Comparative Analysis of Real-Emitted Particulate Matter and PM-Bound Chemicals from Residential and Automotive Sources: A Case Study in Poland
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collecting of Samples of Real-Emitted Particulate Matter from Different Combustion-Related Sources
2.1.1. Sampling of Particulate Matter Emitted from Residential Solid Fuels Combustion
2.1.2. Sampling of Particulate Matter Emitted from Industrial Coal Combustion
2.1.3. Sampling of Particulate Matter Emitted from Diesel-Type Engines
2.2. Determination of the Content of PM-Bound Chemicals
3. Results and Discussion
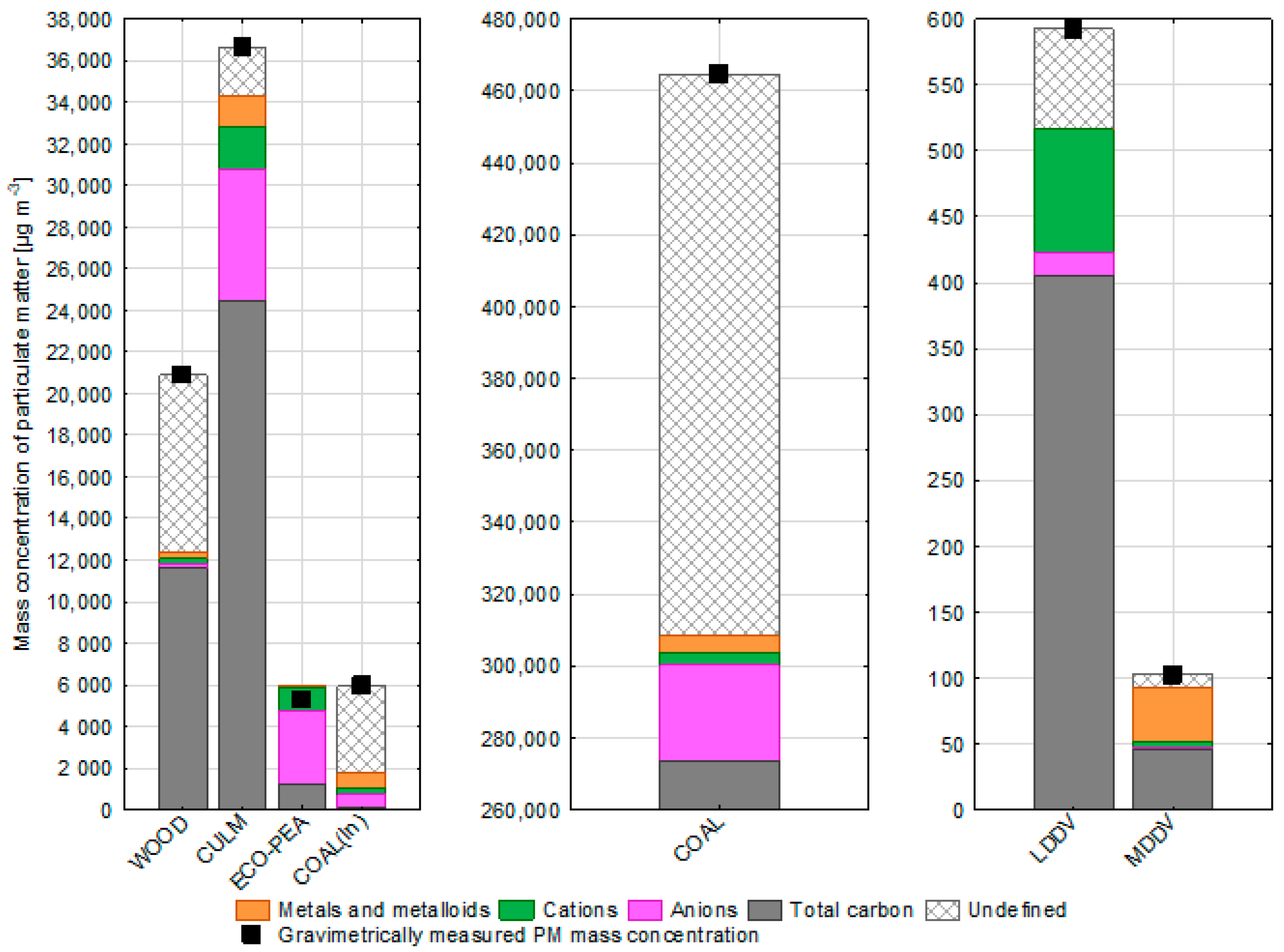
3.1. Real-Emission of Particulate Matter from Different Combustion-Related Sources
3.2. PM-Bound Carbonaceous Compounds
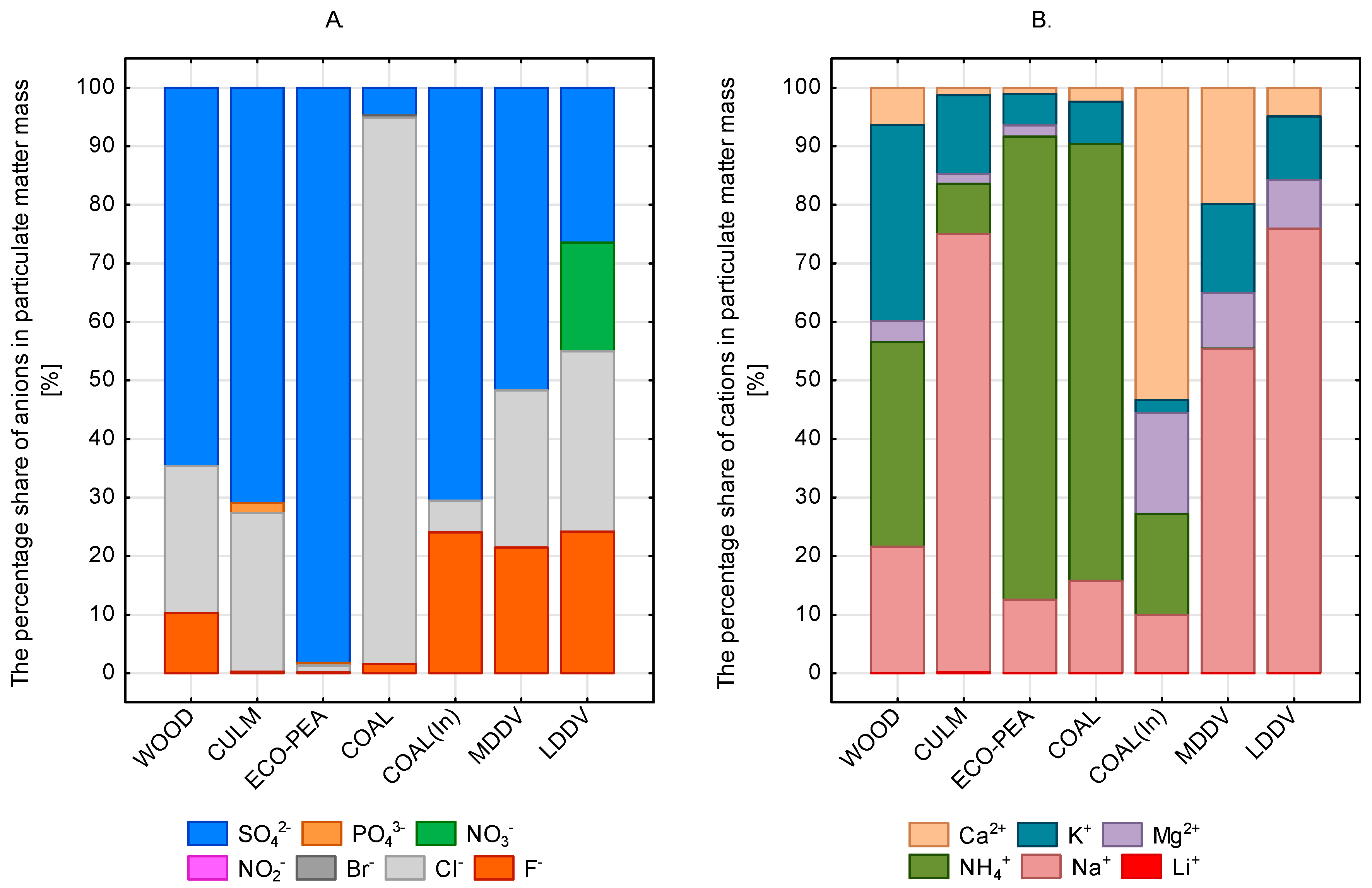
3.3. PM-Bound Inorganic Ions
- Within particulate matter stemming from softwood combustion, sulphate ions took the lead, contributing a significant 60% of the overall anion mass. Chloride ions, though slightly lower, still held substantial prominence, contributing 28% to the anion mass.
- The presence of fluorine ions (F−) was identified in particulate matter arising both from residential coal combustion and the industrial process of coal burning. In the former, the concentration of fluorine ions with a concentration of 500 µg m−3 accounted for 1.5% of the total anion mass, while in the latter, it rose to 25%. The similar contribution (21% and 24%) of F− mass was also noticed for low-duty and medium-duty diesel vehicles, respectively.
- In the context of residential coal combustion, chlorine ions dominated the anion composition, contributing a substantial 95% of the anion mass. In coal burning in a DHU, sulphates shared a smaller proportion, comprising 2% of the anion mass.
- Ecopea coal combustion and bituminous coal combustion in power plants displayed the lowest contributions of chloride ions. In particulate matter from ecopea coal combustion, chlorides contributed a mere 25% of the anion mass. Meanwhile, the anions balance leaned heavily towards sulphate ions, making up almost 100% of the anion mass.
