CO2 Mineralization Methods in Cement and Concrete Industry
Abstract
1. Introduction
1.1. Clinker Reduction Measures
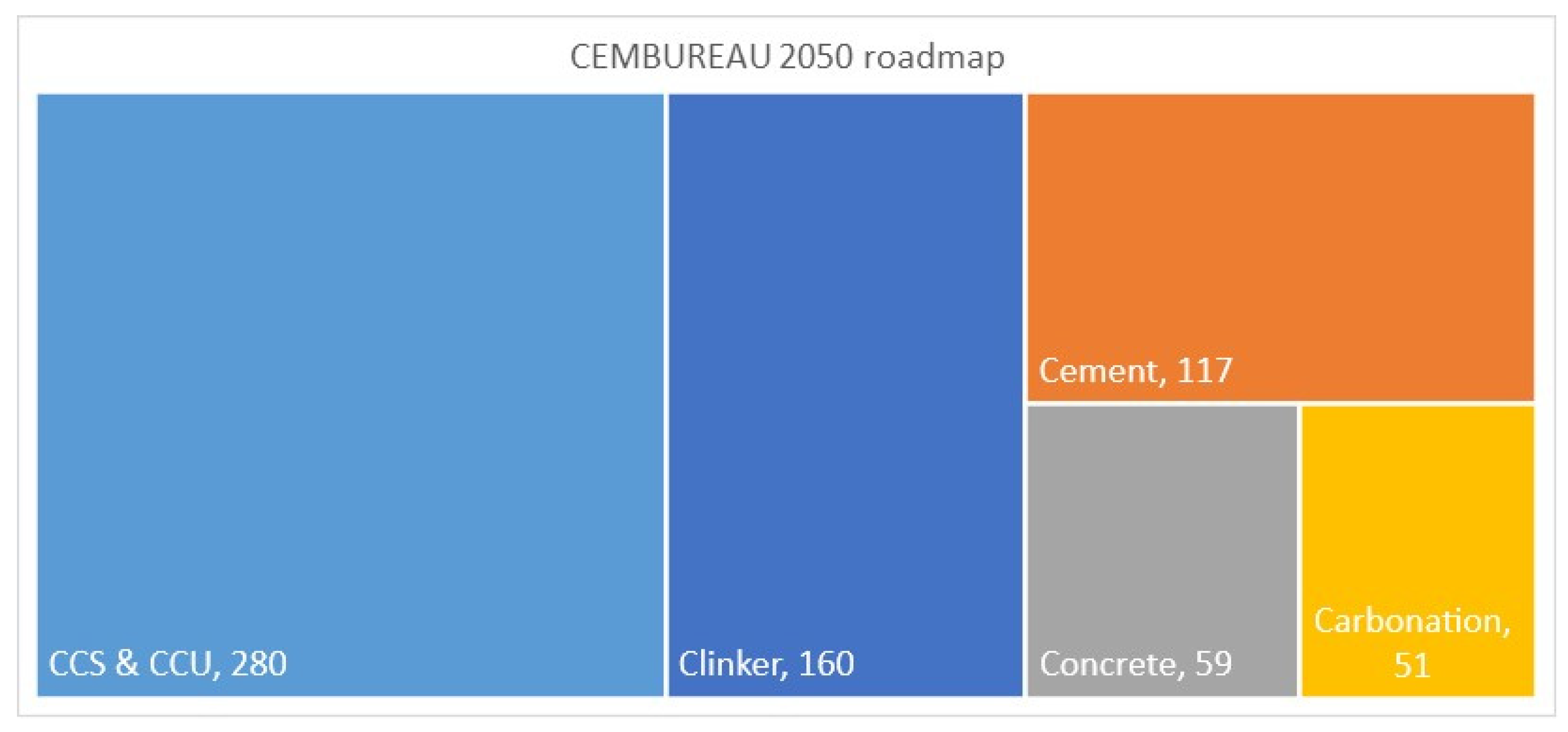
1.2. Carbon Capture and Storage, Carbon Capture and Utilization
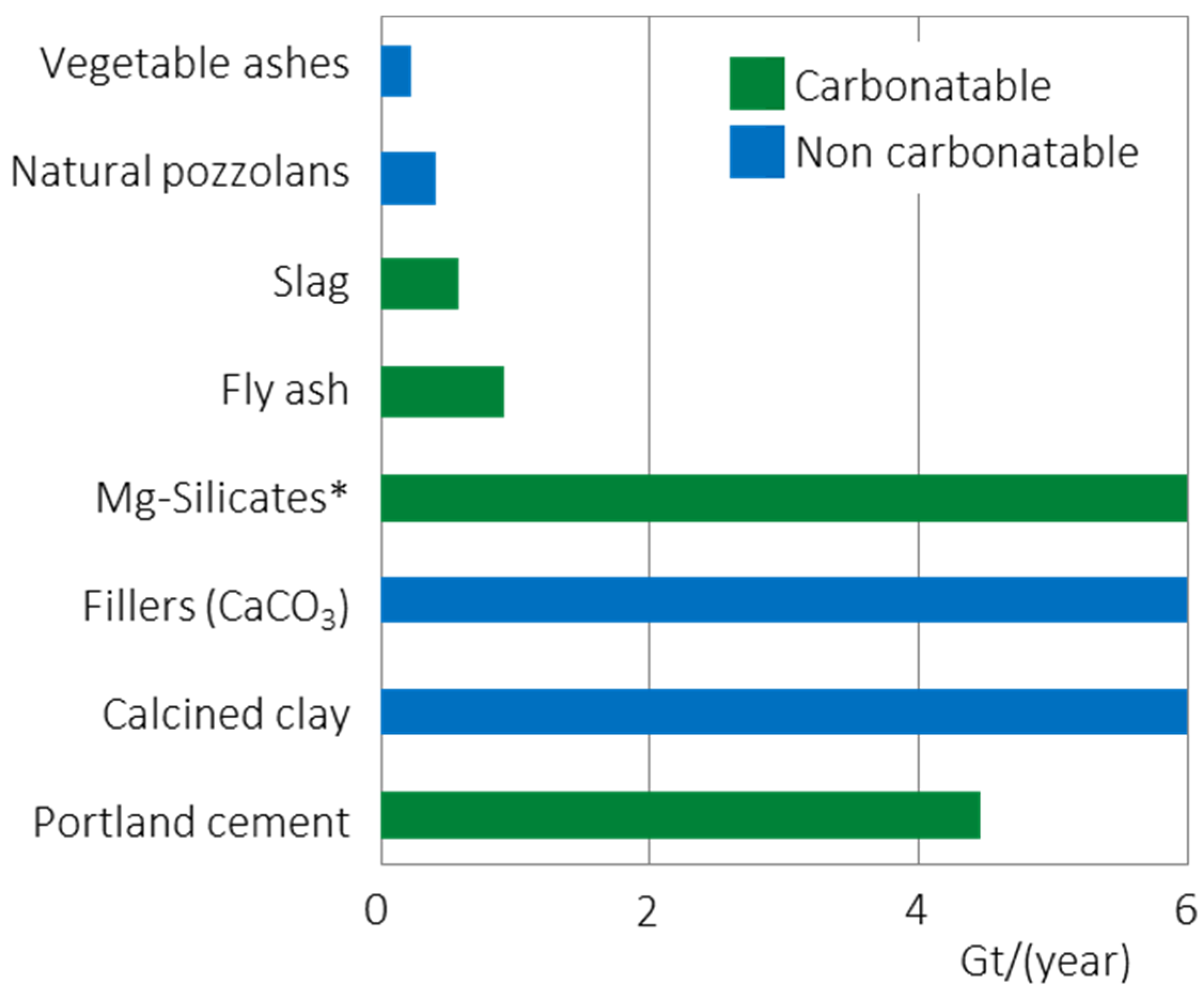
1.3. Role of CO2 Mineralization in Decarbonization of Cement Production
- direct carbonation,
- indirect carbonation.
