Abstract
The drivers underpinning the emergence of SARS-CoV-2 and climate change attest to the fact that we are now living in the Anthropocene Epoch, with human activities significantly impacting and altering the global ecosystem. Here, we explore the historical context of zoonoses, the effect of anthropogenic climate change and interrelated drivers on the emergence of, and response to emerging infectious diseases. We call attention to an urgent need for inculcating a One Health research agenda that acknowledges the primary interconnection between animals, humans, pathogens, and their collective milieus to foster long term resilience across all systems within our shared planetary environment.
1. Introduction
In late 2019, Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), the causal agent of the Coronavirus disease 2019 (COVID-19) emerged from a wildlife wet market in the city of Wuhan in the Hubei province of China [1,2]. The virus spread across the world reaching a pandemic status with an estimated global case count of 197 million and at least 4 million deaths as of July 2021 [3]. An estimated 7.8 billion people on Earth today are witnessing a pandemic unlike any other since the 1918–1919 Spanish Flu that had infected one-third of the world’s population (500 million people) [4] at the time. Against the backdrop of ever-widening social inequities [4,5], globalization [4,6,7], resource over-exploitation [8,9] and biodiversity loss [10,11], the COVID-19 pandemic has highlighted a global gap in governance and pandemic preparedness [12]. After more than a year since the onset of COVID-19, and despite sustained travel restrictions, social distancing, lockdown measures and vaccine development, there is still no stage for a discussion on when or how the pandemic will end. While many have claimed that a pandemic of this scale was impossible to predict, let alone prevent, public health experts, conservationists and disease ecologists alike had been issuing warnings of a coronavirus pandemic since 2003 [13]. First with the identification of SARS-CoV-1 in China in 2004 [14] and then with the Middle Eastern Respiratory Syndrome (MERS-CoV) in Saudi Arabia, 2012 [15]. COVID-19′s origin at the human–environment–animal interface, its accelerated expansion and the collateral damage from a trillion-dollar economic fallout [16], warrants an urgent and renewed examination into the mechanistic causes responsible for such events.
Here, we explore the historical context of zoonoses, the effect of anthropogenic climate change and interrelated drivers on the emergence of, and response to Emerging Infectious Diseases (EIDs). We call attention to an urgent need for inculcating a One Health research agenda to mobilise multisectoral stakeholder groups and foster long term resilience across all systems within our shared planetary environment.
2. Disease Emergence in a Novel Geological Epoch
Zoonoses are infectious diseases caused by pathogens including viruses, bacteria, parasites, prions, and fungi that have potential of transmission between human and non-human vertebrates [17]. Whether due to an isolated event of pathogen spill over from non-human animals to humans later adapting spreading through person-person transmission or due to sustained interspecies transmission, zoonoses have existed within human society since time immemorial. From rabies-the first ever recorded zoonotic disease in 2000 BC [18] to the currently ongoing COVID-19 pandemic, the vast majority (up to 70%) of emerging diseases (e.g., Nipah encephalitis, Zika, Ebola, Variant Creutzfeldt-Jakob disease (vCJD), Lyme disease, etc.) and all known pandemics throughout the history (e.g., influenza such as H5N1 or H1N1, SARS, MERS, HIV/AIDS) have been zoonoses [19].
Over the past few decades, the number of EIDs, especially those belonging to the Coronaviridae family with pandemic potential and a high probability of transmission to humans, has been rapidly increasing [20]. A recent spatiotemporal distribution analysis of 12,102 outbreaks of 215 human infectious diseases across 219 nations adjusted for disease surveillance, communications, geography, and host availability concluded that the total number of infectious disease outbreaks across continents have increased significantly (p < 0.0001) since 1980 [21].
This increase is, in part attributable to the ever-expanding global interconnected nature of modern society and a novel geological epoch–the Anthropocene–where humans exert the greatest influence in shaping changes to environments and systems on a planetary scale [22]. The initial SARS-CoV-2 transmission from an animal to a human host, widely believed to have been facilitated in a Huanan South China seafood wet market, is exemplary of this epoch [23]. Wet markets across Southeast Asia, known for selling and serving a myriad of fresh and alive exotic wildlife for human consumption such as poultry, bats, snakes, marmots, pangolins, racoons, etc. serve as a merging point for millions of microbes, across species that have historically and ecologically never had close contact. The recurrent intermixing of animal fluids such as blood, saliva, and urine during culling, in conjunction to the large human crowds that visit these markets, orchestrates conditions for potential spill over events on a daily basis [23]. Such contact also increases chances for a potential pathogen to acquire novel virulence traits and increased transmissibility as a consequence of genetic recombination and reassortment. The SARS-CoV-1 pandemic of the early 2000s, caused by human consumption of the highly traded civet cat in a similar wet market, underscores the stark similarity between such spill over events [24]. The occurrence of zoonoses, however, is not merely restricted to wet markets, it is potentiated in imbalanced environments existing at the interface of the human-wildlife-domestic animal triad. Such environments include hunting grounds [25], indiscriminately encroached upon natural lands, wildlife trading camps [26], and more commercial habitats commonly visited such as beaches, forests, lakes etc.
