Cytotoxicity of Light-Cured Dental Materials according to Different Sample Preparation Methods
Abstract
:1. Introduction
2. Materials and Methods
2.1. Test Cells
2.2. Test Materials and Sample Preparations
2.3. Degree of Conversion (DC)
2.4. Eluent Preparation
2.5. Agar Diffusion Test
2.6. MTT Assay
2.7. Cytotoxicity Evaluation
2.8. Statistics
3. Results
3.1. Degree of Conversion (DC)
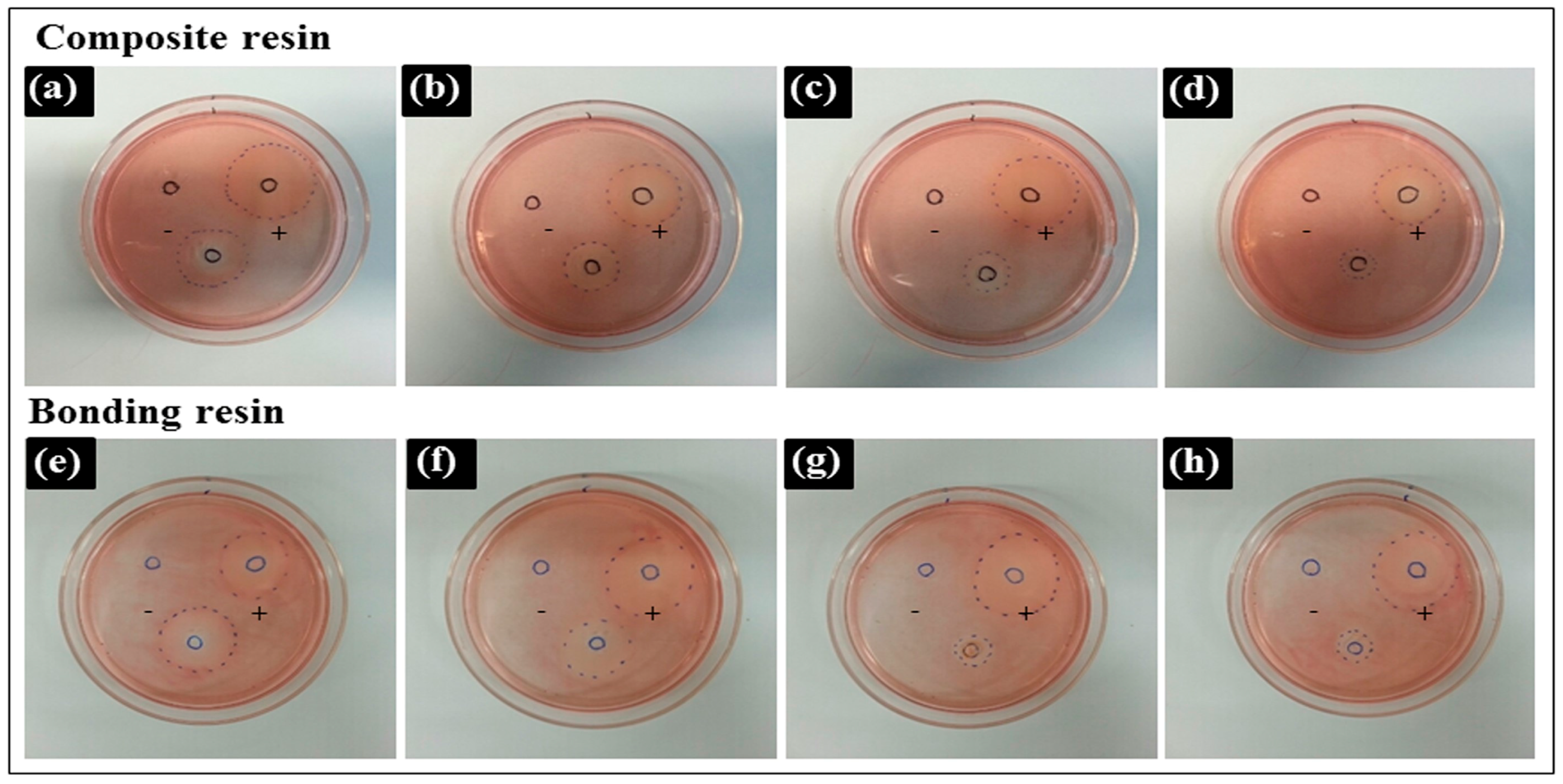
3.2. Agar Diffusion Test
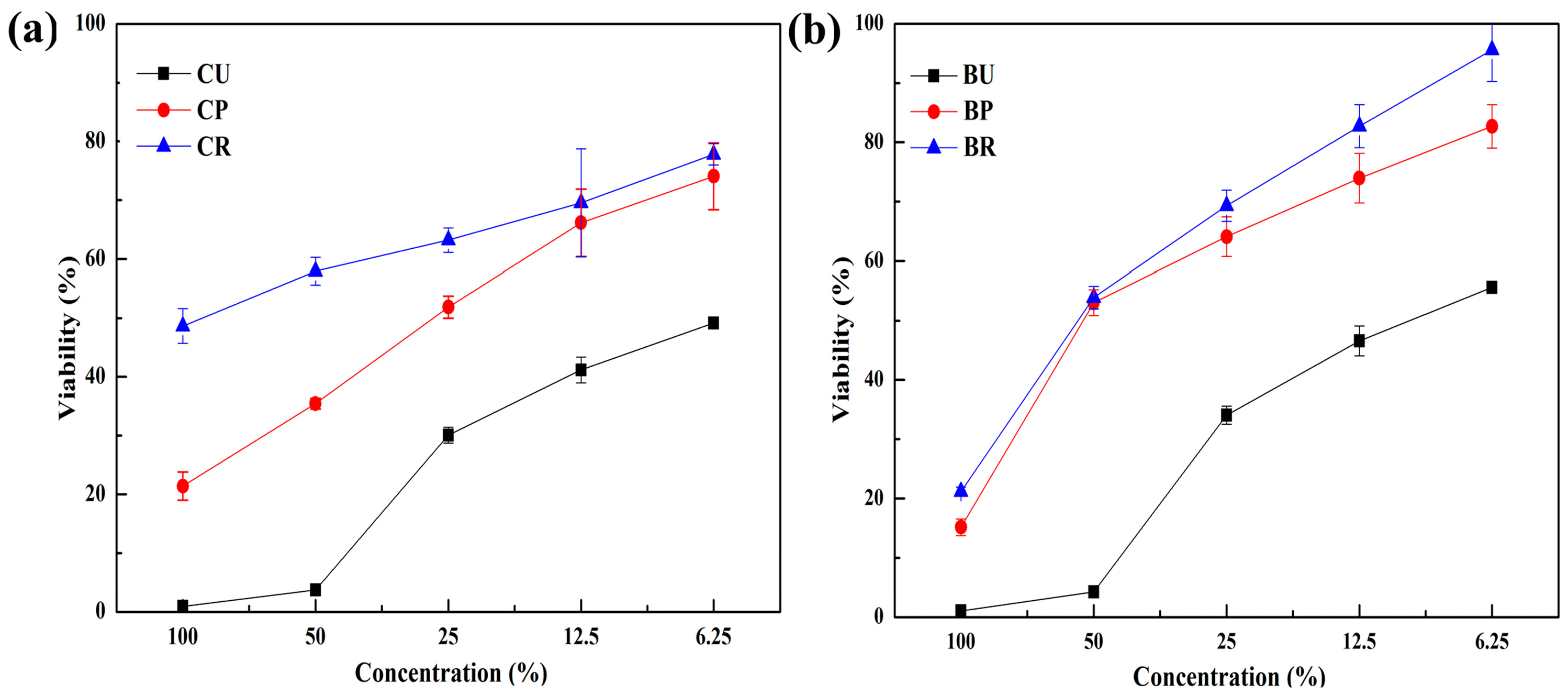
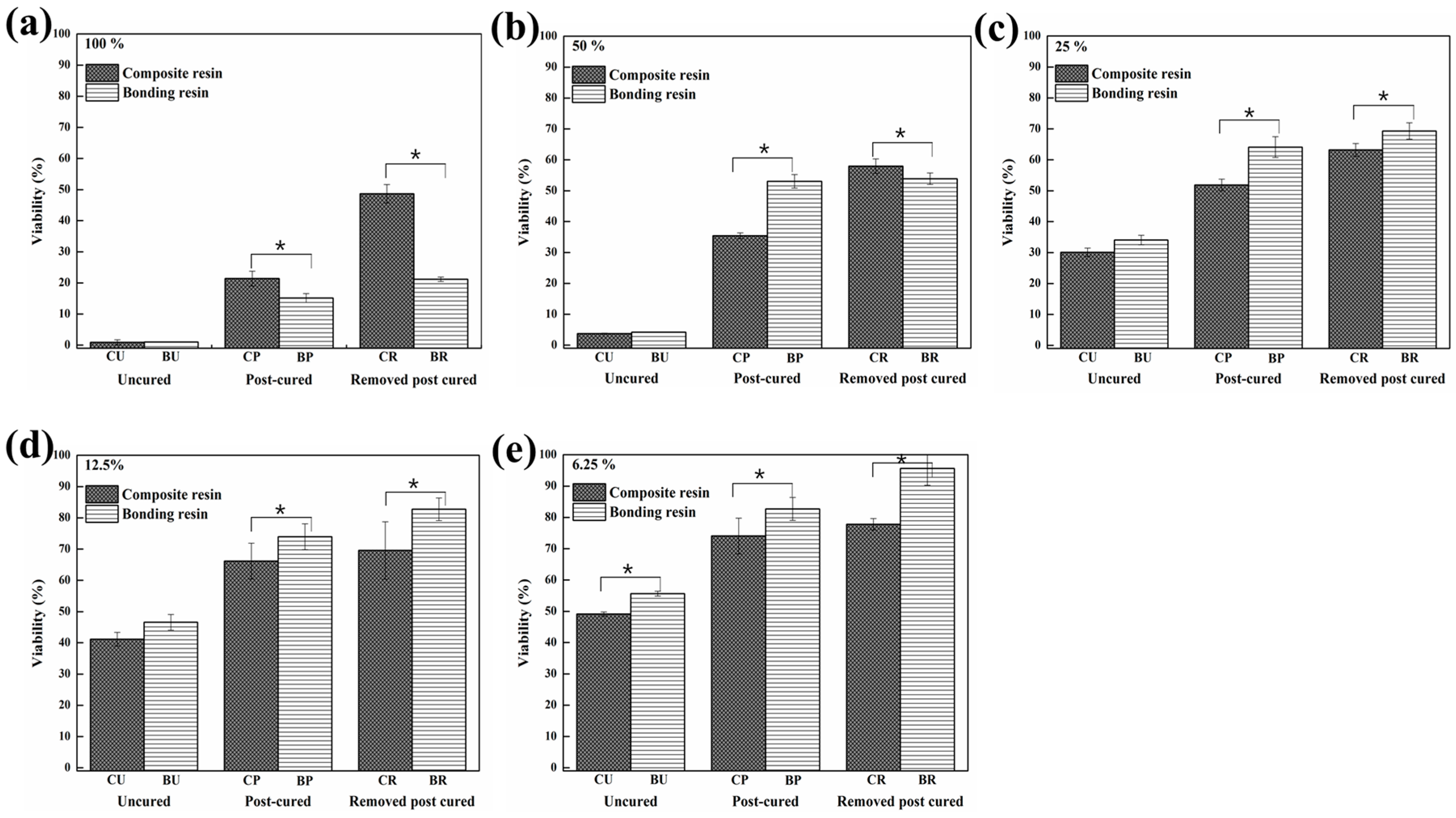
3.3. MTT Assay
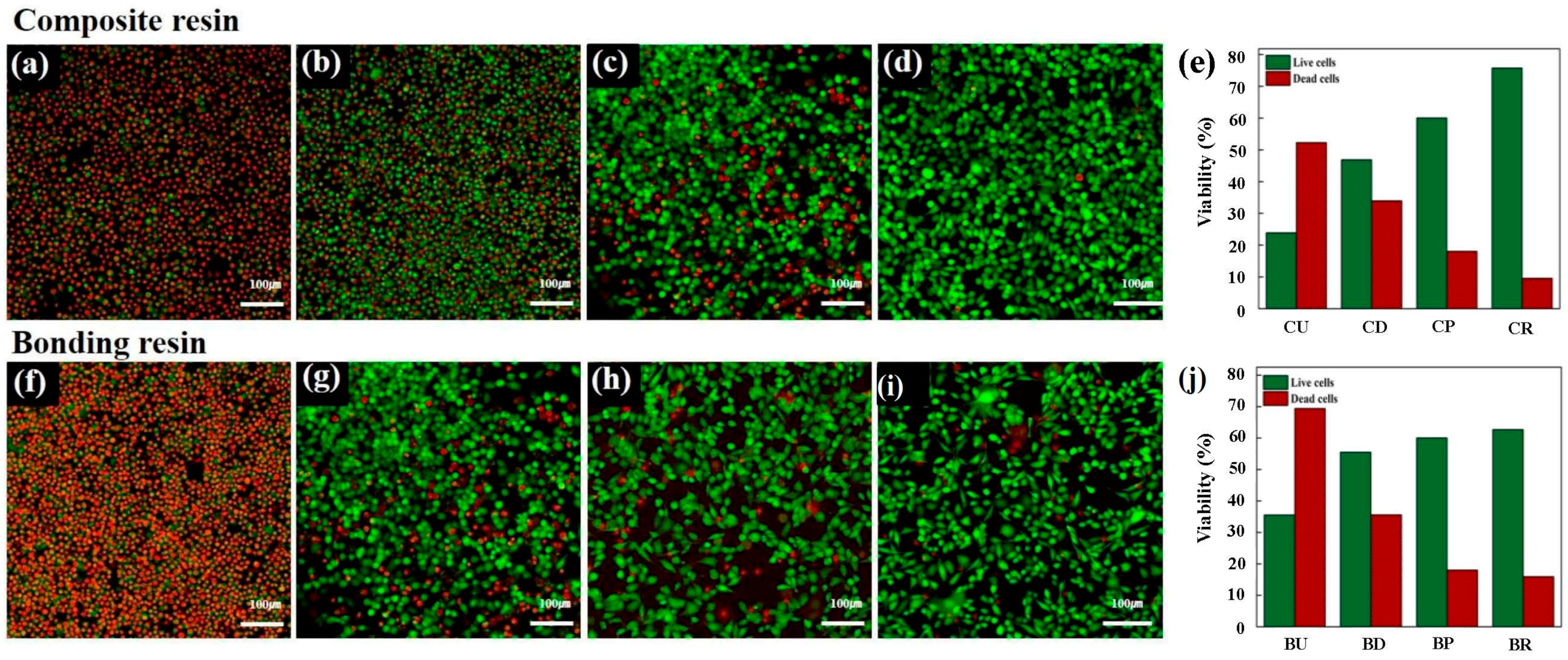
3.4. Live/Dead Image Assay®
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xia, Y.; Zhang, F.; Xie, H.; Gu, N. Nanoparticle-reinforced resin-based dental composites. J. Dent. 2008, 36, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Jandt, K.D.; Sigusch, B.W. Future perspectives of resin-based dental materials. Dent. Mater. 