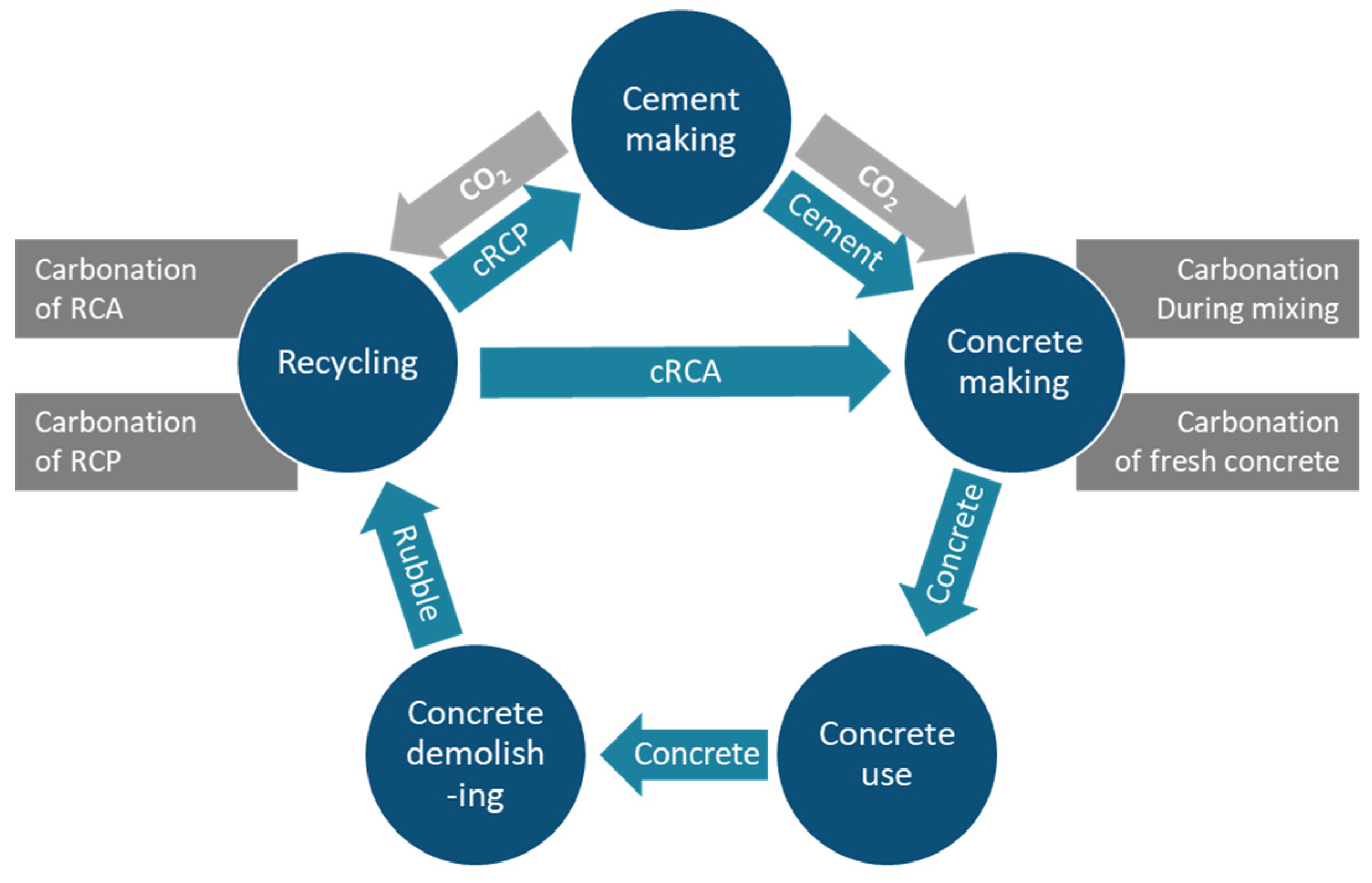
2. CO2 Mineralization within Cement and Concrete Production
2.1. Carbonation Concrete during Mixing
2.2. Carbonation of Fresh Waste Concrete
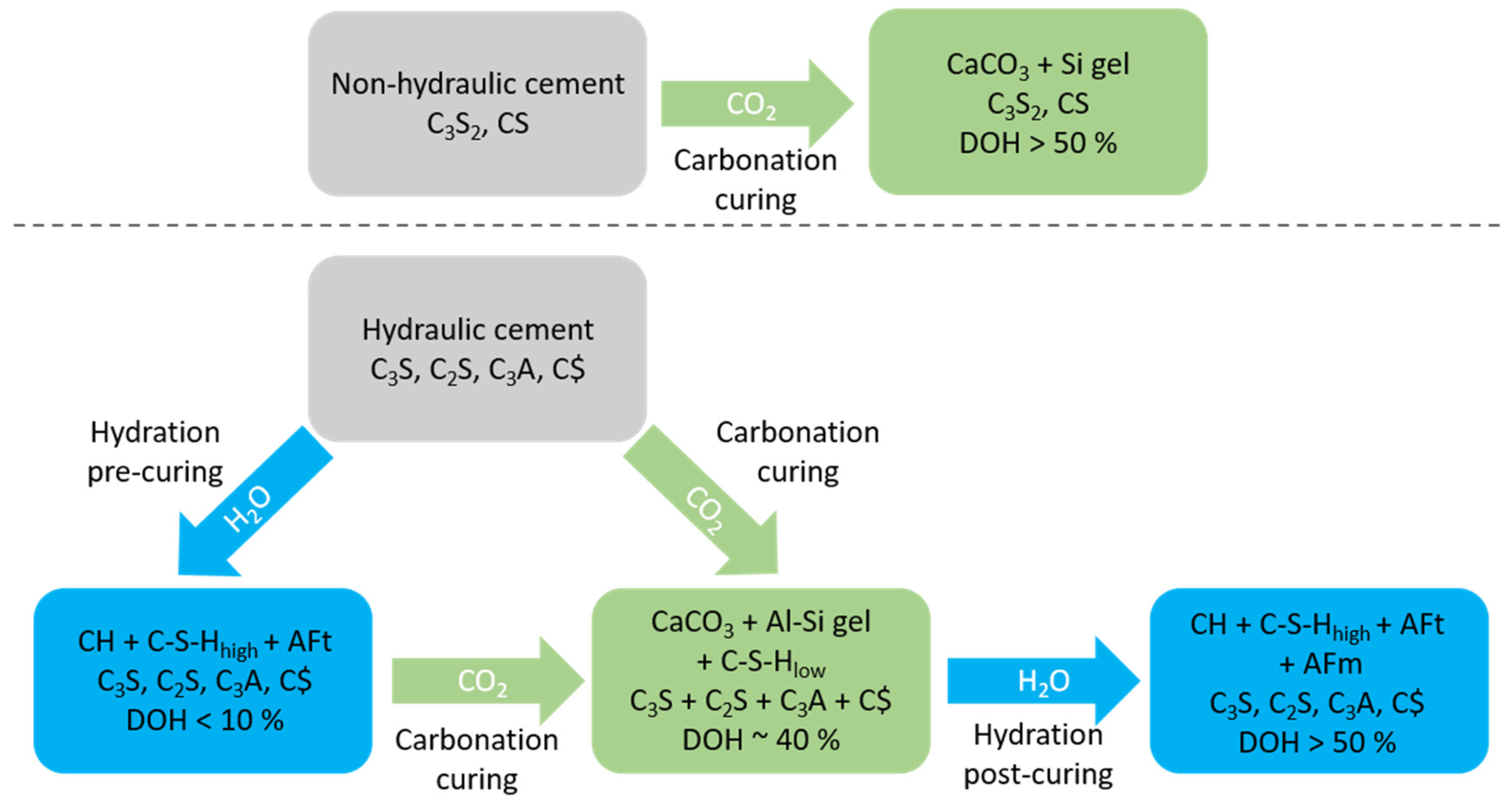
2.3. Carbonation Hardening
- Wollastonite and Rankinite type systems with Ca/Si of approximatively 1.5 and lower. These phases are not hydraulic.
- Portland clinker-based systems that are dominated by alite (C3S) with Ca/Si of approximatively 3. Alite is rapidly reacting with water.
- Concrete mixing and moulding
- Pre-curing
- Carbonation
- Post curing applies only to the hydraulic binders
2.4. Carbonation of Recycled Concrete Aggregates
2.5. Carbonation of Recycled Concrete Paste
2.5.1. Direct Carbonation Methods of Recycled Concrete Paste
- (i)
- An initial stage, which is limited by the rate of the dissolution of CO2 into the solution and precipitation of carbonation products. This stage takes place until the main peak in the evolution of pH is reached. Furthermore, the kinetics of the calcium carbonate precipitation plays an important role in the first kinetics stage [171].
- (ii)
- The second stage concerns the time after the main pH peak where the rate is limited by the dissolution kinetics of the hydrates.
2.5.2. Indirect Carbonation Methods of Recycled Concrete Paste
3. Discussion
3.1. Circular Economy and CO2 Mineralization
3.2. Deployment of CO2 Mineralization and Related Challenges
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Monteiro, P.J.M.; Miller, S.A.; Horvath, A. Towards sustainable concrete. Nat. Mater. 2017, 16, 698–699. [Google Scholar] [CrossRef] [PubMed]
- Gartner, E.M.; Macphee, D.E. A physico-chemical basis for novel cementitious binders. Cem. Concr. Res. 2011, 41, 736–749. [Google Scholar] [CrossRef]
- Scrivener, K. Options for the future of cement. Indian Concr. J. 2014, 88, 11–21. [Google Scholar]
- Barcelo, L.; Kline, J.; Walenta, G.; Gartner, E. Cement and carbon emissions. Mater. Struct. 2014, 47, 1055–1065. [Google Scholar] [CrossRef]
- Schneider, M. The cement industry on the way to a low-carbon future. Cem. Concr. Res. 2019, 124, 105792. [Google Scholar] [CrossRef]
- Habert, G.; Miller, S.A.; John, V.M.; Provis, J.L.; Favier, A.; Horvath, A.; Scrivener, K.L. Environmental impacts and decarbonization strategies in the cement and concrete industries. Nat. Rev. Earth Environ. 2020, 1, 559–573. [Google Scholar] [CrossRef]
- Klee, H.; Hunziker, R.; Van Der Meer, R.; Westaway, R. Getting the numbers right: A database of energy performance and carbon dioxide emissions for the cement industry. Greenh. Gas. Meas. Manag. 2011, 1, 109–118. [Google Scholar] [CrossRef]
- Tokheim, L.-A.; Mathisen, A.; Øi, L.E.; Jayarathna, C.K.; Eldrup, N.H.; Gautestad, T. Combined calcination and CO2 capture in cement clinker production by use of electrical energy. In TCCS–10. CO2 Capture, Transport and Storage, Trondheim, Norway, 17–19 June 2019, Proceedings of the 10th International Trondheim CCS Conference; SINTEF: Trondheim, Norway, 2019. [Google Scholar]
- Madeddu, S.; Ueckerdt, F.; Pehl, M.; Peterseim, J.; Lord, M.; Kumar, K.A.; Krüger, C.; Luderer, G. The CO2 reduction potential for the European industry via direct electrification of heat supply (power-to-heat). Environ. Res. Lett. 2020, 15, 124004. [Google Scholar] [CrossRef]
- CEMBUREAU. Cements for a Low-Carbon Europe. 2013. Available online: https://cembureau.eu/library/reports/ (accessed on 10 February 2022).
- IEA Paris. Cement. 2021. Available online: https://Www.Iea.Org/Reports/Cement (accessed on 10 February 2022).
- Scrivener, K.L.; John, V.M.; Gartner, E.M. Eco-efficient cements: Potential economically viable solutions for a low-CO2 cement-based materials industry. Cem. Concr. Res. 2018, 114, 2–26. [Google Scholar] [CrossRef]
- CEMBUREAU. Cementing the European Green Dea. Reaching Climate Neutrality along the Cement and Concrete Value Chain by 2050. 2020. Available online: https://cembureau.eu/media/kuxd32gi/cembureau-2050-roadmap_final-version_web.pdf (accessed on 10 February 2022).
