Polyolefin Pyrolysis in Multilayer Fluidized Beds: An Innovative Approach to Obtain Valuable Alternative Fuels
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
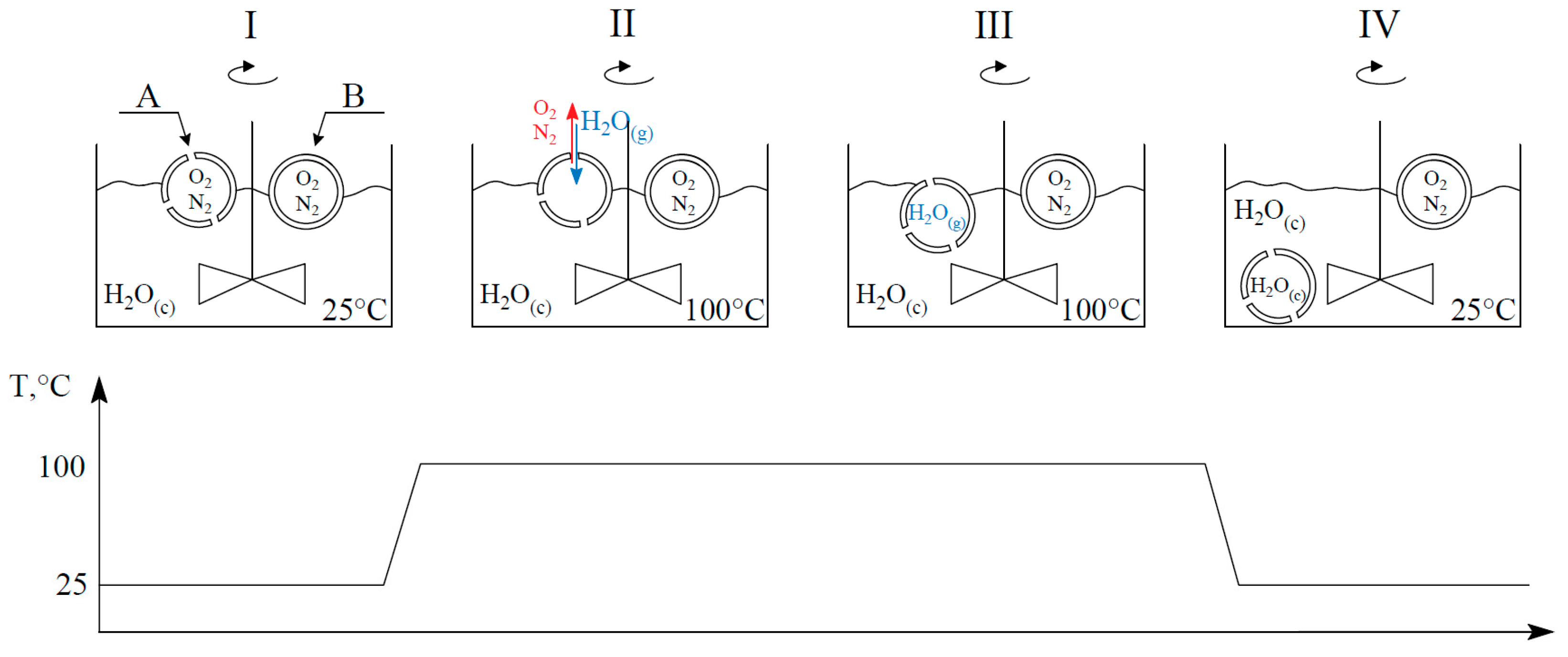
- Step I: Perforated (A) and undamaged (B) cenospheres float on the water surface. Water is mixed continuously.
- Step II: In the boiling process, water vapor fills the interior of the perforated cenospheres due to gas exchange. Oxygen and nitrogen diffuse out of the damaged particles.
- Step III: Cenospheres are completely filled with water vapor.
- Step IV: During the cooling process, the water vapor condenses inside the perforated grains, generating a vacuum and filling damaged cenospheres with liquid water, which leads these cenospheres to sink to the bottom of the vessel.
