Experimental and Numerical Study on the Sooting Behaviors of Furanic Biofuels in Laminar Counterflow Diffusion Flames
Abstract
:1. Introduction
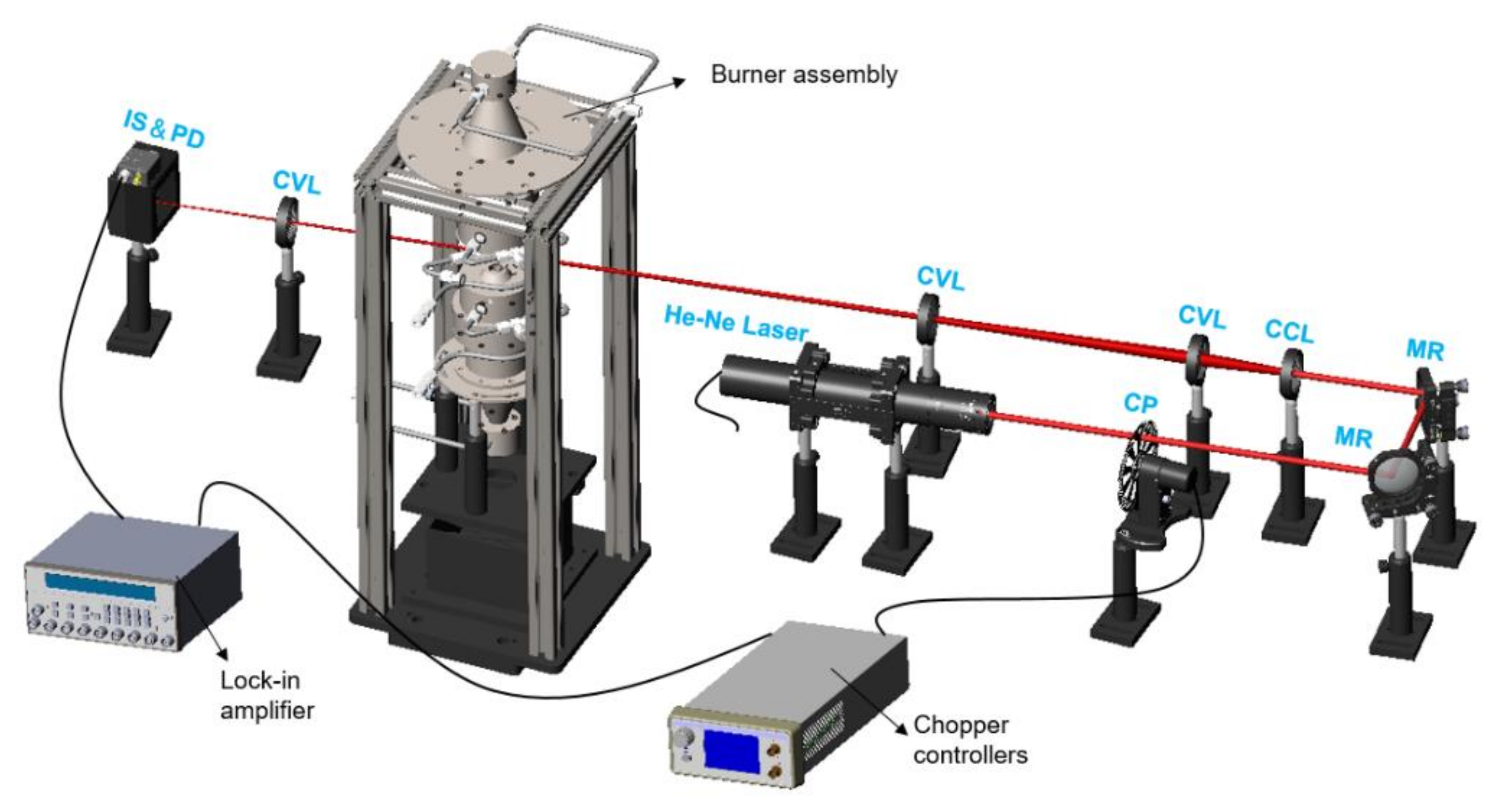
2. Experiments
3. Numerical Simulation
4. Results and Discussion
4.1. General Flame Structure of the Present Counterflow Flames
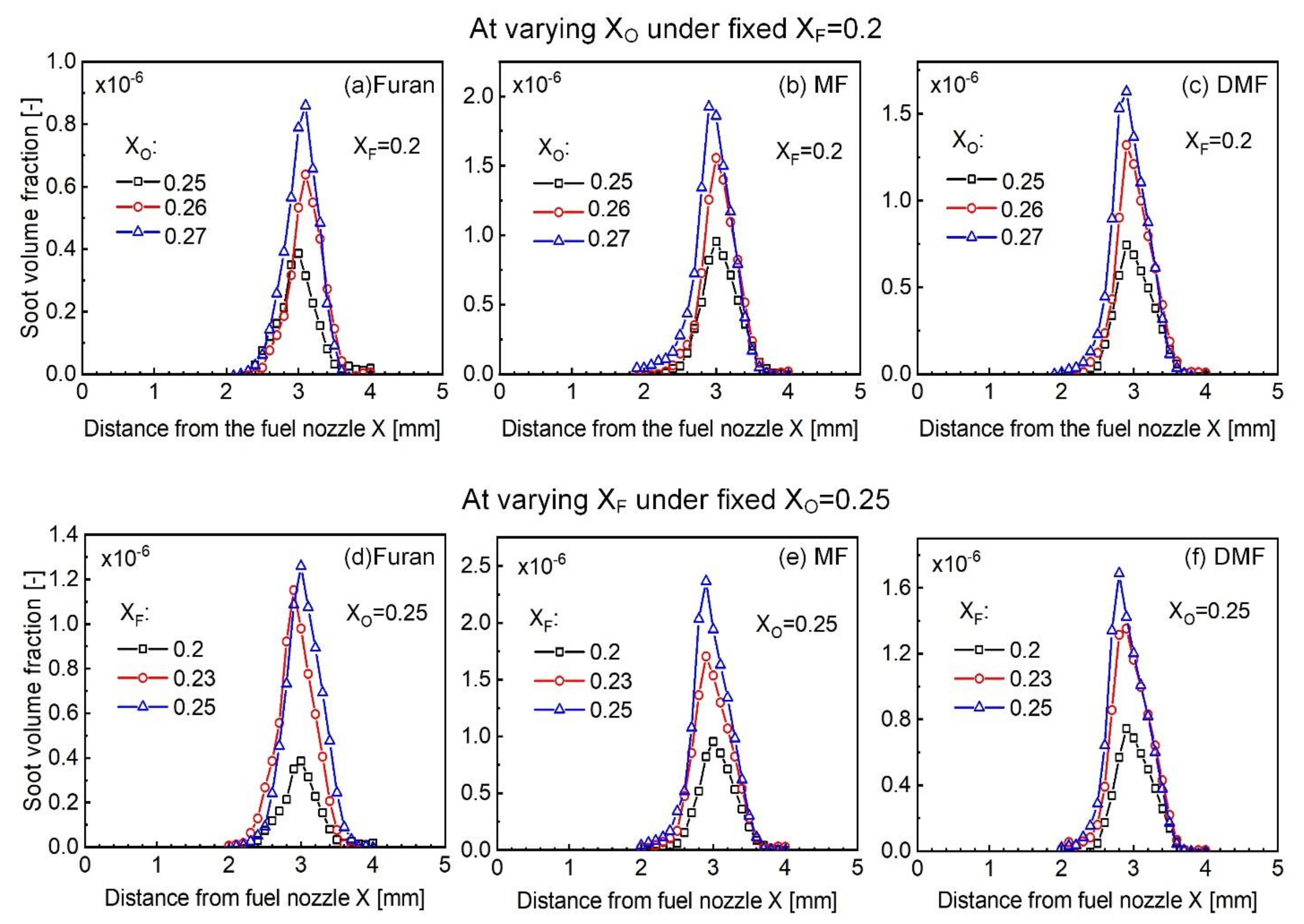
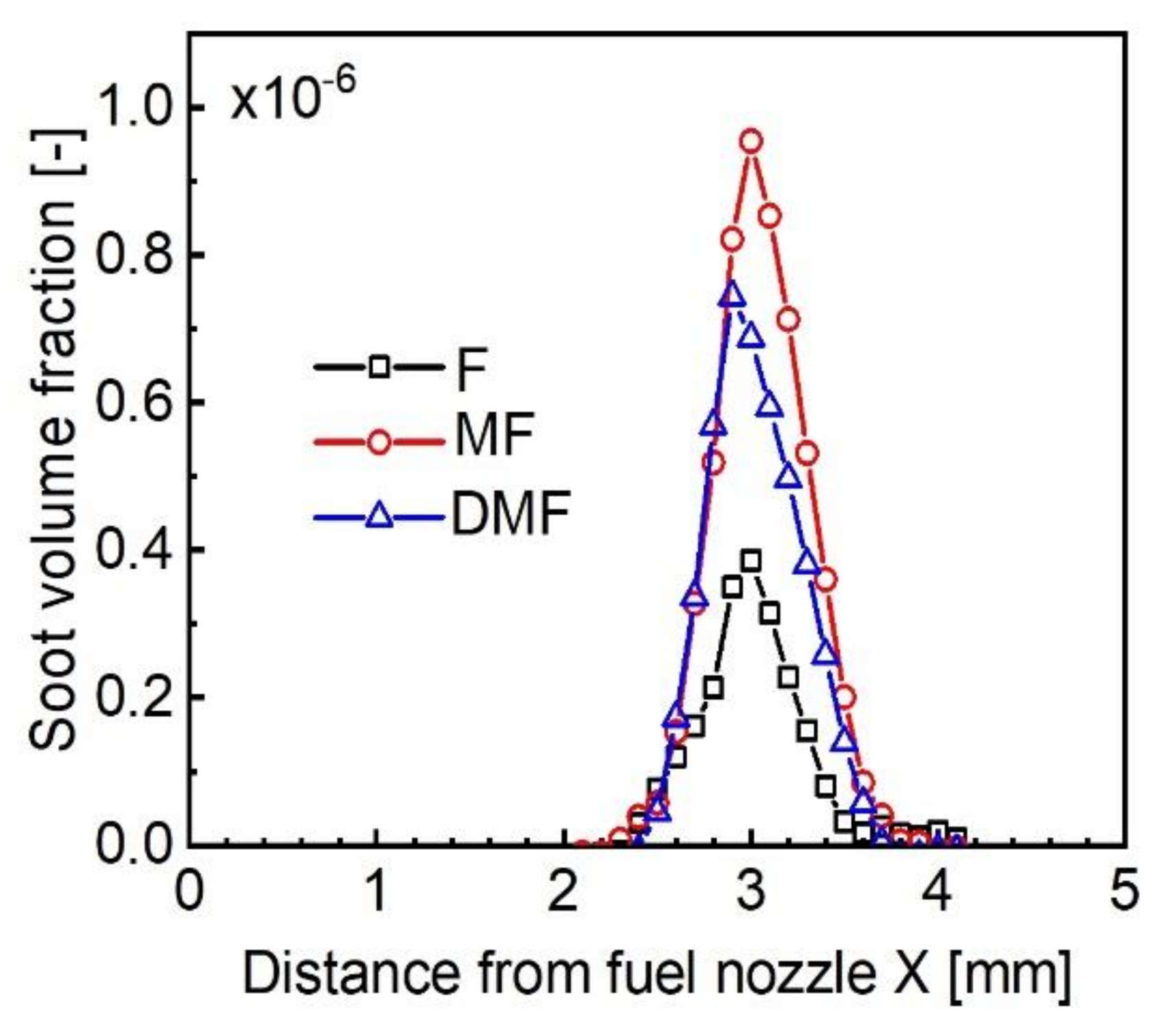
4.2. Experimental Results
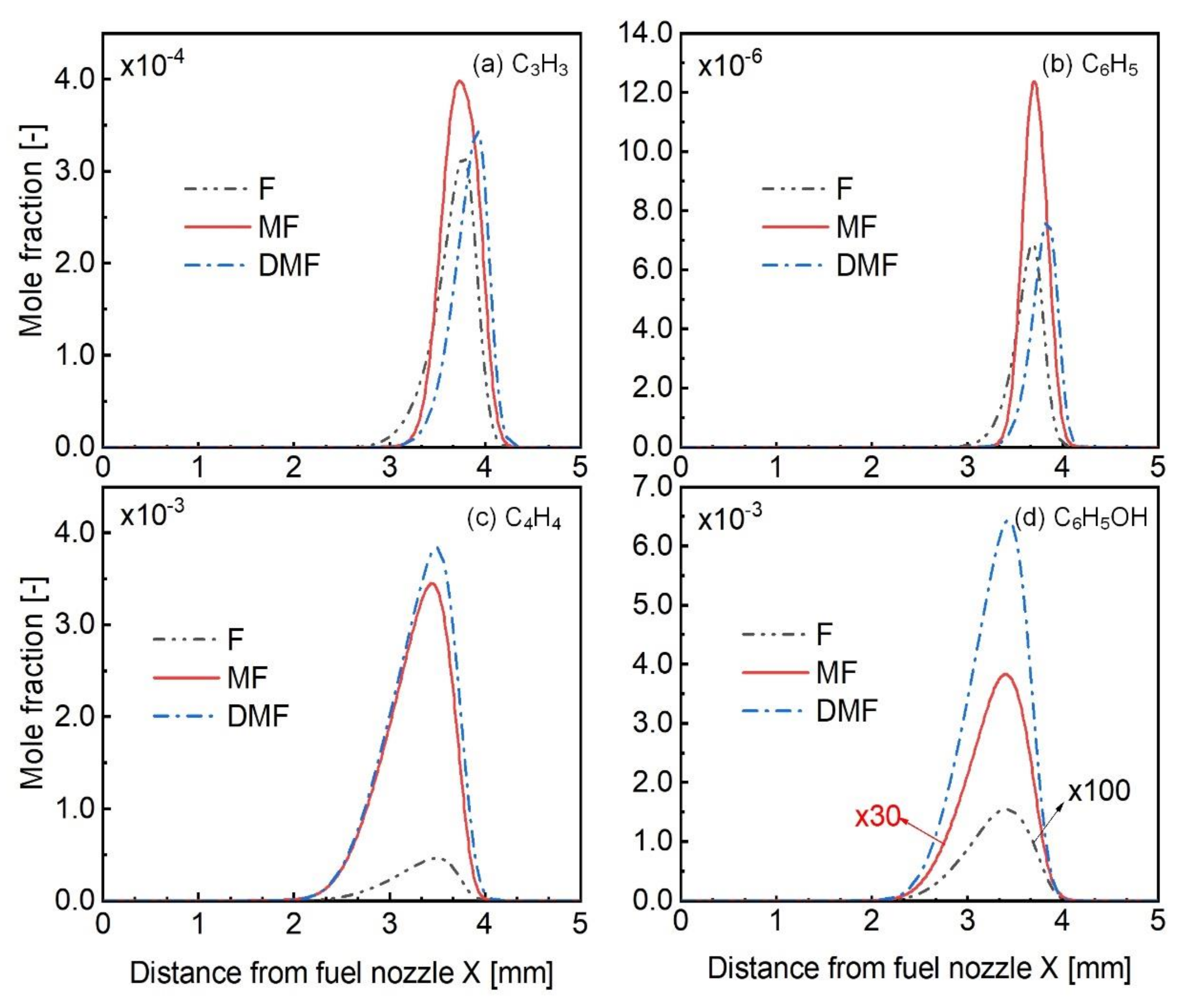
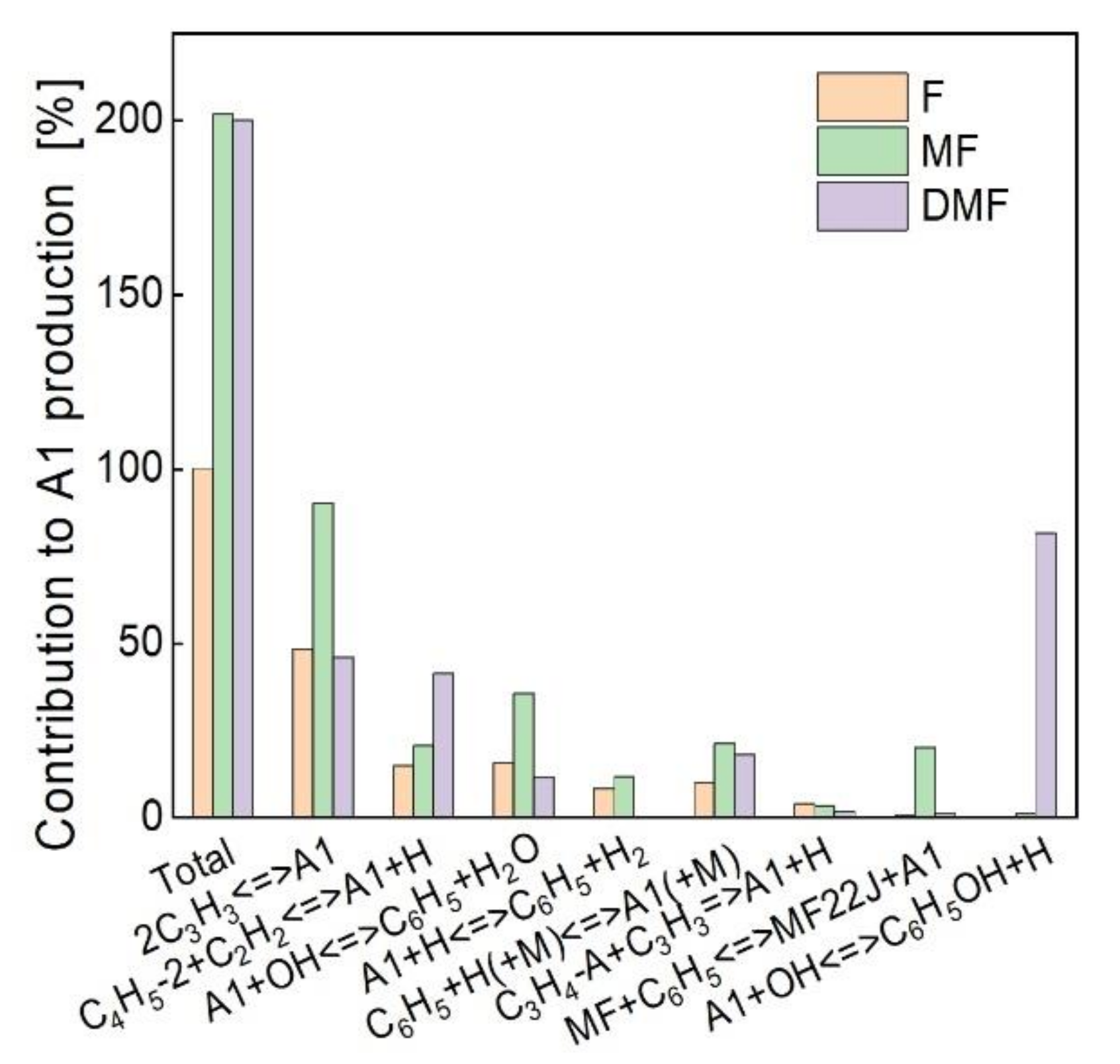
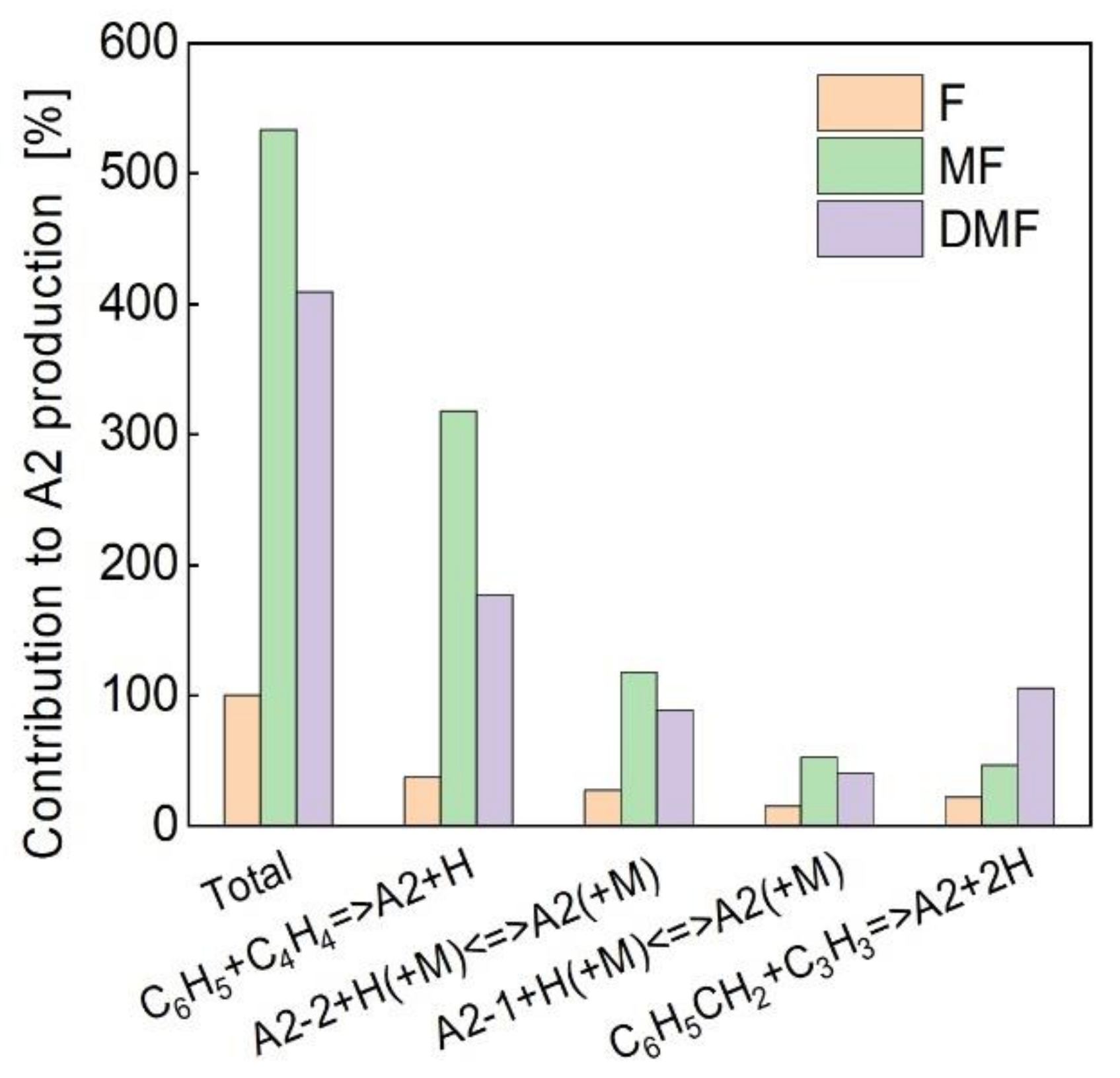
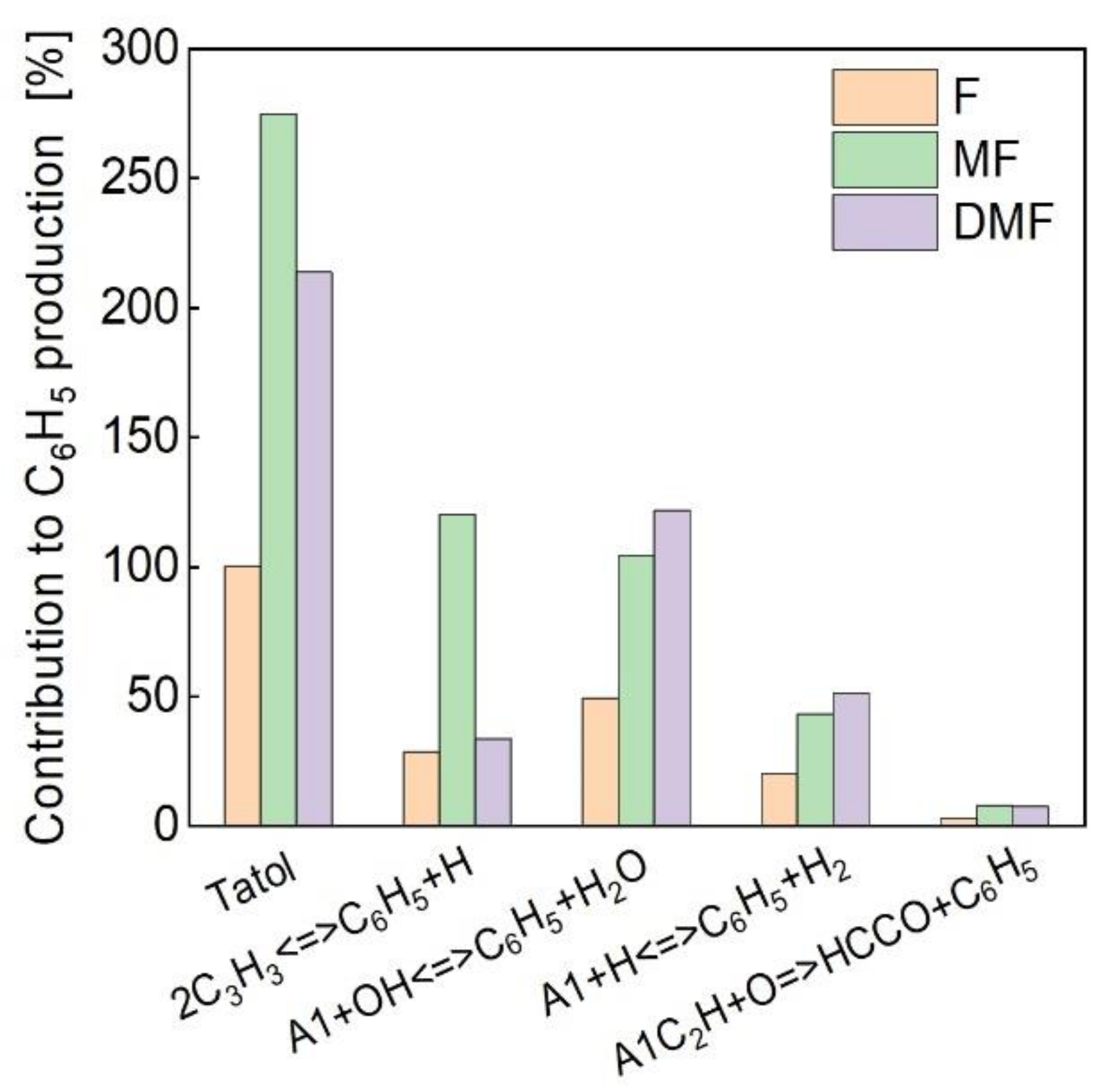
4.3. Kinetic Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kohse-Höinghaus, K. Combustion in the future: The importance of chemistry. Proc. Combust. Inst. 2021, 38, 1–56. [Google Scholar] [CrossRef]
- Xu, H.; Wang, C.; Ma, X.; Sarangi, A.K.; Weall, A.; Krueger-Venus, J. Fuel injector deposits in direct-injection spark-ignition engines. Prog. Energy Combust. Sci. 2015, 50, 63–80. [Google Scholar] [CrossRef]
- Bond, T.C.; Doherty, S.J.; Fahey, D.W.; Forster, P.M.; Berntsen, T.; Deangelo, B.J.; Flanner, M.G.; Ghan, S.; Kärcher, B.; Koch, D. Bounding the role of black carbon in the climate system: A scientific assessment. J. Geophys. Res. Atmos. 2013, 118, 5380–5552. [Google Scholar] [CrossRef]
- Kennedy, I.M. The health effects of combustion-generated aerosols. Proc. Combust. Inst. 2007, 31, 2757–2770. [Google Scholar] [CrossRef]
- Román-Leshkov, Y.; Barrett, C.J.; Liu, Z.Y.; Dumesic, J.A. Production of dimethylfuran for liquid fuels from biomass-derived carbohydrates. Nature 2007, 447, 982–985. [Google Scholar] [CrossRef] [PubMed]
