Bio-Refining of Carbohydrate-Rich Food Waste for Biofuels
Abstract
:1. Introduction
2. Materials and Methods
2.1. R. glutinis Culturing and Acclimatization
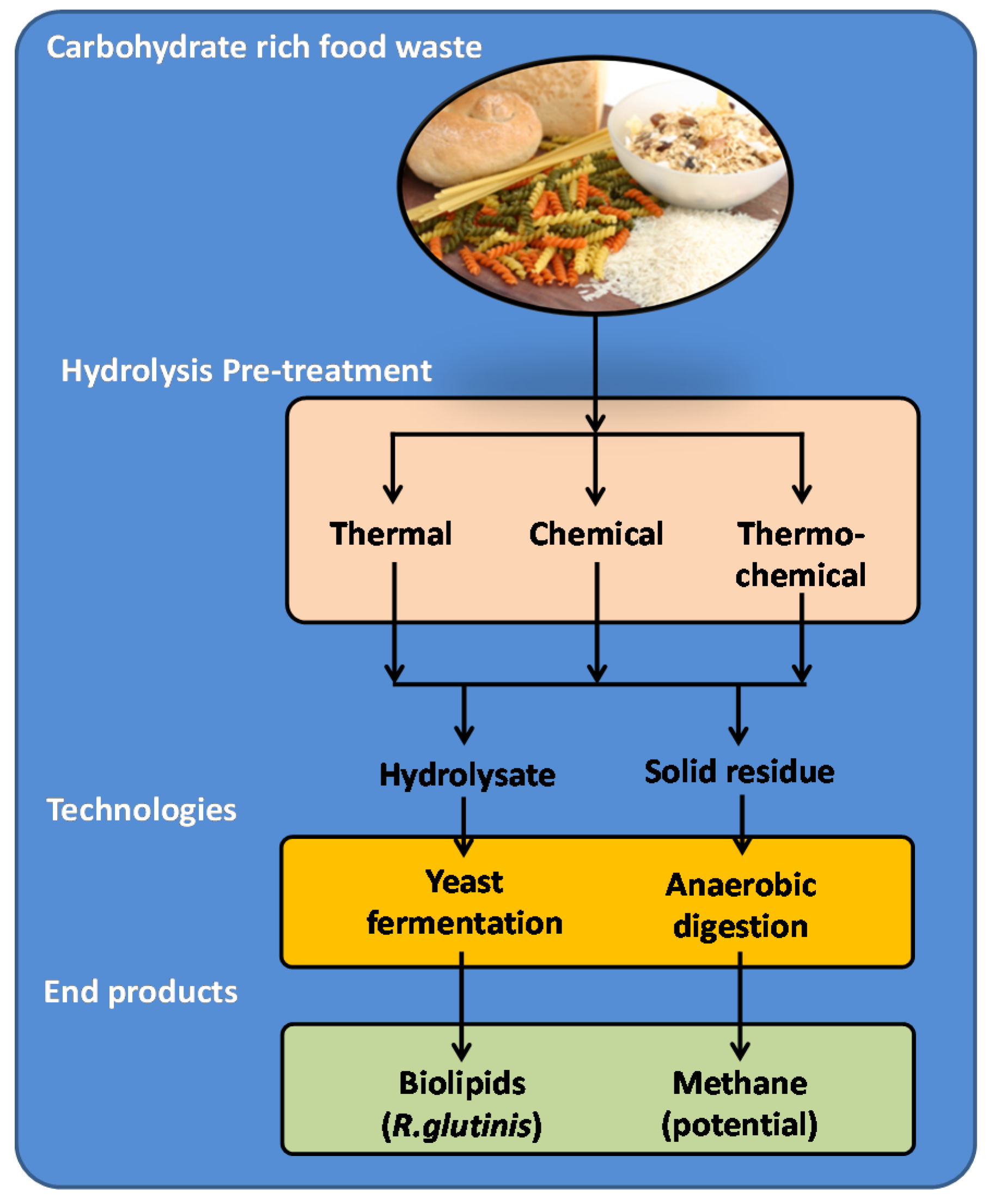
2.2. Bio-Refining of Carbohydrate-Rich Food Waste
2.2.1. Hydrolysis of CRFW
- (i)
- Chemical hydrolysis (Ch)—acidic hydrolysis of the CRFW slurry at a pH of 3 ± 0.01 adjusted with 2 M HCl (Sigma Aldrich, Australia) for 24 h at room temperature (25 °C);
- (ii)
- Thermal hydrolysis (Th)—autoclaving of the CRFW slurry using a standard moisture-heat procedure of 121 °C at 1013.25 hPa for 30 min (Tomy, VWR International, Murarrie, QLD 4172, Australia);
- (iii)
- Thermochemical hydrolysis (TCh)—a combined double hydrolysing procedure, where the chemical hydrolysis of the slurry preceded the thermal hydrolysis.
