A Paradox over Electric Vehicles, Mining of Lithium for Car Batteries
Abstract
:1. Introduction
2. Scope of the Review
3. General Aspects of EVs
4. Lithium Sources, Other Metals, and Production
4.1. Lithium Reserves
4.2. Other Precious Metals
4.3. Suppliers of Lithium and Lithium Reserves
5. Types of Lithium Batteries and Their Characteristics
5.1. Types of Batteries
5.2. Battery Life
5.3. Extra Energy and CO2 Released for Making EVs
6. Technical Challenges and Social Aspects
6.1. Recycling of Spent LiBs
6.2. Dark Sides of Lithium Mining and Extraction
7. Trends and Future Possibilities
7.1. Advances in Battery Technology
7.2. Research in Battery Manufacturing Technology
7.3. Second-Life Applications of Spent LiBs
7.4. Safety Issues and State-of-Charge (SOC)
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Lithium Battery Safety (University of Washington). Available online: https://www.ehs.washington.edu/system/files/resources/lithium-battery-safety.pdf (accessed on 25 October 2022).
- Straubel, J.B. Driving Range for the Model S Family. Available online: https://www.tesla.com/en_CA/blog/driving-range-model-s-family (accessed on 18 October 2022).
- Berjoza, D.; Jurgena, I. Effects of change in the weight of electric vehicles on their performance characteristics. Agron. Res. 2017, 15, 952–963. [Google Scholar]
- Carbon Dioxide Emissions from Electricity (World Nuclear Association). Available online: https://www.world-nuclear.org/information-library/energy-and-the-environment/carbon-dioxide-emissions-from-electricity.aspx (accessed on 18 October 2022).
- Yang, S.; Zhang, F.; Ding, H.; He, P.; Zhou, H. Lithium metal extraction from seawater. Joule 2018, 2, 1648–1651. [Google Scholar] [CrossRef] [Green Version]
- Go All out with the All-New 2023 Outlander PHEV. Available online: https://www.mitsubishicars.com/2023-outlander-phev-release-date (accessed on 6 October 2022).
- Plötz, P.; Moll, C.; Bieker, G.; Mock, P.; Li, Y. Real-World Usage of Plug-In Hybrid Electric Vehicles: Fuel Consumption, Electric Driving, and CO2 Emissions; Technical Report; International Council on Clean Transportation Europe (ICCT): Washington, DC, USA, 2020; Available online: https://theicct.org/publication/real-world-usage-of-plug-in-hybrid-electric-vehicles-fuel-consumption-electric-driving-and-co2-emissions/ (accessed on 6 October 2022).
- The Car Guide. 2014 Toyota Prius PHV: To Plug in or Not to Plug in? 2014. Available online: https://www.guideautoweb.com/en/articles/21152/2014-toyota-prius-phv-to-plug-in-or-not-to-plug-in/ (accessed on 6 October 2021).
- Hyundai NEXO Press Kit. Available online: https://www.hyundai.news/eu/models/electrified/nexo/press-kit.html (accessed on 6 October 2022).
- insideEVs. 2019 BMW i3, i3 REx, i3s & i3s REx: Full Specs. 2019. Available online: https://insideevs.com/news/339970/2019-bmw-i3-i3-rex-i3s-amp-i3s-rex-full-specs/ (accessed on 6 October 2022).
- Yong, J.Y.; Ramachandaramurthy, V.K.; Tan, K.M.; Mithulananthan, N. A review on the state-of-the-art technologies of electric vehicle, its impacts and prospects. Renew. Sustain. Energy Rev. 2015, 49, 365–385. [Google Scholar] [CrossRef]
- Richardson, D.B. Electric vehicles and the electric grid: A review of modeling approaches, Impacts, and renewable energy integration. Renew. Sustain. Energy Rev. 2013, 19, 247–254. [Google Scholar] [CrossRef]
- Habib, S.; Kamran, M.; Rashid, U. Impact analysis of vehicle-to-grid technology and charging strategies of electric vehicles on distribution networks—A review. J. Power Sources 2015, 277, 205–214. [Google Scholar] [CrossRef]
- Liu, L.; Kong, F.; Liu, X.; Peng, Y.; Wang, Q. A review on electric vehicles interacting with renewable energy in smart grid. Renew. Sustain. Energy Rev. 2015, 51, 648–661. [Google Scholar] [CrossRef]
- Hawkins, T.R.; Gausen, O.M.; Strømman, A.H. Environmental impacts of hybrid and electric vehicles—A review. Int. J. Life Cycle Assess. 2012, 17, 997–1014. [Google Scholar] [CrossRef]
- Shuai, W.; Maillé, P.; Pelov, A. Charging electric vehicles in the smart city: A survey of economy-driven approaches. IEEE Trans. Intell. Transp. Syst. 2016, 17, 2089–2106. [Google Scholar] [CrossRef] [Green Version]
- Hu, J.; Morais, H.; Sousa, T.; Lind, M. Electric vehicle fleet management in smart grids: A review of services, optimization and control aspects. Renew. Sustain. Energy Rev. 2016, 56, 1207–1226. [Google Scholar] [CrossRef] [Green Version]
- Tan, K.M.; Ramachandaramurthy, V.K.; Yong, J.Y. Integration of electric vehicles in smart grid: A review on vehicle to grid technologies and optimization techniques. Renew. Sustain. Energy Rev. 2016, 53, 720–732. [Google Scholar] [CrossRef]
- Rahman, I.; Vasant, P.M.; Singh, B.S.M.; Abdullah-Al-Wadud, M.; Adnan, N. Review of recent trends in optimization techniques for plug-in hybrid, and electric vehicle charging infrastructures. Renew. Sustain. Energy Rev. 2016, 58, 1039–1047. [Google Scholar] [CrossRef]
- Mahmud, K.; Town, G.E.; Morsalin, S.; Hossain, M. Integration of electric vehicles and management in the internet of energy. Renew. Sustain. Energy Rev. 2018, 82, 4179–4203. [Google Scholar] [CrossRef]
- Das, H.; Rahman, M.; Li, S.; Tan, C. Electric vehicles standards, charging infrastructure, and impact on grid integration: A technological review. Renew. Sustain. Energy Rev. 2020, 120, 109618. [Google Scholar] [CrossRef]
- Li, Y.; Liu, K.; Foley, A.M.; Zülke, A.; Berecibar, M.; Nanini-Maury, E.; Van Mierlo, J.; Hoster, H.E. Data-driven health estimation and lifetime prediction of lithium-ion batteries: A review. Renew. Sustain. Energy Rev. 2019, 113, 109254. [Google Scholar] [CrossRef]
- Liu, K.; Li, Y.; Hu, X.; Lucu, M.; Widanage, W.D. Gaussian process regression with automatic relevance determination kernel for calendar aging prediction of lithium-ion batteries. IEEE Trans. Ind. Inform. 2020, 16, 3767–3777. [Google Scholar] [CrossRef] [Green Version]
- Hu, X.; Zhang, K.; Liu, K.; Lin, X.; Dey, S.; Onori, S. Advanced fault diagnosis for lithium-ion battery systems: A review of fault mechanisms, fault features, and diagnosis procedures. IEEE Ind. Electron. Mag. 2020, 14, 65–91. [Google Scholar] [CrossRef]
- The Barents Observer. Available online: https://thebarentsobserver.com/en/industry-and-energy/2021/01/longyearbyen-shut-down-coal-power-plant (accessed on 6 October 2022).
- Energy in India Today. Available online: https://www.iea.org/reports/india-energy-outlook-2021/energy-in-india-today (accessed on 6 October 2022).
- China Sector Analysis: Energy. Available online: https://www.globalxetfs.com/china-sector-analysis-energy/#:~:text=Background%20of%20the%20Energy%20Sector%20in%20China&text=This%20distribution%20network%20relies%20on,China’s%20power%20generation%20of%202021 (accessed on 6 October 2022).
- Electricity Production and Distribution. Available online: https://afdc.energy.gov/fuels/electricity_production.html#:~:text=According%20to%20the%20U.S.%20Energy,biomass%2C%20wind%2C%20and%20geothermal (accessed on 6 October 2022).