- Chlorines and sulphates contributed in 28% and 70%, respectively, to anions fractions of particulate matter from culm combustion.
- Nitrates were observed only in PM derived from medium-duty diesel vehicles.
- The cumulative concentration of cations in particulate matter from residential coal combustion amounted to 3.600 µg m−3, while from ecopea coal burning, it stood at 1.050 µg m−3. These figures contributed to 0.8% and 20.0% of the particulate matter’s mass, respectively. Notably, cation mass originating from combustion exceeded that from ecopea coal burning by a factor of 2.0, constituting 6% of PM mass.
- The lowest concentrations of cations were registered in particles emitted from softwood combustion (200 µg m−3) and bituminous coal combustion in power plants (346 µg m−3).
- Inorganic ions contributed 2% of PM mass for light-duty diesel vehicles (LDDV) and 8% for medium-duty diesel vehicles (MDDV). Notably, among the cations, sodium cations held the highest contribution, comprising 76% of the cation mass for LDDV and 55% for MDDV. Potassium and calcium also held substantial proportions within this ionic balance.
- In PM from coal and ecopea coal combustion in DHUs, ammonium ions (NH4+) were predominant, contributing to over 80% of the cation mass. Lower NH4+ content was observed in PM from culm (10%), softwood (38%), and power plant emissions (20%).