2.2.2. Cultivation of R. Glutinis in Hydrolysates for Bio-Product Development
2.3. Analytical Procedure
2.3.1. Reagents and Standard Calibration Gases
2.3.2. Characterization of CRFW
2.3.3. Calculating Bio-diesel and Bio-energy Potential of Bio-refined CRFW
3. Results and Discussion
3.1. Characteristics of Hydrolysates from Pre-treated CRFW
3.2. Growth of R. glutinis in CRFW Hydrolysates

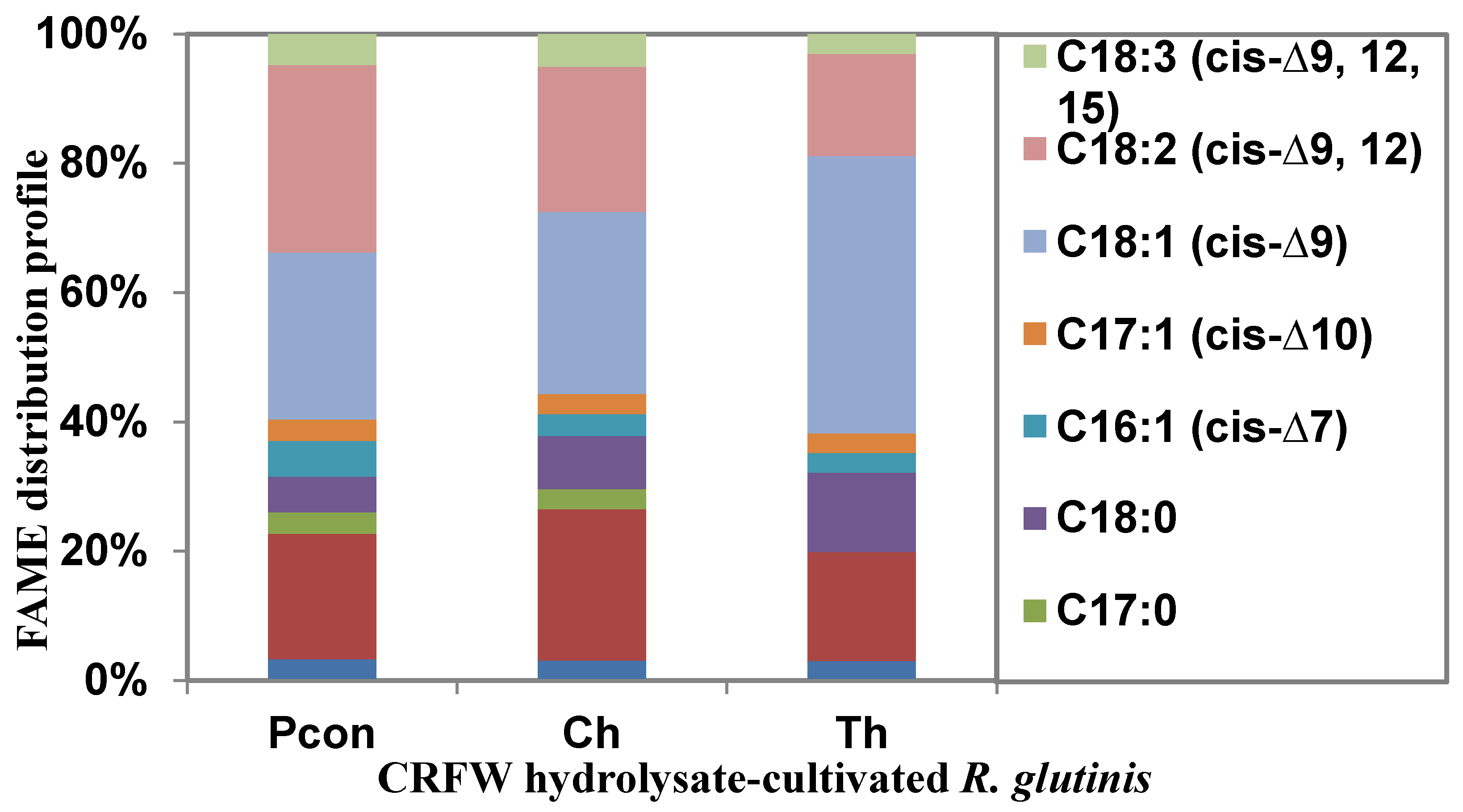
3.2.1. FAME Profile of R. glutinis Cultivated in CRFW Hydrolysates
| Particulars | Pre-treatment of CRFW | ||
|---|---|---|---|
| PCon | Ch | Th | |
| Fatty Acids (mg g−1 DW) | 41.79 ± 12.67 | 38.05 ± 8.23 | 65.56 ± 30.91 |
| Branching Fatty Acid (%) | 9.88 ± 6.02 | 3.79 ± 2.18 | <3 |
| Saturated Fatty Acid (%) | 37.81 ± 7.41 | 39.5 ± 2.78 | 32.53 ± 11.50 |
| Monounsaturated Fatty Acid (%) | 28.64 ± 12.90 | 31.82 ± 12.77 | 51.65 ± 34.85 |
| Polyunsaturated Fatty Acid (%) | 33.55 ± 20.81 | 28.67 ± 12.82 | 15.82 ± 0.98 |
3.2.2. Biodiesel Potential of R. glutinis Enriched from CRFW Hydrolysates
| CN | ʋ | ρ | HHV | |
|---|---|---|---|---|
| ASTM D6751-02 | ≥47 | 1.9–6.0 | 0.86 * | NA |
| EN 14214 | ≥51 | 3.5–5.0 | 0.86–0.90 | 35 ** |
| PCon | 51.06 ± 0.12 | 3.59 ± 0.05 | 0.79 ± 0.03 | 35.76 ± 1.23 |
| Ch | 56.92 ± 0.36 | 3.99 ± 0.9 | 0.84 ± 0.02 | 38.19 ± 1.11 |
| Th | 62.35 ± 0.45 | 4.46 ± 0.12 | 0.86 ± 0.06 | 39.36 ± 1.36 |
3.3. Characteristics of Residual Solids and Bioenergy Potential
| Parameters | Pcon | Ch | Th | TCh |
|---|---|---|---|---|
| Carbon | 41.86 ± 0.81 | 43.02 ± 0.32 | 42.27 ± 0.28 | 44.13 ± 0.31 |
| Nitrogen | 2.68 ± 0.22 | 2.83 ± 0.18 | 2.29 ± 0.26 | 3.06 ± 0.31 |
| Hydrogen | 6.41 ± 0.23 | 6.4 ± 0.37 | 6.46 ± 0.18 | 6.63 ± 0.09 |
| Sulphur | 0.12 ± 0.02 | 0.14 ± 0.01 | <0.1 | 0.19 ± 0.01 |
| Oxygen | 48.95 ± 1.11 | 47.62 ± 0.59 | 48.99 ± 0.83 | 46 ± 1.21 |
| C/N ratio | 15.62 ± 0.58 | 15.23 ± 0.67 | 18.46 ± 0.38 | 14.42 ± 0.79 |
| Total solids | 74.13 ± 2.71 | 75.92 ± 1.63 | 74.68 ± 1.42 | 75.15 ± 2.35 |
| Volatile solids | 96.76 ± 0.97 | 95.75 ± 1.25 | 95.97 ± 1.54 | 97.1 ± 1.80 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cherubinia, F.; Ulgiatib, S. Crop residues as raw materials for biorefinery systems—A LCA case study. Appl. Energy 2010, 87, 47–57. [Google Scholar] [CrossRef]
- Demirbas, M.F. Biorefineries for biofuel upgrading: A critical review. Appl. Energy 2009, 86, S151–S161. [Google Scholar] [CrossRef]
- Sadhukhan, J.; Ng, K.S.; Hernandez, E.M. Biorefineries and Chemical Processes: Design, Integration and Sustainability Analysis; Wiley: Chichester, West Sussex, UK, 2014. [Google Scholar]
- Wright, M.M.; Brown, R.C. Comparative economics of biorefi neries based on the biochemical and thermochemical platforms. Biofuel. Bioprod. Bior. 2007, 1, 49–56. [Google Scholar] [CrossRef]
