The Incremental Capacity Curves and Frequency Response Characteristic Evolution of Lithium Titanate Battery during Ultra-High-Rate Discharging Cycles
Abstract
1. Introduction
- Perform aging cycles at ultra-high-rate current (50 C) on 20 LTO cells;
- Obtain the evolution of IC curves during the ultra-high-rate current cycles;
- Use EIS to analyze the frequency response characteristic;
- Obtain a key indicator of high-power capacity fading of LTO cells.
2. Materials and Methods
2.1. Experiment Condition
2.2. Experiment Methods
- Aging cycle experimentCharge the cells with a constant current of 1 C (2 A) to 2.8 V, then turn it into a constant voltage step until the current decreases to 0.05 C or lower. Rest for 30 min, then discharge the cell at 50 C (100 A) to 1.5 V. Rest for 3 h to eliminate the polarization caused by high-rate discharging. This experiment is performed at 55 °C.
- Capacity calibrating experimentCharge the cells as described in aging cycle experiment, then rest for 30 min. Discharge the cells with a constant current to 1.5 V, then rest for 3 h and start over with a different discharging current rate. In a capacity calibrating experiment, the discharging current would be 0.5, 1, 5, 10, 20, 30, 40, and 50 C. This experiment is performed at 23 °C.
- Charging response experimentDischarge the cells with a constant current of 0.1 C (0.2 A) to 1.5 V. The SOC is considered to be 0%. Charge the cells with a constant current to 2.8 V from 0% SOC, then start over with a different charging current.The charging current used in this experiment is 0.05, 0.1, 0.25, 0.5, 1, 1.5, and 2 C to obtain the charging response at different charging current rates. This experiment is performed at 23 °C.
- EISPerform EIS on each cell with a voltage excitation of 4 mV. The frequency range is 0.01 Hz–100 kHz. Each cell is tested at three various SOC: 30, 50, and 100%. This experiment is performed at 23 °C.
- Charging response experiment;
- EIS;
- Capacity calibrating experiment;
- Aging cycle experiment, for 50 cycles;
- Capacity calibrating experiment;
- Back to Step. 1.
3. Results
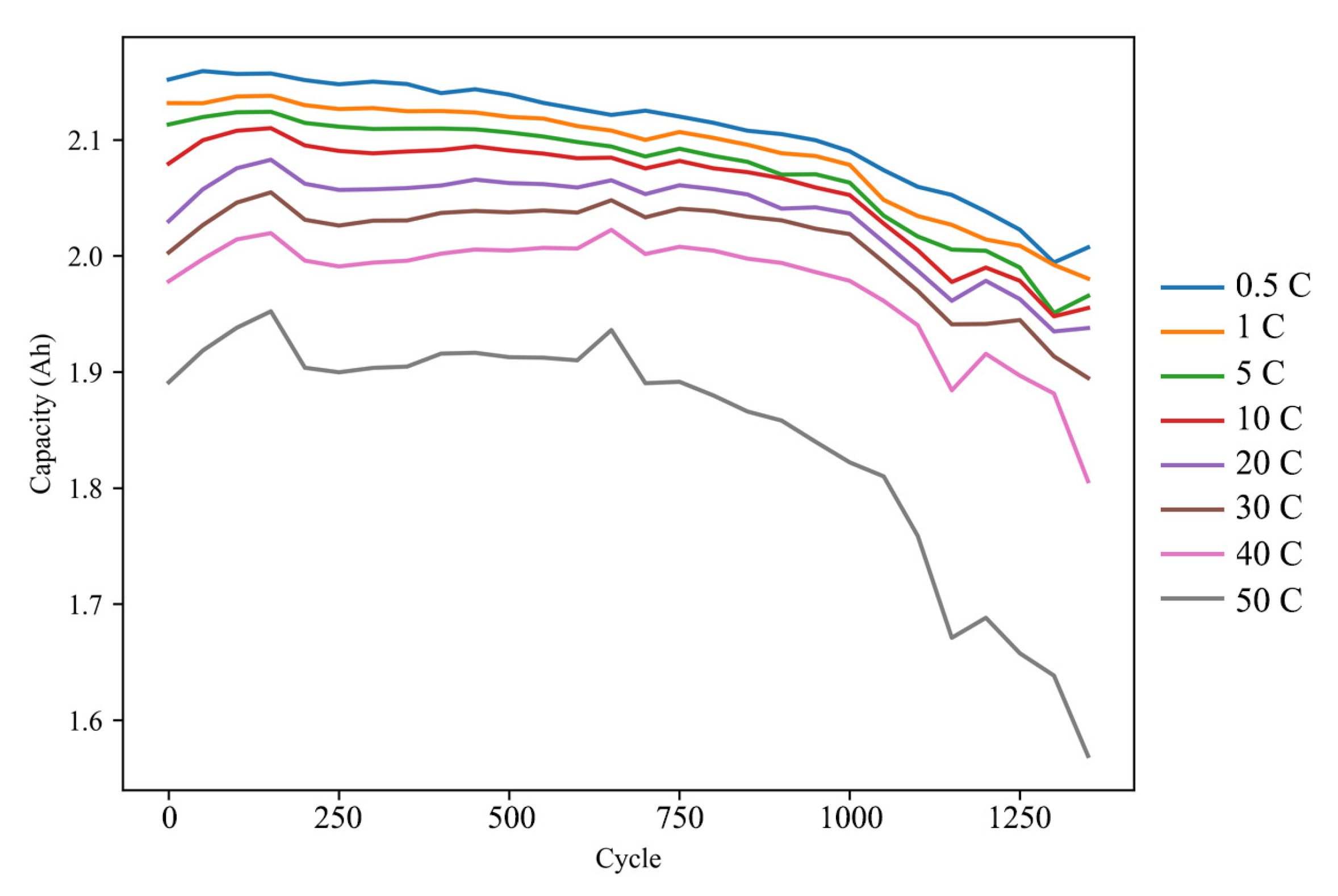
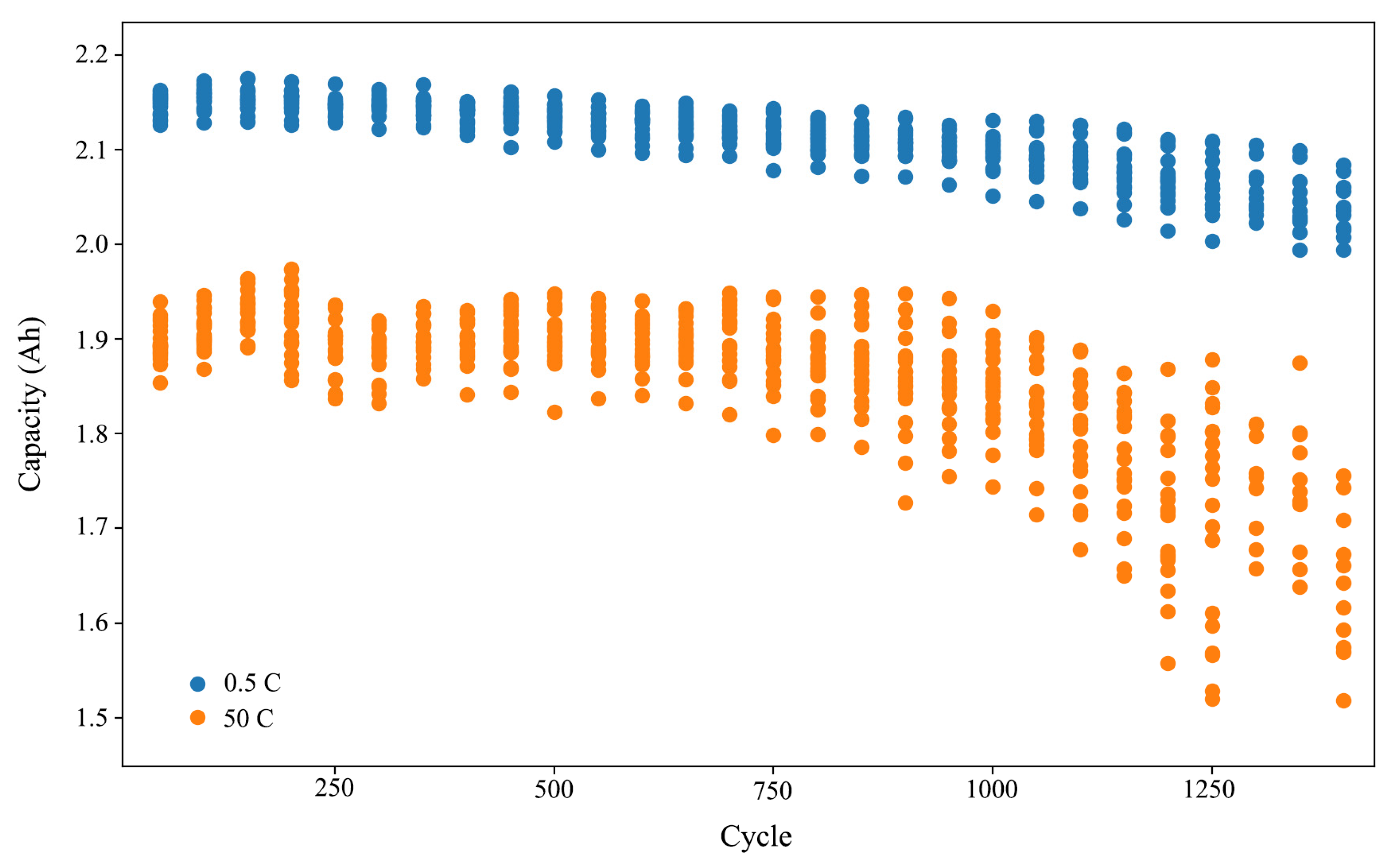
3.1. Aging Cycle Experiments
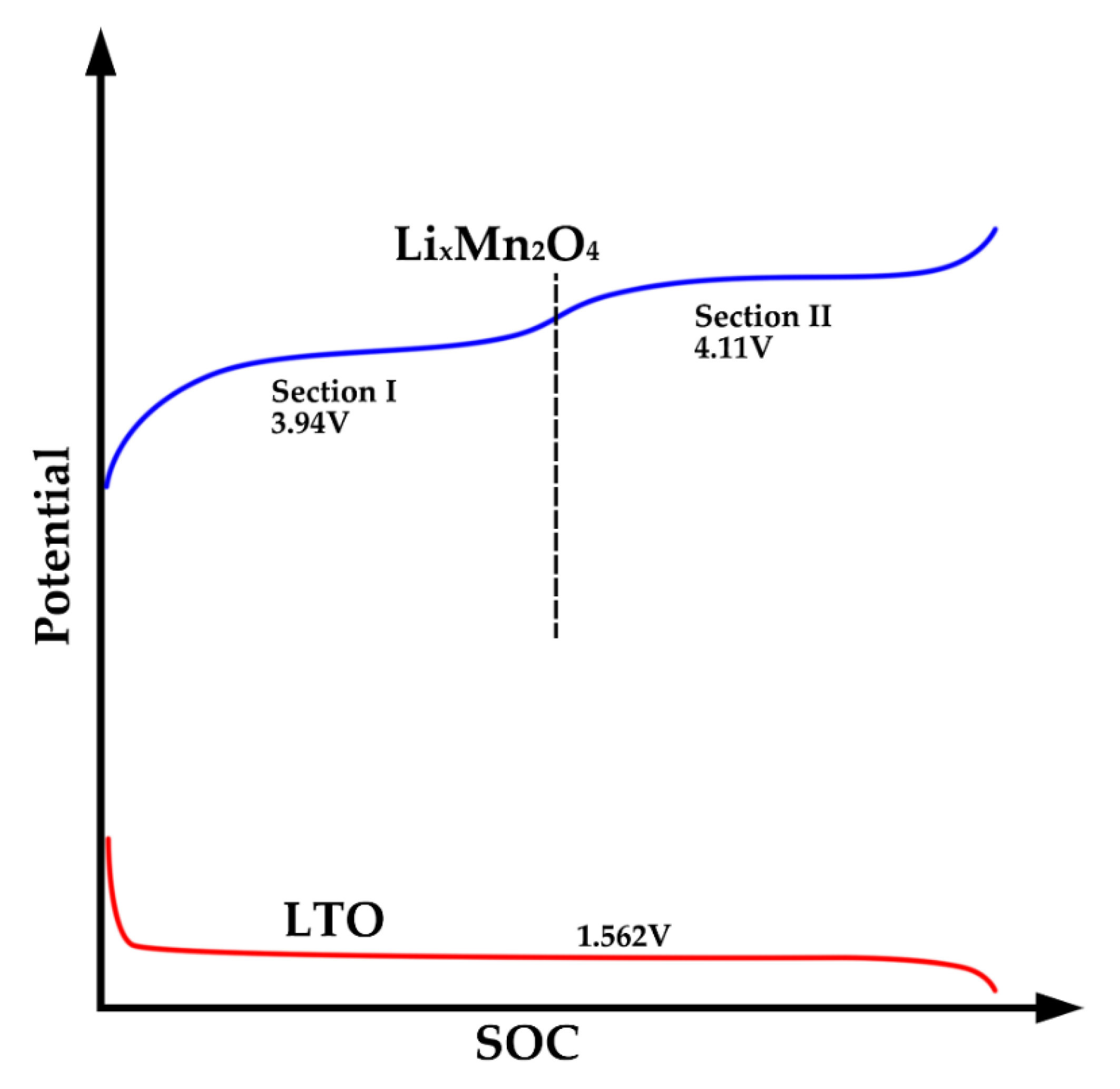
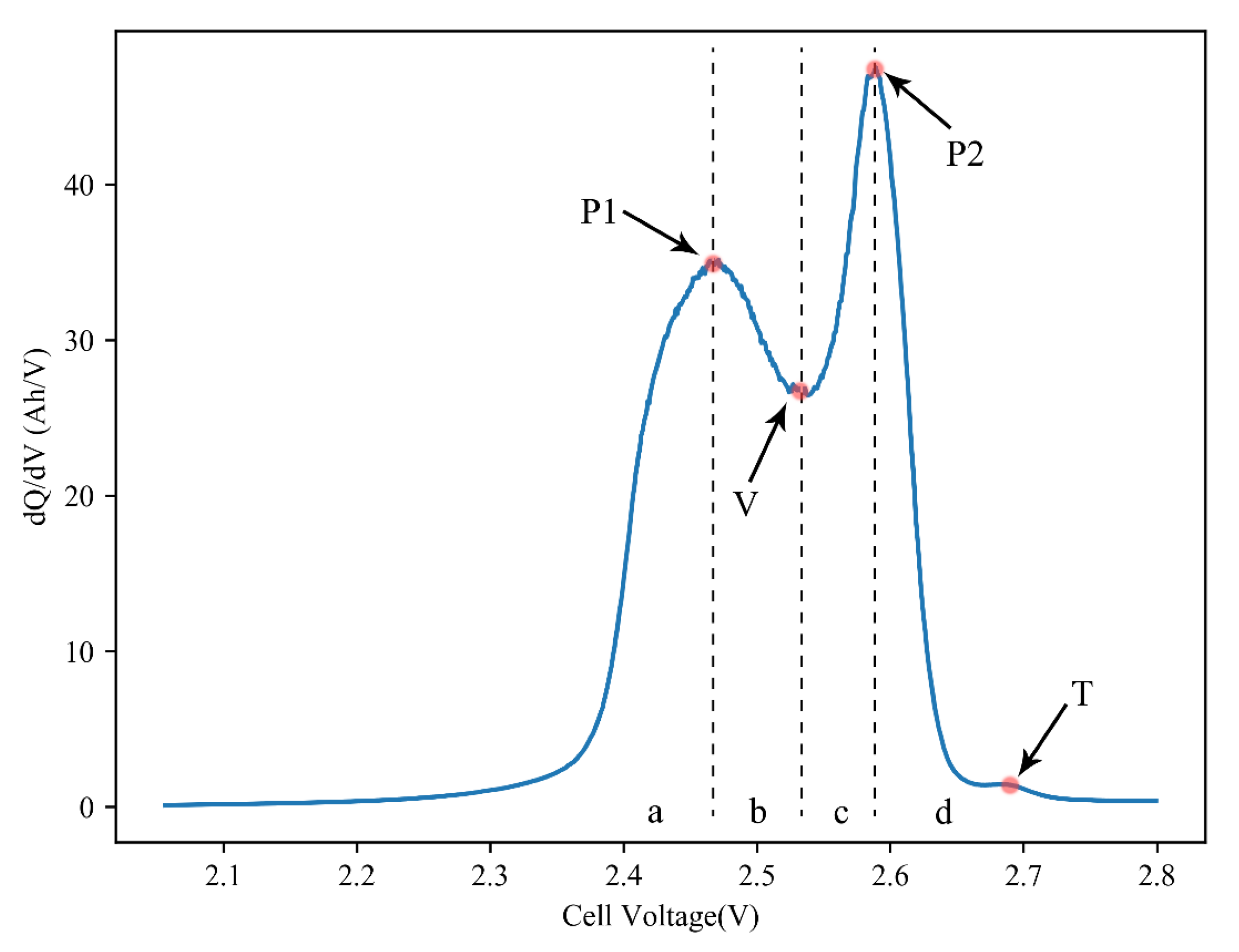
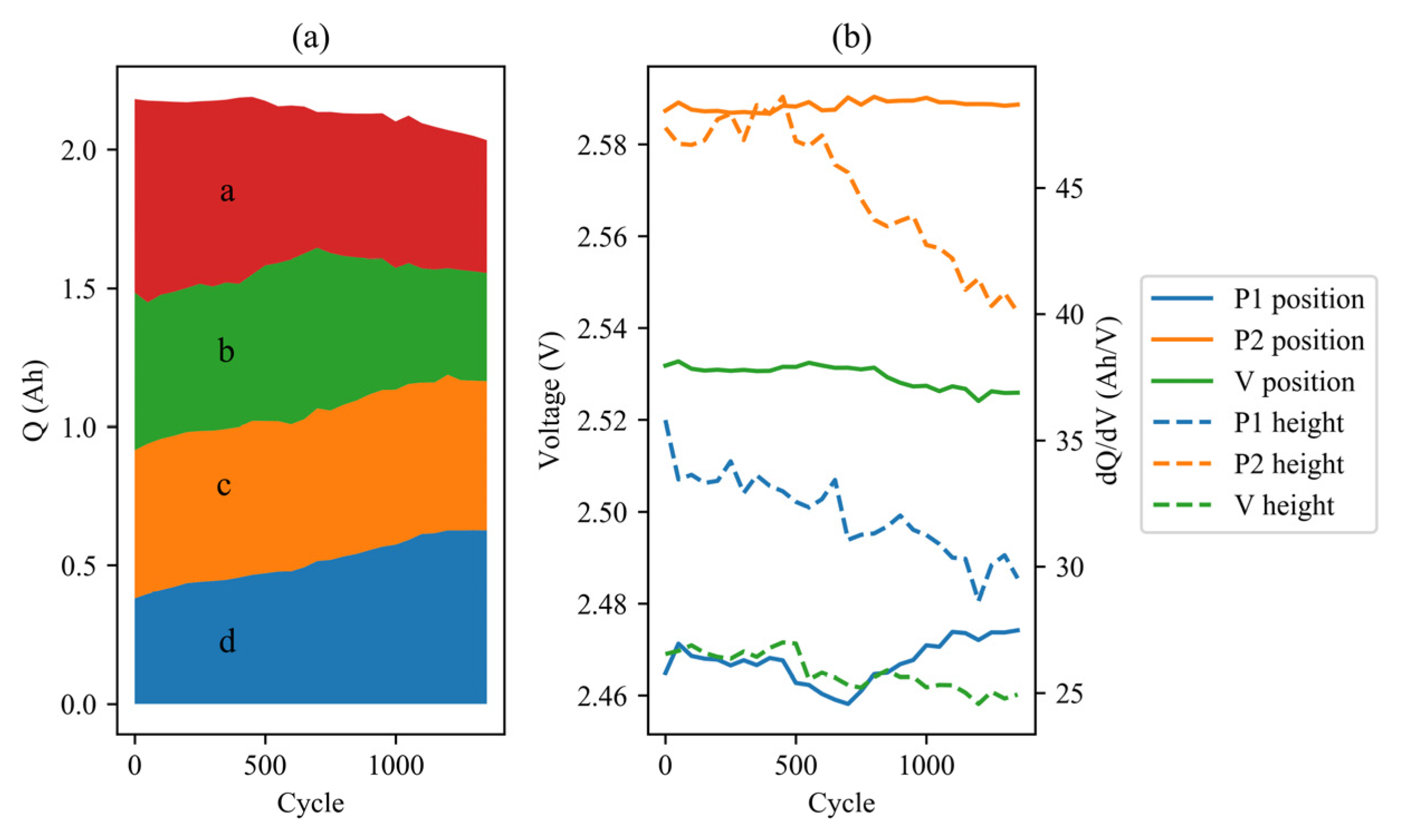
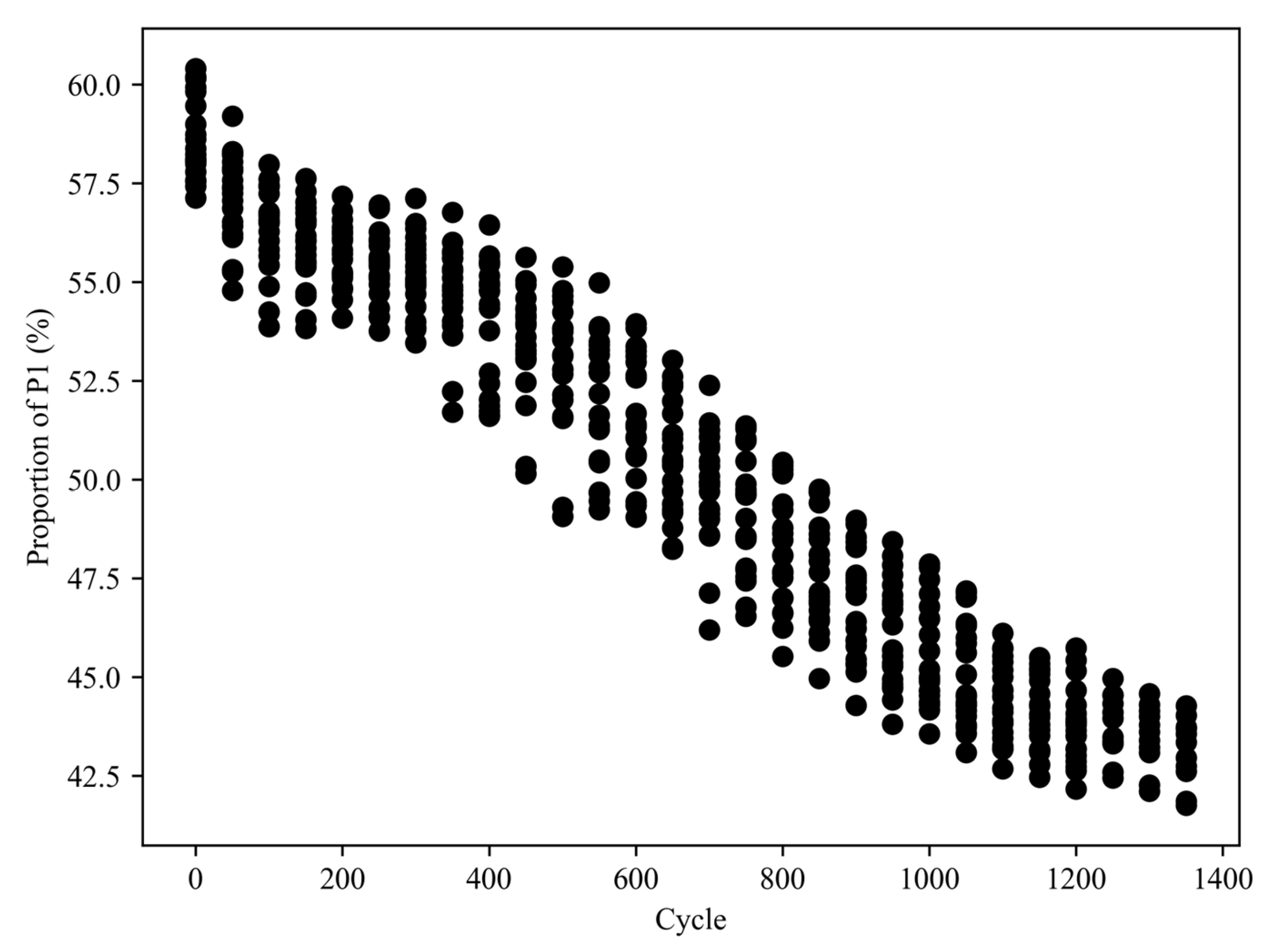
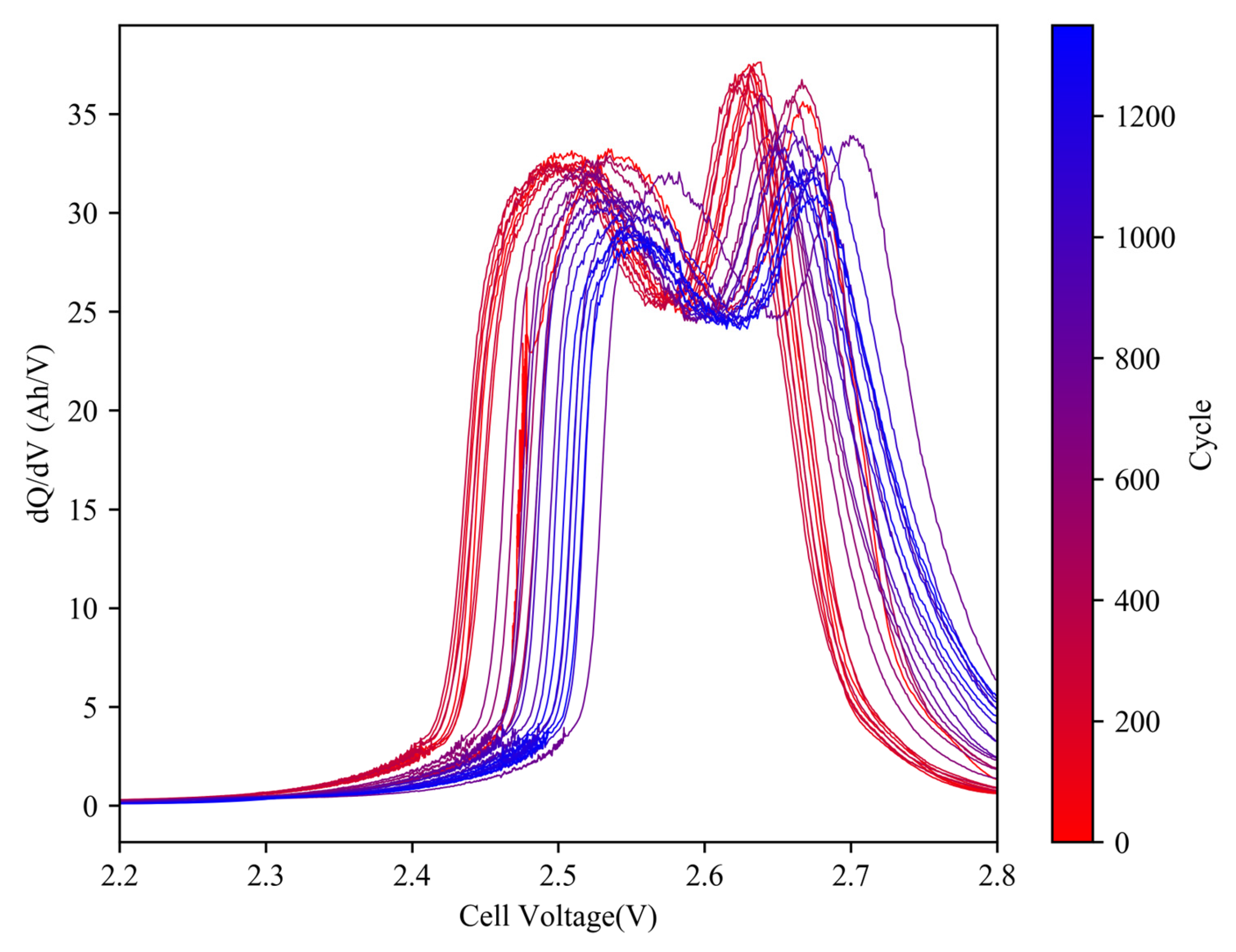
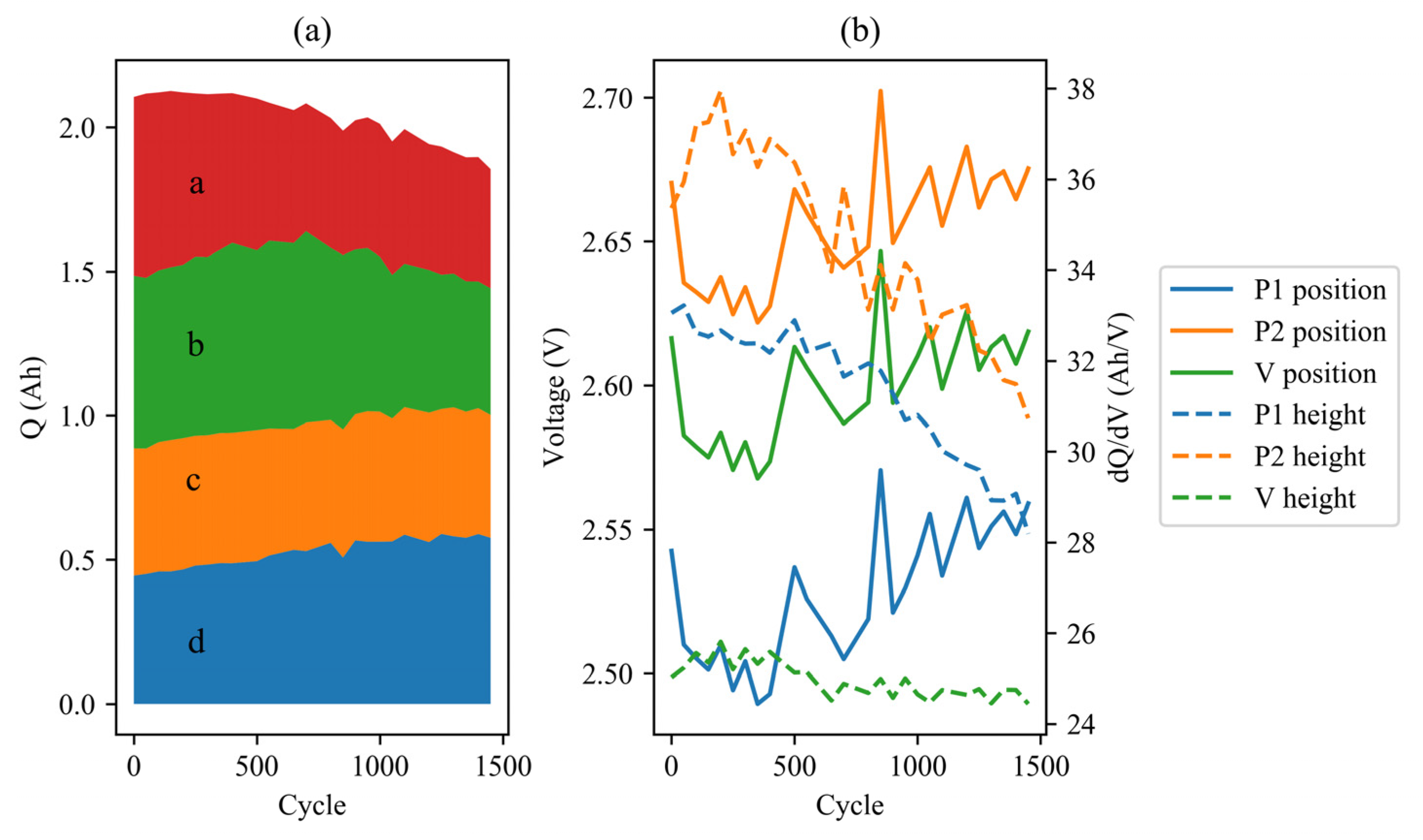
3.2. IC Analysis
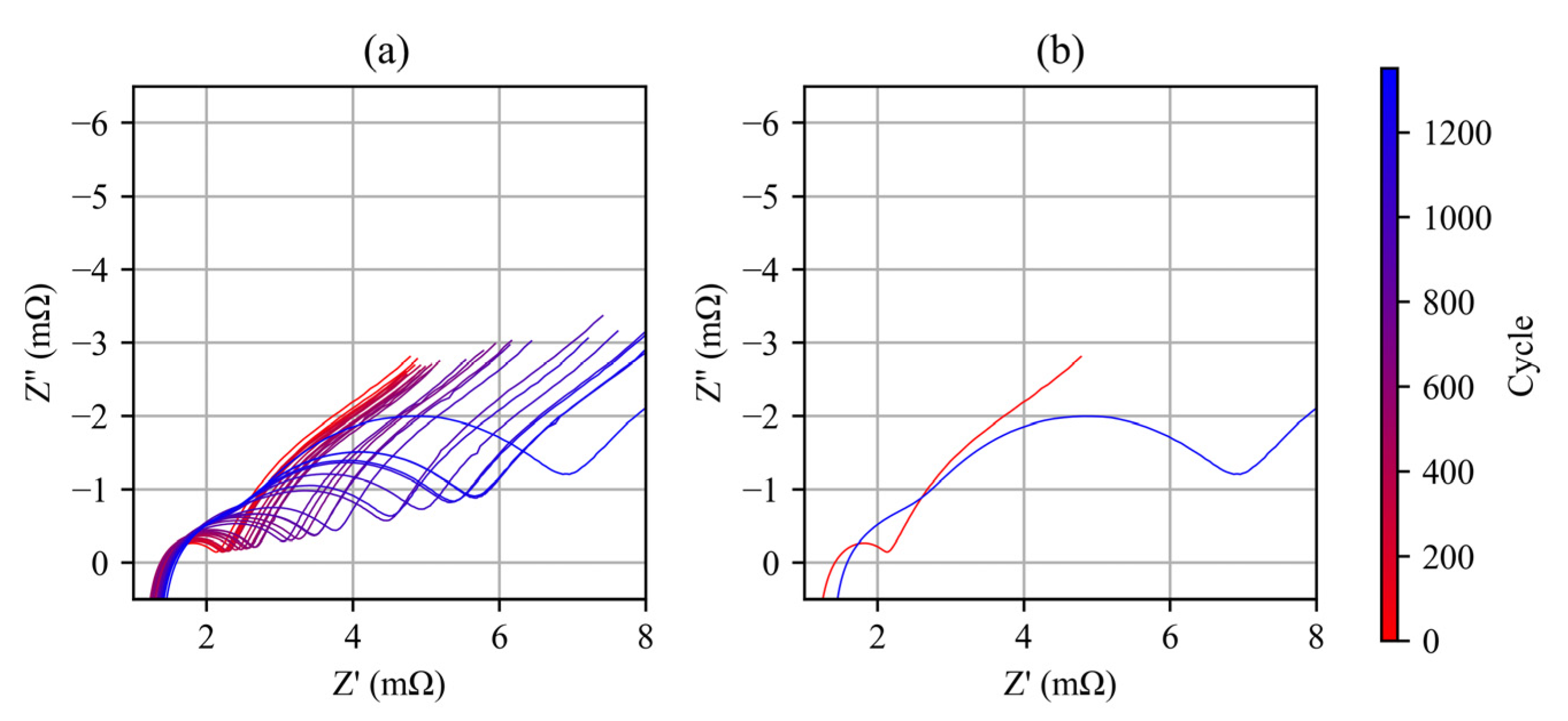
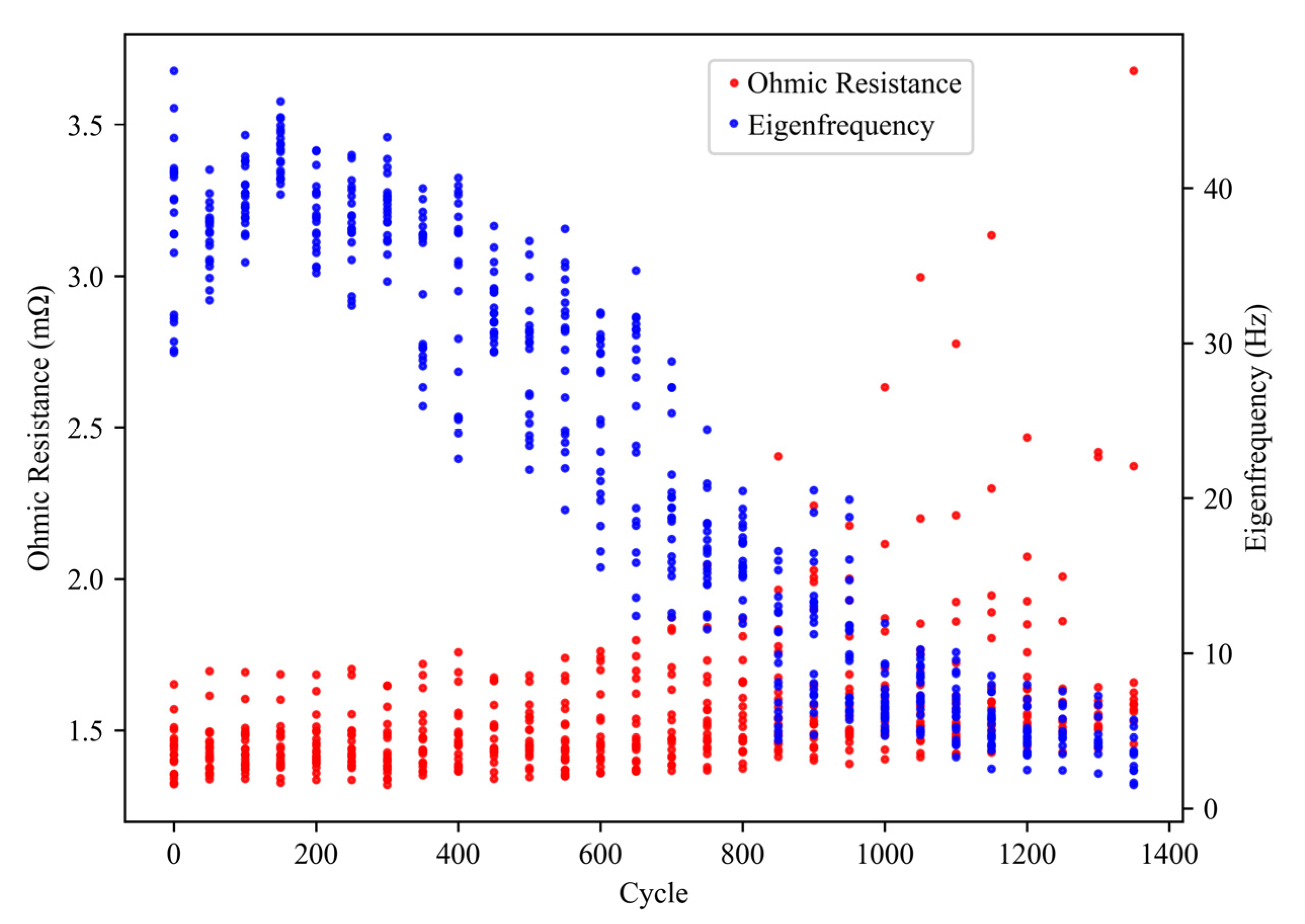
3.3. EIS
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Nishi, Y. Lithium ion secondary batteries; past 10 years and the future. J. Power Sour. 2001, 100, 101–106. [Google Scholar] [CrossRef]