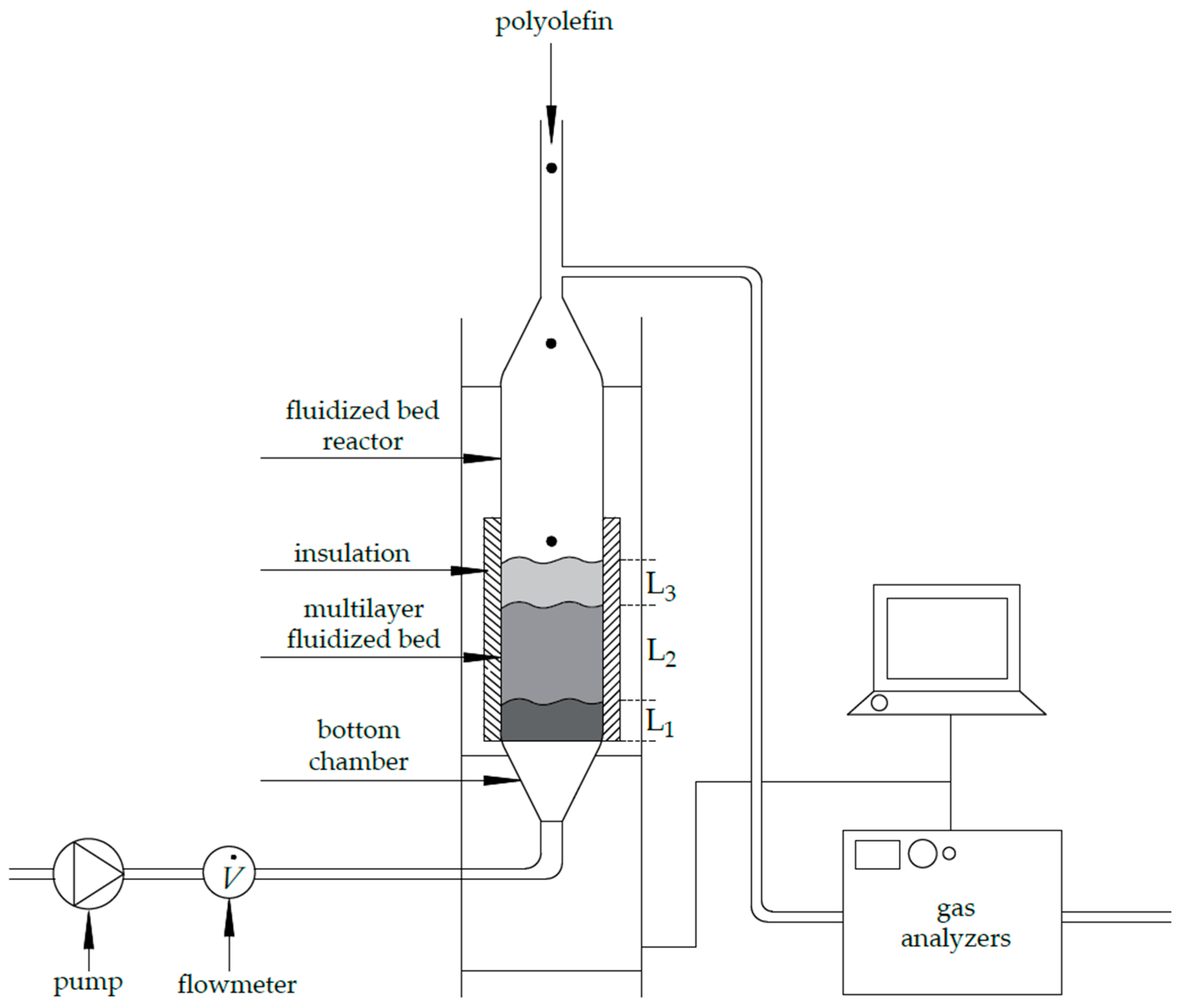
2.2. Methods
2.3. Experimental Procedure
3. Results
3.1. The Minimum Flduization Velocity of the Multialyer Fluidized Beds
- (a)
- Initiating the data acquisition, starting the flow of nitrogen, and setting the flow regulator to the lowest possible value (to prevent powder grains from overflowing through the holes in the bottom sieve);
- (b)
- Placing the lower layer of the multilayer fluidized bed (quartz sand) into the reactor;
- (c)
- Placing the upper layer of the fluidized bed (cenospheres) into the reactor;
- (d)
- Gradually increasing the gas flow while recording the pressure drop in the fluidized bed at a given flow rate through the pressure drop sensor located in the bottom chamber;
- (e)
- Slowly reducing the gas flow to the minimum value (after achieving the fluidized state).