- Menon, V.; Rao, M. Trends in bioconversion of lignocellulose: Biofuels, platform chemicals & biorefinery concept. Prog. Energy Combust. Sci. 2012, 38, 522–550. [Google Scholar]
- Kamm, B.; Gruber, P.R.; Kamm, M. Biorefineries-Industrial Processes and Products: Status quo and Future Directions; Wiley-VCH: Weinheim, Germany, 2006. [Google Scholar]
- Parfitt, J.; Barthel, M.; Macnaughton, S. Food waste within food supply chains: Quantification and potential for change to 2050. Philos. T. R. Soc. B 2010, 365, 3065–3081. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.J. Production of bioenergy and bioproducts from food processing wastes: A review. T. Am. Soc. Agr. Biol. Eng. 2013, 56, 217–229. [Google Scholar]
- Smith, K.; O'Farrel, K.; Brindley, F. Waste and recycling in Australia 2011; Department of Sustainability, Environment, Water, Population and Communities: Sydney, Australia, 2012. [Google Scholar]
- Grosso, M.; Motta, A.; Rigamonti, L. Efficiency of energy recovery from waste incineration, in the light of the new Waste Framework Directive. Waste Manage. 2010, 30, 1238–1243. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; McDonald, A.G.; Coats, E.R. Lactic acid production with undefined mixed culture fermentation of potato peel waste. Waste Manage. 2014, 34, 2022–2027. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Li, M.; Qi, Q.; Gao, C.; Lin, C.S. Mixed food waste as renewable feedstock in succinic acid fermentation. Appl. Biochem. Biotechnol. 2014, 174, 1822–1833. [Google Scholar] [CrossRef] [PubMed]
- Koutinas, A.A.; Vlysidis, A.; Pleissner, D.; Kopsahelis, N.; Lopez Garcia, I.; Kookos, I.K.; Papanikolaou, S.; Kwan, T.H.; Lin, C.S. Valorization of industrial waste and by-product streams via fermentation for the production of chemicals and biopolymers. Chem. Soc. Rev. 2014, 43, 2587–2627. [Google Scholar] [CrossRef] [PubMed]
- Cheirsilp, B.; Suwammarat, W.; Niyomdecha, R. Mixed culture of oleaginous yeast Rhodotorula glutinis and microalga Chlorella vulgaris for lipid production from industrial wastes and its use as biodiesel feedstock. New Biotechnol. 2011, 28, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Saengea, C.; Cheirsilpb, B.; Suksarogea, T.T.; Bourtoomc, T. Potential use of oleaginous red yeast Rhodotorula glutinis for the bioconversion of crude glycerol from biodiesel plant to lipids and carotenoids. Proc. Biochem. 2011, 46, 210–218. [Google Scholar] [CrossRef]
- Schneider, T.; Graeff-Hönninger, S.; French, W.T.; Hernandez, R.; Claupein, W.; Holmes, W.E.; Merkt, N. Screening of Industrial Wastewaters as Feedstock for the Microbial Production of Oils for Biodiesel Production and High-Quality Pigments. J. Combust. 2012, 153410:1–153410:9. [Google Scholar] [CrossRef]
