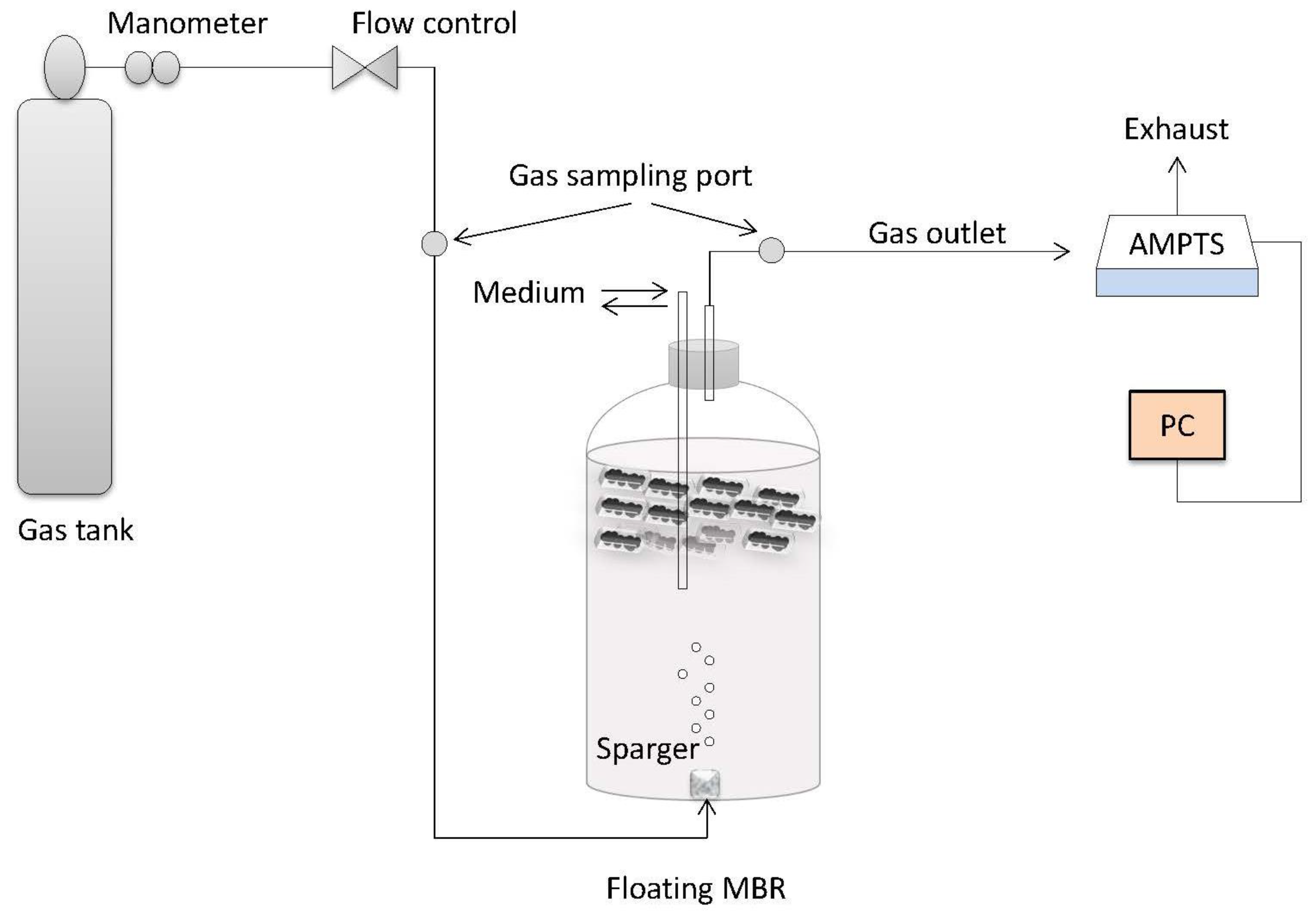
A main challenge during syngas fermentation is the low gas-to-liquid mass transfer, which can be improved by increasing the gas hold-up inside the liquid medium of bioreactors. For this purpose, anaerobic cells were encased in membrane sachets which were then heat-sealed and immersed inside a bubble column bioreactor. During the fermentation process, syngas was introduced to the bottom of the bioreactor with a sparger and the bubble ascent of syngas components was delayed because of the membrane bed, which operated as a mechanical barrier. This delay increased the gas hold-up in the liquid phase. Consequently, the syngas components diffused in the liquid phase and through the 0.1 μm membrane pores. The total interfacial area for gas mass transfer (α) increases with higher gas hold-up and smaller bubble size. Thus, higher mass transfer rates are achieved in systems with higher gas hold-up and smaller bubble size [
14]. In addition, the position of the membrane bed under the liquid surface and the gas passing through the membrane pores and into the cell-contained area favoured the gas-to-cell contact without agitation.