- In the case of softwood combustion, potassium cations (K+) exhibited a prevalence similar to that of ammonium ions. Sodium cations (Na+) shared about 20% of PM originating from softwood. Culm combustion particles were dominated by sodium ions (75%), while the contributions of K+ and NH4+ were notably lower at 12% and 10%, respectively. In PM from the power plant, cation contributions differed, with the majority of the mass comprising mineral components such as Ca2+ (55%), Mg2+ (20%), NH4+ (19%), and Na+ (10%).
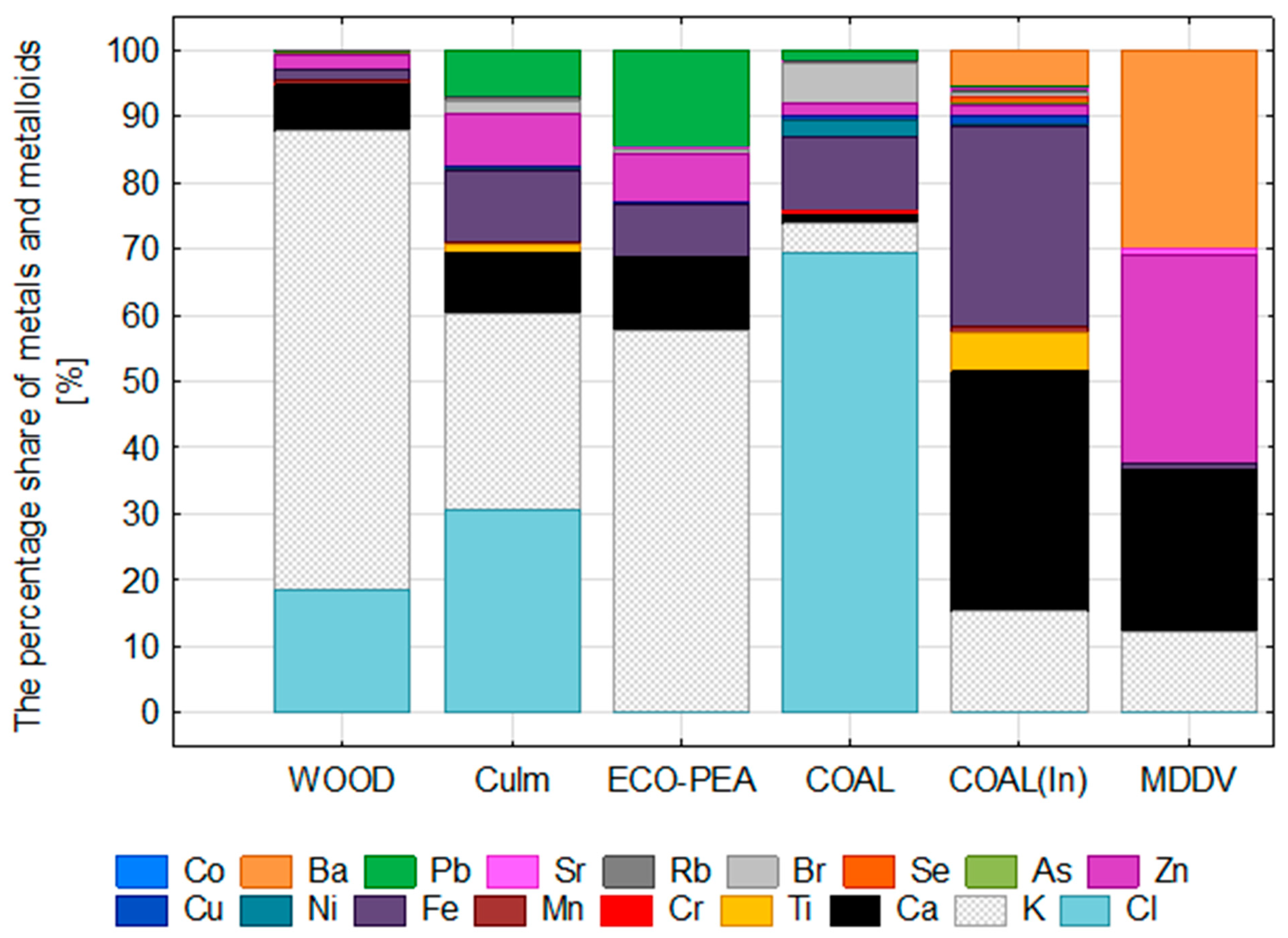
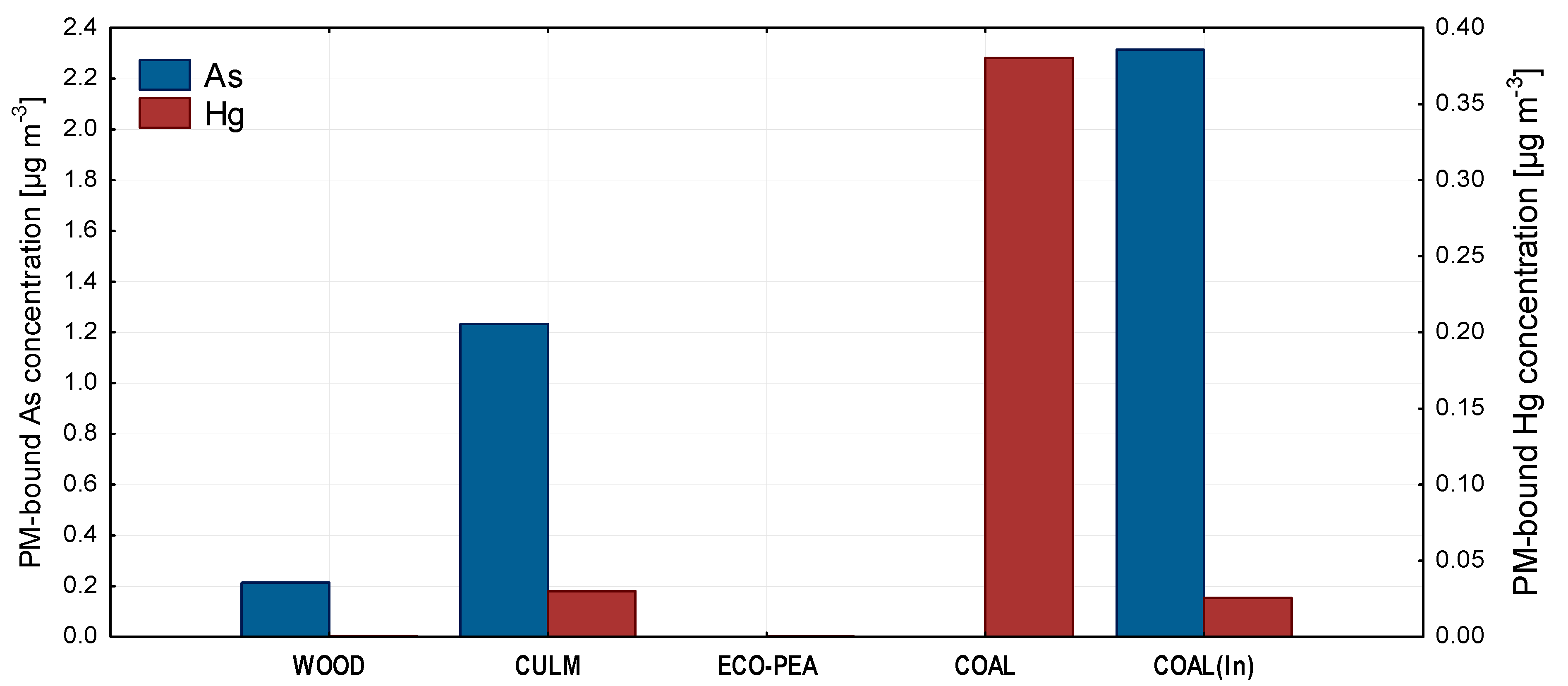
3.4. PM-Bound Metals and Metalloids
4. Identification of Characteristic PM-Bound Chemical for Different Combustion-Related Sources
- High PM emission.
- Dominance of carbonaceous compounds, including organic carbon and elemental carbon, contributing to a significant portion of PM mass.
- Anion contribution around 5.7%, with chloride ions being prominent.
- Low contribution of metals and metalloids (<1% of PM mass), but specific elements like lead, zinc, iron, calcium, and bromide are present.
- Lower PM emission compared to coal combustion.
