Hydrogen-Based Energy Systems: Current Technology Development Status, Opportunities and Challenges
Abstract
:1. Introduction
1.1. Motivation
1.2. Objectives and Methodology
- Title included—“Hydrogen”;
- Document type—“Review Article”;
- Web of science category—“Energy Fuels”;
- Publication year—“2022 or 2021 or 2020”.
1.3. Technology Readiness Level
2. Hydrogen
2.1. Introduction
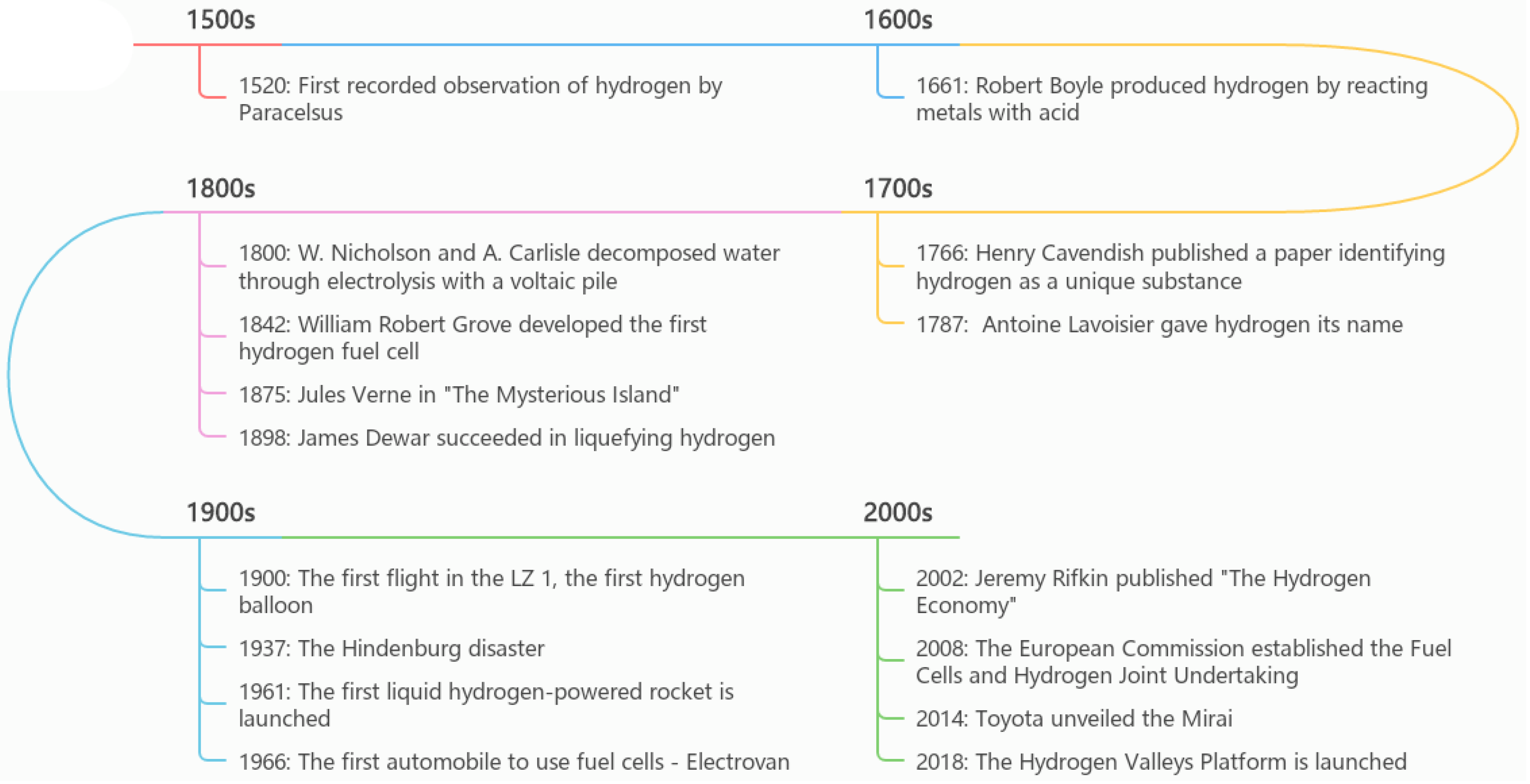
2.2. The History of Hydrogen
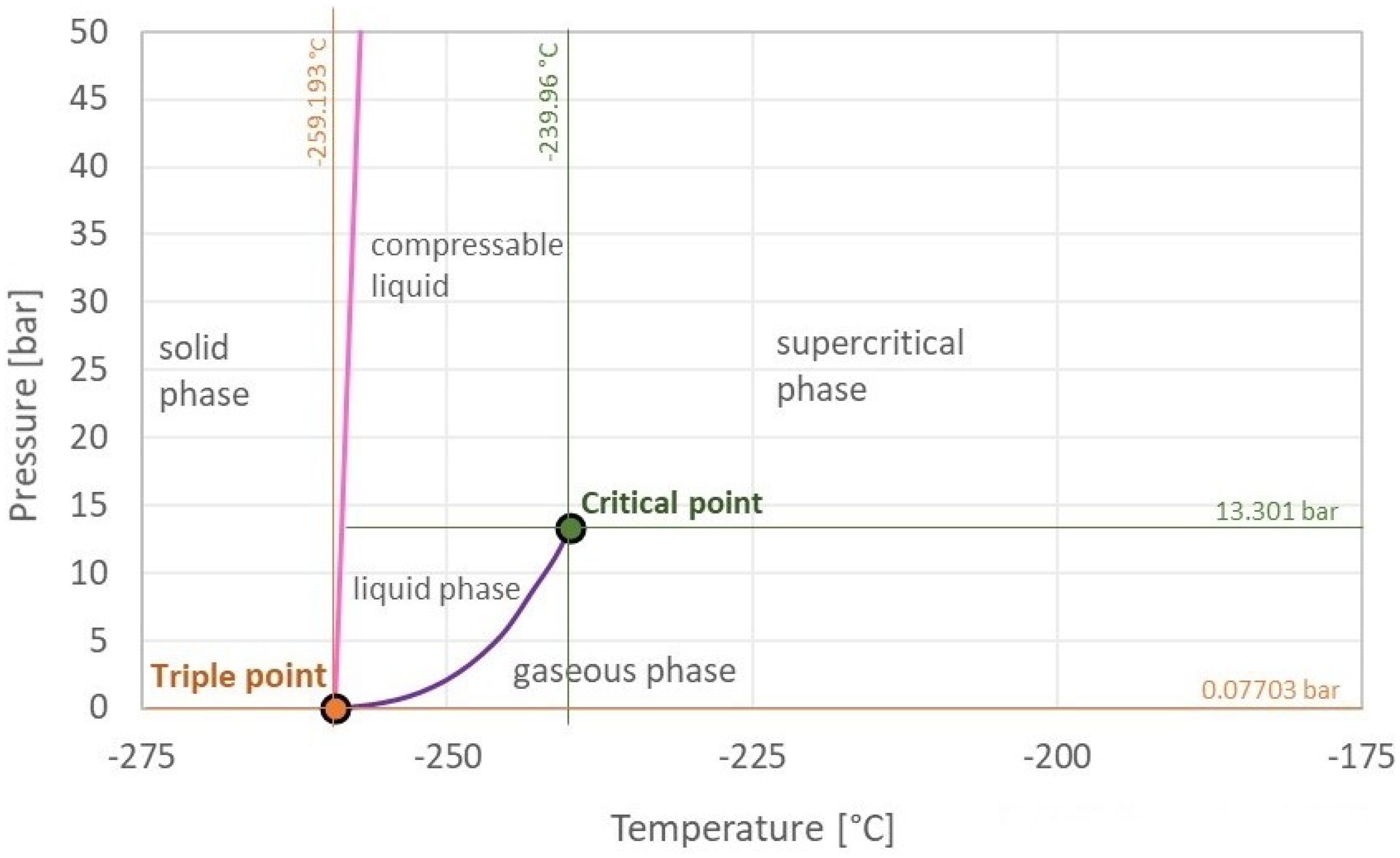
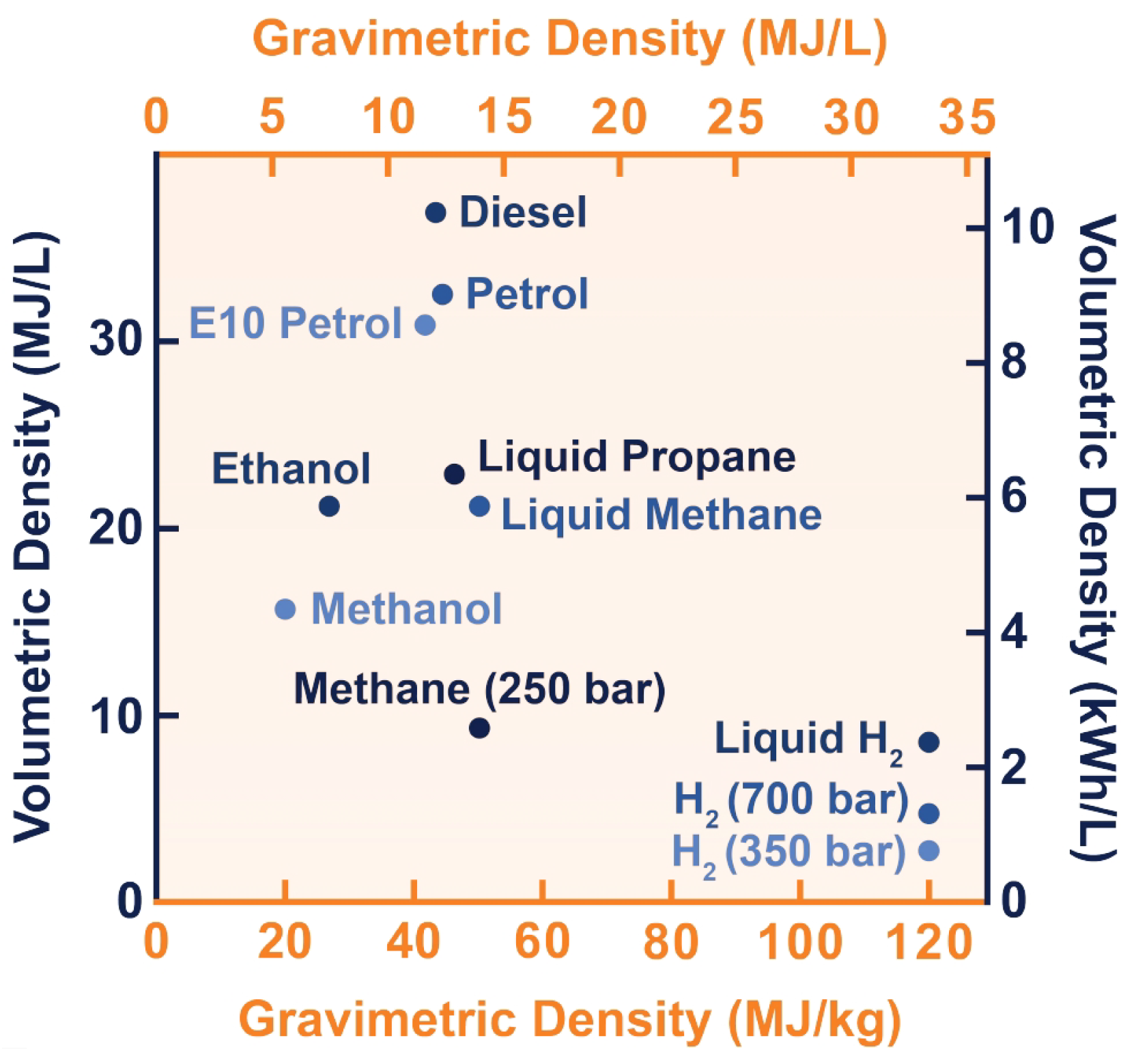
2.3. Hydrogen Properties
2.4. Safety and Standards
- ISO/TR 15916:2015 specifies guidelines for the use and storage of gaseous or liquid H2. The standard identifies the fundamental safety concerns, hazards, and risks, as well as H2 properties that are relevant to safety [41].
- The minimum quality characteristics of hydrogen fuel as dispensed for use in mobile and stationary applications are specified in ISO 14687:2019 [42].
- ISO 22734:2019 specifies the design, safety, and performance requirements for electrochemical hydrogen generators that electrolyse water to produce H2 [43].
- ISO 23273:2013 specifies the essential requirements for fuel cell vehicles (FCV) in terms of protecting people and the environment from hydrogen-related hazards both inside and outside the vehicle [46].
3. Green Hydrogen Production Processes
3.1. Introduction
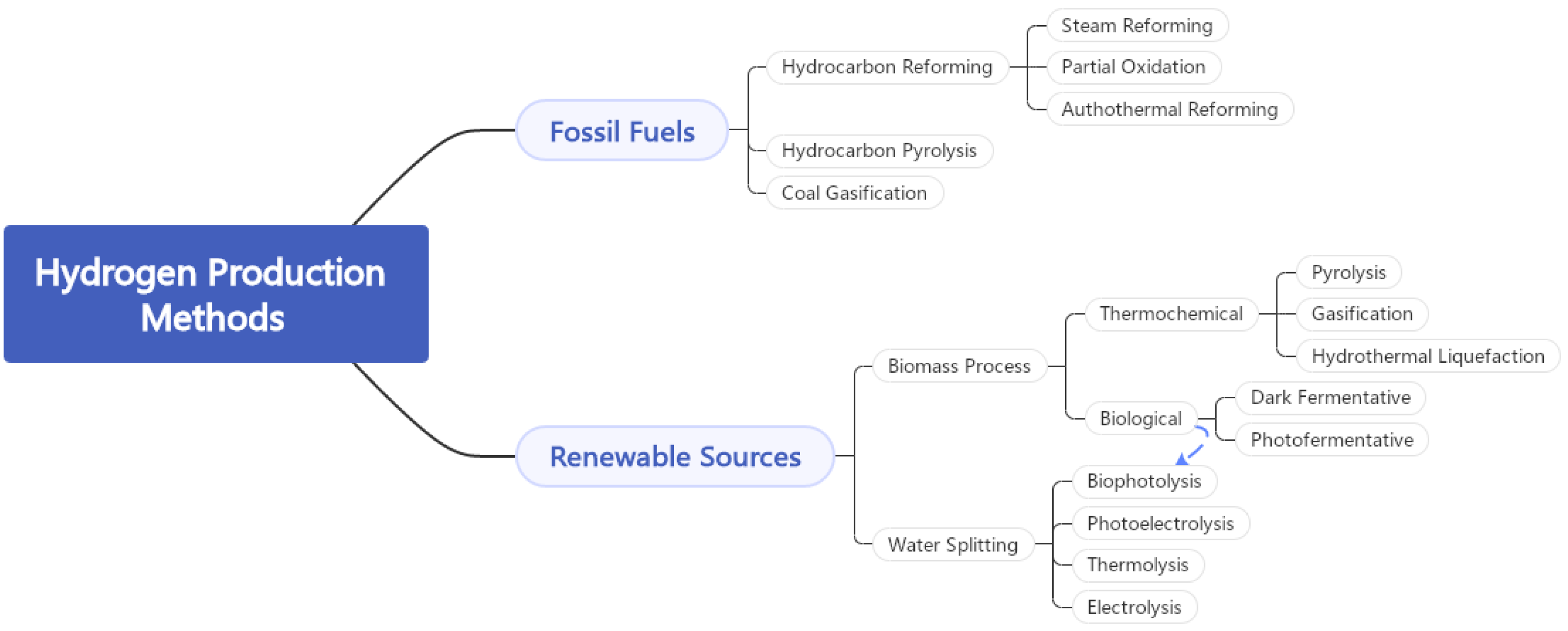
3.2. Hydrogen Production Processes
3.2.1. Hydrogen Production from Fossil Fuels
3.2.2. Hydrogen Production from Biomass
3.2.3. Hydrogen Production from Water
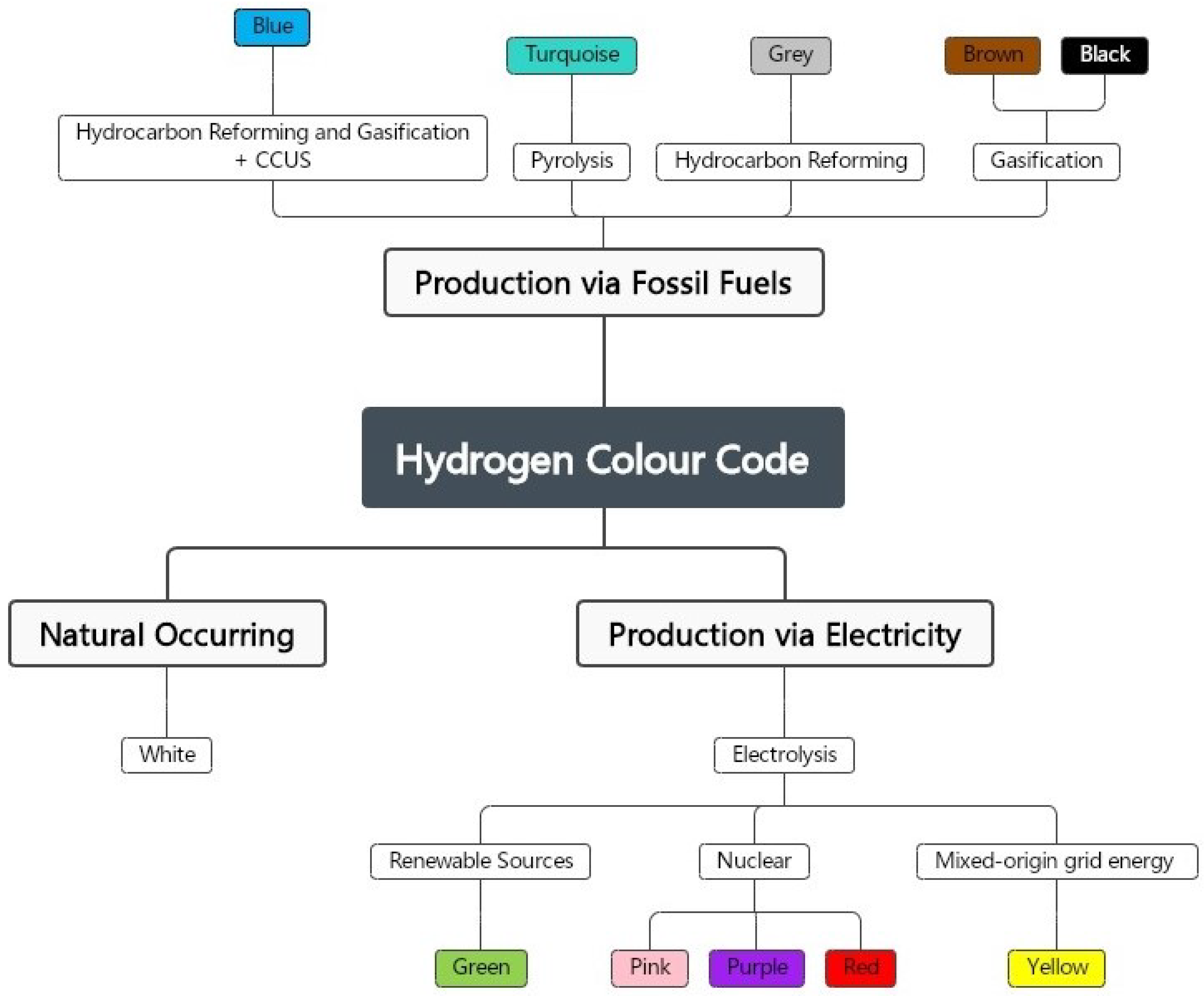
3.3. Hydrogen Colour Code
3.3.1. Black and Brown Hydrogen
3.3.2. Grey Hydrogen
3.3.3. Turquoise Hydrogen
3.3.4. Blue Hydrogen
3.3.5. Yellow Hydrogen
3.3.6. Pink, Red, and Purple Hydrogen
3.3.7. White Hydrogen
3.3.8. Green Hydrogen
Green hydrogen is hydrogen produced through the electrolysis of water with 100% or near 100% renewable energy with close to zero greenhouse gas emissions.
