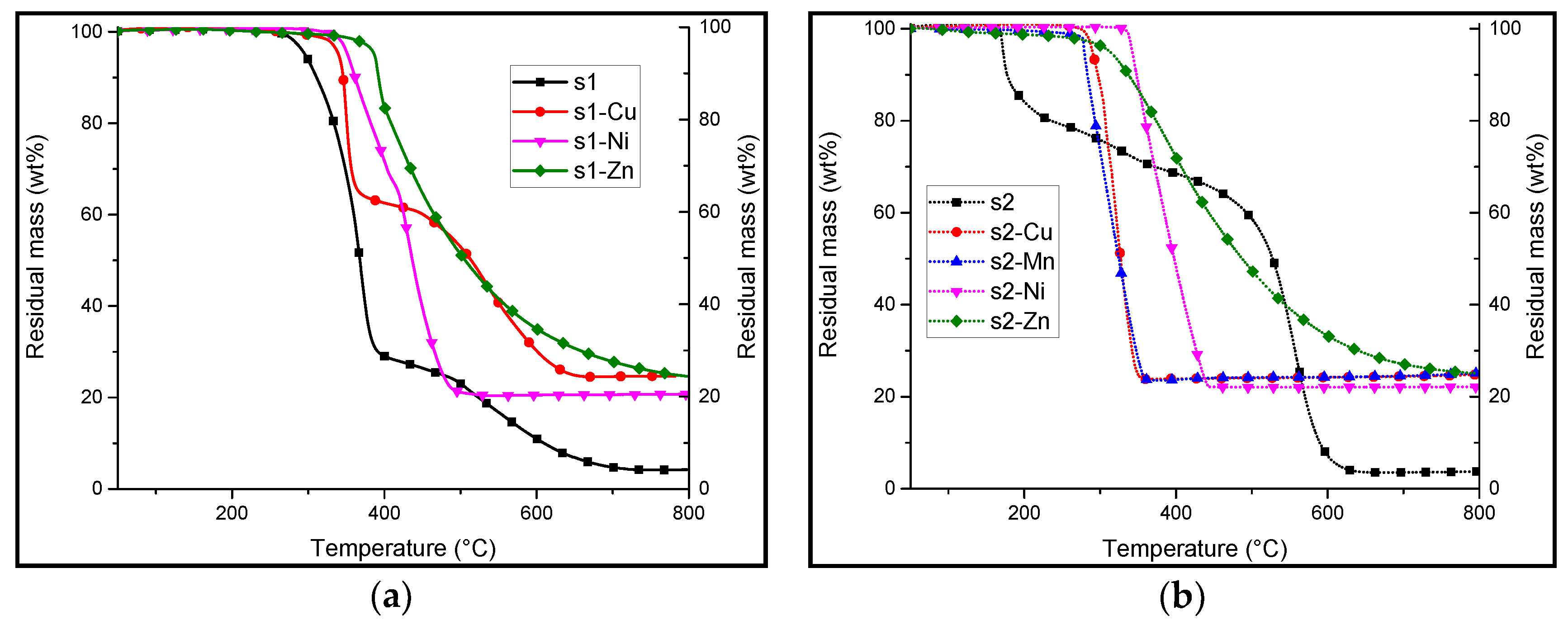
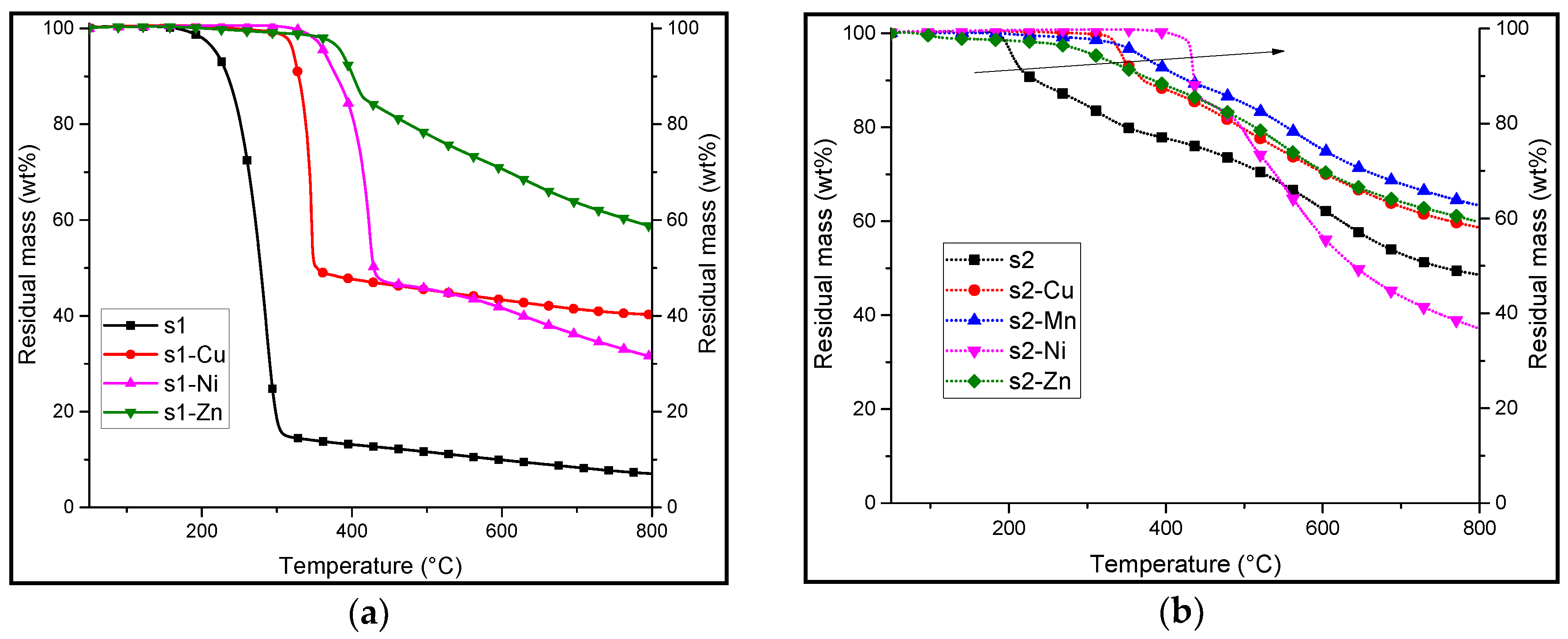
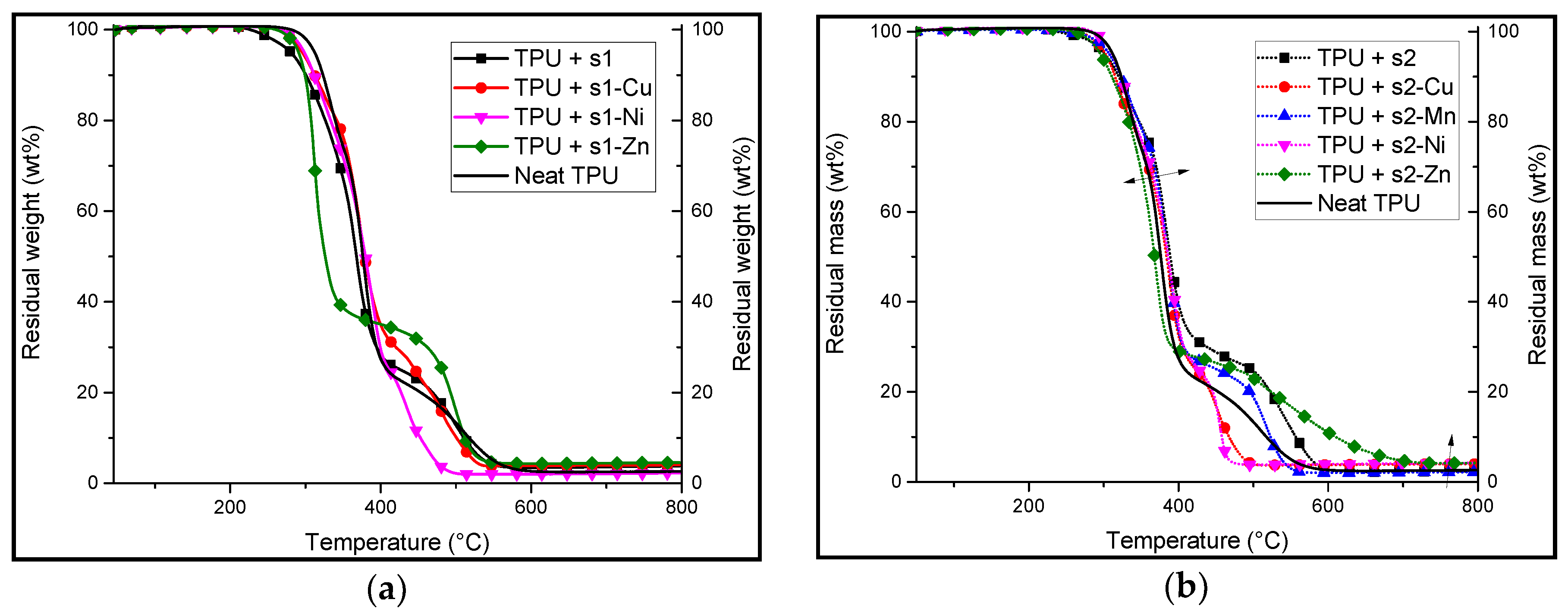
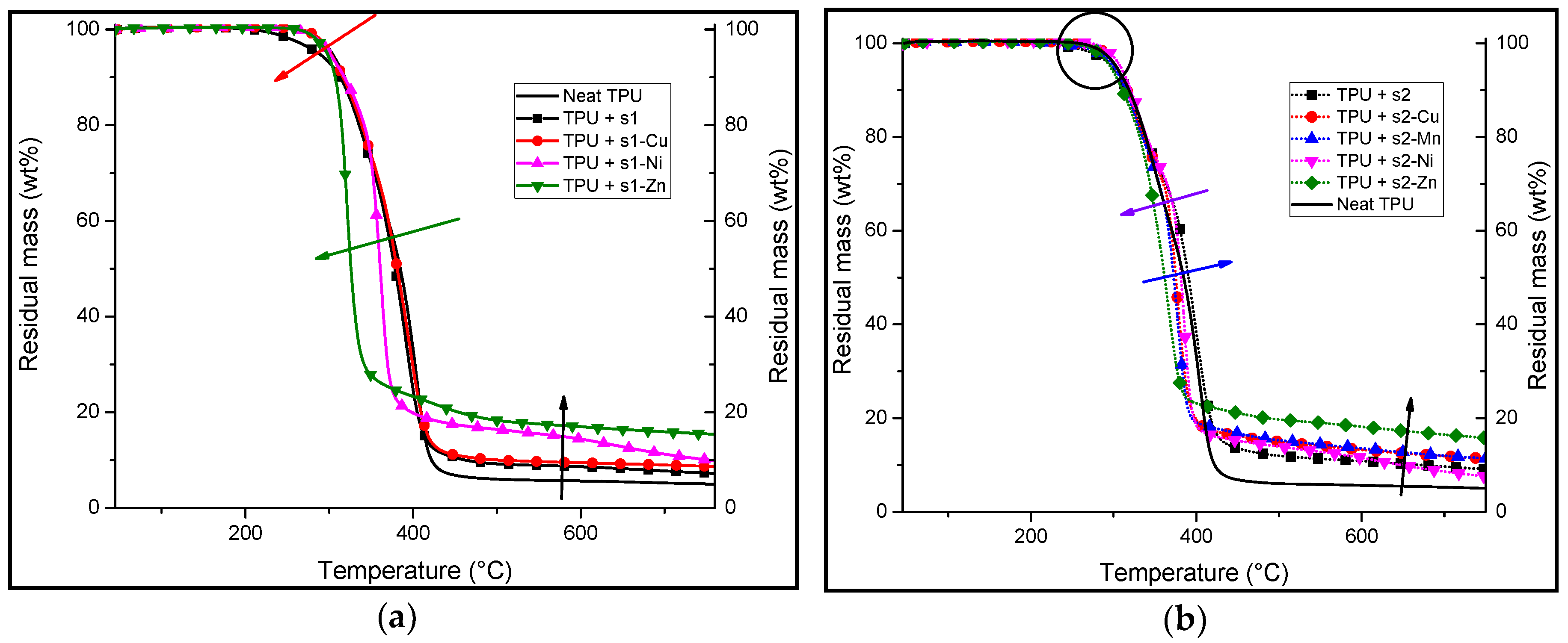
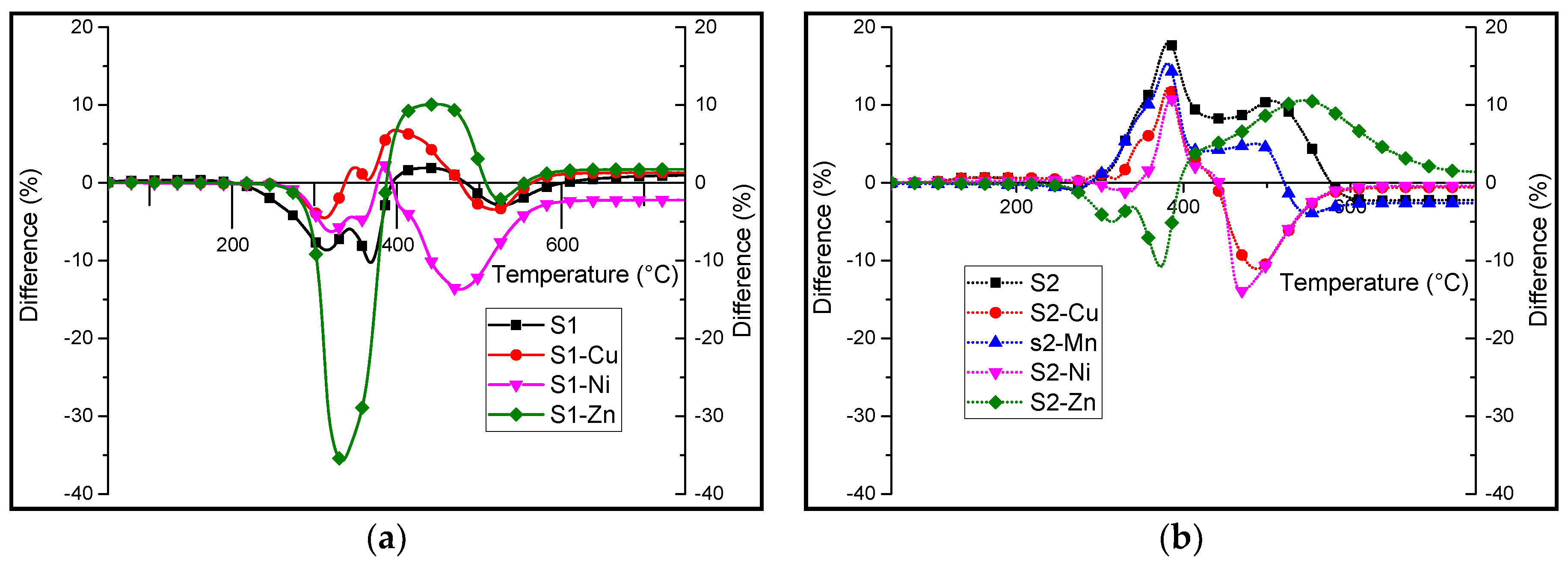
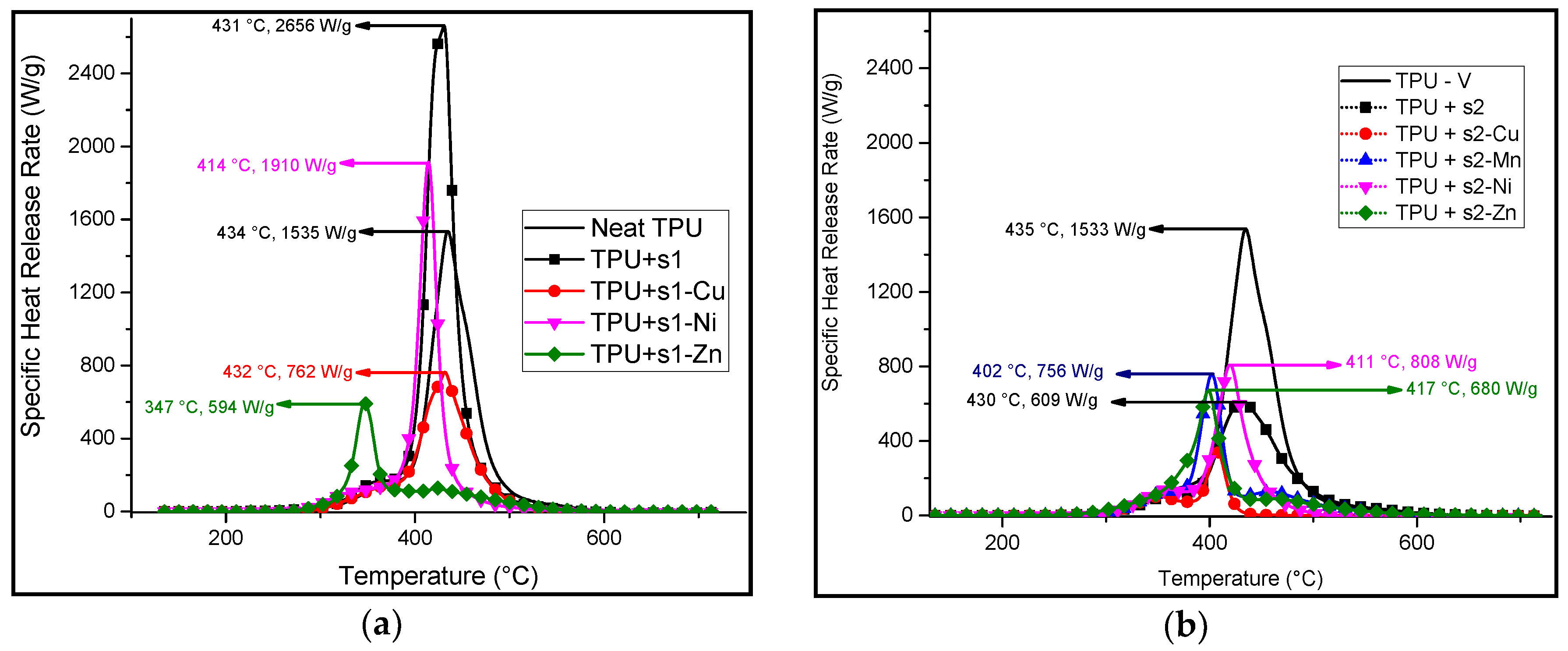
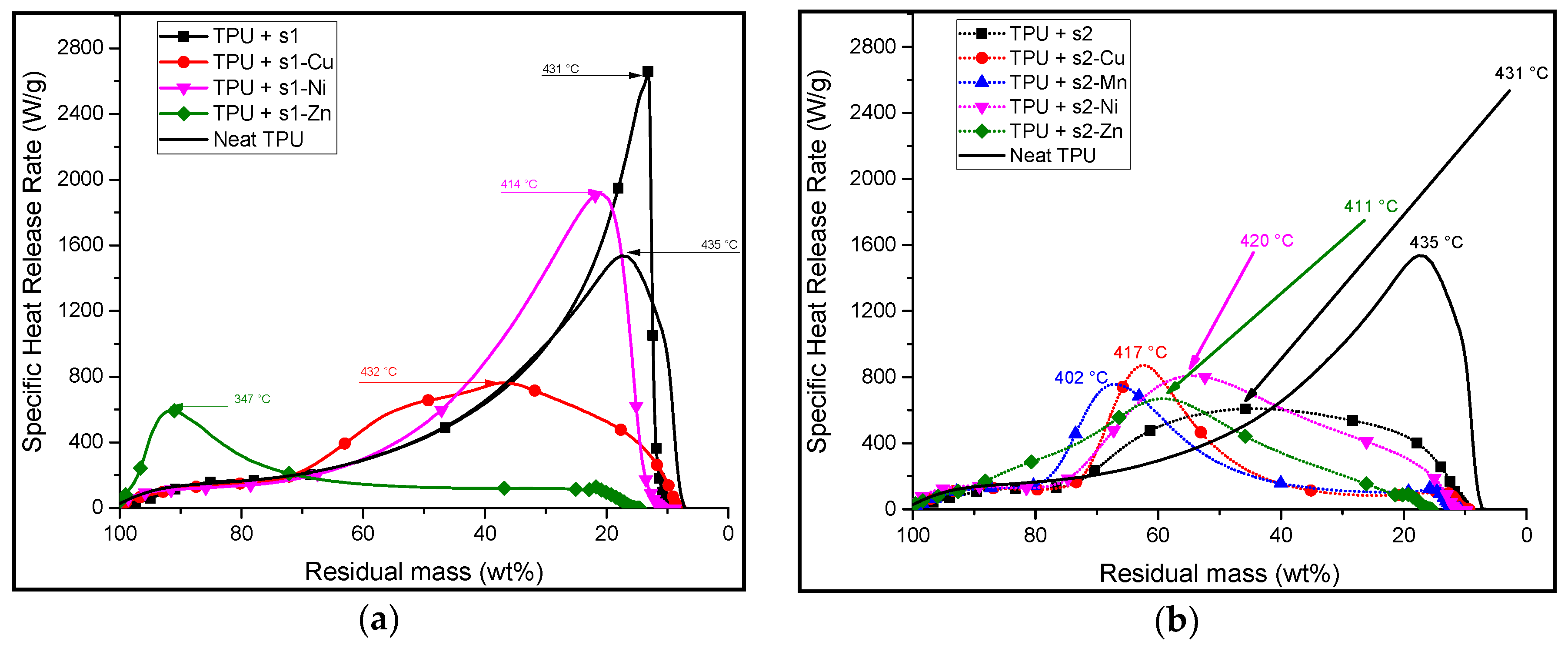
3.2. Synthesis and Characterisation
Salen and hydroxysalens