- Predominance of carbonaceous compounds, with organic carbon constituting a major fraction.
- Anion contribution around 17.3%, with chloride ions being significant.
- Relatively higher contribution of metals and metalloids (1.7–3.8% of PM mass), including lead, zinc, iron, calcium, and bromide.
- Lower PM emission compared to coal combustion.
- Presence of carbonaceous compounds, but less significant compared to coal and culm combustion.
- High anion contribution (67.5% of PM mass), predominantly sulphate ions.
- Moderate contribution of metals and metalloids (1.7–3.8% of PM mass), including lead, zinc, iron, calcium, and bromide.
- Moderate PM emission.
- Dominance of carbonaceous compounds, with a higher contribution of organic carbon.
- Anion contribution around 1.2%, mainly sulphate ions.
- Moderate contribution of metals and metalloids (1.7% of PM mass), including lead, zinc, iron, calcium, and bromide.
- Significant PM emission, meeting emission standards due to effective exhaust aftertreatment.
- Low presence of carbonaceous compounds due to efficient removal mechanisms.
- Anion contribution around 11%, with sulphate ions being prominent.
- Higher contribution of metals and metalloids (>11% of PM mass), including calcium, iron, potassium, titanium, barium, copper, chloride, selenium, and manganese.
- Lower PM emission compared to solid fuel combustion.
- Presence of carbonaceous compounds, with both organic carbon and elemental carbon contributing.
- Anion contribution around 8%, dominated by chloride ions.
- Contribution of metals and metalloids (2% of PM mass), with potassium, calcium, barium, and zinc being dominant.
- Lower PM emission compared to solid fuel combustion.
- Presence of carbonaceous compounds, with both organic carbon and elemental carbon contributing.
- Anion contribution around 2%, mainly chloride ions.
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Styszko, K.; Samek, L.; Szramowiat, K.; Korzeniewska, A.; Kubisty, K.; Rakoczy-Lelek, R.; Kistler, M.; Giebl, A.K. Oxidative Potential of PM10 and PM2.5 Collected at High Air Pollution Site Related to Chemical Composition: Krakow Case Study. Air Qual. Atmos. Health 2017, 10, 1123–1137. [Google Scholar] [CrossRef]
- European Environmental Agency. Europe’s Air Quality Status 2023; European Environmental Agency: Copenhagen, Denmark, 2023. [Google Scholar]
- Szramowiat-Sala, K.; Styszko, K.; Kistler, M.; Kasper-Giebl, A.; Golas, J. Macro Tracer Model as a Technique for Source Apportionment of Particulate Matter in Krakow Agglomeration—An Optimization Approach. In Proceedings of the 24th ETH-Conference on Combustion Generated Nanoparticles, Zurich, Switzerland, 22–24 June 2021. [Google Scholar]
- Samek, L.; Stegowski, Z.; Furman, L.; Styszko, K.; Szramowiat, K.; Fiedor, J. Quantitative Assessment of PM2.5 Sources and Their Seasonal Variation in Krakow. Water Air Soil Pollut. 2017, 228, 290. [Google Scholar] [CrossRef] [PubMed]
- Samek, L.; Stegowski, Z.; Styszko, K.; Furman, L.; Fiedor, J. Seasonal Contribution of Assessed Sources to Submicron and Fine Particulate Matter in a Central European Urban Area. Environ. Pollut. 2018, 241, 406–411. [Google Scholar] [CrossRef]
- Samek, L.; Turek-Fijak, A.; Skiba, A.; Furman, P.; Styszko, K.; Furman, L.; Stegowski, Z. Complex Characterization of Fine Fraction and Source Contribution to PM2.5 Mass at an Urban Area in Central Europe. Atmosphere 2020, 11, 1085. [Google Scholar] [CrossRef]
- Samek, L.; Styszko, K.; Stegowski, Z.; Zimnoch, M.; Skiba, A.; Turek-Fijak, A.; Gorczyca, Z.; Furman, P.; Kasper-Giebl, A.; Rozanski, K. Comparison of PM10 Sources at Traffic and Urban Background Sites Based on Elemental, Chemical and Isotopic Composition: Case Study from Krakow, Southern Poland. Atmosphere 2021, 12, 1364. [Google Scholar] [CrossRef]
- Kaczmarczyk, M.; Kaczmarczyk, M.; Pełka, G.; Luboń, W.; Będkowska, A.; Piechowicz, Ł.; Ciapała, B.; Blok, M. Low Emission: From Its Sources to the Means of Elimination; Kaczmarczyk, M., Ed.; Geosystem Burek, Kotyza S. C.: Kraków, Poland, 2015; ISBN 978-83-64339-02-8. [Google Scholar]
- Zawada, M.; Starostka-Patyk, M. Energy Efficiency in the Context of Low-Stack Emissions Reduction on the Example of the City of Czestochowa. Transp. Res. Procedia 2016, 16, 587–597. [Google Scholar] [CrossRef]
