Engineering Plant Biomass Lignin Content and Composition for Biofuels and Bioproducts
Abstract
:1. Introduction
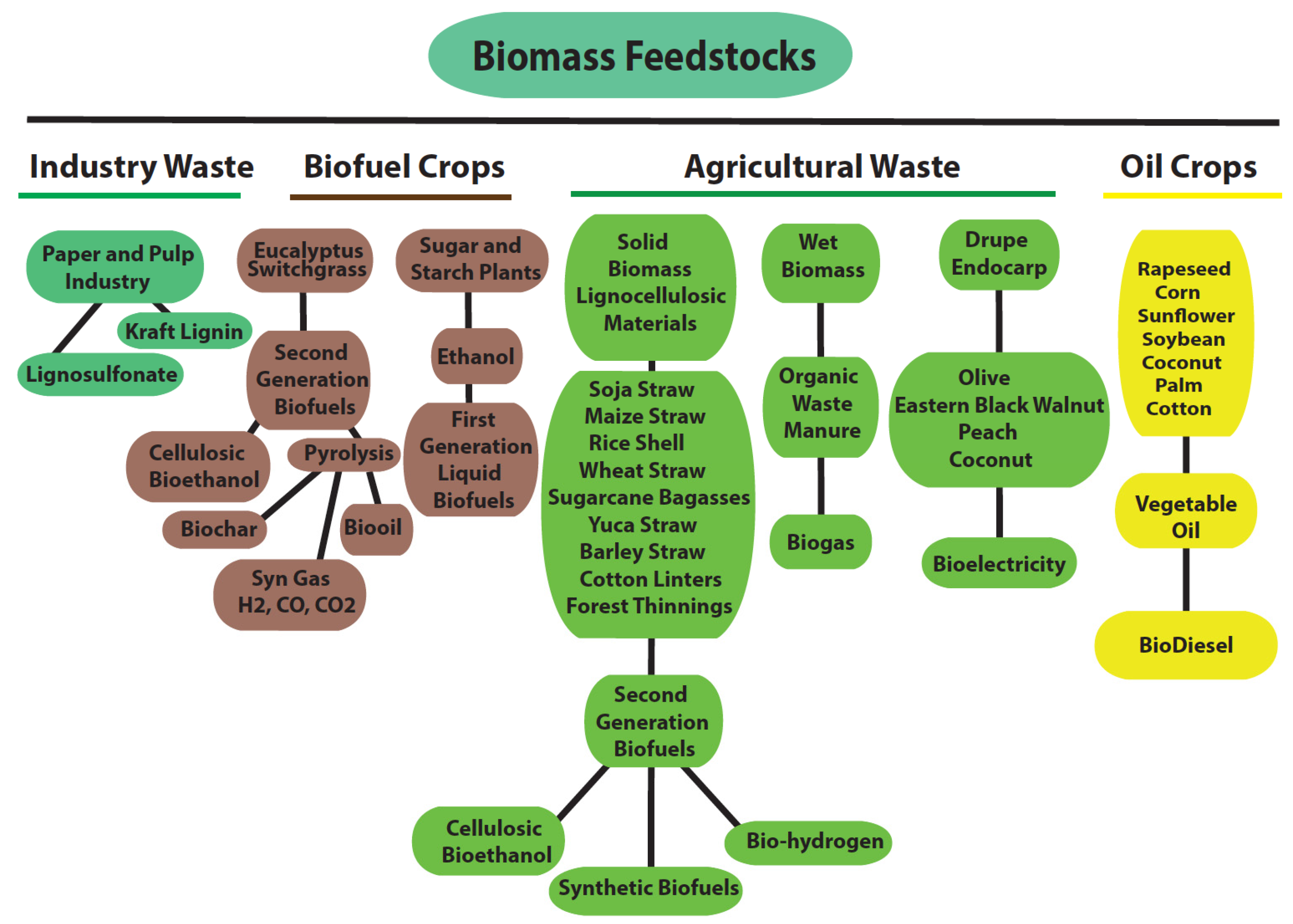
2. Lignocellulosic Biomass, the Focus of Second-Generation Biofuels
3. Lignin Biosynthesis and Composition
| Division | Lignin Occurrence and Composition | Interspecies Variation | Cell Wall Composition (%) | References | ||
|---|---|---|---|---|---|---|
| Cellulose | Hemicellulose | Lignin | ||||
| Algae | Vary in the composition of G, H and S subunits over time | Though lignin is not present in algae, some exceptions exist:
| ~70 | ~43 | NA | [32,33,34,35,36] |
| Bryophytes | Mostly intermediate compounds like 1,3,5-hydroxybenzene | Most bryophytes lack lignin
| NA | NA | NA | [32,37,38] |
| Pteridophytes | Present in the vascular system (tracheary elements). Mostly G and H subunits and rarely S subunits | High variation is seen in S subunit composition:
| ~50 | NA | ~14 | [32,39,40,41] |
| Gymnosperms | Present in the vascular system (tracheary elements). Mostly G and H subunits and lesser amounts of S subunits | High variation is seen in S subunit composition:
| NA | NA | ~30 | [32,39,42] |
| Angiosperms (Monocots) | Present in vascular system (tracheary element and fibers). Significant amounts of all three G, S and H subunits | Variation is seen in phenolic acid content of lignin:
| 20–45 | 20–50 | 20 | [32,43,44,45] |
| Angiosperms (Dicots) | Present in vascular system (tracheary element and fibers). Mostly G and S subunits and minor level of H subunits | Varies between 10%–20% in different species | 15–50 | 5–30 | 7–10 | [32,34,45] |
3.1. Lignin Composition and Structure Analysis
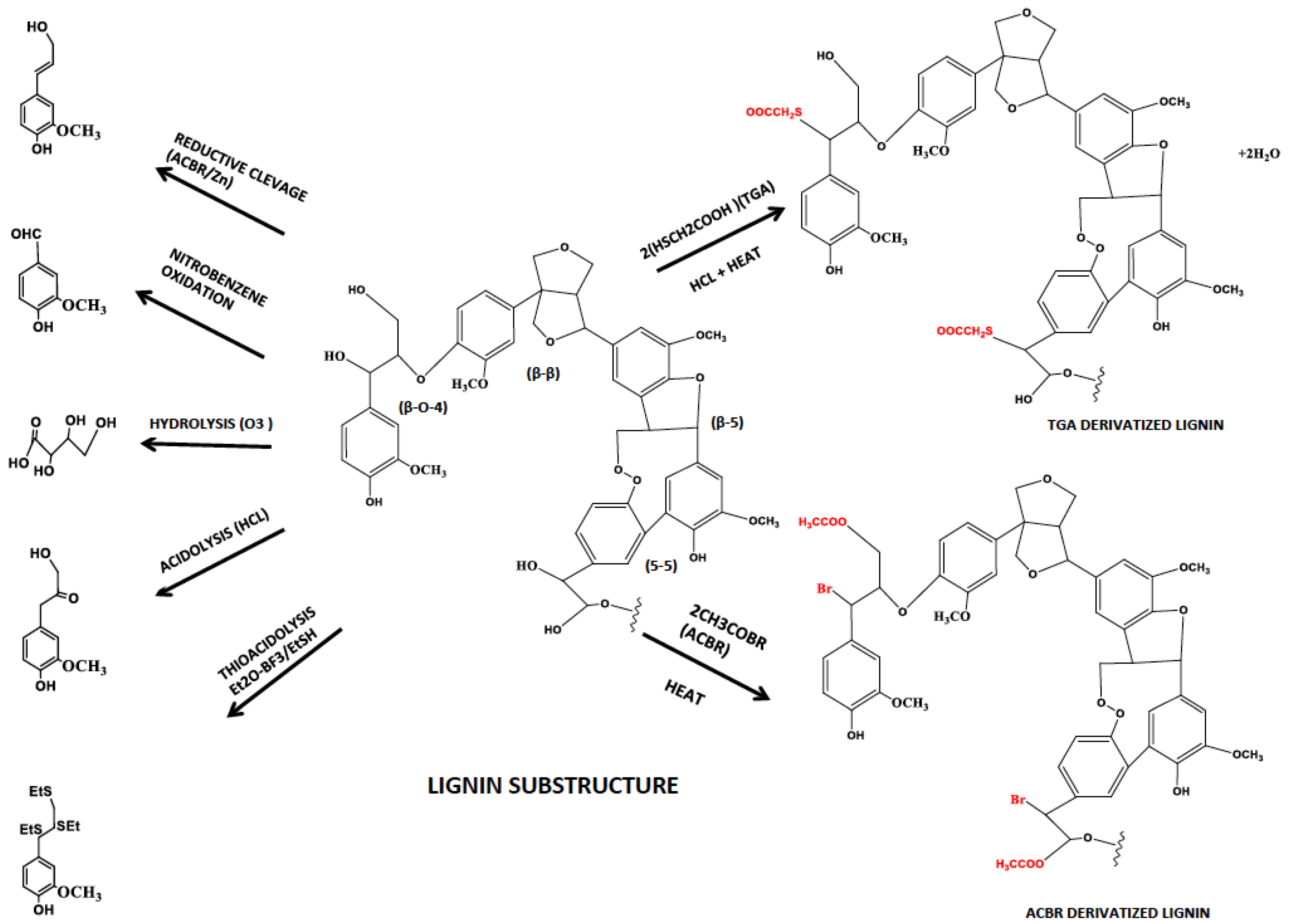
3.1.1. Degradative Analytical Techniques (Oxidation, Reduction, Hydrolysis and Acidolysis)
3.1.2. Non-Degradative Analytical Techniques (Thioglycolic Acid–TGA and Acetyl Bromide–ACBR)
| Derivatization Method | Targeted Substrate | Product | Reference |
|---|---|---|---|
| Nitrobenzene oxidation | Side chains of phenyl ring | Breaks side chains and leaves aromatic ring | [54] |
| Derivatization followed by Reductive Cleavage | α- & β-aryl ether bonds | 4-acetoxy cinnamylacetate Coniferyl diacetate Sinapyl diacetate | [57] |
| Ozonolysis | Unsaturated bonds of phenyl ring | Carboxylic acid | [55] |
| Acidolysis | Aryl glycerol-β-aryl ether | Single ring aromatic compounds | [56] |
| Thio acidolysis | Aryl glycerol-β-aryl ether | Single ring aromatic compounds & dimers with 5-5 & 5-O-4 links | [56] |
| ThioGlycolic Acid | β-aryl ether bonds | Thioglycolic group substituted lignin polymer | [58] |
| Acetyl bromide | α- & β-aryl ether bonds | Acetyl bromide group substituted lignin polymer | [59] |
4. Lignin Bioengineering for Efficient Utilization of Plant Biomass
4.1. Reducing the Lignin Content/Altering the Composition for Easy Separation of Polysaccharides
4.2. Enhancing Lignin Content to Produce High Energy Dense Feedstocks
5. Separation of Lignin from Other Polymers in Plant Biomass (Chemical and Biological)
5.1. Chemical-Based Lignin Separation
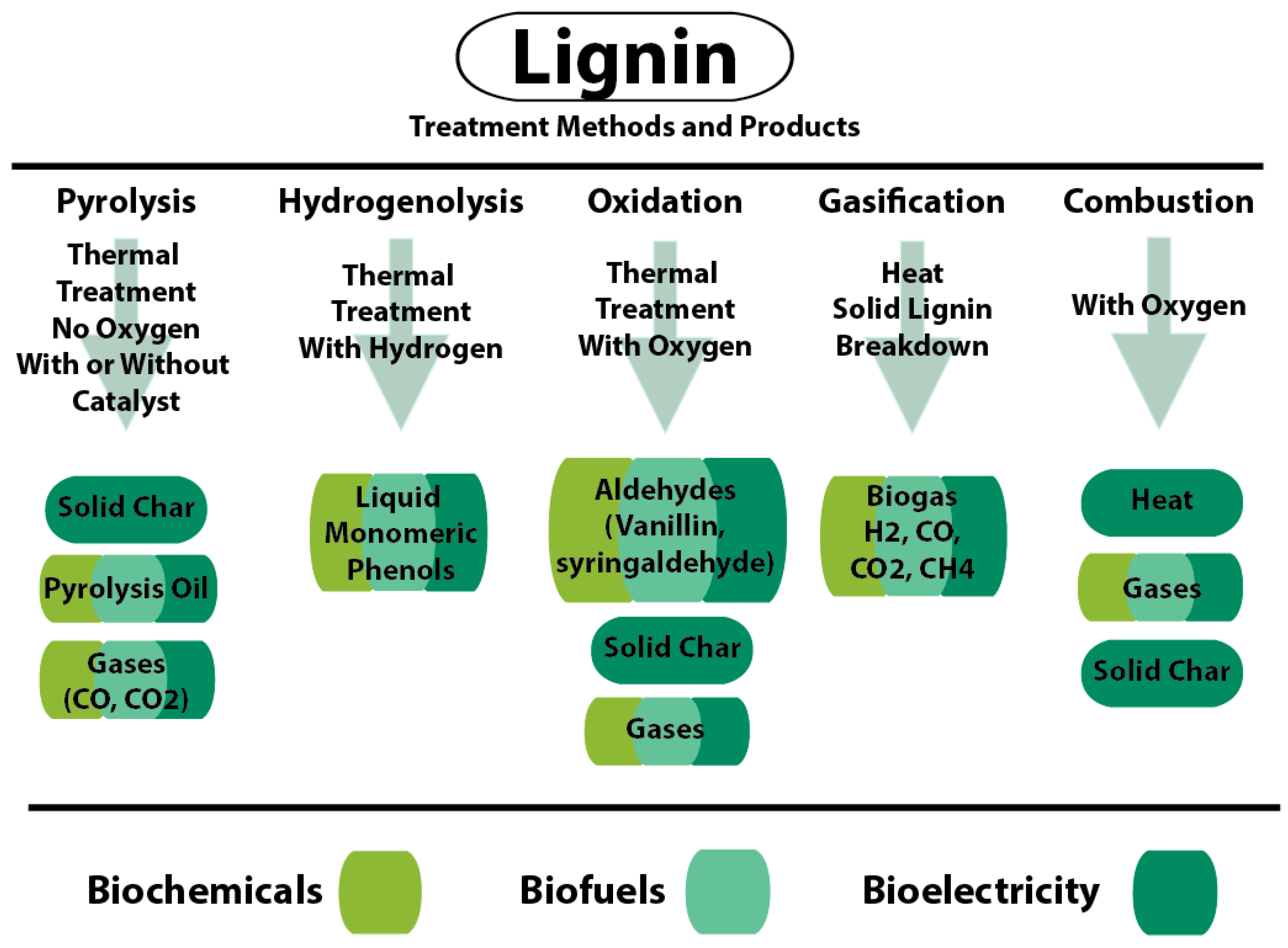
5.2. Biological Degradation of Lignin
6. Applications of Lignin for Biofuel and Bioproducts
7. Conclusions
Acknowledgments
Conflict of interest
References
- Alexandratos, N.; Bruinsm, J. World Agriculture towards 2030/2050: The 2012 Revision; ESA working paper No: 12-03; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2012. [Google Scholar]
- Alexandratos, N. The Mediterranean diet in a world context. Public Health Nutr. 2006, 9, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Perlack, R.D.; Wright, L.L.; Turhollow, A.F.; Graham, R.L.; Stokes, B.J.; Erbach, D.C. Biomass as Feedstock for a Bioenergy and Bioproducts Industry: The Technical Feasibility of a Billion-Ton Annual Supply; Technical Report; Oak Ridge National Laboratory (ORNL): Oak Ridge, TN, USA, 2005. [Google Scholar]