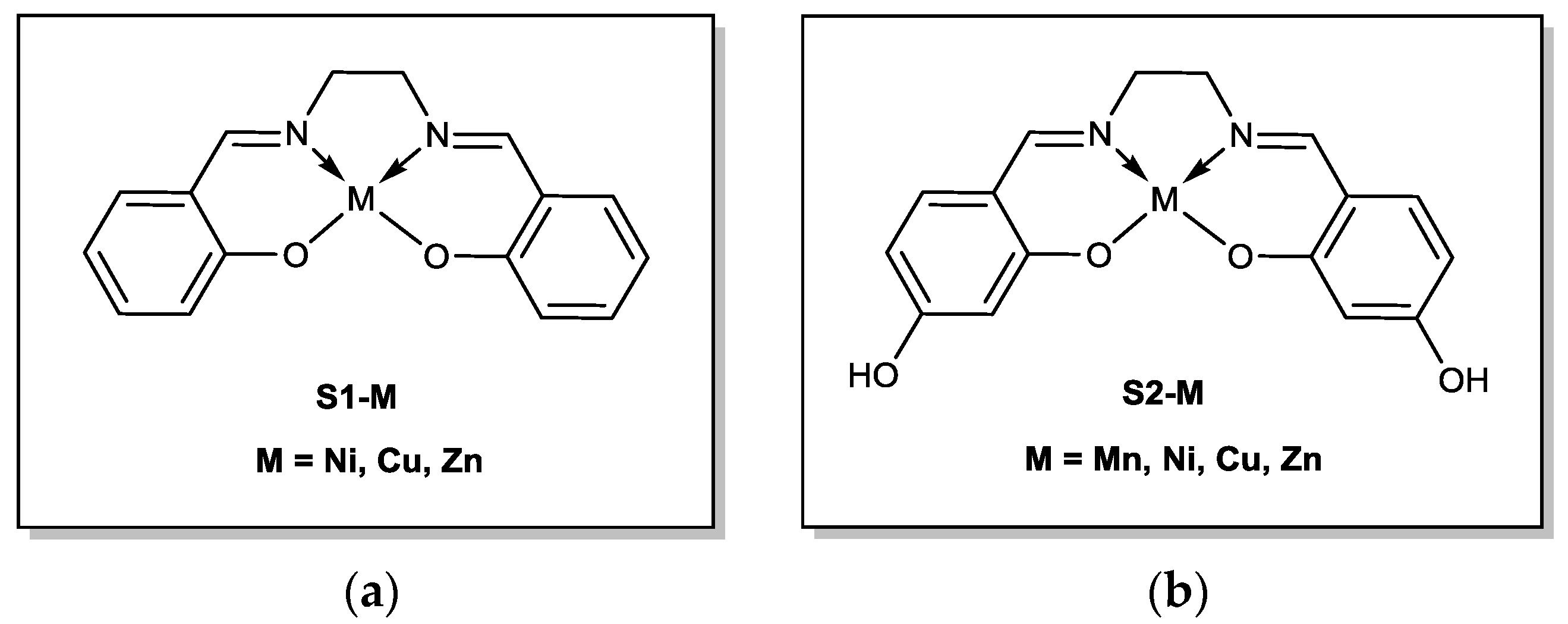
s1 and
s2 were obtained using a conventional procedure for salen synthesis based on the condensation of salicylaldehyde or its hydroxy-derivatives with diamine [
6] Most salen complexes were synthesized according to procedure described in [
5]. Zinc–salen complexes were prepared following the procedure described in [
8].
