On the Morphology of the SDS Film on the Surface of Borosilicate Glass
Abstract
:1. Introduction
2. Results
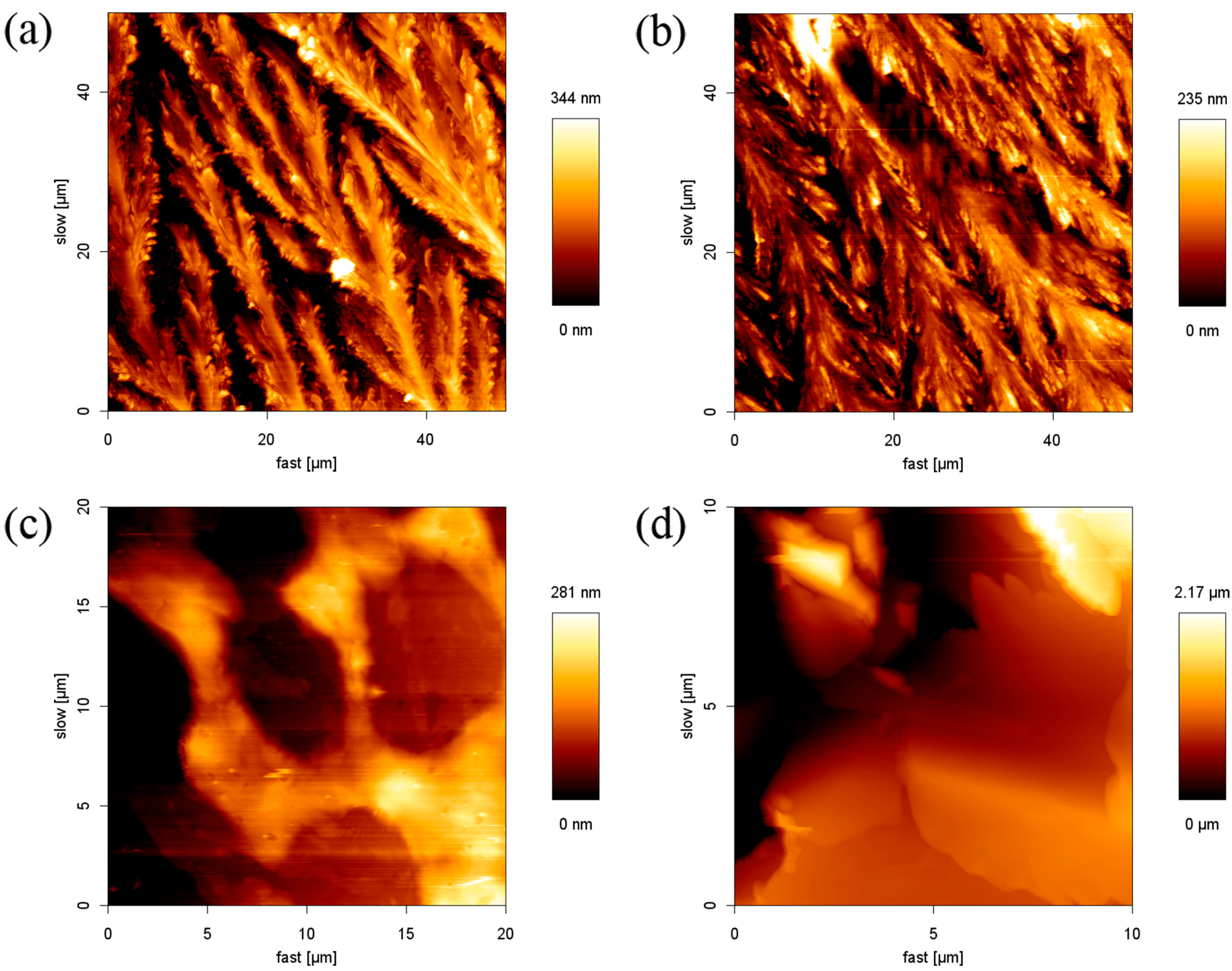
2.1. Structure of the SDS Aggregate
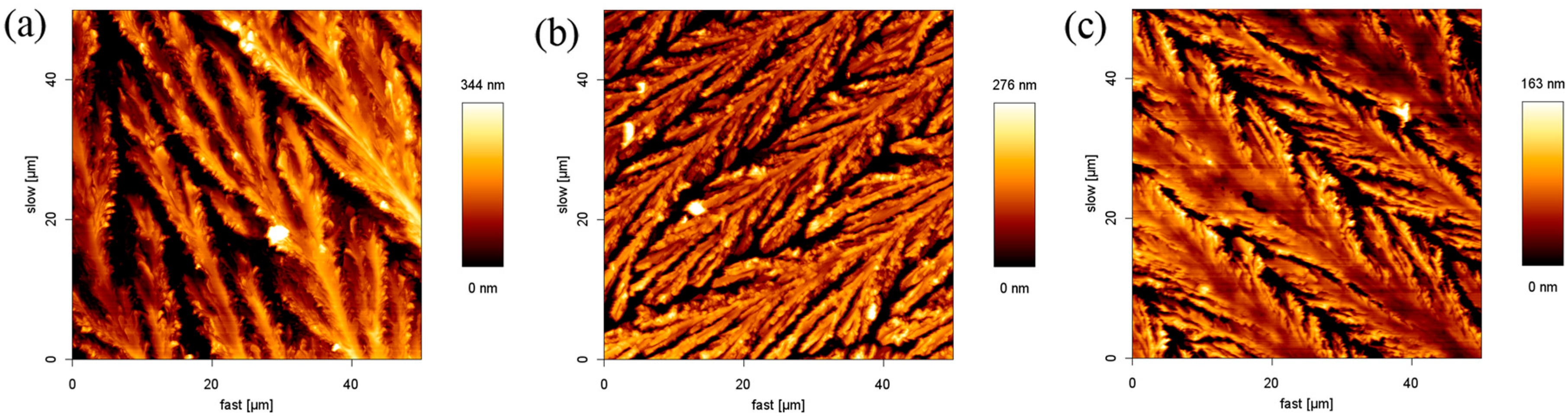
2.2. The Effect of the Cations on the Structure
2.3. The Effect of Anions on the Structure
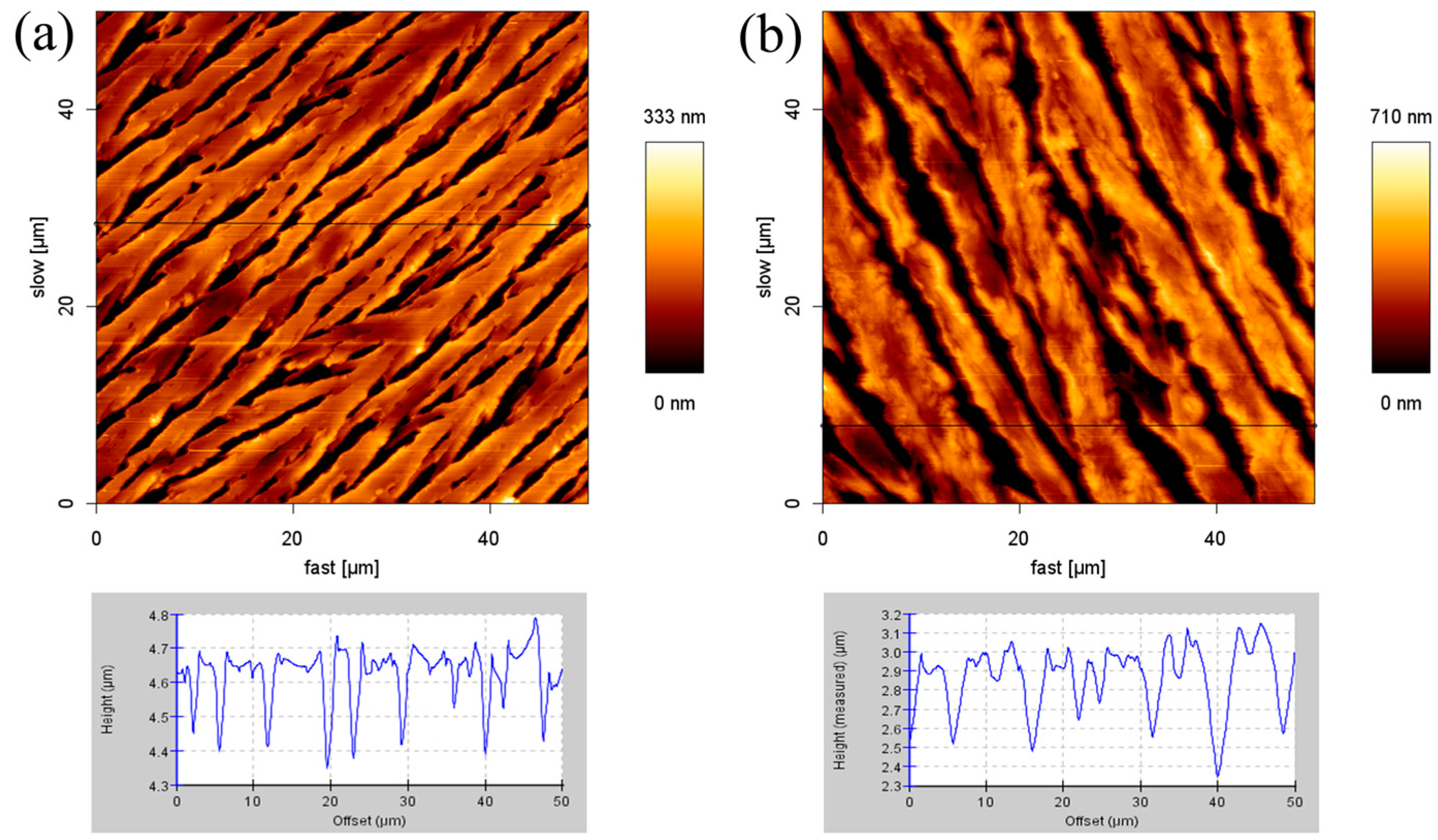
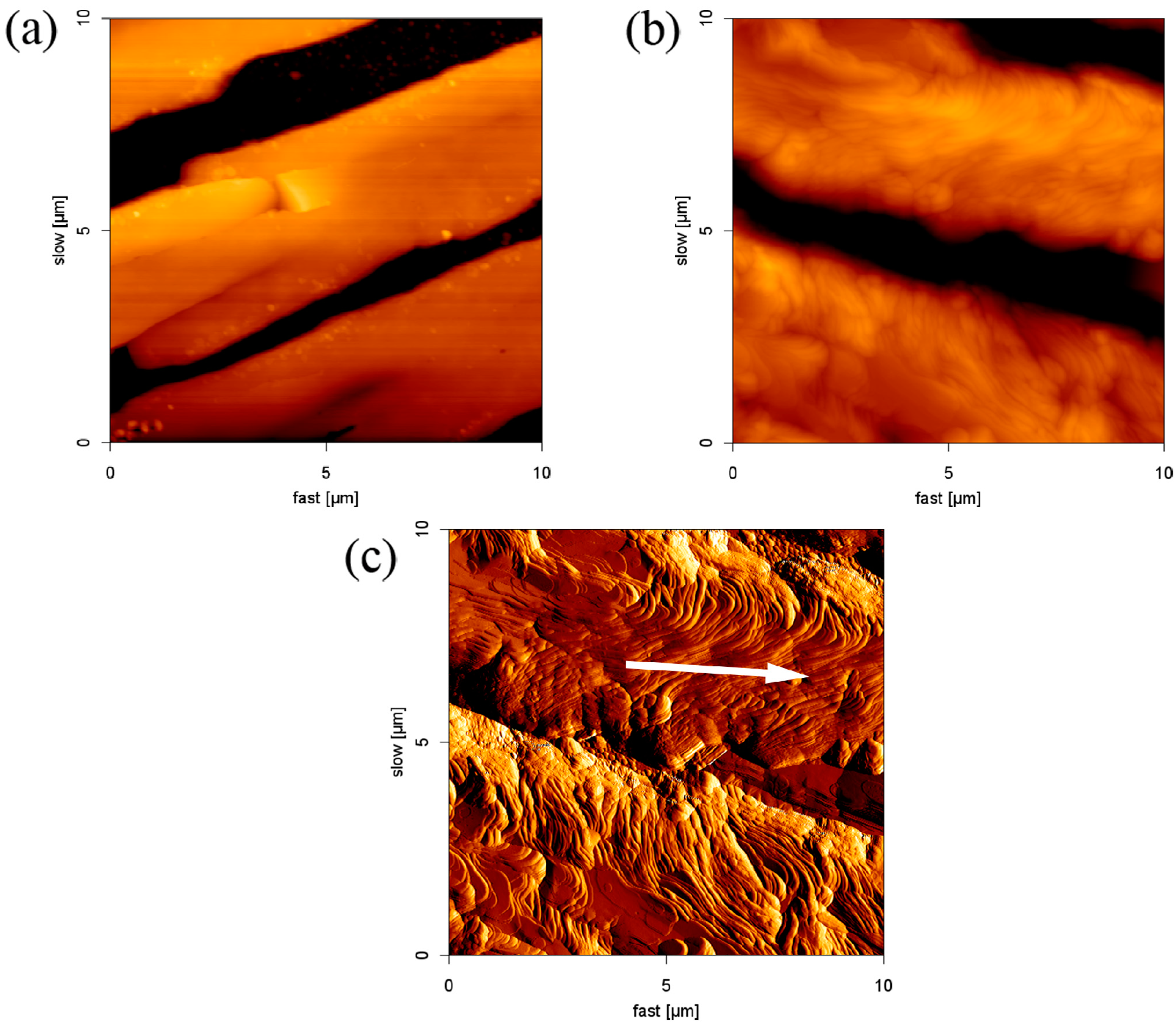
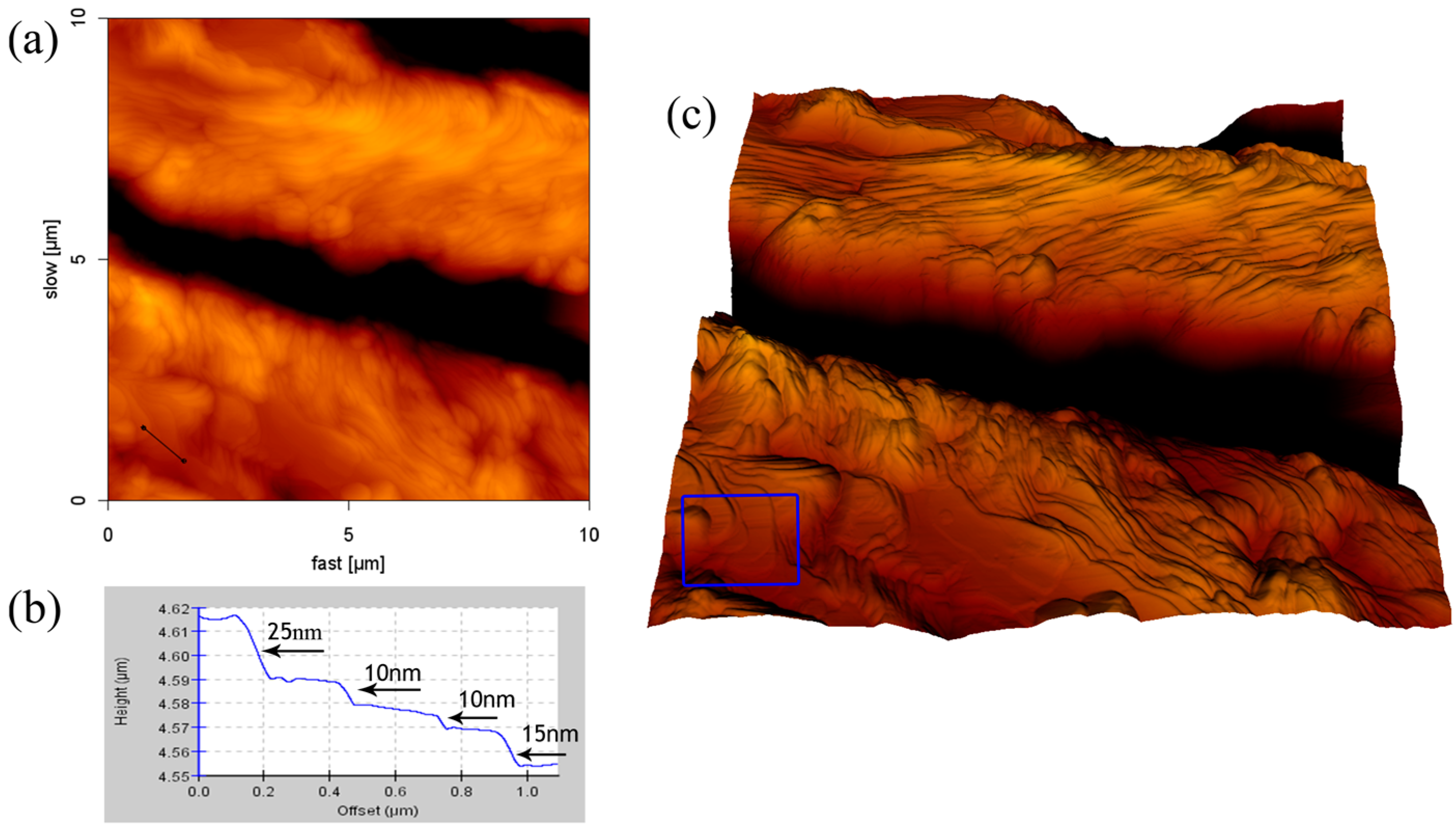
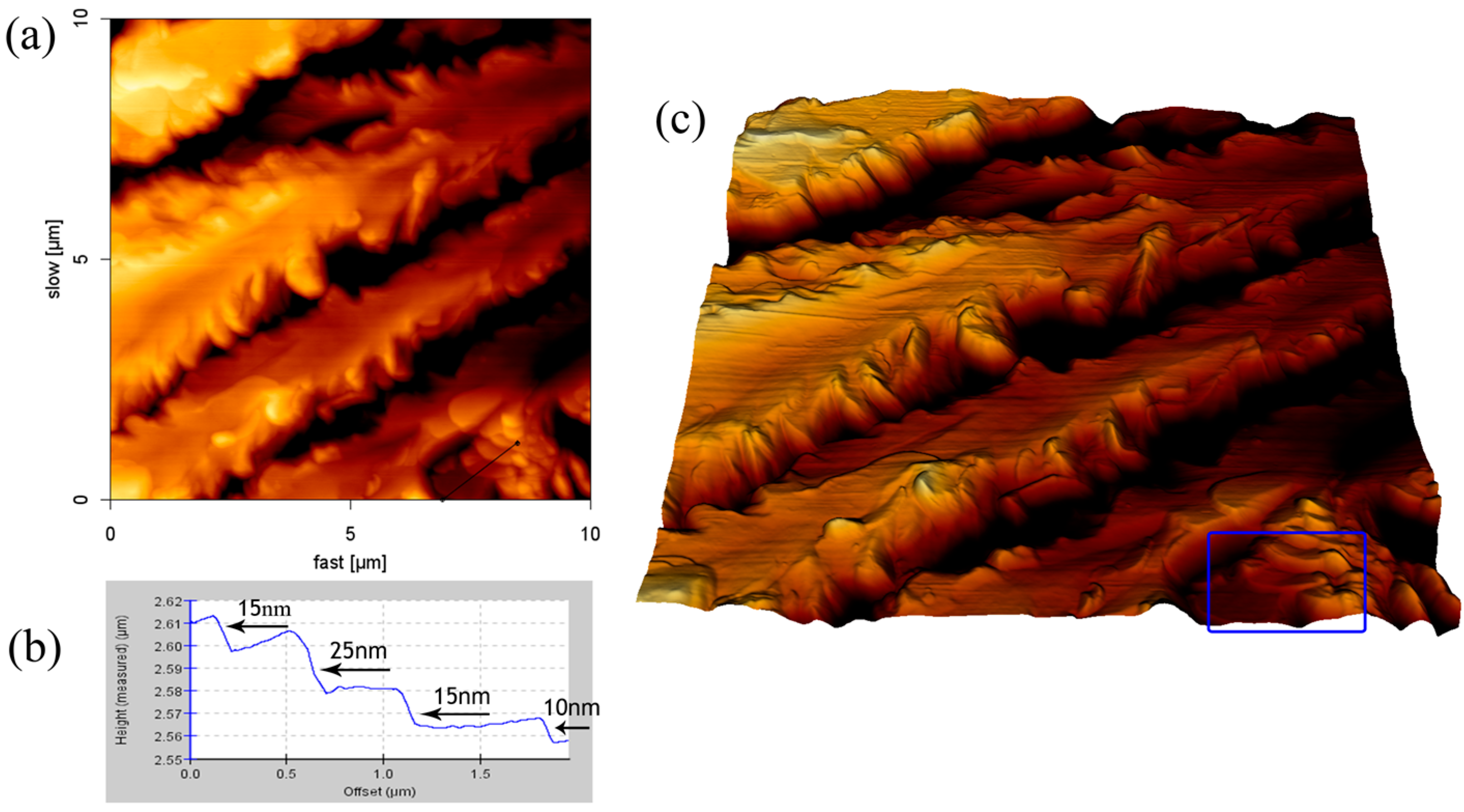
2.4. Formation of SDS Multilayers
3. Discussion
4. Materials and Methods
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Atkin, R.; Craig, V.; Wanless, E.J.; Biggs, S. Mechanism of cationic surfactant adsorption at the solid–aqueous interface. Adv. Colloid Interface Sci. 2003, 103, 219–304. [Google Scholar] [CrossRef]
- Paria, S.; Khilar, K.C. A review on experimental studies of surfactant adsorption at the hydrophilic solid–water interface. Adv. Colloid Interface Sci. 2004, 110, 75–95. [Google Scholar] [CrossRef] [PubMed]