- Elzinga, D.; Bennett, S.; Best, D.; Burnard, K.; Cazzola, P.; D’Ambrosio, D.; Dulac, J.; Fernandez Pales, A.; Hood, C.; LaFrance, M.; et al. Energy Technology Perspectives 2015: Mobilising Innovation to Accelerate Climate Action; International Energy Agency: Paris, France, 2015. [Google Scholar]
- McKinsey, Laying the Foundation for a Zero-Carbon Cement Industry. 2020. Available online: https://www.mckinsey.com/industries/chemicals/our-insights/laying-the-foundation-for-zero-carbon-cement (accessed on 10 February 2022).
- Global Cement and Concrete Associatio, Concrete future. The GCCA 2050 Cement and Concrete Industry Roadmap for Net Zero Concrete. 2021. Available online: https://gccassociation.org/concretefuture/wp-content/uploads/2021/10/GCCA-Concrete-Future-Roadmap.pdf (accessed on 10 February 2022).
- Rubin, E.; De Coninck, H. IPCC special report on carbon dioxide capture and storage. In TNO (2004): Cost Curves for CO2 Storage; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Kearns, D.; Liu, H.; Consoli, C. Technology Readiness and Costs of CCS; Global CCS Institute: Brussels, Belgium, 2021. [Google Scholar]
- Page, B.; Turan, G.; Zapantis, A.; Burrows, J.; Consoli, C.; Erikson, J.; Havercroft, I.; Kearns, D.; Liu, H.; Rassool, D.; et al. The Global Status of CCS 2020: Vital to Achieve Net Zero; Hydrogen Knowledge Centre: Derby, UK, 2020. [Google Scholar]
- Global Cement. Update on Carbon Capture in Cement, September 2021, (n.d.). Available online: https://www.globalcement.com/news/item/13035-update-on-carbon-capture-in-cement-september-2021 (accessed on 23 November 2021).
- Global Cement. LafargeHolcim US Reveals More Detail on Carbon Capture Study at Ste. Genevieve Cement Plant, (n.d.). Available online: https://www.globalcement.com/news/item/13220-lafargeholcim-us-reveals-more-detail-on-carbon-capture-study-at-ste-genevieve-cement-plant (accessed on 24 November 2021).
- Global Cement. Cemex Zement and Carbon Clean to Install Carbon Capture System at Rüdersdorf Cement Plant, (n.d.). Available online: https://www.globalcement.com/news/item/13204-cemex-zement-and-carbon-clean-to-install-carbon-capture-system-at-ruedersdorf-cement-plant (accessed on 24 November 2021).
- Global Cement. Holcim Deutschland to Build a Pilot CO2 Capture Unit at Höver Cement Plant, (n.d.). Available online: https://www.globalcement.com/news/item/13152-holcim-deutschland-to-build-a-pilot-co2-capture-unit-at-hoever-cement-plant (accessed on 24 November 2021).
- Global Cement. Boral to Pilot Carbon Capture and Storage at Berrima Cement Plant, (n.d.). Available online: https://www.globalcement.com/news/item/13092-boral-to-pilot-carbon-capture-and-storage-at-berrima-cement-plant (accessed on 24 November 2021).
- Hills, T.; Leeson, D.; Florin, N.; Fennell, P. Carbon Capture in the Cement Industry: Technologies, Progress, and Retrofitting. Environ. Sci. Technol. 2016, 50, 368–377. [Google Scholar] [CrossRef]
- Plaza, M.G.; Martínez, S.; Rubiera, F. CO2 Capture, Use, and Storage in the Cement Industry: State of the Art and Expectations. Energies 2020, 13, 5692. [Google Scholar] [CrossRef]
- Global Cement. Norwegian Parliament Approves Norcem’s Brevik Carbon Capture and Storage Plans (n.d.). Available online: https://www.globalcement.com/news/item/11741-norwegian-parliament-approves-norcem-s-brevik-carbon-capture-and-storage-plans (accessed on 23 February 2022).
- ZKG Cement. Aker Solutions‘ Carbon Capture and Storage Technology Gets DNV GL Approval, Aker Solutions‘ Carbon Capture and Storage Technology Gets DNV GL Approval, (n.d.). Available online: https://www.zkg.de/en/artikel/zkg_Aker_Solutions_carbon_capture_and_storage_technology_gets_DNV_GL_3552180.html (accessed on 23 February 2022).
- Gorset, O.; Knudsen, J.N.; Bade, O.M.; Askestad, I. Results from Testing of Aker Solutions Advanced Amine Solvents at CO2 Technology Centre Mongstad. Energy Procedia 2014, 63, 6267–6280. [Google Scholar] [CrossRef]
- Heidelberg Cement. Carbon Capture and Storage (CCS), (n.d.). Available online: https://www.heidelbergcement.com/en/carbon-capture-and-storage-ccs (accessed on 23 February 2022).
- Carrasco-Maldonado, F.; Spörl, R.; Fleiger, K.; Hoenig, V.; Maier, J.; Scheffknecht, G. Oxy-fuel combustion technology for cement production—State of the art research and technology development. Int. J. Greenh. Gas. Control. 2016, 45, 189–199. [Google Scholar] [CrossRef]
- LEILAC. Low Emissions Intensity Lime & Cement—LEILAC, (n.d.). Available online: https://www.project-leilac.eu/ (accessed on 21 March 2022).
- Global Cement. LEILAC-1 Study Concludes and Puts a Price Tag on Carbon Capture, (n.d.). Available online: https://www.globalcement.com/news/item/13130-leilac-1-study-concludes-and-puts-a-price-tag-on-carbon-capture (accessed on 24 November 2021).
- Project Accsess. Providing Access to Cost-Efficient, Replicable, Safe and Flexible CCUS, ACCSESS, (n.d.). Available online: https://www.projectaccsess.eu/ (accessed on 24 November 2021).
- Project Porthos. CO2 Reduction through Storage Beneath the North Sea, (n.d.). Available online: https://www.porthosco2.nl/en/ (accessed on 15 November 2021).
- Global Cement. Vicat and Hynamics to Produce Methanol from Captured CO2 at Montalieu-Vercieu Cement Plant, Vicat and Hynamics to Produce Methanol from Captured CO2 at Montalieu-Vercieu Cement Plant, (n.d.). Available online: https://www.globalcement.com/news/item/13007-vicat-and-hynamics-to-produce-methanol-from-captured-co2-at-montalieu-vercieu-cement-plant (accessed on 23 February 2022).
- Hepburn, C.; Adlen, E.; Beddington, J.; Carter, E.A.; Fuss, S.; Mac Dowell, N.; Minx, J.C.; Smith, P.; Williams, C.K. The technological and economic prospects for CO2 utilization and removal. Nature 2019, 575, 87–97. [Google Scholar] [CrossRef]
- Bobicki, E.R.; Liu, Q.; Xu, Z.; Zeng, H. Carbon capture and storage using alkaline industrial wastes. Prog. Energy Combust. Sci. 2012, 38, 302–320. [Google Scholar] [CrossRef]
- Ho, H.-J.; Iizuka, A.; Shibata, E. Carbon Capture and Utilization Technology without Carbon Dioxide Purification and Pressurization: A Review on Its Necessity and Available Technologies. Ind. Eng. Chem. Res. 2019, 58, 8941–8954. [Google Scholar] [CrossRef]
- Cole, I.S.; Corrigan, P.; Sim, S.; Birbilis, N. Corrosion of pipelines used for CO2 transport in CCS: Is it a real problem? Int. J. Greenh. Gas. Control. 2011, 5, 749–756. [Google Scholar] [CrossRef]
- Sanna, A.; Uibu, M.; Caramanna, G.; Kuusik, R.; Maroto-Valer, M.M. A review of mineral carbonation technologies to sequester CO2. Chem. Soc. Rev. 2014, 43, 8049–8080. [Google Scholar] [CrossRef]
- Kelemen, P.; Benson, S.M.; Pilorgé, H.; Psarras, P.; Wilcox, J. An Overview of the Status and Challenges of CO2 Storage in Minerals and Geological Formations. Front. Clim. 2019, 1, 9. [Google Scholar] [CrossRef]
- Ziock, H.-J.; Butt, D.P.; Lackner, K.S.; Wendt, C.H. Chapter 5–The need and options available for permanent CO2 disposal. In Reaction Engineering for Pollution Prevention; Abraham, M.A., Hesketh, R.P., Eds.; Elsevier Science: Amsterdam, The Netherlands, 2000; pp. 41–49. [Google Scholar] [CrossRef]
- Whipple, G.C.; Mayer, A. The Solubility of Calcium Carbonate and of Magnesium Hydroxide and the Precipitation of These Salts with Lime Water. J. Infect. Dis. 1906, 3, S151–S165. [Google Scholar] [CrossRef][Green Version]
- Heidelberg Cement. Breakthrough in New CCU Technology with Recycled Concrete Paste, (n.d.). Available online: https://blog.heidelbergcement.com/en/ccu-brevik-norcem-recycled-concrete (accessed on 23 February 2022).