- Ohzuku, T.; Ueda, A.; Yamamoto, N. Zero-strain insertion material of Li [Li1/3Ti5/3] O4 for rechargeable lithium cells. J. Electrochem. Soc. 1995, 142, 1431. [Google Scholar] [CrossRef]
- Colbow, K.M.; Dahn, J.R.; Haering, R.R. Structure and electrochemistry of the spinel oxides LiTi2O4 and Li43Ti53O4. J. Power Sour. 1989, 26, 397–402. [Google Scholar] [CrossRef]
- Takami, N.; Inagaki, H.; Tatebayashi, Y.; Saruwatari, H.; Honda, K.; Egusa, S. High-power and long-life lithium-ion batteries using lithium titanium oxide anode for automotive and stationary power applications. J. Power Sour. 2013, 244, 469–475. [Google Scholar] [CrossRef]
- Belharouak, I.; Koenig, G.M., Jr.; Amine, K. Electrochemistry and safety of Li4Ti5O12 and graphite anodes paired with LiMn2O4 for hybrid electric vehicle Li-ion battery applications. J. Power Sour. 2011, 196, 10344–10350. [Google Scholar] [CrossRef]
- Nemeth, T.; Schröer, P.; Kuipers, M.; Sauer, D.U. Lithium titanate oxide battery cells for high-power automotive applications–Electro-thermal properties, aging behavior and cost considerations. J. Energy Storage 2020, 31, 101656. [Google Scholar] [CrossRef]
- Lu, J.; Chen, Z.; Pan, F.; Cui, Y.; Amine, K. High-performance anode materials for rechargeable lithium-ion batteries. Electrochem. Energy Rev. 2018, 1, 35–53. [Google Scholar] [CrossRef]
- Zhao, B.; Ran, R.; Liu, M.; Shao, Z. A comprehensive review of Li4Ti5O12-based electrodes for lithium-ion batteries: The latest advancements and future perspectives. Mater. Sci. Eng. R Rep. 2015, 98, 1–71. [Google Scholar]