- Lal, S.; Gautam, D.; Razeeb, K.M. The impact of surfactant sodium dodecyl sulfate on the microstructure and thermoelectric properties of p-type (Sb1−xBix)2Te3 electrodeposited films. ECS J. Solid State Sci. Technol. 2017, 6, N3017–N3021. [Google Scholar] [CrossRef]
- Naylor, A.J.; Koukharenko, E.; Nandhakumar, I.S.; White, N.M. Surfactant-mediated electrodeposition of bismuth telluride films and its effect on microstructural properties. Langmuir 2012, 28, 8296–8299. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Jelinek, R. Dramatic shape modulation of surfactant/diacetylene microstructures at the air–water interface. Chem. Eur. J. 2014, 20, 16747–16752. [Google Scholar] [CrossRef] [PubMed]
- Plawsky, J.; Fedorov, A.; Garimella, S.; Ma, H.; Maroo, S.; Chen, L.; Nam, Y. Nano-and microstructures for thin-film evaporation—A review. Nanoscale Microscale Thermophys. Eng. 2014, 18, 251–269. [Google Scholar] [CrossRef]
- Fatona, A.; Chen, Y.; Reid, M.; Brook, M.A.; Moran-Mirabal, J.M. One-step in-mould modification of pdms surfaces and its application in the fabrication of self-driven microfluidic channels. Lab Chip 2015, 15, 4322–4330. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Yadav, N.; Chowdhury, P.K.; Ganguli, A.K. Controlling the microstructure of reverse micelles and their templating effect on shaping nanostructures. J. Phys. Chem. B 2015, 119, 11295–11306. [Google Scholar] [CrossRef] [PubMed]
- Manne, S.; Cleveland, J.; Gaub, H.; Stucky, G.; Hansma, P. Direct visualization of surfactant hemimicelles by force microscopy of the electrical double layer. Langmuir 1994, 10, 4409–4413. [Google Scholar] [CrossRef]
- Manne, S.; Gaub, H.E. Molecular organization of surfactants at solid-liquid interfaces. Science 1995, 270, 1480. [Google Scholar] [CrossRef]
- Ducker, W.A.; Grant, L.M. Effect of substrate hydrophobicity on surfactant surface−aggregate geometry. J. Phys. Chem. 1996, 100, 11507–11511. [Google Scholar] [CrossRef]
- Lamont, R.; Ducker, W. Organized structure of lithium perfluorooctanesulfonate at the graphite–solution interface. J. Colloid Interface Sci. 1997, 191, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.; Nagarajan, R. Modeling self-assembly of surfactants at solid–liquid interfaces. II. Hydrophilic surfaces. Coll. Surf. A Physicochem. Eng. Asp. 2000, 167, 21–30. [Google Scholar] [CrossRef]