- Helwani, Z.; Wiheeb, A.D.; Kim, J.; Othman, M.R. In-situ mineralization of carbon dioxide in a coal-fired power plant. Energy Sources Part. A Recover. Util. Environ. Eff. 2016, 38, 606–611. [Google Scholar] [CrossRef]
- Bosoaga, A.; Mašek, O.; Oakey, J.E. CO2 Capture Technologies for Cement Industry. Energy Procedia 2009, 1, 133–140. [Google Scholar] [CrossRef]
- Ali, M.; Saidur, R.; Hossain, M. A review on emission analysis in cement industries. Renew. Sustain. Energy Rev. 2011, 15, 2252–2261. [Google Scholar] [CrossRef]
- Pan, S.-Y.; Chiang, A.; Chang, E.-E.; Lin, Y.-P.; Kim, H.; Chiang, P.-C. An Innovative Approach to Integrated Carbon Mineralization and Waste Utilization: A Review. Aerosol Air Qual. Res. 2015, 15, 1072–1091. [Google Scholar] [CrossRef]
- Li, J.; Hitch, M. Mechanical activation of magnesium silicates for mineral carbonation, a review. Miner. Eng. 2018, 128, 69–83. [Google Scholar] [CrossRef]
- Gerdemann, S.J.; O’Connor, W.K.; Dahlin, D.C.; Penner, L.R.; Rush, H. Ex Situ Aqueous Mineral Carbonation. Environ. Sci. Technol. 2007, 41, 2587–2593. [Google Scholar] [CrossRef] [PubMed]
- Ostovari, H.; Müller, L.; Skocek, J.; Bardow, A. From Unavoidable CO2 Source to CO2 Sink? A Cement Industry Based on CO2 Mineralization. Environ. Sci. Technol. 2021, 55, 5212–5223. [Google Scholar] [CrossRef] [PubMed]
- Gadikota, G.; Park, A.-H.A. Accelerated Carbonation of Ca- and Mg-Bearing Minerals and Industrial Wastes Using CO2. In Carbon Dioxide Utilisation; Elsevier: Amsterdam, The Netherlands, 2015; pp. 115–137. [Google Scholar]
- Zajac, M.; Lechevallier, A.; Durdzinski, P.; Bullerjahn, F.; Skibsted, J.; Ben Haha, M. CO2 mineralisation of Portland cement: Towards understanding the mechanisms of enforced carbonation. J. CO2 Util. 2020, 38, 398–415. [Google Scholar] [CrossRef]
- Pan, S.-Y.; Chang, E.; Chiang, P.-C. CO2 Capture by Accelerated Carbonation of Alkaline Wastes: A Review on Its Principles and Applications. Aerosol Air Qual. Res. 2012, 12, 770–791. [Google Scholar] [CrossRef]
- Lothenbach, B.; Scrivener, K.; Hooton, R.D. Supplementary cementitious materials. Cem. Concr. Res. 2011, 41, 1244–1256. [Google Scholar] [CrossRef]
- Veetil, S.P.; Hitch, M. Recent developments and challenges of aqueous mineral carbonation: A review. Int. J. Environ. Sci. Technol. 2020, 17, 4359–4380. [Google Scholar] [CrossRef]
- Andrew, R. Global CO2 Emissions from Cement Production. 2019. Available online: https://essd.copernicus.org/articles/10/195/2018/ (accessed on 10 February 2022).
- Monkman, S.; Kenward, P.A.; Dipple, G.; Macdonald, M.; Raudsepp, M. Activation of cement hydration with carbon dioxide. J. Sustain. Cem.-Based Mater. 2018, 7, 160–181. [Google Scholar] [CrossRef]
- Monkman, S.; Lee, B.E.; Grandfield, K.; MacDonald, M.; Raki, L. The impacts of in-situ carbonate seeding on the early hydration of tricalcium silicate. Cem. Concr. Res. 2020, 136, 106179. [Google Scholar] [CrossRef]
- Monkman, S.; MacDonald, M.; Hooton, R.D.; Sandberg, P. Properties and durability of concrete produced using CO2 as an accelerating admixture. Cem. Concr. Compos. 2016, 74, 218–224. [Google Scholar] [CrossRef]
- Xuan, D.; Zhan, B.; Poon, C.S.; Zheng, W. Innovative reuse of concrete slurry waste from ready-mixed concrete plants in construction products. J. Hazard. Mater. 2016, 312, 65–72. [Google Scholar] [CrossRef]
- Monkman, S.; MacDonald, M.; Sutter, L. Beneficiation of concrete wash water with carbon dioxide. Mater. Struct. 2021, 54, 1–20. [Google Scholar] [CrossRef]
- Keppert, M.; Davidová, V.; Doušová, B.; Scheinherrová, L.; Reiterman, P. Recycling of fresh concrete slurry waste as supplementary cementing material: Characterization, application and leaching of selected elements. Constr. Build. Mater. 2021, 300, 124061. [Google Scholar] [CrossRef]
- Vieira, L.d.B.P.; de Figueiredo, A.D.; John, V.M. Evaluation of the use of crushed returned concrete as recycled aggregate in ready-mix concrete plant. J. Build. Eng. 2020, 31, 101408. [Google Scholar] [CrossRef]
- Vieira, L.d.B.P.; de Figueiredo, A.D.; Moriggi, T.; John, V.M. Waste generation from the production of ready-mixed concrete. Waste Manag. 2019, 94, 146–152. [Google Scholar] [CrossRef]
- Meyer, V.; Sahu, S.; Dunster, A. Properties of Solidia Cement and Concrete. In Proceedings of the 1st International Conference on Innovation in Lowcarbon Cement & Concrete Technology, London, UK, 24–26 June 2019; pp. 1–4. [Google Scholar]
- Monkman, S.; Shao, Y. Integration of carbon sequestration into curing process of precast concrete. Can. J. Civ. Eng. 2010, 37, 302–310. [Google Scholar] [CrossRef]
- Rostami, V.; Shao, Y.; Boyd, A.J. Carbonation Curing versus Steam Curing for Precast Concrete Production. J. Mater. Civ. Eng. 2012, 24, 1221–1229. [Google Scholar] [CrossRef]
- Zhang, D.; Shao, Y. Early age carbonation curing for precast reinforced concretes. Constr. Build. Mater. 2016, 113, 134–143. [Google Scholar] [CrossRef]
- Berger, R.L.; Young, J.F.; Leung, K. Acceleration of Hydration of Calcium Silicates by Carbon Dioxide Treatment. Nat. Phys. Sci. 1972, 240, 16–18. [Google Scholar] [CrossRef]
- Klemm, W.; Berger, R. Accelerated curing of cementitious systems by carbon dioxide: Part I. Portland cement. Cem. Concr. Res. 1972, 2, 567–576. [Google Scholar] [CrossRef]
- Berger, R.; Klemm, W. Accelerated curing of cementitious systems by carbon dioxide: Part II. Hydraulic calcium silicates and aluminates. Cem. Concr. Res. 1972, 2, 647–652. [Google Scholar] [CrossRef]
- Zhang, D.; Ghouleh, Z.; Shao, Y. Review on carbonation curing of cement-based materials. J. CO2 Util. 2017, 21, 119–131. [Google Scholar] [CrossRef]
- Ashraf, W. Carbonation of cement-based materials: Challenges and opportunities. Constr. Build. Mater. 2016, 120, 558–570. [Google Scholar] [CrossRef]
- Liu, Z.; Meng, W. Fundamental Understanding of Carbonation Curing and Durability of Carbonation-Cured Cement-Based Composites: A Review. J. CO2 Util. 2021, 44, 101428. [Google Scholar] [CrossRef]
- You, X.; Hu, X.; He, P.; Liu, J.; Shi, C. A review on the modelling of carbonation of hardened and fresh cement-based materials. Cem. Concr. Compos. 2022, 125, 104315. [Google Scholar] [CrossRef]
- Ashraf, W.; Olek, J. Carbonation behavior of hydraulic and non-hydraulic calcium silicates: Potential of utilizing low-lime calcium silicates in cement-based materials. J. Mater. Sci. 2016, 51, 6173–6191. [Google Scholar] [CrossRef]
- Ashraf, W.; Olek, J. Carbonation activated binders from pure calcium silicates: Reaction kinetics and performance controlling factors. Cem. Concr. Compos. 2018, 93, 85–98. [Google Scholar] [CrossRef]
- Mu, Y.; Liu, Z.; Wang, F. Comparative Study on the Carbonation-Activated Calcium Silicates as Sustainable Binders: Reactivity, Mechanical Performance, and Microstructure. ACS Sustain. Chem. Eng. 2019, 7, 7058–7070. [Google Scholar] [CrossRef]
- Mu, Y.; Liu, Z.; Wang, F.; Huang, X. Carbonation characteristics of γ-dicalcium silicate for low-carbon building material. Constr. Build. Mater. 2018, 177, 322–331. [Google Scholar] [CrossRef]
- Chang, J.; Fang, Y.; Shang, X. The role of β-C2S and γ-C2S in carbon capture and strength development. Mater. Struct. 2016, 49, 4417–4424. [Google Scholar] [CrossRef]
- Song, Q.; Guo, M.-Z.; Wang, L.; Ling, T.-C. Use of steel slag as sustainable construction materials: A review of accelerated carbonation treatment. Resour. Conserv. Recycl. 2021, 173, 105740. [Google Scholar] [CrossRef]