- Barré, A.; Deguilhem, B.; Grolleau, S.; Gérard, M.; Suard, F.; Riu, D. A review on lithium-ion battery ageing mechanisms and estimations for automotive applications. J. Power Sour. 2013, 241, 680–689. [Google Scholar] [CrossRef]
- Jaguemont, J.; Boulon, L.; Dubé, Y. A comprehensive review of lithium-ion batteries used in hybrid and electric vehicles at cold temperatures. Appl. Energy 2016, 164, 99–114. [Google Scholar] [CrossRef]
- Qian, K.; Li, Y.; He, Y.B.; Liu, D.; Zheng, Y.; Luo, D. Abuse tolerance behavior of layered oxide-based Li-ion battery during overcharge and over-discharge. RSC Adv. 2016, 6, 76897–76904. [Google Scholar] [CrossRef]
- Vetter, J.; Novák, P.; Wagner, M.R.; Veit, C.; Möller, K.C.; Besenhard, J.O. Ageing mechanisms in lithium-ion batteries. J. Power Sour. 2005, 147, 269–281. [Google Scholar] [CrossRef]
- Hang, T.; Mukoyama, D.; Nara, H.; Takami, N.; Momma, T.; Osaka, T. Electrochemical impedance spectroscopy analysis for lithium-ion battery using Li4Ti5O12 anode. J. Power Sour. 2013, 222, 442–447. [Google Scholar] [CrossRef]
- Han, X.; Ouyang, M.; Lu, L.; Li, J.; Zheng, Y.; Li, Z. A comparative study of commercial lithium ion battery cycle life in electrical vehicle: Aging mechanism identification. J. Power Sour. 2014, 251, 38–54. [Google Scholar] [CrossRef]