- Shafique, M.; Azam, A.; Rafiq, M.; Luo, X. Life cycle assessment of electric vehicles and internal combustion engine vehicles: A case study of Hong Kong. Res. Transp. Econ. 2022, 91, 101112. [Google Scholar] [CrossRef]
- Shafique, M.; Luo, X. Environmental life cycle assessment of battery electric vehicles from the current and future energy mix perspective. J. Environ. Manag. 2022, 303, 114050. [Google Scholar] [CrossRef] [PubMed]
- The DRC Mining Industry: Child Labor and Formalization of Small-Scale Mining. Available online: https://www.wilsoncenter.org/blog-post/drc-mining-industry-child-labor-and-formalization-small-scale-mining#:~:text=Cobalt%20is%20an%20essential%20raw,%2C%20environmental%20abuses%2C%20and%20corruption (accessed on 6 October 2022).
- Shafique, M.; Rafiq, M.; Azam, A.; Luo, X. Material flow analysis for end-of-life lithium-ion batteries from battery electric vehicles in the USA and China. Resour. Conser. Recyc. 2022, 178, 106061. [Google Scholar] [CrossRef]
- Nickel–Metal Hydride Battery. Available online: https://en.wikipedia.org/wiki/Nickel%E2%80%93metal_hydride_battery (accessed on 18 October 2022).
- BU-705: How to Recycle Batteries (Battery University). Available online: https://batteryuniversity.com/article/bu-705-how-to-recycle-batteries (accessed on 18 October 2022).
- Chen, Y.; Kang, Y.; Zhao, Y.; Wang, L.; Liu, J.; Li, Y.; Liang, Z.; He, X.; Li, X.; Tavajohi, N.; et al. A review of lithium-ion battery safety concerns: The issues, strategies, and testing standards. J. Energy Chem. 2021, 59, 83–99. [Google Scholar] [CrossRef]
- Tadesse, B.; Makuei, F.; Albijanic, B.; Dyer, L. The beneficiation of lithium minerals from hard rock ores: A review. Miner. Eng. 2019, 131, 170–184. [Google Scholar] [CrossRef]
- Meng, F.; McNeice, J.; Zadeh, S.S.; Ghahreman, A. Review of lithium production and recovery from minerals, brines, and lithium-ion batteries. Miner. Process. Extr. Met. Rev. 2019, 42, 123–141. [Google Scholar] [CrossRef]
- US Geological Survey. Mineral Commodities Summary 2021; U.S. Geological Survey: Reston, VA, USA, 2021.
- Castor, S.B.; Henry, C.D. Lithium-rich claystone in the McDermitt Caldera, Nevada, USA: Geologic, mineralogical, and geochemical characteristics and possible origin. Minerals 2020, 10, 68. [Google Scholar] [CrossRef] [Green Version]
- US Geological Survey. Mineral Commodities Summary 2020; U.S. Geological Survey: Reston, VA, USA, 2020; p. 204.
- Mohr, S.H.; Mudd, G.M.; Giurco, D. Lithium resources and production: Critical assessment and global projections. Minerals 2012, 2, 65–84. [Google Scholar] [CrossRef]
- Xu, P.; Hong, J.; Qian, X.M.; Xu, Z.W.; Xia, H.; Tao, X.C.; Xu, Z.Z.; Ni, Q.Q. Materials for lithium recovery from salt lake brine. J. Mater. Sci. 2021, 56, 16–63. [Google Scholar] [CrossRef]
- Harvard International Review. Available online: https://hir.harvard.edu/lithium-triangle/ (accessed on 18 October 2022).
- Australian Mineral Facts. Available online: https://www.ga.gov.au/education/classroom-resources/minerals-energy/australian-mineral-facts (accessed on 18 October 2022).
- STT, Storage & Transfer. Available online: https://www.sttsystems.com/solutions/lithium-extraction/ (accessed on 18 October 2022).
- Albemarle Corporation. Lithium Resources. 2020. Available online: https://www.albemarle.com/businesses/lithium/resources--recycling/lithium-resources (accessed on 6 October 2020).
- What Is Lithium Extraction and How Does It Work? Samco: Buffalo, NY, USA, 2018; Available online: https://samcotech.com/what-is-lithium-extraction-and-how-does-it-work/ (accessed on 6 October 2022).
- Boryta, D.A. Removal of boron from lithium chloride brine. U.S. Patent 4,261,960, 14 April 1981. [Google Scholar]
- Brown, P.M.; Jacob, S.R.; Boryta, D.A. Production of Highly Pure Lithium Chloride from Impure Brines. U.S. Patent 4,271,131, 2 June 1981. [Google Scholar]
- Aral, A.; Vecchio-Sadus, A. Lithium: Environmental Pollution and Health Effects; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Dapcevich, M. Does Viral Photo Show a ‘Toxic’ Lithium Extraction Field? 2022. Available online: https://www.snopes.com/fact-check/photo-lithium-extraction-mines/ (accessed on 6 October 2022).
- Els, F. All the Mines Tesla Needs to Build 20 Million Cars a Year. Available online: https://www.mining.com/all-the-mines-tesla-needs-to-build-20-million-cars-a-year/ (accessed on 18 October 2022).
- US Cobalt Spot Price. Available online: https://ycharts.com/indicators/us_cobalt_spot_price (accessed on 18 October 2022).
- The Cost of Cobalt. Available online: https://www.aljazeera.com/program/people-power/2021/4/1/the-cost-of-cobalt (accessed on 18 October 2022).
- Li, W.; Lee, S.; Manthiram, A. High-nickel NMA: A cobalt-free alternative to NMC and NCA cathodes for lithium-ion batteries. Adv. Mater. 2020, 32, 200271. [Google Scholar] [CrossRef] [PubMed]
- Electrek. Available online: https://electrek.co/2022/04/22/tesla-using-cobalt-free-lfp-batteries-in-half-new-cars-produced/#:~:text=April%2022,Tesla%20is%20already%20using%20cobalt%2Dfree%20LFP%20batteries%20in,of%20its%20new%20cars%20produced&text=Tesla%20confirmed%20that%20nearly%20half,%2Dphosphate%20(LFP)%20batteries (accessed on 6 October 2022).
- Reserves of Cobalt Worldwide in 2021, by Country (in Metric Tons). Available online: https://www.statista.com/statistics/264930/global-cobalt-reserves/ (accessed on 6 October 2022).
- Volkswagen. Available online: https://www.volkswagenag.com/en/news/stories/2020/03/lithium-mining-what-you-should-know-about-the-contentious-issue.html#:~:text=The%20total%20global%20reserves%20are,the%20production%20volume%20in%202018.&text=Where%20is%20the%20most%20lithium,and%20Argentina%20(6%2C200%20tons) (accessed on 6 October 2022).
- Nickel Reserves: Top 8 Countries (Updated 2022). Available online: https://investingnews.com/daily/resource-investing/base-metals-investing/nickel-investing/nickel-reserves-by-country/ (accessed on 6 October 2022).
- Manganese Reserves by Country. Available online: https://investingnews.com/daily/resource-investing/battery-metals-investing/manganese-investing/manganese-reserves/ (accessed on 6 October 2022).
- Lithium Facts. Available online: https://www.nrcan.gc.ca/our-natural-resources/minerals-mining/minerals-metals-facts/lithium-facts/24009 (accessed on 18 October 2022).
- Lithium Data Sheet—Mineral Commodity Summaries 2020. Available online: https://pubs.usgs.gov/periodicals/mcs2020/mcs2020-lithium.pdf (accessed on 18 October 2022).
- Stringfellow, W.T.; Dobson, P.F. Technology for the recovery of lithium from geothermal brines. Energies 2021, 14, 6805. [Google Scholar] [CrossRef]
- Lithium Mining in China. Available online: http://www.lithiummine.com/lithium-mining-in-china (accessed on 6 October 2022).
- California’s Lithium Rush for ev Batteries Hinges on Taming Toxic, Volcanic Brine. Available online: https://www.forbes.com/sites/alanohnsman/2022/08/31/californias-lithium-rush-electric-vehicles-salton-sea/?sh=79f919694f63 (accessed on 6 October 2022).
- Lithium Shortages: Threat or Opportunity? Available online: https://www.mining-technology.com/analysis/lithium-price-challenges/#:~:text=Supply%20and%20demand,300%25%20between%202021%20and%202030 (accessed on 6 October 2022).