4. Interrelationship between Climate Change, COVID-19, and AMR
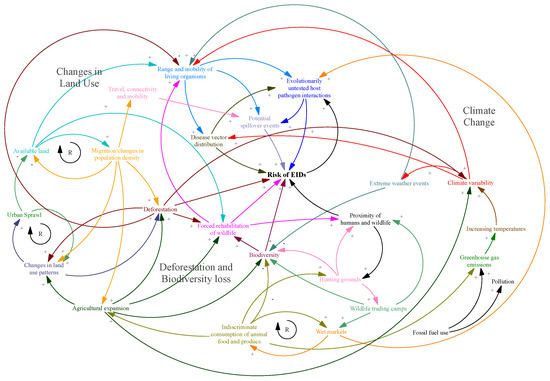
The co-occurrence of the pandemic in the context of climate change and vice versa has showcased the overlapping vulnerability of communities that have contributed to the already high morbidity and mortality burden. A minimum of 92 of the estimated 132 unique extreme weather events that occurred in 2020, have overlapped with the pandemic [55]. While the specific nature of threats changed over seasons and regions, these hazards jeopardized sensitive public health work pertaining to pandemic response. In Zimbabwe, millions were left without access to clean water and at risk of acute food insecurity due to drought. Several southern African countries, heavily dependent on hydropower, experienced large scale power cuts. This led to a hampered coordination of response to the pandemic. In 2020, an estimated 437.1 million people in vulnerable groups were exposed to extreme heat [55], the impacts of which were glaringly apparent in Australia [56], where adverse health impacts because of air pollution due to an extreme bushfire season likely worsened COVID-19 morbidity [57]. On a broader scale, a combination of geopolitical stressors and climate catalysed large scale forced migrations [58,59,60]. Displaced populations, such as the ones residing in refugee camps in Yemen and Rohingya, developed an increased risk of contracting COVID-19 due to living conditions incompatible with social distancing, improper access to testing facilities and an acute lack of healthcare facilities. Cases of other infectious diseases such as diphtheria also skyrocketed in these camps [61,62]. EIDs with the potential to become pandemics and climate change along with their underlying drivers occur on distinct temporal scales. They are, in many ways, like the complex disease interaction(s), shared pathways and underlying social forces serving as interrelated impressions shaping their collective exhibition (Figure 1).
Both EIDs and climate risks are systemic, i.e., their direct manifestations and knock-on effects proliferate rapidly in an interconnected world. Both are nonstationary, i.e., their past probabilities and distribution of occurrence changes rapidly and is usually inadequate to base future projections. Both are nonlinear, with disproportionate socioeconomic impacts once certain thresholds are breached (surface temperature thresholds in case of climate change and hospital capacities in case of COVID 19). They both constitute risk multipliers, i.e., they underscore and aggravate previously untested susceptibilities intrinsic to human society and systems. Both are regressive, i.e., the most vulnerable communities across the world are disproportionately affected and neither can be considered a ‘black swan’ event that experts have been consistently warning against for many years. Addressing both requires a fundamental shift, from optimizing largely for the shorter-term performance of systems to building correspondingly in their longer-term resiliency [63]. The ongoing pandemic has demonstrated, healthcare systems, agricultural practices, supply chains, and cities because of their design, are struggling to function within operational conditions. The measures being implemented to respond to the pandemic, including vaccine development, research, genotyping, personal protective equipment, tracking, pop-up hospitals, mass vaccination and changes to fiscal policies (government stimulus packages approaching several trillion dollars) are illustrative of how expensive the failure to build systemic resiliency can ultimately prove to be [63]. For both climate change and pandemics, the cost of controlling a global crisis inevitably exceeds investments toward their prevention.
Over the next few decades, climate change and antimicrobial resistance (AMR) will rise in prominence as two imminent threats to global public health, accelerated by globalisation. AMR is widely acknowledged as the next big pandemic, and in the US alone, drug-resistant infections are the third leading cause of mortality–estimated to account for ~162,000 deaths annually [64]. The complex transmission dynamics of AMR combined with large interregional inequalities in surveillance systems and reporting frameworks pose grave challenges in estimating the true burden of AMR infections worldwide [65]. However, as an evolutionary feature, it is inevitable that microbes develop resistance traits that will be maintained and propagated so long as the selection pressure induced by antimicrobials persists.
Warmer temperatures can broaden the endemicity of pathogens that host resistance determinants by increasing their growth rates, hence widening their scope, and expanding their pool of susceptible hosts. In the absence of alternatives to replace the existing repertoire of antimicrobials, the increased incidence of resistant infections can place a burden on health systems to prescribe more antimicrobials, propagating resistance further [66]. As AMR becomes more pervasive, healthcare infrastructure inadequacies highlighted during the COVID-19 pandemic will be compounded, as existing treatments against a multitude of infections become obsolete. Langford et al. conducted a meta-analysis into the current extent of irrational prescription practices, finding that 74.6% of 30,623 patients presenting with COVID-19 were incorrectly prescribed antibiotics in the first instance [67,68]. These figures warrant the development of rapid AMR diagnostics and stringent prescription guidelines to mitigate the risk of increasing the morbidity and mortality associated with resistant pathogens that are already selected for.
Several studies have also discussed the positive correlation between the incidence of bacterial infections with rising temperatures, humidity, and monthly precipitation [69]. A study of three clinically relevant pathogens (E. coli, K. pneumoniae and S. aureus) across 41 states in the US confirmed that warmer temperatures can drive bacterial growth and also potentially drive resistance acquisition through horizontal gene transfer. The study revealed that minimum temperatures rising by 10 °C can increase resistance acquisition rates between 2.2 and 4.2% (p < 0.05) [70]. This would provide a further explanation for the higher prevalence of extended-spectrum beta-lactamase-producing Enterobacteriaceae in Southern Europe, previously solely attributed to irrational prescription practices in the region. A study by Arias-Andres et al. also unearthed the role of microplastics in freshwater ecosystems in facilitating HGT between phylogenetically distant bacterial taxa, which can have health implications for communities that rely on these water sources [71].