- Johnson, R.A.; Nagarajan, R. Modeling self-assembly of surfactants at solid/liquid interfaces. I. Hydrophobic surfaces. Coll. Surf. A Physicochem. Eng. Asp. 2000, 167, 31–46. [Google Scholar] [CrossRef]
- Warr, G.G. Surfactant adsorbed layer structure at solid/solution interfaces: Impact and implications of AFM imaging studies. Curr. Opin. Colloid Interface Sci. 2000, 5, 88–94. [Google Scholar] [CrossRef]
- Xu, S.L.; Wang, C.; Zeng, Q.D.; Wu, P.; Wang, Z.G.; Yan, H.K.; Bai, C.L. Self-assembly of cationic surfactants on a graphite surface studied by STM. Langmuir 2002, 18, 657–660. [Google Scholar] [CrossRef]
- Ferrari, M.; Ravera, F.; Viviani, M.; Liggieri, L. Characterization of surfactant aggregates at solid–liquid surfaces by atomic force microscopy. Coll. Surf. A Physicochem. Eng. Asp. 2004, 249, 63–67. [Google Scholar] [CrossRef]
- Tulpar, A.; Ducker, W.A. Surfactant adsorption at solid–aqueous interfaces containing fixed charges: Experiments revealing the role of surface charge density and surface charge regulation. J. Phys. Chem. B 2004, 108, 1667–1676. [Google Scholar] [CrossRef]