- A Guide to the 6 Main Types of Lithium Batteries. Available online: https://dragonflyenergy.com/types-of-lithium-batteries-guide/ (accessed on 6 October 2022).
- Tesla Model S. Available online: https://en.wikipedia.org/wiki/Tesla_Model_S (accessed on 6 October 2022).
- Plungis, J. How Long Do Electric Car Batteries Last? 2021. Available online: https://www.carfax.com/blog/how-long-do-electric-car-batteries-last#:~:text=All%20EV%20batteries%20will%20lose,years%20to%20about%20132%20miles (accessed on 6 October 2022).
- Tucker, S. Warranty Coverage for Hybrid and EV Batteries. 2022. Available online: https://www.kbb.com/car-advice/hybrid-ev-battery-warranty/#:~:text=Federal%20law%20requires%20automakers%20to,standard%20in%20all%2050%20states (accessed on 6 October 2022).
- BU-802: What Causes Capacity Loss? Available online: https://batteryuniversity.com/article/bu-802-what-causes-capacity-loss (accessed on 6 October 2022).
- Pender, J.P.; Jha, G.; Youn, D.H.; Ziegler, J.M.; Andoni, I.; Choi, E.J.; Heller, A.; Dunn, B.S.; Weiss, P.S.; Penner, R.M.; et al. Electrode degradation in lithium-ion batteries. ACS Nano 2020, 14, 1243–1295. [Google Scholar] [CrossRef] [Green Version]
- Manufacturing the Battery for One Electric Car Produces the Same Amount of Carbon Dioxide as Running a Petrol Car for Eight Years. Available online: https://www.politifact.com/factchecks/2021/may/11/viral-image/producing-electric-cars-battery-does-not-emit-same/ (accessed on 22 October 2022).
- Kukreja, B. Life Cycle Analysis of Electric Vehicles; Quantifying the Impact. 2018. Available online: https://sustain.ubc.ca/sites/default/files/2018-63%20Lifecycle%20Analysis%20of%20Electric%20Vehicles_Kukreja.pdf (accessed on 6 October 2022).
- Canada’s Renewable Power—British Columbia. Available online: https://www.cer-rec.gc.ca/en/data-analysis/energy-commodities/electricity/report/canadas-renewable-power/provinces/renewable-power-canada-british-columbia.html (accessed on 18 October 2022).
- Lutsey, N.P.; Hall, D. Effects of Battery Manufacturing on Electric Vehicle Life-Cycle Greenhouse Gas Emissions. February 2018. Project: Electric Vehicle Life Cycle. International Council on Clean Transportation. Available online: https://www.researchgate.net/publication/323118874_Effects_of_battery_manufacturing_on_electric_vehicle_life-cycle_greenhouse_gas_emissions (accessed on 6 October 2022).
- Bruna Alves, B. Distribution of Electricity Production in Norway 2020, by Source. 2022. Available online: https://www.statista.com/statistics/1025497/distribution-of-electricity-production-in-norway-by-source/#:~:text=In%202020%2C%20hydro%20accounted%20for,142%20terawatt%2Dhours%20in%202020 (accessed on 6 October 2022).
- Nuclear Energy in France. 2007. Available online: https://franceintheus.org/spip.php?article637 (accessed on 6 October 2022).
- Electricity Sector in Germany. Available online: https://en.wikipedia.org/wiki/Electricity_sector_in_Germany#:~:text=The%20top%20producers%20were%20the,%25%20biomass%2C%203.7%25%20hydroelectricity (accessed on 6 October 2022).
- Bieker, G. A global Comparison of the Life-Cycle Greenhouse Gas Emissions of Combustion Engine and Electric Passenger Cars. 2021. Available online: https://theicct.org/publication/a-global-comparison-of-the-life-cycle-greenhouse-gas-emissions-of-combustion-engine-and-electric-passenger-cars/ (accessed on 6 October 2022).
- Castelvecchi, D. Electric cars and batteries: How will the world produce enough? Nature 2021, 596, 336–339. [Google Scholar] [CrossRef]
- Lei, C.; Aldous, I.; Hartley, J.M.; Thompson, D.L.; Scott, S.; Hanson, R.; Anderson, P.A.; Kendrick, E.; Sommerville, R.; Ryder, K.S.; et al. Lithium ion battery recycling using high-intensity ultrasonication. Green Chem. 2021, 23, 4710–4715. [Google Scholar] [CrossRef]
- Jacoby, M. It’s Time to Get Serious about Recycling Lithium-Ion Batteries. C&EN 2019, 97. Available online: https://cen.acs.org/materials/energy-storage/time-serious-recycling-lithium/97/i28 (accessed on 4 October 2022).
- Sanguesa, J.A.; Torres-Sanz, V.; Garrido, P.; Martinez, F.J.; Marquez-Barja, J.M. A review on electric vehicles: Technologies and challenges. Smart Cities 2021, 4, 372–404. [Google Scholar] [CrossRef]
- Dunn, J.B.; Gaines, L.; Sullivan, J.; Wang, M.Q. Greenhouse gas emissions of automotive lithium-ion batteries. Environ. Sci. Technol. 2012, 46, 12704–12710. [Google Scholar] [CrossRef] [PubMed]
- Lithium Recovery from Brines. Available online: https://www.lenntech.com/processes/lithium-recovery.htm (accessed on 6 October 2022).
- Understanding Cobalt’s Human Cost. 2021. Available online: https://www.sciencedaily.com/releases/2021/12/211217113232.htm#:~:text=Waste%20generated%20from%20mining%20cobalt,were%20unsafe%2C%20unfair%20and%20stressful (accessed on 6 October 2022).
- Apple and Google Named in US Lawsuit over Congolese Child Cobalt Mining Deaths. Available online: https://www.theguardian.com/global-development/2019/dec/16/apple-and-google-named-in-us-lawsuit-over-congolese-child-cobalt-mining-deaths (accessed on 6 October 2022).
- Portuguese Community Files Legal Action against Lithium Mining Company. Available online: https://www.reuters.com/article/portugal-lithium-idUSL8N2Z33JZ (accessed on 6 October 2022).
- The Spiralling Environmental Cost of Our Lithium Battery Addiction. 2018. Available online: https://www.wired.co.uk/article/lithium-batteries-environment-impact (accessed on 6 October 2022).
- Davis, E.P. EU Faces Green Paradox over Electric Vehicles and Lithium Mining. 2018. Available online: https://dialogochino.net/en/uncategorised/eu-faces-green-paradox-over-electric-vehicles-lithium-mining/ (accessed on 6 October 2022).
- Meshram, P.; Pandey, B.D.; Mankhand, T.R. Extraction of lithium from primary and secondary sources by pre-treatment, leaching and separation: A comprehensive review. Hydrometallurgy 2014, 150, 192–208. [Google Scholar] [CrossRef]
- Abe, Y. Rare Metal Series—Current Status of Lithium Resources; JOGMEC Mineral Resources Report; Japan Oil, Gas and Metals National Corporation (JOGMEC): Chiba, Japan, 2010.