- Saha, B.; Abu-Omar, M.M. Current Technologies, Economics, and Perspectives for 2,5-Dimethylfuran Production from Biomass-Derived Intermediates. ChemSusChem 2015, 8, 1133–1142. [Google Scholar] [CrossRef]
- Xu, N.; Gong, J.; Huang, Z. Review on the production methods and fundamental combustion characteristics of furan derivatives. Renew. Sustain. Energy Rev. 2016, 54, 1189–1211. [Google Scholar] [CrossRef]
- Wang, H.; Zhu, C.; Li, D.; Liu, Q.; Tan, J.; Wang, C.; Cai, C.; Ma, L. Recent advances in catalytic conversion of biomass to 5-hydroxymethylfurfural and 2, 5-dimethylfuran. Renew. Sustain. Energy Rev. 2019, 103, 227–247. [Google Scholar] [CrossRef]
- Tuan Hoang, A.; Viet Pham, V. 2-Methylfuran (MF) as a potential biofuel: A thorough review on the production pathway from biomass, combustion progress, and application in engines. Renew. Sustain. Energy Rev. 2021, 148, 111265. [Google Scholar] [CrossRef]
- Alexandrino, K. Comprehensive Review of the Impact of 2,5-Dimethylfuran and 2-Methylfuran on Soot Emissions: Experiments in Diesel Engines and at Laboratory-Scale. Energy Fuels 2020, 34, 6598–6623. [Google Scholar] [CrossRef]
- Chen, G.; Shen, Y.; Zhang, Q.; Yao, M.; Zheng, Z.; Liu, H. Experimental study on combustion and emission characteristics of a diesel engine fueled with 2,5-dimethylfuran–diesel, n-butanol–diesel and Gasoline–Diesel blends. Energy 2013, 54, 333–342. [Google Scholar] [CrossRef]
- Zhong, S.; Daniel, R.; Xu, H.; Zhang, J.; Turner, D.; Wyszynski, M.L.; Richards, P. Combustion and Emissions of 2,5-Dimethylfuran in a Direct-Injection Spark-Ignition Engine. Energy Fuels 2010, 24, 2891–2899. [Google Scholar] [CrossRef]