As biomedical research begins to appreciate the complex involvement of the human microbiome and its varied roles in maintaining homeostatic function, the effects of antimicrobial-induced disruptions to the gut microbiota become increasingly clear [72]. Researchers have discovered relationships between dysbiosis of the gut microbiota and pathogenesis of non-communicable diseases such as heart disease, obesity, diabetes and even some cancers, although direct causality is yet to be established. Newer studies have also substantiated the complex dynamics between the gut microbiome and mental health disorders through the gut-brain axis [73]. Dysregulation of this bidirectional signalling pathway via antimicrobial use promotes the pathogenesis of psychiatric disorders such as anxiety and depression, with an opposite prophylactic effect observed in patients taking probiotic supplements to replenish the microbiota.
The climate has a profound effect on the incidence and transmission of communicable diseases. At a population level, warming temperatures can have an impact on climate-sensitive infectious diseases. Extreme weather events and pollution can also affect clean water sources, hygiene, and sanitation infrastructure, which can act as reservoirs for resistance genes, inadvertently driving AMR at the genomic and molecular level within the microbiota [74]. These effects take time to accumulate and are likely to go unnoticed, gradually increasing our propensity to develop non-communicable diseases at the individual level. This demonstrates the potential direct and indirect impacts of climate change on global public health through varying degrees of granularity and should create an incentive to increase public healthcare expenditure–associated with lower levels of aggregate resistance overall.
5. A One Health Paradigm for Policy
The current pandemic is merely a microcosmic, short-term reflection of the dynamics underpinning a global, long-term interrelated sustainability crisis—biodiversity loss, indiscriminate consumption patterns and climate change—that humanity is slated to face during the twenty-first century. However, it provides us with a window of opportunity to address future crises in a proactive manner and re-examine our current over-consumptive trajectory with nature. A collaborative, multisectoral and transdisciplinary One Health approach that recognizes the interconnectedness between, animals, microorganisms, humans, and their shared planetary environment is essential. It must work across the local, regional, national, and global levels with a singular goal of accomplishing optimal health. A One Health paradigm underscores the importance of interdisciplinary and transdisciplinary approaches that go beyond conventional boundaries of environmental sustainability and public health [75] (Figure 2). A One Health approach to both pandemics and climate change, is doubly advantageous as it potentiates significant contributions to the multiple contexts and issues including (but not restricted to) food security [76], animal food sources [76], livestock systems [77], environmental sanitation [78], and in establishing global integrated syndromic surveillance and response systems [79]. Research based on this understanding must assimilate evidence to inform a framework within which interpreting and applying evidence from a One Health approach for preventing further adverse global catastrophes would be key.
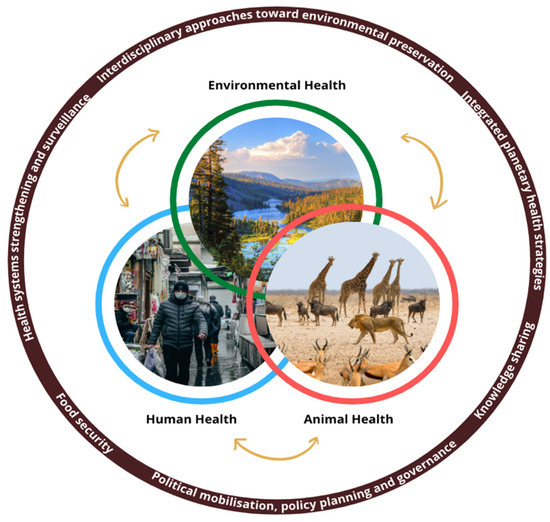
Figure 2.
The One Health Approach. A collaborative, multisectoral and transdisciplinary approach that recognizes the interconnectedness between, animals, humans, and their shared planetary environment. Arrows are indicative of the synergy across shared environments. Pictures source: Shutterstock (URL: https://www.shutterstock.com, accessed on 10 September 2021).
The United Nations Framework Convention on Climate Change (UNFCCC) is a formal multilateral agreement which was first established in 1992 and falls under the United Nations Environment Programme (UNEP). The UNEP has attempted to act as the leading global authority for environmental governance, with a focus on sustainability. The UNFCCC takes into consideration the heterogeneity of populations and geographies and sets out a framework to help participating states anticipate or mitigate climate effects by helping tailor strategic action plans to specific states depending on their vulnerability to environmental catastrophes, accounting for limitations posed by varying socio-economic contexts [80]. As the effects of globalisation were deemed to accelerate the rate of climate change, the original UNFCCC has since been updated with amendments to reflect shifting priorities based on evidence, recognised in the Kyoto protocol of 1997 and the Paris Agreement in 2015, which was adopted by 196 signatory states, with the United States joining in January 2021.
A key stakeholder and collaborator, specifically in contextualising the effects of these interventions within the public health landscape has been the World Health Organisation (WHO). The support of the WHO has been documented in the ‘COP24 Special Report’, written up following the 23rd Conference of the Parties (COP23) for the UNFCCC [81]. The report emphasises the complex and delicate interplay between climate change and worsening health outcomes, in the context of both communicable and non-communicable diseases. The report also highlights the economic and political co-benefits to various social sectors if management plans for climate change are established in advance to promote intersectional compliance and long-term investments for climate-adaptive infrastructure. The UNFCCC provides a solid foundation to base this action.