- Martins, A.C.P.; de Carvalho, J.M.F.; Costa, L.C.B.; Andrade, H.D.; de Melo, T.V.; Ribeiro, J.C.L.; Pedroti, L.G.; Peixoto, R.A.F. Steel slags in cement-based composites: An ultimate review on characterization, applications and performance. Constr. Build. Mater. 2021, 291, 123265. [Google Scholar] [CrossRef]
- Na, H.; Wang, Y.; Zhang, X.; Li, J.; Zeng, Y.; Liu, P. Hydration Activity and Carbonation Characteristics of Dicalcium Silicate in Steel Slag: A Review. Metals 2021, 11, 1580. [Google Scholar] [CrossRef]
- Xuan, D.; Zhan, B.; Poon, C.S.; Zheng, W. Carbon dioxide sequestration of concrete slurry waste and its valorisation in construction products. Constr. Build. Mater. 2016, 113, 664–672. [Google Scholar] [CrossRef]
- Kaliyavaradhan, S.K.; Ling, T.-C.; Mo, K.H. CO2 sequestration of fresh concrete slurry waste: Optimization of CO2 uptake and feasible use as a potential cement binder. J. CO2 Util. 2020, 42, 101330. [Google Scholar] [CrossRef]
- Khan, R.I.; Ashraf, W. Effects of ground wollastonite on cement hydration kinetics and strength development. Constr. Build. Mater. 2019, 218, 150–161. [Google Scholar] [CrossRef]
- Hou, G.; Chen, J.; Lu, B.; Chen, S.; Cui, E.; Naguib, H.M.; Guo, M.-Z.; Zhang, Q. Composition design and pilot study of an advanced energy-saving and low-carbon rankinite clinker. Cem. Concr. Res. 2020, 127, 105926. [Google Scholar] [CrossRef]
- Lu, B.; Shi, C.; Hou, G. Strength and microstructure of CO2 cured low-calcium clinker. Constr. Build. Mater. 2018, 188, 417–423. [Google Scholar] [CrossRef]
- Leemann, A.; Winnefeld, F.; Münch, B.; Läng, F. Carbonated wollastonite—An effective supplementary cementitious material? J. Microsc. 2021, 286, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Siddique, S.; Naqi, A.; Jang, J.G. Influence of water to cement ratio on CO2 uptake capacity of belite-rich cement upon exposure to carbonation curing. Cem. Concr. Compos. 2020, 111, 103616. [Google Scholar] [CrossRef]
- Wang, J.; Xu, H.; Xu, D.; Du, P.; Zhou, Z.; Yuan, L.; Cheng, X. Accelerated carbonation of hardened cement pastes: Influence of porosity. Constr. Build. Mater. 2019, 225, 159–169. [Google Scholar] [CrossRef]
- Chen, T.; Gao, X. Use of Carbonation Curing to Improve Mechanical Strength and Durability of Pervious Concrete. ACS Sustain. Chem. Eng. 2020, 8, 3872–3884. [Google Scholar] [CrossRef]
- Ellingboe, E.; Arehart, J.H.; Srubar, W. On the Theoretical CO2 Sequestration Potential of Pervious Concrete. Infrastructures 2019, 4, 12. [Google Scholar] [CrossRef]
- Chen, T.; Gao, X. Effect of carbonation curing regime on strength and microstructure of Portland cement paste. J. CO2 Util. 2019, 34, 74–86. [Google Scholar] [CrossRef]
- Shi, C.; He, F.; Wu, Y. Effect of pre-conditioning on CO2 curing of lightweight concrete blocks mixtures. Constr. Build. Mater. 2012, 26, 257–267. [Google Scholar] [CrossRef]
- Zhang, D.; Cai, X.; Jaworska, B. Effect of pre-carbonation hydration on long-term hydration of carbonation-cured cement-based materials. Constr. Build. Mater. 2020, 231, 117122. [Google Scholar] [CrossRef]
- Zhan, B.J.; Xuan, D.; Poon, C.S.; Shi, C.J. Mechanism for rapid hardening of cement pastes under coupled CO2-water curing regime. Cem. Concr. Compos. 2019, 97, 78–88. [Google Scholar] [CrossRef]
- Liu, M.; Hong, S.; Wang, Y.; Zhang, J.; Hou, D.; Dong, B. Compositions and microstructures of hardened cement paste with carbonation curing and further water curing. Constr. Build. Mater. 2021, 267, 121724. [Google Scholar] [CrossRef]
- Zajac, M.; Irbe, L.; Bullerjahn, F.; Hilbig, H.; Ben Haha, M. Mechanisms of carbonation hydration hardening in Portland cements. Cem. Concr. Res. 2022, 152, 106687. [Google Scholar] [CrossRef]
- Zhang, D.; Li, V.C.; Ellis, B.R. Optimal Pre-hydration Age for CO2 Sequestration through Portland Cement Carbonation. ACS Sustain. Chem. Eng. 2018, 6, 15976–15981. [Google Scholar] [CrossRef]
- Smigelskyte, A.; Siauciunas, R.; Hilbig, H.; Decker, M.; Urbonas, L.; Skripkiunas, G. Carbonated rankinite binder: Effect of curing parameters on microstructure, strength development and durability performance. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, L.; Xuan, D.; Shen, P.; Zhu, J.; Guan, X.; Shi, C. Enhanced carbonation reactivity of wollastonite by rapid cooling process: Towards an ultra-low calcium CO2 sequestration binder. Constr. Build. Mater. 2021, 299, 124336. [Google Scholar] [CrossRef]
- He, Z.; Wang, S.; Mahoutian, M.; Shao, Y. Flue gas carbonation of cement-based building products. J. CO2 Util. 2020, 37, 309–319. [Google Scholar] [CrossRef]
- Xian, X.; Zhang, D.; Shao, Y. Flue gas carbonation curing of cement paste and concrete at ambient pressure. J. Clean. Prod. 2021, 313, 127943. [Google Scholar] [CrossRef]
- Van Balen, K.; Van Gemert, D.; Elsen, J. Competition between Carbonation and Hydration on the Hardening of Calcium Hydroxide and Calcium Silicate Binders; WTA Publications: Karlsruhe, Germany, 2009. [Google Scholar]
- Scrivener, K.; Ouzia, A.; Juilland, P.; Mohamed, A.K. Advances in understanding cement hydration mechanisms. Cem. Concr. Res. 2019, 124, 105823. [Google Scholar] [CrossRef]
- Chen, T.; Xu, P.; Gao, X.; Wang, T.; Qin, L. New sights in early carbonation of calcium silicates: Performance, mechanism and nanostructure. Constr. Build. Mater. 2022, 314, 125622. [Google Scholar] [CrossRef]
- Juilland, P.; Gallucci, E.; Flatt, R.; Scrivener, K. Dissolution theory applied to the induction period in alite hydration. Cem. Concr. Res. 2010, 40, 831–844. [Google Scholar] [CrossRef]
- Nicoleau, L.; Nonat, A.; Perrey, D. The di- and tricalcium silicate dissolutions. Cem. Concr. Res. 2013, 47, 14–30. [Google Scholar] [CrossRef]
- Ashraf, W.; Olek, J. Elucidating the accelerated carbonation products of calcium silicates using multi-technique approach. J. CO2 Util. 2018, 23, 61–74. [Google Scholar] [CrossRef]
- Young, J.F.; Berger, R.L.; Breese, J. Accelerated Curing of Compacted Calcium Silicate Mortars on Exposure to CO2. J. Am. Ceram. Soc. 1974, 57, 394–397. [Google Scholar] [CrossRef]
- Mehdizadeh, H.; Jia, X.; Mo, K.H.; Ling, T.-C. Effect of water-to-cement ratio induced hydration on the accelerated carbonation of cement pastes. Environ. Pollut. 2021, 280, 116914. [Google Scholar] [CrossRef]
- Rostami, V.; Shao, Y.; Boyd, A.J.; He, Z. Microstructure of cement paste subject to early carbonation curing. Cem. Concr. Res. 2012, 42, 186–193. [Google Scholar] [CrossRef]
- Li, X.; Ling, T.-C. Instant CO2 curing for dry-mix pressed cement pastes: Consideration of CO2 concentrations coupled with further water curing. J. CO2 Util. 2020, 38, 348–354. [Google Scholar] [CrossRef]
- Ashraf, W.; Olek, J.; Jain, J. Microscopic features of non-hydraulic calcium silicate cement paste and mortar. Cem. Concr. Res. 2017, 100, 361–372. [Google Scholar] [CrossRef]
- Kashef-Haghighi, S.; Ghoshal, S. Physico–Chemical Processes Limiting CO2 Uptake in Concrete during Accelerated Carbonation Curing. Ind. Eng. Chem. Res. 2013, 52, 5529–5537. [Google Scholar] [CrossRef]
- Zhan, B.J.; Xuan, D.X.; Poon, C.S.; Scrivener, K.L. Multi-scale investigation on mechanical behavior and microstructural alteration of C-S-H in carbonated Alite paste. Cem. Concr. Res. 2021, 144, 106448. [Google Scholar] [CrossRef]
- Stripple, H.; Ljungkrantz, C.; Gustafsson, T.; Andersson, R. CO2 Uptake in Cement-Containing Products; IVL Swedish Environmental Research Institute: Stockholm, Sweden, 2018; Available online: https://www.ivl.se (accessed on 10 February 2022).