Reagents were purchased from Aldrich (Sigma-Aldrich Chimie S.a.r.l., Lyon, France) or Lancaster Synthesis (Alfa Aesar, Haverhill, MA, USA) and were used without further purification. Diethyl ether, methanol and absolute ethanol were reagent grade commercial solvents and were used without further purification.
NMR spectra were recorded on a Bruker Avance 300 spectrometer (Billerica, MA, USA). Chemical shifts (δ) are referenced to internal solvent and given in ppm. Coupling constants (J) are given in Hz. The following abbreviations apply to spin multiplicity: s (singlet), d (doublet), t (triplet), q (quartet), m (multiplet) and bs (broad singlet).
3.2.1. N,N′-Bis(salicylidene)ethylenediamine (s1)
To a solution of ethylenediamine (5.34 mL; 4.8 g; 0.08 mol) in 60 mL of absolute ethanol, salicylaldehyde (16.7 mL; 19.5 g; 0.16 mol) in 80 mL of ethanol was added dropwise with vigorous stirring. The product precipitated immediately and the mixture was refluxed for two hours, and then kept at room temperature and filtered. The product recrystallized from EtOH was filtered, washed with cold ethanol and ethyl ether, and then dried at 80 °C to give the desired Schiff base s1 as yellow crystals, 20.2 g (94%). 1H-NMR (DMSO-d6) δ 3.92 (s, 4 H), 6.81–6.95 (m, 4 H), 7.35 (m, 4 H), 8.57 (s, 2 H); 13C-NMR (DMSO-d6) δ 58.8 (CH2), 116.4 (CAr), 118.5 (CAr), 118.6 (CAr), 131.6 (CAr), 132.3 (CAr), 160.4 (CAr), 166.8 (CN). MS (NanoESI) m/z 269.1 (M + H)+. Mp = 130 °C.
