Promoting Effect of Inorganic Alkali on Carbon Dioxide Adsorption in Amine-Modified MCM-41
Abstract
:1. Introduction
2. Experimental
2.1. Materials Preparation
2.2. Materials Characterization
2.3. CO2 Adsorption
3. Results and Discussion
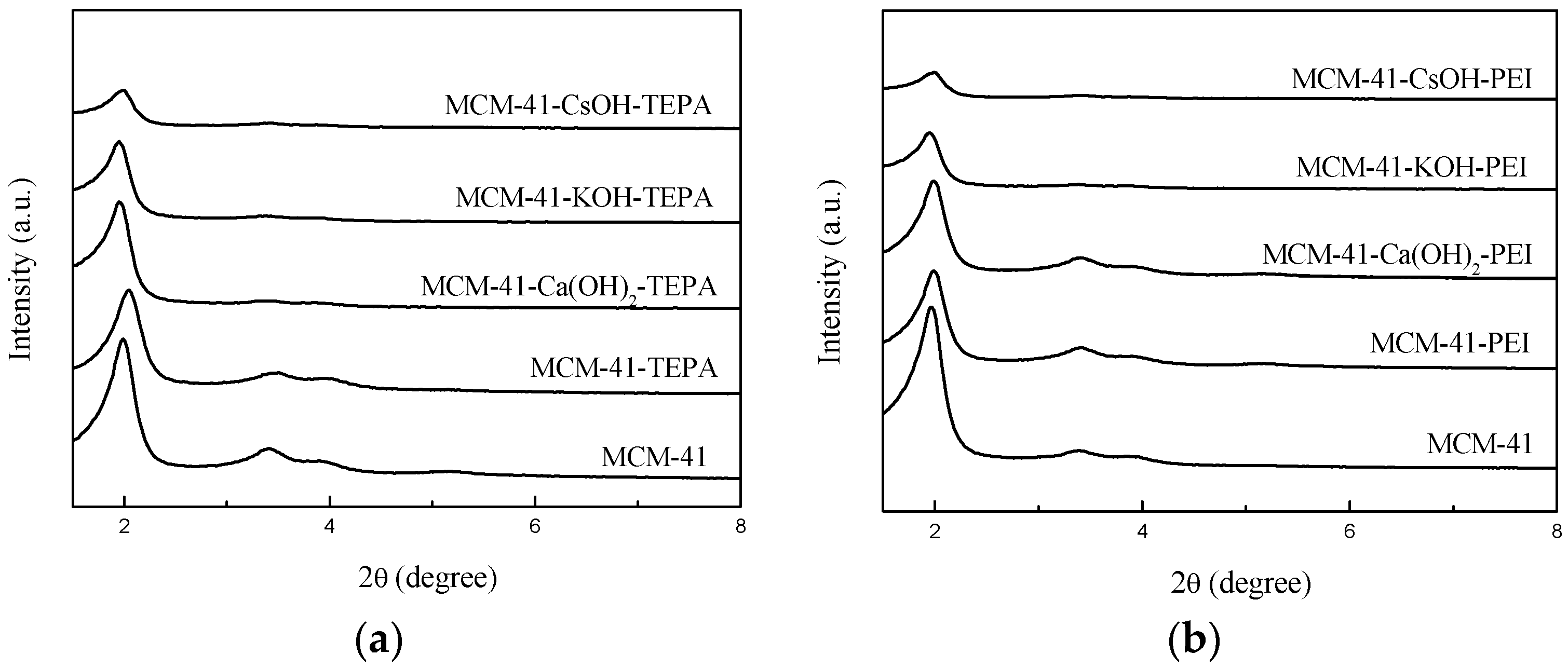
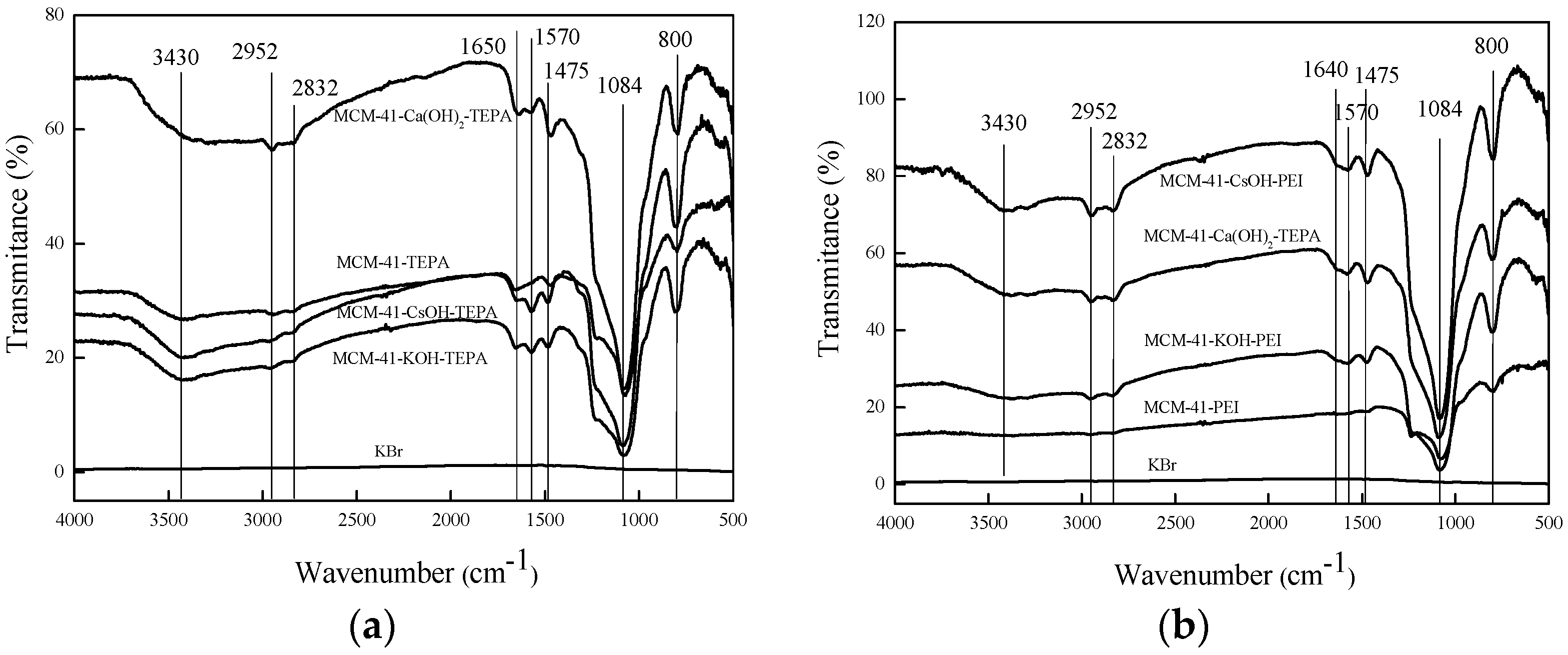
3.1. Preparation and Characterization of the Adsorbents
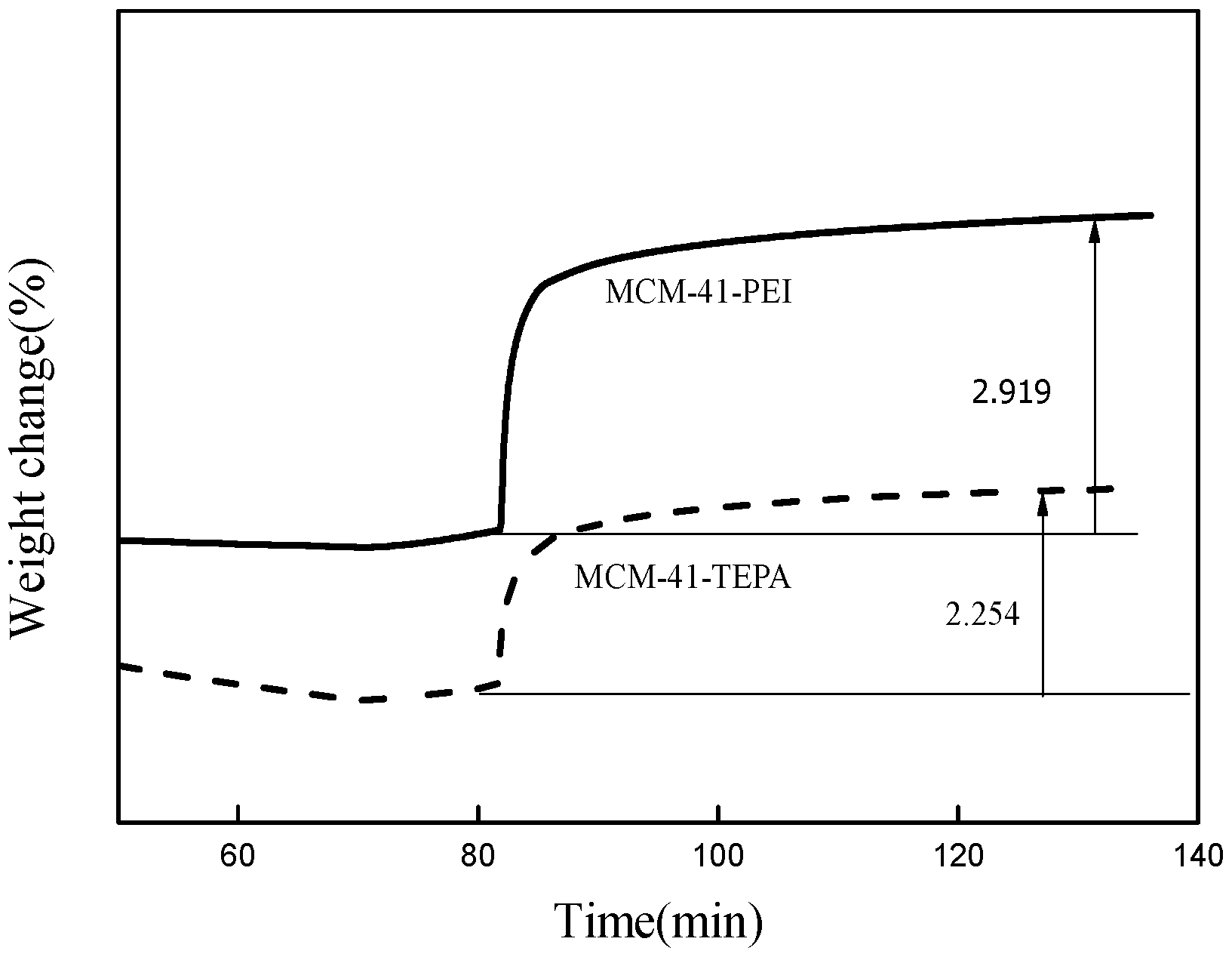
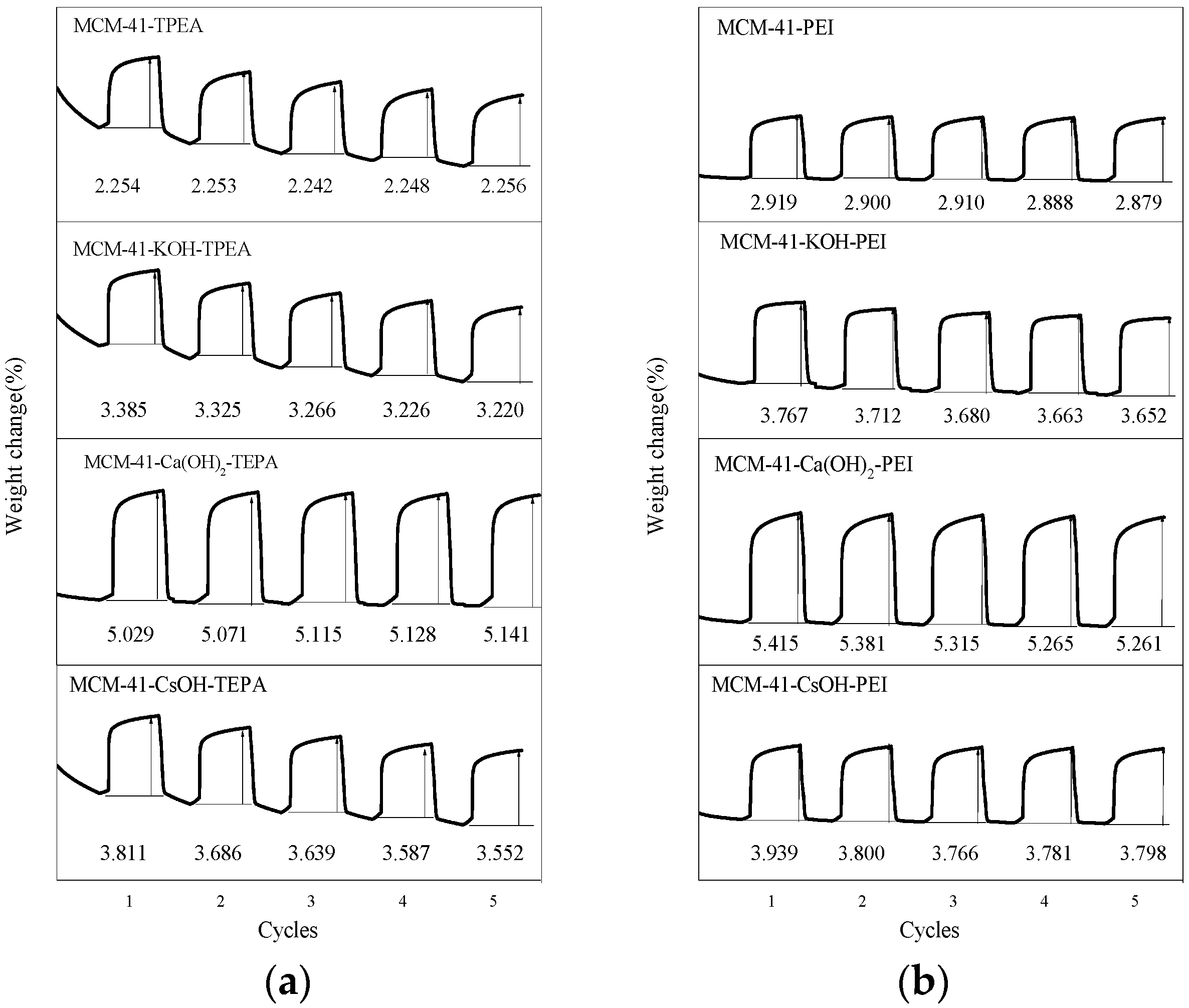
3.2. CO2 Adsorption Performance of Adsorbents
3.3. Promoting Effect of Inorganic Alkali
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| TEPA | Tetraethylenepentamine |
| PEI | Polyethyleneimine |
| FT-IR | Fourier transform infrared |
| TGA | Thermo-gravimetric analysis |
| CTAB | Cetyltrimethylammonium Bromide |
| TEOS | Tetraethylorthosilicate |
| DEA | Diethanol amine |
| DTEA | Diethylenetriamine |
| TETA | Triethylenetetramine |
| AMP | 2-amino-2-methyl-1-propanol |
References
- Gibbins, J.; Chalmers, H. Carbon capture and storage. Energy Policy 2008, 36, 4317–4322. [Google Scholar] [CrossRef]
- Schrag, D.P. Preparing to capture carbon. Science 2007, 315, 812–813. [Google Scholar] [CrossRef] [PubMed]
- Koronaki, I.P.; Prentza, L.; Papaefthimiou, V. Modeling of CO2 capture via chemical absorption processes—An extensive literature review. Renew. Sustain. Energy Rev. 2015, 50, 547–566. [Google Scholar] [CrossRef]
