Laser-Induced Breakdown Spectroscopy (LIBS) for Monitoring the Formation of Hydroxyapatite Porous Layers
Abstract
:1. Introduction
2. Experimental
2.1. Sample Fabrication
2.2. Characterization Techniques
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hench, L.L.; Splinter, R.J.; Greenle, T.K.; Allen, W.C. Bonding mechanisms at the interface of ceramic prosthetic materials. J. Biomed. Mater. Res. 1971, 2, 117–141. [Google Scholar] [CrossRef]
- Hench, L.L.; Wilson, J. An Introduction to Bioceramics, 2nd ed.; Imperial College Press: London, UK, 2013. [Google Scholar]
- Best, S.M.; Porter, A.E.; Thian, E.S.; Huan, J. Bioceramics: Past, present and for the future. J. Eur. Ceram. Soc. 2008, 28, 1319–1327. [Google Scholar] [CrossRef]
- Lu, J.X.; Flautre, B.; Anselme, K.; Hardouin, P.; Gallur, A.; Descamps, M.; Thierry, B. Role of interconnections in porous bioceramics on bone recolonization in vitro and in vivo. J. Mater. Sci. Mater. Med. 1999, 10, 111–120. [Google Scholar] [CrossRef] [PubMed]
- De Groot, K.; Le Geros, R. Significance of Porosity and Physical Chemistry of Calcium Phosphate Ceramics; Ducheyne, P., Ed.; Annal New York Academy Sciences: New York, NY, USA, 1988. [Google Scholar]
- Von Doernberg, M.C.; von Rechenberg, B.; Bohner, M.; Grunenfelder, S.; van Lenthe, G.H.; Muller, R.; Gasser, B.; Mathys, R.; Baroud, G.; Auer, J. In vivo behaviour of calcium phosphate scaffolds with four different pore sizes. Biomaterials 2006, 27, 5186–5198. [Google Scholar] [CrossRef] [PubMed]
- Bungo, O.; Mitsuru, T.; Shunsuke, F.; Masashi, N.; Tadashi, K.; Takashi, N. Pore throat size and connectivity determine bone and tissue ingrowth into porous implants: Tree-dimensional micro-CT based structural analyses of porous bioactive titanium implants. Biomaterial 2006, 27, 5892–5900. [Google Scholar]