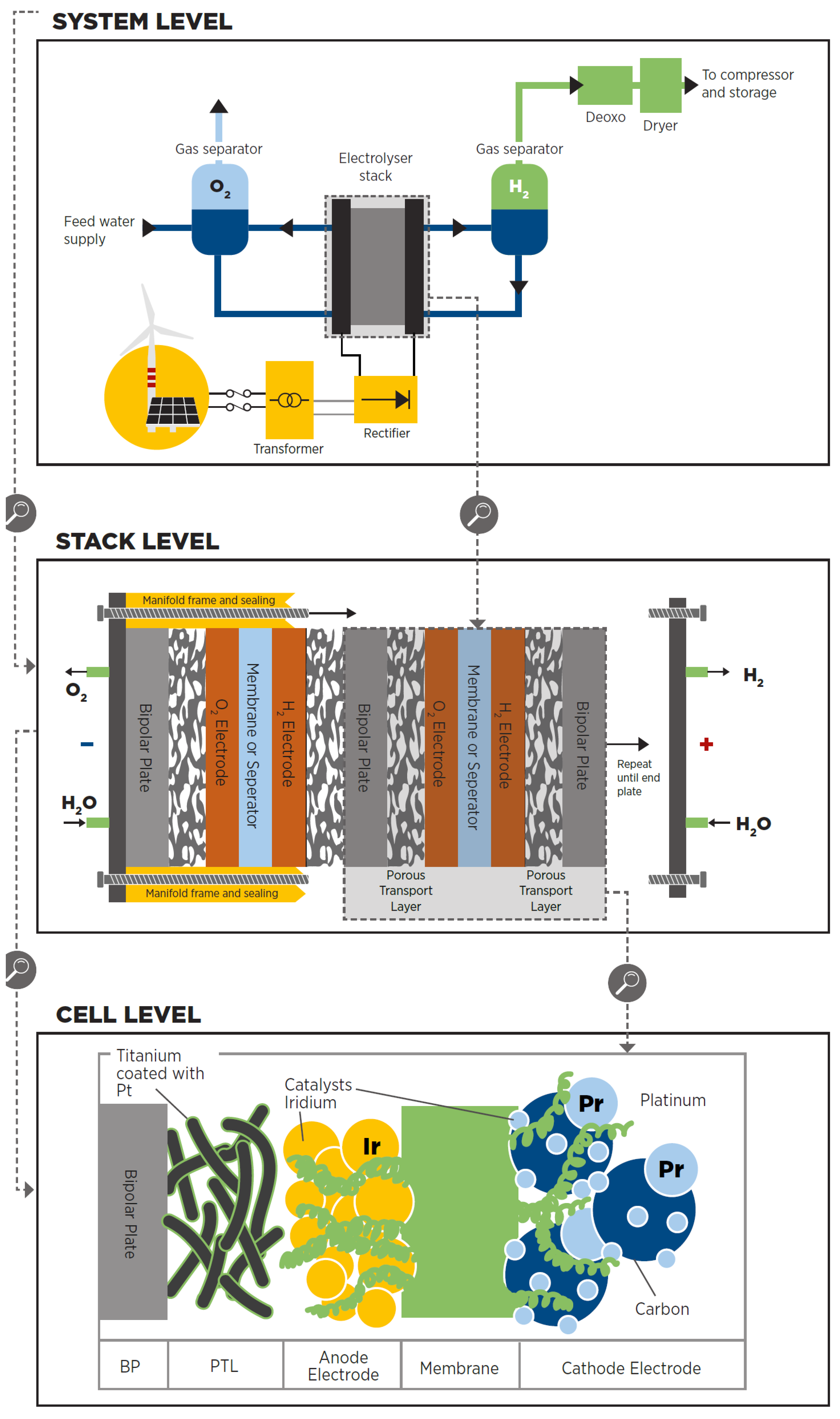
3.4. Water Electrolysis
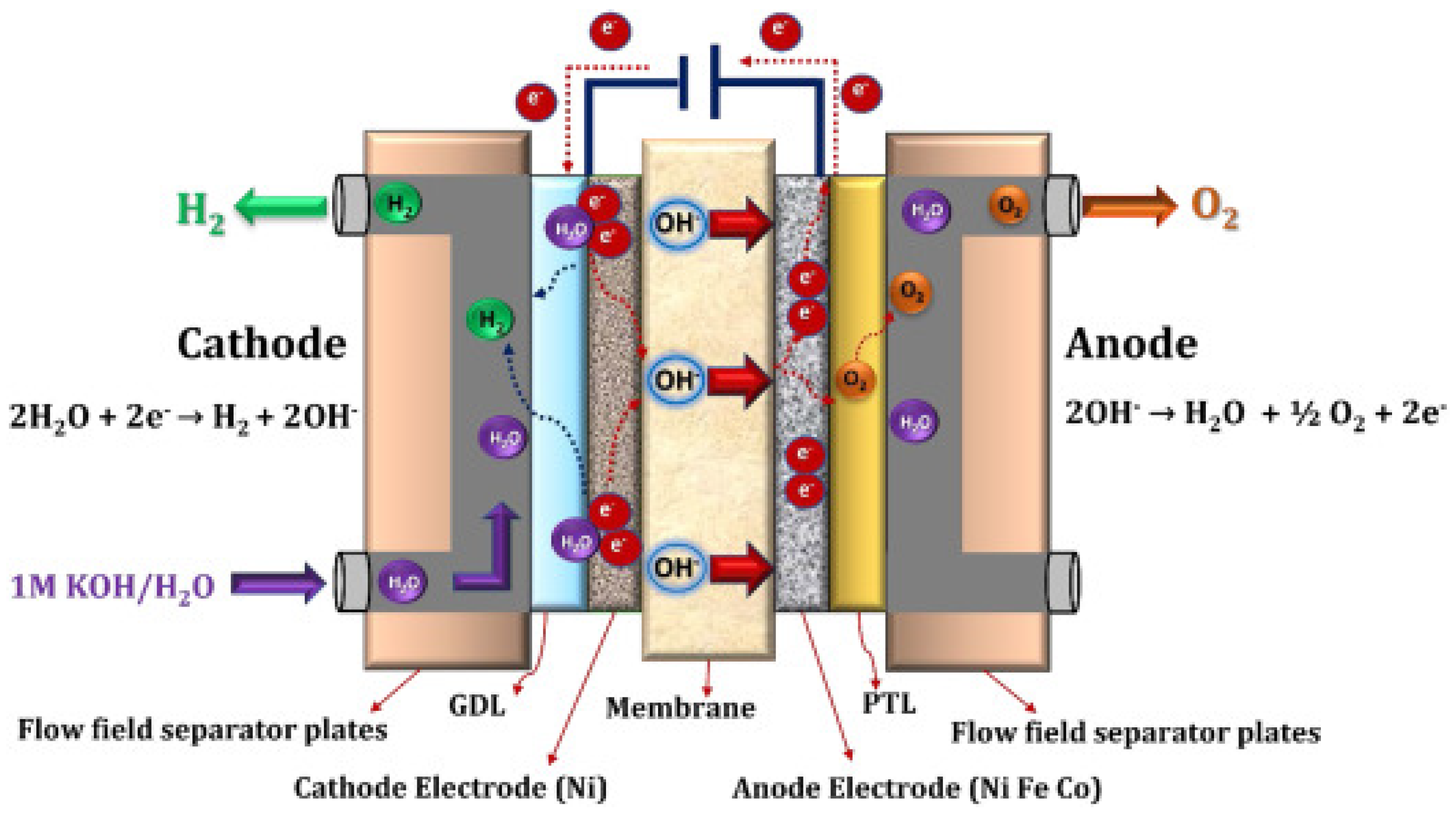
- The cell is the electrolyser’s heart and the site of the electrochemical process. Common cells consist of two electrodes—anode and cathode—immersed in a liquid electrolyte or adjacent to a solid electrolyte membrane, two porous transport layers (PTLs) that facilitate reactant transport and product removal, and bipolar plates (BPs) that provide mechanical support and flow distribution.
- The stack generally serves a broader purpose by incorporating multiple cells connected in series, insulating material spacers between opposing electrodes, seals, frames for mechanical support, and end plates to prevent leakage and collect fluids.
- The system level usually includes cooling equipment, hydrogen processing (e.g., for purity and compression), electricity input conversion (e.g., transformer and rectifier), water supply treatment (e.g., deionisation), and gas output (e.g., from oxygen output).
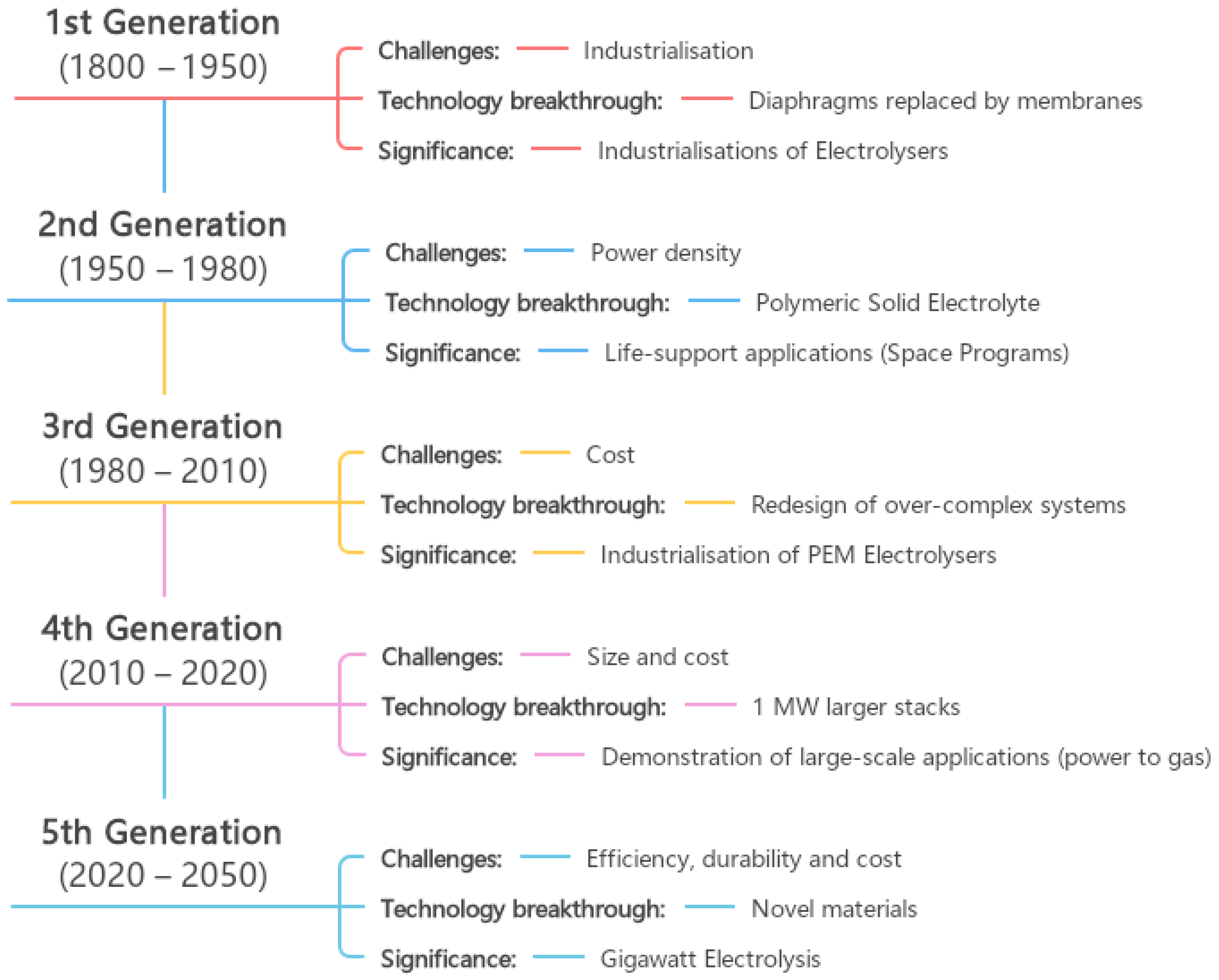
- 1st generation (1800–1950)
- 2nd generation (1950–1980)
- 3rd generation (1980–2010)
- 4th generation (2010–2020)
- 5th generation (post-2020)
3.4.1. Alkaline Electrolysis
- Cathode reaction (HER):
- Anode reaction (OER):
3.4.2. Anion Exchange Membrane Electrolysis
3.4.3. Proton Exchange Membrane Electrolysis
- Cathode reaction (HER):
- Anode reaction (OER):
3.4.4. Solid Oxide Electrolysis
- Cathode reaction (HER):
- Anode reaction (OER):
3.5. Analysis of Green Hydrogen Production Processes
3.5.1. Alkaline Water Electrolysis
- Current density: One of the major barriers to alkaline electrolysis of water is low current density [79]. They are currently between 0.2 and 0.8 A/cm2 and should be capable of reaching 2 to 3 A/cm2. This increase in value, however, should not come at the expense of decreased energy efficiency. These values can be obtained by reducing the diaphragm thickness and using electrode materials with high specific areas.
- Diaphragm: The thinner the diaphragm, the less resistance there is when transporting OH− ions from the cathode to the anode. This reduction has the potential to improve cell efficiency and reduce energy consumption. At the limit, this decrease can result in an increase in gas permeation, which raises safety concerns. Another issue is that there is less durability because orifices in the diaphragm are more likely to form, and there is less mechanical robustness. The thickness of the diaphragms of alkaline electrolysers is currently around 460 m; reducing this value to 50 m would increase the energy efficiency from 53% to 75% at 1 A/cm2.
3.5.2. Anion Exchange Membrane Water Electrolysis
- Membrane: Researchers are currently studying AEM membranes that possess advantageous characteristics, including strong mechanical, thermal, and chemical stability, as well as high ionic conductivity [12]. Additionally, they aim to develop membranes with reduced permeability to electrons and gases [12]. As a result, a trade-off between the desirable properties of the membrane and its cost is required. An additional notable drawback of an AEM involves the deterioration of polymers due to corrosion in alkaline electrolysers. This corrosion leads to a swift decline in the conductivity of both the membrane and ionomer present in the catalyst layer [12]. The ionic conductivity of an AEM has a significant impact on its performance because higher levels of ionic conductivity allow for much higher current densities and thus higher energy efficiency [12].
3.5.3. Proton Exchange Membrane Water Electrolysis
- Membrane: The membrane is a critical component of the PEM water electrolyser, and significant innovations or improvements in this area are required to increase energy efficiency and durability while lowering costs [79]. Reduced membrane thickness allows for increased energy efficiency, which leads to reduced electricity consumption. Currently, a Nafion N117 membrane is approximately 180 m thick, resulting in an energy efficiency loss of 25%, with a current density of 2 A/cm2 [12,103]. There are membranes with a thickness of 20 m, but they lack the required design for an electrolyser. At a current density of 2 A/cm2, such a reduction could result in a 6% reduction in energy efficiency losses. It is crucial to remember that a decrease in membrane thickness is related to a reduction in durability, as there may be a drop in mechanical properties and a higher likelihood of defects.
- Electrode materials: As another key component of the PEM electrolyser, electrodes require significant innovation, precious materials being a major barrier in their cost and scale-up [79]. As a result, significant solutions to replace or reduce such materials are required, including using abundant non-noble materials.
- Cell stack: Anodic PTLs use platinum as a coating to protect titanium from passivation, a process in which a film forms on the electrode, decreasing its activity and providing optimal interface strength. Titanium bipolar plates also have platinum and gold protective layers on the anode and cathode. Alternatives to titanium plates are required, based on materials such as niobium, tantalum, and eventually stainless steel approximations, but with stable protective coatings and no platinum or gold, which are expensive materials [12].
3.5.4. Solid Oxide Water Electrolysis
- Durability: Durability can be improved by increasing electrode activity at lower temperatures and optimising chemical and mechanical stability. Furthermore, the oxidation state of the electrode and nickel accumulation must be controlled, as well as the delamination issues associated with the use of LSM and LSF of the electrolyte.
4. Hydrogen Storage Processes
4.1. Introduction

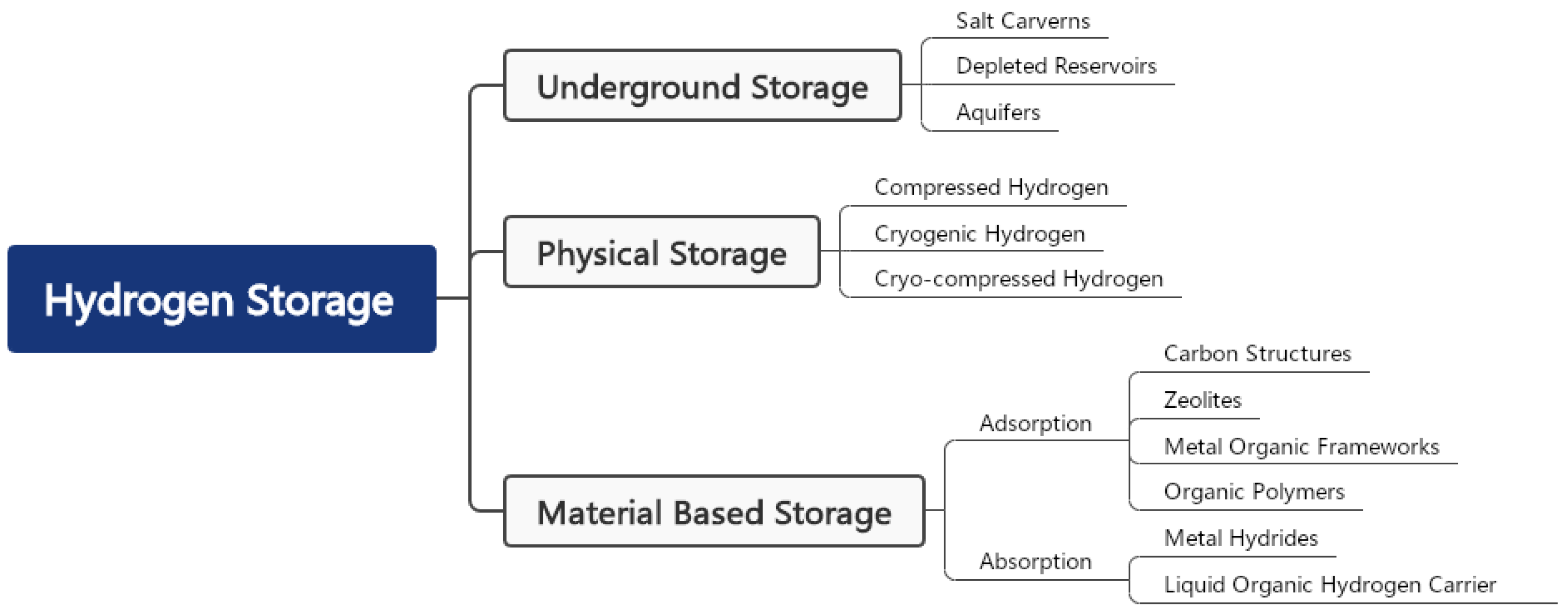
4.2. Underground Hydrogen Storage
4.2.1. Salt Caverns
Challenges
- Cavern development: The leaching and salt dissolution mining techniques encounter difficulties in managing resources and process-related problems. The procedure requires the use of low-salinity water to dissolve the salt, creating a geographical restriction, and the extracted brine needs to be managed due to its surplus production. Closed-loop systems are used by mineral processing operations to comply with regulations, but they may not be practical for significant amounts of brine. The creation of uneven caverns in subterranean hydrogen storage units poses difficulties regarding safety and efficiency. There are three factors that play a role in this process—geological parameters, construction techniques, and issues with tubing—and high levels of salt impurities are a significant contributor, as they can cause additional undesirable layers to form.