Recently, a growing chorus of calls for establishing inclusive and transparent One Health research coalitions to strengthen linkages with the evolving climate change between planetary health research communities, medical professionals, veterinarians, anthropologists, ecologists, politicians, laboratory-based scientists, economists, and others have gathered traction [82,83,84]. Mediation and sustenance of such partnerships will be critical in informing our collective and sustainable approach to tackling future challenges.
Beyond research, multisectoral collaborations mediated under the One Health lens can act as an enabler for the health in all policies (HiAP) approach and engage key stakeholders that have chronically been underrepresented in the discourse surrounding research uptake in policy. Working off the foundations of the Alma Ata Declaration (1978) and the Ottawa Charter (1968), the one health paradigm could facilitate the HiAP approach that seeks synergies in taking into consideration the implications of intersectoral policy on the social determinants of health and health systems, encouraging health equity and promotion [85]. Such an approach would encompass a pragmatic and versatile set of policies employed systematically across different fora to achieve the same underlying goal of healthy living and wellbeing. Complemented by the One Health lens, HiAP could support the development of cross-sectoral partnerships by highlighting the co-benefits of factoring in health as a priority in the process of societal development to all parties involved–emphasising on the cost-effectiveness of these measures, the creation of jobs and the greater economic stability offered as a result of long-term investments [86]. Moreover, focussed evidence generation through the one health coalitions helps facilitate the political cooperation required between health, economic and environment actors to mobilize resources to address the effects of anthropogenic activity on the environment and ensures sustainable development for future generations.
By fostering an increased collaboration between the environmental and public health research community and by sharing cross sectoral expertise, especially in the sphere of policy, ownership of actions at various levels could be expedited. This could go a long way in bridging the gap in evidence informed policy making, which, incidentally, is often contested despite being central to health and environmental litigation [87].
6. Conclusions
While global pandemic preparedness has marginally improved overall by learning from past experiences, response to pandemics has always been reactionary. Going forward, adoption of a focused, preventative approach is required instead. However, to achieving this, there remain barriers to cooperation and collaboration required to address the root causes and underlying drivers of EIDs. Traditional, siloed approaches alone can neither be relied upon to predict spill over events leading to epidemics and pandemics nor they can inform strategies for addressing systemic and endemic problems. While the complex and dynamic nature of climate change makes direct cause-effect relationships difficult to establish, most currently available evidence points to a fast-evolving climate crisis and warrants urgent action. Therefore, a precautionary approach towards addressing climate change, acknowledging the long-term outcomes of exploiting existing ecosystem on human health and wellbeing, is worth the investment despite the risks involved. Governments need to start conducting proactive cost-benefit analyses and develop appropriately guided policy frameworks. A One Health approach could, with sufficient goodwill, conviction, political provision, and financial support, substantially reduce the dual threat of a climate crisis and future pandemics. An essential element of global pandemic preparedness would be to recognise and act upon the timely recognized interests of the scientific and political agendas, in a hope to eventually harmonise our shared existence with nature.
Author Contributions
A.M. (Ankur Mutreja) framed and supervised the work. S.S., F.K., A.M. (Archana Madhav), B.P. and V.S. wrote the first draft. A.M. (Ankur Mutreja) and V.S. reviewed and edited the submitted manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
A.M. (Ankur Mutreja) is funded by the NIHR Cambridge Biomedical Research Centre and NIHR AMR Research Capital Funding Scheme [NIHR200640]. The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
Acknowledgments
The authors thank Mamatha Ballal, Kasturba Medical College, Manipal, India for her advice.
Conflicts of Interest
The authors declare no conflict of interest.
Glossary
| SARS-CoV-2 | Severe Acute Respiratory Syndrome Coronavirus 2 |
| COVID-19 | Coronavirus Disease 2019 |
| MERS-CoV | Middle Eastern Respiratory Syndrome |
| vCJD | Variant Creutzfeldt-Jakob disease |
| SARS | Severe Acute Respiratory Syndrome |
| HIV | Human Immunodeficiency Virus |
| AMR | Antimicrobial resistance |
| NCDs | Non-communicable diseases |
| AIDS | Acquired immune deficiency syndrome |
| EID | Emerging infectious disease |
| SARS-CoV-1 | Severe Acute Respiratory Syndrome |
References
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.-M.; Wang, W.; Song, Z.-G.; Hu, Y.; Tao, Z.-W.; Tian, J.-H.; Pei, Y.-Y.; et al. A new coronavirus associated with human respiratory disease in China. Nature 2020, 579, 265–269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johns Hopkins Coronavirus Resource Center. Global Map. Available online: https://coronavirus.jhu.edu/map.html. (accessed on 6 October 2020).
- 1918 Pandemic (H1N1 Virus)|Pandemic Influenza (Flu)|CDC. Available online: https://www.cdc.gov/flu/pandemic-resources/1918-pandemic-h1n1.html (accessed on 14 June 2021).