- Neupane, G.; Wendt, D.S. Assessment of mineral resources in geothermal brines in the US. In Proceedings of the 42nd Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 13–15 February 2017; Stanford University: Stanford, CA, USA, 2017. [Google Scholar]
- Neupane, G.; Wendt, D.S. Potential economic values of minerals in brines of identified hydrothermal systems in the US. Trans. Geotherm. Resour. Counc. 2017, 41, 1938–1956. [Google Scholar]
- Simmons, S.; Kirby, S.; Verplanck, P.; Kelley, K. Strategic and critical elements in produced geothermal fluids from Nevada and Utah. In Proceedings of the 43rd Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 12–14 February 2018; Stanford University: Stanford, CA, USA, 2018. [Google Scholar]
- McKibben, M.; Elders, W.A.; Raju, A.S.K. Chapter 7—Lithium and other geothermal mineral and energy resources beneath the Salton Sea. In Crisis at the Salton Sea: The Vital Role of Science; University of California Riverside Salton Sea Task Force, Environmental Dynamics and GeoEcology (EDGE) Institute, University of California, Riverside: Riverside, CA, USA, 2021. [Google Scholar]
- Zhang, Y.; Wang, L.; Sun, W.; Hu, Y.H.; Tang, H.H. Membrane technologies for Li+/Mg2+ separation from salt-lake brines and seawater: A comprehensive review. J. Ind. Eng. Chem. 2020, 81, 7–23. [Google Scholar] [CrossRef]
- Tian, L.; Ma, W.; Han, M. Adsorption behavior of Li+ onto nano-lithium ion sieve from hybrid magnesium/lithium manganese oxide. Chem. Eng. J. 2010, 156, 134–140. [Google Scholar] [CrossRef]
- Li, W.; Shi, C.; Zhou, A.; He, X.; Sun, Y.; Zhang, J. A positively charged composite nanofiltration membrane modified by EDTA for LiCl/MgCl2 separation. Sep. Purif. Technol. 2017, 186, 233–242. [Google Scholar]
- Somrani, A.; Hamzaoui, A.; Pontie, M. Study on lithium separation from salt lake brines by nanofiltration (NF) and low pressure reverse osmosis (LPRO). Desalination 2013, 317, 184–192. [Google Scholar] [CrossRef]
- Wen, X.; Ma, P.; Zhu, C.; He, Q.; Deng, X. Preliminary study on recovering lithium chloride from lithium-containing waters by nanofiltration. Sep. Purif. Technol. 2006, 49, 230–236. [Google Scholar] [CrossRef]
- Bi, Q.; Zhang, Z.; Zhao, C.; Tao, Z. Study on the recovery of lithium from high Mg2+/Li+ ratio brine by nanofiltration. Water Sci. Technol. 2014, 70, 1690–1694. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.-Y.; Cai, L.-J.; Nie, X.-Y.; Song, X.; Yu, J.-G. Separation of magnesium and lithium from brine using a Desal nanofiltration membrane. J. Water Process Eng. 2015, 7, 210–217. [Google Scholar] [CrossRef]
- Yang, G.; Shi, H.; Liu, W.; Xing, W.; Xu, N. Investigation of Mg2+/Li+ separation by nanofiltration. Chin. J. Chem. Eng. 2011, 19, 586–591. [Google Scholar] [CrossRef]
- Tesla Roadster. Available online: https://ev-database.uk/car/1167/Tesla-Roadster#:~:text=The%20battery%20of%20the%20Tesla,on%20a%20fully%20charged%20battery (accessed on 6 October 2022).
- Thompson, L.M.; Harlow, J.E.; Eldesoky, A.; Bauer, M.K.G.; Cheng, J.H.; Stone, W.S.; Taskovic, T.; McFarlane, C.R.M.; Dahn, J.R. Study of electrolyte and electrode composition changes vs time in aged Li-ion cells. J. Electrochem. Soc. 2021, 168, 020532. [Google Scholar] [CrossRef]
- Henschel, J.; Peschel, C.; Günter, F.; Reinhart, G.; Winter, M.; Nowak, S. Reaction product analysis of the most active “inactive” material in lithium-ion batteries—The electrolyte. ii: Battery operation and additive impact. Chem. Mater. 2019, 31, 9977–9983. [Google Scholar] [CrossRef]
- Geantil, P. The Importance of Industrial Battery Temperature Control. 2021. Available online: https://www.fluxpower.com/blog/the-importance-of-industrial-battery-temperature-control#:~:text=In%20general%2C%20lithium%2Dion%20batteries%20can%20be%20charged%20in%20an,high%20as%20140%C2%B0F (accessed on 6 October 2022).
- Global EV Outlook 2018. Available online: https://www.iea.org/reports/global-ev-outlook-2018 (accessed on 6 October 2022).
- NANOMYTE® Solid Electrolytes. Available online: https://www.neicorporation.com/products/batteries/solid-state-electrolyte/ (accessed on 6 October 2022).
- Nichols, D. Next Gen EV Batteries Will Deliver 500-Mile Range. Available online: https://www.greencars.com/news/next-gen-ev-batteries-will-deliver-500-mile-range (accessed on 6 October 2022).
- Chandler, D. New Electrode Design May Lead to More Powerful Batteries. MIT News. 2020. Available online: https://news.mit.edu/2020/solid-batteries-lithium-metal-electrode-0203 (accessed on 6 October 2022).
- Schweiger, H.G.; Obeidi, O.; Komesker, O.; Raschke, A.; Schiemann, M.; Zehner, C.; Gehnen, M.; Keller, M.; Birke, P. Comparison of several methods for determining the internal resistance of lithium ion cells. Sensors 2010, 10, 5604–5625. [Google Scholar] [CrossRef] [Green Version]
- Oliveri, R.L.; Insinga, M.G.; Pisana, S.; Patella, B.; Aiello, G.; Inguanta, R. High-performance lead-acid batteries enabled by Pb and PbO2: Nanostructured electrodes: Effect of operating temperature. Appl. Sci. 2021, 11, 6357. [Google Scholar] [CrossRef]
- Vashist, S.K.; Luong, J.H.T. Recent advances in electrochemical biosensing schemes using graphene and graphene-based nanocomposites. Carbon 2015, 84, 519–550. [Google Scholar] [CrossRef]
- García, F. La Española Graphenano Presenta una Batería que Dura 800 Kilómetros. 2016. Available online: https://www.elmundo.es/motor/2016/02/11/56bc7d6aca4741e31e8b461f.html (accessed on 6 October 2022).
- Anthony, S.; Extremetech. Aluminium-Air Battery Can Power Electric Vehicles for 1000 Miles will Come to Production Cars in 2017. Available online: https://www.extremetech.com/extreme/151801-aluminium-air-battery-can-power-electric-vehicles-for-1000-miles-will-come-to-production-cars-in-2017 (accessed on 6 October 2022).
- Energy Storage Inter-Platform Group. State of the Art of Energy Storage Regulations and Technology. Available online: http://www.futured.es/wp-content/uploads/2016/06/GIA-Maqueta_eng.pdf (accessed on 6 October 2022).
- Kosivi, J.; Gomez, J.; Nelson, R.; Kalu, E.E.; Weatherspoon, M.H. Non-Paste Based Composite Cathode Electrode for Lithium Air Battery; ECS Meeting Abstracts; The Electrochemical Society: Pennington, NJ, USA, 2014; p. 206. [Google Scholar]
- Brown, N.; Cleantechnica. Sodium-Air Batteries May Best Lithium-Air Batteries. 2013. Available online: https://cleantechnica.com/2013/03/20/sodium-air-batteries-may-best-lithium-air-batteries/ (accessed on 6 October 2022).
- Bi, X.; Wang, R.; Yuan, Y.; Zhang, D.; Zhang, T.; Ma, L.; Wu, T.; Shahbazian-Yassar, R.; Amine, K.; Lu, J. From sodium–oxygen to sodium–air battery: Enabled by sodium peroxide dihydrate. Nano Lett. 2020, 20, 4681–4686. [Google Scholar] [CrossRef]
- Zhao-Karger, Z.; Fichtner, M. Magnesium–Sulfur Battery: Its Beginning and Recent Progress. MRS Commun. 2017, 7, 770–784. [Google Scholar]
- Seeker. Supercharged! Battery Power for the Future. Available online: https://www.seeker.com/supercharged-battery-power-for-the-future-1766230400.html (accessed on 6 October 2022).
- Andrews, J. A Multiscale Approach to Magnesium Intercalation Batteries: Safer, Lighter, and Longer-Lasting. Available online: https://www.nasa.gov/directorates/spacetech/strg/nstrf_2017/Magnesium_Intercalation_Batteries/ (accessed on 6 October 2022).
- Edmondson, J. Will Rare-Earths be Eliminated in Electric Vehicle Motors? Available online: https://www.idtechex.com/en/research-article/will-rare-earths-be-eliminated-in-electric-vehicle-motors/21972 (accessed on 6 October 2022).
- Nzereogu, P.U.; Omah, A.D.; Ezema, F.I.; Iwuoha, E.I.; Nwanya, A.C. Anode materials for lithium-ion batteries: A review. Appl. Surf. Sci. Adv. 2022, 9, 100233. [Google Scholar] [CrossRef]
- Rangarajan, S.S.; Sunddararaj, S.P.; Sudhakar, A.V.V.; Shiva, C.K.; Subramaniam, U.; Collins, R.; Senjyu, T. Lithium-ion batteries—The crux of electric vehicles with opportunities and challenges. Clean Technol. 2022, 4, 908–930. [Google Scholar] [CrossRef]
- J1850_201510; Vehicle Architecture for Data Communications Standards—Class B Data Communications Network Interface. SAE International: Warrendale, PA, USA, 2009.