- Operation of cavern: The use of caverns in cycles presents difficulties in maintaining their stability due to the effects of mechanical and thermal loading on the surrounding structure. The frequency of operation cycles affects the fracture stress of the structure, faster cycles causing quicker stress changes. The complexity of the stability criteria is increased by the heterogeneity and variable mechanical properties of the structure. Injecting gas at high temperatures can additionally cause thermal stress, leading to micro-fractures or even roof collapse in extreme cases. Thus, temperature fluctuations of the injected hydrogen must be taken into account during both the injection and withdrawal cycles.
Geological Criteria
- Biogeochemical reactions: Despite the fact that salt does not chemically react with hydrogen, impurities that are present within the formation may react with hydrogen, decreasing the amount and quality of the stored hydrogen. Anhydrite, which is found in salt formations, is extremely hygroscopic, and in the presence of hydrogen, can produce hydrogen sulphide, which is both toxic and corrosive, and can contaminate the stored hydrogen.
4.2.2. Aquifers
Challenges
- Site characterisation: The cost of characterising a site, which includes drilling and analysing data, is estimated to make up approximately 20% of CAPEX, and this cost is even higher for offshore aquifers. However, there is an economic risk associated with drilling before determining whether a site is suitable for hydrogen storage. To reduce costs, 3D-printed cores could potentially be used to replicate samples from previous wells. Borehole drilling, as for natural gas storage, is assumed to follow the best practices. Creating an open-source database of previous drilled sites could also help reduce costs by providing parameters for case development.
- Cushion hydrogen gas reduction: Cushion hydrogen gas provides for 45 to 80% of the aquifer’s volumetric storage capacity and is considered to be responsible for 52% of the costs. As a result, cost-cutting measures are required. One possible approach is to strategically place many shallow extraction wells on the reservoir’s roof. This arrangement has the potential to recover up to 78% of the initial H2 injection. While successful, accurate modelling requires exact site knowledge. Alternatively, employing nitrogen as a cushion gas could reduce costs, but the decision should take into account the higher complexity of gas mixing as well as the additional expenses of separation operations during hydrogen gas extraction.
- Viscous fingering: Viscous fingering can lead to hydrogen loss during storage and is a potential issue that needs to be addressed. This occurs when the mobility ratio of hydrogen is higher than that of saline water, resulting in lateral migration beyond the spill point of the plume. Several methods are recommended to prevent this, including adjusting the gas injection rate, using a more dense cushion gas, and selecting deeply steeping anticline structures. Decreasing the gas injection rate can allow gravitational and capillary forces to overcome viscous forces, but this process can take several years to complete. The use of a denser cushion gas during the initial displacement of reservoir water would lower the chance of finger development. Additionally, selecting deeply steeping anticline structures can also help mitigate viscous fingering.
- Upconing: Aquifer hydrogen storage is often limited by upconing, which occurs when most of the fluid withdrawn is water. This phenomenon happens due to an increase in the pressure gradient between the interface of water/hydrogen and the well, leading to the upward movement of the water phase and consequent withdrawal of hydrogen mixed with water. This can result in more challenging separation and potential well shutdown. To mitigate the effects of upconing, alternative cushion gases with higher density and viscosity than hydrogen gas and well configurations with extraction points placed higher up can be considered.
Geological Criteria
- Permeability of reservoir rock: The inherent permeability of a reservoir rock is established by factors like grain size and packing and is typically calculated through Darcy’s Law. Nevertheless, in real-world situations, other variables like heterogeneous reservoirs and multi-phase interactions must be accounted for. The permeability of the reservoir rock can impact the speed at which fluids are injected/withdrawn, the amount of storage space available, and the impact of gas mixing. Still, even if the permeability is enhanced, the thermophysical phenomena/process may limit the hydrogen injection/withdrawal rates.
- Caprock sealing capacity: Unlike depleted oil and gas deposits, aquifers require thorough evaluation to ensure they are impermeable. The effectiveness of caprock sealing is determined by the caprock’s permeability and the presence of faults, which can create pathways for hydrogen gas migration. A low permeability layer is crucial to prevent hydrogen gas leakage, and faults can facilitate the movement of gas. Tectonic traps containing aquifers in steep domes are ideal for obtaining high-quality hydrogen storage. Ignoring existing faults may result in a significant amount of unrecoverable hydrogen. Therefore, fault information should be taken into account during surveys, which increases costs but maximises hydrogen gas recovery.