- Xu, X.; Song, C.; Andersen, J.M.; Miller, B.G.; Scaroni, A.W. Preparation and characterization of novel CO2 “molecular basket” adsorbents based on polymer-modified mesoporous molecular sieve MCM-41. Microporous Mesoporous Mater. 2003, 62, 29–45. [Google Scholar] [CrossRef]
- Yue, M.B.; Chun, Y.; Cao, Y.; Dong, X.; Zhu, J.H. CO2 capture by as-prepared SBA-15 with an occluded organic template. Adv. Funct. Mater. 2006, 16, 1717–1722. [Google Scholar] [CrossRef]
- Chen, C.; Son, W.J.; You, K.S.; Ahn, J.W.; Ahn, W.S. Carbon dioxide capture using amine-impregnated HMS having textural mesoporosity. Chem. Eng. J. 2010, 161, 46–52. [Google Scholar] [CrossRef]
- Brunetti, A.; Scura, F.; Barbieri, G.; Drioli, E. Membrane technologies for CO2 separation. J. Membr. Sci. 2010, 359, 115–125. [Google Scholar] [CrossRef]
- Xu, G.; Li, L.; Yang, Y.; Tian, L.; Liu, T.; Zhang, K. A novel CO2 cryogenic liquefaction and separation system. Energy 2012, 42, 522–529. [Google Scholar] [CrossRef]
- Auta, M.; Amat Darbis, N.D.; Mohd, A.T.; Hameed, B.H. Fixed-bed column adsorption of carbon dioxide by sodium hydroxide modified activated alumina. Chem. Eng. J. 2013, 233, 80–87. [Google Scholar] [CrossRef]