- Han, X.; Ouyang, M.; Lu, L.; Li, J. Cycle life of commercial lithium-ion batteries with lithium titanium oxide anodes in electric vehicles. Energies 2014, 7, 4895–4909. [Google Scholar] [CrossRef]
- Stroe, A.I.; Knap, V.; Stroe, D.I. Comparison of lithium-ion battery performance at beginning-of-life and end-of-life. Microelectron. Reliab. 2018, 88, 1251–1255. [Google Scholar] [CrossRef]
- De Hoog, J.; Timmermans, J.M.; Ioan-Stroe, D.; Swierczynski, M.; Jaguemont, J.; Goutam, S. Combined cycling and calendar capacity fade modeling of a Nickel-Manganese-Cobalt Oxide Cell with real-life profile validation. Appl. Energy 2017, 200, 47–61. [Google Scholar] [CrossRef]
- Liu, S.; Winter, M.; Lewerenz, M.; Becker, J.; Sauer, D.U.; Ma, Z.; Jiang, J. Analysis of cyclic aging performance of commercial Li4Ti5O12-based batteries at room temperature. Energy 2019, 173, 1041–1053. [Google Scholar] [CrossRef]
- Wang, C.; Liu, Z.; Sun, Y.; Gao, Y.; Yan, P. Aging behavior of lithium titanate battery under high-rate discharging cycle. Energies 2021, 14, 5482. [Google Scholar] [CrossRef]
- Weng, C.; Cui, Y.; Sun, J.; Peng, H. On-board state of health monitoring of lithium-ion batteries using incremental capacity analysis with support vector regression. J. Power Sour. 2013, 235, 36–44. [Google Scholar] [CrossRef]