2009, 25, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Chai, Z.; Li, F.; Fang, M.; Wang, Y.; Ma, S.; Xiao, Y.; Huang, L.; Chen, J. The bonding property and cytotoxicity of a dental adhesive incorporating a new antibacterial monomer. J. Oral Rehabil. 2011, 38, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Sigusch, B.W.; Pflaum, T.; Volpel, A.; Schinkel, M.; Jandt, K.D. The influence of various light curing units on the cytotoxicity of dental adhesives. Dent. Mater. 2009, 25, 1446–1452. [Google Scholar] [CrossRef] [PubMed]
- Sigusch, B.W.; Volpel, A.; Braun, I.; Uhl, A.; Jandt, K.D. Influence of different light curing units on the cytotoxicity of various dental composites. Dent. Mater. 2007, 23, 1342–1348. [Google Scholar] [CrossRef] [PubMed]
- Moharamzadeh, K.; Van Noort, R.; Brook, I.M.; Scutt, A.M. Cytotoxicity of resin monomers on human gingival fibroblasts and HaCaT keratinocytes. Dent. Mater. 2007, 23, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Ferracane, J.L. Elution of leachable components from composites. J. Oral Rehabil. 1994, 21, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.M.; Chang, Y.C. Cytotoxicity of resin-based restorative materials on human pulp cell cultures. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2002, 94, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Geurtsen, W.; Lehmann, F.; Spahl, W.; Leyhausen, G. Cytotoxicity of 35 dental resin composite monomers/additives in permanent 3T3 and three human primary fibroblast cultures. J. Biomed. Mater. Res. 1998, 41, 474–480. [Google Scholar] [CrossRef]
- Wataha, J.C.; Lockwood, P.E.; Bouillaguet, S.; Noda, M. In vitro biological response to core and flowable dental restorative materials. Dent. Mater. 2003, 19, 25–31. [Google Scholar] [CrossRef]
- Schmalz, G. The biocompatibility of non-amalgam dental filling materials. Eur. J. Oral Sci. 1998, 106, 696–706. [Google Scholar] [CrossRef] [PubMed]