- Sorek, N.; Yeats, T.H.; Szemenyei, H.; Youngs, H.; Somerville, C.R. The Implications of Lignocellulosic Biomass Chemical Composition for the Production of Advanced Biofuels. BioScience 2014, 64. [Google Scholar] [CrossRef]
- van Duren, I.; Voinov, A.; Arodudu, O.; Firrisa, M.T. Where to produce rapeseed biodiesel and why? Mapping European rapeseed energy efficiency. Renew. Energy 2015, 74, 49–59. [Google Scholar] [CrossRef]
- Doherty, W.O.S.; Mousavioun, P.; Fellows, C.M. Value-adding to cellulosic ethanol: Lignin polymers. Industrial Crop. Prod. 2011, 33, 259–276. [Google Scholar] [CrossRef] [Green Version]
- Searchinger, T.; Edwards, R.; Mulligan, D.; Heimlich, R.; Plevin, R. Do biofuel policies seek to cut emissions by cutting food? Science 2015, 347, 1420–1422. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, D.; Burgess, M. Biofuel production using food. In Environment, Development and Sustainability; Springer: Berlin, Germany, 2014; Volume 16, pp. 1–3. [Google Scholar]
- Galbe, M.; Zacchi, G. Pretreatment of lignocellulosic materials for efficient bioethanol production. Adv. Biochem. Eng. Biotechnol. 2007, 108, 41–65. [Google Scholar] [PubMed]
- Mendu, V.; Shearin, T.; Campbell, J.E.; Stork, J.; Jae, J.; Crocker, M.; Huber, G.; DeBolt, S. Global bioenergy potential from high-lignin agricultural residue. Proc. Natl. Acad. Sci. 2012, 109, 4014–4019. [Google Scholar] [CrossRef] [PubMed]
- Stigka, E.K.; Paravantis, J.A.; Mihalakakou, G.K. Social acceptance of renewable energy sources: A review of contingent valuation applications. Renew. Sustain. Energy Rev. 2014, 32, 100–106. [Google Scholar] [CrossRef]
- Sticklen, M.B. Plant genetic engineering for biofuel production: towards affordable cellulosic ethanol. Nat. Rev. Genet. 2008, 9, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Kajikawa, Y.; Takeda, Y. Structure of research on biomass and bio-fuels: A citation-based approach. Technol. Forecast. Soc. Chang. 2008, 75, 1349–1359. [Google Scholar] [CrossRef]
- Lark, T.J.; Salmon, J.M.; Gibbs, H.K. Cropland expansion outpaces agricultural and biofuel policies in the United States. Environ. Res. Lett. 2015, 10, 044003. [Google Scholar] [CrossRef]
- Qin, Z.; Zhuang, Q.; Zhu, X.; Cai, X.; Zhang, X. Carbon Consequences and Agricultural Implications of Growing Biofuel Crops on Marginal Agricultural Lands in China. Environ. Sci. Technol. 2011, 45, 10765–10772. [Google Scholar] [CrossRef] [PubMed]
- Mendu, V.; Harman-Ware, A.E.; Crocker, M.; Jae, J.; Stork, J.; Morton, S., 3rd; Placido, A.; Huber, G.; Debolt, S. Identification and thermochemical analysis of high-lignin feedstocks for biofuel and biochemical production. Biotechnol. Biofuels 2011, 4. [Google Scholar] [CrossRef] [PubMed]
- McKendry, P. Energy production from biomass (part 2): Conversion technologies. Bioresour. Technol. 2002, 83, 47–54. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K. Recent advances in green hydrogels from lignin: a review. Int. J. Biol. Macromol. 2015, 72, 834–847. [Google Scholar] [CrossRef] [PubMed]
- Sluiter, J.B.; Ruiz, R.O.; Scarlata, C.J.; Sluiter, A.D.; Templeton, D.W. Compositional Analysis of Lignocellulosic Feedstocks. 1. Review and Description of Methods. J. Agric. Food Chem. 2010, 58, 9043–9053. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Owens, V.N.; Boe, A.; Jeranyama, P. Composition of Herbaceous Biomass Feedstocks; North Central Sun Grant Center, South Dakota State University: Brookings, SD, USA, 2007; pp. 1–16. [Google Scholar]
- Pandey, M.P.; Kim, C.S. Lignin Depolymerization and Conversion: A Review of Thermochemical Methods. Chem. Eng. Technol. 2011, 34, 29–41. [Google Scholar] [CrossRef]
- Klinke, H.B.; Thomsen, A.B.; Ahring, B.K. Inhibition of ethanol-producing yeast and bacteria by degradation products produced during pre-treatment of biomass. Appl. Microbiol. Biotechnol. 2004, 66, 10–26. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Arancon, R.A.D.; Labidi, J.; Luque, R. Lignin depolymerisation strategies: towards valuable chemicals and fuels. Chem. Soc. Rev. 2014, 43, 7485–7500. [Google Scholar] [CrossRef] [PubMed]