- Li, Y.; Abdel-Monem, M.; Gopalakrishnan, R.; Berecibar, M.; Nanini-Maury, E.; Omar, N. A quick on-line state of health estimation method for Li-ion battery with incremental capacity curves processed by Gaussian filter. J. Power Sour. 2018, 373, 40–53. [Google Scholar] [CrossRef]
- Tang, X.; Zou, C.; Yao, K.; Chen, G.; Liu, B.; He, Z.; Gao, F. A fast estimation algorithm for lithium-ion battery state of health. J. Power Sour. 2018, 396, 453–458. [Google Scholar] [CrossRef]
- Dubarry, M.; Liaw, B.Y.; Chen, M.S.; Chyan, S.S.; Han, K.C.; Sie, W.T.; Wu, S.H. Identifying battery aging mechanisms in large format Li ion cells. J. Power Sour. 2011, 196, 3420–3425. [Google Scholar] [CrossRef]
- Yu, Z.; Zhang, X.; Yang, G.; Liu, J.; Wang, J.; Wang, R.; Zhang, J. High rate capability and long-term cyclability of Li4Ti4.9V0.1O12 as anode material in lithium ion battery. Electrochim. Acta 2011, 56, 8611–8617. [Google Scholar] [CrossRef]
- Wong, D.N.; Wetz, D.A.; Heinzel, J.M.; Mansour, A.N. Characterizing rapid capacity fade and impedance evolution in high rate pulsed discharged lithium iron phosphate cells for complex, high power loads. J. Power Sour. 2016, 328, 81–90. [Google Scholar] [CrossRef]
- Hall, F.; Touzri, J.; Wußler, S.; Buqa, H.; Bessler, W.G. Experimental investigation of the thermal and cycling behavior of a lithium titanate-based lithium-ion pouch cell. J. Energy Storage 2018, 17, 109–117. [Google Scholar] [CrossRef]