- Zheng, Z.; Wang, X.; Zhong, X.; Hu, B.; Liu, H.; Yao, M. Experimental study on the combustion and emissions fueling biodiesel/n-butanol, biodiesel/ethanol and biodiesel/2,5-dimethylfuran on a diesel engine. Energy 2016, 115, 539–549. [Google Scholar] [CrossRef]
- Michelsen, H.A. Probing soot formation, chemical and physical evolution, and oxidation: A review of in situ diagnostic techniques and needs. Proc. Combust. Inst. 2017, 36, 717–735. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Chung, S.H. Soot formation in laminar counterflow flames. Prog. Energy Combust. Sci. 2019, 74, 152–238. [Google Scholar] [CrossRef]
- Alexandrino, K.; Baena, C.; Millera, Á.; Bilbao, R.; Alzueta, M.U. 2-methylfuran pyrolysis: Gas-phase modelling and soot formation. Combust. Flame 2018, 188, 376–387. [Google Scholar] [CrossRef]
- Cheng, Z.; Xing, L.; Zeng, M.; Dong, W.; Zhang, F.; Qi, F.; Li, Y. Experimental and kinetic modeling study of 2,5-dimethylfuran pyrolysis at various pressures. Combust. Flame 2014, 161, 2496–2511. [Google Scholar] [CrossRef]
- Alexandrino, K.; Millera, Á.; Bilbao, R.; Alzueta, M.U. Novel aspects in the pyrolysis and oxidation of 2,5-dimethylfuran. Proc. Combust. Inst. 2015, 35, 1717–1725. [Google Scholar] [CrossRef]
- Togbé, C.; Tran, L.-S.; Liu, D.; Felsmann, D.; Oßwald, P.; Glaude, P.-A.; Sirjean, B.; Fournet, R.; Battin-Leclerc, F.; Kohse-Höinghaus, K. Combustion chemistry and flame structure of furan group biofuels using molecular-beam mass spectrometry and gas Chromatography—Part III: 2,5-Dimethylfuran. Combust. Flame 2014, 161, 780–797. [Google Scholar] [CrossRef] [Green Version]