- Hao, G.P.; Li, W.C.; Lu, A.H. Novel porous solids for carbon dioxide capture. J. Mater. Chem. 2011, 21, 6447–6451. [Google Scholar] [CrossRef]
- Siriwardane, R.N.; Shen, M.S.; Fisher, E.P. Adsorption of CO2, N2, and O2 on natural zeolites. Energy Fuels 2003, 17, 571–576. [Google Scholar] [CrossRef]
- Bezerra, D.P.; Oliveira, R.S.; Vieira, R.S.; Cavalcante, C.L.; Azevedo, D.C.S. Adsorption of CO2 on nitrogen-enriched activated carbon and zeolite 13X. Adsorption 2011, 17, 235–246. [Google Scholar] [CrossRef]
- Herm, Z.R.; Swisher, J.A.; Smit, B.; Krishna, R.; Long, J.R. Metal-organic frameworks as adsorbents for hydrogen purification and precombustion carbon dioxide capture. J. Am. Chem. Soc. 2011, 133, 5664–5667. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Chen, X.; Anthony, E.J.; Jiang, X.; Duan, L.; Wu, Y.; Dong, W.; Zhao, C. Capturing CO2 in flue gas from fossil fuel-fired power plants using dry regenerable alkali metal-based sorbent. Prog. Energ. Combust. 2013, 39, 515–534. [Google Scholar] [CrossRef]
- Xu, X.; Song, C.; Andersen, J.M.; Mille, B.G.; Scaroni, A.W. Novel polyethylenimine-modified mesoporousmolecular sieve of MCM-41 type as high-capacity adsorbent for CO2 capture. Energy Fuels 2002, 16, 1463–1469. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, L.; Fu, X.; Sun, Y.; Su, W.; Zhou, Y. Adsorption and regeneration study of the mesoporous adsorbent SBA-15 adapted to the capture/separation of CO2 and CH4. Chem. Eng. Sci. 2007, 62, 1101–1110. [Google Scholar] [CrossRef]