- Nawrath, C.; Schreiber, L.; Franke, R.B.; Geldner, N.; Reina-Pinto, J.J.; Kunst, L. Apoplastic Diffusion Barriers in Arabidopsis. Arab. Book 2013, 11. [Google Scholar] [CrossRef] [PubMed]
- Boyce, C.K.; Zwieniecki, M.A.; Cody, G.D.; Jacobsen, C.; Wirick, S.; Knoll, A.H.; Holbrook, N.M. Evolution of xylem lignification and hydrogel transport regulation. Proc. Natl. Acad. Sci. USA 2004, 101, 17555–17558. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, N.H.; Selvaraj, G.; Wei, Y.; King, J. Role of lignification in plant defense. Plant Signal. Behav. 2009, 4, 158–159. [Google Scholar] [CrossRef] [PubMed]
- Liljegren, S.J.; Ditta, G.S.; Eshed, Y.; Savidge, B.; Bowman, J.L.; Yanofsky, M.F. SHATTERPROOF MADS-box genes control seed dispersal in Arabidopsis. Nature 2000, 404, 766–770. [Google Scholar] [PubMed]
- Weng, J.K.; Chapple, C. The origin and evolution of lignin biosynthesis. New Phytol. 2010, 187, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Uzal, E.N.; Gomez Ros, L.V.; Pomar, F.; Bernal, M.A.; Paradela, A.; Albar, J.P.; Ros Barcelo, A. The presence of sinapyl lignin in Ginkgo biloba cell cultures changes our views of the evolution of lignin biosynthesis. Physiol. Plant. 2009, 135, 196–213. [Google Scholar] [CrossRef] [PubMed]
- Boerjan, W.; Ralph, J.; Baucher, M. Lignin biosynthesis. Annu. Rev. Plant Biol. 2003, 54, 519–546. [Google Scholar]
- Ragauskas, A.J.; Beckham, G.T.; Biddy, M.J.; Chandra, R.; Chen, F.; Davis, M.F.; Davison, B.H.; Dixon, R.A.; Gilna, P.; Keller, M.; et al. Lignin valorization: Improving lignin processing in the biorefinery. Science 2014, 344. [Google Scholar] [CrossRef] [PubMed]
- Lewis, N.G.; Yamamoto, E. Lignin: Occurrence, biogenesis and biodegradation. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1990, 41, 455–496. [Google Scholar] [CrossRef] [PubMed]
- Martone, P.T.; Estevez, J.M.; Lu, F.; Ruel, K.; Denny, M.W.; Somerville, C.; Ralph, J. Discovery of lignin in seaweed reveals convergent evolution of cell-wall architecture. Curr. Biol. 2009, 19, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Woolhouse, H.W. Advances in Botanical Research; Academic Press: Waltham, MA, USA, 1981; Volume 8, p. 285. [Google Scholar]
- Estevez, J.M.; Leonardi, P.I.; Alberghina, J.S. Cell wall carbohydrate epitopes in the green alga Oedogonium bharuchae F. minor (oedogoniales, chlorophyta). . J. Phycol. 2008, 44, 1257–1268. [Google Scholar] [CrossRef]
- Kloareg, B.; Quatrano, R.S. Structure of the cell walls of marine algae and ecophysiological functions of the matrix polysaccharides. Oceanogr. Mar. Biol. Annu. Rev. 1988, 26, 259–315. [Google Scholar]
- Wilson, M.A.; Sawyer, J.; Hatcher, P.G.; Lerch, H.E., III. 1,3,5-Hydroxybenzene structures in mosses. Phytochemistry 1989, 28, 1395–1400. [Google Scholar] [CrossRef]
- Siegel, S.M. Evidence for the Presence of Lignin in Moss Gametophytes. Am. J. Bot. 1969, 56, 175–179. [Google Scholar] [CrossRef]
- Ros, L.V.G.; Gabaldón, C.; Pomar, F.; Merino, F.; Pedreño, M.A.; Barceló, A.R. Structural motifs of syringyl peroxidases predate not only the gymnosperm–angiosperm divergence but also the radiation of tracheophytes. New Phytol. 2007, 173, 63–78. [Google Scholar] [CrossRef] [PubMed]
- Silva, G.B.; Ionashiro, M.; Carrara, T.B.; Crivellari, A.C.; Tiné, M.A.S.; Prado, J.; Carpita, N.C.; Buckeridge, M.S. Cell wall polysaccharides from fern leaves: Evidence for a mannan-rich Type III cell wall in Adiantum raddianum. Phytochemistry 2011, 72, 2352–2360. [Google Scholar] [CrossRef] [PubMed]
- Moozhiyil, M.; Pallauf, J. Chemical composition of the water fern, salvinia molesta, and its potential as feed source for ruminants. Econ. Bot. 1986, 40, 375–383. [Google Scholar] [CrossRef]