- Ahmed, H.; Tiznobaik, M.; Huda, S.B.; Islam, M.S.; Alam, M.S. Recycled aggregate concrete from large-scale production to sustainable field application. Constr. Build. Mater. 2020, 262, 119979. [Google Scholar] [CrossRef]
- Poon, C.S.; Chan, D. A review on the use of recycled aggregate in concrete in Hong Kong. In Proceedings of the International Conference on Sustainable Construction Materials and Technologies, Coventry, UK, 11–13 June 2007; pp. 144–155. [Google Scholar]
- Nedeljković, M.; Visser, J.; Šavija, B.; Valcke, S.; Schlangen, E. Use of fine recycled concrete aggregates in concrete: A critical review. J. Build. Eng. 2021, 38, 102196. [Google Scholar] [CrossRef]
- Safiuddin; Alengaram, U.J.; Rahman, M.; Salam, A.; Jumaat, M.Z. Use of recycled concrete aggregate in concrete: A review. J. Civ. Eng. Manag. 2013, 19, 796–810. [Google Scholar] [CrossRef]
- Dhir, R.; Paine, K.; Ho, N.; Poon, C.; Etxeberria, M.; De Brito, J.; Tam, V. Use of Recycled and Secondary Aggregates in Concrete: An Overview. In Proceedings of the International UKIERI Concrete Congress on New Developments in Concrete Construction, Delhi, India, 8–10 March 2011. [Google Scholar] [CrossRef]
- Shi, C.; Li, Y.; Zhang, J.; Li, W.; Chong, L.; Xie, Z. Performance enhancement of recycled concrete aggregate–A review. J. Clean. Prod. 2016, 112, 466–472. [Google Scholar] [CrossRef]
- Mistri, A.; Bhattacharyya, S.K.; Dhami, N.K.; Mukherjee, A.; Barai, S.V. A review on different treatment methods for enhancing the properties of recycled aggregates for sustainable construction materials. Constr. Build. Mater. 2020, 233, 117894. [Google Scholar] [CrossRef]
- Ouyang, K.; Shi, C.; Chu, H.; Guo, H.; Song, B.; Ding, Y.; Guan, X.; Zhu, J.; Zhang, H.; Wang, Y.; et al. An overview on the efficiency of different pretreatment techniques for recycled concrete aggregate. J. Clean. Prod. 2020, 263, 121264. [Google Scholar] [CrossRef]
- Bertos, M.F.; Simons, S.J.R.; Hills, C.D.; Carey, P.J. A review of accelerated carbonation technology in the treatment of cement-based materials and sequestration of CO2. J. Hazard. Mater. 2004, 112, 193–205. [Google Scholar] [CrossRef]
- Liang, C.; Pan, B.; Ma, Z.; He, Z.; Duan, Z. Utilization of CO2 curing to enhance the properties of recycled aggregate and prepared concrete: A review. Cem. Concr. Compos. 2020, 105, 103446. [Google Scholar] [CrossRef]
- Tam, V.W.; Butera, A.; Le, K.N.; Li, W. Utilising CO2 technologies for recycled aggregate concrete: A critical review. Constr. Build. Mater. 2020, 250, 118903. [Google Scholar] [CrossRef]
- Liu, K.; Xu, W.; Sun, D.; Tang, J.; Wang, A.; Chen, D. Carbonation of recycled aggregate and its effect on properties of recycled aggregate concrete: A review. Mater. Express 2021, 11, 1439–1452. [Google Scholar] [CrossRef]
- Pu, Y.; Li, L.; Wang, Q.; Shi, X.; Luan, C.; Zhang, G.; Fu, L.; El-Fatah Abomohra, A. Accelerated carbonation technology for enhanced treatment of recycled concrete aggregates: A state-of-the-art review. Constr. Build. Mater. 2021, 282, 122671. [Google Scholar] [CrossRef]
- Zhan, B.; Poon, C.S.; Liu, Q.; Kou, S.; Shi, C. Experimental study on CO2 curing for enhancement of recycled aggregate properties. Constr. Build. Mater. 2013, 67, 3–7. [Google Scholar] [CrossRef]
- Xuan, D.; Zhan, B.; Poon, C.S. Assessment of mechanical properties of concrete incorporating carbonated recycled concrete aggregates. Cem. Concr. Compos. 2016, 65, 67–74. [Google Scholar] [CrossRef]
- Lu, B.; Shi, C.; Cao, Z.; Guo, M.; Zheng, J. Effect of carbonated coarse recycled concrete aggregate on the properties and microstructure of recycled concrete. J. Clean. Prod. 2019, 233, 421–428. [Google Scholar] [CrossRef]
- Auroy, M.; Poyet, S.; Le Bescop, P.; Torrenti, J.M.; Charpentier, T.; Moskura, M.; Bourbon, X. Impact of carbonation on unsaturated water transport properties of cement-based materials. Cem. Concr. Res. 2015, 74, 44–58. [Google Scholar] [CrossRef]
- Morandeau, A.; Thiéry, M.; Dangla, P. Investigation of the carbonation mechanism of CH and C-S-H in terms of kinetics, microstructure changes and moisture properties. Cem. Concr. Res. 2014, 56, 153–170. [Google Scholar] [CrossRef]
- Justnes, H.; Skocek, J.; Østnor, T.A.; Engelsen, C.J.; Skjølsvold, O. Microstructural changes of hydrated cement blended with fly ash upon carbonation. Cem. Concr. Res. 2020, 137, 106192. [Google Scholar] [CrossRef]
- Wang, D.; Noguchi, T.; Nozaki, T. Increasing efficiency of carbon dioxide sequestration through high temperature carbonation of cement-based materials. J. Clean. Prod. 2019, 238, 117980. [Google Scholar] [CrossRef]
- Wang, D.; Noguchi, T.; Nozaki, T.; Higo, Y. Investigation of the carbonation performance of cement-based materials under high temperatures. Constr. Build. Mater. 2021, 272, 121634. [Google Scholar] [CrossRef]
- Gholizadeh-Vayghan, A.; Bellinkx, A.; Snellings, R.; Vandoren, B.; Quaghebeur, M. The effects of carbonation conditions on the physical and microstructural properties of recycled concrete coarse aggregates. Constr. Build. Mater. 2020, 257, 119486. [Google Scholar] [CrossRef]
- Zhan, M.; Pan, G.; Wang, Y.; Fu, M.; Lu, X. Effect of presoak-accelerated carbonation factors on enhancing recycled aggregate mortars. Mag. Concr. Res. 2017, 69, 838–849. [Google Scholar] [CrossRef]
- Zhan, B.J.; Xuan, D.; Poon, C.S. Enhancement of recycled aggregate properties by accelerated CO2 curing coupled with limewater soaking process. Cem. Concr. Compos. 2018, 89, 230–237. [Google Scholar] [CrossRef]
- Fang, X.; Zhan, B.; Poon, C.S. Enhancing the accelerated carbonation of recycled concrete aggregates by using reclaimed wastewater from concrete batching plants. Constr. Build. Mater. 2020, 239, 117810. [Google Scholar] [CrossRef]
- Zajac, M.; Skibsted, J.; Skocek, J.; Durdzinski, P.; Bullerjahn, F.; Ben Haha, M. Phase assemblage and microstructure of cement paste subjected to enforced, wet carbonation. Cem. Concr. Res. 2020, 130, 105990. [Google Scholar] [CrossRef]
- Liu, S.; Shen, P.; Xuan, D.; Li, L.; Sojobi, A.; Zhan, B.; Poon, C.S. A comparison of liquid-solid and gas-solid accelerated carbonation for enhancement of recycled concrete aggregate. Cem. Concr. Compos. 2021, 118, 103988. [Google Scholar] [CrossRef]
- Tang, W.; Zhan, B.; Wu, C.; Kou, S.-C. Experimental investigation and mathematical modelling of the carbon dioxide sequestration of cement pastes during pressurized CO2 curing. Constr. Build. Mater. 2021, 302, 124383. [Google Scholar] [CrossRef]
- Xuan, D.; Zhan, B.; Poon, C.S. Durability of recycled aggregate concrete prepared with carbonated recycled concrete aggregates. Cem. Concr. Compos. 2017, 84, 214–221. [Google Scholar] [CrossRef]
- Tam, V.W.; Butera, A.; Le, K. Carbon-conditioned recycled aggregate in concrete production. J. Clean. Prod. 2016, 133, 672–680. [Google Scholar] [CrossRef]
- Luo, S.; Ye, S.; Xiao, J.; Zheng, J.; Zhu, Y. Carbonated recycled coarse aggregate and uniaxial compressive stress-strain relation of recycled aggregate concrete. Constr. Build. Mater. 2018, 188, 956–965. [Google Scholar] [CrossRef]
- Fang, X.; Xuan, D.; Poon, C.S. Empirical modelling of CO2 uptake by recycled concrete aggregates under accelerated carbonation conditions. Mater. Struct. 2017, 50, 200. [Google Scholar] [CrossRef]
- Kou, S.-C.; Zhan, B.-J.; Poon, C.-S. Use of a CO2 curing step to improve the properties of concrete prepared with recycled aggregates. Cem. Concr. Compos. 2014, 45, 22–28. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, C.; Li, Y.; Pan, X.; Poon, C.S.; Xie, Z. Performance Enhancement of Recycled Concrete Aggregates through Carbonation. J. Mater. Civ. Eng. 2015, 27, 04015029. [Google Scholar] [CrossRef]
- Pu, Y.; Li, L.; Wang, Q.; Shi, X.; Fu, L.; Zhang, G.; Luan, C.; Abomohra, A.E.-F. Accelerated carbonation treatment of recycled concrete aggregates using flue gas: A comparative study towards performance improvement. J. CO2 Util. 2021, 43, 101362. [Google Scholar] [CrossRef]
- Abate, S.Y.; Song, K.-I.; Song, J.-K.; Lee, B.Y.; Kim, H.-K. Internal curing effect of raw and carbonated recycled aggregate on the properties of high-strength slag-cement mortar. Constr. Build. Mater. 2018, 165, 64–71. [Google Scholar] [CrossRef]
- Wang, D.; Xiao, J.; Duan, Z. Strategies to accelerate CO2 sequestration of cement-based materials and their application prospects. Constr. Build. Mater. 2022, 314, 125646. [Google Scholar] [CrossRef]