- IEC 62196-1:2014; Plugs, Socket-Outlets, Vehicle Couplers and Vehicle Inlets—Conductive Charging of Electric Vehicles—Part 1: General Requirements. IEC: Geneva, Switzerland, 2014.
- GB/T 20234.3-2015 (GB/T20234.3-2015). Available online: https://www.chinesestandard.net/PDF.aspx/GBT20234.3-2015 (accessed on 6 October 2022).
- Tesla. Tesla Official Website. 2019. Available online: https://www.tesla.com/en_eu/supercharger (accessed on 6 October 2022).
- Manshadi, S.D.; Khodayar, M.E.; Abdelghany, K.; Üster, H. Wireless charging of electric vehicles in electricity and transportation networks. IEEE Trans. Smart Grid 2018, 9, 4503–4512. [Google Scholar] [CrossRef]
- Dai, J.; Ludois, D.C. A Survey of wireless power transfer and a critical comparison of inductive and capacitive coupling for small gap applications. IEEE Trans. Power Electron. 2015, 30, 6017–6029. [Google Scholar] [CrossRef]
- Li, L.; Wang, Z.; Gao, F.; Wang, S.; Deng, J. A family of compensation topologies for capacitive power transfer converters for wireless electric vehicle charger. Appl. Energy 2020, 260, 114156. [Google Scholar] [CrossRef]
- Nohara, J.; Omori, H.; Yamamoto, A.; Kimura, N.; Morizane, T. A miniaturized single-ended wireless ev charger with new high power-factor drive and natural cooling structure. In Proceedings of the 2018 IEEE International Power Electronics and Application Conference and Exposition (PEAC), Shenzhen, China, 4–7 November 2018; pp. 1–6. [Google Scholar]
- Bhatti, A.R.; Salam, Z.; Aziz, M.J.B.A.; Yee, K.P.; Ashique, R.H. Electric vehicles charging using photovoltaic: Status and technological review. Renew. Sustain. Energy Rev. 2016, 54, 34–47. [Google Scholar] [CrossRef]
- Calise, F.; Cappiello, F.L.; Cartenì, A.; d’Accadia, M.D.; Vicidomini, M. A novel paradigm for a sustainable mobility based on electric vehicles, photovoltaic panels and electric energy storage systems: Case studies for Naples and Salerno (Italy). Renew. Sustain. Energy Rev. 2019, 111, 97–114. [Google Scholar] [CrossRef]
- Zakeri, B.; Syri, S. Electrical energy storage systems: A comparative life cycle cost analysis. Renew. Sustain. Energy Rev. 2015, 42, 569–596. [Google Scholar] [CrossRef]
- Liu, J.; Hu, C.; Kimber, A.; Wang, Z. Uses, Cost-benefit analysis, and markets of energy storage systems for electric grid applications. J. Energy Storage 2020, 32, 101731. [Google Scholar] [CrossRef]
- Elio, J.; Phelan, P.; Villalobos, R.; Milcarek, R.J. A review of energy storage technologies for demand-side management in industrial facilities. J. Clean. Prod. 2021, 307, 127322. [Google Scholar] [CrossRef]
- Wang, W.; Yuan, B.; Sun, Q.; Wennersten, R. Application of energy storage in integrated energy systems—A solution to fluctuation and uncertainty of renewable energy. J. Energy Storage 2022, 52, 104812. [Google Scholar] [CrossRef]
- Andujar, J.M.; Segura, F.; Rey, J.; Vivas, F.J. Batteries and hydrogen storage: Technical analysis and commercial revision to select the best option. Energies 2022, 15, 6196. [Google Scholar] [CrossRef]
- Chen, D.; Shao, G.-Q.; Li, B.; Zhao, G.-G.; Li, J.; Liu, J.-H.; Gao, Z.-S.; Zhang, H.-F. Synthesis, crystal structure and electrochemical properties of LiFePO4F cathode material for Li-ion batteries. Electrochim. Acta 2014, 147, 663–668. [Google Scholar] [CrossRef]
- Ramesh, T.N.; Lee, K.T.; Ellis, B.L.; Nazar, L.F. Tavorite lithium iron fluorophosphate cathode materials: Phase transition and electrochemistry of LiFePO4F–Li2FePO4F. Electrochem. Solid-State Lett. 2010, 13, A43–A47. [Google Scholar] [CrossRef] [Green Version]
- Kwade, A.; Haselrieder, W.; Leithoff, R.; Modlinger, A.; Dietrich, F.; Droeder, K. Current status and challenges for automotive battery production technologies. Nat. Energy 2018, 3, 290–300. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, R.; Wang, J.; Wang, Y. Current and future lithium-ion battery manufacturing. iScience 2021, 24, 102332. [Google Scholar] [CrossRef]
- U.S. Department of Energy. Energy Storage Grand Challenge Roadmap. 2020. Available online: https://www.energy.gov/energy-storage-grandchallenge/downloads/energy-storage-grandchallenge-roadmap (accessed on 18 October 2022).
- Gao, Y.; Jiang, J.; Zhang, C.; Zhang, W.; Ma, Z.; Jiang, Y. Lithium-ion battery aging mechanisms and life model under different charging stresses. J. Power Source 2017, 356, 103–114. [Google Scholar] [CrossRef]
- Hawkins, T.R.; Singh, B.; Majeau-Bettez, G.; Strømman, A.H. Comparative environmental life cycle assessment of conventional and electric vehicles. J. Ind. Ecol. 2012, 17, 53–64. [Google Scholar] [CrossRef]
- Saxena, S.; Le Floch, C.; MacDonald, J.; Moura, S. Quantifying EV battery end-of-life through analysis of travel needs with vehicle powertrain models. J. Power Source 2015, 282, 265–276. [Google Scholar] [CrossRef] [Green Version]
- Sanghai, B.; Sharma, D.; Baidya, K.; Raja, M. Refurbished and Repower: Second Life of Batteries from Electric Vehicles for Stationary Application; SAE Technical Paper 2019-26-0156; SAE: Warrendale, PA, USA, 2019. [Google Scholar]
- Robb, J. Making Stadiums and Arenas More Resilient and Energy Efficient, Publication No. WP701001EN/CSSC-868, Eaton xStorage Buildings White Paper. March 2018. Available online: https://www.eaton.com/content/dam/eaton/markets/buildings/resiliency/documents/xstorage-peak-shaving-whitepaper.pdf (accessed on 25 October 2022).
- Melin, H.E. Circular Opportunities in the Lithium-Ion Industry. Creation Inn: London, UK, 2017. [Google Scholar]
- Lv, W.; Wang, Z.; Cao, H.; Sun, Y.; Zhang, Y.; Sun, Z. A critical review and analysis on the recycling of spent lithium-ion batteries. ACS Sustain. Chem. Eng. 2018, 6, 1504–1521. [Google Scholar] [CrossRef]
- Xia, X.; Li, P. A review of the life cycle assessment of electric vehicles: Considering the influence of batteries. Sci. Total Environ. 2022, 814, 152870. [Google Scholar] [CrossRef]
- Jiang, C.; Wang, S.; Wu, B.; Fernandez, C.; Xiong, X.; Coffieken, J. A state-of-charge estimation method of the power lithium-ion battery in complex conditions based on adaptive square root extended Kalman filter. Energy 2021, 219, 119603. [Google Scholar] [CrossRef]
- Wang, Q.; Ping, P.; Zhao, X.; Chu, G.; Sun, J.; Chen, C. Thermal runaway caused fire and explosion of lithium ion battery. J. Power Sources 2012, 208, 210–224. [Google Scholar] [CrossRef]
- Lei, B.; Zhao, W.; Ziebert, C.; Uhlmann, N.; Rohde, M.; Seifert, H. Experimental analysis of thermal runaway in 18650 cylindrical Li-Ion cells using an accelerating rate calorimeter. Batteries 2017, 3, 14. [Google Scholar] [CrossRef] [Green Version]
- Jiang, F.; Liu, K.; Wang, Z.; Tong, X.; Guo, L. Theoretical analysis of lithium-ion battery failure characteristics under different states of charge. Fire Mater. 2018, 42, 680–686. [Google Scholar] [CrossRef]
- Crafts, C.C.; Doughty, D.H.; McBreen, J.; Roth, E.P. Advanced Technology Development Program for Lithium-Ion Batteries: Thermal Abuse Performance of 18650 Li-ion Cells. 2004. Available online: https://doi.org/10.2172/918751 (accessed on 19 October 2022).