- Biogeochemical reactions: In the initial assessment stage of assessing an aquifer, it is crucial to confirm the absence of bacteria that could either consume or contaminate hydrogen. Methanogenic bacteria (MB) can still make use of CO2 and do not necessarily impede the utilisation of the formation if they are present. On the other hand, if sulphate-reducing bacteria (SRB) are detected, additional equipment for desulphurisation may be necessary, and if such equipment is not present, it is not advisable to continue with development due to the resulting increasing costs.
4.2.3. Depleted Oil and Gas Reservoirs
Challenges
- Re-purposing of infrastructure: Re-purposing the current infrastructure for hydrogen storage presents various obstacles that involve the formation of internal flaws, hydride creation, and steel embrittlement. Materials commonly used in the oil and gas sector, including low and high-alloy steels, plain carbon steels, and stainless steels, are vulnerable to the negative effects of hydrogen-induced failure mechanisms.
- Multiphase–multicomponent mixing: The mixing of different components and phases in a reservoir occurs due to various processes and is affected by factors like fluid pressure and velocity. Such mixing can cause the extracted hydrogen to be contaminated and lead to increased costs for separation. Therefore, it is important to have accurate models that can describe these processes accurately, and this is a key area of research for many research groups.
Geological Criteria
- Methanogenic and sulphate-reducing bacteria: Hydrogen reservoirs can host methanogenic bacteria and SRB, which can negatively impact the quantity and quality of stored hydrogen through biogeochemical reactions. MB consume CO2 and hydrogen, producing methane and water, indicating that the use of CO2 as a cushion gas can lead to increased methane production. The presence of SRB in reservoirs can lead to a decrease in the quality and quantity of stored hydrogen due to the production of toxic sulphide, erosion of structural steel, and degradation of the hydrogen quality. High temperatures promote the SRB growth, which is why deep reservoirs are preferred for hydrogen storage. Reservoir rocks cemented with anhydrite and gypsum should be avoided, as they promote the SRB growth.
- Caprock tightness: While it is generally assumed that gas storage over a long period of time results in caprock tightness, the level of tightness is not entirely guaranteed due to the differing properties of fluids. In order to obtain an accurate understanding of interfacial properties, it is important to take into account additional factors such as contact angle, wettability, and capillary pressure within the pores. Furthermore, the salinity of the water in the reservoir can impact the interfacial tension and therefore should be taken into consideration in the used models.
4.3. Physical Storage
4.3.1. Compressed Hydrogen

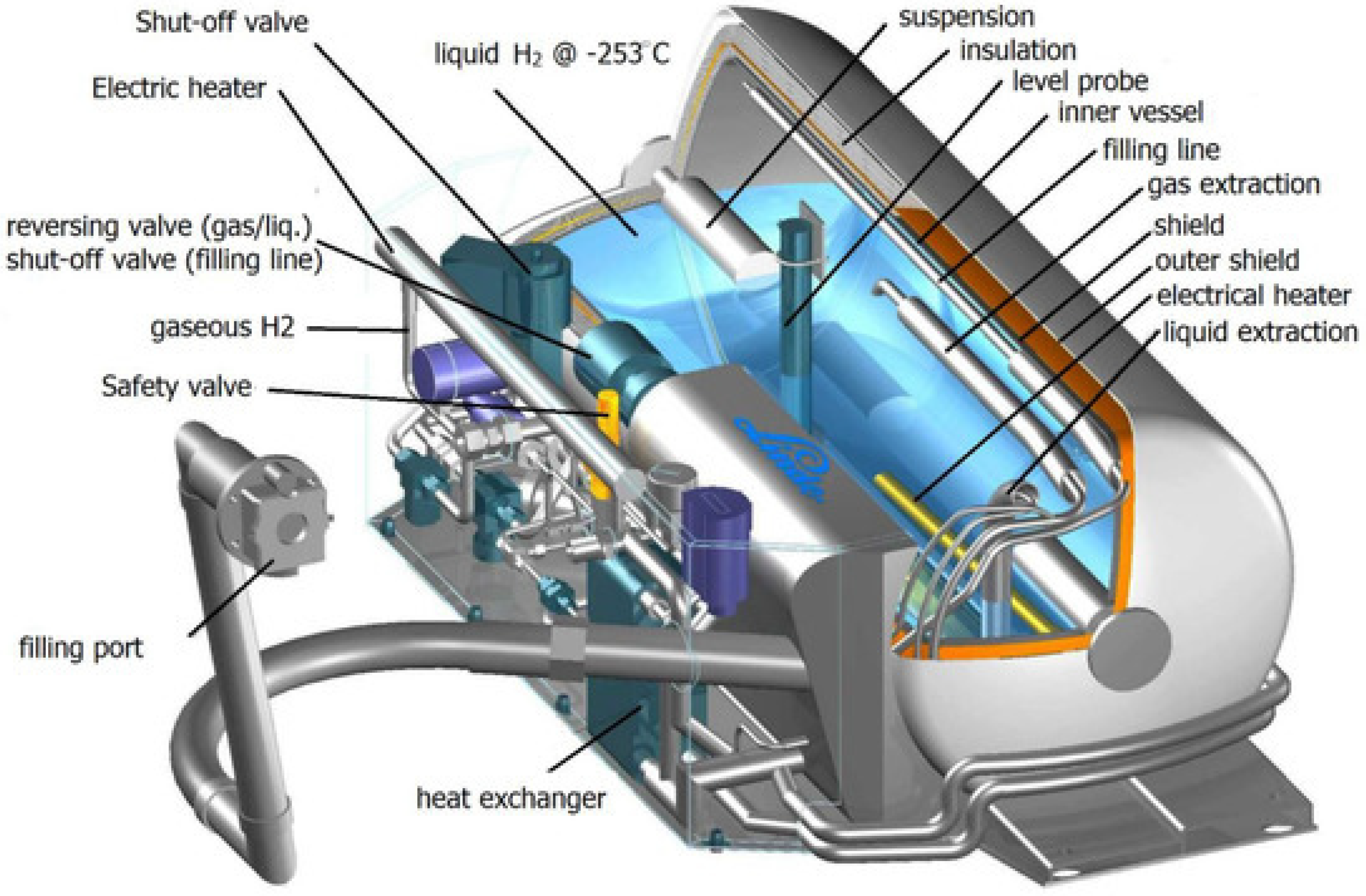
4.3.2. Cryogenic Hydrogen
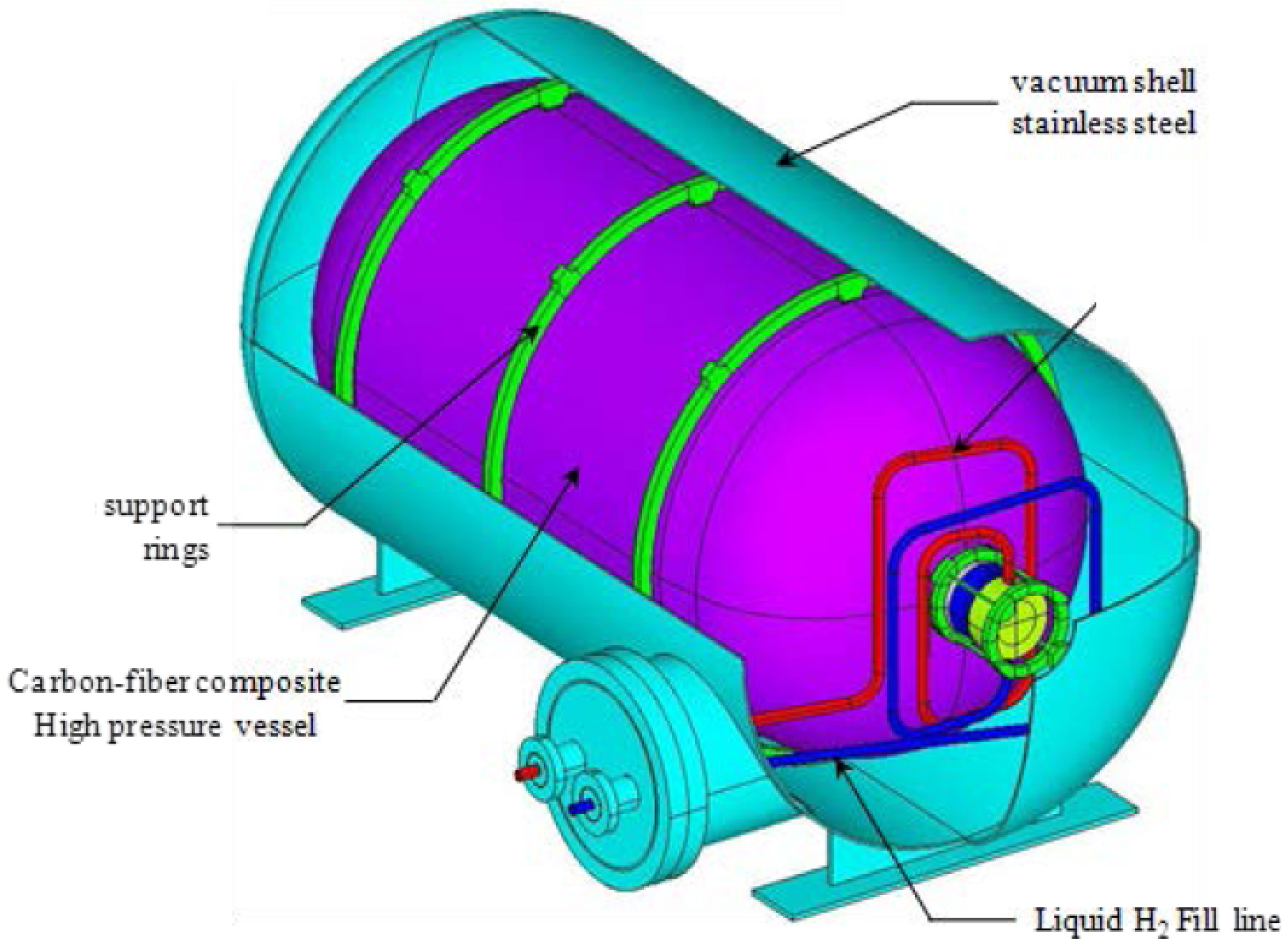
4.3.3. Cryo-Compressed Hydrogen
4.4. Material-Based Hydrogen Storage
4.4.1. Adsorbents
Carbon Structures
Zeolites
Metal–Organic Frameworks
4.4.2. Metal Hydrides
4.4.3. Liquid Organic Hydrogen Carriers
4.5. Analysis of Hydrogen Storage Processes
5. Hydrogen Transportation Processes
5.1. Introduction
5.2. Trucks
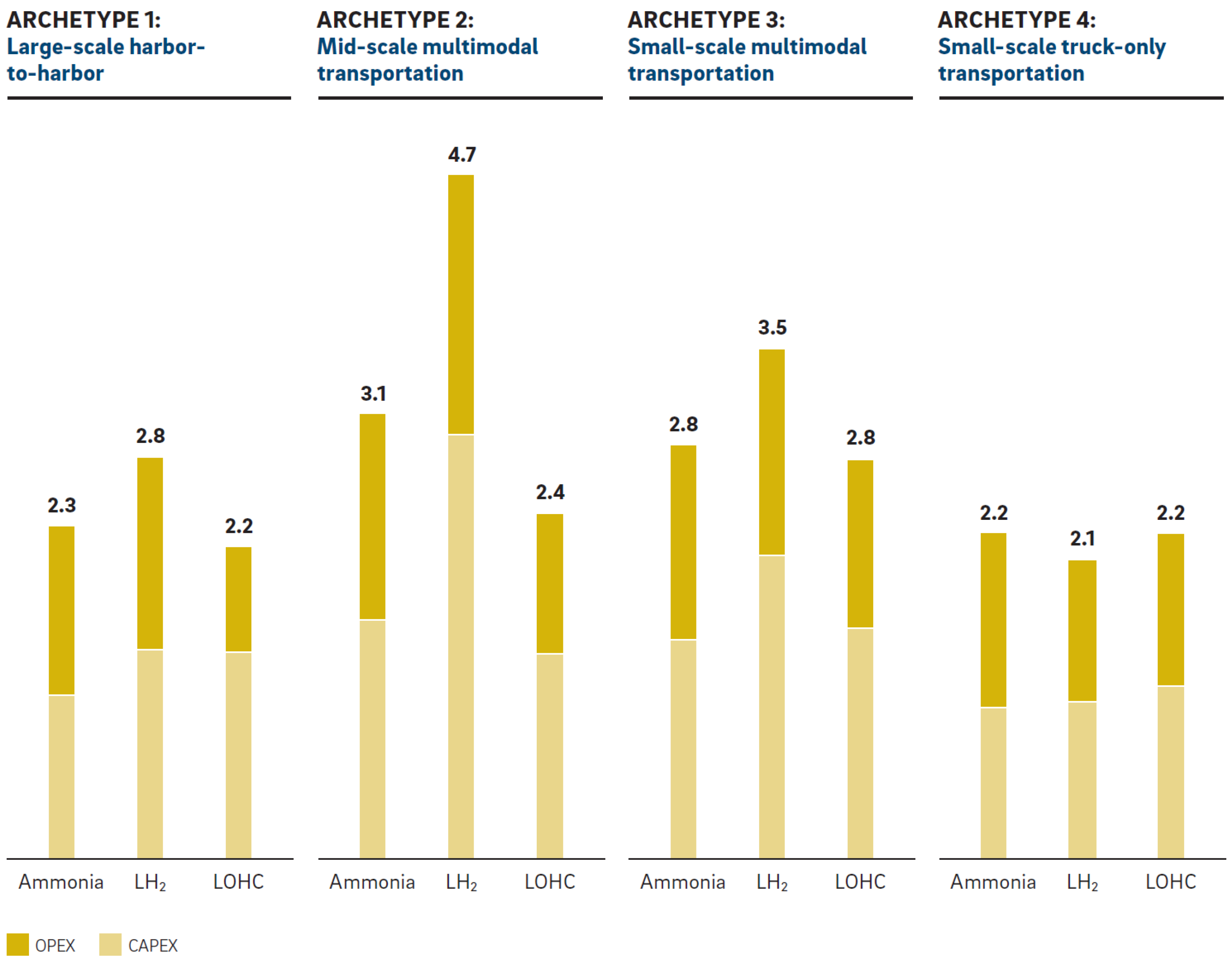
5.3. Ship
5.3.1. Ammonia
5.3.2. Liquid Hydrogen

5.3.3. Liquid Organic Hydrogen Carriers
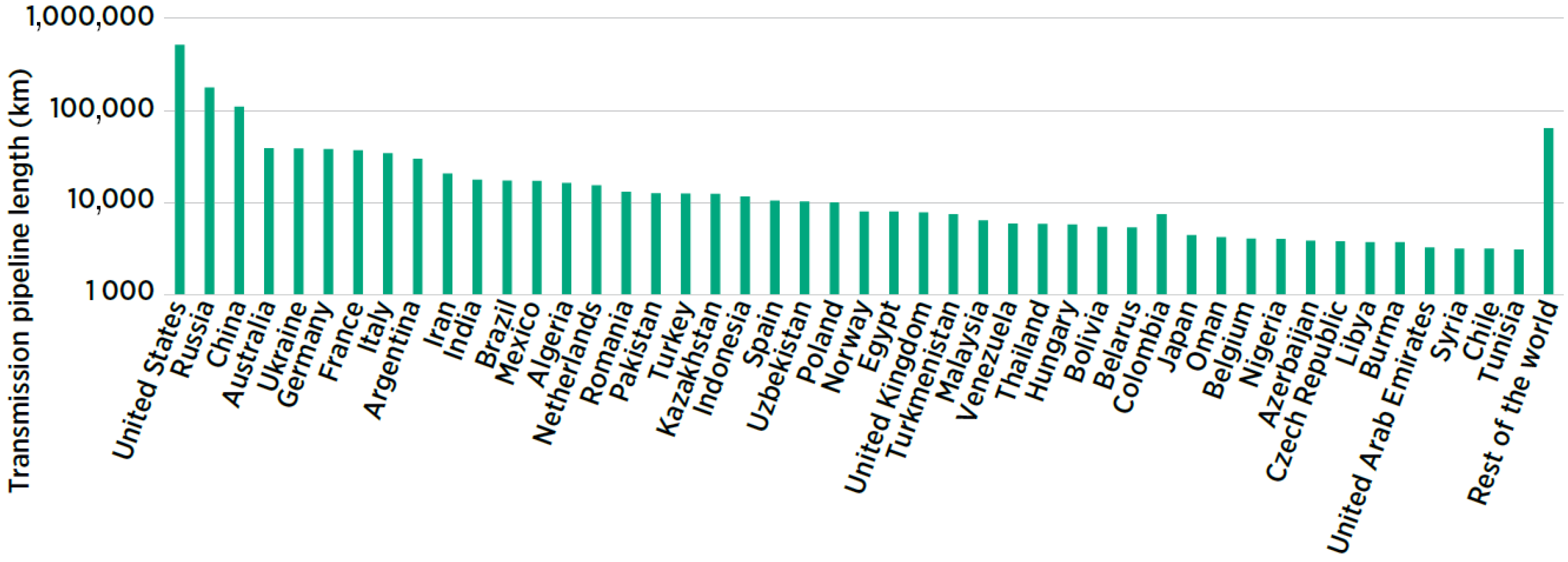
5.4. Pipelines
5.4.1. Repurposing Natural Gas Pipelines
5.4.2. Blending with Natural Gas
5.5. Analysis of Hydrogen Transportation Processes
6. Use of Hydrogen in Energy Conversion Processes
6.1. Introduction
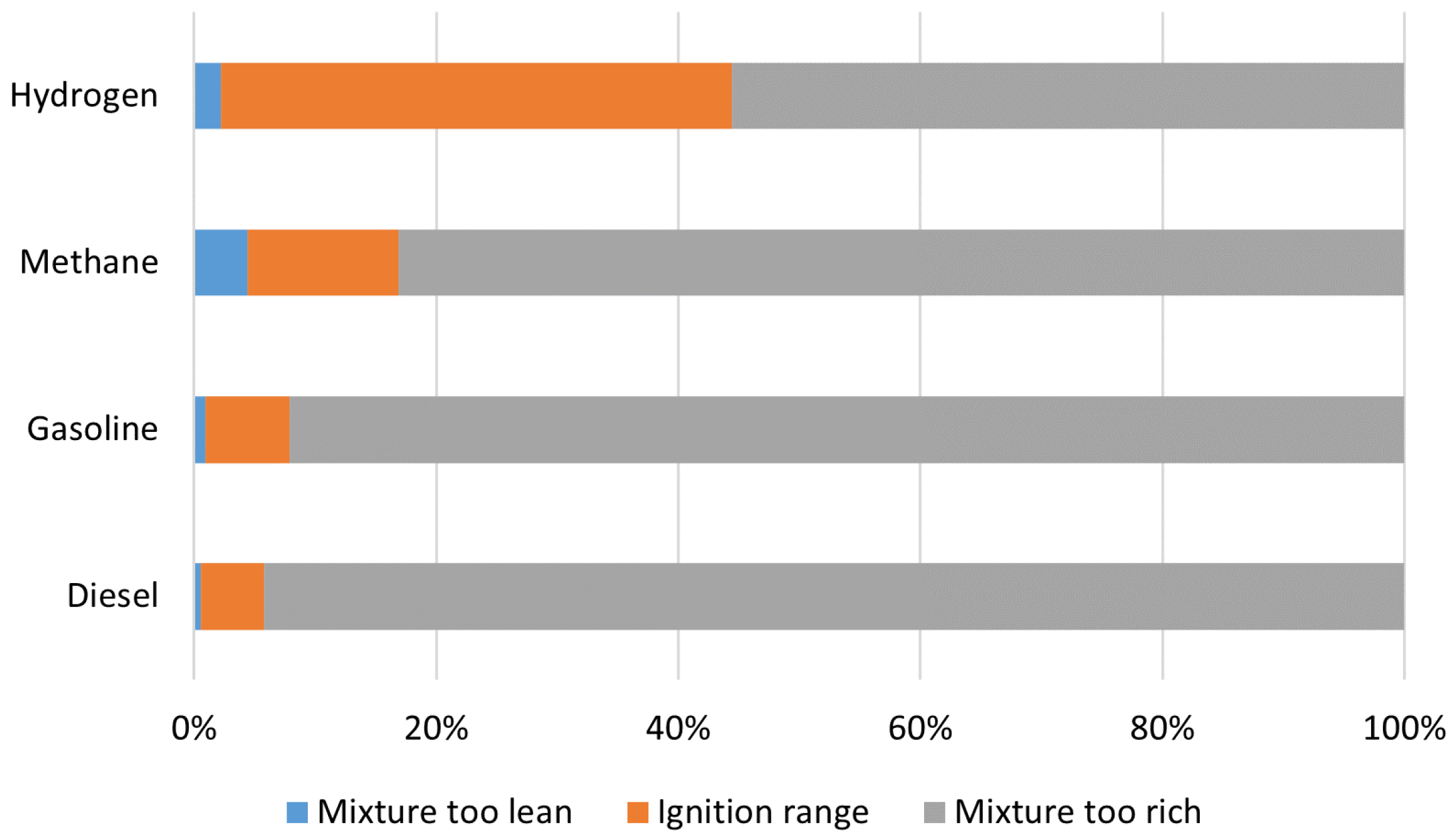
6.2. Hydrogen Internal Combustion Engines
6.3. Hydrogen Gas Turbines
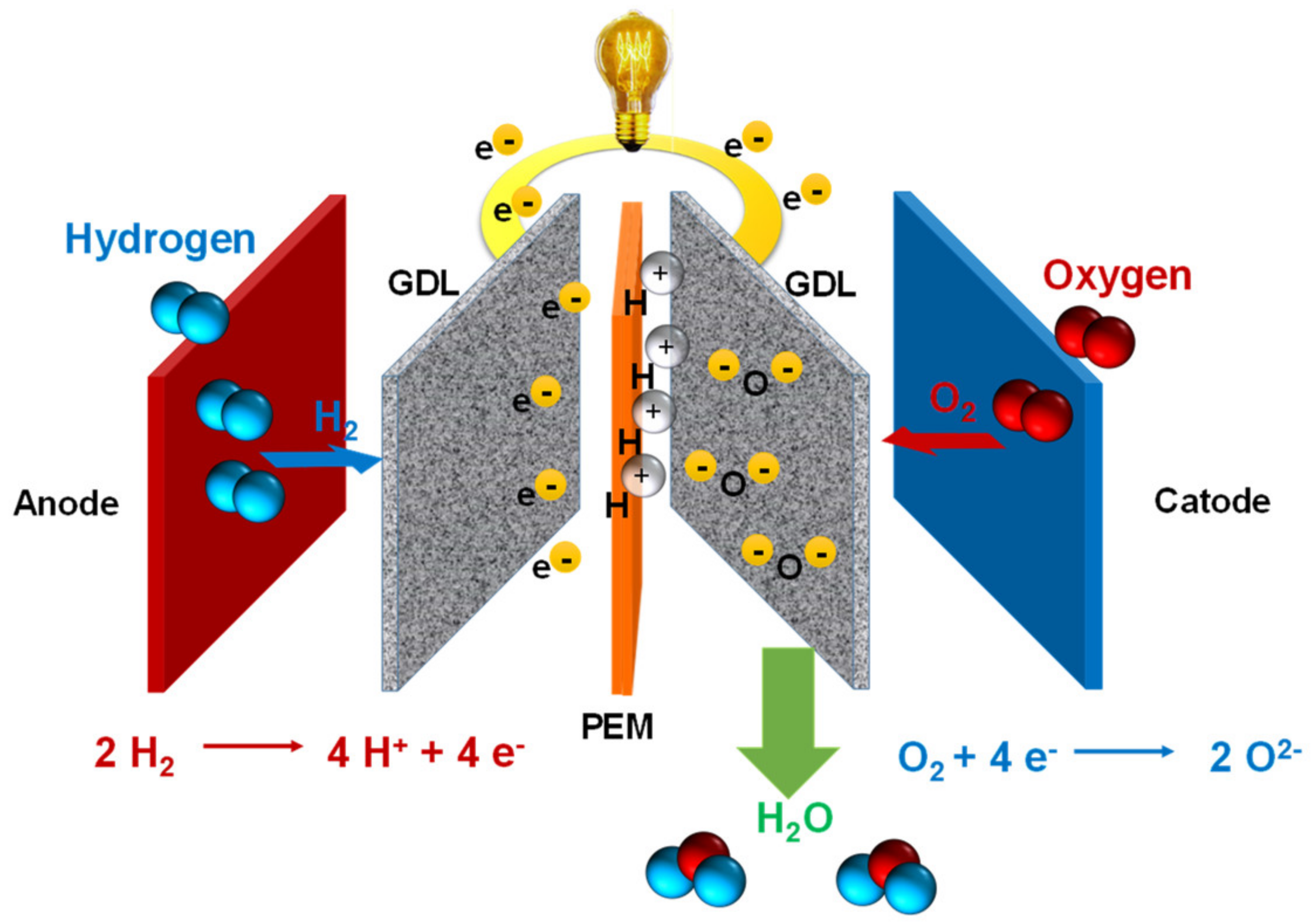
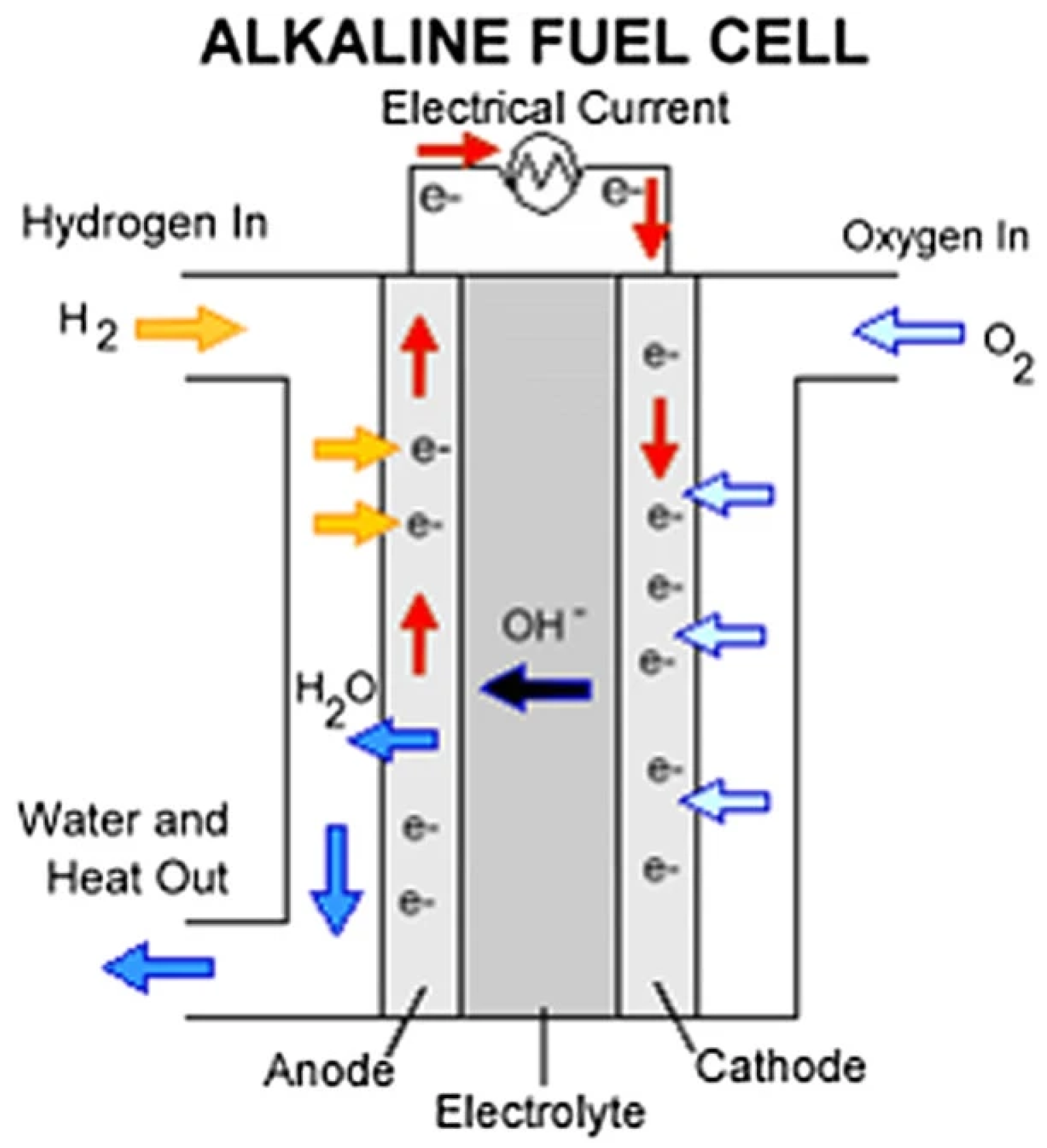
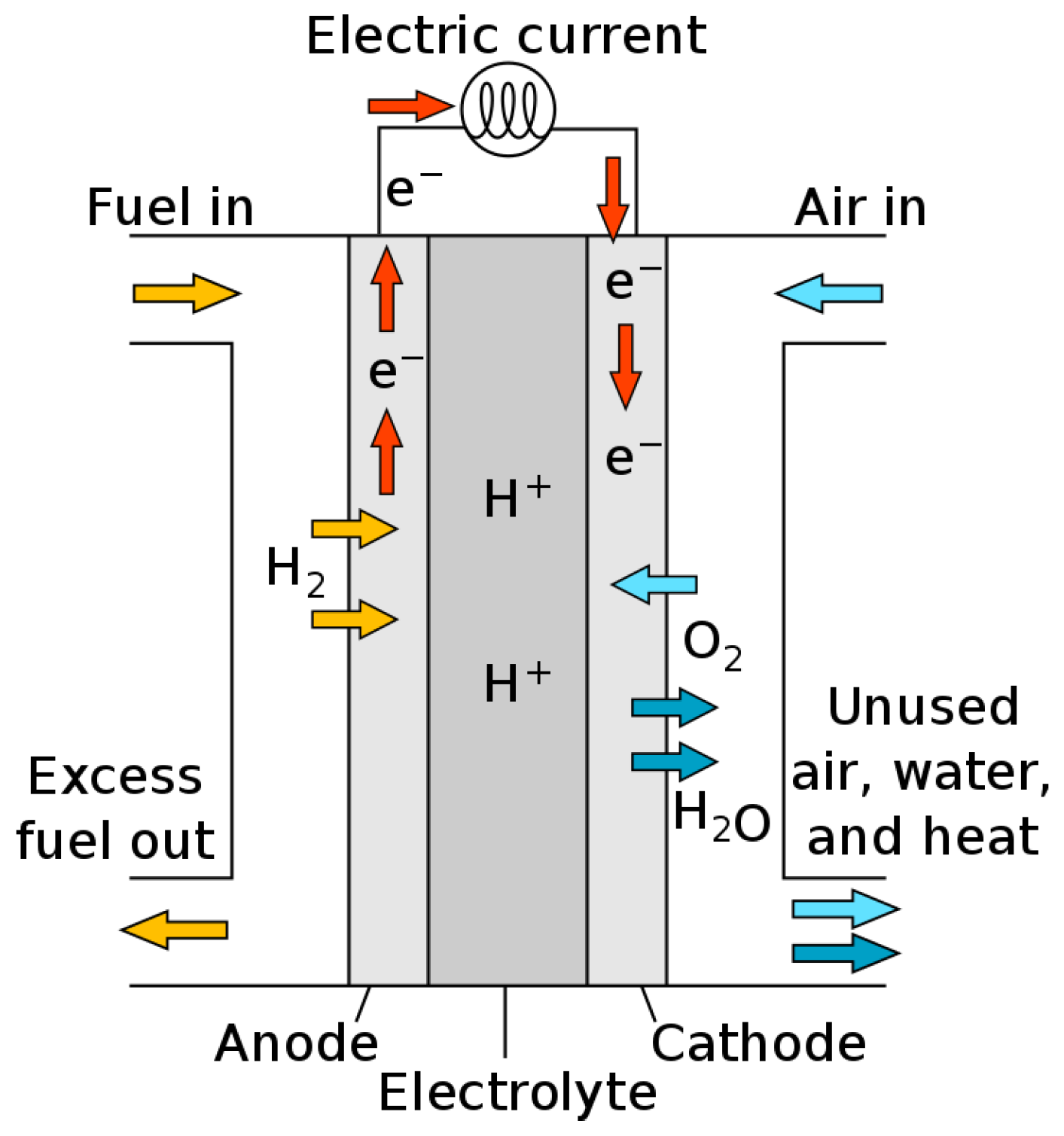
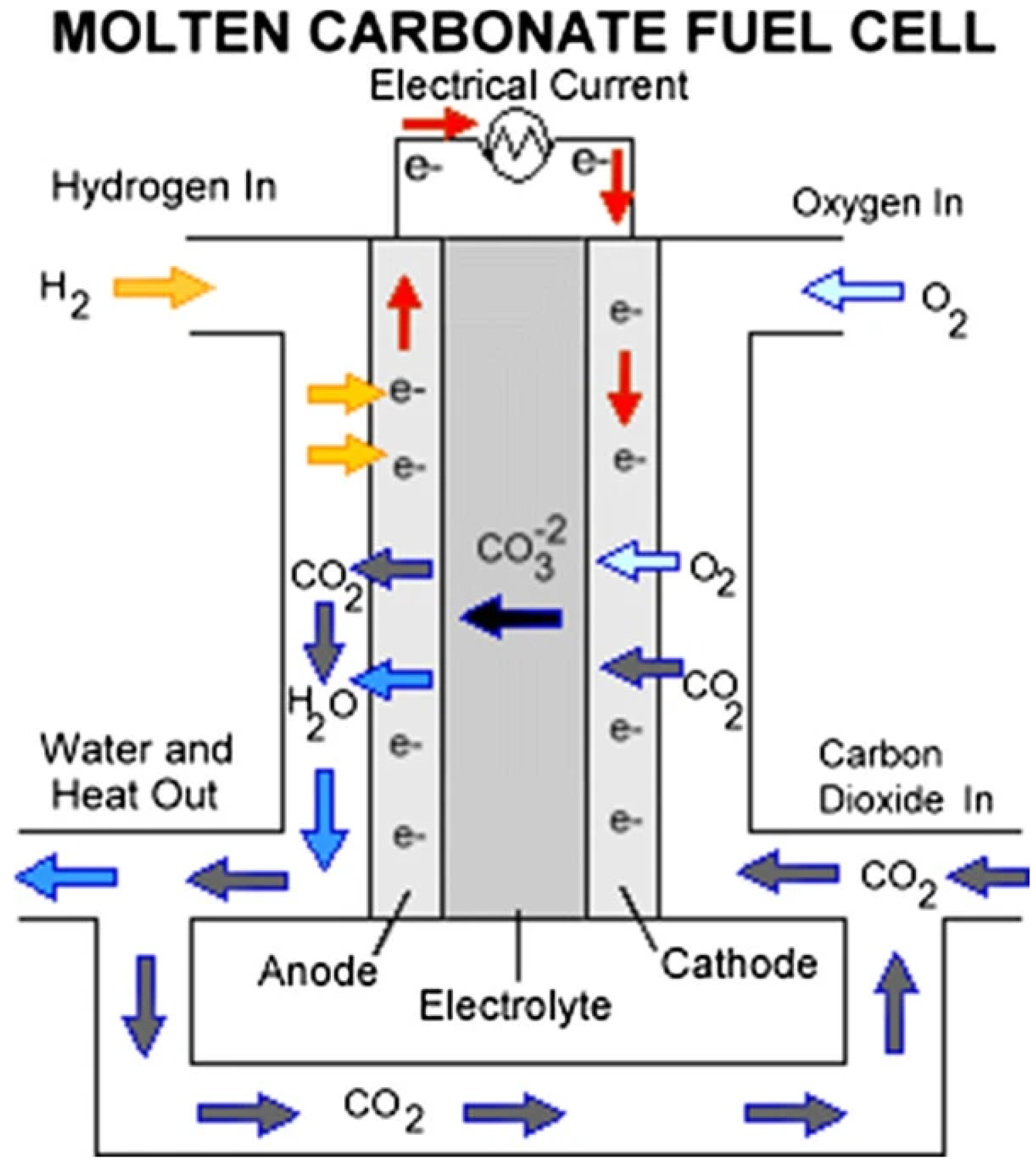
6.4. Fuel Cells
6.4.1. Alkaline Fuel Cells
6.4.2. Proton Exchange Membrane Fuel Cells
6.4.3. Direct Methanol Fuel Cells
6.4.4. Phosphoric Acid Fuel Cells
6.4.5. Molten Carbonate Fuel Cells
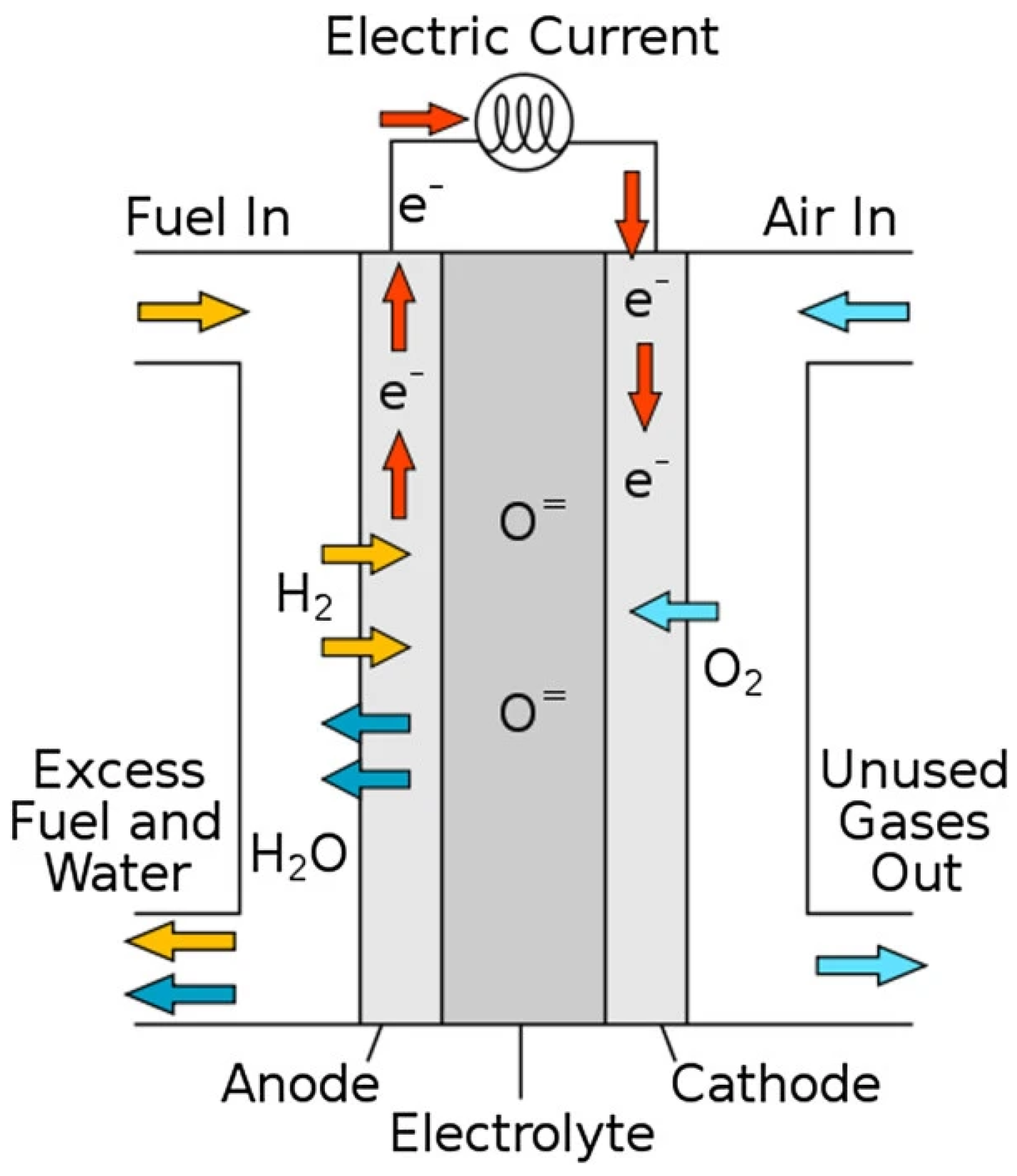
6.4.6. Solid Oxide Fuel Cells
6.4.7. Recent Research in PEMFCs
6.5. Discussion on the Processes of Energy Conversion
7. Conclusions and Outlook
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| CH4 | Methane |
| CO2 | Carbon Dioxide |
| CO | Carbon Monoxide |
| C | Carbon |
| H2 | Hydrogen (molecular) |
| H | Hydrogen (atomic) |
| K2CO3 | Potassium Carbonate |
| KOH | Potassium Hydroxide |
| NaOH | Sodium Hydroxide |
| O2 | Oxygen (molecular) |
| O2− | Oxide |
| OH− | Hydroxide |
| YSZ | Yttria-Stabilized Zirconia |
| ZrO2 | Zirconium Dioxide |
| ATR | Autothermal Reforming |