- Yue, M.B.; Sun, L.B.; Cao, Y.; Wang, Z.J.; Wang, Y.; Yu, Q.; Zhu, J.H. Promoting the CO2 adsorption in the amine-containing SBA-15 by hydroxyl group. Microporous Mesoporous Mater. 2008, 114, 74–81. [Google Scholar] [CrossRef]
- Wei, J.; Liao, L.; Xiao, Y.; Zhang, P.; Shi, Y. Capture of carbon dioxide by amine-impregnated as-synthesized MCM-41. J. Environ. Sci. 2010, 22, 1558–1563. [Google Scholar] [CrossRef]
- Su, F.; Lu, C.; Kuo, S.C.; Zeng, W. Adsorption of CO2 on amine-functionalized Y-type zeolites. Energy Fuels 2010, 24, 1441–1448. [Google Scholar] [CrossRef]
- Liu, Z.; Teng, Y.; Zhang, K.; Cao, Y.; Pan, W. CO2 adsorption properties and thermal stability of different amine-impregnated MCM-41 materials. J. Fuel. Chem. Technol. 2013, 41, 469–475. [Google Scholar] [CrossRef]
- Liu, Z.; Teng, Y.; Zhang, K.; Chen, H.; Yang, Y. CO2 adsorption performance of different amine-based siliceous MCM-41 materials. J. Energy Chem. 2015, 24, 322–330. [Google Scholar] [CrossRef]
- Kamarudin, K.N.; Alias, N. Adsorption performance of MCM-41 impregnated with amine for CO2 removal. Fuel Process. Technol. 2013, 106, 332–337. [Google Scholar] [CrossRef]