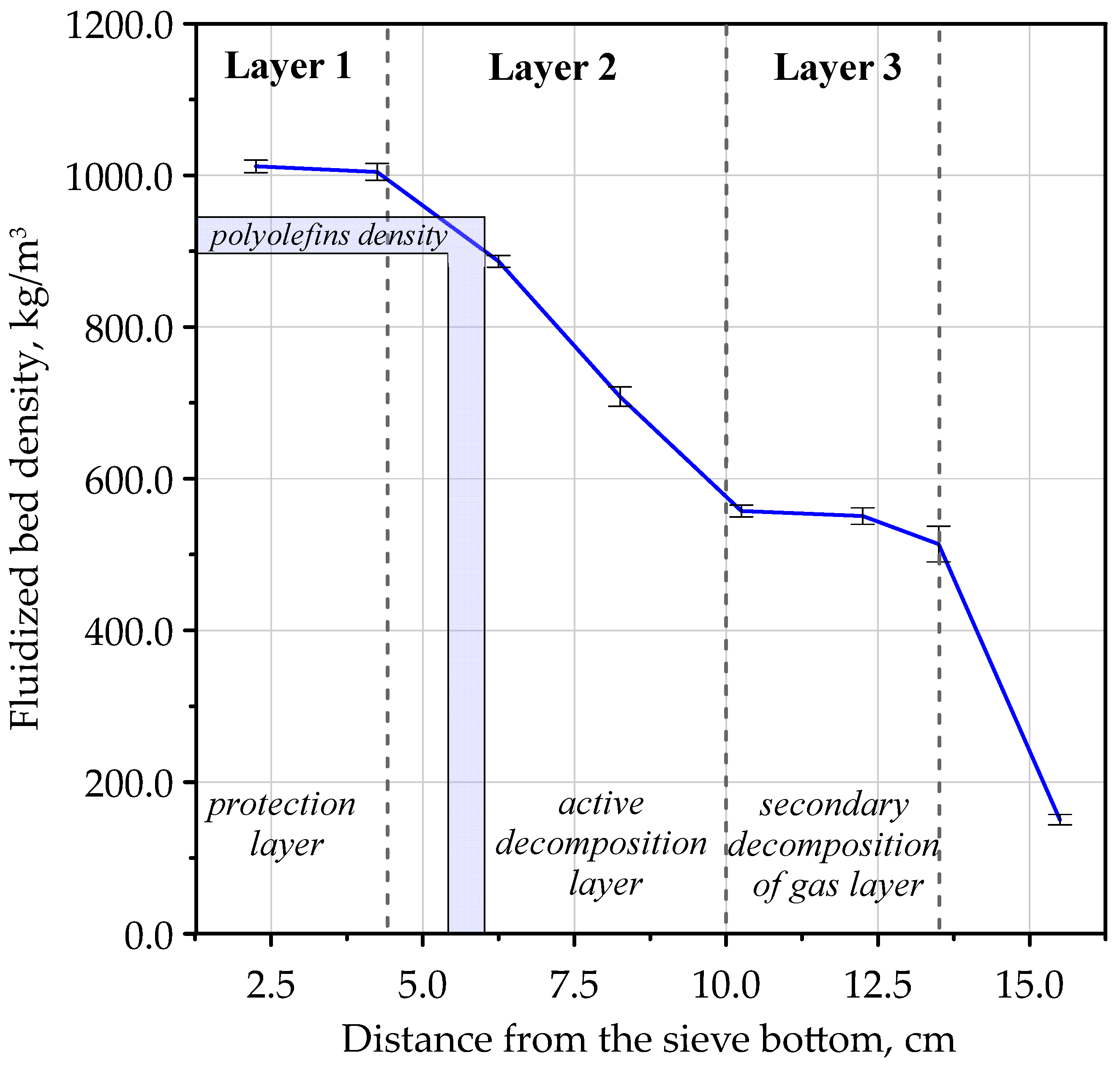
3.2. The Density Profile of the Multilayer Fluidized Beds
- (a)
- Initiating the data acquisition and setting the nitrogen flow to the lowest possible value;
- (b)
- Placing of lower and upper layers of the multilayer fluidized bed into the reactor;
- (c)
- Adjusting the gas flow to achieve u/umf, sand = 1.6 and fluidizing the multilayer fluidized beds for 10 min to achieve the desired mixing;
- (d)
- Recording pressure drop values at different heights inside the fluidized bed, for 10 min, at a frequency of 10 Hz;
- (e)
- Slowly reducing the gas flow to the minimum value (after achieving the fluidized state).
- ρ(h)—fluidized bed density at height h [kg/m3];
- dp(h)—change of differential pressure in the fluidized bed at height h [Pa];
- dh—height difference between two heights in the fluidized bed [m].