- Clouston, S.A.; Natale, G.; Link, B.G. Socioeconomic inequalities in the spread of coronavirus-19 in the United States: A examination of the emergence of social inequalities. Soc. Sci. Med. 2021, 268, 113554. [Google Scholar] [CrossRef]
- Mas-Coma, S.; Jones, M.K.; Marty, A.M. COVID-19 and globalization. One Health 2020, 9, 100132. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, N.; Shad, M.Y.; Ulvi, O.; Khan, M.H.; Karamehic-Muratovic, A.; Nguyen, U.-S.D.; Baghbanzadeh, M.; Wardrup, R.; Aghamohammadi, N.; Cervantes, D.; et al. The impact of COVID-19 on globalization. One Health 2020, 11, 100180. [Google Scholar] [CrossRef]
- Schwab, N.; Berger, E.; Sanjayan, M.; Gough, M.; President, K.R. Rodriguez: Policy brief, COVID 19 Response and Recovery Recommendations for Policymakers, National Capitals Coalition. 2020. Available online: https://capitalscoalition.org/covid-19-response-recovery-nature-based-solutions-for-people-planet-prosperity/CEO (accessed on 14 June 2021).
- McNeely, J.A. Nature and COVID-19: The pandemic, the environment, and the way ahead. Ambio 2021, 50, 767–781. [Google Scholar] [CrossRef]
- Akinsorotan, O.A.; Olaniyi, O.E.; Adeyemi, A.A.; Olasunkanmi, A.H. Corona Virus Pandemic: Implication on Biodiversity Conservation. Front. Water 2021, 3, 635529. [Google Scholar] [CrossRef]
- Platto, S.; Zhou, J.; Wang, Y.; Wang, H.; Carafoli, E. Biodiversity loss and COVID-19 pandemic: The role of bats in the origin and the spreading of the disease. Biochem. Biophys. Res. Commun. 2021, 538, 2–13. [Google Scholar] [CrossRef]
- Levy, D.L. COVID-19 and Global Governance. J. Manag. Stud. 2021, 58, 562–566. [Google Scholar] [CrossRef]
- Environmental Justice Foundation|Viral Diseases from Wildlife in China: Could SARS Happen Again? Available online: https://ejfoundation.org/reports/viral-diseases-from-wildlife-in-china-could-sars-happen-again (accessed on 28 June 2021).
- Xu, R.-H.; He, J.-F.; Evans, M.R.; Peng, G.-W.; Field, H.E.; Yu, D.-W.; Lee, C.-K.; Luo, H.-M.; Lin, W.-S.; Lin, P.; et al. Epidemiologic Clues to SARS Origin in China. Emerg. Infect. Dis. 2004, 10, 1030–1037. [Google Scholar] [CrossRef]
- Middle East Respiratory Syndrome Coronavirus (MERS-CoV). Available online: https://www.who.int/health-topics/middle-east-respiratory-syndrome-coronavirus-mers#tab=tab_1 (accessed on 14 June 2021).
- Total Stimulus for the COVID-19 Crisis Already Triple That for the Entire 2008–2009 Recession|McKinsey & Company. Available online: https://www.mckinsey.com/featured-insights/coronavirus-leading-through-the-crisis/charting-the-path-to-the-next-normal/total-stimulus-for-the-covid-19-crisis-already-triple-that-for-the-entire-2008-09-recession# (accessed on 14 June 2021).
- Zoonotic Diseases|One Health|CDC. Available online: https://www.cdc.gov/onehealth/basics/zoonotic-diseases.html (accessed on 14 June 2021).
- Tarantola, A. Four Thousand Years of Concepts Relating to Rabies in Animals and Humans, Its Prevention and Its Cure. Trop. Med. Infect. Dis. 2017, 2, 5. [Google Scholar] [CrossRef] [Green Version]
- Pappaioanou, M.; Gomez, T.; Drenzek, C. New and Emerging Zoonoses. Emerg. Infect. Dis. 2004, 10, e28. [Google Scholar] [CrossRef]
- Sabin, N.S.; Calliope, A.S.; Simpson, S.V.; Arima, H.; Ito, H.; Nishimura, T.; Yamamoto, T. Implications of human activities for (re)emerging infectious diseases, including COVID-19. J. Physiol. Anthropol. 2020, 39, 1–12. [Google Scholar] [CrossRef]
- Smith, K.F.; Goldberg, M.; Rosenthal, S.; Carlson, L.; Chen, J.; Chen, C.; Ramachandran, S. Global rise in human infectious disease outbreaks. J. R. Soc. Interface 2014, 11, 20140950. [Google Scholar] [CrossRef] [PubMed]
- Steffen, W.; Rockström, J.; Richardson, K.; Lenton, T.M.; Folke, C.; Liverman, D.; Summerhayes, C.P.; Barnosky, A.D.; Cornell, S.E.; Crucifix, M.; et al. Trajectories of the Earth System in the Anthropocene. Proc. Natl. Acad. Sci. USA 2018, 115, 8252–8259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mizumoto, K.; Kagaya, K.; Chowell, G. Effect of a wet market on coronavirus disease (COVID-19) transmission dynamics in China, 2019–2020. Int. J. Infect. Dis. 2020, 97, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.-F.; Eaton, B.T. Bats, Civets and the Emergence of SARS. Curr. Top. Microbiol. Immunol. 2007, 315, 325–344. [Google Scholar] [CrossRef]
- Keatts, L.O.; Robards, M.; Olson, S.H.; Hueffer, K.; Insley, S.J.; Joly, D.O.; Kutz, S.; Lee, D.S.; Chetkiewicz, C.-L.B.; Lair, S.; et al. Implications of Zoonoses from Hunting and Use of Wildlife in North American Arctic and Boreal Biomes: Pandemic Potential, Monitoring, and Mitigation. Front. Public Health 2021, 9, 451. [Google Scholar] [CrossRef] [PubMed]
- National Research Council (US); Committee on Achieving Sustainable Global Capacity for Surveillance and Response to Emerging Diseases of Zoonotic Origin. Sustaining Global Surveillance and Response to Emerging Zoonotic Diseases; Keusch, G.T., Pappaioanou, M., Gonzalez, M.C., Eds.; National Academies Press (US): Washington, DC, USA, 2009. Available online: https://www.ncbi.nlm.nih.gov/books/NBK215318/ (accessed on 14 June 2021).
