Evaluation of CO2 Storage in a Shale Gas Reservoir Compared to a Deep Saline Aquifer in the Ordos Basin of China
Abstract
1. Introduction
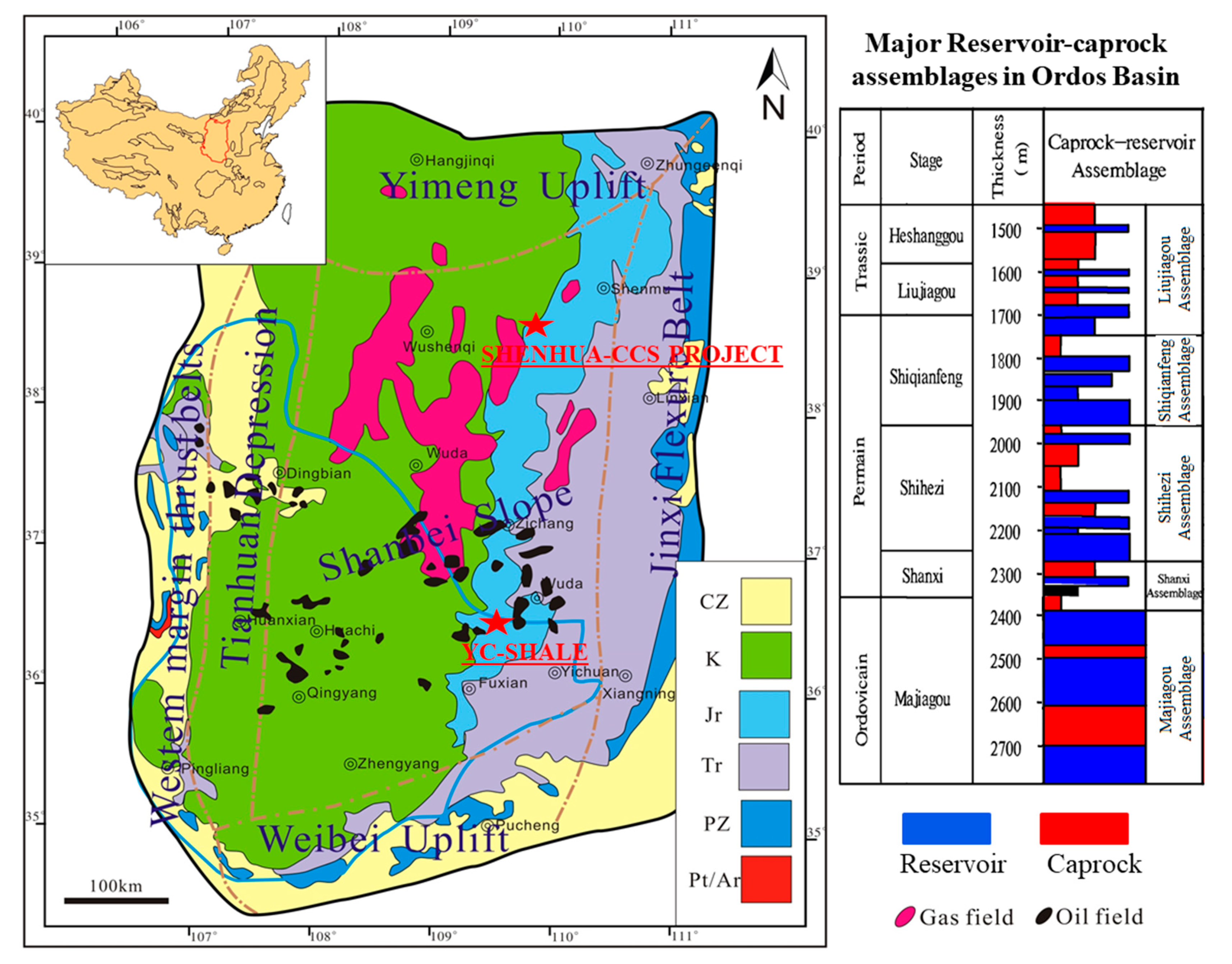
2. Geological Background
3. Material and Methods
3.1. Governing Equations for Different Trapping Mechanisms
3.1.1. Multi-Component Adsorption
3.1.2. Dissolution
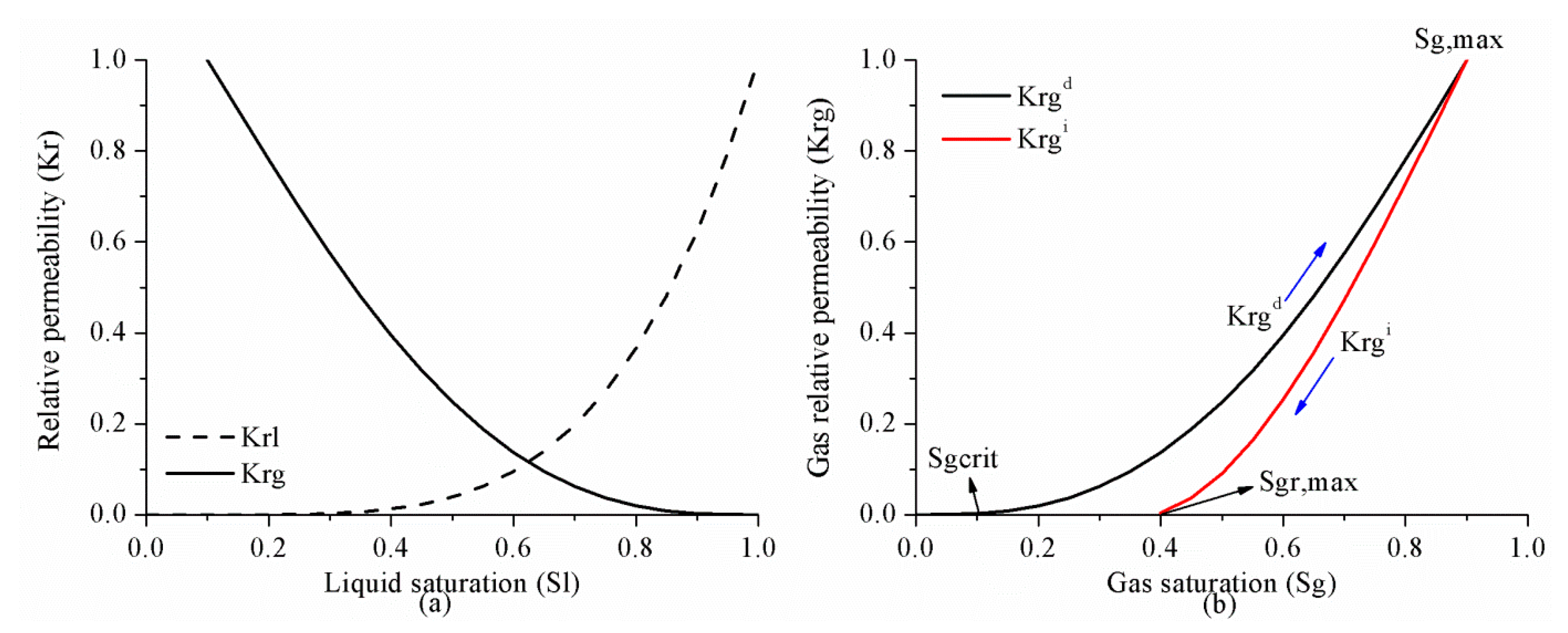
3.1.3. Trapping and Relative Permeability Hysteresis