3.2.2. N,N′-Bis(4-hydroxysalicylidene)ethylenediamine (s2)
According to the method described for s1, 21.9 g (0.16 mol) of 2,4-dihydroxybenzaldehyde and 5.34 mL (4.8 g, 0.08 mol) of ethylenediamine yielded compound s2 (28.1 g; 94%) orange solid; 1H-NMR (DMSO-d6) δ 3.77 (s, 4 H), 6.15 (d, J = 2.4 Hz, 2 H), 6.26 (dd, J = 2.4, 8.6 Hz, 2 H), 7.16 (d, J = 8.6 Hz, 2 H), 8.35 (s, 2 H); 13C-NMR (DMSO-d6) δ 57.8 (CH2), 102.5 (CAr), 106.9 (CAr), 111.2 (CAr), 133.4 (CAr), 161.7 (CAr), 164.3 (CAr), 165.7 (CN). MS (NanoESI) m/z 301.2 (M + H)+. Mp > 190 °C (decomposition).
3.2.3. N,N′-Bis(salicylidene)ethylenediamine Copper (II) Complex, s1-Cu
N,N′-bis(salicylidene)ethylenediamine s1 (8.1 g, 0.03 mol) was dissolved in 70 mL absolute ethanol; then, a solution of copper (II) acetate (6.0 g, 0.03 mol) in 40 mL of water was added dropwise and the mixture was refluxed under vigorous stirring for 2.5 h. The green precipitate was collected by filtration, washed thoroughly with ethanol and then dried at 80 °C to give N,N′-bis(salicylidene)ethylenediamine copper (II) complex s1-Cu (6.96 g, 70%) as a fine green powder. NMR spectra of the complex was not recorded due to paramagnetism of Cu(II) (this well-known phenomenon leads to poor resolution of the spectra). MS (NanoESI) m/z 330.0 (M + H)+, 659.1 (dimer + H)+. Mp > 260 °C.
3.2.4. N,N′-Bis(salicylidene)ethylenediamine Nickel (II) Complex, s1-Ni
N,N′-bis(salicylidene)ethylenediamine s1 (6.01 g, 0.022 mol) was dissolved in 70 mL absolute ethanol, then a solution of nickel (II) acetate (5.51 g, 0.022 mol) in 40 mL of water was added dropwise and the mixture was refluxed under vigorous stirring for 2.5 h. The orange precipitate was collected by filtration, washed thoroughly with ethanol, and then dried at 80 °C to give N,N′-bis(salicylidene)ethylenediamine Nickel (II) complex s1-Ni (5.87 g, 80.6%) as an orange powder. 1H-NMR (300 MHz, DMSO-d6) δ ppm 3.29–3.35 (m, 1 H) 3.42 (s, 4 H) 6.51 (t, J = 7.25 Hz, 2 H) 6.70 (d, J = 8.67 Hz, 2 H) 7.17 (ddd, J = 8.43, 6.92, 1.79 Hz, 2 H) 7.25 (dd, J = 7.82, 1.60 Hz, 2 H) 7.89 (s, 2 H). 13C-NMR (75 MHz, DMSO-d6) δ ppm 58.45 (s, 1 C) 114.75 (s, 1 C) 120.22 (s, 1 C) 120.79 (s, 1 C) 133.22 (s, 1 C) 133.98 (s, 1 C) 163.07 (s, 1 C) 164.40 (s, 1 C). MS (NanoESI) m/z 324.0 M+. Mp > 260 °C.
3.2.5. N,N′-Bis(salicylidene)ethylenediamine Zinc (II) Complex, s1-Zn
The complex s1-Zn was prepared by first treating salicylaldehyde (2.15 mL, 0.02 mol) with zinc (II) acetate (2.26 g, 0.01 mol) in methanol (100 mL) for 30 min at room temperature. Then, ethylenediamine (0.68 mL, 0.01 mol) was added to the solution, and stirring was maintained at room temperature overnight. During that time, a light yellow precipitate formed. The precipitate was collected by filtration, washed with cold methanol and ether, and dried at 80 °C, affording 3.00 g, 80%. 1H-NMR (300 MHz, DMSO-d6) δ ppm 3.72 (s, 4 H) 3.92 (s, 1 H) 6.34–6.50 (m, 2 H) 6.55–6.68 (m, 1 H) 6.61 (d, J = 8.29 Hz, 1 H) 7.09–7.44 (m, 4 H) 8.43 (s, 2 H) 13.34–13.45 (m, 1 H). 13C-NMR (75 MHz, DMSO-d6) δ ppm 55.75 (s, 1 C) 112.15 (s, 1 C) 119.31 (s, 1 C) 122.68 (s, 1 C) 132.77 (s, 1 C) 134.73 (s, 1 C) 167.97 (s, 1 C) 171.01 (s, 1 C). MS (NanoESI) m/z 331.0 (M + H)+. Mp > 260 °C.