3.3. Polyolefin Thermogravimetric Analysis
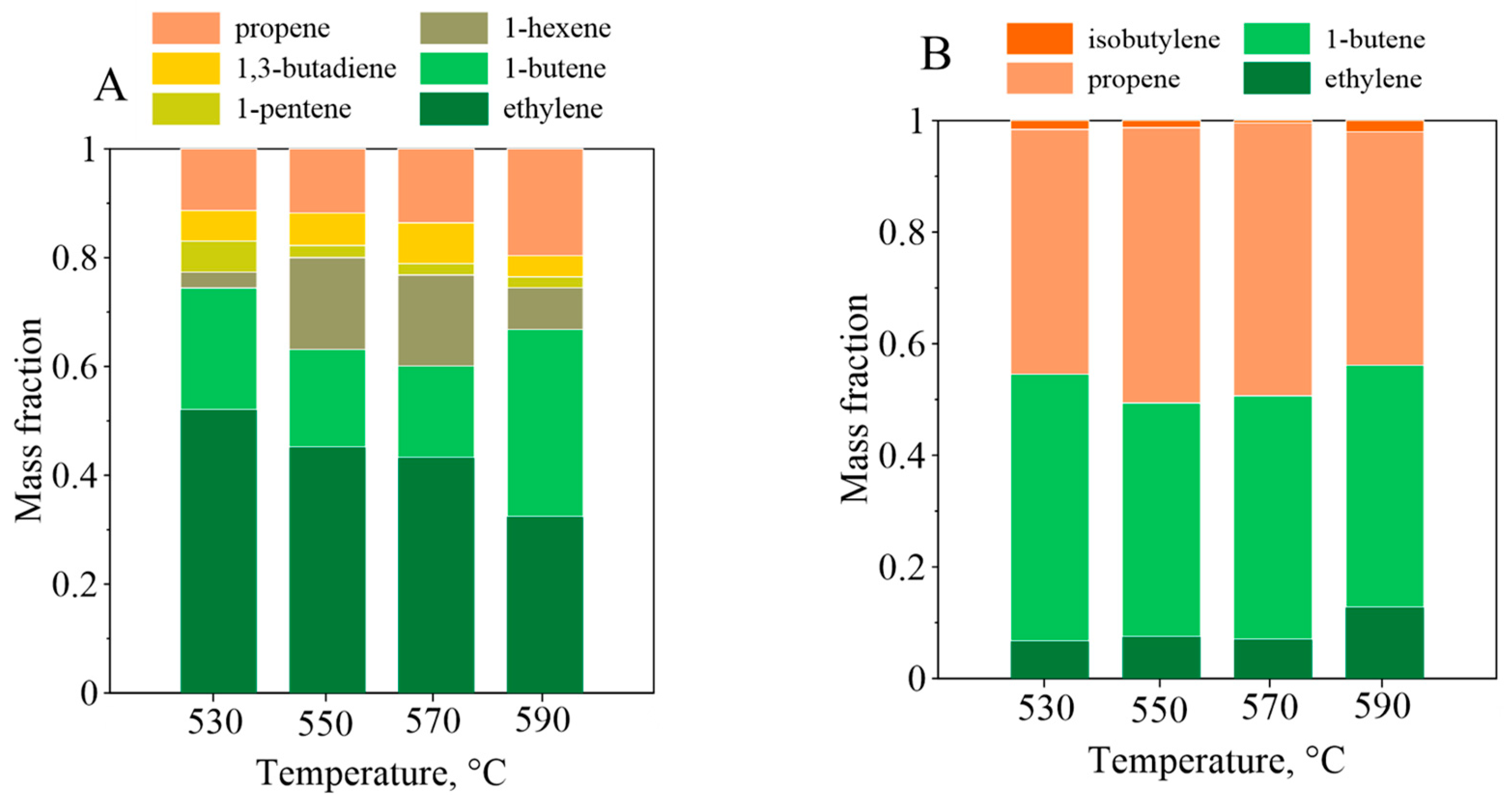
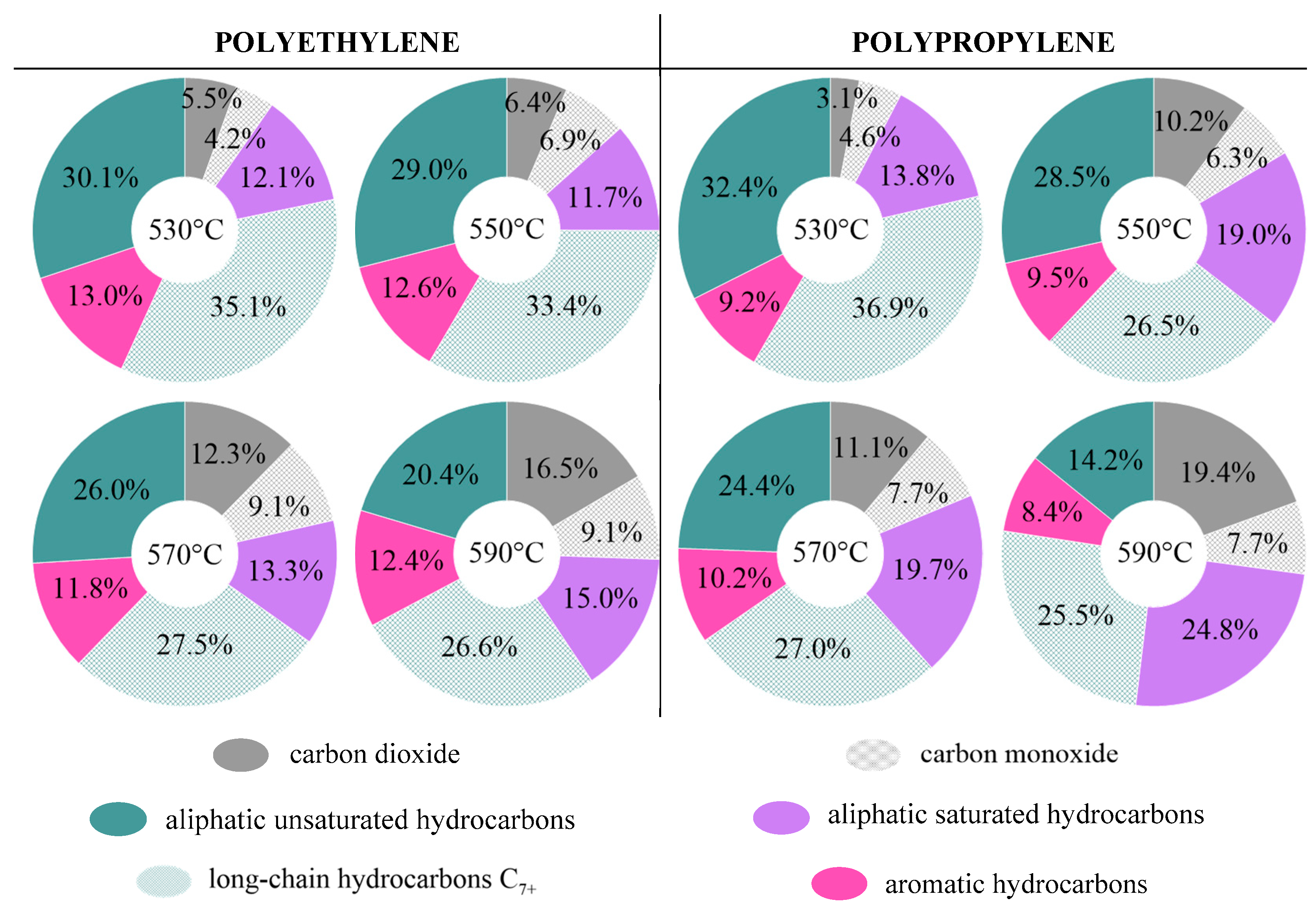
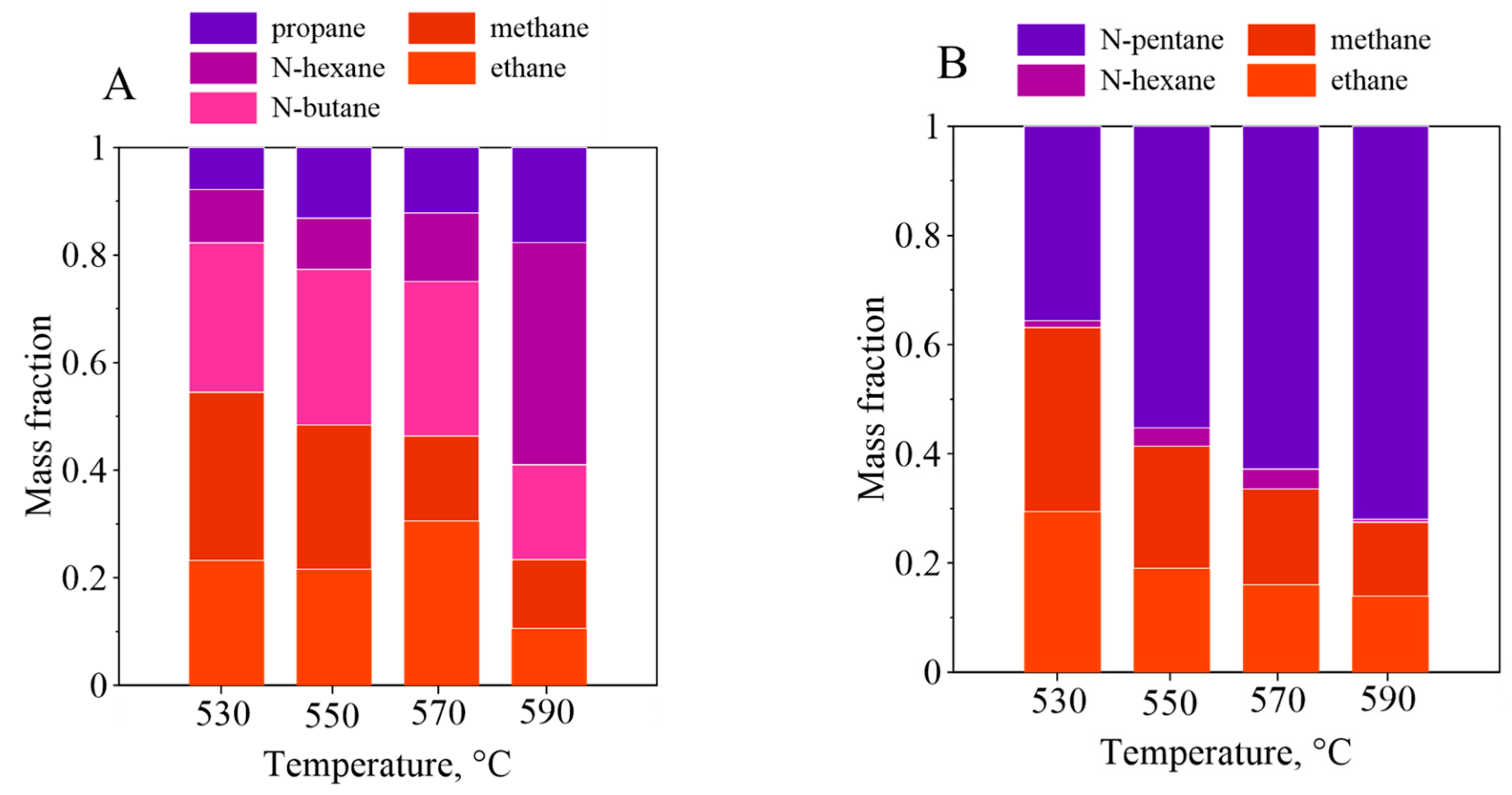
3.4. Polyolefin Pyrolysis in the Fluidized Beds
4. Conclusions
- The paper introduces an alternative method for conducting the polyolefin pyrolysis process in a fluidized bed reactor.
- 2.
- The new approach used in the investigation avoids the formation of a solid phase, a common challenge in pyrolysis processes.
- 3.
- Hydrocarbon mixtures containing CO and CO2 were obtained in the case of a multilayer fluidized bed made out of modified cenospheres.
- 4.
- The gas mixtures obtained during pyrolysis are suitable for various catalytic processes. Depending on the chosen catalytic process for gas mixtures, saturated hydrocarbons and hydrogen can be produced.
- 5.
- The general benefits of using multilayer fluidized beds.
- (I)
- The low-density (upper) layer of the multilayer fluidized beds allows for the free dosing of fuel samples into the fluidized bed volume without the use of special feeders. The premature fuel degradation and nozzle clogging in the feeders are eliminated.
- (II)
- The variable-density (middle) layer acts as a space for material pyrolysis, characterized by even temperature, good mass and heat exchange, and intensive mixing, and imposing a specific residence time for reagents inside the fluidized beds. According to Archimedes’ principle, polyolefins freely sink into the multilayer fluidized beds’ depth, where the density is like that of PE and PP. The decomposition process then takes place inside the fluidized bed layer.