- Jouanin, L.; Lapierre, C. Lignins: Biosynthesis, Biodegradation and Bioengineering. In Advances in Botanical Research; Academic Press: Waltham, MA, USA, 2012; Volume 61, pp. 2–391. [Google Scholar]
- Ralph, J.; Hatfield, R.D.; Quideau, S.; Helm, R.F.; Grabber, J.H.; Jung, H.-J.G. Pathway of p-Coumaric Acid Incorporation into Maize Lignin as Revealed by NMR. J. Am. Chem. Soc. 1994, 116, 9448–9456. [Google Scholar] [CrossRef]
- Saulnier, L.; Crépeau, M.-J.; Lahaye, M.; Thibault, J.-F.; Garcia-Conesa, M.T.; Kroon, P.A.; Williamson, G. Isolation and structural determination of two 5,5′-diferuloyl oligosaccharides indicate that maize heteroxylans are covalently cross-linked by oxidatively coupled ferulates. Carbohydr. Res. 1999, 320, 82–92. [Google Scholar] [CrossRef]
- Vogel, J. Unique aspects of the grass cell wall. Curr. Opin. Plant Biol. 2008, 11, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Alejandro, S.; Lee, Y.; Tohge, T.; Sudre, D.; Osorio, S.; Park, J.; Bovet, L.; Lee, Y.; Geldner, N.; Fernie, A.R.; et al. AtABCG29 is a monolignol transporter involved in lignin biosynthesis. Curr. Biol. 2012, 22, 1207–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eudes, A.; Liang, Y.; Mitra, P.; Loque, D. Lignin bioengineering. Curr. Opin. Biotechnol. 2014, 26, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Terashima, N.; Yoshida, M.; Hafrén, J.; Fukushima, K.; Westermark, U. Proposed supramolecular structure of lignin in softwood tracheid compound middle lamella regions. In Holzforschung; Walter de Gruyter: Berlin, Germany, 2012; Volume 66, pp. 907–915. [Google Scholar]
- Giardina, P.; Sannia, G. Laccases: Old enzymes with a promising future. Cell. Mol. Life Sci. 2015, 72, 855–856. [Google Scholar] [CrossRef] [PubMed]
- Berthet, S.; Demont-Caulet, N.; Pollet, B.; Bidzinski, P.; Cezard, L.; Le Bris, P.; Borrega, N.; Herve, J.; Blondet, E.; Balzergue, S.; et al. Disruption of LACCASE4 and 17 results in tissue-specific alterations to lignification of Arabidopsis thaliana stems. Plant. Cell. 2011, 23, 1124–1137. [Google Scholar] [CrossRef] [PubMed]
- Schuetz, M.; Benske, A.; Smith, R.A.; Watanabe, Y.; Tobimatsu, Y.; Ralph, J.; Demura, T.; Ellis, B.; Samuels, A.L. Laccases direct lignification in the discrete secondary cell wall domains of protoxylem. Plant Physiol. 2014, 166, 798–807. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Bouchabké-Coussa, O.; Le Bris, P.; Antelme, S.; Soulhat, C.; Gineau, E.; Dalmais, M.; Bendahmane, A.; Morin, H.; Mouille, G.; et al. LACCASE 5 is required for lignification of the Brachypodium distachyon culm. Plant Physiol. 2015, 168, 192–204. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Nakashima, J.; Chen, F.; Yin, Y.; Fu, C.; Yun, J.; Shao, H.; Wang, X.; Wang, Z.-Y.; Dixon, R.A. LACCASE Is Necessary and Nonredundant with PEROXIDASE for Lignin Polymerization during Vascular Development in Arabidopsis. Plant Cell 2013, 25, 3976–3987. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.L. Nitrobenzene and Cupric Oxide Oxidations. In Methods in Lignin Chemistry; Lin, S., Dence, C., Eds.; Springer: Berlin/Heidelberg, Germany, 1992; pp. 301–321. [Google Scholar]
- Sarkanen, K.V.; Islam, A.; Anderson, C.D. Ozonation. In Methods in Lignin Chemistry; Lin, S., Dence, C., Eds.; Springer: Berlin/Heidelberg, Germany, 1992; pp. 387–406. [Google Scholar]
- Lundquist, K.; Kirk, T.K. Acid degradation of lignin. IV. Analysis of lignin acidolysis products by gas chromatography, using trimethylsilyl derivatives. Acta Chem. Scand. 1971, 25, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Fachuang, L.; John, R. Efficient Ether Cleavage in Lignins: The Derivatization Followed by Reductive Cleavage Procedure as a Basis for New Analytical Methods. In Lignin and Lignan Biosynthesis; American Chemical Society: Washington, DC, USA, 1998; Volume 697, pp. 294–322. [Google Scholar]