- Hu, C.; Zhao, X.; Zhao, J.; Wu, S.; Zhao, Z.K. Effects of biomass hydrolysis by-products on oleaginous yeast Rhodosporidium toruloides. Bioresource Technol. 2009, 4843–4847. [Google Scholar] [CrossRef] [PubMed]
- Ratledge, C. Yeasts, moulds, algae and bacteria as sources of lipids. In Technological Advances in Improved and Alternative Sources of Lipids; Kamel, B.S., Kakuda, Y., Eds.; Blackie Academic and Professional: London, UK, 1994. [Google Scholar]
- Beopoulos, A.; Desfougeres, T.; Sabirova, J.; Zinjarde, S.; Neuveglise, C.; Nicaud, J.M. The hydrocarbon-degrading oleaginous yeast Yarrowia lipolytica. In Handbook of Hydrocarbon and Lipid Microbiology; Timmis, K.N., Ed.; Springer: New York, NY, USA, 2009. [Google Scholar]
- Chi, Z.; Zheng, Y.; Jiang, A.; Chen, S. Lipid production by culturing oleaginous yeast and algae with food waste and municipal wastewater in an integrated process. Appl. Biochem. Biotechnol. 2011, 165, 442–453. [Google Scholar] [CrossRef] [PubMed]
- Xue, F.; Gao, B.; Zhu, Y.; Zhangm, X.; Feng, W.; Tan, T. Pilot-scale production of microbial lipid using starch wastewater as raw material. Bioresource Technol. 2010, 101, 6092–6095. [Google Scholar] [CrossRef] [PubMed]
- Beopoulos, A.; Nicaud, J.M.; Gaillardin, C. An overview of lipid metabolism in yeasts and its impact on biotechnological processes. Appl. Microbiol. Biotechnol. 2011, 90, 1193–1206. [Google Scholar] [CrossRef] [PubMed]
- Braunwald, T.; Schwemmlein, L.; Graeff-Hönninger, S.; French, W.T.; Hernandez, R.; Holmes, W.E.; Claupain, W. Effect of different C/N ratios on carotenoid and lipid production by Rhodotorula glutinis. Appl. Microbiol. Biotechnol. 2013, 97, 6581–6588. [Google Scholar] [CrossRef] [PubMed]
- Saenge, C.; Cheirsilp, B.; Suksaroge, T.T.; Bourtoom, T. Effificent concomitant production of lipids and carotenoids by oleaginous red yeast Rhodotorula glutinis cultured in palm oil mill effluent and application of lipids for biodiesel production. Biotechnol. Bioproc. Eng. 2011, 16, 23–33. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Organization: Washington DC, USA, 2005. [Google Scholar]
- Albalasmeh, A.A.; Berhe, A.A.; Ghezzehei, T.A. A new method for rapid determination of carbohydrate and total carbon concentrations using UV spectrophotometry. Carbohydr. Polym. 2013, 97, 253–261. [Google Scholar] [CrossRef] [PubMed]
- von Alvensleben, N.; Stookey, K.; Magnusson, M.; Heimann, K. Salinity tolerance of Picochlorum atomus and the use of salinity for contamination control by the freshwater cyanobacterium Pseudanabaena limnetica. PLoS ONE 2013, 8, e63569. [Google Scholar] [CrossRef] [PubMed]
- Davoli, P.; Mierau, V.; Weber, R.W.S. Carotenoids and fatty acids in red yeasts Sporobolomyces roseus and Rhodotorula glutinis. Appl. Biochem. Microbiol. 2004, 40, 392–397. [Google Scholar] [CrossRef]