- (III)
- The high-density (bottom) layer protects the reactor from the falling of the polyolefins to the bottom sieve during the operation and from the clogging of the gas inlets.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Golub, G.; Tsyvenkova, N.; Kukharets, S.; Holubenko, A.; Omarov, I.; Klymenko, O.; Mudryk, K.; Hutsol, T. European Green Deal: An Experimental Study of the Biomass Filtration Combustion in a Downdraft Gasifier. Energies 2023, 16, 7490. [Google Scholar] [CrossRef]
- Nitkiewicz, T.; Ociepa-Kubicka, A. Eco-Investments-Life Cycle Assessment of Different Scenarios of Biomass Combustion. Ecol. Chem. Eng. S 2018, 25, 307–322. [Google Scholar] [CrossRef]
- Garcia-cortes, S. Repurposing End-of-Life Coal Mines with Business Models. Energies 2023, 16, 7617. [Google Scholar]
- Barmparitsas, N.; Karellas, S.; Pallis, P.; Thanopoulos, S.; Kobelt, D. An Innovative Heating, Ventilation, Air Conditioning and Refrigeration Circular Economy System for Reducing Carbon Dioxide Emissions in Europe via Extensive Reuse of Existing Fluorinated Gases. Energies 2023, 16, 7705. [Google Scholar] [CrossRef]
- Leski, K.; Berkowicz-Płatek, G. Pyrolysis of Plastic Wastes as a Way of Obtaining Valuable Chemical Raw Materials. Chem. Rev. Lett. 2021, 4, 92–97. [Google Scholar] [CrossRef]
- Holubčík, M.; Klačková, I.; Ďurčanský, P. Pyrolysis Conversion of Polymer Wastes to Noble Fuels in Conditions of the Slovak Republic. Energies 2020, 13, 4849. [Google Scholar] [CrossRef]
- Anuar Sharuddin, S.D.; Abnisa, F.; Wan Daud, W.M.A.; Aroua, M.K. A Review on Pyrolysis of Plastic Wastes. Energy Convers. Manag. 2016, 115, 308–326. [Google Scholar] [CrossRef]
- Harussani, M.M.; Sapuan, S.M.; Rashid, U.; Khalina, A.; Ilyas, R.A. Pyrolysis of Polypropylene Plastic Waste into Carbonaceous Char: Priority of Plastic Waste Management amidst COVID-19 Pandemic. Sci. Total Environ. 2022, 803, 149911. [Google Scholar] [CrossRef]
- López, A.; de Marco, I.; Caballero, B.M.; Laresgoiti, M.F.; Adrados, A. Pyrolysis of Municipal Plastic Wastes: Influence of Raw Material Composition. Waste Manag. 2010, 30, 620–627. [Google Scholar] [CrossRef]
- Schwartz, N.R.; Paulsen, A.D.; Blaise, M.J.; Wagner, A.L.; Yelvington, P.E. Analysis of Emissions from Combusting Pyrolysis Products. Fuel 2020, 274, 117863. [Google Scholar] [CrossRef]
- Sciazko, M.; Mertas, B.; Kosyrczyk, L.; Sobolewski, A. A Predictive Model for Coal Coking Based on Product Yield and Energy Balance. Energies 2020, 13, 4953. [Google Scholar] [CrossRef]
- Brown, R.M.; Hoover, A.N.; Klinger, J.L.; Wahlen, B.D.; Hartley, D.; Lee, H.; Thompson, V.S. Decontamination of Mixed Paper and Plastic Municipal Solid Waste Increases Low and High Temperature Conversion Yields. Front. Energy Res. 2022, 10, 834832. [Google Scholar] [CrossRef]
- Ganguly, A.; Brown, R.C.; Wright, M.M. Investigating the Impacts of Feedstock Variability on a Carbon-Negative Autothermal Pyrolysis System Using Machine Learning. Front. Clim. 2022, 4, 842650. [Google Scholar] [CrossRef]
- Sanito, R.C.; Chen, C.H.; You, S.J.; Yang, H.H.; Wang, Y.F. Volatile Organic Compounds (VOCs) Analysis from Plasma Pyrolysis of Printed Circuit Boards (PCB) with the Addition of CaCO3 from Natural Flux Agents. Environ. Technol. Innov. 2023, 29, 103011. [Google Scholar] [CrossRef]
- European Commission A Green Deal Industrial Plan for the Net-Zero Age. 2023. Available online: https://commission.europa.eu/system/files/2023-02/COM_2023_62_2_EN_ACT_A%20Green%20Deal%20Industrial%20Plan%20for%20the%20Net-Zero%20Age.pdf (accessed on 21 February 2024).