- Tran, L.-S.; Sirjean, B.; Glaude, P.-A.; Kohse-Höinghaus, K.; Battin-Leclerc, F. Influence of substituted furans on the formation of Polycyclic Aromatic Hydrocarbons in flames. Proc. Combust. Inst. 2015, 35, 1735–1743. [Google Scholar] [CrossRef]
- Cheng, Z.; He, S.; Xing, L.; Wei, L.; Li, W.; Li, T.; Yan, B.; Ma, W.; Chen, G. Experimental and Kinetic Modeling Study of 2-Methylfuran Pyrolysis at Low and Atmospheric Pressures. Energy Fuels 2017, 31, 896–903. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, P.; Liu, X.; Chen, B.; Geng, C.; Li, B.; Wang, H.; Li, Z.; Yao, M. Laser diagnostics and chemical kinetic analysis of PAHs and soot in co-flow partially premixed flames using diesel surrogate and oxygenated additives of n -butanol and DMF. Combust. Flame 2018, 188, 129–141. [Google Scholar] [CrossRef]
- Conturso, M.; Sirignano, M.; D’Anna, A. Effect of 2,5-dimethylfuran doping on particle size distributions measured in premixed ethylene/air flames. Proc. Combust. Inst. 2017, 36, 985–992. [Google Scholar] [CrossRef]
- Conturso, M.; Sirignano, M.; D’Anna, A. Effect of furanic biofuels on particles formation in premixed Ethylene–Air flames: An experimental study. Fuel 2016, 175, 137–145. [Google Scholar] [CrossRef]
- Sirignano, M.; Conturso, M.; D’Anna, A. Effect of furans on particle formation in diffusion flames: An experimental and modeling study. Proc. Combust. Inst. 2015, 35, 525–532. [Google Scholar] [CrossRef]
- Chu, H.; Ya, Y.; Nie, X.; Qiao, F.; Jiaqiang, E. Effects of adding cyclohexane, n-hexane, ethanol, and 2,5-dimethylfuran to fuel on soot formation in laminar coflow n-heptane/iso-octane diffusion flame. Combust. Flame 2021, 225, 120–135. [Google Scholar] [CrossRef]