- Bruce, R.J.; West, C.A. Elicitation of lignin biosynthesis and isoperoxidase activity by pectic fragments in suspension cultures of castor bean. Plant Physiol. 1989, 91, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Morrison, I.M. Improvements in the acetyl bromide technique to determine lignin and digestibility and its application to legumes. J. Sci. Food Agric. 1972, 23, 1463–1469. [Google Scholar] [CrossRef]
- Gidh, A.V.; Decker, S.R.; Vinzant, T.B.; Himmel, M.E.; Williford, C. Determination of lignin by size exclusion chromatography using multi angle laser light scattering. J. Chromatogr. A 2006, 1114, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Jorgenson, L.R. Brown Midrib in Maize and its Linkage Relations. Agron. J. 1931, 23, 549–557. [Google Scholar] [CrossRef]
- Kuc, J.; Nelson, O.E. The abnormal lignins produced by the brown-midrib mutants of maize: I. The brown-midrib-1 mutant. Arch. Biochemi. Biophys. 1964, 105, 103–113. [Google Scholar] [CrossRef]
- Kuc, J.; Nelson, O.E.; Flanagan, P. Degradation of abnormal lignins in the brown-midrib mutants and double mutants of maize. Phytochemistry 1968, 7, 1435–1436. [Google Scholar] [CrossRef]
- Saballos, A.; Vermerris, W.; Rivera, L.; Ejeta, G. Allelic Association, Chemical Characterization and Saccharification Properties of brown midrib Mutants of Sorghum (Sorghum bicolor (L.) Moench). Bioenergy Res. 2008, 1, 193–204. [Google Scholar] [CrossRef]
- Petti, C.; Harman-Ware, A.; Tateno, M.; Kushwaha, R.; Shearer, A.; Downie, A.; Crocker, M.; DeBolt, S. Sorghum mutant RG displays antithetic leaf shoot lignin accumulation resulting in improved stem saccharification properties. Biotechnol. Biofuels 2013, 6. [Google Scholar] [CrossRef] [PubMed]
- Mir Derikvand, M.; Sierra, J.; Ruel, K.; Pollet, B.; Do, C.-T.; Thévenin, J.; Buffard, D.; Jouanin, L.; Lapierre, C. Redirection of the phenylpropanoid pathway to feruloyl malate in Arabidopsis mutants deficient for cinnamoyl-CoA reductase 1. Planta 2008, 227, 943–956. [Google Scholar] [CrossRef] [PubMed]
- Poovaiah, C.R.; Nageswara-Rao, M.; Soneji, J.R.; Baxter, H.L.; Stewart, C.N., Jr. Altered lignin biosynthesis using biotechnology to improve lignocellulosic biofuel feedstocks. Plant Biotechnol. J. 2014, 12, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Franke, R.; McMichael, C.M.; Meyer, K.; Shirley, A.M.; Cusumano, J.C.; Chapple, C. Modified lignin in tobacco and poplar plants over-expressing the Arabidopsis gene encoding ferulate 5-hydroxylase. Plant J. 2000, 22, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Sangha, A.K.; Parks, J.M.; Standaert, R.F.; Ziebell, A.; Davis, M.; Smith, J.C. Radical Coupling Reactions in Lignin Synthesis: A Density Functional Theory Study. J. Phys. Chem. B 2012, 116, 4760–4768. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ximenes, E.; Kim, Y.; Slininger, M.; Meilan, R.; Ladisch, M.; Chapple, C. Lignin monomer composition affects Arabidopsis cell-wall degradability after liquid hot water pretreatment. Biotechnol. Biofuels 2010, 3. [Google Scholar] [CrossRef] [PubMed]
- Sewalt, V.; Ni, W.; Blount, J.W.; Jung, H.G.; Masoud, S.A.; Howles, P.A.; Lamb, C.; Dixon, R.A. Reduced Lignin Content and Altered Lignin Composition in Transgenic Tobacco Down-Regulated in Expression of l-Phenylalanine Ammonia-Lyase or Cinnamate 4-Hydroxylase. Plant Physiol. 1997, 115, 41–50. [Google Scholar] [PubMed]
- Eudes, A.; George, A.; Mukerjee, P.; Kim, J.S.; Pollet, B.; Benke, P.I.; Yang, F.; Mitra, P.; Sun, L.; Cetinkol, O.P.; et al. Biosynthesis and incorporation of side-chain-truncated lignin monomers to reduce lignin polymerization and enhance saccharification. Plant Biotechnol. J. 2012, 10, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Reiss, R.; Ihssen, J.; Richter, M.; Eichhorn, E.; Schilling, B.; Thöny-Meyer, L. Laccase versus Laccase-Like Multi-Copper Oxidase: A Comparative Study of Similar Enzymes with Diverse Substrate Spectra. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Davis, E.J.; Ballif, J.; Liang, M.; Bushman, E.; Haroldsen, V.; Torabinejad, J.; Wu, Y. Mutant identification and characterization of the laccase gene family in Arabidopsis. J. Exp. Bot. 2006, 57, 2563–2569. [Google Scholar] [CrossRef] [PubMed]