- De Aza, P.N.; Guitian, F.; de Aza, S. Phase diagram of wollastonite-tricalcium phosphate. J. Am. Ceram. Soc. 1995, 78, 1653–1656. [Google Scholar] [CrossRef]
- De Aza, P.N.; Guitian, F.; de Aza, S. Bioeutectic: A new ceramic material for human bone replacement. Biomaterials 1997, 18, 1285–1291. [Google Scholar] [CrossRef]
- Magallanes-Perdomo, M.; de Aza, A.H.; Sobrados, I.; Sanz, J.; Pena, P. Structure and properties of bioactive eutectic glasses based on the Ca3(PO4)2–CaSiO3–CaMg(SiO3)2 system. Acta Biomater. 2012, 8, 820–829. [Google Scholar] [CrossRef] [PubMed]
- Llorca, J.; Orera, V.M. Directionally-solidified eutectic ceramic oxides. Prog. Mater. Sci. 2006, 51, 711–809. [Google Scholar] [CrossRef]
- Sola, D.; Ester, F.J.; Oliete, P.B.; Peña, J.I. Study of the stability of the molten zone and the stresses induced during the growth of Al2O3–Y3Al5O12 eutectic composite by the laser floating zone technique. J. Eur. Ceram. Soc. 2011, 31, 1211–1218. [Google Scholar] [CrossRef]
- Ester, F.J.; Sola, D.; Peña, J.I. Thermal stresses in the Al2O3-ZrO2(Y2O3) eutectic composite during the growth by the laser floating zone technique. Bol. Soc. Esp. Ceram. 2008, 47, 352–357. [Google Scholar] [CrossRef]
- Sola, D.; Peña, J.I. Laser machining of Al2O3-ZrO2 (3%Y2O3) eutectic composite. J. Eur. Ceram. Soc. 2012, 32, 807–814. [Google Scholar] [CrossRef]
- Orera, V.M.; Peña, J.I.; Merino, R.I.; Lazaro, J.A.; Valles, J.A.; Rebolledo, M.A. Prospects of new planar optical waveguides based on the eutectic microcomposites of insulating crystals: ZrO2 (c)-CaZrO3 erbium doped system. Appl. Phys. Lett. 1997, 71, 2746–2748. [Google Scholar] [CrossRef]