- Winkler, K.; Fuchs, R.; Rounsevell, M.; Herold, M. Global land use changes are four times greater than previously estimated. Nat. Commun. 2021, 12, 1–10. [Google Scholar] [CrossRef]
- Update: Outbreak of Nipah Virus—Malaysia and Singapore. 1999. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/00057012.htm (accessed on 14 June 2021).
- Rahman, M.; Chakraborty, A. Nipah virus outbreaks in Bangladesh: A deadly infectious disease. WHO South-East Asia J. Public Health 2012, 1, 208–212. [Google Scholar] [CrossRef] [Green Version]
- Chadha, M.S.; Comer, J.A.; Lowe, L.; Rota, P.A.; Rollin, P.; Bellini, W.J.; Ksiazek, T.G.; Mishra, A.C. Nipah Virus-associated Encephalitis Outbreak, Siliguri, India. Emerg. Infect. Dis. 2006, 12, 235–240. [Google Scholar] [CrossRef]
- Rulli, M.C.; D’Odorico, P.; Galli, N.; Hayman, D.T.S. Land-use change and the livestock revolution increase the risk of zoonotic coronavirus transmission from rhinolophid bats. Nat. Food 2021, 2, 409–416. [Google Scholar] [CrossRef]
- Living Planet Report 2020|Official Site|WWF. Available online: https://livingplanet.panda.org/en-us/ (accessed on 14 June 2021).
- Lecompte, E.; Fichet-Calvet, E.; Daffis, S.; Koulemou, K.; Sylla, O.; Kourouma, F.; Dore, A.; Soropogui, B.; Aniskin, V.; Allali, B.; et al. Mastomys natalensis and Lassa Fever, West Africa. Emerg. Infect. Dis. 2006, 12, 1971–1974. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.H.; Goba, A.; Chu, M.; Roth, C.; Healing, T.; Marx, A.; Fair, J.; Guttieri, M.C.; Ferro, P.; Imes, T.; et al. Mano River Union Lassa Fever Network. New opportunities for field research on the pathogenesis and treatment of Lassa fever. Antivir. Res. 2008, 78, 103–115. [Google Scholar] [CrossRef]
- Suk, J.E.; Van Cangh, T.; Beaute, J.; Bartels, C.; Tsolova, S.; Pharris, A.; Ciotti, M.; Semenza, J.C. The interconnected and cross-border nature of risks posed by infectious diseases. Glob. Health Action 2014, 10, 25287, Erratum in: 2015, 8, 27635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tynkkynen, K. Lontoon neljä 1800-luvun koleraepidemiaa [Four cholera epidemics in nineteenth-century London]. Hippokrates 1995, 12, 62–88. [Google Scholar] [PubMed]
- Alwan, A.; Armstrong, T.; Bettcher, D.; Branca, F.; Chisholm, D.; Ezzati, M.; Garfield, R.; MacLean, D.; Mendis, S.; Riley, L.; et al. Global Status Report on Noncommunicable Diseases; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Friel, S.; Bowen, K.; Campbell-Lendrum, D.; Frumkin, H.; McMichael, A.; Rasanathan, K. Climate Change, Noncommunicable Diseases, and Development: The Relationships and Common Policy Opportunities. Annu. Rev. Public Health 2011, 32, 133–147. [Google Scholar] [CrossRef] [Green Version]
- Roundtable on Environmental Health Sciences, Research, and Medicine; Board on Population Health and Public Health Practice; Institute of Medicine. Public Health Linkages with Sustainability: Workshop Summary; National Academies Press (US): Washington, DC, USA, 2013. Available online: https://www.ncbi.nlm.nih.gov/books/NBK202306/ (accessed on 14 June 2021).
- FAO. Technical Report: The State of Food and Agriculture 2019. Moving Forward on Food Loss and Waste Reduction. 2019. Available online: http://www.fao.org/3/ca6030en/ca6030en.pdf (accessed on 14 June 2021).
- Warren, C.J.; Sawyer, S.L. How host genetics dictates successful viral zoonosis. PLoS Biol. 2019, 17, e3000217. [Google Scholar] [CrossRef]
- Springmann, M.; Godfray, H.C.J.; Rayner, M.; Scarborough, P. Analysis and valuation of the health and climate change cobenefits of dietary change. Proc. Natl. Acad. Sci. USA 2016, 113, 4146–4151. [Google Scholar] [CrossRef] [Green Version]
- Why Earth Is Warming|UCAR Center for Science Education. Available online: https://scied.ucar.edu/learning-zone/how-climate-works/why-earth-warming (accessed on 14 June 2021).
- Global Climate Report—Annual 2020|2020 Year-To-Date Temperatures versus Previous Years|State of the Climate|National Centers for Environmental Information (NCEI). Available online: https://www.ncdc.noaa.gov/sotc/global/202013/supplemental/page-1 (accessed on 15 June 2021).