- Thiery, M.; Dangla, P.; Belin, P.; Habert, G.; Roussel, N. Carbonation kinetics of a bed of recycled concrete aggregates: A laboratory study on model materials. Cem. Concr. Res. 2013, 46, 50–65. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, S.; Wang, R.; Zhao, Y.; Men, C. Effects of carbonation treatment on the crushing characteristics of recycled coarse aggregates. Constr. Build. Mater. 2019, 201, 408–420. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, J.; Cao, D.; Dang, H.; Ding, B. Comparison of recycled aggregate treatment methods on the performance for recycled concrete. Constr. Build. Mater. 2020, 234, 117366. [Google Scholar] [CrossRef]
- Sojobi, A.O.; Xuan, D.; Li, L.; Liu, S.; Poon, C.S. Optimization of gas-solid carbonation conditions of recycled aggregates using a linear weighted sum method. Dev. Built Environ. 2021, 7, 100053. [Google Scholar] [CrossRef]
- Yahui, Y.; Gang, X.; Bin, T. Carbonation characteristics of cement-based materials under the uniform distribution of pore water. Constr. Build. Mater. 2021, 275, 121450. [Google Scholar] [CrossRef]
- Cui, H.; Tang, W.; Liu, W.; Dong, Z.; Xing, F. Experimental study on effects of CO2 concentrations on concrete carbonation and diffusion mechanisms. Constr. Build. Mater. 2015, 93, 522–527. [Google Scholar] [CrossRef]
- Dos Reis, G.S.; Cazacliu, B.; Artoni, R.; Torrenti, J.-M.; Hoffmann, C.S.; Lima, E.C. Coupling of attrition and accelerated carbonation for CO2 sequestration in recycled concrete aggregates. Clean. Eng. Technol. 2021, 3, 100106. [Google Scholar] [CrossRef]
- Li, L.; Xiao, J.; Xuan, D.; Poon, C.S. Effect of carbonation of modeled recycled coarse aggregate on the mechanical properties of modeled recycled aggregate concrete. Cem. Concr. Compos. 2018, 89, 169–180. [Google Scholar] [CrossRef]
- Shi, C.; Wu, Z.; Cao, Z.; Ling, T.-C.; Zheng, J. Performance of mortar prepared with recycled concrete aggregate enhanced by CO2 and pozzolan slurry. Cem. Concr. Compos. 2018, 86, 130–138. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, C.; Li, Y.; Pan, X.; Poon, C.-S.; Xie, Z. Influence of carbonated recycled concrete aggregate on properties of cement mortar. Constr. Build. Mater. 2015, 98, 1–7. [Google Scholar] [CrossRef]
- Li, L.; Poon, C.S.; Xiao, J.; Xuan, D. Effect of carbonated recycled coarse aggregate on the dynamic compressive behavior of recycled aggregate concrete. Constr. Build. Mater. 2017, 151, 52–62. [Google Scholar] [CrossRef]
- Guo, H.; Shi, C.; Guan, X.; Zhu, J.; Ding, Y.; Ling, T.-C.; Zhang, H.; Wang, Y. Durability of recycled aggregate concrete—A review. Cem. Concr. Compos. 2018, 89, 251–259. [Google Scholar] [CrossRef]
- Zajac, M.; Skocek, J.; Skibsted, J.; Ben Haha, M. CO2 mineralization of demolished concrete wastes into a supplementary cementitious material—A new CCU approach for the cement industry. RILEM Tech. Lett. 2021, 6, 53–60. [Google Scholar] [CrossRef]
- Zajac, M.; Skibsted, J.; Durdzinski, P.; Bullerjahn, F.; Skocek, J.; Ben Haha, M. Kinetics of enforced carbonation of cement paste. Cem. Concr. Res. 2020, 131, 106013. [Google Scholar] [CrossRef]
- Ben Ghacham, A.; Cecchi, E.; Pasquier, L.-C.; Blais, J.-F.; Mercier, G. CO2 sequestration using waste concrete and anorthosite tailings by direct mineral carbonation in gas–solid–liquid and gas–solid routes. J. Environ. Manag. 2015, 163, 70–77. [Google Scholar] [CrossRef]
- Kong, Y.; Song, Y.; Kurumisawa, K.; Wang, T.; Yan, D.; Zeng, Q.; Zhou, X.; Ruan, S. Use of hydrated cement pastes (HCP) as a CO2 sponge. J. CO2 Util. 2022, 55, 101804. [Google Scholar] [CrossRef]
- Wang, Y.; Lu, B.; Hu, X.; Liu, J.; Zhang, Z.; Pan, X.; Xie, Z.; Chang, J.; Zhang, T.; Nehdi, M.L.; et al. Effect of CO2 surface treatment on penetrability and microstructure of cement-fly ash–slag ternary concrete. Cem. Concr. Compos. 2021, 123, 104194. [Google Scholar] [CrossRef]
- Iizuka, A.; Fujii, M.; Yamasaki, A.; Yanagisawa, Y. A Novel Reduction Process of CO2 Fixation by Waste Concrete Treatment. Kagaku Kogaku Ronbunshu 2002, 28, 587–592. [Google Scholar] [CrossRef]
- Katsuyama, Y.; Yamasaki, A.; Iizuka, A.; Fujii, M.; Kumagai, K.; Yanagisawa, Y. Development of a process for producing high-purity calcium carbonate (CaCO3) from waste cement using pressurized CO2. Environ. Prog. 2005, 24, 162–170. [Google Scholar] [CrossRef]
- Iizuka, A.; Fujii, M.; Yamasaki, A.; Yanagisawa, Y. Development of a New CO2 Sequestration Process Utilizing the Carbonation of Waste Cement. Ind. Eng. Chem. Res. 2004, 43, 7880–7887. [Google Scholar] [CrossRef]
- Ho, H.-J.; Iizuka, A.; Shibata, E.; Tomita, H.; Takano, K.; Endo, T. CO2 Utilization via Direct Aqueous Carbonation of Synthesized Concrete Fines under Atmospheric Pressure. ACS Omega 2020, 5, 15877–15890. [Google Scholar] [CrossRef]
- Ho, H.-J.; Iizuka, A.; Shibata, E.; Tomita, H.; Takano, K.; Endo, T. Utilization of CO2 in direct aqueous carbonation of concrete fines generated from aggregate recycling: Influences of the solid–liquid ratio and CO2 concentration. J. Clean. Prod. 2021, 312, 127832. [Google Scholar] [CrossRef]
- Zajac, M.; Skibsted, J.; Ben Haha, M. Effect of alkalis on enforced carbonation of cement paste: Mechanism of reaction. J. Am. Ceram. Soc. 2021, 104, 1076–1087. [Google Scholar] [CrossRef]
- Zajac, M.; Skibsted, J.; Durdzinski, P.; Ben Haha, M. Effect of alkalis on products of enforced carbonation of cement paste. Constr. Build. Mater. 2021, 291, 123203. [Google Scholar] [CrossRef]
- Nedunuri, A.S.S.S.; Mohammed, A.Y.; Muhammad, S. Carbonation potential of concrete debris fines and its valorisation through mineral carbonation. Constr. Build. Mater. 2021, 310, 125162. [Google Scholar] [CrossRef]
- Abe, M.; Tanaka, S.; Noguchi, M.; Yamasaki, A. Investigation of Mineral Carbonation with Direct Bubbling into Concrete Sludge. ACS Omega 2021, 6, 15564–15571. [Google Scholar] [CrossRef] [PubMed]
- Zajac, M.; Skibsted, J.; Lothenbach, B.; Bullerjahn, F.; Skocek, J.; Ben Haha, M. Effect of sulfate on CO2 binding efficiency of recycled alkaline materials. Cem. Concr. Res. 2022, 157, 106804. [Google Scholar] [CrossRef]
- Pan, S.-Y.; Ling, T.-C.; Park, A.-H.A.; Chiang, P.-C. An Overview: Reaction Mechanisms and Modelling of CO2 Utilization via Mineralization. Aerosol Air Qual. Res. 2018, 18, 829–848. [Google Scholar] [CrossRef]
- Yoo, J.; Shin, H.; Ji, S. An Eco-Friendly Neutralization Process by Carbon Mineralization for Ca-Rich Alkaline Wastewater Generated from Concrete Sludge. Metals 2017, 7, 371. [Google Scholar] [CrossRef]
- Oelkers, E.H.; Declercq, J.; Saldi, G.D.; Gislason, S.R.; Schott, J. Olivine dissolution rates: A critical review. Chem. Geol. 2018, 500, 1–19. [Google Scholar] [CrossRef]
- Wang, F.; Dreisinger, D.; Jarvis, M.; Hitchins, T. Kinetic evaluation of mineral carbonation of natural silicate samples. Chem. Eng. J. 2021, 404, 126522. [Google Scholar] [CrossRef]
- Ben Ghacham, A.; Pasquier, L.-C.; Cecchi, E.; Blais, J.-F.; Mercier, G. Valorization of waste concrete through CO2 mineral carbonation: Optimizing parameters and improving reactivity using concrete separation. J. Clean. Prod. 2017, 166, 869–878. [Google Scholar] [CrossRef]
- Pasquier, L.-C.; Kemache, N.; Mocellin, J.; Blais, J.-F.; Mercier, G. Waste Concrete Valorization; Aggregates and Mineral Carbonation Feedstock Production. Geosciences 2018, 8, 342. [Google Scholar] [CrossRef]
- Auroy, M.; Poyet, S.; Le Bescop, P.; Torrenti, J.M.; Charpentier, T.; Moskura, M.; Bourbon, X. Comparison between natural and accelerated carbonation (3% CO2): Impact on mineralogy, microstructure, water retention and cracking. Cem. Concr. Res. 2018, 109, 64–80. [Google Scholar] [CrossRef]
- Shah, V.; Parashar, A.; Bishnoi, S. Changes in pore structure properties of cement paste and concrete on carbonation. Adv. Constr. Mater. Syst. 2018, 109, 184–197. [Google Scholar] [CrossRef]
- Li, Y.; Liu, W.; Xing, F.; Wang, S.; Tang, L.; Lin, S.; Dong, Z. Carbonation of the synthetic calcium silicate hydrate (C-S-H) under different concentrations of CO2: Chemical phases analysis and kinetics. J. CO2 Util. 2020, 35, 303–313. [Google Scholar] [CrossRef]
- Sevelsted, T.F.; Skibsted, J. Carbonation of C–S–H and C–A–S–H samples studied by 13 C, 27 Al and 29 Si MAS NMR spectroscopy. Cem. Concr. Res. 2015, 71, 56–65. [Google Scholar] [CrossRef]
- C2inCO2—Calcium Carbonatisierung Zur Industriellen Nutzung von CO2, C2inCO2—Calcium Carbonatisierung Zur Industriellen Nutzung von CO2. (n.d.). Available online: https://co2-utilization.net/de/projekte/co2-mineralisation/c2inco2/ (accessed on 23 February 2022).