- Sola, D.; Oliete, P.B.; Peña, J.I. Directionally solidified fabrication in planar geometry of Al2O3-Er3Al5O12 eutectic composite for thermophotovoltaic devices. Opt. Express 2016, 24, A823–A831. [Google Scholar] [CrossRef] [PubMed]
- Pardo, J.A.; Peña, J.I.; Merino, R.I.; Cases, R.; Larrea, A.; Orera, V.M. Spectroscopic properties of Er3+ and Nd3+ doped glasses with 0.8CaSiO3–0.2Ca3(PO4)2 eutectic composition. J. Non-Cryst. Solids 2002, 298, 23–31. [Google Scholar] [CrossRef]
- Sola, D.; Balda, R.; Peña, J.I.; Fernandez, J. Site-selective laser spectroscopy of Nd3+ ions in 0.8CaSiO3–0.2Ca3(PO4)2 biocompatible eutectic glass-ceramics. Opt. Express 2012, 20, 10701–10711. [Google Scholar] [CrossRef] [PubMed]
- Sola, D.; Balda, R.; Al-Saleh, M.; Peña, J.I.; Fernandez, J. Time-resolved fluorescence line-narrowing of Eu3+ in biocompatible eutectic glass-ceramics. Opt. Express 2013, 21, 6561–6571. [Google Scholar] [CrossRef] [PubMed]
- De Mendivil, J.M.; Sola, D.; de Aldana, J.R.V.; Lifante, G.; De Aza, A.H.; Pena, P.; Peña, J.I. Ultrafast direct laser writing of cladding waveguides in the 0.8CaSiO3–0.2Ca3(PO4)2 eutectic glass doped with Nd3+ ions. J. Appl. Phys. 2015, 117, 043104. [Google Scholar] [CrossRef]
- Sola, D.; De Mendibil, J.M.; De Aldana, J.R.V.; Lifante, G.; Balda, R.; De Aza, A.H.; Pena, P.; Fernandez, J. Stress-induced buried waveguides in the 0.8CaSiO3–0.2Ca 3(PO4)2 eutectic glass doped with Nd3+ ions. Appl. Surf. Sci. 2013, 278, 289–294. [Google Scholar] [CrossRef]
- Carrodeguas, R.G.; de Aza, S. α-Tricalcium phosphate: Synthesis, properties and biomedical applications. Acta Biomater. 2011, 7, 3536–3546. [Google Scholar] [CrossRef] [PubMed]
