3.2. Gas Composition
The complex composition of pyrolysis gases, their production volumes and heating values are displayed in
Table 2. The main sludge pyrolysis gas components are CO
2, CO, H
2, and CH
4 with their sum being almost 90 vol. %. The rest of the pyrolysis gases are composed of hydrocarbons, nitrogen, and other minor compounds.
The carbon dioxide content decreased sharply when the pyrolysis temperature rose from 400 to 800 °C. On the other hand, the contents of carbon monoxide and hydrogen increased significantly with the increase in temperature within this range. Methane increased first and decreased slightly at temperatures over 600 °C. These findings are, to a large extent, in agreement with the other literature results [
4,
5,
7,
13,
18].
Tomasi Morgano et al. [
4] consider CO
2 in the gas as a primary pyrolytic product [
19] and CO as a product of cracking reactions of oxygenated compounds, whereas Fan et al. [
13] attribute the formation of both CO and CO
2 to the decomposition of oxygenated groups, such as decarboxylation and decarbonylation reactions. Both pathways are probably responsible for the presence of CO
2. Moreover, the presence of CO
2 results in a steeper increase in the CO content at temperatures above 600 °C due to the Boudouard reaction [
16,
20] which may as well be partially catalyzed by the ash components of sludge-chars formed during pyrolysis.
The hydrogen content of the gas increased sharply when the process temperature increased from 400 to 600 °C and then the increase continued slightly up to 800 °C. Sun et al. [
18] found that the hydrogen content even decreased when pyrolysis temperature exceeded 600 °C, which is mainly attributed to the inhibitory effect of the reverse water–gas shift reaction when hydrogen reacts with carbon dioxide, and carbon monoxide and steam are formed.
Apart from the major gas components, other hydrocarbons are present in pyrolysis gases with their sum being up to 9 vol. % of the pyrolysis gas. It is clear (see
Table 2) that the content of the saturated hydrocarbons such as ethane, propane, butanes, pentanes and hexanes decreased with increasing pyrolysis temperature. On the other hand, we may observe the increase in the content of unsaturated hydrocarbons, preferably having double covalent bonds between adjacent carbon atoms, such as ethylene, propene, buta-1,3-diene. That suggests ongoing elimination reactions where hydrogen atoms are released to form hydrogen or water molecules. Nevertheless, their direct release from the sludge organic matter, which is very complex compared to conventional biomass sources, at higher temperatures, cannot be ruled out. It must as well be noted that there was a significant decline in the content of hydrocarbons (methane—C
6) when pyrolysis temperature increased from 700 to 800 °C, which is responsible for the decline in the heating value of the pyrolysis gas.
In addition to the above-mentioned gas compounds, a remarkable amount of nitrogen was analyzed in the pyrolysis gases when the process temperature exceeded 500 °C, and its content increased with further increases in the temperature. The primary gaseous nitrogen species present in the devolatilized pyrolysis gas are NH
3 (ammonia) and HCN (hydrogen cyanide) [
21,
22], however, some small amount of molecular nitrogen (N
2) is present in the gas as well [
23]. The increasing content of N
2 is most likely the result of the enhanced decomposition of NH
3 (and HCN) at higher temperatures catalyzed by metal oxides [
24] present in the ash of the sludge, and of the enhanced conversion of NH
3 and HCN by oxygen-mediated pathways at high temperatures [
23].