- Harvey, J.A.; Malcicka, M. Climate Change, Range Shifts and Multitrophic Interactions. Biodivers. Ecosyst. 2015, 1, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Ruscio, B.A.; Brubaker, M.; Glasser, J.; Hueston, W.; Hennessy, T.W. One Health—A strategy for resilience in a changing arctic. Int. J. Circumpolar Health 2015, 74. [Google Scholar] [CrossRef] [PubMed]
- Semenza, J.C.; Suk, J.E. Vector-borne diseases and climate change: A European perspective. FEMS Microbiol. Lett. 2018, 365, fnx244. [Google Scholar] [CrossRef]
- Githeko, A.K.; Lindsay, S.W.; Confalonieri, U.E.; Patz, J.A. Climate change and vector-borne diseases: A regional analysis. Bull. World Health Organ. 2000, 78, 1136–1147. [Google Scholar] [PubMed]
- Brooks, D.R.; Hoberg, E.P. How will global climate change affect parasite–host assemblages? Trends Parasitol. 2007, 23, 571–574. [Google Scholar] [CrossRef] [Green Version]
- Asadgol, Z.; Mohammadi, H.; Kermani, M.; Badirzadeh, A.; Gholami, M. The effect of climate change on cholera disease: The road ahead using artificial neural network. PLoS ONE 2019, 14, e0224813. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Climate Change: Regional Impacts|UCAR Center for Science Education. Available online: https://scied.ucar.edu/learning-zone/climate-change-impacts/regional (accessed on 15 June 2021).
- Colombo, E.; Masera, D.; Bologna, S. Renewable Energies to Promote Local Development. Renew. Energy Unleashing Sustain. Dev. 2013, 1, 3–25. [Google Scholar] [CrossRef]
- Ogwu, M.C. Towards Sustainable Development in Africa: The Challenge of Urbanization and Climate Change Adaptation. Geogr. Clim. Change Adapt. Urban Afr. 2019, 29–55. [Google Scholar] [CrossRef]
- Energy and Climate Change—European Environment Agency. Available online: https://www.eea.europa.eu/signals/signals-2017/articles/energy-and-climate-change (accessed on 7 August 2021).
- Walton, D.; Van Aalst, M. Climate-Related Extreme Weather Events and COVID-19 A First Look at the Number of People Affected by Intersecting Disasters: Assessment Report. 2020. Available online: https://reliefweb.int/report/world/climate-related-extreme-weather-events-and-covid-19-first-look-number-people-affected (accessed on 14 June 2021).
- Jan van Oldenborgh, G.; Krikken, F.; Lewis, S.; Leach, N.J.; Lehner, F.; Saunders, K.R.; van Weele, M.; Haustein, K.; Li, S.; Wallom, D. Attribution of the Australian bushfire risk to anthropogenic climate change. Hazards Earth Syst. Sci 2021, 21, 941–960. [Google Scholar] [CrossRef]
- Arriagada, N.B.; Palmer, A.J.; Bowman, D.M.; Morgan, G.; Jalaludin, B.B.; Johnston, F.H. Unprecedented smoke-related health burden associated with the 2019–20 bushfires in eastern Australia. Med. J. Aust. 2020, 213, 282. [Google Scholar] [CrossRef]
- The Impact of Coronavirus (COVID-19) on Forcibly Displaced Persons in Developing Countries. Available online: https://www.oecd.org/coronavirus/policy-responses/the-impact-of-coronavirus-covid-19-on-forcibly-displaced-persons-in-developing-countries-88ad26de/ (accessed on 15 June 2021).
- Impact of COVID-19 on Migrants and Refugees in the Arab Region, Technical Paper, United Nations Economic and Social Commission for Western Asia (ESCWA), the United Nations High Commissioner for Refugees (UNHCR) and the International Labour Organization (ILO). 2020. Available online: https://www.ilo.org/beirut/publications/WCMS_764756/lang--en/index.htm (accessed on 14 June 2021).
- A Virus That Respects no Borders: Protecting Refugees and Migrants during COVID-19. Available online: https://www.who.int/news-room/feature-stories/detail/a-virus-that-respects-no-borders-protecting-refugees-and-migrants-during-covid-19 (accessed on 15 June 2021).
- Rahman, M.R.; Islam, K. Massive diphtheria outbreak among Rohingya refugees: Lessons learnt. J. Travel Med. 2019, 26, 1–3. [Google Scholar] [CrossRef]
- Dureab, F.; Müller, O.; Jahn, A. Resurgence of diphtheria in Yemen due to population movement. J. Travel Med. 2018, 25, tay094. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Addressing Climate Change Post-Coronavirus|McKinsey. Available online: https://www.mckinsey.com/business-functions/sustainability/our-insights/addressing-climate-change-in-a-post-pandemic-world (accessed on 15 June 2021).