- Domínguez, H. Self-aggregation of the sds surfactant at a solid–liquid interface. J. Phys. Chem. B 2007, 111, 4054–4059. [Google Scholar] [CrossRef] [PubMed]
- Schniepp, H.C.; Shum, H.C.; Saville, D.A.; Aksay, I.A. Surfactant aggregates at rough solid–liquid interfaces. J. Phys. Chem. B 2007, 111, 8708–8712. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Burgess, I.; Lipkowski, J. Potential controlled surface aggregation of surfactants at electrode surfaces—A molecular view. Surf. Sci. 2009, 603, 1878–1891. [Google Scholar] [CrossRef]
- Bernardes, J.S.; Rezende, C.A.; Galembeck, F. Morphology and self-arraying of SDS and DTAB dried on mica surface. Langmuir 2010, 26, 7824–7832. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Chorover, J. Adsorption of sodium dodecyl sulfate (SDS) at ZnSe and α-Fe2O3 surfaces: Combining infrared spectroscopy and batch uptake studies. J. Colloid Interface Sci. 2010, 348, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, P.M.; Anderson, M.W.; Palmqvist, A.E. Adsorption of sodium dodecyl sulfate and sodium dodecyl phosphate at the surface of aluminium oxide studied with AFM. Corros. Sci. 2010, 52, 1103–1105. [Google Scholar] [CrossRef]