3.1.4. CO2-Water-Rock Reactions
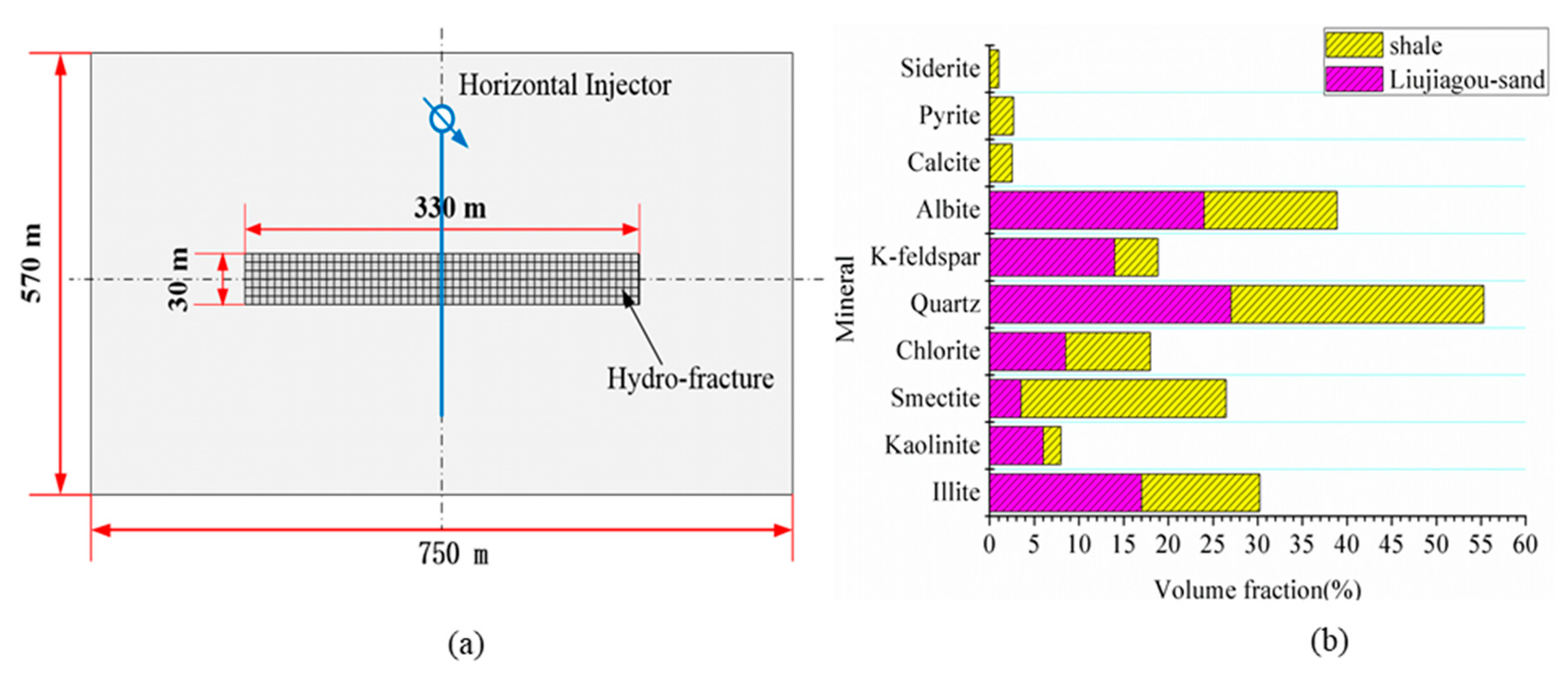
3.2. Model Description
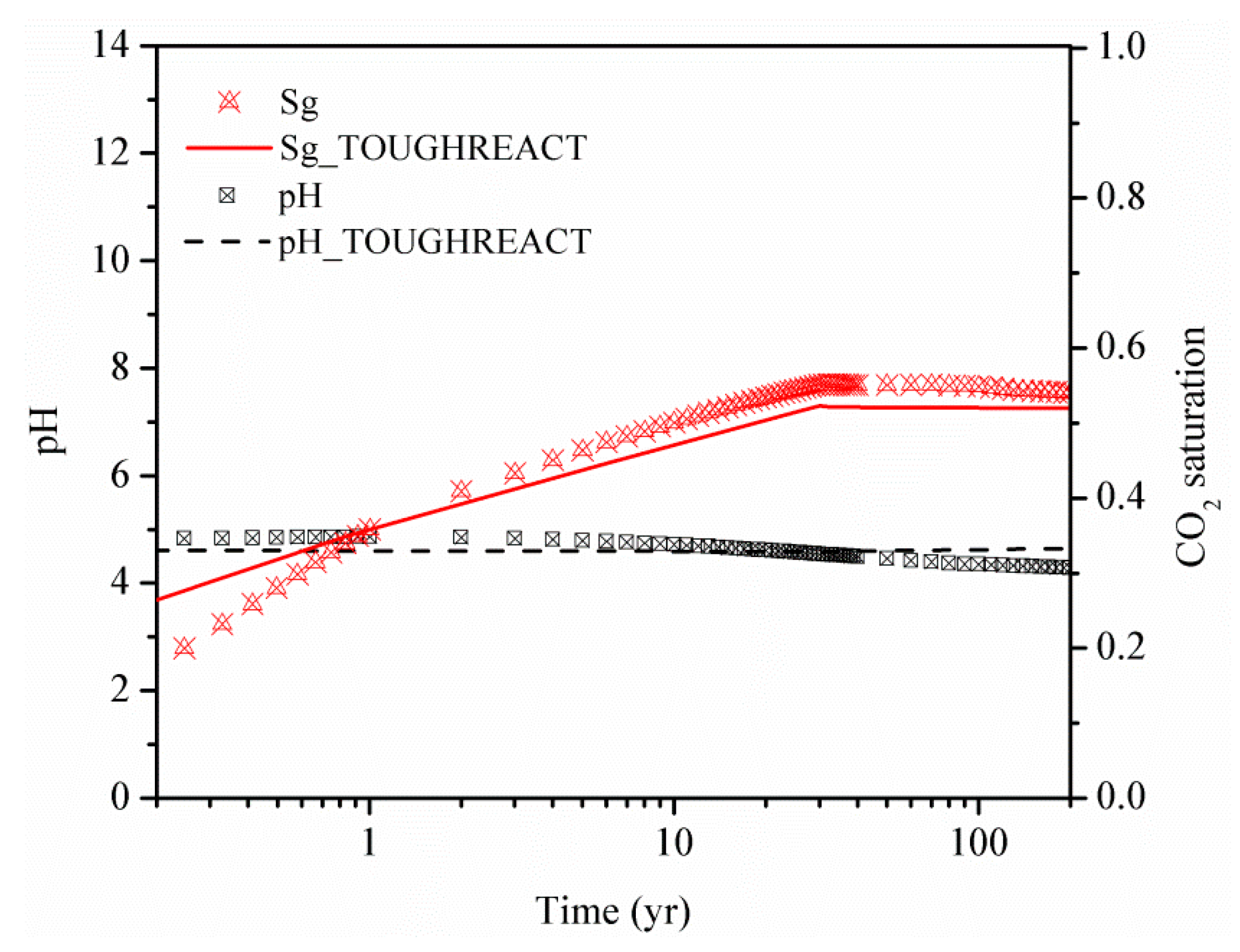
3.3. Simulation Cases and Validation
4. Results and Discussion
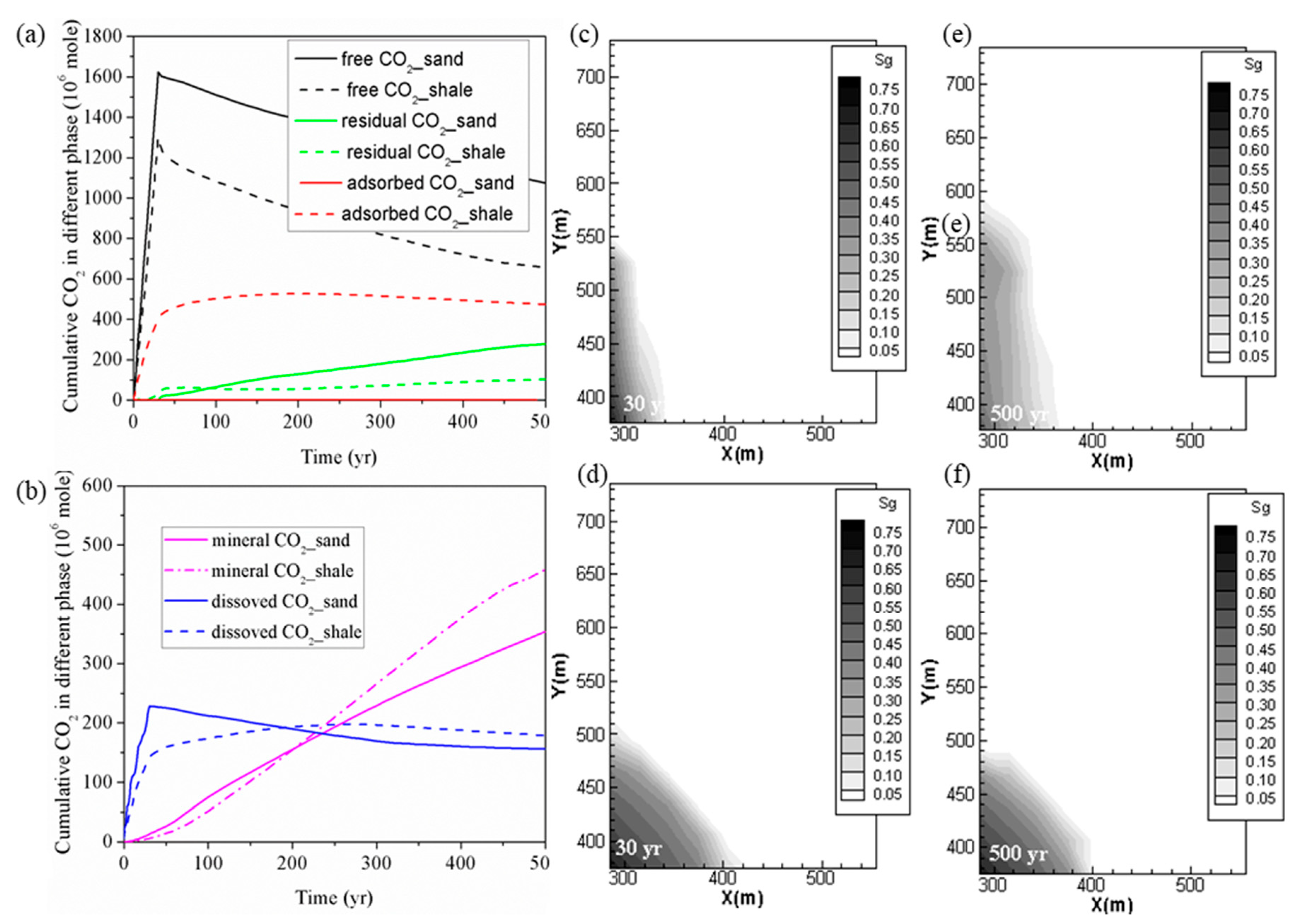
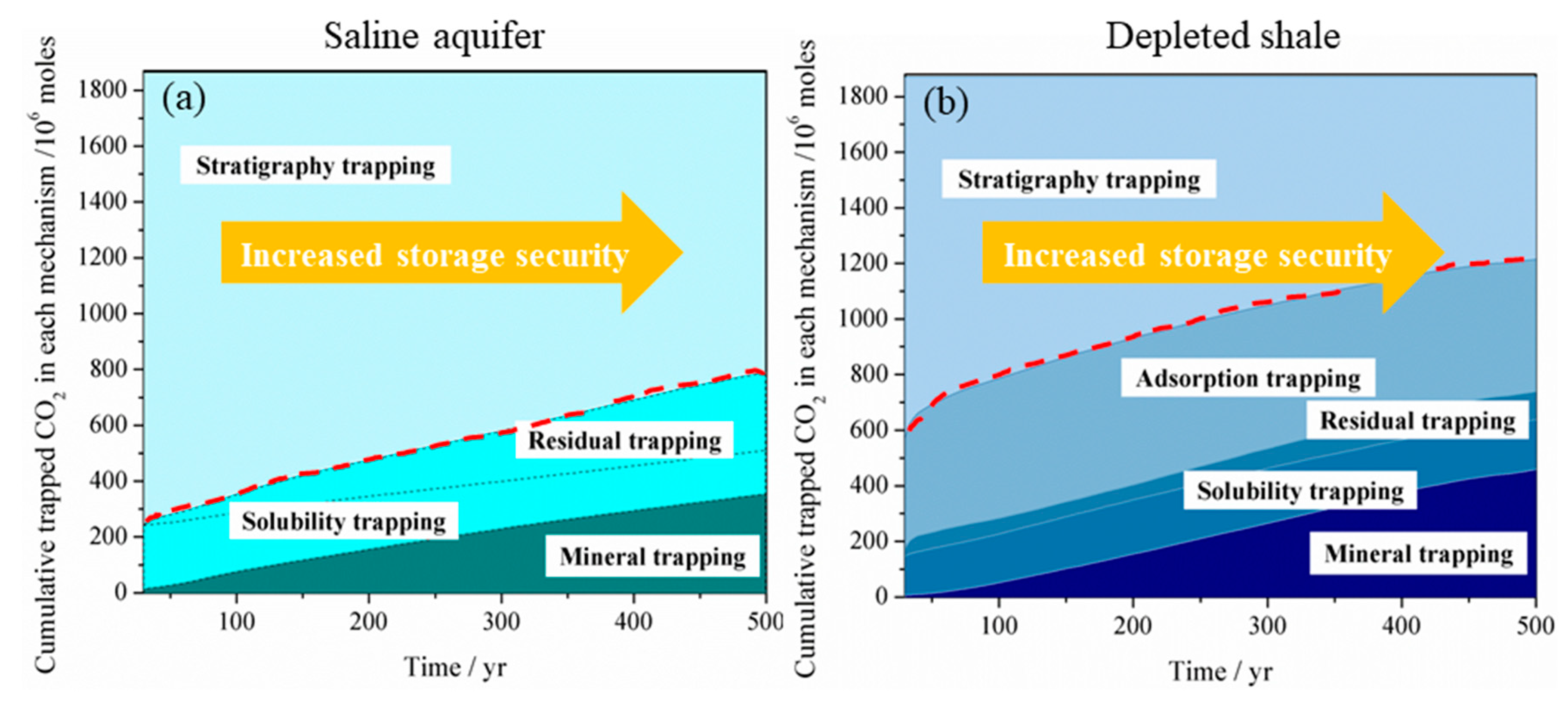
4.1. Comparison of CO2 Storage in Shale and Deep Saline Aquifer
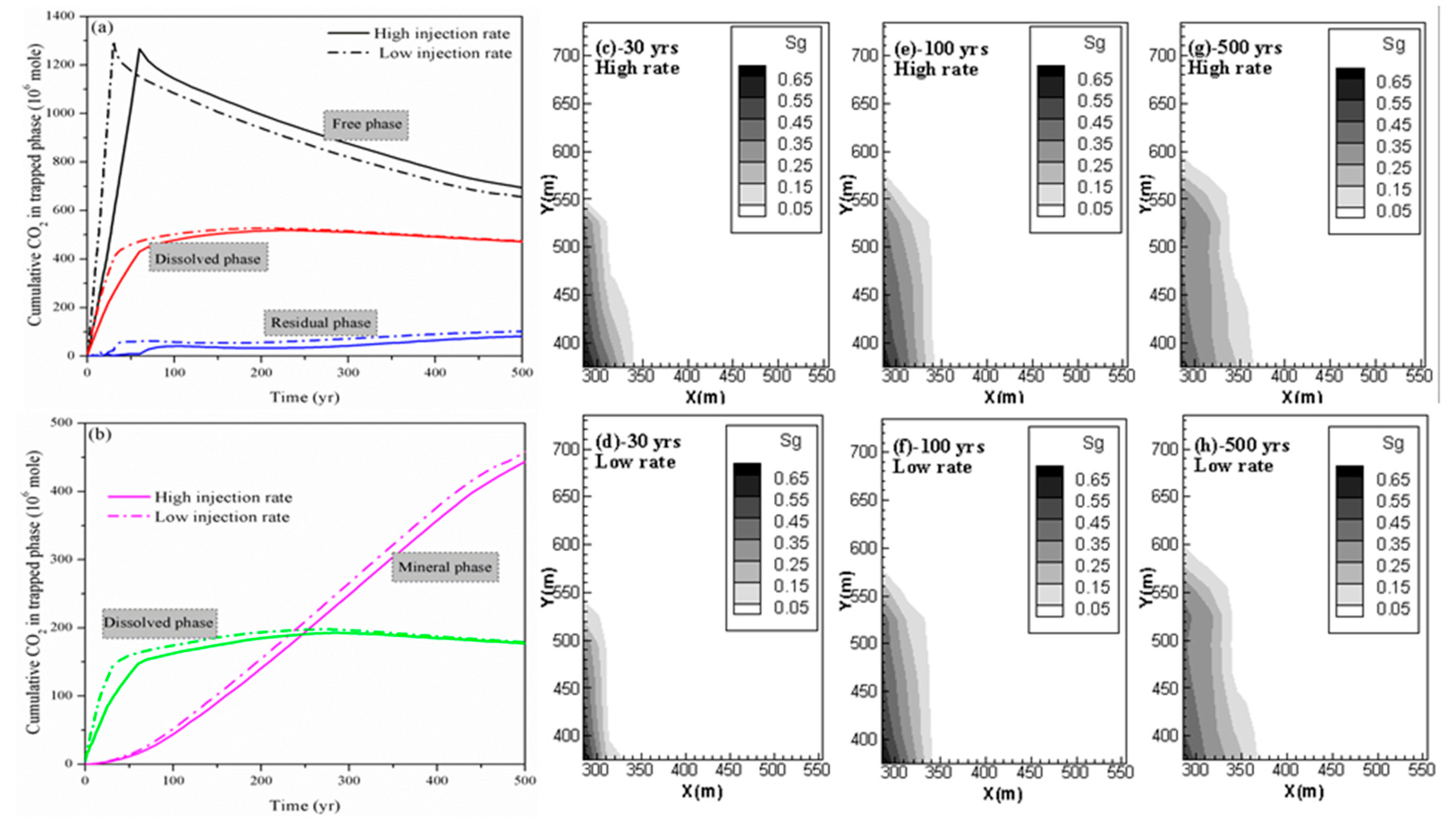