- Liu, L.; Li, H.; Zhang, Y. A comparative study on catalytic performances of chromium incorporated and supported mesoporous MSU-x catalysts for the oxide hydrogenation of ethane to ethylene with carbon dioxide. Catal. Today 2006, 115, 235–241. [Google Scholar] [CrossRef]
- Khatri, R.A.; Chuang, S.S.C.; Soong, Y.; Gray, M. Carbon dioxide capture by diamine-grafted SBA-15: A combined fourier transform infrared and mass spectrometry study. Ind. Eng. Chem. Res. 2005, 44, 3702–3708. [Google Scholar] [CrossRef]
- Serna-Guerrero, R.; Da’na, E.; Sayari, A. New insights into the interactions of CO2 with amine-functionalized silica. Ind. Eng. Chem. Res. 2008, 47, 9406–9412. [Google Scholar] [CrossRef]
- Zelenak, V.; Halamova, D.; Gaberova, L.; Bloch, E.; Llewellyn, P. Amine-modified SBA-12 mesoporous silica for carbon dioxide capture: Effect of amine basicity on sorption properties. Microporous Mesoporous Mater. 2008, 116, 358–364. [Google Scholar] [CrossRef]
- Leal, O.; Bolívar, C.; Ovalles, C.; García, J.J.; Espidel, Y. Reversible adsorption of carbon dioxide on amine surface-bonded silica gel. Igorg. Chim. Acta 1995, 240, 183–189. [Google Scholar] [CrossRef]




| Materials | Surface Areas (m2/g) | Pore Volume (cm3/g) | Average Pore Diameters (nm) |
|---|---|---|---|
| MCM-41 | 865 | 1.00 | 4.64 |
| MCM-41-TEPA | 431 | 0.83 | 2.21 |