- Henriksen, M.; Vaagsaether, K.; Lundberg, J.; Forseth, S.; Bjerketvedt, D. Explosion characteristics for Li-ion battery electrolytes at elevated temperatures. J. Hazard. Mater. 2019, 371, 1–7. [Google Scholar] [CrossRef]
- Hess, S.; Wohlfahrt-Mehrens, M.; Wachtler, M. Flammability of Li-Ion battery electrolytes: Flash point and self-extinguishing time measurements. J. Electrochem. Soc. 2015, 162, A3084–A3097. [Google Scholar] [CrossRef]
- LG Chem Develops Fire Retardant Plastic for EV Batteries. Available online: https://www.just-auto.com/news/lg-chem-develops-fire-retardant-plastic-for-ev-batteries/#:~:text=New%20material%20has%20the%20longest,in%20electric%20vehicles%20(EVs) (accessed on 20 October 2022).
- Zhu, J.; Darma, M.S.D.; Knapp, M.; Sørensen, D.R.; Heere, M.; Fang, Q.; Wang, X.; Dai, H.; Mereacre, L.; Senyshyn, A.; et al. Investigation of lithium-ion battery degradation mechanisms by combining differential voltage analysis and alternating current impedance. J. Power Sources 2020, 448, 227575. [Google Scholar] [CrossRef]
- Zhu, J.; Knapp, M.; Darma, M.S.D.; Fang, Q.; Wang, X.; Dai, H.; Wei, X.; Ehrenberg, H. An improved electro-thermal battery model complemented by current dependent parameters for vehicular low temperature application. Appl. Energy 2019, 248, 149–161. [Google Scholar] [CrossRef]
- Wang, Q.-K.; He, Y.-J.; Shen, J.-N.; Hu, X.-S.; Ma, Z.-F. State of charge-dependent polynomial equivalent circuit modeling for electrochemical impedance spectroscopy of lithium-ion batteries. IEEE Trans. Power Electron. 2018, 33, 8449–8460. [Google Scholar] [CrossRef]
- Han, L.; Jiao, X.H.; Zhang, Z. Recurrent neural network-based adaptive energy management control strategy of plug-in hybrid electric vehicles considering battery aging. Energies 2020, 13, 202. [Google Scholar] [CrossRef] [Green Version]
- Liu, K.L.; Hu, X.; Wei, Z.; Li, Y.; Jiang, Y. Modified Gaussian process regression models for cyclic capacity prediction of lithium-ion batteries. IEEE Trans. Transp. Electrif. 2019, 5, 1225–1236. [Google Scholar] [CrossRef]
- Tagade, P.; Hariharan, K.S.; Ramachandran, S.; Khandelwal, A.; Naha, A.; Kolake, S.M.; Han, S.H. Deep Gaussian process regression for lithium-ion battery health prognosis and degradation mode diagnosis. J. Power Sources 2020, 445, 227281. [Google Scholar] [CrossRef]
- Dong, G.; Yang, F.; Wei, Z.; Wei, J.; Tsui, K.-L. Data-driven battery health prognosis using adaptive Brownian motion model. IEEE Trans. Ind. Inf. 2020, 16, 4736–4746. [Google Scholar] [CrossRef]
- Liu, K.; Ashwin, T.R.; Hu, X.; Lucu, M.; Dhammika, W.W. An evaluation study of different modelling techniques for calendar ageing prediction of lithium-ion batteries. Renew. Sustain. Energy Rev. 2020, 131, 110017. [Google Scholar] [CrossRef]
- Tian, J.P.; Xiong, R.; Yu, Q.Q. Fractional-Order model-based incremental capacity analysis for degradation state recognition of lithium-ion batteries. IEEE Trans. Ind. Electron. 2019, 66, 1576–1584. [Google Scholar] [CrossRef]
- Xiong, R.; Li, L.; Li, Z.; Yu, Q.; Mu, H. An electrochemical model based degradation state identification method of lithium-ion battery for all-climate electric vehicles application. Appl. Energy 2018, 219, 264–275. [Google Scholar] [CrossRef]
- Ouyang, T.; Xu, P.; Chen, J.; Su, Z.; Huang, G.; Chen, N. A novel state of charge estimation method for lithium-ion batteries based on bias compensation. Energy 2021, 226, 120348. [Google Scholar] [CrossRef]
- Ghannoum, A.; Nieva, P. Graphite lithiation and capacity fade monitoring of lithium ion batteries using optical fibers. J. Energy Storage 2020, 28, 101233. [Google Scholar] [CrossRef]
- Park, K.Y.; Park, J.-W.; Seong, W.M.; Yoon, K.; Hwang, T.-H.; Ko, K.-H.; Han, J.H.; Yang, J.; Kang, K. Understanding capacity fading mechanism of thick electrodes for lithium-ion rechargeable batteries. J. Power Sources 2020, 468, 228369. [Google Scholar] [CrossRef]
- Li, X.Y.; Huang, Z.; Tian, J.; Tian, Y. State-of-charge estimation tolerant of battery aging based on a physics-based model and an adaptive cubature Kalman filter. Energy 2021, 220, 119767. [Google Scholar] [CrossRef]
- Guo, Y.J.; Yang, Z.; Liu, K.; Zhang, Y.; Feng, W. A compact and optimized neural network approach for battery state-of-charge estimation of energy storage system. Energy 2021, 219, 119529. [Google Scholar] [CrossRef]
- Kadiyala, P.G.; Bryson, A.E.; Schmidt, S.F. Discrete square root filtering: A survey of current techniques. IEEE Trans. Automat. Control 1971, 16, 727–735. [Google Scholar]
- Wang, S.; Takyi-Aninakwa, P.; Jin, S.; Yu, C.; Fernandez, C.; Stroe, D.-I. An improved feedforward-long short-term memory modeling method for the whole-life-cycle state of charge prediction of lithium-ion batteries considering current-voltage-temperature variation. Energy 2022, 254, 124224. [Google Scholar] [CrossRef]