- Hume, W.R.; Gerzina, T.M. Bioavailability of components of resin-based materials which are applied to teeth. Crit. Rev. Oral Biol. Med. 1996, 7, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Santerre, J.P.; Shajii, L.; Leung, B.W. Relation of dental composite formulations to their degradation and the release of hydrolyzed polymeric-resin-derived products. Crit. Rev. Oral Biol. Med. 2001, 12, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.S.; Liu, C.C.; Tseng, W.Y.; Jeng, J.H.; Lin, C.P. Cytotoxicity of three dentin bonding agents on human dental pulp cells. J. Dent. 2003, 31, 223–229. [Google Scholar] [CrossRef]
- Franz, A.; Konig, F.; Lucas, T.; Watts, D.C.; Schedle, A. Cytotoxic effects of dental bonding substances as a function of degree of conversion. Dent. Mater. 2009, 25, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Mohsen, N.M.; Craig, R.G.; Hanks, C.T. Cytotoxicity of urethane dimethacrylate composites before and after aging and leaching. J. Biomed. Mater. Res. 1998, 39, 252–260. [Google Scholar] [CrossRef]
- Ergun, G.; Egilmez, F.; Cekic-Nagas, I. The cytotoxicity of resin composites cured with three light curing units at different curing distances. Med. Oral Patol. Oral Cir. Bucal. 2011, 16, e252–e259. [Google Scholar] [CrossRef] [PubMed]
- Yalcin, M.; Ulker, M.; Ulker, E.; Sengun, A. Evaluation of cytotoxicity of six different flowable composites with the methyl tetrazolium test method. Eur. J. Gen. Dent. 2013, 2, 292. [Google Scholar] [CrossRef]
- Schedle, A.; Franz, A.; Rausch-Fan, X.; Spittler, A.; Lucas, T.; Samorapoompichit, P.; Sperr, W.; Boltz-Nitulescu, G. Cytotoxic effects of dental composites, adhesive substances, compomers and cements. Dent. Mater. 1998, 14, 429–440. [Google Scholar] [CrossRef]
- Dentistry-Evaluation of Biocompatibility of Medical Devices Used in Dentistry; ISO 7405; International Standard: Geneva, Switzerland, 2013.