- Seiedi, O.; Rahbar, M.; Nabipour, M.; Emadi, M.A.; Ghatee, M.H.; Ayatollahi, S. Atomic force microscopy (afm) investigation on the surfactant wettability alteration mechanism of aged mica mineral surfaces. Energy Fuels 2010, 25, 183–188. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Y.E.; Fan, Q.; Zuo, Y.Y. On the low surface tension of lung surfactant. Langmuir 2011, 27, 8351. [Google Scholar] [CrossRef] [PubMed]
- Song, S.H.; Koelsch, P.; Weidner, T.; Wagner, M.S.; Castner, D.G. Sodium dodecyl sulfate adsorption onto positively charged surfaces: Monolayer formation with opposing headgroup orientations. Langmuir 2013, 29. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.F.; Ducker, W.A. Surface-induced phase behavior of alkyltrimethylammonium bromide surfactants adsorbed to mica, silica, and graphite. J. Phys. Chem. B 1999, 103, 8558–8567. [Google Scholar] [CrossRef]
- Ducker, W.A.; Wanless, E.J. Adsorption of hexadecyltrimethylammonium bromide to mica: Nanometer-scale study of binding-site competition effects. Langmuir 1999, 15, 160–168. [Google Scholar] [CrossRef]
- Lamont, R.E.; Ducker, W.A. Surface-induced transformations for surfactant aggregates. J. Am. Chem. Soc. 1998, 120, 7602–7607. [Google Scholar] [CrossRef]
- Wanless, E.J.; Ducker, W.A. Organization of sodium dodecyl sulfate at the graphite−solution interface. J. Phys. Chem. 1996, 100, 3207–3214. [Google Scholar] [CrossRef]
- Domínguez, H. Structure of the sodium dodecyl sulfate surfactant on a solid surface in different nacl solutions. Langmuir 2009, 25, 9006–9011. [Google Scholar] [CrossRef] [PubMed]
- Wanless, E.J.; Ducker, W.A. Weak influence of divalent ions on anionic surfactant surface-aggregation. Langmuir 1997, 13, 1463–1474. [Google Scholar] [CrossRef]
- Deegan, R.D.; Bakajin, O.; Dupont, T.F.; Huber, G.; Nagel, S.R.; Witten, T.A. Capillary flow as the cause of ring stains from dried liquid drops. Nature 1997, 389, 827–829. [Google Scholar] [CrossRef]
- Goodwin, J. Colloids and Interfaces with Surfactants and Polymers: An Introduction, 1st ed.; John Wiley & Sons Ltd: West Sussex, UK, 2004. [Google Scholar]
- Cosgrove, T. Colloid Science: Principles, Methods and Applications, 1st ed.; Wiley-Blackwell: Oxford, UK, 2005. [Google Scholar]