- Pourcel, L.; Routaboul, J.-M.; Kerhoas, L.; Caboche, M.; Lepiniec, L.Ø.; Debeaujon, I. TRANSPARENT TESTA10 Encodes a Laccase-Like Enzyme Involved in Oxidative Polymerization of Flavonoids in Arabidopsis Seed Coat. Plant Cell 2005, 17, 2966–2980. [Google Scholar] [CrossRef] [PubMed]
- Liang, M.; Davis, E.; Gardner, D.; Cai, X.; Wu, Y. Involvement of AtLAC15 in lignin synthesis in seeds and in root elongation of Arabidopsis. Planta 2006, 224, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Bhinu, V.S.; Li, R.; Huang, J.; Kaminskyj, S.; Sharpe, A.; Hannoufa, A. Perturbation of lignin biosynthesis pathway in Brassica napus (canola) plants using RNAi. Can. J. Plant Sci. 2009, 89, 441–453. [Google Scholar] [CrossRef]
- Guo, D.; Chen, F.; Inoue, K.; Blount, J.W.; Dixon, R.A. Downregulation of caffeic acid 3-O-methyltransferase and caffeoyl CoA 3-O-methyltransferase in transgenic alfalfa. impacts on lignin structure and implications for the biosynthesis of G and S lignin. Plant Cell 2001, 13, 73–88. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.H.; Kim, H.T.; Hwang, I.; Han, K.H. Tissue-type-specific transcriptome analysis identifies developing xylem-specific promoters in poplar. Plant Biotechnol. J. 2012, 10, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, S.; Mitsuda, N. Reconstitution of a secondary cell wall in a secondary cell wall-deficient Arabidopsis mutant. Plant Cell Physiol. 2015, 56, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Mitra, P.; Zhang, L.; Prak, L.; Verhertbruggen, Y.; Kim, J.S.; Sun, L.; Zheng, K.; Tang, K.; Auer, M.; et al. Engineering secondary cell wall deposition in plants. Plant Biotechnol. J. 2013, 11, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Gaj, T.; Gersbach, C.A.; Barbas, C.F., 3rd. ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol. 2013, 31, 397–405. [Google Scholar] [CrossRef] [PubMed]
- White, R.H. Effect of Lignin Content and Extractives on the Higher Heating Value of Wood; Society of Wood Science and Technology: Madison, WI, USA, 1987; Volume 19, pp. 446–452. [Google Scholar]
- Marsh, G. Biofuels: Aviation alternative? Renew. Energy Focus 2008, 9, 48–51. [Google Scholar] [CrossRef]
- Regalbuto, J.R. Cellulosic Biofuels—Got Gasoline? Science 2009, 325, 822–824. [Google Scholar] [CrossRef] [PubMed]
- Van Acker, R.; Vanholme, R.; Storme, V.; Mortimer, J.C.; Dupree, P.; Boerjan, W. Lignin biosynthesis perturbations affect secondary cell wall composition and saccharification yield in Arabidopsis thaliana. Biotechnol. Biofuels 2013, 6, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cano-Delgado, A.; Penfield, S.; Smith, C.; Catley, M.; Bevan, M. Reduced cellulose synthesis invokes lignification and defense responses in Arabidopsis thaliana. Plant J. 2003, 34, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.A.; Dubos, C.; Surman, C.; Willment, J.; Cullis, I.F.; Mansfield, S.D.; Campbell, M.M. Comparison of lignin deposition in three ectopic lignification mutants. New Phytol. 2005, 168, 123–140. [Google Scholar] [CrossRef] [PubMed]
- Hématy, K.; Sado, P.-E.; Van Tuinen, A.; Rochange, S.; Desnos, T.; Balzergue, S.; Pelletier, S.; Renou, J.-P.; Höfte, H. A Receptor-like Kinase Mediates the Response of Arabidopsis Cells to the Inhibition of Cellulose Synthesis. Curr. Biol. 2007, 17, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Moura, J.C.; Bonine, C.A.; de Oliveira Fernandes Viana, J.; Dornelas, M.C.; Mazzafera, P. Abiotic and biotic stresses and changes in the lignin content and composition in plants. J. Integr. Plant Biol. 2010, 52, 360–376. [Google Scholar] [CrossRef] [PubMed]