- Prasad, M.; Mohamed, S.; Nayak, K.; Shetty, S.K.; Talapaneni, A.K. Effect of moisture, saliva, and blood contamination on the shear bond strength of brackets bonded with a conventional bonding system and self-etched bonding system. J. Nat. Sci. Biol. Med. 2014, 5, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Hong, S.; Choi, Y.; Park, S. The effect of saliva decontamination procedures on dentin bond strength after universal adhesive curing. Restor. Dent. Endod. 2015, 40, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Durner, J.; Obermaier, J.; Draenert, M.; Ilie, N. Correlation of the degree of conversion with the amount of elutable substances in nano-hybrid dental composites. Dent. Mater. 2012, 28, 1146–1153. [Google Scholar] [CrossRef] [PubMed]
- Evaluation of Medical Devices-Part 5: Tests for in Vitro Cytotoxicity; ISO 10993-5; International Standard: Geneva, Switzerland, 2009.
- Goon, A.T.; Isaksson, M.; Zimerson, E.; Goh, C.L.; Bruze, M. Contact allergy to (meth)acrylates in the dental series in southern Sweden: Simultaneous positive patch test reaction patterns and possible screening allergens. Contact Derm. 2006, 55, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Terakado, M.; Yamazaki, M.; Tsujimoto, Y.; Kawashima, T.; Nagashima, K.; Ogawa, J.; Fujita, Y.; Sugiya, H.; Sakai, T.; Furuyama, S. Lipid peroxidation as a possible cause of benzoyl peroxide toxicity in rabbit dental pulp—A microsomal lipid peroxidation in vitro. J. Dent. Res. 1984, 63, 901–905. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.A.; Vaerten, M.A.; Edwards, C.A.; Hanks, C.T. Cytotoxic effects of current dental adhesive systems on immortalized odontoblast cell line MDPC-23. Dent. Mater. 1999, 15, 434–441. [Google Scholar] [CrossRef]
- Anand, V.S.; Balasubramanian, V. Effect of resin chemistry on depth of cure and cytotoxicity of dental resin composites. Mater. Sci. Eng. B 2014, 181, 33–38. [Google Scholar] [CrossRef]
- Kwon, J.S.; Lee, S.B.; Kim, C.K.; Kim, K.N. Modified cytotoxicity evaluation of elastomeric impression materials while polymerizing with reduced exposure time. Acta Odontol. Scand. 2012, 70, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Schmalz, G.; Hiller, K.A.; Dörter-Aslan, F. New developments in the filter test system for cytotoxicity testing. J. Mater. Sci. Mater. Med. 1994, 5, 43–51. [Google Scholar] [CrossRef]
- Chen, F.; Wu, T.; Cheng, X. Cytotoxic effects of denture adhesives on primary human oral keratinocytes, fibroblasts and permanent L929 cell lines. Gerodontology 2014, 31, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Schweikl, H.; Hiller, K.A.; Bolay, C.; Kreissl, M.; Kreismann, W.; Nusser, A.; Steinhauser, S.; Wieczorek, J.; Vasold, R.; Schmalz, G. Cytotoxic and mutagenic effects of dental composite materials. Biomaterials 2005, 26, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Galvão, M.; Costa, S.; Victorino, K.; Ribeiro, A.; Menezes, F.; Rastelli, A.N.D.S.; Bagnato, V.S.; Andrade, M. Influence of light guide tip used in the photo-activation on degree of conversion and hardness of one nanofilled dental composite. Laser Phys. 2010, 20, 2050–2055. [Google Scholar] [CrossRef]
- Boaro, L.C.; Goncalves, F.; Guimaraes, T.C.; Ferracane, J.L.; Pfeifer, C.S.; Braga, R.R. Sorption, solubility, shrinkage and mechanical properties of “low-shrinkage” commercial resin composites. Dent. Mater. 2013, 29, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, M.A.; Stangel, I.; Ellis, T.H.; Zhu, X.X. Oxygen inhibition in dental resins. J. Dent. Res. 2005, 84, 725–729. [Google Scholar] [CrossRef] [PubMed]