- Easterling, E.R.; French, W.T.; Hernandez, R.; Licha, M. The effect of glycerol as a sole and secondary substrate on the growth and fatty acid composition of Rhodotorula glutinis. Bioresource Technol. 2009, 100, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.A.; Magnusson, M.; Brown, R.J.; Ayoko, G.A.; Nabi, M.N.; Heimann, K. Microalgal species selection for biodiesel production based on fuel properties derived from fatty acid profiles. Energies 2013, 6, 5676–5702. [Google Scholar] [CrossRef]
- Ramírez-Verduzco, L.F.; Rodríguez-Rodríguez, J.E.; Jaramillo-Jacob, A.R. Predicting cetane number, kinematic viscosity, density and higher heating value of biodiesel from its fatty acid methyl ester composition. Fuel 2012, 91, 102–111. [Google Scholar] [CrossRef]
- Roati, C.; Fiore, S.; Ruffino, B.; Marchese, F.; Novarino, D.; Zanetti, M.C. Preliminary evaluation of the potential biogas production of food-processing industrial wastes. Am. J. Environ. Sci. 2012, 8, 291–296. [Google Scholar]
- Angelidaki, I.; Alves, M.; Bolzonella, D.; Borzacconi, L.; Campos, J.L.; Guwy, A.J. Defining the biomethane potential (BMP) of solid organic wastes and energy crops: A proposed protocol for batch assays. Water Sci. Technol. 2009, 59, 927–934. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, D. Evaluation of pre-treatment processes for increasing biodegradability of agro-food waste. Environ. Technol. 2012, 33, 1497–1503. [Google Scholar] [CrossRef] [PubMed]
- Velmurugan, B.; Ramanujam, R.A. Anaerobic digestion of vegetable wastes for biogas production in a fed-batch reactor. Int. J. Emerg. Sci. 2011, 1, 478–486. [Google Scholar]
- Li, J.H.; Vasathan, T.; Rossnagel, B.; Hoover, R. Starch from hull-less barley: II. Thermal, rheological and acid hydrolysis characteristics. Food Chem. 2001, 74, 407–415. [Google Scholar] [CrossRef]
- Ariunbaatar, J.; Panico, A.; Esposito, G.; Pirozzi, F.; Lens, P.N.L. Pretreatment methods to enhance anaerobic digestion of organic solid waste. Appl. Energy 2014, 123, 143–156. [Google Scholar] [CrossRef]
- Li, Y.H.; Liu, B.; Sun, Y.; Zhao, Z.B.; Bai, F.W. Screening of oleaginous yeasts for broad-spectrum carbohydrates assimilating capacity. Chin. J. Biotechnol. 2005, 25, 39–43. [Google Scholar]
- Sahu, A.; Pancha, I.; Jain, D.; Paliwal, C.; Ghosh, G.; Patidar, S.; Bhattacharya, S.; Mishra, S. Fatty acids as biomarkers of microalgae. Phytochemistry 2013, 89, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Berner, F.; Heimann, K.; Sheehan, M. Microalgasl biofilms for biomass production. J. Appl. Phycol. 2014. [Google Scholar] [CrossRef]
- Papanikolaou, S.; Aggelis, G. Lipids of oleaginous yeast. Part I. Biochemistry related with single cell oil production. Eur. J. Lipid Sci. Technol. 2011, 113, 1031–1051. [Google Scholar] [CrossRef]