- Szramowiat, K.; Woodburn, J.; Pacura, W.; Berent, K.; Bielaczyc, P.; Gołaś, J. Engine-Generated Solid Particles—A Case Study. Combust. Engines 2018, 174, 33–39. [Google Scholar] [CrossRef]
- Whitby, K.T. The Physical Characteristics of Sulfur Aerosols. Atmos. Environ. 1978, 12, 135–159. [Google Scholar] [CrossRef]
- Szramowiat-Sala, K.; Korzeniewska, A.; Sornek, K.; Marczak, M.; Wierońska, F.; Berent, K.; Gołaś, J.; Filipowicz, M. The Properties of Particulate Matter Generated during Wood Combustion in In-Use Stoves. Fuel 2019, 253, 792–801. [Google Scholar] [CrossRef]
- Maricq, M.M. Engine, Aftertreatment, Fuel Quality and Non-Tailpipe Achievements to Lower Gasoline Vehicle PM Emissions: Literature Review and Future Prospects. Sci. Total Environ. 2023, 866, 161225. [Google Scholar] [CrossRef]
- Macherzyński, M. Reduction of Mercury Emission to the Environment—Selected Problems in the Aspect of Laboratory and Industrial Research [Original Title: Redukcja Emisji Rtęci Do Środowiska—Wybrane Problemy w Świetle Badań Laboratoryjnych i Przemysłowych]; AGH University of Science and Technology Press: Kraków, Poland, 2018. [Google Scholar]
- Sani, F.M.; Oyelaran, O.A. Exhaust Gas Treatment in Thermal Power Plants: A Review. Int. J. Adv. Sci. Res. Eng. 2019, 5, 227–233. [Google Scholar] [CrossRef]
- Ryšavý, J.; Serenčíšová, J.; Horák, J.; Ochodek, T. The Co-Combustion of Pellets with Pistachio Shells in Residential Units Additionally Equipped by Pt-Based Catalyst. Biomass Convers. Biorefinery 2023, 1–17. [Google Scholar] [CrossRef]
- Ryšavý, J.; Horák, J.; Hopan, F.; Kuboňová, L.; Krpec, K.; Molchanov, O.; Garba, M.; Ochodek, T. Influence of Flue Gas Parameters on Conversion Rates of Honeycomb Catalysts. Sep. Purif. Technol. 2021, 278, 119491. [Google Scholar] [CrossRef]
- Kuye, A.; Kumar, P. A Review of the Physicochemical Characteristics of Ultrafine Particle Emissions from Domestic Solid Fuel Combustion during Cooking and Heating. Sci. Total Environ. 2023, 886, 163747. [Google Scholar] [CrossRef]
- Dusiło, M. Energy Transformation in Poland; AmCham.PL: Warszawa, Poland, 2022. [Google Scholar]
- Zyśk, J.; Wyrwa, A.; Suwała, W.; Pluta, M.; Olkuski, T.; Raczyński, M. The Impact of Decarbonization Scenarios on Air Quality and Human Health in Poland—Analysis of Scenarios up to 2050. Atmosphere 2020, 11, 1222. [Google Scholar] [CrossRef]
- Wang, G.; Deng, J.; Zhang, Y.; Zhang, Q.; Duan, L.; Hao, J.; Jiang, J. Air Pollutant Emissions from Coal-Fired Power Plants in China over the Past Two Decades. Sci. Total Environ. 2020, 741, 140326. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Jiang, J.; Ma, Z.; Wang, S.; Duan, L. Effect of Selective Catalytic Reduction (SCR) on Fine Particle Emission from Two Coal-Fired Power Plants in China. Atmos. Environ. 2015, 120, 227–233. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, J.; Yu, X.; Sun, P.; Zhao, Y.; Zhang, J.; Chen, G.; Yao, H.; Zheng, C. Migration and Emission Characteristics of Hg in Coal-Fired Power Plant of China with Ultra Low Emission Air Pollution Control Devices. Fuel Process. Technol. 2017, 158, 272–280. [Google Scholar] [CrossRef]
- Wang, C.; Liu, X.; Li, D.; Si, J.; Zhao, B.; Xu, M. Measurement of Particulate Matter and Trace Elements from a Coal-Fired Power Plant with Electrostatic Precipitators Equipped the Low Temperature Economizer. Proc. Combust. Inst. 2015, 35, 2793–2800. [Google Scholar] [CrossRef]
- Colvile, R.; Hutchinson, E.; Mindell, J.; Warren, R. The Transport Sector as a Source of Air Pollution. Atmos. Environ. 2001, 35, 1537–1565. [Google Scholar] [CrossRef]
- European Automobile Manufacturers Association. The Automobile Industry—Pocket Guide 2022/2023; ACEA: Brussels, Belgium, 2023. [Google Scholar]
- Jung, J.C.; Sharon, E. The Volkswagen Emissions Scandal and Its Aftermath. Glob. Bus. Organ. Excell. 2019, 38, 6–15. [Google Scholar] [CrossRef]
- Harris, S.J.; Maricq, M.M. Signature Size Distributions for Diesel and Gasoline Engine Exhaust Particulate Matter. J. Aerosol Sci. 2001, 32, 749–764. [Google Scholar] [CrossRef]
- CSN EN 13284-1; Stationary Sources Emissions—Determination of Low Range Mass Concentration of Dust—Part 1: Manual Gravimetric Method. European Standards: Plzen, Czech Republic, 2017.