- Tezvergil-Mutluay, A.; Lassila, L.V.; Vallittu, P.K. Degree of conversion of dual-cure luting resins light-polymerized through various materials. Acta Odontol. Scand. 2007, 65, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, I.; Otsuki, M.; Sadr, A.; Nomura, T.; Kishikawa, R.; Tagami, J. Effect of filler content of flowable composites on resin-cavity interface. Dent. Mater. J. 2009, 28, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Denizot, F.; Lang, R. Rapid colorimetric assay for cell growth and survival: Modifications to the tetrazolium dye procedure giving improved sensitivity and reliability. J. Immunol. Methods 1986, 89, 271–277. [Google Scholar] [CrossRef]

| Product | Manufacturer | Type | Formulation |
|---|---|---|---|
| Filtek™ Z-350XT | 3M ESPE, St. Paul, MN, USA | Composite resin | Bis-GMA, UDMA, Bis-EMA, PEGDMA, TEGDMA resins, combination of 20 nm silica filler, 4 to 11 nm zirconia filler, zirconia/silica cluster filler |
| Adper Scotchbond™ | 3M ESPE, St. Paul, MN, USA | Bonding resin | Bis-GMA, UDMA, HEMA, glycerol dimethacrylate (GDMA), modified polyacrylic acid, ethanol, water |
| Product | Material | Group | Code | Method of Sample Preparation | Curing Condition |
|---|---|---|---|---|---|
| Filtek™ | Composite resin | C | CU | Uncured | No polymerization |
| CD | Direct cured | Direct light curing | |||
| Z-350XT | CP | Post-cured | Indirect light curing | ||
| CR | Removed unreacted layer | Indirect light curing | |||
| Adper | Bonding resin | B | BU | Uncured | No polymerization |
| BD | Direct cured | Direct light curing | |||
| Scotchbond™ | BP | Post-cured | Indirect light curing | ||
| BR | Removed unreacted layer | Indirect light curing |
| Test Materials | Degree of Conversion (%) |
|---|---|
| CU | 0 |
| CD | 74.52 ± 2.28 |
| CP | 87.59 ± 1.51 |
| CR | 95.96 ± 0.07 |
| BU | 0 |
| BD | 48.71 ± 2.09 |
| BP | 61.14 ± 3.48 |
| BR | 94.45 ± 0.22 |
| Test Materials | Decolorization Index | Lysis Index | Interpretation |
|---|---|---|---|
| Positive control | 4 | 5 | Severely cytotoxic |
| Negative control | 0 | 0 | Non-cytotoxic |
| CU | 4 | 4 | Severely cytotoxic |
| CD | 4 | 5 | Severely cytotoxic |
| CP | 3 | 4 | Moderately cytotoxic |
| CR | 2 | 3 | Moderately cytotoxic |
| BU | 4 | 4 | Severely cytotoxic |
| BD | 4 | 5 | Severely cytotoxic |
| BP | 3 | 4 | Moderately cytotoxic |
| BR | 2 | 3 | Moderately cytotoxic |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, M.-J.; Kim, M.-J.; Kwon, J.-S.; Lee, S.-B.; Kim, K.-M. Cytotoxicity of Light-Cured Dental Materials according to Different Sample Preparation Methods. Materials 2017, 10, 288. https://doi.org/10.3390/ma10030288
Lee M-J, Kim M-J, Kwon J-S, Lee S-B, Kim K-M. Cytotoxicity of Light-Cured Dental Materials according to Different Sample Preparation Methods. Materials. 2017; 10(3):288. https://doi.org/10.3390/ma10030288
Chicago/Turabian StyleLee, Myung-Jin, Mi-Joo Kim, Jae-Sung Kwon, Sang-Bae Lee, and Kwang-Mahn Kim. 2017. "Cytotoxicity of Light-Cured Dental Materials according to Different Sample Preparation Methods" Materials 10, no. 3: 288. https://doi.org/10.3390/ma10030288
APA StyleLee, M.-J., Kim, M.-J., Kwon, J.-S., Lee, S.-B., & Kim, K.-M. (2017). Cytotoxicity of Light-Cured Dental Materials according to Different Sample Preparation Methods. Materials, 10(3), 288. https://doi.org/10.3390/ma10030288