Sulfur is the other element of concern in the gas, mainly due to its oxidation to SO
2 during the combustion process. The main sulfur species analyzed in the gas were H
2S (hydrogen sulfide), COS (carbonyl sulfide) and CH
3SH (methanethiol) (
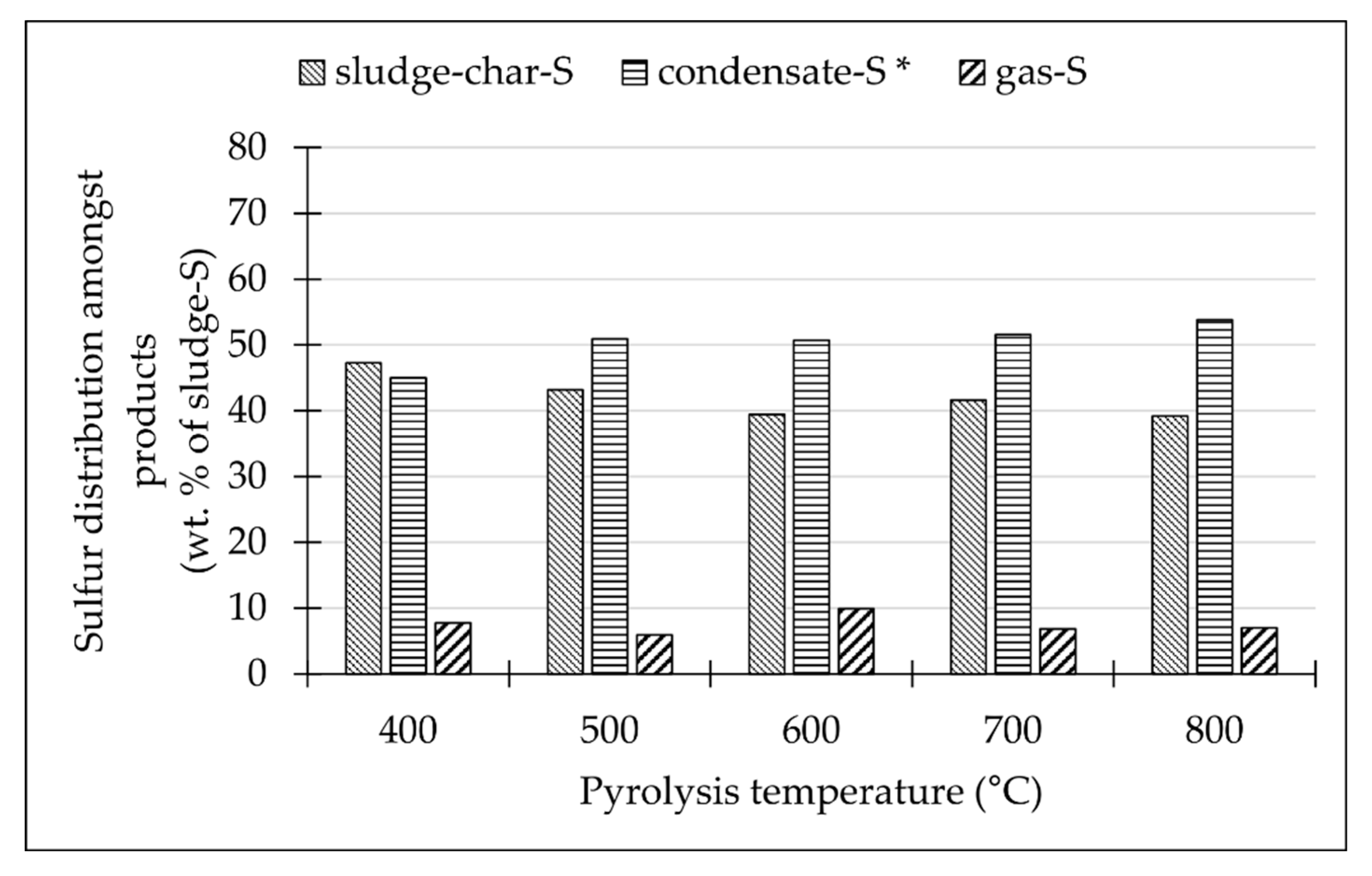
Table 3). Despite a slight increase in their content at a pyrolysis temperature of 600 °C, we may conclude that their content in the gas generally decreases when the pyrolysis temperature increases. Combined with the sulfur content in other pyrolysis products, we observed that the sulfur balance and distribution among pyrolysis products (
Figure 3) is relatively independent of any pyrolysis temperature above 500 °C [
25]. Therefore, the decreasing sulfur content is the result of its dilution in the increased gas volume as the S-species are mostly released from the sludge to the gas phase up to pyrolysis temperatures of 400–500 °C [
26,
27]. Zhang et al. [
26] observed an increase in S conversion to the gas phase during conventional demineralized sludge pyrolysis. However, when using CaO as the conditioner, the increase was only minimal. Therefore, the nature of the sludge (high ash and alkali metals content) must be considered, since the presence of alkali metals may help to preserve S in the sludge-char, preventing S release into gaseous phase and its oxidation into SO
2 when burning the gas and condensate [
28,
29].