- Chu, H.; Ya, Y.; Nie, X.; Zhou, Y.; Hu, J.; Dong, S.; Jiaqiang, E. Effects of ethanol and 2, 5-dimethylfuran addition on the morphology and nanostructure evolution of soot in gasoline primary reference fuel-air coflow diffusion flames. Fuel 2020, 281, 118711. [Google Scholar] [CrossRef]
- Xu, L.; Yan, F.; Zhou, M.; Wang, Y.; Chung, S.H. Experimental and soot modeling studies of ethylene counterflow diffusion flames: Non-monotonic influence of the oxidizer composition on soot formation. Combust. Flame 2018, 197, 304–318. [Google Scholar] [CrossRef]
- Yan, F.; Zhou, M.; Xu, L.; Wang, Y.; Chung, S.H. An experimental study on the spectral dependence of light extinction in sooting ethylene counterflow diffusion flames. Exp. Therm. Fluid Sci. 2019, 100, 259–270. [Google Scholar] [CrossRef] [Green Version]
- Yi, X.; Lee, C.F.F. Forward-illumination light-extinction technique for soot measurement. Appl. Opt. 2006, 45, 2046–2057. [Google Scholar]
- Xu, Y.; Lee, C. Study of Soot Formation of Oxygenated Diesel Fuels Using Forward Illumination Light Extinction (FILE) Technique. SAE Tech. Pap. 2006. [Google Scholar] [CrossRef]
- Bi, L.; Yang, P.; Kattawar, G.W. Edge-effect contribution to the extinction of light by dielectric disks and cylindrical particles. Appl. Opt. 2010, 49, 4641–4646. [Google Scholar] [CrossRef]
- Santoro, R.J.; Semerjian, H.G.; Dobbins, R.A. Soot particle measurements in diffusion flames. Combust. Flame 1983, 51, 203–218. [Google Scholar] [CrossRef]