| AFC | Alkaline Fuel Cell |
| bgs | Below Ground Surface |
| BP | Bipolar Plate |
| BEV | Battery Electric Vehicle |
| CAPEX | Capital Expenditure |
| CCUS | Carbon Capture, Utilisation, and Storage |
| CcH2 | Cryo-Compressed Hydrogen |
| CGH2 | Compressed Gas Hydrogen |
| DI | Direct Injection |
| ESS | Energy Storage System |
| FCV | Fuel Cell Vehicles |
| GDL | Gas Diffusion Layer |
| GH2 | Green Hydrogen |
| GHG | Greenhouse Gas |
| HER | Hydrogen Evolution Reaction |
| HHV | Higher Heating Value |
| ISO | International Standards Organisation |
| ICE | Internal Combustion Engine |
| LH2 | Liquid Hydrogen |
| LHV | Lower Heating Value |
| LNG | Liquefied Natural Gas |
| LPG | Liquefied Petroleum Gas |
| LOHC | Liquid Organic Hydrogen Carrier |
| MB | Methanogenic Bacteria |
| MOF | Metal–Organic Framework |
| OER | Oxygen Evolution Reaction |
| PEM | Proton Exchange Membrane |
| PEMFC | Proton Exchange Membrane Fuel Cell |
| POX | Partial Oxidation |
| PTL | Porous Transport Layer |
| PFI | Port-Fuel Injection |
| R&D | Research and Development |
| RES | Renewable Energy Sources |
| SMR | Steam Methane Reforming |
| SOEC | Solid Oxide Electrolysis Cell |
| SRB | Sulfate-Reducing Bacteria |
| TC | Technical Committee |
| UHS | Underground Hydrogen Storage |
References
- UN—United Nations. United Nations Framework Convention on Climate Change. 1992. Available online: https://unfccc.int/resource/docs/convkp/conveng.pdf (accessed on 10 July 2023).
- UNFCCC—United Nations Framework Convention on Climate Change. Kyoto Protocol; UNFCCC: Bonn, Germany, 1997. [Google Scholar]
- UNFCCC—United Nations Framework Convention on Climate Change. Paris Agreement; UNFCCC: Bonn, Germany, 2015. [Google Scholar]
- UNFCCC—United Nations Framework Convention on Climate Change. Glasgow Climate Pact; UNFCCC: Bonn, Germany, 2021. [Google Scholar]
- Ishaq, H.; Dincer, I.; Crawford, C. A review on hydrogen production and utilization: Challenges and opportunities. Int. J. Hydrogen Energy 2022, 47, 26238–26264. [Google Scholar] [CrossRef]
- Razi, F.; Dincer, I. Challenges, opportunities and future directions in hydrogen sector development in Canada. Int. J. Hydrogen Energy 2022, 47, 9083–9102. [Google Scholar] [CrossRef]
- Hjeij, D.; Biçer, Y.; Koç, M. Hydrogen strategy as an energy transition and economic transformation avenue for natural gas exporting countries: Qatar as a case study. Int. J. Hydrogen Energy 2022, 47, 4977–5009. [Google Scholar] [CrossRef]
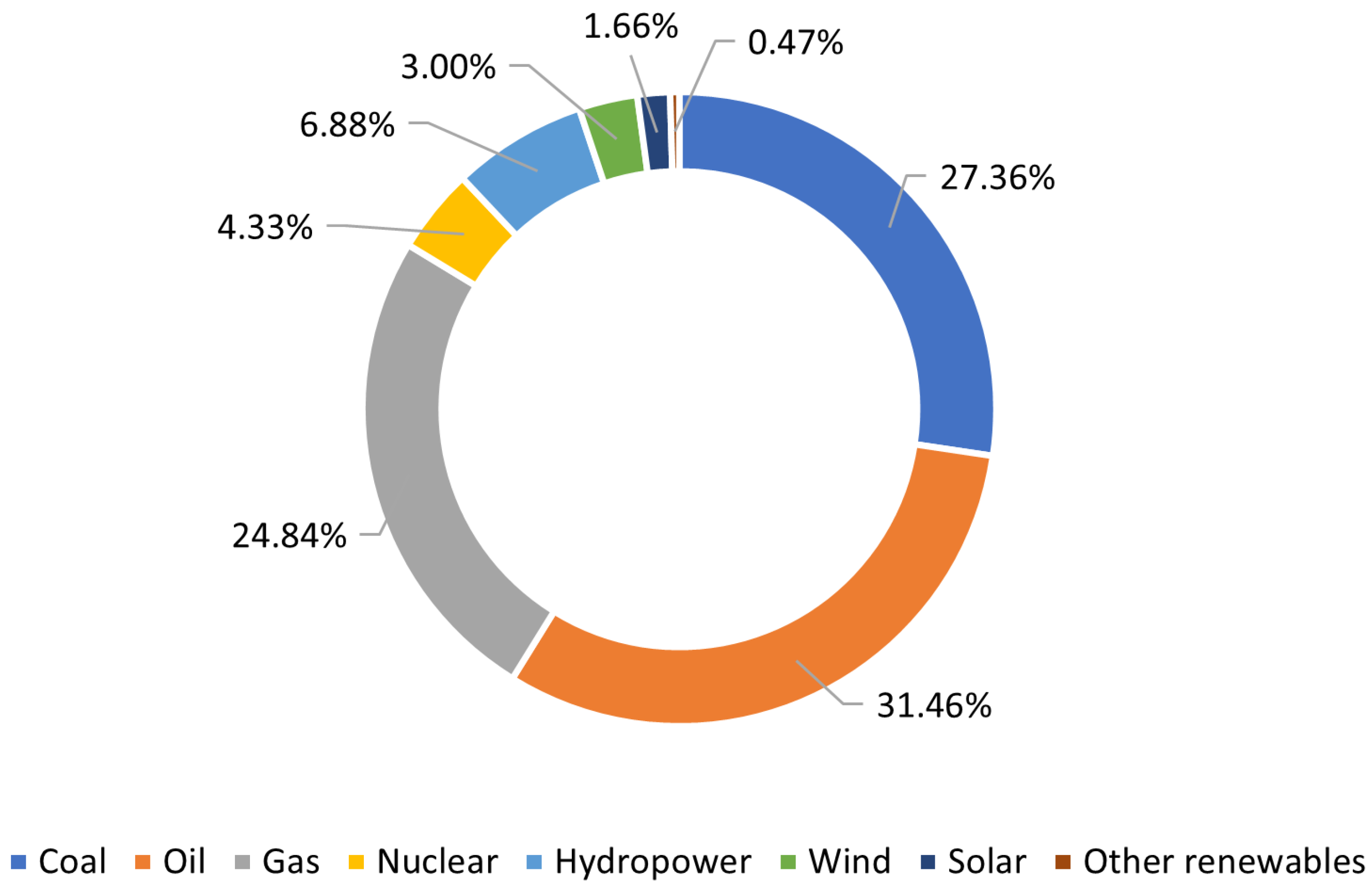
- Our World in Data. Primary Energy Consumption by Source. 2021. Available online: https://ourworldindata.org/grapher/primary-energy-source-bar?stackMode=absolute&time=latest&country=~OWID_WRL (accessed on 20 December 2022).
- Edwards, R.L.; Font-Palma, C.; Howe, J. The status of hydrogen technologies in the UK: A multi-disciplinary review. Sustain. Energy Technol. Assess. 2021, 43, 100901. [Google Scholar] [CrossRef]
- IEA—International Energy Agency. Global Hydrogen Demand by Sector in the Net Zero Scenario, 2019–2030. 2022. Available online: https://www.iea.org/data-and-statistics/charts/global-hydrogen-demand-by-sector-in-the-net-zero-scenario-2019-2030 (accessed on 20 December 2022).
- Capurso, T.; Stefanizzi, M.; Torresi, M.; Camporeale, S.M. Perspective of the role of hydrogen in the 21st century energy transition. Energy Convers. Manag. 2022, 251, 114898. [Google Scholar] [CrossRef]
- IRENA—International Renewable Energy Agency. Green Hydrogen Cost Reduction: Scaling Up Electrolysers to Meet the 1.5 °C Climate Goal, 2020. Available online: https://www.irena.org/-/media/Files/IRENA/Agency/Publication/2020/Dec/IRENA_Green_hydrogen_cost_2020.pdf (accessed on 10 July 2023).
- IEA—International Energy Agency. Electrolysers. 2022. Available online: https://www.iea.org/reports/electrolysers (accessed on 20 December 2022).
- Mankins, J.C. Technology Readiness Levels: A White Paper. 1995. Available online: https://www.researchgate.net/publication/247705707_Technology_Readiness_Level_-_A_White_Paper (accessed on 10 July 2023).
- International Energy Agency. Energy Technology Perspectives—Clean Energy Technology Guide. 2020. Available online: https://iea.blob.core.windows.net/assets/355d9b26-b38c-476c-b9fa-0afa34742800/iea_technology-guide-poster.pdf (accessed on 14 October 2022).
- Dawood, F.; Anda, M.; Shafiullah, G.M. Hydrogen production for energy: An overview. Int. J. Hydrogen Energy 2020, 45, 3847–3869. [Google Scholar] [CrossRef]
- Hydropole. About Hydrogen. Available online: https://hydropole.ch/en/hydrogen/abouth2/ (accessed on 21 December 2022).
- Shell Deutschland Oil GmbH. Shell Hydrogen Study: Energy of the Future? Sustainable Mobility through Fuel Cells and H2. 2017. Available online: https://epub.wupperinst.org/frontdoor/deliver/index/docId/6786/file/6786_Hydrogen_Study.pdf (accessed on 10 July 2023).
- Hydrogen Europe. Hydrogen: Enabling a Zero-Emission Society. 2022. Available online: https://hydrogen.revolve.media/2022/ (accessed on 10 July 2023).
- Hydropole. The History of Hydrogen. Available online: https://hydropole.ch/en/hydrogen/ (accessed on 21 December 2022).