- Besseau, S.; Hoffmann, L.; Geoffroy, P.; Lapierre, C.; Pollet, B.; Legrand, M. Flavonoid accumulation in Arabidopsis repressed in lignin synthesis affects auxin transport and plant growth. Plant Cell 2007, 19, 148–162. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhu, M.L.; Wei, Z.M. Cotton laccase gene overexpression in transgenic Populus alba var. pyramidalis and its effects on the lignin biosynthesis in transgenic plants. J. Mol. Cell Biol. 2008, 41, 11–18. (In Chinese) [Google Scholar]
- Stewart, J.J.; Akiyama, T.; Chapple, C.; Ralph, J.; Mansfield, S.D. The effects on lignin structure of overexpression of ferulate 5-hydroxylase in hybrid poplar. Plant Physiol. 2009, 150, 621–635. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Dixon, R.A. Transcriptional networks for lignin biosynthesis: More complex than we thought? Trends Plant Sci. 2011, 16, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Lee, C.; Zhong, R.; Ye, Z.-H. MYB58 and MYB63 Are Transcriptional Activators of the Lignin Biosynthetic Pathway during Secondary Cell Wall Formation in Arabidopsis. Plant Cell 2009, 21, 248–266. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.S.; Wickneswari, R. Characterization of microRNAs expressed during secondary wall biosynthesis in Acacia mangium. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Sitbon, F.; Hennion, S.; Little, C.H.A.; Sundberg, B. Enhanced ethylene production and peroxidase activity in IAA-overproducing transgenic tobacco plants is associated with increased lignin content and altered lignin composition. Plant Sci. 1999, 141, 165–173. [Google Scholar] [CrossRef]
- Rhodes, M.J.C.; Wooltorton, L.S.C. Stimulation of phenolic acid and lignin biosynthesis in swede root tissue by ethylene. Phytochemistry 1973, 12, 107–118. [Google Scholar] [CrossRef]
- Peng, D.; Chen, X.; Yin, Y.; Lu, K.; Yang, W.; Tang, Y.; Wang, Z. Lodging resistance of winter wheat (Triticum aestivum L.): Lignin accumulation and its related enzymes activities due to the application of paclobutrazol or gibberellin acid. Field Crops Res. 2014, 157, 1–7. [Google Scholar] [CrossRef]
- Elumalai, S.; Tobimatsu, Y.; Grabber, J.H.; Pan, X.; Ralph, J. Epigallocatechin gallate incorporation into lignin enhances the alkaline delignification and enzymatic saccharification of cell walls. Biotechnol. Biofuels 2012, 5. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Tucker, M.; Ji, Y. Recent Development in Chemical Depolymerization of Lignin: A Review. J. Appl. Chem. 2013, 2013. [Google Scholar] [CrossRef]
- Toledano, A.; García, A.; Mondragon, I.; Labidi, J. Lignin separation and fractionation by ultrafiltration. Sep. Purif. Technol. 2010, 71, 38–43. [Google Scholar] [CrossRef]
- López-Abelairas, M.; Álvarez Pallín, M.; Salvachúa, D.; Lú-Chau, T.; Martínez, M.J.; Lema, J.M. Optimisation of the biological pretreatment of wheat straw with white-rot fungi for ethanol production. Bioprocess Biosyst. Eng. 2013, 36, 1251–1260. [Google Scholar] [CrossRef] [PubMed]
- Rubin, E.M. Genomics of cellulosic biofuels. Nature 2008, 454, 841–845. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C. Lignocellulosic residues: Biodegradation and bioconversion by fungi. Biotechnol. Adv. 2009, 27, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Li, Y. Effectiveness of microbial pretreatment by Ceriporiopsis subvermispora on different biomass feedstocks. Bioresour. Technol. 2011, 102, 7507–7512. [Google Scholar] [CrossRef] [PubMed]
- Dhiman, S.S.; Haw, J.-R.; Kalyani, D.; Kalia, V.C.; Kang, Y.C.; Lee, J.-K. Simultaneous pretreatment and saccharification: Green technology for enhanced sugar yields from biomass using a fungal consortium. Bioresour.Technol. 2015, 179, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Kellner, H.; Luis, P.; Zimdars, B.; Kiesel, B.; Buscot, F. Diversity of bacterial laccase-like multicopper oxidase genes in forest and grassland Cambisol soil samples. Soil Biol. Biochem. 2008, 40, 638–648. [Google Scholar] [CrossRef]