- Eremin, A.; Gurentsov, E.; Popova, E.; Priemchenko, K. Size dependence of complex refractive index function of growing nanoparticles. Appl. Phys. B 2011, 104, 285–295. [Google Scholar] [CrossRef]
- Krishnan, S.S.; Lin, K.-C.; Faeth, G.M. Extinction and Scattering Properties of Soot Emitted From Buoyant Turbulent Diffusion Flames. J. Heat Trans. 2000, 123, 331–339. [Google Scholar] [CrossRef]
- Diego, C.P.R.D.S. CHEMKIN-PRO 15131; Reaction Design: San Diego, CA, USA, 2011. [Google Scholar]
- Somers, K.P.; Simmie, J.M.; Metcalfe, W.K.; Curran, H.J. The pyrolysis of 2-methylfuran: A quantum chemical, statistical rate theory and kinetic modelling study. Phys. Chem. Chem. Phys. 2014, 16, 5349–5367. [Google Scholar] [CrossRef] [Green Version]
- Somers, K.P.; Simmie, J.M.; Gillespie, F.; Conroy, C.; Black, G.; Metcalfe, W.K.; Battin-Leclerc, F.; Dirrenberger, P.; Herbinet, O.; Glaude, P.A.; et al. A comprehensive experimental and detailed chemical kinetic modelling study of 2,5-dimethylfuran pyrolysis and oxidation. Combust. Flame 2013, 160, 2291–2318. [Google Scholar] [CrossRef] [Green Version]
- Somers, K.P.; Simmie, J.M.; Gillespie, F.; Burke, U.; Connolly, J.; Metcalfe, W.K.; Battin-Leclerc, F.; Dirrenberger, P.; Herbinet, O.; Glaude, P.A.; et al. A high temperature and atmospheric pressure experimental and detailed chemical kinetic modelling study of 2-methyl furan oxidation. Proc. Combust. Inst. 2013, 34, 225–232. [Google Scholar] [CrossRef] [Green Version]
- Yu, W.; Raj, A.; Chung, S.H. A PAH growth mechanism and synergistic effect on PAH formation in counterflow diffusion flames. Combust. Flame 2013, 160, 1667–1676. [Google Scholar]