- Ozturk, M.; Dincer, I. A comprehensive review on power-to-gas with hydrogen options for cleaner applications. Int. J. Hydrogen Energy 2021, 46, 31511–31522. [Google Scholar] [CrossRef]
- Webster, D. What Really Felled the Hindenburg? 2017. Available online: https://www.smithsonianmag.com/smithsonian-institution/80th-anniversary-hindenburg-disaster-mysteries-remain-180963107/ (accessed on 23 December 2022).
- Clean Hydrogen Partnership. Our Story. 2021. Available online: https://wayback.archive-it.org/12090/20220602144938/https://www.clean-hydrogen.europa.eu/about-us/our-story_en (accessed on 23 December 2022).
- Toyota. Toyota Ushers in the Future with Launch of ‘Mirai’ Fuel Cell Sedan. 2014. Available online: https://global.toyota/en/detail/4198334 (accessed on 23 December 2022).
- Hydrogen Valleys. The Hydrogen Valley Platform. Available online: https://h2v.eu/about-us (accessed on 23 December 2022).
- Avargani, V.M.; Zendehboudi, S.; Saady, N.M.C.; Dusseault, M.B. A comprehensive review on hydrogen production and utilization in North America: Prospects and challenges. Energy Convers. Manag. 2022, 269, 115927. [Google Scholar] [CrossRef]
- Rizi, H.A.Y.; Shin, D. Green Hydrogen Production Technologies from Ammonia Cracking. Energies 2022, 15, 8246. [Google Scholar] [CrossRef]
- Aziz, M. Liquid hydrogen: A review on liquefaction, storage, transportation, and safety. Energies 2021, 14, 5917. [Google Scholar] [CrossRef]
- Engineering ToolBox. Hydrogen—Thermophysical Properties. 2008. Available online: https://www.engineeringtoolbox.com/hydrogen-d_1419.html (accessed on 5 January 2023).
- Stępień, Z. A comprehensive overview of hydrogen-fueled internal combustion engines: Achievements and future challenges. Energies 2021, 14, 6504. [Google Scholar] [CrossRef]
- Qazi, U.Y. Future of Hydrogen as an Alternative Fuel for Next-Generation Industrial Applications; Challenges and Expected Opportunities. Energies 2022, 15, 4741. [Google Scholar] [CrossRef]
- Martins, J.; Brito, F.P. Alternative Fuels for Internal Combustion Engines. Energies 2020, 13, 4086. [Google Scholar] [CrossRef]
- Gao, W.; Fu, Z.; Li, Y.; Li, Y.; Zou, J. Progress of Performance, Emission, and Technical Measures of Hydrogen Fuel Internal-Combustion Engines. Energies 2022, 15, 7401. [Google Scholar] [CrossRef]
- Pemberton, R.; Giannis, S. Unravelling the Hydrogen Regulatory Framework: Approval Process for Type IV Onboard Vehicle Hydrogen Fuel Containers; NPL Report 10.47120/npl.ENG70; National Physics Laboratory: Middlesex, UK, 2021. [CrossRef]
- Hoecke, L.V.; Laffineur, L.; Campe, R.; Perreault, P.; Verbruggen, S.W.; Lenaerts, S. Challenges in the use of hydrogen for maritime applications. Energy Environ. Sci. 2021, 14, 815–843. [Google Scholar] [CrossRef]
- Lan, H.; Wang, G.; Zhao, K.; He, Y.; Zheng, T. Review on the Hydrogen Dispersion and the Burning Behavior of Fuel Cell Electric Vehicles. Energies 2022, 15, 7295. [Google Scholar] [CrossRef]
- Ustolin, F.; Paltrinieri, N.; Berto, F. Loss of integrity of hydrogen technologies: A critical review. Int. J. Hydrogen Energy 2020, 45, 23809–23840. [Google Scholar] [CrossRef]
- Mahant, B.; Linga, P.; Kumar, R. Hydrogen Economy and Role of Hythane as a Bridging Solution: A Perspective Review. Energy Fuels 2021, 35, 15424–15454. [Google Scholar] [CrossRef]
- Li, H.; Cao, X.; Liu, Y.; Shao, Y.; Nan, Z.; Teng, L.; Peng, W.; Bian, J. Safety of hydrogen storage and transportation: An overview on mechanisms, techniques, and challenges. Energy Rep. 2022, 8, 6258–6269. [Google Scholar] [CrossRef]
- ISO/TC 197; Hydrogen Technologies. ISO—International Organization for Standardization: Geneva, Switzerland, 2023. Available online: https://www.iso.org/committee/54560.html (accessed on 8 February 2023).
- ISO/TR 15916:2015; Basic Considerations for the Safety of Hydrogen Systems. ISO—International Organization for Standardization: Geneva, Switzerland, 2020. Available online: https://www.iso.org/standard/56546.html?browse=tc (accessed on 8 February 2023).
- ISO 14687:2019; Hydrogen Fuel Quality—Product Specification. ISO—International Organization for Standardization: Geneva, Switzerland, 2021. Available online: https://www.iso.org/standard/69539.html?browse=tc (accessed on 8 February 2023).
- ISO 22734:2019; Hydrogen Generators Using Water Electrolysis—Industrial, Commercial, and Residential Applications. ISO—International Organization for Standardization: Geneva, Switzerland, 2021. Available online: https://www.iso.org/standard/69212.html?browse=tc (accessed on 8 February 2023).
- ISO 19881:2018; Gaseous Hydrogen—Land Vehicle Fuel Containers. ISO—International Organization for Standardization: Geneva, Switzerland, 2021. Available online: https://www.iso.org/standard/65029.html?browse=tc (accessed on 8 February 2023).
- ISO 13985:2006; Liquid Hydrogen— Land Vehicle Fuel Tanks. ISO—International Organization for Standardization: Geneva, Switzerland, 2022. Available online: https://www.iso.org/standard/39892.html?browse=tc (accessed on 8 February 2023).
- ISO 23273:2013; Fuel Cell Road Vehicles—Safety Specifications—Protection against Hydrogen Hazards for Vehicles Fuelled with Compressed Hydrogen. ISO—International Organization for Standardization: Geneva, Switzerland, 2019. Available online: https://www.iso.org/standard/64047.html (accessed on 8 February 2023).
- IEA—International Energy Agency. Energy Demand for Hydrogen Production by Fuel in the Net Zero Scenario, 2019–2030. 2022. Available online: https://www.iea.org/data-and-statistics/charts/energy-demand-for-hydrogen-production-by-fuel-in-the-net-zero-scenario-2019-2030 (accessed on 13 February 2023).
- Megia, P.J.; Vizcaino, A.J.; Calles, J.A.; Carrero, A. Hydrogen Production Technologies: From Fossil Fuels toward Renewable Sources. A Mini Review. Energy Fuels 2021, 35, 16403–16415. [Google Scholar] [CrossRef]
- Amin, M.; Shah, H.H.; Fareed, A.G.; Khan, W.U.; Chung, E.; Zia, A.; Farooqi, Z.U.R.; Lee, C. Hydrogen production through renewable and non-renewable energy processes and their impact on climate change. Int. J. Hydrogen Energy 2022, 47, 33112–33134. [Google Scholar] [CrossRef]
- Chau, K.; Djire, A.; Khan, F. Review and analysis of the hydrogen production technologies from a safety perspective. Int. J. Hydrogen Energy 2022, 47, 13990–14007. [Google Scholar] [CrossRef]
- von Döllen, A.; Hwang, Y.; Schlüter, S. The future is colorful—an analysis of the co2 bow wave and why green hydrogen cannot do it alone. Energies 2021, 14, 5720. [Google Scholar] [CrossRef]
- Suer, J.; Traverso, M.; Jäger, N. Carbon Footprint Assessment of Hydrogen and Steel. Energies 2022, 15, 9468. [Google Scholar] [CrossRef]
- Jan, B.M.; Dahari, M.B.; Abro, M.; Ikram, R. Exploration of waste-generated nanocomposites as energy-driven systems for various methods of hydrogen production; A review. Int. J. Hydrogen Energy 2022, 47, 16398–16423. [Google Scholar] [CrossRef]
- Kannah, R.Y.; Kavitha, S.; Preethi; Karthikeyan, O.P.; Kumar, G.; Dai-Viet, N.V.; Banu, J.R. Techno-economic assessment of various hydrogen production methods—A review. Bioresour. Technol. 2021, 319, 124175. [Google Scholar] [CrossRef]
- Cho, H.H.; Strezov, V.; Evans, T.J. A review on global warming potential, challenges and opportunities of renewable hydrogen production technologies. Sustain. Mater. Technol. 2023, 35, e00567. [Google Scholar] [CrossRef]
- Rambhujun, N.; Salman, M.S.; Wang, T.; Pratthana, C.; Sapkota, P.; Costalin, M.; Lai, Q.; Aguey-Zinsou, K.F. Renewable hydrogen for the chemical industry. MRS Energy Sustain. 2020, 7, 33. [Google Scholar] [CrossRef]
- Sharma, S.; Agarwal, S.; Jain, A. Significance of hydrogen as economic and environmentally friendly fuel. Energies 2021, 14, 7389. [Google Scholar] [CrossRef]
- Pipitone, G.; Zoppi, G.; Pirone, R.; Bensaid, S. A critical review on catalyst design for aqueous phase reforming. Int. J. Hydrogen Energy 2022, 47, 151–180. [Google Scholar] [CrossRef]
- Zoppi, G.; Pipitone, G.; Pirone, R.; Bensaid, S. Aqueous phase reforming process for the valorization of wastewater streams: Application to different industrial scenarios. Catal. Today 2022, 387, 224–236. [Google Scholar] [CrossRef]
- Saenz de Miera, B.; Oliveira, A.; Baeza, J.; Calvo, L.; Rodriguez, J.; Gilarranz, M. Treatment and valorisation of fruit juice wastewater by aqueous phase reforming: Effect of pH, organic load and salinity. J. Clean. Prod. 2020, 252, 119849. [Google Scholar] [CrossRef]
- Oliveira, A.; Cordero-Lanzac, T.; Baeza, J.; Calvo, L.; Heras, F.; Rodriguez, J.; Gilarranz, M. Continuous aqueous phase reforming of a synthetic brewery wastewater with Pt/C and PtRe/C catalysts for biohydrogen production. Chemosphere 2021, 281, 130885. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zuo, H.; Wang, J.; Xue, Q.; Ren, B.; Yang, F. The production and application of hydrogen in steel industry. Int. J. Hydrogen Energy 2021, 46, 10548–10569. [Google Scholar] [CrossRef]
- Hosseini, S.E.; Wahid, M.A. Hydrogen from solar energy, a clean energy carrier from a sustainable source of energy. Int. J. Energy Res. 2020, 44, 4110–4131. [Google Scholar] [CrossRef]
- Pervaiz, E.; Ali, M.; Abbasi, M.A.; Noor, T.; Said, Z.; Alawadhi, H. Unfolding essence of nanoscience for improved water splitting hydrogen generation in the light of newly emergent nanocatalysts. Int. J. Hydrogen Energy 2022, 47, 26915–26955. [Google Scholar] [CrossRef]
- Oruc, O.; Dincer, I. Assessing the potential of thermochemical water splitting cycles: A bridge towards for clean and sustainable hydrogen generation. Fuel 2021, 286, 119325. [Google Scholar] [CrossRef]
- Epelle, E.I.; Desongu, K.S.; Obande, W.; Adeleke, A.A.; Ikubanni, P.P.; Okolie, J.A.; Gunes, B. A comprehensive review of hydrogen production and storage: A focus on the role of nanomaterials. Int. J. Hydrogen Energy 2022, 47, 20398–20431. [Google Scholar] [CrossRef]
- Ravi, S.S.; Aziz, M. Clean hydrogen for mobility—Quo vadis? Int. J. Hydrogen Energy 2022, 47, 20632–20661. [Google Scholar] [CrossRef]
- Vodovozov, V.; Raud, Z.; Petlenkov, E. Review of Energy Challenges and Horizons of Hydrogen City Buses. Energies 2022, 15, 6945. [Google Scholar] [CrossRef]
- Chakraborty, S.; Dash, S.K.; Elavarasan, R.M.; Kaur, A.; Elangovan, D.; Meraj, S.T.; Kasinathan, P.; Said, Z. Hydrogen Energy as Future of Sustainable Mobility. Front. Energy Res. 2022, 10, 893475. [Google Scholar] [CrossRef]
- Yue, M.; Lambert, H.; Pahon, E.; Roche, R.; Jemei, S.; Hissel, D. Hydrogen energy systems: A critical review of technologies, applications, trends and challenges. Renew. Sustain. Energy Rev. 2021, 146, 111180. [Google Scholar] [CrossRef]
- Hermesmann, M.; Müller, T.E. Green, Turquoise, Blue, or Grey? Environmentally friendly Hydrogen Production in Transforming Energy Systems. Prog. Energy Combust. Sci. 2022, 90, 111180. [Google Scholar] [CrossRef]
- Sircar, A.; Solanki, K.; Bist, N.; Yadav, K.; Mahanta, K. Green hydrogen: Alternate fuel for Indian energy basket. MRS Energy Sustain. 2022, 90, 100996. [Google Scholar] [CrossRef]
- Tarkowski, R.; Uliasz-Misiak, B. Towards underground hydrogen storage: A review of barriers. Renew. Sustain. Energy Rev. 2022, 162, 112451. [Google Scholar] [CrossRef]
- Raza, A.; Arif, M.; Glatz, G.; Mahmoud, M.; Kobaisi, M.A.; Alafnan, S.; Iglauer, S. A holistic overview of underground hydrogen storage: Influencing factors, current understanding, and outlook. Fuel 2022, 330, 125636. [Google Scholar] [CrossRef]
- Lebrouhi, B.E.; Djoupo, J.J.; Lamrani, B.; Benabdelaziz, K.; Kousksou, T. Global hydrogen development—A technological and geopolitical overview. Int. J. Hydrogen Energy 2022, 47, 7016–7048. [Google Scholar] [CrossRef]
- Germscheidt, R.L.; Moreira, D.E.B.; Yoshimura, R.G.; Gasbarro, N.P.; Datti, E.; dos Santos, P.L.; Bonacin, J.A. Hydrogen Environmental Benefits Depend on the Way of Production: An Overview of the Main Processes Production and Challenges by 2050. Adv. Energy Sustain. Res. 2021, 2, 2100093. [Google Scholar] [CrossRef]
- Okonkwo, E.C.; Al-Breiki, M.; Bicer, Y.; Al-Ansari, T. Sustainable hydrogen roadmap: A holistic review and decision-making methodology for production, utilisation and exportation using Qatar as a case study. Int. J. Hydrogen Energy 2021, 46, 35525–35549. [Google Scholar] [CrossRef]
- Elmanakhly, F.; DaCosta, A.; Berry, B.; Stasko, R.; Fowler, M.; Wu, X.Y. Hydrogen economy transition plan: A case study on Ontario. AIMS Energy 2021, 9, 775–811. [Google Scholar] [CrossRef]
- Kumar, S.S.; Lim, H. An overview of water electrolysis technologies for green hydrogen production. Energy Rep. 2022, 8, 13793–13813. [Google Scholar] [CrossRef]
- Ji, M.; Wang, J. Review and comparison of various hydrogen production methods based on costs and life cycle impact assessment indicators. Int. J. Hydrogen Energy 2021, 46, 38612–38635. [Google Scholar] [CrossRef]
- Jayakumar, A.; Madheswaran, D.K.; Kannan, A.M.; Sureshvaran, U.; Sathish, J. Can hydrogen be the sustainable fuel for mobility in India in the global context? Int. J. Hydrogen Energy 2022, 47, 33571–33596. [Google Scholar] [CrossRef]
- The Green Hydrogen Organisation. Green Hydrogen Standard. 2023. Available online: www.greenhydrogenstandard.org (accessed on 20 December 2022).
- Green Hydrogen Standard. GH2 Statement on New EU Rules Defining Green Hydrogen. 2023. Available online: https://www.greenhydrogenstandard.org/news/gh2-statement-new-eu-rules-defining-green-hydrogen (accessed on 14 February 2023).
- European Commission. Commission Sets Out Rules for Renewable Hydrogen. 2023. Available online: https://ec.europa.eu/commission/presscorner/detail/en/IP_23_594 (accessed on 14 February 2023).
- Grot, W.G. Applications. In Electrochemistry in Industry: New Directions; Springer: Boston, MA, USA, 1982; pp. 73–87. [Google Scholar] [CrossRef]
- Lokesh, S.; Srivastava, R. Advanced Two-Dimensional Materials for Green Hydrogen Generation: Strategies toward Corrosion Resistance Seawater Electrolysis—Review and Future Perspectives. Energy Fuels 2022, 36, 13417–13450. [Google Scholar] [CrossRef]
- Nel Hydrogen. A Series—Atmospheric Alkaline Electrolyser. 2022. Available online: https://nelhydrogen.com/product/atmospheric-alkaline-electrolyser-a-series/ (accessed on 16 March 2023).
- Thyssenkrupp Nucera. Green Hydrogen Solutions. 2023. Available online: https://thyssenkrupp-nucera.com/green-hydrogen-solutions/ (accessed on 16 March 2023).
- Cummins. Hydrogen: The Next Generation. 2021. Available online: https://www.cummins.com/sites/default/files/2021-08/cummins-hydrogen-generation-brochure-20210603.pdf (accessed on 10 July 2023).
- McPhy. New Generation of Pressurized Alkaline Electrolysis for Large-Scale Platforms (Multi-MW/GW). 2019. Available online: https://mcphy.com/en/equipment-services/electrolyzers/augmented/ (accessed on 16 March 2023).
- Sunfire. Hydrogen—The Renewable Feedstock and Energy Carrier. 2020. Available online: https://www.sunfire.de/en/hydrogen (accessed on 16 March 2023).
- Wu, X.; Scott, K. CuxCo3−xO4 (0 ≤ x < 1) nanoparticles for oxygen evolution in high performance alkaline exchange membrane water electrolysers. J. Mater. Chem. 2011, 21, 12344–12351. [Google Scholar] [CrossRef]
- Xiao, L.; Zhang, S.; Pan, J.; Yang, C.; He, M.; Zhuang, L.; Lu, J. First implementation of alkaline polymer electrolyte water electrolysis working only with pure water. Energy Environ. Sci. 2012, 5, 7869–7871. [Google Scholar] [CrossRef]
- Enapter. Technical Integration. 2023. Available online: https://handbook.enapter.com/knowledge_base/technical_integration.html#how-is-the-end-of-lifetime-of-enapter-s-electrolysers-defined (accessed on 16 March 2023).
- Enapter. AEM Multicore. 2021. Available online: https://www.enapter.com/app/uploads/2022/10/Enapter-AEM-Multicore%E2%84%A2-Datasheet-EN.pdf (accessed on 10 July 2023).
- Salehmin, M.N.I.; Husaini, T.; Goh, J.; Sulong, A.B. High-pressure PEM water electrolyser: A review on challenges and mitigation strategies towards green and low-cost hydrogen production. Energy Convers. Manag. 2022, 268, 115985. [Google Scholar] [CrossRef]
- Nel Hydrogen. M Series—PEM Electrolyser. 2023. Available online: https://nelhydrogen.com/product/m-series-3/ (accessed on 20 March 2023).