- Al-Saadi, M.; Olmos, J.; Saez-de-Ibarra, A.; Van Mierlo, J.; Berecibar, M. Fast charging impact on the lithium-ion batteries’ lifetime and cost-effective battery sizing in heavy-duty electric vehicles applications. Energies 2022, 15, 1278. [Google Scholar] [CrossRef]
- Hasan, M.; Avramis, N.; Ranta, M.; Saez-De-Ibarra, A.; El Baghdadi, M.; Hegazy, O. Multi-objective energy management and charging strategy for electric bus fleets in cities using various ECO strategies. Sustainability 2021, 13, 47865. [Google Scholar] [CrossRef]
- Mathieu, R.; Briat, O.; Gyan, P.; Vinassa, J.-M. Comparison of the impact of fast charging on the cycle life of three lithium-ion cells under several parameters of charge protocol and temperatures. Appl. Energy 2020, 283, 116344. [Google Scholar] [CrossRef]
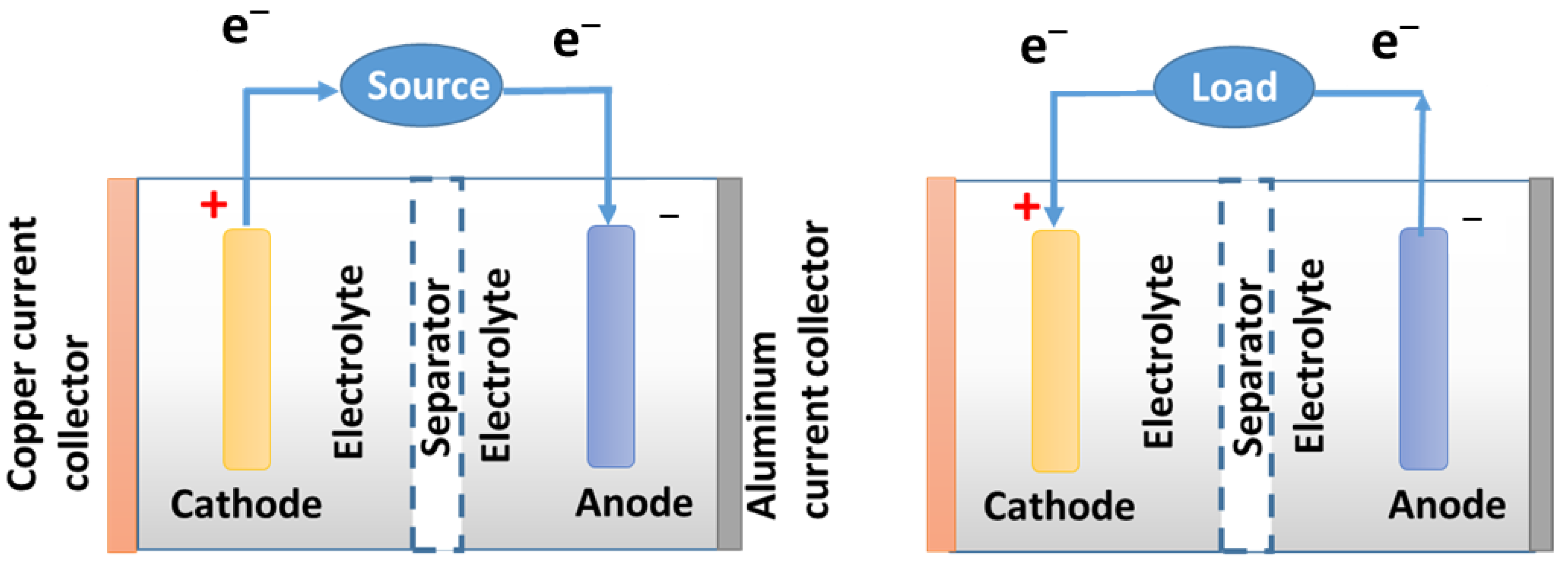
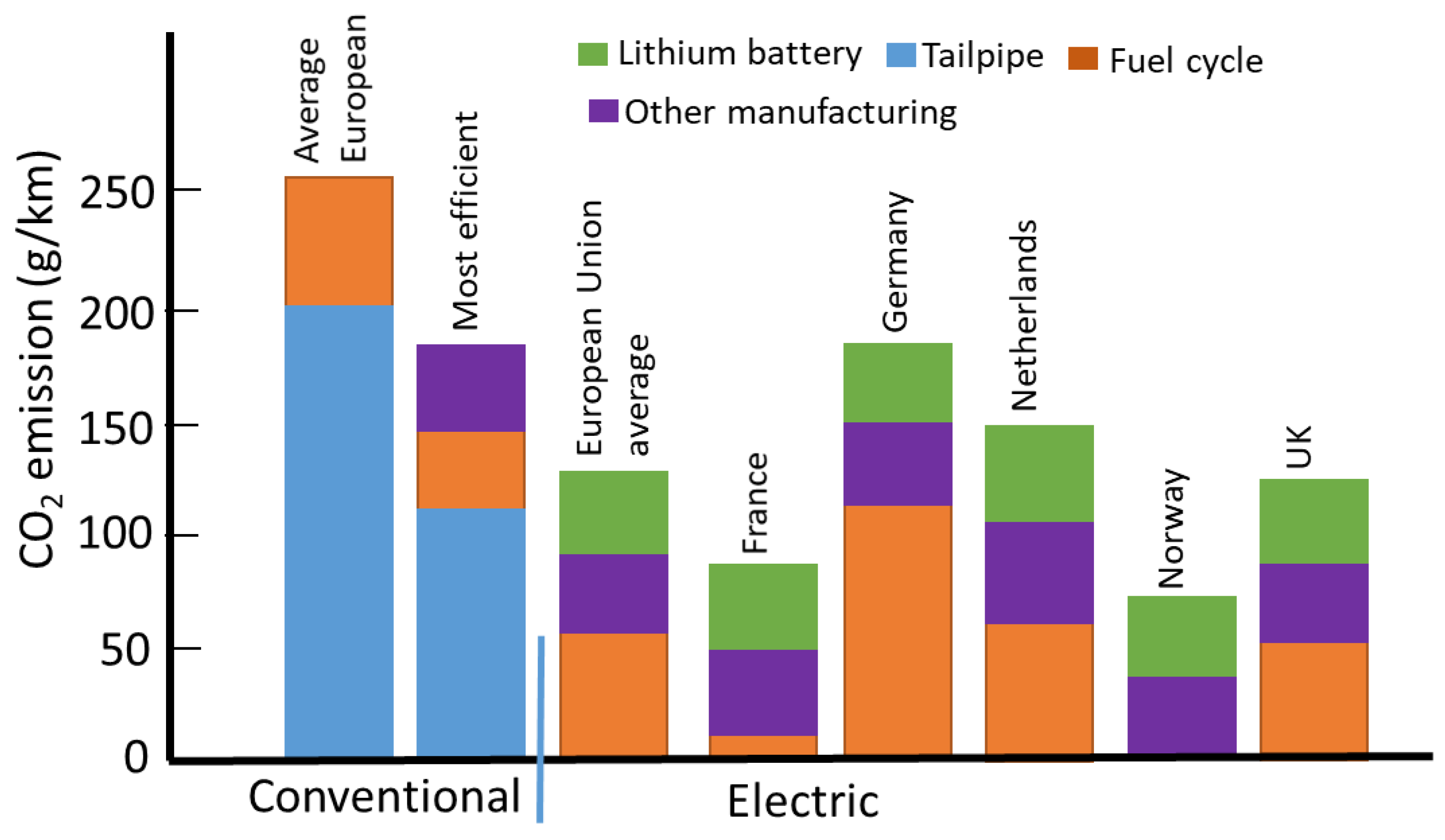

| Advantages | Disadvantages |
|---|---|
| -High reliability with few breakdowns as EVs have simple components | -Limited driving range: 200–350 km after a single full charge. -Tesla Model S range: up to 434 km (270 miles) [2] -Limited recharged points/stations (still in the development stages) |
| -No engine explosion, vibration, or fuel corrosion. -No vibration or engine noise: more comfortable. | -Lengthy charging time, 4–8 h -Fast charge, 80% in 30 min -Supercharge, 50% in 20 min and 80% in 30 min (similar to fast charge). -Not suitable for cities with a shortage of electricity. -Home fast recharge is not available for many EV drivers. |
| -Low maintenance costs and low operation costs the electricity. -Government subsidies in many countries. | -Expensive large battery package. Replacement of lithium batteries is very expensive -Heavy battery packs, up to 200 kg [3], occupy significant vehicle space. -Li-ion batteries tend to overheat and can be damaged at high voltages [1]. -Reduction of CO2 emission is only meaningful if electricity is generated from carbon-free sources. Nuclear power emits 12 g of CO2 equivalent per kWh of electricity produced, compared with 12 CO2/kWh from wind offshore, and 11 CO2/kWh from wind onshore [4]. Coal generates 820 g CO2/kWh. -Technology for the recovery of spent batteries remains to be developed. -The initial investment for an EV is significantly higher than for a conventional petrol car. -An EV battery pack can go into thermal runaway, leading to the release of toxic and flammable gases and fires start once the cell’s internal temperature rises above a certain level. |
| Topics | Ref. |
|---|---|
| Creation of EVs from the 19th century to the present | Yong et al. [11] |
| Production of EVs: Economic and environmental impacts | Richarson [12] |
| Charging methods versus the power distribution systems | Habib [13] |
| Incorporation of renewable energy into EVs | Liu et al. [14] |
| Environmental impacts of hybrid EVs and battery EVs (EVs or BEVs). | Hawkins [15] |
| A general vision of EVs: unidirectional charging versus bidirectional energy | Shuai [16] |
| Revised intelligent charging of electric vehicles | Hu et al. [17] |
| Vehicle technology: benefits and challenges | Tan et al. [18] |
| Charging infrastructure of PHEVs and BEVs | Rahman [19] |
| EV: charging and grid integration | Mahmud [20] |
| Autonomous driving versus EV charging and grid integration | Das et al. [21] |
| Data-driven battery health estimation | Li et al. [22] |
| Prediction of lithium-ion batteries aging | Liu et al. [23] |
| Advanced fault diagnosis techniques for battery | [24] |
| Lithium Chemistry and Resources | General Information and Description | Ref. |
|---|---|---|
| Lithium atom | Density = 0.53 g/cm3 and an ionic diameter of = 1.58 Å. In solution, lithium is highly soluble as lithium ions (Li+). | |
| Lithium resources | -Saline subsurface waters (continental brines) -Hydrothermally altered clays (sedimentary deposits) -Crystalline hard rock (pegmatites) -Lithium-bearing minerals are LiPO4 and complex aluminosilicates. -Potential lithium-bearing minerals are spodumene (lithium aluminosilicate), lepidolite (K(Li,Al)3(Al,Si,Rb)4O10(F,OH)2), petalite (LiAlSi4O10), and amblygonite ((Li,Na)AlPO4(F,OH)). | [36,37] |
| Lithium quantity | -World lithium reserves = 21 million tons (MTs). -World lithium resource = 86 MTs. -The US lithium resource = 7.9 MTs from continental-geothermal brines, sedimentary deposits, and pegmatites [12]. -Thacker Pass (Humboldt County, Nevada): potentially 22.4 MTs | [38,39] |
| Commercial lithium production | -Hard rock deposits in Australia and China. -Brine deposits in Argentina, Chile, and China -Brine deposits = 50 and 75% of the world’s lithium production -Brine lithium is produced mainly in South America | [38,40,41,42] |
| Lithium [57] | Cobalt [58] | Nickel [59] | Manganese [60] |
| 14 | 7.1 | 95 | 1500 |