- De Aza, P.N.; Luklinska, Z.B.; Anseau, M.R.; Hector, M.; Guitian, F.; De Aza, S. Reactivity of a wollastonite-tricalcium phosphate Bioeutectic® ceramic in human parotid saliva. Biomaterials 2000, 21, 1735–1741. [Google Scholar] [CrossRef]
- De Aza, P.N.; Guitian, F.; de Aza, S. A new bioactive material which transforms in situ into hydroxyapatite. Acta Mater. 1998, 46, 2541–2549. [Google Scholar] [CrossRef]
- Magallanes-Perdomo, M.; Pena, P.; de Aza, P.N.; Carrodeguas, R.G.; Rodríguez, M.A.; Turrillas, X.; de Aza, S.; de Aza, A.H. Devitrification studies of wollastonite–tricalcium phosphate eutectic glass. Acta Biomater. 2009, 5, 3057–3066. [Google Scholar] [CrossRef] [PubMed]
- De Aza, P.N.; Peña, J.I.; Luklinska, Z.B.; Meseguer-Olmo, L. Bioeutectic® ceramics for biomedical application obtained by Laser Floating Zone method. In vivo Evaluation. Materials 2014, 7, 2395–2410. [Google Scholar] [CrossRef] [PubMed]
- Magallanes-Perdomo, M.; De Aza, A.H.; Sobrados, I.; Sanz, J.; Luklinska, Z.B.; Pena, P. Structural changes during crystallization of apatite and wollastonite in the eutectic glass of Ca3(PO4)2-CaSiO3 system. J. Am. Ceram. Soc. 2017, 100, 4288–4304. [Google Scholar] [CrossRef]
- Paules, D.; Hamida, S.; Lasheras, R.J.; Escudero, M.; Benouali, D.; Cáceres, J.O.; Anzano, J. Characterization of natural and treated diatomite by Laser-Induced Breakdown Spectroscopy (LIBS). Microchem. J. 2018, 137, 1–7. [Google Scholar] [CrossRef]
- Cremers, D.A.; Radziemski, L.J. Handbook of Laser-Induced Breakdown Spectroscopy, 2nd ed.; Wiley-Blackwell: Oxford, UK, 2013. [Google Scholar]