- Wang, J.; Sun, W.; Wang, G.; Fan, X.; Lee, Y.-Y.; Law, C.K.; Qi, F.; Yang, B. Understanding benzene formation pathways in pyrolysis of two C6H10 isomers: Cyclohexene and 1,5-hexadiene. Proc. Combust. Inst. 2019, 37, 1091–1098. [Google Scholar] [CrossRef]
- Hansen, N.; Kasper, T.; Yang, B.; Cool, T.A.; Li, W.; Westmoreland, P.R.; Oßwald, P.; Kohse-Höinghaus, K. Fuel-structure dependence of benzene formation processes in premixed flames fueled by C6H12 isomers. Proc. Combust. Inst. 2011, 33, 585–592. [Google Scholar] [CrossRef]
- Zhang, H.R.; Eddings, E.G.; Sarofim, A.F.; Westbrook, C.K. Fuel dependence of benzene pathways. Proc. Combust. Inst. 2009, 32, 377–385. [Google Scholar] [CrossRef]
- Xu, L.; Yan, F.; Wang, Y.; Chung, S.H. Chemical effects of hydrogen addition on soot formation in counterflow diffusion flames: Dependence on fuel type and oxidizer composition. Combust. Flame 2020, 213, 14–25. [Google Scholar] [CrossRef]
- Gleason, K.; Carbone, F.; Sumner, A.J.; Drollette, B.D.; Plata, D.L.; Gomez, A. Small aromatic hydrocarbons control the onset of soot nucleation. Combust. Flame 2021, 223, 398–406. [Google Scholar] [CrossRef]
- Yan, F.; Xu, L.; Wang, Y.; Park, S.; Sarathy, S.M.; Chung, S.H. On the opposing effects of methanol and ethanol addition on PAH and soot formation in ethylene counterflow diffusion flames. Combust. Flame 2019, 202, 228–242. [Google Scholar] [CrossRef]
- Frenklach, M. Reaction mechanism of soot formation in flames. Phys. Chem. Chem. Phys. 2002, 4, 2028–2037. [Google Scholar] [CrossRef]
- Frenklach, M.; Singh, R.I.; Mebel, A.M. On the low-temperature limit of HACA. Proc. Combust. Inst. 2019, 37, 969–976. [Google Scholar] [CrossRef]