- Lu, B.; Shi, C.; Zhang, J.; Wang, J. Effects of carbonated hardened cement paste powder on hydration and microstructure of Portland cement. Constr. Build. Mater. 2018, 186, 699–708. [Google Scholar] [CrossRef]
- Zajac, M.; Skocek, J.; Durdzinski, P.; Bullerjahn, F.; Skibsted, J.; Ben Haha, M. Effect of carbonated cement paste on composite cement hydration and performance. Cem. Concr. Res. 2020, 134, 106090. [Google Scholar] [CrossRef]
- Kong, Y.; Song, Y.; Weng, Y.; Kurumisawa, K.; Yan, D.; Zhou, X.; Wang, S.; Ruan, S. Influences of CO2-cured cement powders on hydration of cement paste. Greenh. Gases Sci. Technol. 2022, 12, 249–262. [Google Scholar] [CrossRef]
- Mehdizadeh, H.; Ling, T.-C.; Cheng, X.; Pan, S.-Y.; Mo, K.H. CO2 Treatment of Hydrated Cement Powder: Characterization and Application Consideration. J. Mater. Civ. Eng. 2021, 33, 04021041. [Google Scholar] [CrossRef]
- Ho, H.-J.; Iizuka, A.; Shibata, E. Chemical recycling and use of various types of concrete waste: A review. J. Clean. Prod. 2021, 284, 124785. [Google Scholar] [CrossRef]
- Iizuka, A.; Honma, M.; Hayakawa, Y.; Yamasaki, A.; Yanagisawa, Y. Aqueous Mineral Carbonation Process via Concrete Sludge. Kagaku Kogaku Ronbunshu 2012, 38, 129–134. [Google Scholar] [CrossRef]
- Iizuka, A.; Sakai, Y.; Yamasaki, A.; Honma, M.; Hayakawa, Y.; Yanagisawa, Y. Bench-Scale Operation of a Concrete Sludge Recycling Plant. Ind. Eng. Chem. Res. 2012, 51, 6099–6104. [Google Scholar] [CrossRef]
- Iizuka, A.; Sasaki, T.; Honma, M.; Yoshida, H.; Hayakawa, Y.; Yanagisawa, Y.; Yamasaki, A. Pilot-Scale Operation of a Concrete Sludge Recycling Plant and Simultaneous Production of Calcium Carbonate. Chem. Eng. Commun. 2017, 204, 79–85. [Google Scholar] [CrossRef]
- Shuto, D.; Nagasawa, H.; Iizuka, A.; Yamasaki, A. A CO2fixation process with waste cement powder via regeneration of alkali and acid by electrodialysis. RSC Adv. 2014, 4, 19778–19788. [Google Scholar] [CrossRef]
- Shuto, D.; Igarashi, K.; Nagasawa, H.; Iizuka, A.; Inoue, M.; Noguchi, M.; Yamasaki, A. CO2 Fixation Process with Waste Cement Powder via Regeneration of Alkali and Acid by Electrodialysis: Effect of Operation Conditions. Ind. Eng. Chem. Res. 2015, 54, 6569–6577. [Google Scholar] [CrossRef]
- Mun, M.; Cho, H.; Kwon, J.; Kim, K.; Kim, R. Investigation of the CO2 Sequestration by Indirect Aqueous Carbonation of Waste Cement. Am. J. Clim. Chang. 2017, 6, 132–150. [Google Scholar] [CrossRef]
- Vanderzee, S.; Zeman, F. Recovery and carbonation of 100% of calcium in waste concrete fines: Experimental results. J. Clean. Prod. 2018, 174, 718–727. [Google Scholar] [CrossRef]
- Rim, G.; Roy, N.; Zhao, D.; Kawashima, S.; Stallworth, P.; Greenbaum, S.G.; Park, A.-H.A. CO2 utilization in built environment via the P CO2 swing carbonation of alkaline solid wastes with different mineralogy. Faraday Discuss. 2021, 230, 187–212. [Google Scholar] [CrossRef]
- Lee, M.-G.; Kang, D.; Jo, H.; Park, J. Carbon dioxide utilization with carbonation using industrial waste-desulfurization gypsum and waste concrete. J. Mater. Cycles Waste Manag. 2016, 18, 407–412. [Google Scholar] [CrossRef]
- Iizuka, A.; Yoshida, H.; Hayakawa, Y. High-Performance Phosphorus Adsorbent Based on Concrete Sludge. In Phosphorus Recovery and Recycling; Springer: Berlin/Heidelberg, Germany, 2018; pp. 449–456. [Google Scholar] [CrossRef]
- Fang, X.; Xuan, D.; Zhan, B.; Li, W.; Poon, C.S. A novel upcycling technique of recycled cement paste powder by a two-step carbonation process. J. Clean. Prod. 2021, 290, 125192. [Google Scholar] [CrossRef]
- Fang, X.; Xuan, D.; Zhan, B.; Li, W.; Poon, C.S. Characterization and optimization of a two-step carbonation process for valorization of recycled cement paste fine powder. Constr. Build. Mater. 2021, 278, 122343. [Google Scholar] [CrossRef]
- Benhelal, E.; Rashid, M.; Rayson, M.; Oliver, T.; Brent, G.; Stockenhuber, M.; Kennedy, E. “ACEME”: Synthesis and characterization of reactive silica residues from two stage mineral carbonation Process. Environ. Prog. Sustain. Energy 2019, 38, e13066. [Google Scholar] [CrossRef]
- Benhelal, E.; Rashid, M.I.; Rayson, M.S.; Prigge, J.-D.; Molloy, S.; Brent, G.F.; Cote, A.; Stockenhuber, M.; Kennedy, E.M. Study on mineral carbonation of heat activated lizardite at pilot and laboratory scale. J. CO2 Util. 2018, 26, 230–238. [Google Scholar] [CrossRef]
- Benhelal, E.; Rashid, M.I.; Holt, C.; Rayson, M.S.; Brent, G.; Hook, J.M.; Stockenhuber, M.; Kennedy, E.M. The utilisation of feed and byproducts of mineral carbonation processes as pozzolanic cement replacements. J. Clean. Prod. 2018, 186, 499–513. [Google Scholar] [CrossRef]
- Sáez, P.V.; Osmani, M. A diagnosis of construction and demolition waste generation and recovery practice in the European Union. J. Clean. Prod. 2019, 241, 118400. [Google Scholar] [CrossRef]
- Ruiz, L.A.L.; Ramón, X.R.; Domingo, S.G. The circular economy in the construction and demolition waste sector—A review and an integrative model approach. J. Clean. Prod. 2020, 248, 119238. [Google Scholar] [CrossRef]
- Zhang, J.; Gu, F.; Zhang, Y. Use of building-related construction and demolition wastes in highway embankment: Laboratory and field evaluations. J. Clean. Prod. 2019, 230, 1051–1060. [Google Scholar] [CrossRef]
- Zheng, L.; Wu, H.; Zhang, H.; Duan, H.; Wang, J.; Jiang, W.; Dong, B.; Liu, G.; Zuo, J.; Song, Q. Characterizing the generation and flows of construction and demolition waste in China. Constr. Build. Mater. 2017, 136, 405–413. [Google Scholar] [CrossRef]
- Ghisellini, P.; Ji, X.; Liu, G.; Ulgiati, S. Evaluating the transition towards cleaner production in the construction and demolition sector of China: A review. J. Clean. Prod. 2018, 195, 418–434. [Google Scholar] [CrossRef]
- Tam, V.W.Y.; Soomro, M.; Evangelista, A.C.J. A review of recycled aggregate in concrete applications (2000–2017). Constr. Build. Mater. 2018, 172, 272–292. [Google Scholar] [CrossRef]
- Noguchi, T.; Kitagaki, R.; Tsujino, M. Minimizing environmental impact and maximizing performance in concrete recycling. Struct. Concr. 2011, 12, 36–46. [Google Scholar] [CrossRef]
- Gebremariam, A.T.; Di Maio, F.; Vahidi, A.; Rem, P. Innovative technologies for recycling End-of-Life concrete waste in the built environment. Resour. Conserv. Recycl. 2020, 163, 104911. [Google Scholar] [CrossRef]
- Ostovari, H.; Sternberg, A.; Bardow, A. Rock ‘n’ use of CO2: Carbon footprint of carbon capture and utilization by mineralization. Sustain. Energy Fuels 2020, 4, 4482–4496. [Google Scholar] [CrossRef]
- Kamyab, H.K.; Nielsen, P.; Van Mierloo, P.; Horckmans, L. Carbstone Pavers: A Sustainable Solution for the Urban Environment. Appl. Sci. 2021, 11, 6418. [Google Scholar] [CrossRef]



| Carbonation Method | Direct | In Direct | ||||
|---|---|---|---|---|---|---|
| Starting material | Ca source | Recycled Concrete paste | Recycled aggregates | Fresh concrete products | Concrete during mixing | Recycled Concrete paste |
| CO2 concentration | 5–100% | 5–100% | 5–100% | ~100% | 5–100% | |
| Process | Aqueous or dry | Aqueous or dry | Dry | Aqueous | Aqueous | |
| Conditions | Possible at normal temperature and pressure | |||||
| Sequestration potential (% process emissions) | 30–95% | ~30% | 30–90% | Few % | 40–100% | |
| Products | SCM | Improved aggregates | Hardened concrete | Fresh concrete | Added value products | |
| TRL | 4 | 8 | 8 | 9 | 4–9 | |
| Challenge | Technology not yet available Logistics | Low efficiency CO2 infrastructure Low value of products | Reinforcement Corrosion Missing experience | Limited sequestration CO2 infrastructure | Cost | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zajac, M.; Skocek, J.; Ben Haha, M.; Deja, J. CO2 Mineralization Methods in Cement and Concrete Industry. Energies 2022, 15, 3597. https://doi.org/10.3390/en15103597
Zajac M, Skocek J, Ben Haha M, Deja J. CO2 Mineralization Methods in Cement and Concrete Industry. Energies. 2022; 15(10):3597. https://doi.org/10.3390/en15103597
Chicago/Turabian StyleZajac, Maciej, Jan Skocek, Mohsen Ben Haha, and Jan Deja. 2022. "CO2 Mineralization Methods in Cement and Concrete Industry" Energies 15, no. 10: 3597. https://doi.org/10.3390/en15103597
APA StyleZajac, M., Skocek, J., Ben Haha, M., & Deja, J. (2022). CO2 Mineralization Methods in Cement and Concrete Industry. Energies, 15(10), 3597. https://doi.org/10.3390/en15103597