- Hahn, D.W.; Omenetto, N. Laser-Induced Breakdown Spectroscopy (LIBS), Part I: Review of Basic Diagnostics and Plasma-Particle Interactions: Still-Challenging Issues within the Analytical Plasma Community. Appl. Spectrosc. 2010, 64, 335–366. [Google Scholar] [CrossRef] [PubMed]
- Hahn, D.W.; Omenetto, N. Laser-Induced Breakdown Spectroscopy (LIBS), Part II: Review of Instrumental and Methodological Approaches to Material Analysis and Applications to Different Fields. Appl. Spectrosc. 2012, 66, 347–419. [Google Scholar] [CrossRef] [PubMed]
- Miziolek, A.W.; Palleschi, V.; Schechter, I. Laser-Induced Breakdown Spectroscopy (LIBS). Fundamentals and Applications; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Parigger, C.G. Atomic and molecular emissions in laser-induced breakdown spectroscopy. Spectrochim. Acta Part B At. Spectrosc. 2013, 79–80, 4–16. [Google Scholar] [CrossRef]
- Sola, D.; Conejos, D.; de Mendivil, J.M.; Ortega-San-Martin, L.; Lifante, G.; Peña, J.I. Directional solidification, thermo-mechanical and optical properties of (MgxCa1−x)3Al2Si3O12 glasses doped with Nd3+ ions. Opt. Express 2015, 23, 26356–26368. [Google Scholar] [CrossRef] [PubMed]
- Arias-Egido, E.; Sola, D.; Pardo, J.A.; Martínez, J.I.; Cases, R.; Peña, J.I. On the control of optical transmission of aluminosilicate glasses manufactured by the laser floating zone technique. Opt. Mater. Express 2016, 6, 2413–2421. [Google Scholar] [CrossRef]
- Oyane, A.; Kim, H.; Furuya, T.; Kokubo, T.; Miyazaki, T.; Nakamura, T. Preparation and assessment of revised simulated body fluid. J. Biomed. Mater. Res. A 2003, 65, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Tariq, U.; Haider, Z.; Hussain, R.; Tufail, K.; Ali, J. LIBS Analysis of Hydroxyapatite Extracted from Bovine Bone for Ca/P Ratio Measurements. In Proceedings of the 9th International Conference on Plasma Science and Applications, Langkawi, Malaysia, 28–30 November 2017; Volume 1824. [Google Scholar]