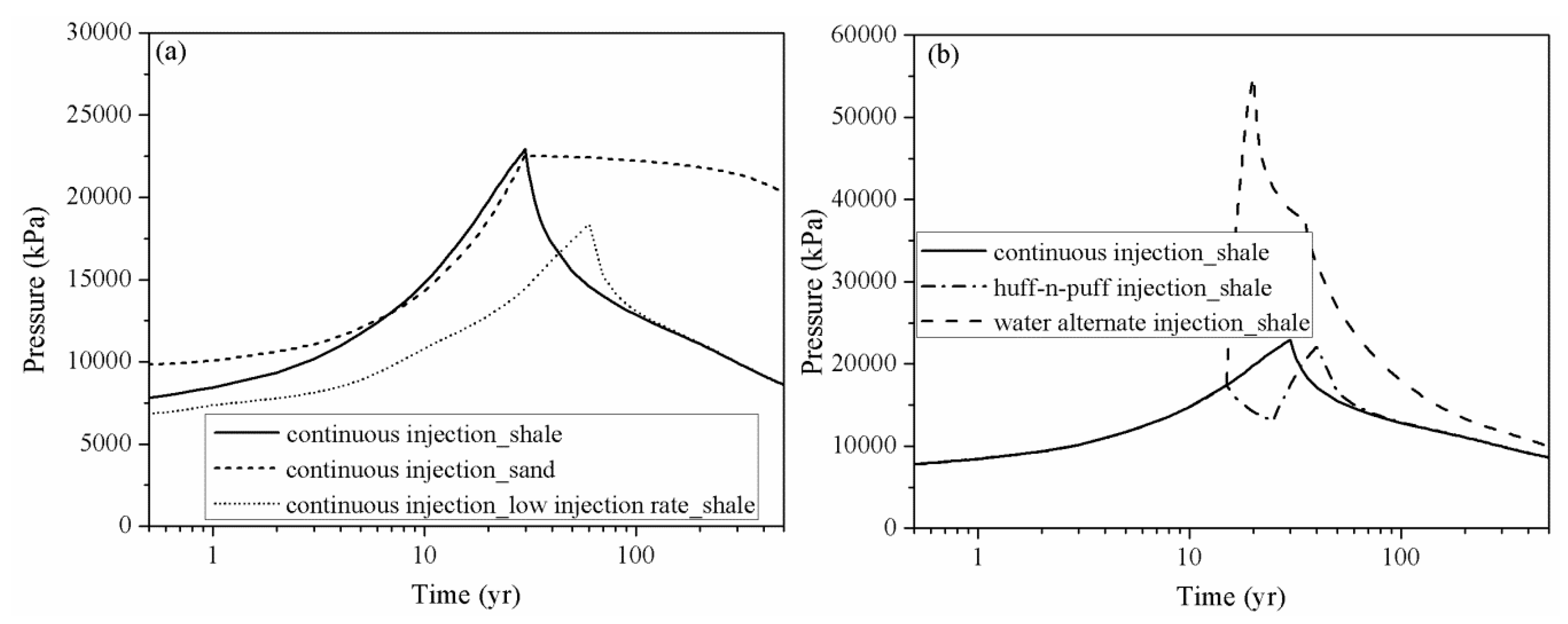
4.2. Effect of CO2 Injection Rate on CO2 Storage in Shale
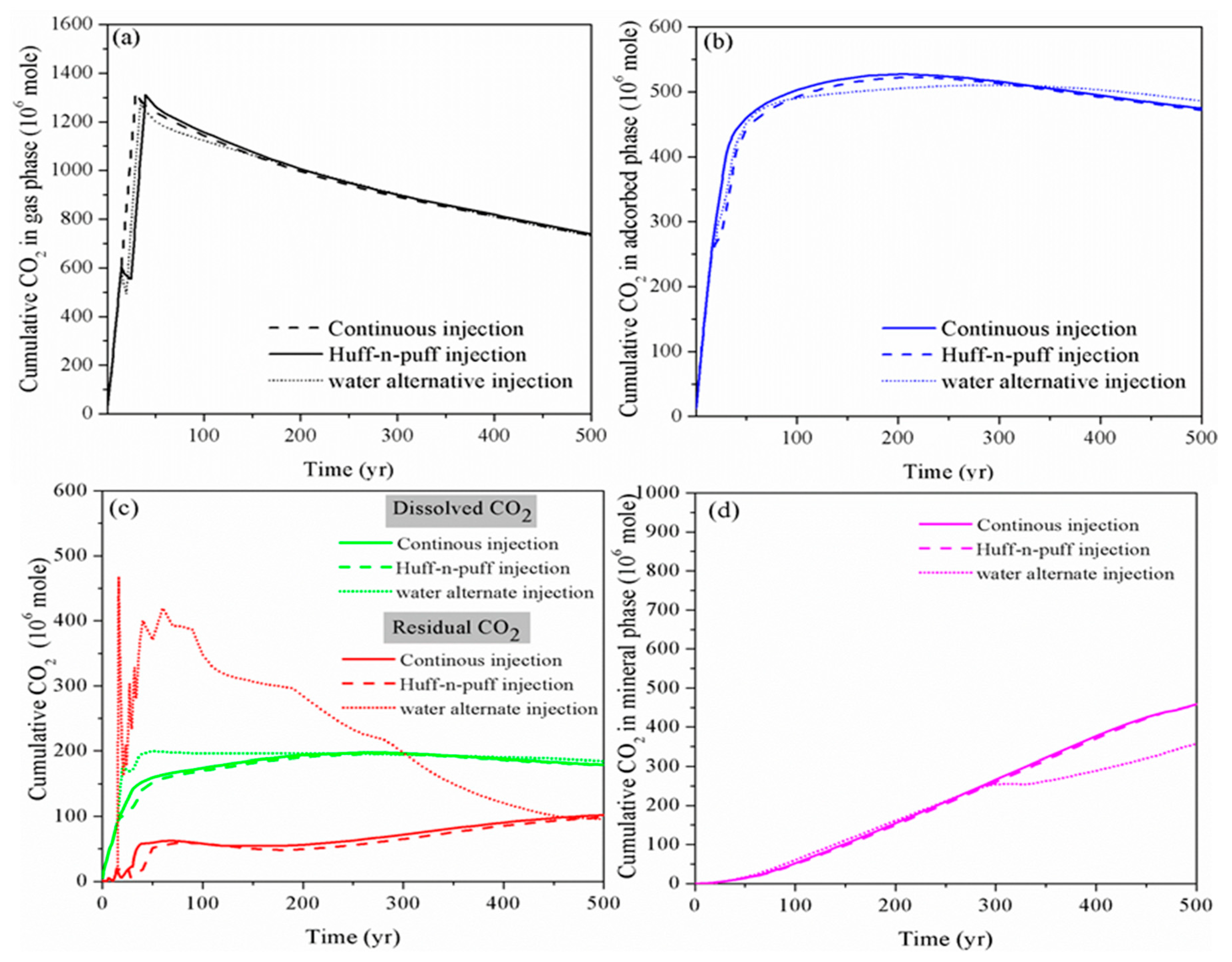
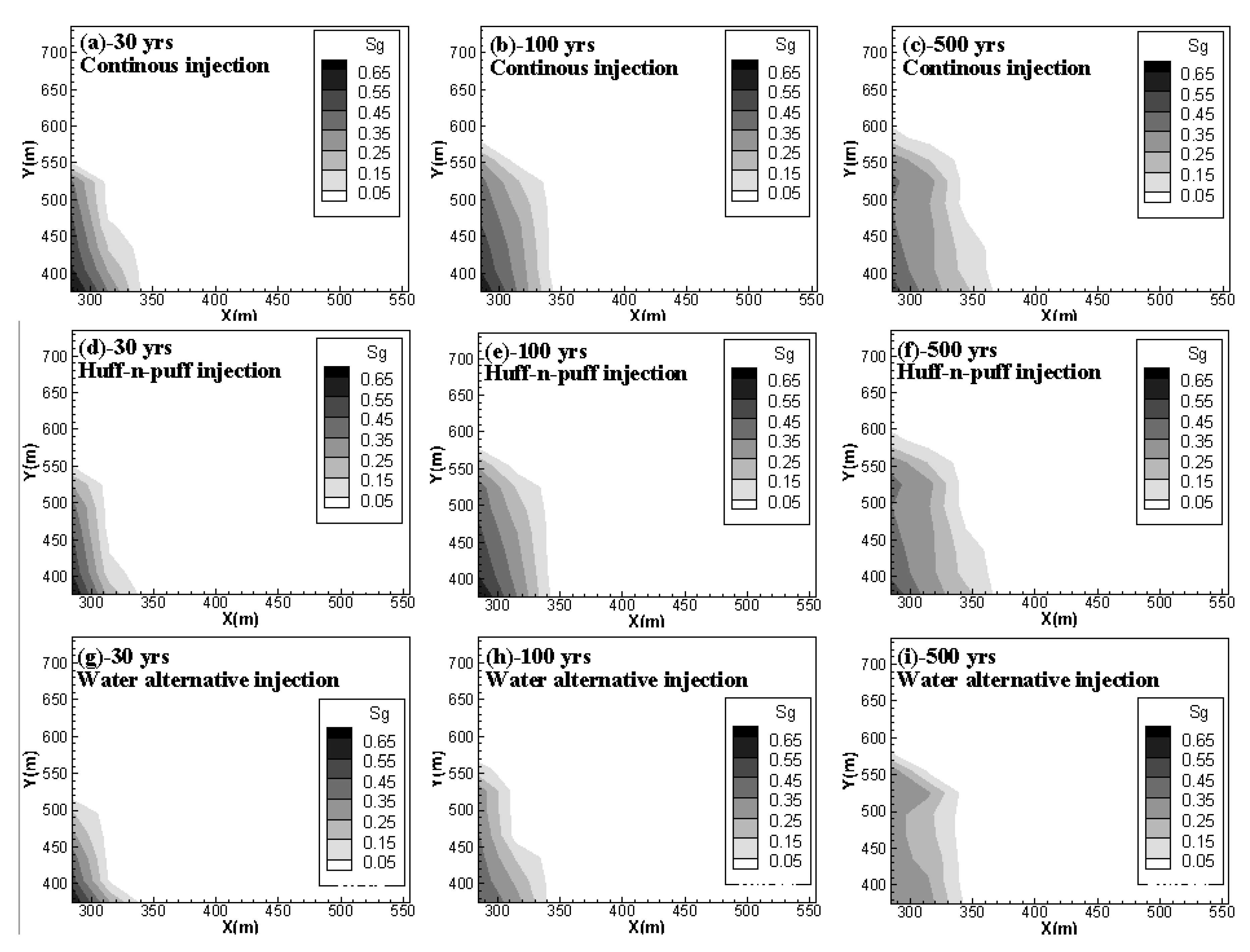
4.3. Effect of Huff-N-Puff Injection and Water Alternating Gas Injection
4.4. Pressure Perturbation Induced by CO2 Injection
4.5. Implication to CO2 Storage Safety and Stability
5. Conclusions
- From the point of view of CO2 phase transformation, CO2 storage in shale can be safer than in saline aquifer by trapping more CO2 in immobile phases, including adsorbed, residual, dissolution and mineral phase with lesser percentage remaining in free mobile phase in longer-term. Although, the saline aquifer has the advantage in trapping more CO2 in the residual, dissolution and mineral phase in the short term.
- The pressure perturbation induced by CO2 injection in the saline aquifer is longer lasting and generally larger than in the shale reservoir. The pressure build-up in shale can be rapidly released when CO2 injection is stopped.
- Although the water alternating injection scheme can significantly increase the dissolution and residual phase of CO2 in short to middle term, the pressure build-up caused by water injection is more drastic than other schemes. For the aim of increasing the fraction of immobile CO2 while maintaining a safe pressure-perturbation, the intermittent injection procedure with multiple slugs of huff-n-puff injection can be employed to replace the continuous CO2 injection. Within the allowable range of pressure increase, the CO2 injection rate can be maximized to increase the CO2 storage capacity and security in a shale reservoir.