- ITM Power. HGas3SP. 2023. Available online: https://itm-power.com/products/hgas3sp (accessed on 20 March 2023).
- Siemens Energy. Silyzer 300: The Next Paradigm of PEM Electrolysis. 2020. Available online: https://assets.siemens-energy.com/siemens/assets/api/uuid:a193b68f-7ab4-4536-abe2-c23e01d0b526/datasheet-silyzer300.pdf?ste_sid=62d73127f359ae7db15e5ca4e4f2c676 (accessed on 20 March 2023).
- Santos, A.L.; Cebola, M.J.; Santos, D.M. Towards the hydrogen economy—A review of the parameters that influence the efficiency of alkaline water electrolyzers. Energies 2021, 14, 3193. [Google Scholar] [CrossRef]
- Sunfire. Sunfire—HyLink SOEC. 2021. Available online: https://www.sunfire.de/files/sunfire/images/content/Sunfire.de%20(neu)/Sunfire-Factsheet-HyLink-SOEC-20210303.pdf (accessed on 10 July 2023).
- Haldor Topsoe. SOEC High-Temperature Electrolysis. 2021. Available online: https://www.topsoe.com/hubfs/DOWNLOADS/DOWNLOADS%20-%20Brochures/SOEC%20high-temperature%20electrolysis%20factsheet.pdf?hsCtaTracking=dc9b7bfd-4709-4e7e-acb5-39e76e956078%7C20d976e0-d884-4c00-9fcf-3af3d0850476 (accessed on 10 July 2023).
- Miller, H.A.; Bouzek, K.; Hnat, J.; Loos, S.; Bernäcker, C.I.; Weißgärber, T.; Röntzsch, L.; Meier-Haack, J. Green hydrogen from anion exchange membrane water electrolysis: A review of recent developments in critical materials and operating conditions. Sustain. Energy Fuels 2020, 4, 2114–2133. [Google Scholar] [CrossRef]
- Andújar, J.M.; Segura, F.; Rey, J.; Vivas, F.J. Batteries and Hydrogen Storage: Technical Analysis and Commercial Revision to Select the Best Option. Energies 2022, 15, 6196. [Google Scholar] [CrossRef]
- Dematteis, E.M.; Barale, J.; Corno, M.; Sciullo, A.; Baricco, M.; Rizzi, P. Solid-state hydrogen storage systems and the relevance of a gender perspective. Energies 2021, 14, 6158. [Google Scholar] [CrossRef]
- Hassan, I.A.; Ramadan, H.S.; Saleh, M.A.; Hissel, D. Hydrogen storage technologies for stationary and mobile applications: Review, analysis and perspectives. Renew. Sustain. Energy Rev. 2021, 149, 111311. [Google Scholar] [CrossRef]
- Thiyagarajan, S.R.; Emadi, H.; Hussain, A.; Patange, P.; Watson, M. A comprehensive review of the mechanisms and efficiency of underground hydrogen storage. J. Energy Storage 2022, 51, 104490. [Google Scholar] [CrossRef]
- Egeland-Eriksen, T.; Hajizadeh, A.; Sartori, S. Hydrogen-based systems for integration of renewable energy in power systems: Achievements and perspectives. Int. J. Hydrogen Energy 2021, 46, 31963–31983. [Google Scholar] [CrossRef]
- Amirthan, T.; Perera, M. The role of storage systems in hydrogen economy: A review. J. Nat. Gas Sci. Eng. 2022, 108, 104843. [Google Scholar] [CrossRef]
- Moore, J.; Shabani, B. A Critical Study of Stationary Energy Storage Policies in Australia in an International Context: The Role of Hydrogen and Battery Technologies. Energies 2016, 9, 674. [Google Scholar] [CrossRef]
- Usman, M.R. Hydrogen storage methods: Review and current status. Renew. Sustain. Energy Rev. 2022, 167, 112743. [Google Scholar] [CrossRef]
- Quintos Fuentes, J.E.; Santos, D.M.F. Technical and Economic Viability of Underground Hydrogen Storage. Hydrogen 2023, 4, 975–1000. [Google Scholar] [CrossRef]
- Małachowska, A.; Łukasik, N.; Mioduska, J.; Gębicki, J. Hydrogen Storage in Geological Formations—The Potential of Salt Caverns. Energies 2022, 15, 5038. [Google Scholar] [CrossRef]
- Sambo, C.; Dudun, A.; Samuel, S.A.; Esenenjor, P.; Muhammed, N.S.; Haq, B. A review on worldwide underground hydrogen storage operating and potential fields. Int. J. Hydrogen Energy 2022, 47, 22840–22880. [Google Scholar] [CrossRef]
- Muhammed, N.S.; Haq, B.; Shehri, D.A.; Al-Ahmed, A.; Rahman, M.M.; Zaman, E. A review on underground hydrogen storage: Insight into geological sites, influencing factors and future outlook. Energy Rep. 2022, 8, 461–499. [Google Scholar] [CrossRef]
- Wallace, R.L.; Cai, Z.; Zhang, H.; Zhang, K.; Guo, C. Utility-scale subsurface hydrogen storage: UK perspectives and technology. Int. J. Hydrogen Energy 2021, 46, 25137–25159. [Google Scholar] [CrossRef]
- Lord, A.S.; Kobos, P.H.; Borns, D.J. Geologic storage of hydrogen: Scaling up to meet city transportation demands. Int. J. Hydrogen Energy 2014, 39, 15570–15582. [Google Scholar] [CrossRef]
- Sainz-Garcia, A.; Abarca, E.; Rubi, V.; Grandia, F. Assessment of feasible strategies for seasonal underground hydrogen storage in a saline aquifer. Int. J. Hydrogen Energy 2017, 42, 16657–16666. [Google Scholar] [CrossRef]
- Matos, C.R.; Carneiro, J.F.; Silva, P.P. Overview of Large-Scale Underground Energy Storage Technologies for Integration of Renewable Energies and Criteria for Reservoir Identification. J. Energy Storage 2019, 21, 241–258. [Google Scholar] [CrossRef]
- Underground Sun Storage. Final Report. 2017. Available online: www.underground-sun-storage.at (accessed on 10 July 2023).
- Jeff Sloan. Composites end Markets: Pressure Vessels (2023). 2023. Available online: https://www.compositesworld.com/articles/composites-end-markets-pressure-vessels-2023 (accessed on 2 May 2023).
- Alves, M.P.; Gul, W.; Junior, C.A.C.; Ha, S.K. A Review on Industrial Perspectives and Challenges on Material, Manufacturing, Design and Development of Compressed Hydrogen Storage Tanks for the Transportation Sector. Energies 2022, 15, 5152. [Google Scholar] [CrossRef]
- U.S. Department of Energy. Target Explanation Document: Onboard Hydrogen Storage for Light-Duty Fuel Cell Vehicles. 2017. Available online: https://www.energy.gov/eere/fuelcells/articles/target-explanation-document-onboard-hydrogen-storage-light-duty-fuel-cell (accessed on 10 July 2023).
- Nachtane, M.; Tarfaoui, M.; Abichou, M.a.; Vetcher, A.; Rouway, M.; Aâmir, A.; Mouadili, H.; Laaouidi, H.; Naanani, H. An Overview of the Recent Advances in Composite Materials and Artificial Intelligence for Hydrogen Storage Vessels Design. J. Compos. Sci. 2023, 7, 119. [Google Scholar] [CrossRef]
- Rivard, E.; Trudeau, M.; Zaghib, K. Hydrogen storage for mobility: A review. Materials 2019, 12, 1973. [Google Scholar] [CrossRef]
- Rao, A.G.; Yin, F.; Werij, H.G. Energy transition in aviation: The role of cryogenic fuels. Aerospace 2020, 7, 181. [Google Scholar] [CrossRef]
- Ahluwalia, R.; Hua, T.; Peng, J.K.; Lasher, S.; McKenney, K.; Sinha, J.; Gardiner, M.; Division, N.E.; LLC, T.; DOE, U.S. Technical assessment of cryo-compressed hydrogen storage tank systems for automotive applications. Int. J. Hydrogen Energy 2010, 35. [Google Scholar] [CrossRef]
- Salvador Aceves, G.P. Performance and Durability Testing of Volumetrically Efficient Cryogenic Vessels and High Pressure Liquid Hydrogen Pump. In Proceedings of the DOE Annual Merit Review and Peer Evaluation Meeting (AMR), Washington, DC, USA, 7 June 2017; Lawrence Livermore National Laboratory: Livermore, CA, USA, 2017. [Google Scholar]
- Simanullang, M.; Prost, L. Nanomaterials for on-board solid-state hydrogen storage applications. Int. J. Hydrogen Energy 2022, 47, 29808–29846. [Google Scholar] [CrossRef]
- Rao, P.C.; Yoon, M. Potential liquid-organic hydrogen carrier (Lohc) systems: A review on recent progress. Energies 2020, 13, 6040. [Google Scholar] [CrossRef]
- Epelle, E.I.; Obande, W.; Udourioh, G.A.; Afolabi, I.C.; Desongu, K.S.; Orivri, U.; Gunes, B.; Okolie, J.A. Perspectives and prospects of underground hydrogen storage and natural hydrogen. Sustain. Energy Fuels 2022, 6, 3324–3343. [Google Scholar] [CrossRef]
- Tarkowski, R. Underground hydrogen storage: Characteristics and prospects. Renew. Sustain. Energy Rev. 2019, 105, 86–94. [Google Scholar] [CrossRef]
- Trattner, A.; Höglinger, M.; Macherhammer, M.G.; Sartory, M. Renewable Hydrogen: Modular Concepts from Production over Storage to the Consumer. Chem. Ing. Tech. 2021, 93, 706–716. [Google Scholar] [CrossRef]
- IEA—International Energy Agency. The Future of Hydrogen: Seizing Today’s Opportunities. 2019. Available online: https://www.iea.org/reports/the-future-of-hydrogen (accessed on 10 July 2023).
- Ma, Y.; Wang, X.R.; Li, T.; Zhang, J.; Gao, J.; Sun, Z.Y. Hydrogen and ethanol: Production, storage, and transportation. Int. J. Hydrogen Energy 2021, 46, 27330–27348. [Google Scholar] [CrossRef]
- File: Hydrogen Tube Trailer.jpg—Wikimedia Commons, the Free Media Repository. 2022. Available online: https://commons.wikimedia.org/wiki/File:Hydrogen_trailer.jpg (accessed on 10 December 2023).
- Faye, O.; Szpunar, J.; Eduok, U. A critical review on the current technologies for the generation, storage, and transportation of hydrogen. Int. J. Hydrogen Energy 2022, 47, 13771–13802. [Google Scholar] [CrossRef]
- IRENA. Global Hydrogen Trade to Meet the 1.5 °C Climate Goal: Part II—Technology Review of Hydrogen Carriers. 2022. Available online: www.irena.org/publications (accessed on 10 July 2023).
- Nordic Innovation. The Nordic Green Ammonia Powered Ships (NoGAPS). 2020. Available online: https://www.nordicinnovation.org/programs/nordic-green-ammonia-powered-ships-nogaps (accessed on 21 May 2023).
- C-Job. LH2 Europe & C-Job | Liquid Hydrogen Tanker—C-Job Naval Architects. 2022. Available online: https://c-job.com/projects/liquid-hydrogen-tanker/ (accessed on 30 May 2023).
- Wikimedia Commons. The Free Media Repository. 2023. Available online: https://commons.wikimedia.org/wiki/File:SUISO_FRONTIER_left_rear_view_at_Kawasaki_Heavy_Industries_Kobe_Shipyard_October_18,_2020_02.jpg (accessed on 10 July 2023).
- Silva, B. Hidrogénio Verde já Chega aos Fogões de Famílias no Seixal Desde Outubro. O que Mudou? 2023. Available online: https://www.jornaldenegocios.pt/empresas/energia/detalhe/hidrogenio-verde-ja-chega-aos-fogoes-de-familias-no-seixal-desde-outubro-o-que-mudou (accessed on 6 June 2023).
- Roland Berger Gmbh. Hydrogen Transportation: The Key to Unlocking the Clean Hydrogen Economy. 2021. Available online: https://www.rolandberger.com/publications/publication_pdf/roland_berger_hydrogen_transport.pdf (accessed on 10 July 2023).
- Sarli, V.D. Stability and Emissions of a Lean Pre-Mixed Combustor with Rich Catalytic/Lean-burn Pilot. Int. J. Chem. React. Eng. 2014, 12, 77–89. [Google Scholar] [CrossRef]
- Garcia-Agreda, A.; Di Sarli, V.; Di Benedetto, A. Bifurcation analysis of the effect of hydrogen addition on the dynamic behavior of lean premixed pre-vaporized ethanol combustion. Int. J. Hydrogen Energy 2012, 37, 6922–6932. [Google Scholar] [CrossRef]
- Di Sarli, V.; Di Benedetto, A.; Long, E.; Hargrave, G. Time-Resolved Particle Image Velocimetry of dynamic interactions between hydrogen-enriched methane/air premixed flames and toroidal vortex structures. Int. J. Hydrogen Energy 2012, 37, 16201–16213. [Google Scholar] [CrossRef]
- Salzano, E.; Cammarota, F.; Di Benedetto, A.; Di Sarli, V. Explosion behavior of hydrogen–methane/air mixtures. J. Loss Prev. Process. Ind. 2012, 25, 443–447. [Google Scholar] [CrossRef]
- Atiyeh, C. Everything You Need to Know about the VW Diesel Emissions Scandal. Car Driv. 2019. Available online: https://www.caranddriver.com/news/a15339250/everything-you-need-to-know-about-the-vw-diesel-emissions-scandal/ (accessed on 10 July 2023).
- Candelaresi, D.; Valente, A.; Iribarren, D.; Dufour, J.; Spazzafumo, G. Comparative life cycle assessment of hydrogen-fuelled passenger cars. Int. J. Hydrogen Energy 2021, 46, 35961–35973. [Google Scholar] [CrossRef]
- Netwrok, E.G.E.T. Hydrogen Gas Turbines: The Path Towards a Zero-Carbon Gas Turbine. 2020. Available online: https://etn.global/wp-content/uploads/2020/01/ETN-Hydrogen-Gas-Turbines-report.pdf (accessed on 10 July 2023).
- Thomas, J.M.; Edwards, P.P.; Dobson, P.J.; Owen, G.P. Decarbonising energy: The developing international activity in hydrogen technologies and fuel cells. J. Energy Chem. 2020, 51, 405–415. [Google Scholar] [CrossRef]
- Tellez-Cruz, M.M.; Escorihuela, J.; Solorza-Feria, O.; Compañ, V. Proton Exchange Membrane Fuel Cells (PEMFCs): Advances and Challenges. Polymers 2021, 13, 3064. [Google Scholar] [CrossRef] [PubMed]
- Vaghari, H.; Jafarizadeh-Malmiri, H.; Berenjian, A.; Anarjan, N. Recent advances in application of chitosan in fuel cells. Sustain. Chem. Process. 2013, 1, 16. [Google Scholar] [CrossRef]
- Arsalis, A.; Georghiou, G.E.; Papanastasiou, P. Recent Research Progress in Hybrid Photovoltaic–Regenerative Hydrogen Fuel Cell Microgrid Systems. Energies 2022, 15, 3512. [Google Scholar] [CrossRef]
- File: Proton Exchange Fuel Cell Diagram.svg—Wikimedia Commons, the Free Media Repository. 2023. Available online: https://en.m.wikipedia.org/wiki/File:Proton_Exchange_Fuel_Cell_Diagram.svg (accessed on 10 December 2023).