- PN-Z-04030-7:1994; Air Protection against Pollution—Dust Concentration Measurement—Measurement of Concentration and Mass Flow of Dust in Flue Gases by the Gravimetric Method. 1994.
- Samek, L.; Stegowski, Z.; Furman, L. Preliminary PM2.5 and PM10 Fractions Source Apportionment Complemented by Statistical Accuracy Determination. Nukleonika 2016, 61, 75–83. [Google Scholar] [CrossRef]
- Samek, L.; Furman, L.; Kawik, T.; Welnogorska, K. Application of X-Ray Fluorescence Method for Elemental Analysis of PM2.5 Fraction. Nukleonika 2015, 60, 621. [Google Scholar] [CrossRef]
- Vekemans, B.; Janssens, K.; Vincze, L.; Adams, F.; Van Espen, P. Analysis of X-Ray Spectra by Iterative Least Squares (AXIL): New Developments. X-Ray Spectrom. 1994, 23, 278–285. [Google Scholar] [CrossRef]
- EPA. Method 7473 (SW-846): Mercury in Solids and Solutions by Thermal Decomposition, Amalgamation, and Atomic Absorption Spectrophotometry; EPA: Washington, DC, USA, 1998. [Google Scholar]
- D6722-19; Standard Test Method for Total Mercury in Coal and Coal Combustion Residues by Direct Combustion Analysis. ASTM: West Conshohocken, PA, USA, 2001.
- Styszko, K.; Szramowiat, K.; Kistler, M.; Kasper-Giebl, A.; Samek, L.; Furman, L.; Pacyna, J.; Gołaś, J. Mercury in Atmospheric Aerosols: A Preliminary Case Study for the City of Krakow, Poland. Comptes Rendus Chim. 2015, 18, 1183–1191. [Google Scholar] [CrossRef]
- Szramowiat, K.; Styszko, K.; Kistler, M.; Kasper-Giebl, A.; Gołaś, J. Carbonaceous Species in Atmospheric Aerosols from the Krakow Area (Malopolska District): Carbonaceous Species Dry Deposition Analysis. E3S Web Conf. 2016, 10, 92. [Google Scholar] [CrossRef]
- Jankowski, N.; Schmidl, C.; Marr, I.L.; Bauer, H.; Puxbaum, H. Comparison of Methods for the Quantification of Carbonate Carbon in Atmospheric PM10 Aerosol Samples. Atmos. Environ. 2008, 42, 8055–8064. [Google Scholar] [CrossRef]
- Pacura, W.; Szramowiat-Sala, K.; Macherzyński, M.; Gołaś, J.; Bielaczyc, P. Analysis of Micro-Contaminants in Solid Particles from Direct Injection Gasoline Vehicles. Energies 2022, 15, 5732. [Google Scholar] [CrossRef]
- Commission Implementing Decision of 9 October 2014 Establishing Best Available Techniques (BAT) Conclusions, under Directive 2010/75/EU of the European Parliament and of the Council on Industrial Emissions, for the Refining of Mineral Oil and Gas. Available online: https://eur-lex.europa.eu/eli/dec_impl/2014/738/oj (accessed on 16 August 2023).