- Xue, F.; Miao, J.; Zhang, X.; Luo, H.; Tan, T. Studies on lipid production by Rhodotorula glutinis fermentation using monosodium glutamate wastewater as culture medium. Bioresource Technol. 2008, 99, 5923–5927. [Google Scholar] [CrossRef] [PubMed]
- Schneider, T.; Graeff-Hönninger, S.; French, W.T.; Hernandez, R.; Merkt, N.; Claupein, W.; Hetrick, M.; Pham, P. Lipid and carotenoid production by oleaginous red yeast Rhodotorula glutinis cultivated on brewery effluents. Energy 2013, 61, 34–43. [Google Scholar] [CrossRef]
- Dai, C.C.; Tao, J.; Xie, F.; Dai, Y.J.; Zhao, M. Biodiesel generation from oleaginous yeast Rhodotorula glutinis with xylose assimilating capacity. Afr. J. Biochem. Res. 2007, 6, 2130–2134. [Google Scholar]
- Lin, L.; Zhou, C.; Vittayapadung, S.; Xiangqian, S.; Mingdong, D. Opportunities and challenges for biodiesel fuel. Appl. Energies 2011, 88, 1020–1031. [Google Scholar] [CrossRef]
- Yu, X.; Zheng, Y.; Dorgan, K.; Chen, S. Oil production by oleaginous yeasts using the hydrolysate from pretreatment of wheat straw with dilute sulfuric acid. Bioresource Technol. 2011, 102, 6134–6140. [Google Scholar] [CrossRef] [PubMed]
- Knothe, G. Fuel properties of highly polyunsaturated fatty acid methyl esters. Prediction of fuel properties of algal biodiesel. Energy Fuel 2012, 26, 5265–5273. [Google Scholar] [CrossRef]
- de Silva, M.J.; de Souza, S.N.M.; Chaves, L.I.; Rosa, H.A.; Secco, D.; Santos, R.F.; Barricatti, R.A.; Nogueira, C.E.C. Comparative analysis of engine generator performance using diesel oil and biodiesels available in Paraná State, Brazil. Renew. Sustain. Energy Rev. 2013, 17, 278–282. [Google Scholar] [CrossRef]
- Karthikeyan, O.P.; Visvanatha, C. Bio-energy recovery from high-solid organic substrates by dry anaerobic bio-conversion processes: A review. Rev. Environ. Sci. Bio/Technol. 2013, 12, 257–284. [Google Scholar] [CrossRef]
- Strong, P.J.; McDonald, B.; Gapes, D.J. Combined thermochemical and fermentative destruction of municipal biosolids: A comparison between thermal hydrolysis and wet oxidative pre-treatment. Bioresource Technol. 2010, 102, 5520–5527. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hao, H.-T.N.; Karthikeyan, O.P.; Heimann, K. Bio-Refining of Carbohydrate-Rich Food Waste for Biofuels. Energies 2015, 8, 6350-6364. https://doi.org/10.3390/en8076350
Hao H-TN, Karthikeyan OP, Heimann K. Bio-Refining of Carbohydrate-Rich Food Waste for Biofuels. Energies. 2015; 8(7):6350-6364. https://doi.org/10.3390/en8076350
Chicago/Turabian StyleHao, Hoang-Tuong Nguyen, Obulisamy Parthiba Karthikeyan, and Kirsten Heimann. 2015. "Bio-Refining of Carbohydrate-Rich Food Waste for Biofuels" Energies 8, no. 7: 6350-6364. https://doi.org/10.3390/en8076350
APA StyleHao, H.-T. N., Karthikeyan, O. P., & Heimann, K. (2015). Bio-Refining of Carbohydrate-Rich Food Waste for Biofuels. Energies, 8(7), 6350-6364. https://doi.org/10.3390/en8076350