- Wang, Y.; Pang, Y.; Xu, H.; Martinez, A.; Chen, K.S. PEM Fuel cell and electrolysis cell technologies and hydrogen infrastructure development—A review. Energy Environ. Sci. 2022, 15, 2288–2328. [Google Scholar] [CrossRef]
- Govindarasu, R.; Somasundaram, S. Studies on Influence of Cell Temperature in Direct Methanol Fuel Cell Operation. Processes 2020, 8, 353. [Google Scholar] [CrossRef]
- Thakkar, N.; Paliwal, P. Hydrogen storage based micro-grid: A comprehensive review on technology, energy management and planning techniques. Int. J. Green Energy 2022, 20, 445–463. [Google Scholar] [CrossRef]
- fan, L.; Tu, Z.; Chan, S.H. Recent development of hydrogen and fuel cell technologies: A review. Energy Rep. 2021, 7, 8421–8446. [Google Scholar] [CrossRef]
- Hezarjaribi, M.; Jahanshahi, M.; Rahimpour, A.; Yaldagard, M. Gas diffusion electrode based on electrospun Pani/CNF nanofibers hybrid for proton exchange membrane fuel cells (PEMFC) applications. Appl. Surf. Sci. 2014, 295, 144–149. [Google Scholar] [CrossRef]
- Middelman, E. Improved PEM fuel cell electrodes by controlled self-assembly. Fuel Cells Bull. 2002, 2002, 9–12. [Google Scholar] [CrossRef]
- Debe, M.K. Tutorial on the Fundamental Characteristics and Practical Properties of Nanostructured Thin Film (NSTF) Catalysts. J. Electrochem. Soc. 2013, 160, F522. [Google Scholar] [CrossRef]
- Majlan, E.; Rohendi, D.; Daud, W.; Husaini, T.; Haque, M. Electrode for proton exchange membrane fuel cells: A review. Renew. Sustain. Energy Rev. 2018, 89, 117–134. [Google Scholar] [CrossRef]
- Scofield, M.E.; Liu, H.; Wong, S.S. A concise guide to sustainable PEMFCs: Recent advances in improving both oxygen reduction catalysts and proton exchange membranes. Chem. Soc. Rev. 2015, 44, 5836–5860. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Pasupathi, S.; Bladergroen, B.; Linkov, V.; Pollet, B.G. Optimization of gas diffusion electrode for polybenzimidazole-based high temperature proton exchange membrane fuel cell: Evaluation of polymer binders in catalyst layer. Int. J. Hydrogen Energy 2013, 38, 11370–11378. [Google Scholar] [CrossRef]
- Sutradhar, S.C.; Rahman, M.M.; Ahmed, F.; Ryu, T.; Yoon, S.; Lee, S.; Kim, J.; Lee, Y.; Jin, Y.; Kim, W. Thermally and chemically stable poly(phenylenebenzophenone) membranes for proton exchange membrane fuel cells by Ni (0) catalyst. J. Ind. Eng. Chem. 2019, 76, 233–239. [Google Scholar] [CrossRef]
- Haragirimana, A.; Ingabire, P.B.; Zhu, Y.; Lu, Y.; Li, N.; Hu, Z.; Chen, S. Four-polymer blend proton exchange membranes derived from sulfonated poly(aryl ether sulfone)s with various sulfonation degrees for application in fuel cells. J. Membr. Sci. 2019, 583, 209–219. [Google Scholar] [CrossRef]
- Oh, K.; Kwon, O.; Son, B.; Lee, D.H.; Shanmugam, S. Nafion-sulfonated silica composite membrane for proton exchange membrane fuel cells under operating low humidity condition. J. Membr. Sci. 2019, 583, 103–109. [Google Scholar] [CrossRef]
- Paul, M.T.; Saha, M.S.; Qi, W.L.; Stumper, J.; Gates, B.D. Microstructured membranes for improving transport resistances in proton exchange membrane fuel cells. Int. J. Hydrogen Energy 2020, 45, 1304–1312. [Google Scholar] [CrossRef]
- Teixeira, F.C.; de Sá, A.I.; Teixeira, A.P.; Rangel, C. Nafion phosphonic acid composite membranes for proton exchange membranes fuel cells. Appl. Surf. Sci. 2019, 487, 889–897. [Google Scholar] [CrossRef]
- Wang, J.; Li, P.; Zhang, Y.; Liu, Y.; Wu, W.; Liu, J. Porous Nafion nanofiber composite membrane with vertical pathways for efficient through-plane proton conduction. J. Membr. Sci. 2019, 585, 157–165. [Google Scholar] [CrossRef]
- Zhang, S.; Yuan, X.Z.; Hin, J.N.C.; Wang, H.; Friedrich, K.A.; Schulze, M. A review of platinum-based catalyst layer degradation in proton exchange membrane fuel cells. J. Power Sources 2009, 194, 588–600. [Google Scholar] [CrossRef]
- Garapati, M.S.; Sundara, R. Highly efficient and ORR active platinum-scandium alloy-partially exfoliated carbon nanotubes electrocatalyst for Proton Exchange Membrane Fuel Cell. Int. J. Hydrogen Energy 2019, 44, 10951–10963. [Google Scholar] [CrossRef]
- Pillai, S.R.; Sonawane, S.H.; Gumfekar, S.P.; Suryawanshi, P.L.; Ashokkumar, M.; Potoroko, I. Continuous flow synthesis of nanostructured bimetallic Pt-Mo/C catalysts in milli-channel reactor for PEM fuel cell application. Mater. Chem. Phys. 2019, 237, 121854. [Google Scholar] [CrossRef]
- Wang, Q.; Zhou, Z.Y.; Lai, Y.J.; You, Y.; Liu, J.G.; Wu, X.L.; Terefe, E.; Chen, C.; Song, L.; Rauf, M.; et al. Phenylenediamine-Based FeNx/C Catalyst with High Activity for Oxygen Reduction in Acid Medium and Its Active-Site Probing. J. Am. Chem. Soc. 2014, 136, 10882–10885. [Google Scholar] [CrossRef]
- Ming, W.; Sun, P.; Zhang, Z.; Qiu, W.; Du, J.; Li, X.; Zhang, Y.; Zhang, G.; Liu, K.; Wang, Y.; et al. A systematic review of machine learning methods applied to fuel cells in performance evaluation, durability prediction, and application monitoring. Int. J. Hydrogen Energy 2023, 48, 5197–5228. [Google Scholar] [CrossRef]
- Lee, K.S.; Spendelow, J.S.; Choe, Y.K.; Fujimoto, C.; Kim, Y.S. An operationally flexible fuel cell based on quaternary ammonium-biphosphate ion pairs. Nat. Energy 2016, 1, 16120. [Google Scholar] [CrossRef]
- Okolie, J.A.; Patra, B.R.; Mukherjee, A.; Nanda, S.; Dalai, A.K.; Kozinski, J.A. Futuristic applications of hydrogen in energy, biorefining, aerospace, pharmaceuticals and metallurgy. Int. J. Hydrogen Energy 2021, 46, 8885–8905. [Google Scholar] [CrossRef]









| Company | Electrolyser Name | Pressure [bar] | H2 Capacity [Nm3/h] | Energy Consumption [kWh/Nm3] | Ref. |
|---|---|---|---|---|---|
| Nel Hydrogen (Oslo, Norway) | A3880 | 200 | 2400–3880 | 3.8–4.4 | [87] |
| Thyssenkrupp Nucera (Dortmund, Germany) | 20 MW AWE unit | 30 | 4000 | 4.5 | [88] |
| Cummins (Columbus, IN, USA) | HySTAT® 100-10 | 10 | 100 | 4.9–5.4 | [89] |
| McPhy (Grenoble, France) | McLyzer 800-30 | 30 | 800 | 4.5 | [90] |
| Sunfire (Dresden, Germany) | HyLink Alkaline | 30 | 2230 | 4.7 | [91] |
| Company | Electrolyser Name | Pressure [bar] | H2 Capacity [Nm3/h] | Energy Consumption [kWh/Nm3] | Ref. |
|---|---|---|---|---|---|
| Enapter | AEM Multicore™ | 35 | 210 | 4.8 | [95] |
| Company | Electrolyser Name | Pressure [bar] | H2 Capacity [Nm3/h] | Energy Consumption [kWh/Nm3] | Ref. |
|---|---|---|---|---|---|
| Nel Hydrogen (Oslo, Norway) | M5000 | 30 | 4920 | 4.5 | [97] |
| ITM Power (Sheffield, UK) | HGas3SP | 30 | 400 | - | [98] |
| Cummins (Columbus, IN, USA) | HyLYZER® 4000-30 | 30 | 4000 | 5 | [89] |
| Siemens Energy (Munich, Germany) | Silyzer 300 | - | 1112–22,236 | - | [99] |
| Company | Electrolyser Name | Pressure [bar] | H2 Capacity [Nm3/h] | Energy Consumption [kWh/Nm3] | Ref. |
|---|---|---|---|---|---|
| Sunfire (Dresden, Germany) | HyLink SOEC | 3.5–5.5 | 750 | 3.6 | [101] |
| Topsoe (Lyngby, Denmark) | H2 SOEC 100 MW | 2 | 32,000 | 3.1 | [102] |
| Alkaline | AEM | PEM | Solid Oxide | |
|---|---|---|---|---|
| Electrolyte | KOH/NaOH (5–7 mol/L) | DVB polymer support with KOH/NaOH (1 mol/L) | PFSA membrane | YSZ |
| Separator | Asbestos, Zirfon, Ni | Fumatech | Nafion® | Solid electrolyte YSZ |
| Electrode/catalyst (oxygen side) | Nickel-coated perforated stainless steel | Nickel or NiFeCo alloys | Iridium oxide | Perovskites-type (LSCF, LSM) |
| Electrode/catalyst (hydrogen side) | Nickel-coated perforated stainless steel | Nickel | Platinum nanoparticles on carbon black | Ni-YSZ |
| PTL anode | Nickel mesh (not always present) | Nickel foam | Platinum-coated sintered porous titanium | Nickel mesh or foam |
| PTL cathode | Nickel mesh | Nickel foam or carbon cloth | Sintered porous titanium or carbon cloth | None |
| BP anode | Nickel-coated stainless steel | Nickel-coated stainless steel | Platinum-coated titanium | None |
| BP cathode | Nickel-coated stainless steel | Nickel-coated stainless steel | Gold-coated titanium | Cobalt-coated stainless steel |
| Operating temperature | 70–90 °C | 40–60 °C | 50–80 °C | 700–850 °C |
| Operating pressure | <30 bar | <35 bar | <30 bar | 1 bar |
| Nominal current density | 0.2–0.8 A/cm2 | 0.2–2 A/cm2 | 1–2 A/cm2 | 0.3–1 A/cm2 |
| Voltage range (limits) | 1.4–3.0 V | 1.4–2.0 V | 1.4–2.5 V | 1.0–1.5 V |
| Electrode area | 10,000–30,000 cm2 | <300 cm2 | 1500 cm2 | 200 cm2 |
| Efficiency | 50–68% | 52–67% | 50–68% | 75–85% |
| H2 purity | 99.9–99.9998% | 99.9–99.999% | 99.9–99.9999% | 99.9% |
| Lifetime (stack) | 60,000 h | >5000 h | 50,000–80,000 h | 20,000 h |
| Cold start | <50 min | <20 min | <20 min | >600 min |
| Stack unit size | 1 MW | 2.5 kW | 1 MW | 5 kW |
| Capital costs (stack) minimum 1 MW | 270 USD/kW | n.d. | 400 USD/kW | >2000 USD/kW |
| Capital costs (stack) minimum 10 MW | 500–1000 USD/kW | n.d. | 700–1400 USD/kW | n.d. |
| Development status | Early Adoption | Large Prototype | Early Adoption | Demonstration |
| TRL Scale | TRL 9 | TRL 6 | TRL 9 | TRL 7 |
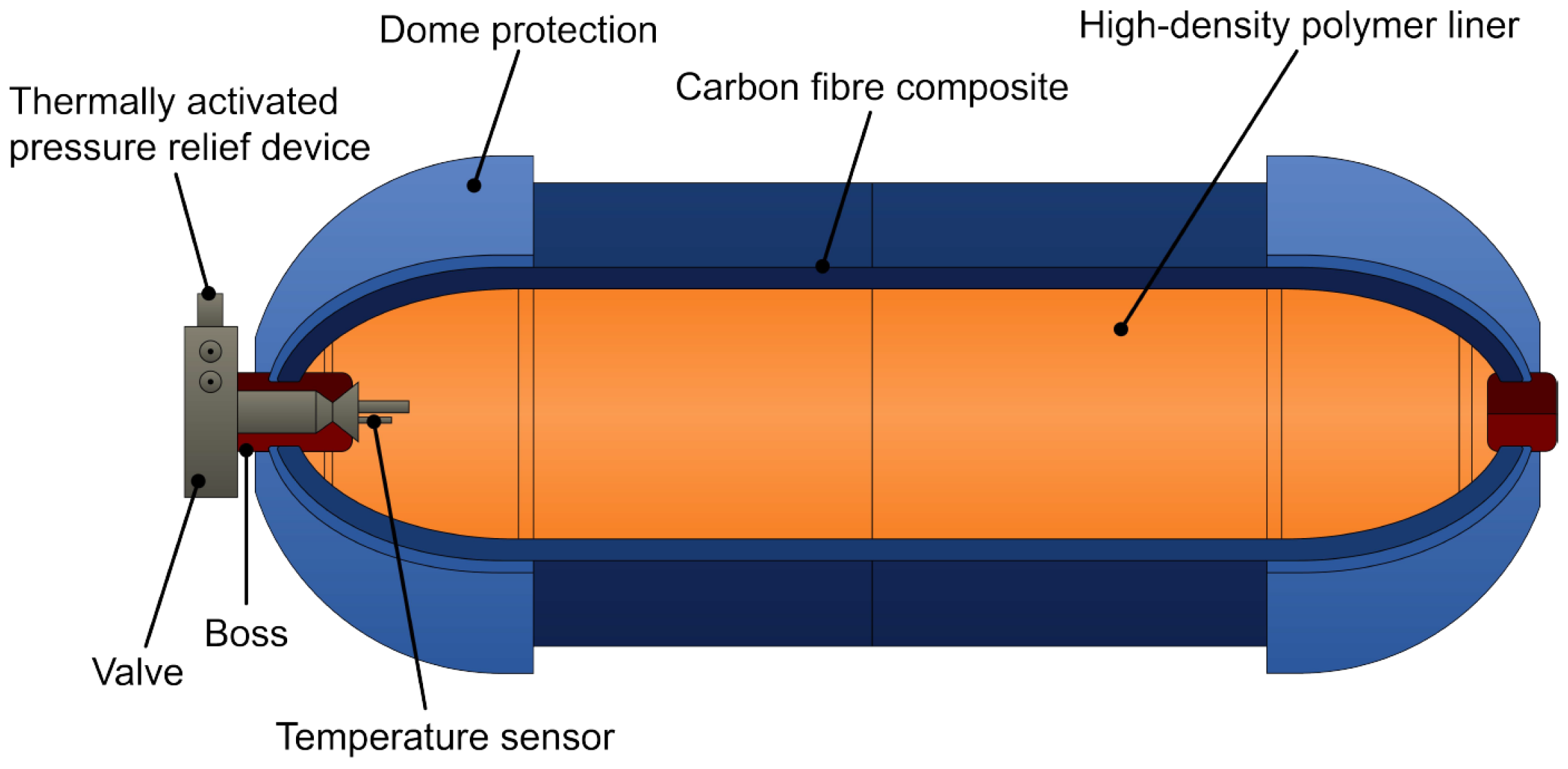
| Type 1 | Type 2 | Type 3 | Type 4 | Type 5 | |
|---|---|---|---|---|---|
| Components | All metal construction | Steel vessel with glass fibre composite | Full composite overwrap with metal (aluminium) liner | Full carbon fibre or carbon–glass fibre composite with HDPE liner and metallic boss | Full composite without liner |
| Structural load | Metal body withstands the whole load | Steel and composite materials share the load equally | The composite structure bears the most load, and the metal liner takes only about 5% mechanical load | Composite material carries the load | Composite material carries the load |
| Storage pressure | Up to 50 MPa | Highest pressure tolerance | Typically, around 45 MPa and showed problems for 70 MPa | Up to 100 MPa | Under development |
| Gravimetric density | ∼1 wt% | ≤2.1 wt% | ≤4.2 wt% | 4.4–5.7% | Under development |
| Weight | Relative weight is high | 30–40% less than Type I | Around half the weight of Type II | Lighter than Type III | It has the potential to be the lightest |
| Cost (differs with application) | 83 USD/kg H2 | 86 USD/kg H2 | 700 USD/kg H2 | 633 USD/kg H2 | Under development |
| Salt Caverns | Depleted Reservoirs | Aquifers | |
|---|---|---|---|
| Depth (m) | 300–1800 | 300–2700 | 400–2300 |
| Operating pressure (bar) | 35–270 | 15–285 | 30–315 |
| Safety | Very good | Poor | Poor |
| Relative Investment Cost | Low | Low | Low |
| Relative Operational Cost | Low | Moderate | Low |
| Injection rate | Moderate | High | Moderate |
| Withdrawal rate | Moderate | High | Moderate |
| Working/Total gas capacity | High | Moderate | Low |
| Feasible cycles per annum | High | Low | Low |
| Current locations | UK and USA | None for H2 Established use with NG | None for H2 Established use with NG |
| Suitability for H2 | Proven | Still under investigation. Proven for town gas with up to 50% of H2. | Under investigation. Proven for CH4 with up to 10% of H2. |
| Technology Maturity | Early Adoption | Small Prototype | Concept |
| TRL Scale | 9–10 | 4 | 3 |
| H2 Storage System | Gravimetric Energy Density, kWh/kg (System) | Gravimetric Energy Density, kWh/kg (Pure Substance) | Volumetric Energy Density, g/L (System) | Volumetric Energy Density, g/L (Pure Substance) | Technology Maturity | TRL Scale | |
|---|---|---|---|---|---|---|---|
| Compressed gas | Type III 35 MPa | 1.9 | 33.3 | 0.5 | 0.8 | Mature | 11 |
| Type III 70 MPa | 1.0 | 33.3 | 0.7 | 1.3 | Mature | 11 | |
| Type IV 70 MPa | 1.6 | 33.3 | 0.7 | 1.3 | Mature | 11 | |
| Liquid | LH2 | 2.0 | 33.3 | 1.2 | 2.2 | Demonstration | 7–9 |
| Cryo- compressed | CcH2 | 2.5 | 33.3 | 1.5 | 2.7 | Demonstration | 6–7 |
| Adsorbent | MOF-5 | 1.0 | 2.7 | 0.7 | 1.8 | Concept | 2–3 |
| Metal hydride | MgH2 | 0.7 | 2.5 | 1.2 | 3.7 | Demonstration | 6–7 |
| TiFe | 0.3 | 0.6 | 0.6 | 4.1 | Large Prototype | 5–6 | |
| TiMn2 | 0.4 | 0.6 | 1.3 | 4.0 | Large Prototype | 5–6 | |
| Complex hydride | Mg(BH4)2 | - | 5.0 | - | 4.9 | Concept | 1–2 |
| NaAIH4 | - | 2.5 | - | 3.1 | Concept | 1–2 | |
| LOHC | N-ethyl carbazole | - | 1.7 | - | 2.2 | Demonstration | 6–7 |
| Dibenzyl toluene | - | 2.0 | - | 1.9 | Demonstration | 7–8 |
| ICE | Gas Turbine | PEMFC | SOFC | |
|---|---|---|---|---|
| Conversion efficiency | 50% | 35% | 52% | 60% |
| System efficiency | 50% | 58% | 56% | 80% |
| Cost (USD/kW) | <500 | – | >1500 | >4500 |
| Partial load efficiency | High | Low | High | High |
| Tolerance to load variations | High | High | Medium | Low |
| Lifetime | High | High | Low | Low |
| Noise/vibration | High | High | Low | Low |
| NOx, CO and hydrocarbon emissions | Medium | Medium | Low | Low |
| Technology Maturity | High | High | Medium | Low |
| TRL Scale | 9 | 8 | 9 | 8 |
| Fuel Cell Type | AFC | PEMFC | DMFC | PAFC | MCFC | SOFC |
|---|---|---|---|---|---|---|
| Temperature °C | 90–100 | 50–100 | 60–200 | 150–200 | 600–700 | 600–1000 |
| Electrolyte | Potassium hydroxide | Polymer membrane | Polymer membrane | Phosphoric acid | Carbonate melt | Solid ceramic oxide |
| Power range | 1 kW to 100 kW | 1 W to 100 kW | 1 W to 100 kW | 200 kW to 10 MW | 500 kW to 10 MW | 1 kW to 2 MW |
| Fuel | Purest H2 | Pure H2 | CH3OH + H2O | H2, COCH3OH | H2, CO, CH4 | H2, CO2CH4 |
| Efficiency | 40–55% | 40–55% | 50–70% | 40–55% | 50–60% | 40–72% |
| Startup time | <5 min | <5 min | <5 min | 1–4 h | 5–10 h | 5–10 h |
| Investment costs USD/kW | 200 to 700 | 3000 to 4000 (stationary) 500 (mobile) | n.a. | 4000 to 5000 | 4000 to 6000 | 3000 to 4000 |
| Life expectancy | 4000 to 8000 h | 60,000 h (stationary) 5000 h (mobile) | n.a. | 30,000 to 60,000 h | 20,000 to 40,000 h | <90,000 h |
| Market development | Early Adoption | Early Adoption | Large Prototype | Demonstration | Demonstration | Demonstration |
| TRL scale | 10 | 9 | 6 | 8 | 7 | 8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rolo, I.; Costa, V.A.F.; Brito, F.P. Hydrogen-Based Energy Systems: Current Technology Development Status, Opportunities and Challenges. Energies 2024, 17, 180. https://doi.org/10.3390/en17010180
Rolo I, Costa VAF, Brito FP. Hydrogen-Based Energy Systems: Current Technology Development Status, Opportunities and Challenges. Energies. 2024; 17(1):180. https://doi.org/10.3390/en17010180
Chicago/Turabian StyleRolo, Inês, Vítor A. F. Costa, and Francisco P. Brito. 2024. "Hydrogen-Based Energy Systems: Current Technology Development Status, Opportunities and Challenges" Energies 17, no. 1: 180. https://doi.org/10.3390/en17010180
APA StyleRolo, I., Costa, V. A. F., & Brito, F. P. (2024). Hydrogen-Based Energy Systems: Current Technology Development Status, Opportunities and Challenges. Energies, 17(1), 180. https://doi.org/10.3390/en17010180








