.; Ruckenstein, E. Miscibility and transesterification of phenoxy with biodegradable poly (3-hydroxybutyrate). Polymer 1998, 39, 1893–1897. [Google Scholar] [CrossRef]
- Eguiazabal, J.I.; Iruin, J.J. Miscibility and thermal decomposition in phenoxy/poly (ethylene terephthalate) and phenoxy/poly (butylene terephthalate) blends. Mater. Chem. Phys. 1987, 18, 147–154. [Google Scholar] [CrossRef]
- Sperling, L.H. Introduction to Physical Polymer Science, 4th ed.; Willey: New York, NY, USA, 2006; pp. 395–406. [Google Scholar]
- Fried, J.R. Polymer Science and Technology, 2nd ed.; Pearson Education Taiwan Ltd.: Taipei, Taiwan, 2004; pp. 172–183. [Google Scholar]
- Coleman, M.M.; Graf, J.F.; Painter, P.C. Specific Interactions and the Miscibility of Polymer Blends; Technomic Publishing Company Inc.: Lancaster, PA, USA, 1991; pp. 65–76. [Google Scholar]
- Chang, L.L.; Woo, E.M. Thermal, morphology, and NMR characterizations on phase behavior and miscibility in blends of isotactic polystyrene with poly (cyclohexyl methacrylate). J. Polym. Sci. Part B Polym. Phys. Ed. 2003, 41, 772–784. [Google Scholar] [CrossRef]
- Su, C.C.; Shieh, C.K. Miscibility and transesterification in ternary blends of poly (ethylene naphthalate)/poly (pentamethylene terephthalate)/poly (ether imide). Colloid Polym. Sci. 2005, 283, 1278–1288. [Google Scholar] [CrossRef]
- Hoffman, J.D.; Weeks, J.J. Rate of Spherulitic Crystallization with Chain Folds in Polychlorotrifluoroethylene. J. Chem. Phys. 1962, 37, 1723–1741. [Google Scholar] [CrossRef]
- Brandrup, J.; Immergut, E.H.; Grulke, E.A.; Abe, A.; Bloch, D.R. Polymer Handbook, 4th ed.; John Wiley & Sons: New York, NY, USA, 1999. [Google Scholar]
- Fernandez-Barridi, M.J.; Valero, M.; Martinez de Ilarduya, A.; Espi, E.; Iruin, J.J. Phenoxy blends: An approach to the miscibility by FTi.r. and chemical modification of the interacting sites. Polymer 1993, 34, 38–42. [Google Scholar] [CrossRef]