- Basu, S.; Patil, R.S.; Ramachandran, S.; Hariharan, K.S.; Kolake, S.M.; Song, T. Non-isothermal electrochemical model for lithium-ion cells with composite cathodes. J. Power Sour. 2015, 283, 132–150. [Google Scholar] [CrossRef]
- Kojima, Y.; Muto, S.; Tatsumi, K.; Kondo, H.; Oka, H.; Horibuchi, K. Degradation analysis of a Ni-based layered positive-electrode active material cycled at elevated temperatures studied by scanning transmission electron microscopy and electron energy-loss spectroscopy. J. Power Sour. 2011, 196, 7721–7727. [Google Scholar] [CrossRef]
- Abraham, D.P.; Reynolds, E.M.; Sammann, E.; Jansen, A.N.; Dees, D.W. Aging characteristics of high-power lithium-ion cells with LiNi0.8Co0.15Al0.05O2 and Li4/3Ti5/3O4 electrodes. Electrochim. Acta 2005, 51, 502–510. [Google Scholar] [CrossRef]
- Watanabe, S.; Kinoshita, M.; Nakura, K. Capacity fade of LiNi(1−x−y)CoxAlyO2 cathode for lithium-ion batteries during accelerated calendar and cycle life test. I. Comparison analysis between LiNi(1−x−y)CoxAlyO2 and LiCoO2 cathodes in cylindrical lithium-ion cells during long term storage test. J. Power Sour. 2014, 247, 412–422. [Google Scholar] [CrossRef]
- Wu, L.; Nam, K.W.; Wang, X.; Zhou, Y.; Zheng, J.C.; Yang, X.Q.; Zhu, Y. Structural origin of overcharge-induced thermal instability of Ni-containing layered-cathodes for high-energy-density lithium batteries. Chem. Mater. 2011, 23, 3953–3960. [Google Scholar] [CrossRef]





| Property | Unit | Graphite | LTO |