- Huang , Q.; Li, S.; Li, G.; Yao, Q. Mechanisms on the Size Partitioning of Sodium in Particulate Matter from Pulverized Coal Combustion. Combust. Flame 2017, 182, 313–323. [Google Scholar] [CrossRef]
- Li, G.; Li, S.; Dong, M.; Yao, Q.; Guo, C.Y.; Axelbaum, R.L. Comparison of Particulate Formation and Ash Deposition under Oxy-Fuel and Conventional Pulverized Coal Combustions. Fuel 2013, 106, 544–551. [Google Scholar] [CrossRef]
- Gao, X.; Rahim, M.U.; Chen, X.; Wu, H. Inorganic PM10 Emission from the Combustion of Individual Mallee Components and Whole-Tree Biomass. Proc. Combust. Inst. 2017, 36, 3313–3319. [Google Scholar] [CrossRef]
- Wang, W.; Wen, C.; Li, C.; Wang, M.; Li, X.; Zhou, Y.; Gong, X. Emission Reduction of Particulate Matter from the Combustion of Biochar via Thermal Pre-Treatment of Torrefaction, Slow Pyrolysis or Hydrothermal Carbonisation and Its Co-Combustion with Pulverized Coal. Fuel 2019, 240, 278–288. [Google Scholar] [CrossRef]
- Kittelson, D.B. Engines and Nanoparticles: A Review. J. Aerosol Sci. 1998, 29, 575–588. [Google Scholar] [CrossRef]
- Shi, J.P.; Harrison, R.M.; Brear, F. Particle Size Distribution from a Modern Heavy Duty Diesel Engine. Sci. Total Environ. 1999, 235, 305–317. [Google Scholar] [CrossRef]
- Pio, C.; Mirante, F.; Oliveira, C.; Matos, M.; Caseiro, A.; Oliveira, C.; Querol, X.; Alves, C.; Martins, N.; Cerqueira, M.; et al. Size-Segregated Chemical Composition of Aerosol Emissions in an Urban Road Tunnel in Portugal. Atmos. Environ. 2013, 71, 15–25. [Google Scholar] [CrossRef]
- He, C.; Li, J.; Wang, Y.; Tan, J.; Song, G.; Jia, D.; Zhao, L. Size-Segregated Particulate Matter Emission Characteristics of a Heavy-Duty Diesel Engine with Oxygenated Fuels. Appl. Therm. Eng. 2017, 125, 1173–1180. [Google Scholar] [CrossRef]
- Nussbaumer, T.; Klippel, N.; Johansson, L. Survey on Measurements and Emission Factors on Particulate Matter from Biomass Combustion in IEA Countries. In Proceedings of the 16th European Biomass Conference and Exhibition, Valencia, Spain, 2–6 June 2008. [Google Scholar]
- Sillanpää, M.; Hillamo, R.; Saarikoski, S.; Frey, A.; Pennanen, A.; Makkonen, U.; Spolnik, Z.; Van Grieken, R.; Braniš, M.; Brunekreef, B.; et al. Chemical Composition and Mass Closure of Particulate Matter at Six Urban Sites in Europe. Atmos. Environ. 2006, 40 (Suppl. S2), 212–223. [Google Scholar] [CrossRef]
- Popovicheva, O.B.; Kireeva, E.D.; Steiner, S.; Rothen-Rutishauser, B.; Persiantseva, N.M.; Timofeev, M.A.; Shonija, N.K.; Comte, P.; Czerwinski, J. Microstructure and Chemical Composition of Diesel and Biodiesel Particle Exhaust. Aerosol Air Qual. Res. 2014, 14, 1392–1401. [Google Scholar] [CrossRef]
| Fuel Kind | DHU Type | Sampling Time |
|---|---|---|
| Soft wood | Stove, nominal power: 24 kW, efficiency: 83.4% | 480–1800 s |
| Culm | Steel heating boiler with water-cooling water, nominal power 30 kW, efficiency 84% | 600 s |
| Ecopea coal | Central heating unit with a stove dedicated for ecopea coal fuelling | 600 s |
| Bituminous coal | Central heating unit a cast iron stove, nominal power 18 kW, efficiency 77% | 600 s |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szramowiat-Sala, K.; Styszko, K.; Samek, L.; Kistler, M.; Macherzyński, M.; Ryšavý, J.; Krpec, K.; Horák, J.; Kasper-Giebl, A.; Gołaś, J. Comparative Analysis of Real-Emitted Particulate Matter and PM-Bound Chemicals from Residential and Automotive Sources: A Case Study in Poland. Energies 2023, 16, 6514. https://doi.org/10.3390/en16186514
Szramowiat-Sala K, Styszko K, Samek L, Kistler M, Macherzyński M, Ryšavý J, Krpec K, Horák J, Kasper-Giebl A, Gołaś J. Comparative Analysis of Real-Emitted Particulate Matter and PM-Bound Chemicals from Residential and Automotive Sources: A Case Study in Poland. Energies. 2023; 16(18):6514. https://doi.org/10.3390/en16186514
Chicago/Turabian StyleSzramowiat-Sala, Katarzyna, Katarzyna Styszko, Lucyna Samek, Magdalena Kistler, Mariusz Macherzyński, Jiří Ryšavý, Kamil Krpec, Jiří Horák, Anne Kasper-Giebl, and Janusz Gołaś. 2023. "Comparative Analysis of Real-Emitted Particulate Matter and PM-Bound Chemicals from Residential and Automotive Sources: A Case Study in Poland" Energies 16, no. 18: 6514. https://doi.org/10.3390/en16186514
APA StyleSzramowiat-Sala, K., Styszko, K., Samek, L., Kistler, M., Macherzyński, M., Ryšavý, J., Krpec, K., Horák, J., Kasper-Giebl, A., & Gołaś, J. (2023). Comparative Analysis of Real-Emitted Particulate Matter and PM-Bound Chemicals from Residential and Automotive Sources: A Case Study in Poland. Energies, 16(18), 6514. https://doi.org/10.3390/en16186514