- Jia, Y.; Mao, Z.; Huang, W.; Zhang, J. Effect of Temperature and Crystallinity on the Thermal Conductivity of Semi-Crystalline Polymers: A Case Study of Polyethylene. Mater. Chem. Phys. 2022, 287, 126325. [Google Scholar] [CrossRef]
- Wang, X.; Jin, Q.; Zhang, J.; Li, Y.; Li, S.; Mikulčić, H.; Vujanović, M.; Tan, H.; Duić, N. Soot Formation during Polyurethane (PU) Plastic Pyrolysis: The Effects of Temperature and Volatile Residence Time. Energy Convers. Manag. 2018, 164, 353–362. [Google Scholar] [CrossRef]
- Onwudili, J.A.; Insura, N.; Williams, P.T. Composition of Products from the Pyrolysis of Polyethylene and Polystyrene in a Closed Batch Reactor: Effects of Temperature and Residence Time. J. Anal. Appl. Pyrolysis 2009, 86, 293–303. [Google Scholar] [CrossRef]
- Font, R.; Aracil, I.; Fullana, A.; Martín-Gullón, I.; Conesa, J.A. Semivolatile Compounds in Pyrolysis of Polyethylene. J. Anal. Appl. Pyrolysis 2003, 68–69, 599–611. [Google Scholar] [CrossRef]
- Antoniou, N.A.; Zorpas, A.A. Quality Protocol and Procedure Development to Define End-of-Waste Criteria for Tire Pyrolysis Oil in the Framework of Circular Economy Strategy. Waste Manag. 2019, 95, 161–170. [Google Scholar] [CrossRef]
- Islam, M.N.; Joardder, M.U.H.; Hoque, S.M.N.; Uddin, M.S. A Comparative Study on Pyrolysis for Liquid Oil from Different Biomass Solid Wastes. Procedia Eng. 2013, 56, 643–649. [Google Scholar] [CrossRef]
- Supriyanto; Usino, D.O.; Ylitervo, P.; Dou, J.; Sipponen, M.H.; Richards, T. Identifying the Primary Reactions and Products of Fast Pyrolysis of Alkali Lignin. J. Anal. Appl. Pyrolysis 2020, 151, 104917. [Google Scholar] [CrossRef]
- Oliveri, L.M.; Chiacchio, F.; D’Urso, D.; Matarazzo, A.; Cutaia, L.; Luciano, A. Circular Economy and Industrial Symbiosis in Sicily. IFIP Adv. Inf. Commun. Technol. 2022, 663 IFIP, 432–439. [Google Scholar] [CrossRef]
- Baral, B.; Raine, R. Performance and Emissions of a Spark Ignition Engine Running on Gasoline Adulterated with Kerosene; SAE Technical Paper; SAE International: Warrendale, PA, USA, 2009; Volume 2009-Decem. [Google Scholar] [CrossRef]
- Igliński, B.; Kujawski, W.; Kiełkowska, U. Pyrolysis of Waste Biomass: Technical and Process Achievements, and Future Development—A Review. Energies 2023, 16, 1829. [Google Scholar] [CrossRef]
- Peters, J.; May, J.; Ströhle, J.; Epple, B. Flexibility of CFB Combustion: An Investigation of Co-Combustion with Biomass and RDF at Part Load in Pilot Scale. Energies 2020, 13, 4665. [Google Scholar] [CrossRef]
- Żukowski, W.; Berkowicz, G. The Combustion of Liquids and Low-Density Solids in a Cenospheric Fluidised Bed. Combust. Flame 2019, 206, 476–489. [Google Scholar] [CrossRef]
- Berkowicz-Płatek, G.; Żukowski, W.; Leski, K. Combustion of Polyethylene and Polypropylene in the Fluidized Bed with a Variable Vertical Density Profile. Energy 2024, 286, 129611. [Google Scholar] [CrossRef]
- Berkowicz, G.; Majka, T.M.; Żukowski, W. The Pyrolysis and Combustion of Polyoxymethylene in a Fluidised Bed with the Possibility of Incorporating CO2. Energy Convers. Manag. 2020, 214, 112888. [Google Scholar] [CrossRef]
- Merrill, P.B.; Madix, R.J. Hydrogenation of Weakly Rehybridized Ethylene on Fe(100)−H: Ethyl Group Formation. J. Am. Chem. Soc. 1996, 118, 5062–5067. [Google Scholar] [CrossRef]
- Vouvoudi, E.C.; Rousi, A.T.; Achilias, D.S. Effect of the Catalyst Type on Pyrolysis Products Distribution of Polymer Blends Simulating Plastics Contained in Waste Electric and Electronic Equipment. Sustain. Chem. Pharm. 2023, 34, 101145. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.K.; Trenary, M. Propyne Hydrogenation over a Pd/Cu(111) Single-Atom Alloy Studied Using Ambient Pressure Infrared Spectroscopy. ACS Catal. 2020, 10, 9716–9724. [Google Scholar] [CrossRef]
- Azzolina-Jury, F.; Thibault-Starzyk, F. Mechanism of Low Pressure Plasma-Assisted CO2 Hydrogenation over Ni-USY by Microsecond Time-Resolved FTIR Spectroscopy. Top. Catal. 2017, 60, 1709–1721. [Google Scholar] [CrossRef]