Author Contributions
Funding
Conflicts of Interest
References
- Bachu, S. Sequestration of CO2 in geological media: Criteria and approach for site selection in response to climate change. Energy Convers. Manag. 2000, 41, 953–970. [Google Scholar] [CrossRef]
- Bachu, S.; Adams, J.J. Sequestration of CO2 in geological media in response to climate change: Capacity of deep saline aquifers to sequester CO2 in solution. Energy Convers. Manag. 2003, 44, 3151–3175. [Google Scholar] [CrossRef]
- Bachu, S.; Bonijoly, D.; Bradshaw, J.; Burruss, R.; Holloway, S.; Christensen, N.P.; Mathiassen, O.M. CO2 storage capacity estimation: Methodology and gaps. Int. J. Greenh. Gas Control 2007, 1, 430–443. [Google Scholar] [CrossRef]
- Busch, A.; Alles, S.; Gensterblum, Y.; Prinz, D.; Dewhurst, D.N.; Raven, M.D.; Stanjek, H.; Krooss, B.M. Carbon dioxide storage potential of shales. Int. J. Greenh. Gas Control 2008, 2, 297–308. [Google Scholar] [CrossRef]
- Nuttal, B.C.; Eble, C.; Bustin, R.M.; Drahovzal, J.A. Analysis of Devonian black shales in kentucky for potential carbon dioxide sequestration and enhanced natural gas production. Greenh. Gas Control Technol. 2005, 7, 2225–2228. [Google Scholar]
- Heller, R.; Zoback, M. Adsorption of methane and carbon dioxide on gas shale and pure mineral samples. J. Unconv. Oil Gas Resour. 2014, 8, 14–24. [Google Scholar] [CrossRef]
- Kang, S.M.; Fathi, E.; Ambrose, R.J.; Akkutlu, I.Y.; Sigal, R.F. Carbon dioxide storage capacity of organic-rich shales. Spe J. 2011, 16, 842–855. [Google Scholar] [CrossRef]
- Zhang, J. Study of Continental Shale Gas Adsorption and Desorption Effect in Fuxian Area of Ordos Basin; Southwest Petroleum University: Chengdu, China, 2013. [Google Scholar]
- Schepers, K.C.; Nuttall, B.; Oudinot, A.Y.; Gonzalez, R. Reservoir Modeling and simulation of the Devonian Gas Shale of Eastern Kentrucky for Enhanced Gas Recovery and CO2 Storage. Soc. Pet. Eng. 2009. [Google Scholar] [CrossRef]
- Liu, F.; Ellett, K.; Xiao, Y.; Rupp, J.A. Assessing the feasibility of CO2 storage in the New Albany Shale (Devonian–Mississippian) with potential enhanced gas recovery using reservoir simulation. Int. J. Greenh. Gas Control 2013, 17, 111–126. [Google Scholar] [CrossRef]
- Tao, Z.; Clarens, A. Estimating the carbon sequestration capacity of shale formations using methane production rates. Environ. Sci. Technol. 2013, 47, 11318–11325. [Google Scholar] [CrossRef]
- Liu, D.; Li, Y.; Agarwal, R.K. Numerical simulation of long-term storage of CO2 in Yanchang shale reservoir of the Ordos basin in China. Chem. Geol. 2016, 440, 288–305. [Google Scholar] [CrossRef]
- Benson, S.; Cook, P.; Anderson, J.; Bachu, S.; Nimir, H.B.; Basu, B. Capter 5—Underground geological storage. In IPCC Special Report on CO2 Capture and Sequestration; Cambridge University Press: New York, NY, USA, 2005. [Google Scholar]
- Hesse, M.A.; Orr, F.M.; Tchelepi, H.A. Gravity currents with residual trapping. J. Fluid Mech. 2008, 611, 35–60. [Google Scholar] [CrossRef]
- Juanes, R.; Spiteri, E.J.; Orr, F.M.; Blunt, M.J. Impact of relative permeability hysteresis on geological CO2 storage. Water Resour. Res. 2006, 42, 395–397. [Google Scholar] [CrossRef]
- Saadatpoor, E.; Bryant, S.L.; Sepehrnoori, K. New Trapping Mechanism in Carbon Sequestration. Transp. Porous Media 2010, 82, 3–17. [Google Scholar] [CrossRef]
- Kim, T.H.; Cho, J.; Lee, K.S. Evaluation of CO2 injection in shale gas reservoirs with multi-component transport and geomechanical effects. Appl. Energy 2017, 190, 1195–1206. [Google Scholar] [CrossRef]
- Liu, D.; Agarwal, R.; Li, Y. Numerical simulation and optimization of CO2 enhanced shale gas recovery using a genetic algorithm. J. Clean. Prod. 2017, 164, 1093–1104. [Google Scholar] [CrossRef]
- Xu, X.; Chen, S.; Zhang, D. Convective stability analysis of the long-term storage of carbon dioxide in deep saline aquifer. Adv. Water Resour. 2006, 29, 397–407. [Google Scholar] [CrossRef]
- Green, C.P.; Ennis-King, J. Steady dissolution rate due to convective mixing in anisotropic porous media. Adv. Water Resour. 2014, 73, 65–73. [Google Scholar] [CrossRef]
- De Paoli, M.; Zonta, F.; Soldati, A. Dissolution in anisotropic porous media: Modelling convection regimes from onset to shutdown. Phys. Fluids 2017, 29, 026601. [Google Scholar] [CrossRef]
- Li, X.; Li, Q.; Bai, B.; Wei, N.; Yuan, W. The geomechanics of Shenhua carbon dioxide capture and storage (CCS) demonstration project in Ordos Basin, China. J. Rock Mech. Geotech. Eng. 2016, 8, 948–966. [Google Scholar] [CrossRef]
- Jiao, Z.; Surdam, R.C.; Zhou, L.; Stauffer, P.H.; Luo, T. A feasibility study of geological CO2 sequestration in the Ordos Basin, China. Energy Procedia 2011, 4, 5982–5989. [Google Scholar] [CrossRef]
- Liu, D.; Li, Y.; Song, S.; Agarwal, R. Simulation and analysis of lithology heterogeneity on CO2 geological sequestration in deep saline aquifer: A case study of the Ordos Basin. Environ. Earth Sci. 2016, 75, 1–13. [Google Scholar] [CrossRef]
- Blunt, M.J. An Empirical Model for Three-Phase Relative Permeability. Spe J. 1999, 5, 435–445. [Google Scholar] [CrossRef]
- Carlson, F.M. Simulation of Relative Permeability Hysteresis to the Nonwetting Phase. In Proceedings of the Spe Technical Conference and Exhibition, San Antonio, TX, USA, 4–7 October 1981. [Google Scholar]
- Land, C.S. Calculation of Imbibition Relative Permeability for Two and Three-Phase Flow from Rock Properties. Soc. Pet. Eng. J. 1968, 8, 149–156. [Google Scholar] [CrossRef]
- Land, C.S. Comparison of Calculated with Experimental Imbibition Relative Permeability. Soc. Pet. Eng. J. 1971, 11, 419–425. [Google Scholar] [CrossRef]
- Lenhard, R.J.; Oostrom, M. A Parametric Model for Predicting Relative Permeability-Saturation-Capillary Pressure Relationships of Oil–Water Systems in Porous Media with Mixed Wettability. Transp. Porous Media 1998, 31, 109–131. [Google Scholar] [CrossRef]