| Chile: 8 Australia: 2.7 Argentina: 2 China: 1 Zimbabwe: 0.07 Portugal: 0.08 Brazil: 0.054 USA: 0.035 | DR Congo: 3.5 Australia: 1.4 Indonesia: 0.6 Cuba: 0.5 Philippine: 0.26 Russia: 0.22 Canada: 0.2 Madagascar: 0.1 China: 0.08 USA: 0.069 | Indonesia: 21 Australia: 21 Brazil: 16 Russia: 7.5 Philippines: 4.8 China: 2.8 Canada: 2 USA: 0.34 | South Africa: 200 Ukraine: 140 Brazil: 116 Australia: 91 India: 52 China: 43 Gabon: 22 Ghana: 12 Kazakhstan: 5 Mexico: 5 |
| Reserve Name | Country | Li+ (%) | Mg2+ (%) | Mg/Li ratio |
|---|---|---|---|---|
| Salar de Atacama | Chile | 0.157 | 0.965 | 6.15 |
| Maricunga | 0.092 | 0.74 | 0.84 | |
| Uyuni | Bolivia | 0.0321 | 0.65 | 20.2 |
| Cauchari | Argentina | 0.062 | 0.18 | 2.9 |
| Olaroz | 0.09 | 0.18 | 2 | |
| Hombre Muerto | 0.062 | 0.089 | 1.44 | |
| Rincon | 0.034 | 0.04 | 1.18 | |
| Silver Peak | USA, Esmeralda, Nevada | 0.03 | 0.04 | 1.33 |
| Salton Sea | California, Riverside | 0.022 | 0.028 | 1.27 |
| Smackover | South Arkansas | 0.038 | 0.75 | 19.7 |
| Clayton Valley | Nevada | 0.0163 | 0.019 | 1.17 |
| East Taijinar | China | 0.085 | 2.99 | 35.2 |
| West Taijinar | 0.021 | 1.28 | 64 | |
| Yiliping | 0.022 | 2 | 91 | |
| Zabuye | 0.97 | 0.001 | 0.001 | |
| Dead Sea | Israel | 0.002 | 3.4 | 1700 |
| Electrode-Structure | General Remarks and Characteristics |
|---|---|
| LCO-lithium cobalt oxide (LiCO2) | -Still widely used in portable electronics with high energy density and cycle life with good reliability. |
| LMO (LiMn2O4)-lithium manganese oxide; spinel | -MnO2 is non-toxic and earth-abundant, low cost, and has ecological appeal. |
| NMC-nickel manganese cobalt layered/spinel (LiNiMnCoC2) | -Most widely used for transportation applications with high specific energy and extra performances concerning specific power, lifetime, and safety. A typical NMC cathode powder is referred to as a 1-1-1 blend (1/3 Ni, 1/3 Mn, and 1/3 Co) -Newly developed NMC 811 has only 10% Co, 80% Ni, and 10% Mn -Its deployment is highly expected in upcoming EVs. |
| LiNiO2-layered | -Cheaper with higher energy density but less stable, compared to LiCoO2 |
| LFP-olivine (LiFePO4, lithium iron phosphate) (Li2FePO4F-olivine) | -Stable but is less dense energy than Ni-based counterparts. -Ideal for stationary applications with low cost and high safety. -A promising electrode material, tavorite-structured lithium-metal-fluorophosphate. |
| NCA, lithium nickel cobalt aluminum oxide | -Based on Ni, Co, and Al as the cathode material, e.g., LiNi0.8Co0.15 Al0.05 O2, i.e., good energy density with minimal Co. Commercial EVs and most Tesla vehicles are powered with NCA. |
| C-LiFePO4 | -Coated with carbon with 4% Li, 32% Fe, 20% PO4, 0.3% manganese, a trace of lead, and 1.5% carbon. Tesla indicated a switch to LFP-type batteries after incorporating mostly NCA types in the past. |
| LTO-Lithium titanate-oxide (Li4Ti5O12) | -Considerably lower energy density than most other lithium batteries—Fast-charging capability and long cycle life. |
| CO2 Emission (g/km) | Mitsubishi-iMiEV | Ford Focus |
|---|---|---|
| Raw material production | 163.7 | 100.9 |
| Manufacturing | 34.1 | 37.3 |
| Transportation | 2.6 | 1.4 |
| Operation | 2.2 | 253 |
| Decommissioning | 0.194 | 0.012 |
| Total | 202.8 | 392.6 |
|
| Country | Financial Packages |
|---|---|
| Belgium | EUR 4000 (purchase) and EUR 74 (for road tax instead of EUR 1900) |
| France | EUR 4000–6000 |
| Germany | EUR 4000 |
| The UK | GBP 4500 without circulation taxes if the car’s value is <GBP 40,000 |
| Spain | EUR 1300–5500 |
| USA | USD 2500 (purchase) and 417 USD/kWh from 4 kWh to a maximum of USD 7500 |
| Quebec, Canada | Up to CAD 7000, depending on the model |
| Type of Batteries | Maximal Lifetime [cycles] | Maximum Specific [W.h/kg] | Specific Power [W/kg] | Power Range [MW] |
|---|---|---|---|---|
| Li-ion | 10,000 [138] | 100–265 [139] | 150–3000 [140] | 0–100 [140] |
| Ni-Cd | 2500 [138,141] | 45–80 [141] | 150–300 [141] | 0–40 [141] |
| Lead acid | ~2500 [138] | ~50 [138] | 75–300 [140] | 0–40 [140] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luong, J.H.T.; Tran, C.; Ton-That, D. A Paradox over Electric Vehicles, Mining of Lithium for Car Batteries. Energies 2022, 15, 7997. https://doi.org/10.3390/en15217997
Luong JHT, Tran C, Ton-That D. A Paradox over Electric Vehicles, Mining of Lithium for Car Batteries. Energies. 2022; 15(21):7997. https://doi.org/10.3390/en15217997
Chicago/Turabian StyleLuong, John H. T., Cang Tran, and Di Ton-That. 2022. "A Paradox over Electric Vehicles, Mining of Lithium for Car Batteries" Energies 15, no. 21: 7997. https://doi.org/10.3390/en15217997
APA StyleLuong, J. H. T., Tran, C., & Ton-That, D. (2022). A Paradox over Electric Vehicles, Mining of Lithium for Car Batteries. Energies, 15(21), 7997. https://doi.org/10.3390/en15217997