|---|---|---|---|
| Theoretical specific capacity | mAh/g | 372 | 175 |
| Potential versus Li/Li+ | V | 0.1 | 1.55 |
| Electronic conductivity | S/cm | 10−4 | 10−13–10−8 |
| Lithium diffusion coefficient | cm/s | 10−11–10−7 | 10−16–10−8 |
| Volume change during lithiation/delithiation | % | 10 | 0.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Sun, Y.; Gao, Y.; Yan, P. The Incremental Capacity Curves and Frequency Response Characteristic Evolution of Lithium Titanate Battery during Ultra-High-Rate Discharging Cycles. Energies 2023, 16, 3434. https://doi.org/10.3390/en16083434
Wang C, Sun Y, Gao Y, Yan P. The Incremental Capacity Curves and Frequency Response Characteristic Evolution of Lithium Titanate Battery during Ultra-High-Rate Discharging Cycles. Energies. 2023; 16(8):3434. https://doi.org/10.3390/en16083434
Chicago/Turabian StyleWang, Chu, Yaohong Sun, Yinghui Gao, and Ping Yan. 2023. "The Incremental Capacity Curves and Frequency Response Characteristic Evolution of Lithium Titanate Battery during Ultra-High-Rate Discharging Cycles" Energies 16, no. 8: 3434. https://doi.org/10.3390/en16083434
APA StyleWang, C., Sun, Y., Gao, Y., & Yan, P. (2023). The Incremental Capacity Curves and Frequency Response Characteristic Evolution of Lithium Titanate Battery during Ultra-High-Rate Discharging Cycles. Energies, 16(8), 3434. https://doi.org/10.3390/en16083434