- Zhang, W.; Li, Y.; Xu, T. Long-term variations of CO2 trapped in different mechanisms in deep saline formations: A case study of the Songliao Basin, China. Int. J. Greenh. Gas Control 2009, 3, 161–180. [Google Scholar] [CrossRef]
- Liu, D.; Agarwal, R.; Li, Y.; Yang, S. Reactive transport modeling of mineral carbonation in unaltered and altered basalts during CO2 sequestration. Int. J. Greenh. Gas Control 2019, 85, 109–210. [Google Scholar] [CrossRef]
- Bethke, C.M. Geochemical Reaction Modeling; Oxford University Press: New York, NY, USA, 1996. [Google Scholar]
- Nghiem, L.; Sammon, P.; Grabenstetter, J.; Ohkuma, H. Modeling CO2 storage in aquifers with a fully-coupled geochemical EOS compositional simulator. Soc. Pet. Eng. 2004. [Google Scholar] [CrossRef]
- Duan, H.; Fang, H.; Zhao, C.; Bai, J.; Feng, Q. Study on adaptability of shale gas network fracturing in Fuxian, Ordos Basin. Sci. Technol. Eng. 2013, 13, 8434–8439. [Google Scholar]
- Vilarrasa, V.; Bolster, D.; Dentz, M.; Olivella, S.; Carrera, J. Effects of CO2 compressibility on CO2 storage in deep saline aquifers. Transp. Porous Media 2010, 85, 619–639. [Google Scholar] [CrossRef]
- Yang, G.; Li, Y.; Atrens, A.; Liu, D.; Wang, Y.; Jia, L.; Lu, Y. Reactive transport modeling of long-term CO2 sequestration mechanisms at the Shenhua CCS demonstration project, China. J. Earth Sci. 2017, 28, 457–472. [Google Scholar] [CrossRef]
- Gaus, I.; Azaroual, M.; Czernichowski-Lauriol, I. Reactive transport modelling of the impact of CO2 injection on the clayey cap rock at Sleipner (North Sea). Chem. Geol. 2005, 217, 319–337. [Google Scholar] [CrossRef]
- Gallo, Y.L.; Trenty, L.; Michel, A.; Vidal-Gilbert, S.; Parra, T. Long-Term Flow Simulations of CO2 Storage in Saline Aquifer. In Proceedings of the GHGT8 Conference, Trondheim, Norway, 18 June 2006. [Google Scholar]
- Carroll, S.A.; Mcnab, W.W.; Torres, S.C. Experimental Study of Cement—Sandstone/Shale—Brine—CO2 Interactions. Geochem. Trans. 2011, 12, 1–19. [Google Scholar] [CrossRef]
- Peng, D.; Robinson, D.B. A new two-constant equation of state. Ind. Eng. Chem. Fundam. 1976, 15, 59–64. [Google Scholar] [CrossRef]
- Jossi, J.A.; Stiel, L.I.; Thodos, G. The viscosity of pure substances in the dense gaseous and liquid phases. AIChe J. 1962, 8, 59–63. [Google Scholar] [CrossRef]
- Xu, T.; Apps, J.A.; Pruess, K. Reactive geochemical transport simulation to study mineral trapping for CO2 disposal in deep arenaceous formations. J. Geophys. Res. Solid Earth 2003, 108, B2. [Google Scholar] [CrossRef]
- Strapoc, D.; Mastalerz, M.; Schimmelmann, A.; Drobniak, A.; Hasenmueller, N.R. Geochemical constraints on the origin and volume of gas in the New Albany Shale (Devonian–Mississippian), eastern Illinois Basin. AAPG Bull. 2010, 94, 1713–1740. [Google Scholar] [CrossRef]
- Vermylen, J.P. Geomechanical Studies of the Barnett Shale; Stanford University: Stanford, CA, USA, 2011. [Google Scholar]
- Xu, R.; Rong, L.; Jin, M.; Di, H.; Jiang, P. Effect of mineral dissolution/precipitation and CO2 exsolution on CO2 transport in Geological Carbon Storage. Acc. Chem. Res. 2017, 50, 2056–2066. [Google Scholar] [CrossRef]
- Birkholzer, J.; Zhou, Q.; Tsang, C. Large-scale impact of CO2 storage in deep saline aquifers: A sensitivity study on pressure response in stratified systems. Int. J. Greenh. Gas Control 2009, 3, 181–194. [Google Scholar] [CrossRef]
- Liu, D.; Agarwal, R.; Li, Y. Numerical simulation and optimization of CO2-enhanced water recovery by employing a genetic algorithm. J. Clean. Prod. 2016, 133, 994–1007. [Google Scholar] [CrossRef]
| Properties | YC Shale | LJG Sand | |
|---|---|---|---|
| Matrix | Fracture | ||
| Pressure | |||
| Temperature/(°C) | 34.15 a | 56 c | |
| Porosity/(%) | 10 a | 9.3 c | |
| Gas Diffusivity/(m2/s) | 1.00 × 10−9 b | 1.00 × 10−9 b | |
| Rock density/(kg/m3) | 2249 a | 2249 a | |
| Permeability/(mD) | 2.53 × 10−4 a | 3.300 × 10−3 a | 5.615 c |
| Salinity(%) | 3 | ||
| TOC/(%) | 5 a | / | |
| Compressibility/(1/Pa) | 4.5 × 10−10 c | 4.5 × 10−9 c | 2.5 × 10−9 d |
| Maximal adsorbed gas(CO2) /(cm3/g) | 1.258 a | / | / |
| Langmuir adsorption constant(CO2) /(1/Pa) | 1.295 × 10−7 a | / | / |
| Maximal adsorbed gas(CH4) /(cm3/g) | 0.224 a | / | / |
| Langmuir adsorption constant(CH4) /(1/Pa) | 6.702 × 10−8 a | / | / |
| Case Description | Description | Lithology | |||
|---|---|---|---|---|---|
| Gas Adsorption | CO2 Injection Rate/(m3/day) * | CO2 Injection Period /(year) | CO2 Injection Scheme | ||
| Case1 | / | 4000 | 30 | Continuous injection | LJG sand |
| Case2 | 4000 | 30 | Continuous injection | YC shale | |
| Case3 | 2000 | 60 | Continuous injection | YC shale | |
| Case4 | 4000 | 30 | Huff-n-puff injection | YC shale | |
| Case5 | 4000 | 30 | Water alternating gas injection | YC shale | |
| Cumulative CO2 Trapped under Different Mechanisms/(106 moles) | |||||
|---|---|---|---|---|---|
| Case | Hydrodynamic Trapping | Adsorbed Trapping | Residual Trapping | Dissolution Trapping | Mineral Trapping |
| Case2 | 656.59 | 474.66 | 102.03 | 179.12 | 458.06 |
| Case3 | 695.15 | 472.18 | 82.11 | 177.39 | 443.53 |
| Deviation | 5.87% | −0.52% | −19.52% | −0.97% | 3.17% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, D.; Li, Y.; Agarwal, R. Evaluation of CO2 Storage in a Shale Gas Reservoir Compared to a Deep Saline Aquifer in the Ordos Basin of China. Energies 2020, 13, 3397. https://doi.org/10.3390/en13133397
Liu D, Li Y, Agarwal R. Evaluation of CO2 Storage in a Shale Gas Reservoir Compared to a Deep Saline Aquifer in the Ordos Basin of China. Energies. 2020; 13(13):3397. https://doi.org/10.3390/en13133397
Chicago/Turabian StyleLiu, Danqing, Yilian Li, and Ramesh Agarwal. 2020. "Evaluation of CO2 Storage in a Shale Gas Reservoir Compared to a Deep Saline Aquifer in the Ordos Basin of China" Energies 13, no. 13: 3397. https://doi.org/10.3390/en13133397
APA StyleLiu, D., Li, Y., & Agarwal, R. (2020). Evaluation of CO2 Storage in a Shale Gas Reservoir Compared to a Deep Saline Aquifer in the Ordos Basin of China. Energies, 13(13), 3397. https://doi.org/10.3390/en13133397