- Miyazaki, T.; Miyazaki, J.; Yamane, H.; Nishiyama, M. alpha-Aminoadipate aminotransferase from an extremely thermophilic bacterium, Thermus thermophilus. Microbiology 2004, 150, 2327–2334. [Google Scholar] [CrossRef] [PubMed]
- Johannes, T.W.; Zhao, H. Directed evolution of enzymes and biosynthetic pathways. Curr. Opin. Microbiol. 2006, 9, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Elegir, G.; Kindl, A.; Sadocco, P.; Orlandi, M. Development of antimicrobial cellulose packaging through laccase-mediated grafting of phenolic compounds. Enzyme Microb. Technol. 2008, 43, 84–92. [Google Scholar] [CrossRef]
- Baurhoo, B.; Ruiz-Feria, C.A.; Zhao, X. Purified lignin: Nutritional and health impacts on farm animals—A review. Anim. Feed Sci. Technol. 2008, 144, 175–184. [Google Scholar] [CrossRef]
- Barber, M.S.; McConnell, V.S.; DeCaux, B.S. Antimicrobial intermediates of the general phenylpropanoid and lignin specific pathways. Phytochemistry 2000, 54, 53–56. [Google Scholar] [CrossRef]
- Haggblom, M.M. Microbial breakdown of halogenated aromatic pesticides and related compounds. FEMS Microbiol. Rev. 1992, 9, 29–71. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Brink, D.L.; Blanch, H.W. Identification of potential fermentation inhibitors in conversion of hybrid poplar hydrolyzate to ethanol. Biomass Bioenergy 2002, 22, 125–138. [Google Scholar] [CrossRef]
- Laurichesse, S.; Avérous, L. Chemical modification of lignins: Towards biobased polymers. Prog. Polym. Sci. 2014, 39, 1266–1290. [Google Scholar] [CrossRef]
- Yang, D.; Qiu, X.; Zhou, M.; Lou, H. Properties of sodium lignosulfonate as dispersant of coal water slurry. Energy Convers. Manag. 2007, 48, 2433–2438. [Google Scholar] [CrossRef]
- Boeriu, C.G.; Bravo, D.; Gosselink, R.J.A.; van Dam, J.E.G. Characterisation of structure-dependent functional properties of lignin with infrared spectroscopy. Ind. Crops Prod. 2004, 20, 205–218. [Google Scholar] [CrossRef]
- Sena-Martins, G.; Almeida-Vara, E.; Duarte, J.C. Eco-friendly new products from enzymatically modified industrial lignins. Ind. Crops Prod. 2008, 27, 189–195. [Google Scholar] [CrossRef]
- Mohan, D.; Pittman, C.U., Jr.; Steele, P.H. Single, binary and multi-component adsorption of copper and cadmium from aqueous solutions on Kraft lignin—A biosorbent. J. Colloid Interface Sci. 2006, 297, 489–504. [Google Scholar] [CrossRef] [PubMed]
- Norgren, M.; Edlund, H. Lignin: Recent advances and emerging applications. Curr. Opin. Colloid Interface Sci. 2014, 19, 409–416. [Google Scholar] [CrossRef]
- Hu, S.; Zhang, S.; Pan, N.; Hsieh, Y.-L. High energy density supercapacitors from lignin derived submicron activated carbon fibers in aqueous electrolytes. J. Power Sources 2014, 270, 106–112. [Google Scholar] [CrossRef]
- Pedersen, J.F.; Vogel, K.P.; Funnell, D.L. Impact of Reduced Lignin on Plant Fitness. Crop. Sci. 2005, 45, 812–819. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Welker, C.M.; Balasubramanian, V.K.; Petti, C.; Rai, K.M.; DeBolt, S.; Mendu, V. Engineering Plant Biomass Lignin Content and Composition for Biofuels and Bioproducts. Energies 2015, 8, 7654-7676. https://doi.org/10.3390/en8087654
Welker CM, Balasubramanian VK, Petti C, Rai KM, DeBolt S, Mendu V. Engineering Plant Biomass Lignin Content and Composition for Biofuels and Bioproducts. Energies. 2015; 8(8):7654-7676. https://doi.org/10.3390/en8087654
Chicago/Turabian StyleWelker, Cassie Marie, Vimal Kumar Balasubramanian, Carloalberto Petti, Krishan Mohan Rai, Seth DeBolt, and Venugopal Mendu. 2015. "Engineering Plant Biomass Lignin Content and Composition for Biofuels and Bioproducts" Energies 8, no. 8: 7654-7676. https://doi.org/10.3390/en8087654
APA StyleWelker, C. M., Balasubramanian, V. K., Petti, C., Rai, K. M., DeBolt, S., & Mendu, V. (2015). Engineering Plant Biomass Lignin Content and Composition for Biofuels and Bioproducts. Energies, 8(8), 7654-7676. https://doi.org/10.3390/en8087654