- Kwei, T.K. The effect of hydrogen bonding on the glass transition temperatures of polymer mixtures. J. Polym. Sci. Polym. Lett. 1984, 22, 307–313. [Google Scholar] [CrossRef]
- Painter, P.C.; Graf, J.F.; Coleman, M.M. Effect of hydrogen bonding on the enthalpy of mixing and the composition dependence of the glass transition temperature in polymer blends. Macromolecules 1991, 24, 5630–5638. [Google Scholar] [CrossRef]
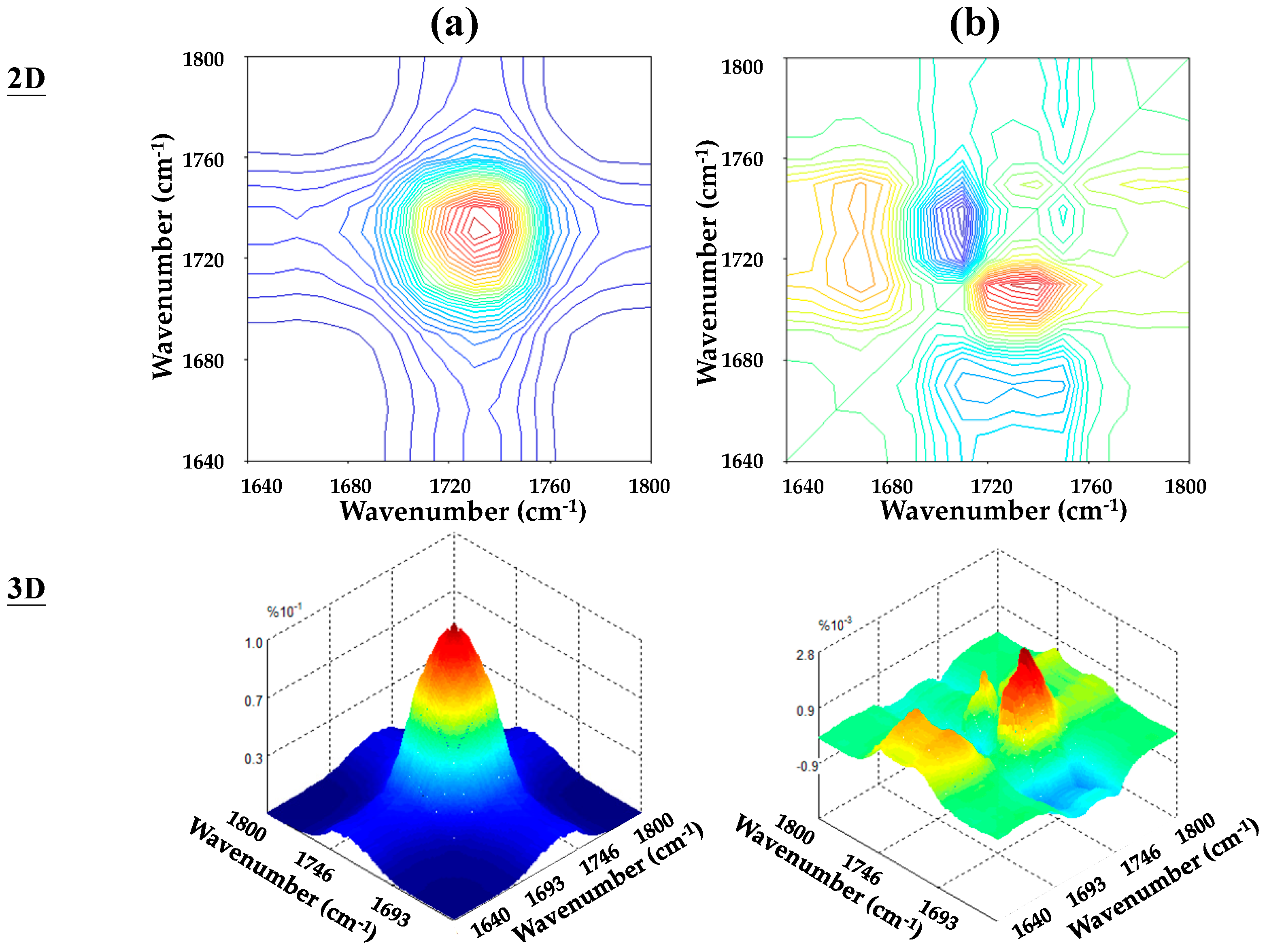
| Ester groups | Wavenumber (cm−1) |
|---|---|
 | 1730 cm−1 |
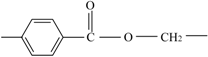 | 1716 cm−1 |
 | 1750 cm−1 |
 | 1735 cm−1 |
| Exothermic Heat of Crystallization ΔHn (J/g) | Annealing Times Per Cycle | ||
|---|---|---|---|
| 2 min | 4 min | 6 min | |
| ΔHn=0 | 70.9 | 61.2 | 39.6 |
| ΔHn=1 | 65.8 | 57.3 | 34.3 |
| ΔHn=2 | 60.8 | 52.2 | 30.2 |
| ΔHn=3 | 55.7 | 49.3 | 15.4 |
| ΔHn=4 | 51.6 | 46.2 | 14.8 |
| ΔHn=5 | 49.6 | 43.0 | 14.6 |
| ΔHn=6 | 45.4 | 42.4 | 14.4 |
| ΔHn=7 | 44.6 | 40.2 | 12.4 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, C.-F.; Wang, H.-C.; Su, C.-C. Enhancing the Compatibility of Poly (1,4-butylene adipate) and Phenoxy Resin in Blends. Materials 2017, 10, 692. https://doi.org/10.3390/ma10070692
Yang C-F, Wang H-C, Su C-C. Enhancing the Compatibility of Poly (1,4-butylene adipate) and Phenoxy Resin in Blends. Materials. 2017; 10(7):692. https://doi.org/10.3390/ma10070692
Chicago/Turabian StyleYang, Cheng-Fu, Hsiang-Ching Wang, and Chean-Cheng Su. 2017. "Enhancing the Compatibility of Poly (1,4-butylene adipate) and Phenoxy Resin in Blends" Materials 10, no. 7: 692. https://doi.org/10.3390/ma10070692
APA StyleYang, C.-F., Wang, H.-C., & Su, C.-C. (2017). Enhancing the Compatibility of Poly (1,4-butylene adipate) and Phenoxy Resin in Blends. Materials, 10(7), 692. https://doi.org/10.3390/ma10070692