3.3. Condensate Composition
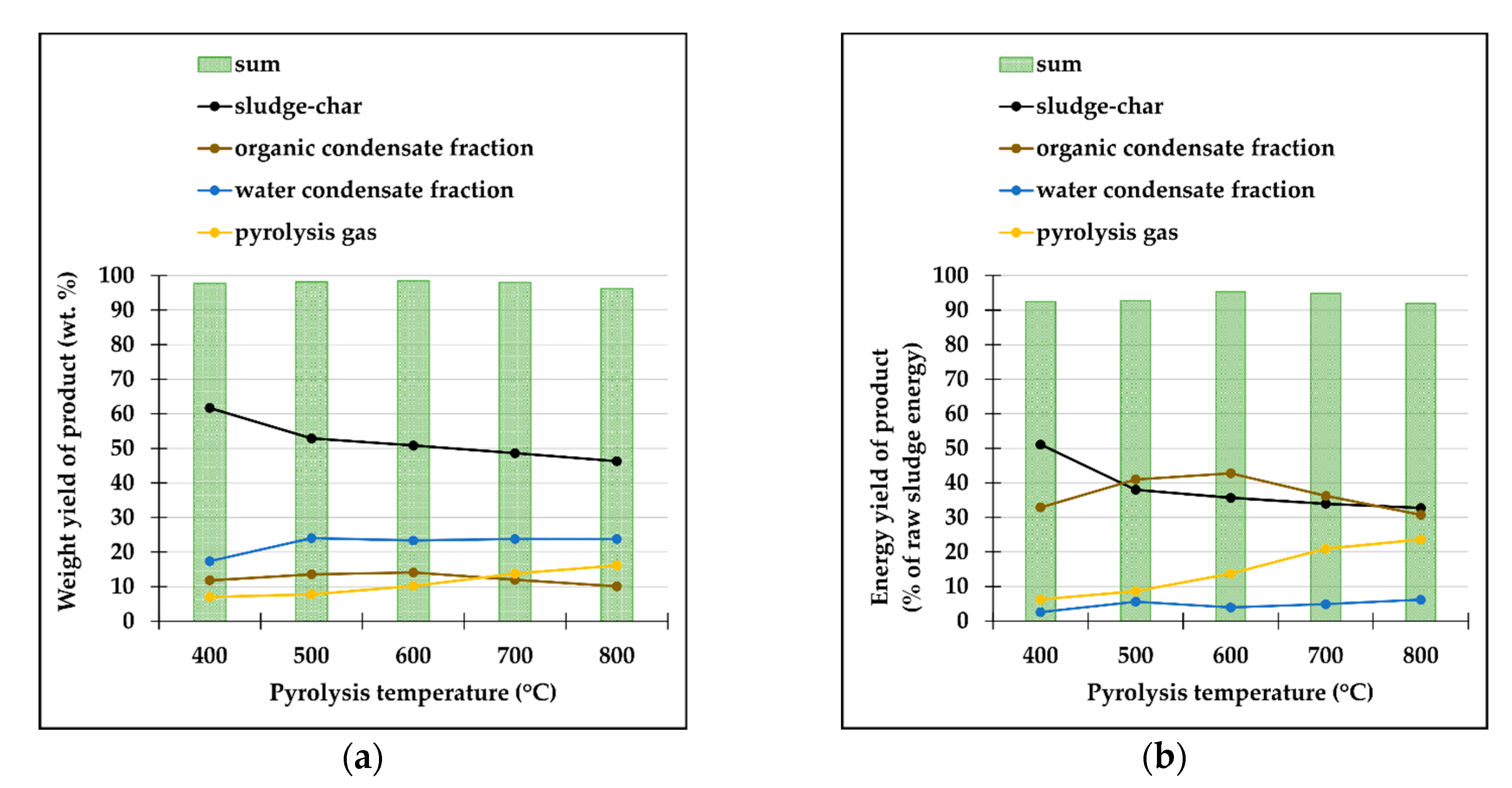
The vast majority of condensate (liquid pyrolysis product) was collected in the first impinger, and only that part was used for further analysis. The condensate was divided into two immiscible parts: the organic fraction (oil) and an inorganic fraction (water fraction containing some hydrosoluble compounds responsible for its partial HHV (
Figure 2b)) which were analyzed individually after their separation.
Bio-oils from the sludge pyrolysis have been widely analyzed to obtain their overall composition and other properties [
5,
12,
13,
18,
30,
31]. Monoaromatics, steroids and aliphatic, oxygenated and nitrogenated compounds [
13] contained in bio-oil can be divided into three groups according to their molecular weight: light, medium, and high [
12]. The increase in pyrolysis temperature and heating rate then results in a higher content of light compounds due to more intense cracking of compounds with higher molecular weight. We studied the elemental composition and heating value of both fractions of condensate (
Table 4 and
Table 5) which is relevant in case of the combustion to obtain energy for pyrolysis is the primary end-point of the condensate. There was an initial change in the composition of condensate when the pyrolysis temperature increased from 400 to 500 °C, resulting from the decomposition of sludge organic matter, followed by steeper sludge-char yield decrease. In contrast, a further increase in the temperature had an insignificant effect on the elemental composition of the condensate and its heating value, as the sludge organic matter was converted to gas rather than condensate, followed by decrease in condensate yield as the higher temperature promotes decomposition of primary pyrolysis products resulting in the “non-changing” composition, as neither diluting nor concentrating occur.