- Nadimpalli, M.L.; Chan, C.W.; Doron, S. Antibiotic resistance: A call to action to prevent the next epidemic of inequality. Nat. Med. 2021, 27, 187–188. [Google Scholar] [CrossRef]
- Pokharel, S.; Raut, S.; Adhikari, B. Tackling antimicrobial resistance in low-income and middle-income countries. BMJ Glob. Health 2019, 4, e002104. [Google Scholar] [CrossRef] [PubMed]
- Godman, B.; Egwuenu, A.; Haque, M.; Malande, O.; Schellack, N.; Kumar, S.; Saleem, Z.; Sneddon, J.; Hoxha, I.; Islam, S.; et al. Strategies to Improve Antimicrobial Utilization with a Special Focus on Developing Countries. Life 2021, 11, 528. [Google Scholar] [CrossRef]
- Langford, B.J.; So, M.; Raybardhan, S.; Leung, V.; Soucy, J.R.; Westwood, D.; Daneman, N.; MacFadden, D.R. Antibiotic prescribing in patients with COVID-19: Rapid review and meta-analysis. Clin. Microbiol. Infect. 2021, 27, 520–531. [Google Scholar] [CrossRef] [PubMed]
- Rawson, T.M.; Ming, D.; Ahmad, R.; Moore, L.S.P.; Holmes, A.H. Antimicrobial use, drug-resistant infections and COVID-19. Nat. Rev. Microbiol. 2020, 18, 409–410. [Google Scholar] [CrossRef]
- Burnham, J.P. Climate change and antibiotic resistance: A deadly combination. Ther. Adv. Infect. Dis. 2021, 8. [Google Scholar] [CrossRef]
- MacFadden, D.R.; McGough, S.F.; Fisman, D.; Santillana, M.; Brownstein, J.S. Antibiotic resistance increases with local temperature. Nat. Clim. Chang. 2018, 8, 510–514. [Google Scholar] [CrossRef]
- Arias-Andres, M.; Klümper, U.; Rojas-Jimenez, K.; Grossart, H. Microplastic pollution increases gene exchange in aquatic ecosystems. Environ. Pollut. 2018, 237, 253–261. [Google Scholar] [CrossRef] [Green Version]
- Ramirez, J.; Guarner, F.; Fernandez, L.B.; Maruy, A.; Sdepanian, V.L.; Cohen, H. Antibiotics as Major Disruptors of Gut Microbiota. Front. Cell. Infect. Microbiol. 2020, 10, 572912. [Google Scholar] [CrossRef]
- Clapp, M.; Aurora, N.; Herrera, L.; Bhatia, M.; Wilen, E.; Wakefield, S. Gut Microbiota’s Effect on Mental Health: The Gut-Brain Axis. Clin. Pract. 2017, 7, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Collignon, P.; Beggs, J.J.; Walsh, T.; Gandra, S.; Laxminarayan, R. Anthropological and socioeconomic factors contributing to global antimicrobial resistance: A univariate and multivariable analysis. Lancet Planet. Health 2018, 2, e398–e405. [Google Scholar] [CrossRef]
- Zinsstag, J.; Crump, L.; Schelling, E.; Hattendorf, J.; Maidane, Y.O.; Ali, K.O.; Muhummed, A.; Umer, A.; Aliyi, F.; Nooh, F.; et al. Climate change and One Health. FEMS Microbiol. Lett. 2018, 365, 85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garcia, S.N.; Osburn, B.I.; Jay-Russell, M.T. One Health for Food Safety, Food Security, and Sustainable Food Production. Front. Sustain. Food Syst. 2020, 4, 1. [Google Scholar] [CrossRef] [Green Version]
- Thumbi, S.M.; Njenga, M.K.; Marsh, T.L.; Noh, S.; Otiang, E.; Munyua, P.; Ochieng, L.; Ogola, E.; Yoder, J.; Audi, A.; et al. Linking Human Health and Livestock Health: A “One-Health” Platform for Integrated Analysis of Human Health, Livestock Health, and Economic Welfare in Livestock Dependent Communities. PLoS ONE 2015, 10, e0120761. [Google Scholar] [CrossRef] [Green Version]
- Musoke, D.; Ndejjo, R.; Atusingwize, E.; Halage, A.A. The role of environmental health in One Health: A Uganda perspective. One Health 2016, 2, 157–160. [Google Scholar] [CrossRef] [Green Version]
- Gardy, J.L.; Loman, N.J. Towards a genomics-informed, real-time, global pathogen surveillance system. Nat. Rev. Genet. 2018, 19, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Tanaka, K.; Matsuoka, S. Environmental and economic effectiveness of the Kyoto Protocol. PLoS ONE 2020, 15, e0236299. [Google Scholar] [CrossRef]
- World Health Organization. COP24 Special Report: Health and Climate Change. 2014. Available online: https://apps.who.int/iris/handle/10665/276405 (accessed on 15 June 2021).
- Amuasi, J.H.; Walzer, C.; Heymann, D.; Carabin, H.; Huong, L.T.; Haines, A.; Winkler, A.S. Calling for a COVID-19 One Health Research Coalition. Lancet 2020, 395, 1543–1544. [Google Scholar] [CrossRef]
- Queenan, K. Roadmap to a One Health agenda 2030. CAB Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2017, 12. [Google Scholar] [CrossRef] [Green Version]
- Ogden, N.H.; Wilson, J.R.U.; Richardson, D.M.; Hui, C.; Davies, S.; Kumschick, S.; Le Roux, J.; Measey, J.; Saul, W.-C.; Pulliam, J.R.C. Emerging infectious diseases and biological invasions: A call for a One Health collaboration in science and management. R. Soc. Open Sci. 2019, 6, 181577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ståhl, T. Health in All Policies: From rhetoric to implementation and evaluation—The Finnish experience. Scand. J. Public Heal. 2018, 46, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Implementing Health in All Policies|The Health Foundation. Available online: https://www.health.org.uk/publications/reports/implementing-health-in-all-policies (accessed on 23 August 2021).
- Bhaumik, S.; Dutta, R.; Sen, B. A call for increased collaboration between environmental health scientists and lawyers. Lancet Planet. Health 2019, 3, e60–e61. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).