- Edward, J.T. Molecular volumes and the stokes-einstein equation. J. Chem. Educ. 1970, 47, 261. [Google Scholar] [CrossRef]
- Coiro, V.; Manigrasso, M.; Mazza, F.; Pochetti, G. Structure of a triclinic phase of sodium dodecyl sulfate monohydrate. A comparison with other sodium dodecyl sulfate crystal phases. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 1987, 43, 850–854. [Google Scholar] [CrossRef]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, Z.-Y.; Lee, M.-T. On the Morphology of the SDS Film on the Surface of Borosilicate Glass. Materials 2017, 10, 555. https://doi.org/10.3390/ma10050555
Shen Z-Y, Lee M-T. On the Morphology of the SDS Film on the Surface of Borosilicate Glass. Materials. 2017; 10(5):555. https://doi.org/10.3390/ma10050555
Chicago/Turabian StyleShen, Zih-Yao, and Maw-Tien Lee. 2017. "On the Morphology of the SDS Film on the Surface of Borosilicate Glass" Materials 10, no. 5: 555. https://doi.org/10.3390/ma10050555
APA StyleShen, Z.-Y., & Lee, M.-T. (2017). On the Morphology of the SDS Film on the Surface of Borosilicate Glass. Materials, 10(5), 555. https://doi.org/10.3390/ma10050555