3.2.6. N,N′-Bis(4-hydroxysalicylidene)ethylenediamine Copper (II) Complex, s2-Cu
N,N′-bis(4-hydroxysalicylidene)ethylenediamine s2 (9.0 g, 0.03 mol) was dissolved in 70 mL absolute ethanol, then a solution of copper (II) acetate (6.0 g, 0.03 mol) in 40 mL of water was added dropwise and the mixture was refluxed under vigorous stirring for 2.5 h. The purple precipitate was collected by filtration, washed thoroughly with ethanol, and then dried at 80 °C to give N,N′-bis(4-hydroxysalicylidene)ethylenediamine copper (II) complex s1-Cu (9.7 g, 89%) as a purple powder. NMR spectra of the complex was not recorded due to paramagnetism of Cu(II) (this well-known phenomenon leads to poor resolution of the spectra). MS (NanoESI) m/z 362.0 (M + H)+. Mp > 260 °C.
3.2.7. N,N′-Bis(4-hydroxysalicylidene)ethylenediamine Manganese (II) Complex, s2-Mn
N,N′-bis(4-hydroxysalicylidene)ethylenediamine s2 (3.60 g, 0.012 mol) was dissolved in 70 mL absolute ethanol, then a solution of manganese (II) acetate (2.94 g, 0.012 mol) in 40 mL of water was added dropwise and the mixture was refluxed under vigorous stirring for 2.5 h. The brown precipitate was collected by filtration, washed thoroughly with ethanol, and then dried at 80 °C to give N,N′-bis(4-hydroxysalicylidene)ethylenediamine manganese (II) complex s1-Mn (3.1 g, 73%) as a brown solid. NMR spectra of the complex was not recorded due to paramagnetism of Mn(II) (this well-known phenomenon leads to poor resolution of the spectra). MS (NanoESI) m/z 353.0 (M)+. Mp > 260 °C.
3.2.8. N,N′-Bis(4-hydroxysalicylidene)ethylenediamine Manganese (II) Complex, s2-Ni
N,N′-bis(4-hydroxysalicylidene)ethylenediamine s2 (7.00 g, 0.022 mol) was dissolved in 50 mL absolute ethanol, then a solution of nickel (II) acetate (5.51 g, 0.022 mol) in 50 mL of ethanol was added dropwise, and the mixture was refluxed under vigorous stirring for 2. The red precipitate was collected by filtration, washed thoroughly with ethanol, and then dried at 80 °C to give N,N′-bis(4-hydroxysalicylidene)ethylenediamine cobalt (II) complex s2-Ni (5.36 g, 89%) as a red powder. 1H-NMR (300 MHz, DMSO-d6) δ ppm 3.25–3.31 (m, 2 H) 6.04 (d, J = 8.69 Hz, 2 H) 6.05 (s, 1 H) 7.05 (d, J = 8.10 Hz, 1 H) 7.62 (s, 1 H) 9.73 (br s, 1 H). 13C-NMR (75 MHz, DMSO-d6) δ ppm 58 (s, 1 C) 104 (s, 1 C) 106 (s, 1 C) 115 (s, 1 C) 135 (s, 1 C) 161 (s, 1 C) 163 (s, 1 C) 166 (s, 1 C). MS (NanoESI) m/z 356.0 (M)+. Mp > 260 °C.
3.2.9. N,N′-bis(4-hydroxysalicylidene)ethylenediamine Manganese (II) Complex, s2-Zn
The complex s2-Zn was prepared by first treating 4-hydroxysalicylaldehyde (2.82 g, 0.020 mol) with Zn(OAc)2·2H2O (2.26 g, 0.010 mol) in methanol (70 mL) for 30 min at room temperature. Then, ethylenediamine (0.68 mL, 0.01 mol) in 35 mL of methanol was added to the solution, and stirring was maintained at room temperature overnight. During that time, a yellow precipitate formed. The precipitate was collected by filtration, washed with cold methanol and ether, and dried under vacuum, affording 3.23 g, 87%. 1H-NMR (300 MHz, DMSO-d6) δ ppm 3.59 (s, 2 H) 5.89–5.96 (m, 1 H) 5.90–5.95 (m, 1 H) 5.95 (s, 1 H) 5.98 (s, 1 H) 6.92 (d, J = 8.48 Hz, 1 H) 8.21 (s, 1 H) 9.39 (br s, 1 H). 13C-NMR (75 MHz, DMSO-d6) δ ppm 56 (s, 2 C) 103 (1 C) 107 (1 C) 113 (1 C) 136 (1 C) 162 (1 C) 167 (1 C) 173 (1 C). MS (NanoESI) m/z 363.1 (M + H)+. Mp > 260 °C.