| MCM-41-KOH-TEPA | 322 | 0.97 | 2.15 |
| MCM-41-Ca(OH)2-TEPA | 405 | 0.94 | 2.31 |
| MCM-41-CsOH-TEPA | 293 | 0.97 | 2.61 |
| MCM-41-PEI | 508 | 0.98 | 2.54 |
| MCM-41-KOH-PEI | 391 | 1.08 | 2.33 |
| MCM-41-Ca(OH)2-PEI | 411 | 1.12 | 2.50 |
| MCM-41-CsOH-PEI | 306 | 0.91 | 2.14 |
| Materials | NaOH Solution (/mL) | Alkali Amounts/(mmol/g) |
|---|---|---|
| MCM-41-TEPA | 11.81 | 0.8812 |
| MCM-41-KOH-TEPA | 8.91 | 1.1607 |
| MCM-41-Ca(OH)2-TEPA | 4.35 | 1.6001 |
| MCM-41-CsOH-TEPA | 8.82 | 1.1693 |
| MCM-41-PEI | 12.48 | 0.7819 |
| MCM-41-KOH-PEI | 6.73 | 1.3708 |
| MCM-41-Ca(OH)2-PEI | 5.86 | 1.4546 |
| MCM-41-CsOH-PEI | 6.35 | 1.4074 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teng, Y.; Li, L.; Xu, G.; Zhang, K.; Li, K. Promoting Effect of Inorganic Alkali on Carbon Dioxide Adsorption in Amine-Modified MCM-41. Energies 2016, 9, 667. https://doi.org/10.3390/en9090667
Teng Y, Li L, Xu G, Zhang K, Li K. Promoting Effect of Inorganic Alkali on Carbon Dioxide Adsorption in Amine-Modified MCM-41. Energies. 2016; 9(9):667. https://doi.org/10.3390/en9090667
Chicago/Turabian StyleTeng, Yang, Lijiao Li, Gang Xu, Kai Zhang, and Kaixi Li. 2016. "Promoting Effect of Inorganic Alkali on Carbon Dioxide Adsorption in Amine-Modified MCM-41" Energies 9, no. 9: 667. https://doi.org/10.3390/en9090667