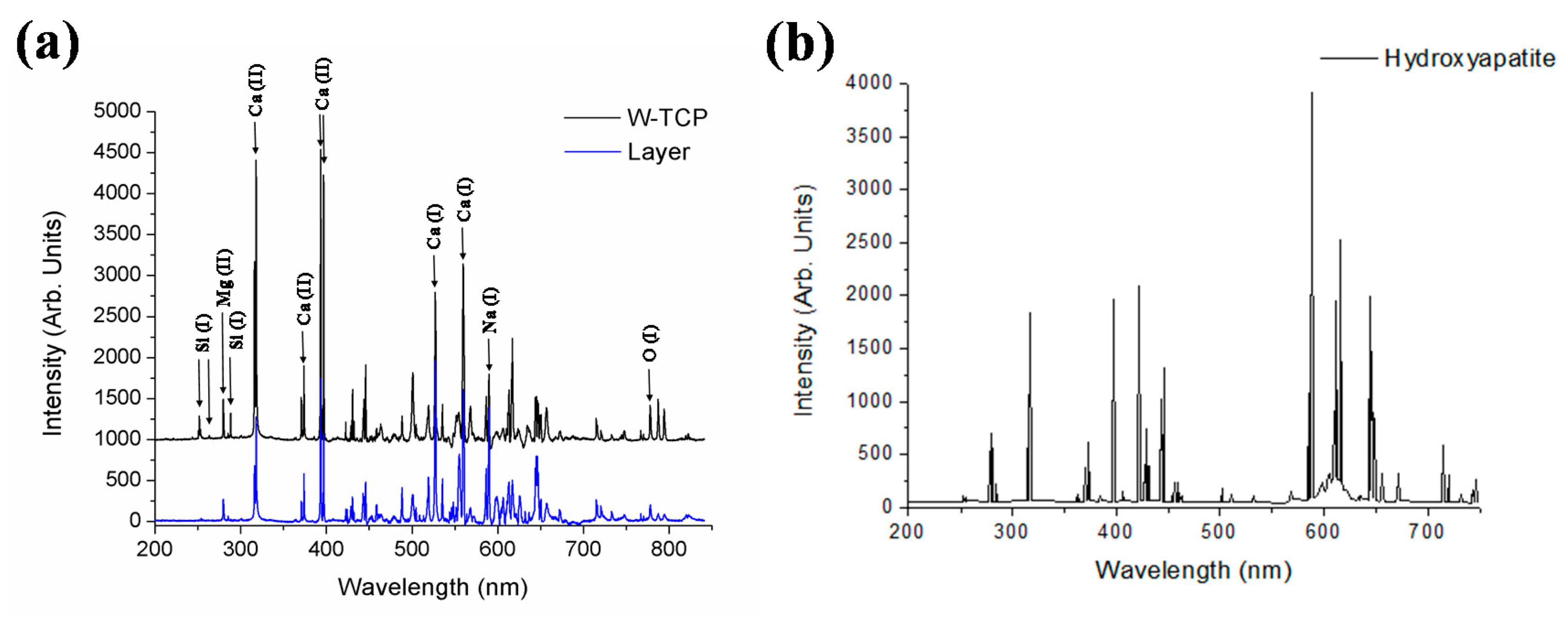
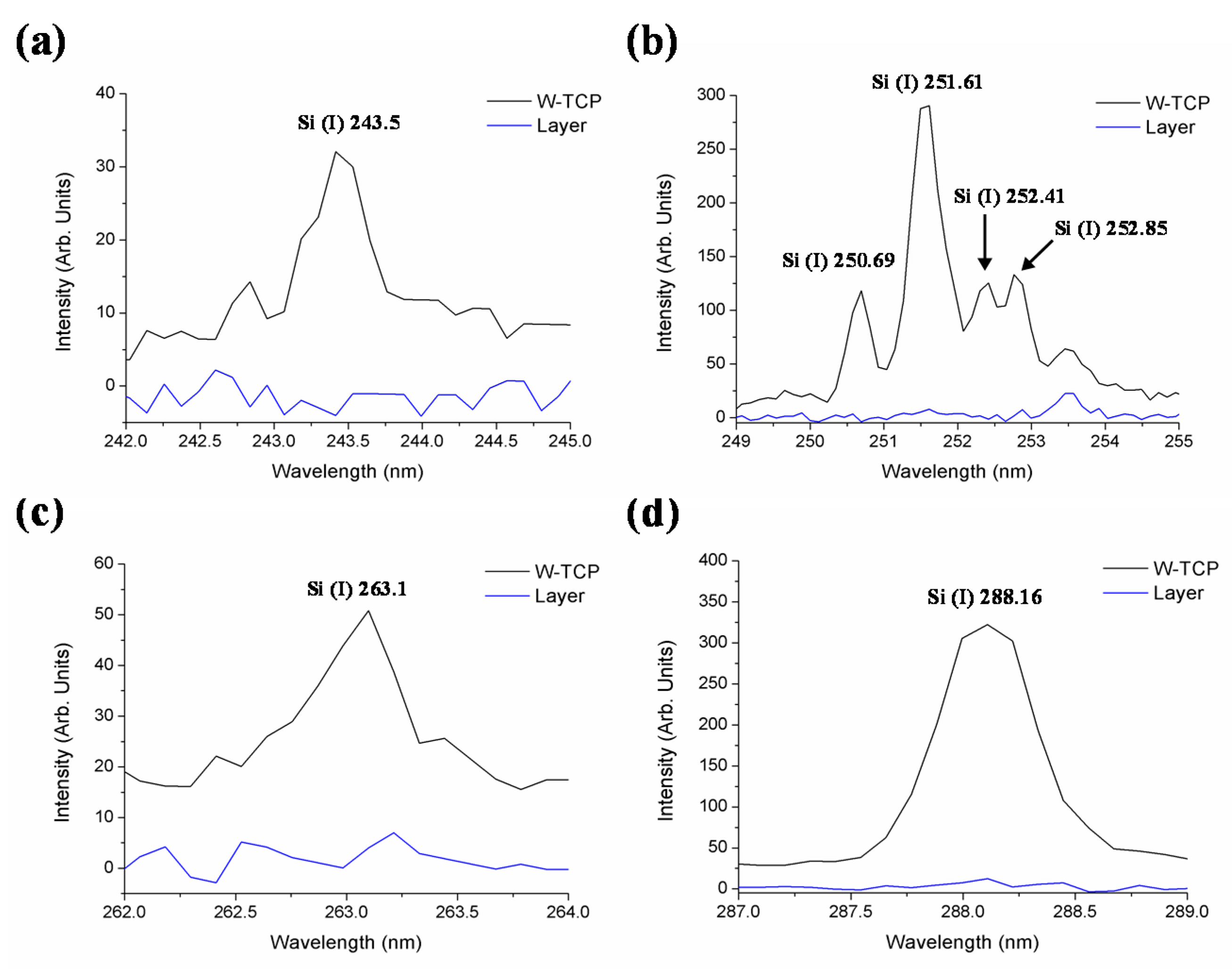
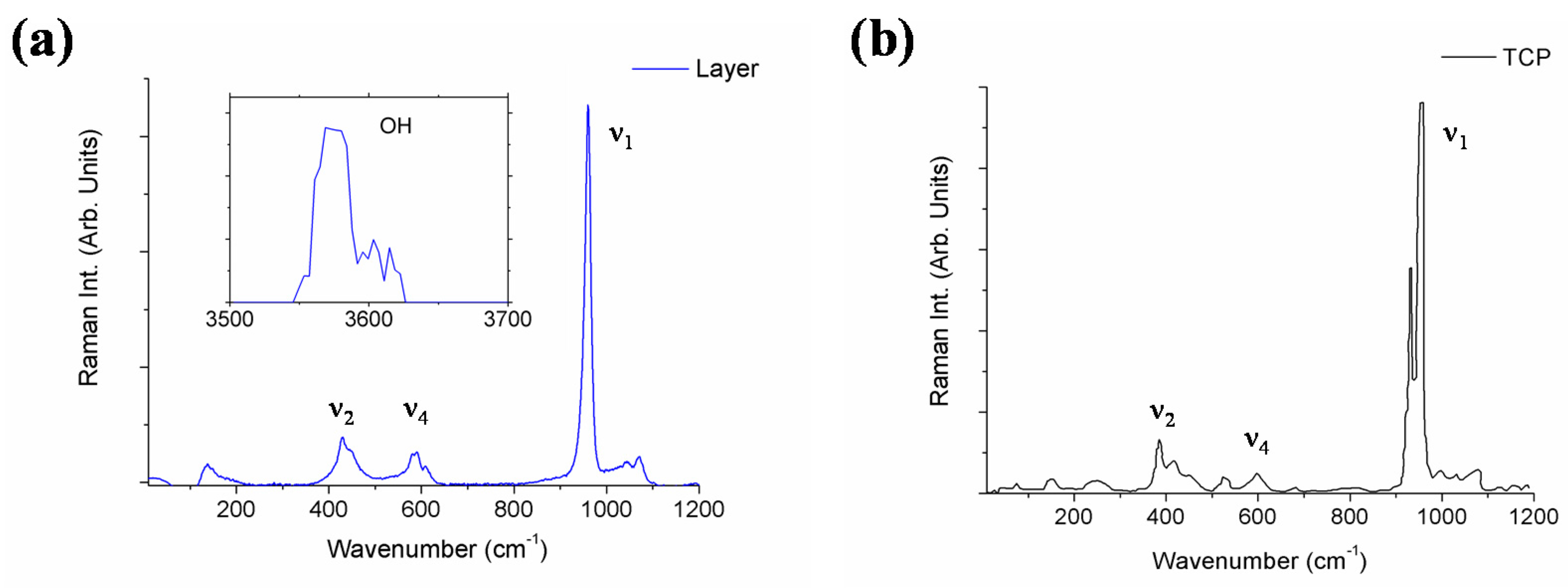

| Element | Sample | Wavelength (nm) |
|---|---|---|
| Si (I) | W-TCP | 243.5, 250.69, 251.61, 252.41, 252.85, 263.1, 288.16 |
| Ca (I) | W-TCP, HA | 527.03, 559.45 |
| Ca (II) | W-TCP, HA | 317.93, 373.69, 393.37, 396.85 |
| Mg (II) | W-TCP, HA | 279.55 |
| Na (I) | W-TCP, HA | 589 |
| O (I) | W-TCP, HA | 777.5 |
| Sample | Si | P | Ca |
|---|---|---|---|
| Glass | 12.73 | 6.27 | 18.86 |
| Layer | – | 16.56 | 21.70 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sola, D.; Paulés, D.; Grima, L.; Anzano, J. Laser-Induced Breakdown Spectroscopy (LIBS) for Monitoring the Formation of Hydroxyapatite Porous Layers. Materials 2017, 10, 1395. https://doi.org/10.3390/ma10121395
Sola D, Paulés D, Grima L, Anzano J. Laser-Induced Breakdown Spectroscopy (LIBS) for Monitoring the Formation of Hydroxyapatite Porous Layers. Materials. 2017; 10(12):1395. https://doi.org/10.3390/ma10121395
Chicago/Turabian StyleSola, Daniel, Daniel Paulés, Lorena Grima, and Jesús Anzano. 2017. "Laser-Induced Breakdown Spectroscopy (LIBS) for Monitoring the Formation of Hydroxyapatite Porous Layers" Materials 10, no. 12: 1395. https://doi.org/10.3390/ma10121395
APA StyleSola, D., Paulés, D., Grima, L., & Anzano, J. (2017). Laser-Induced Breakdown Spectroscopy (LIBS) for Monitoring the Formation of Hydroxyapatite Porous Layers. Materials, 10(12), 1395. https://doi.org/10.3390/ma10121395