| Series | Fuel Stream | Oxidizer Stream | Zst (F/MF/DMF) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| F/MF/DMF | N2 | V (cm/s) | T | O2 | N2 | V (cm/s) | T | ||
| Base | 0.2/0.2/0.2 | 0.8 | 20 | 373 K | 0.25 | 0.75 | 20 | 298 K | 0.256/0.218/0.193 |
| Varying XO | 0.2/0.2/0.2 | 0.8 | 20 | 0.26 | 0.74 | 20 | 0.264/0.225/0.199 | ||
| 0.2/0.2/0.2 | 0.8 | 20 | 0.27 | 0.73 | 20 | 0.271/0.231/0.205 | |||
| Varying XF | 0.23/0.23/0.23 | 0.77 | 20 | 0.25 | 0.75 | 20 | 0.237/0.202/0.179 | ||
| 0.25/0.25/0.25 | 0.75 | 20 | 0.25 | 0.75 | 20 | 0.226/0.193/0.171 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mu, Q.; Yan, F.; Zhang, J.; Xu, L.; Wang, Y. Experimental and Numerical Study on the Sooting Behaviors of Furanic Biofuels in Laminar Counterflow Diffusion Flames. Energies 2021, 14, 5995. https://doi.org/10.3390/en14185995
Mu Q, Yan F, Zhang J, Xu L, Wang Y. Experimental and Numerical Study on the Sooting Behaviors of Furanic Biofuels in Laminar Counterflow Diffusion Flames. Energies. 2021; 14(18):5995. https://doi.org/10.3390/en14185995
Chicago/Turabian StyleMu, Qianqian, Fuwu Yan, Jizhou Zhang, Lei Xu, and Yu Wang. 2021. "Experimental and Numerical Study on the Sooting Behaviors of Furanic Biofuels in Laminar Counterflow Diffusion Flames" Energies 14, no. 18: 5995. https://doi.org/10.3390/en14185995
APA StyleMu, Q., Yan, F., Zhang, J., Xu, L., & Wang, Y. (2021). Experimental and Numerical Study on the Sooting Behaviors of Furanic Biofuels in Laminar Counterflow Diffusion Flames. Energies, 14(18), 5995. https://doi.org/10.3390/en14185995