- Hakkarainen, T.; Mikkola, E.; Laperre, J.; Gensous, F.; Fardell, P.; Le Tallec, Y.; Baiocchi, C.; Paul, K.; Simonson, M.; Deleu, C.; et al. Smoke Gas Analysis by Fourier Transform Infrared Spectroscopy—Summary of the SAFIR Project Results. Fire Mater. 2000, 24, 101–112. [Google Scholar] [CrossRef]
- Plyler, E.K.; Tidwell, E.D.; Blaine, L.R. Infrared Absorption Spectrum of Methane from 2470 to 3200 cm−1. J. Res. Natl. Bur. Stand. Sect. A Phys. Chem. 1960, 64A, 201. [Google Scholar] [CrossRef] [PubMed]
- Kınaytürk, N.K.; Kalaycı, T.; Tunalı, B.; Altuğ, D.T. A Spectroscopic Approach to Compare the Quantum Chemical Calculations and Experimental Characteristics of Some Organic Molecules; Benzene, Toluene, P-Xylene, P-Toluidine. Chem. Phys. 2023, 570, 111905. [Google Scholar] [CrossRef]
- Cho, M.H.; Jung, S.H.; Kim, J.S. Pyrolysis of Mixed Plastic Wastes for the Recovery of Benzene, Toluene, and Xylene (BTX) Aromatics in a Fluidized Bed and Chlorine Removal by Applying Various Additives. Energy Fuels 2010, 24, 1389–1395. [Google Scholar] [CrossRef]
- Namioka, T.; Saito, A.; Inoue, Y.; Park, Y.; Min, T.J.; Roh, S.A.; Yoshikawa, K. Hydrogen-Rich Gas Production from Waste Plastics by Pyrolysis and Low-Temperature Steam Reforming over a Ruthenium Catalyst. Appl. Energy 2011, 88, 2019–2026. [Google Scholar] [CrossRef]
- Maqsood, T.; Dai, J.; Zhang, Y.; Guang, M.; Li, B. Pyrolysis of Plastic Species: A Review of Resources and Products. J. Anal. Appl. Pyrolysis 2021, 159, 105295. [Google Scholar] [CrossRef]
- Calero, M.; Solís, R.R.; Muñoz-Batista, M.J.; Pérez, A.; Blázquez, G.; Ángeles Martín-Lara, M. Oil and Gas Production from the Pyrolytic Transformation of Recycled Plastic Waste: An Integral Study by Polymer Families. Chem. Eng. Sci. 2023, 271, 118569. [Google Scholar] [CrossRef]
- Pal, P.; Ting, J.M.; Agarwal, S.; Ichikawa, T.; Jain, A. The Catalytic Role of D-Block Elements and Their Compounds for Improving Sorption Kinetics of Hydride Materials: A Review. Reactions 2021, 2, 22. [Google Scholar] [CrossRef]
- Ranjbar, N.; Kuenzel, C. Cenospheres: A Review. Fuel 2017, 207, 1–12. [Google Scholar] [CrossRef]
- Chen, Z.; Xu, L.; Zhang, X. Upgrading of Polyethylene to Hydrocarbon Fuels over the Fe-Modified Pt/Al2O3 Catalysts at a Mild Condition without External H2. Chem. Eng. J. 2022, 446, 136213. [Google Scholar] [CrossRef]
- Shen, X.; Zhao, Z.; Li, H.; Gao, X.; Fan, X. Microwave-Assisted Pyrolysis of Plastics with Iron-Based Catalysts for Hydrogen and Carbon Nanotubes Production. Mater. Today Chem. 2022, 26, 101166. [Google Scholar] [CrossRef]







| Fluidized Bed Version | Mass, g | Particle Diameter, μm | Static Height in the FB, cm | Bulk Density, g/cm3 |
|---|---|---|---|---|
| Fluidized bed I | quartz sand | |||
| 360 | 120–200 | 6 | 1.36 | |
| raw cenospheres | ||||
| 160 | 140–160 | 8 | 0.46 | |
| Fluidized bed II | quartz sand | |||
| 360 | 120–200 | 6 | 1.36 | |
| modified cenospheres | ||||
| 160 | 140–160 | 8 | 0.47 | |
| Temperature, °C | umf, multilayer fluidized bed Based on Grace Equation, cm/s | Quartz Sand Fluidization Number u/umf, sand | Nitrogen Velocity under Process Conditions, cm/s |
|---|---|---|---|
| 530 | 1.17 | 1.6 | 1.87 |
| 550 | 1.14 | 1.82 | |
| 570 | 1.08 | 1.73 | |
| 590 | 1.09 | 1.68 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Żukowski, W.; Leski, K.; Berkowicz-Płatek, G.; Wrona, J. Polyolefin Pyrolysis in Multilayer Fluidized Beds: An Innovative Approach to Obtain Valuable Alternative Fuels. Energies 2024, 17, 1034. https://doi.org/10.3390/en17051034
Żukowski W, Leski K, Berkowicz-Płatek G, Wrona J. Polyolefin Pyrolysis in Multilayer Fluidized Beds: An Innovative Approach to Obtain Valuable Alternative Fuels. Energies. 2024; 17(5):1034. https://doi.org/10.3390/en17051034
Chicago/Turabian StyleŻukowski, Witold, Krystian Leski, Gabriela Berkowicz-Płatek, and Jan Wrona. 2024. "Polyolefin Pyrolysis in Multilayer Fluidized Beds: An Innovative Approach to Obtain Valuable Alternative Fuels" Energies 17, no. 5: 1034. https://doi.org/10.3390/en17051034
APA StyleŻukowski, W., Leski, K., Berkowicz-Płatek, G., & Wrona, J. (2024). Polyolefin Pyrolysis in Multilayer Fluidized Beds: An Innovative Approach to Obtain Valuable Alternative Fuels. Energies, 17(5), 1034. https://doi.org/10.3390/en17051034