The membranes were hydrophilic and, therefore, accessible to dissolved syngas components and organic acids in the liquid phase. These components diffused through the membrane pores and thereafter were converted into biogas. The produced biogas built a pressure inside the membranes which increased until it reached the membrane bubble point. Then, biogas exited the inner membrane area by blowing the membrane pores dry. Consequently, the inner pressure of the membrane dropped again, and fresh liquid diffused through the membrane. The fact that the membrane sachets were continuously swollen led also to the conclusion that there was probably minimum gas exchange from the outside to the inside of the sachets. The membranes are accessible to gas when they are dry, however once they are wet, the differential pressure has to be greater than the bubble point for a gas exchange to occur. This means that the membranes worked as conductors of the diffused liquid phase and the produced gas phase. From previous studies, it was observed that the floating effect was directly stirred by the organic loading rate (OLR) of the bioreactors [
15] because the biogas production rate inside the membranes had to be high in order to create the floating effect. The experiments in this work showed that a minimum OLR of approximately 1 g COD/(L·d) was required in order to initiate the membrane floating phenomenon. The undissolved syngas bubbles ascended between the membrane sachets to the surface of the liquid.
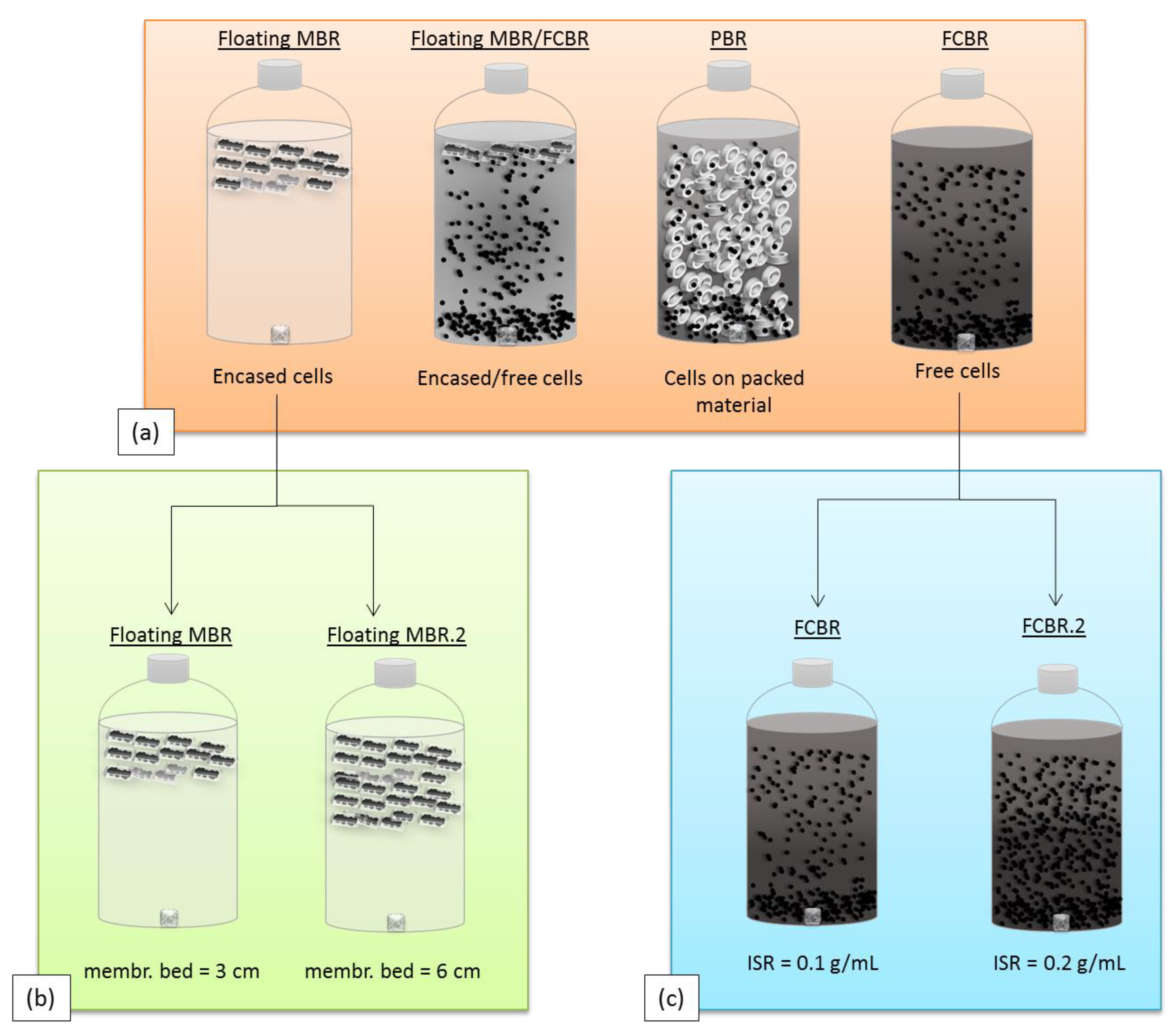
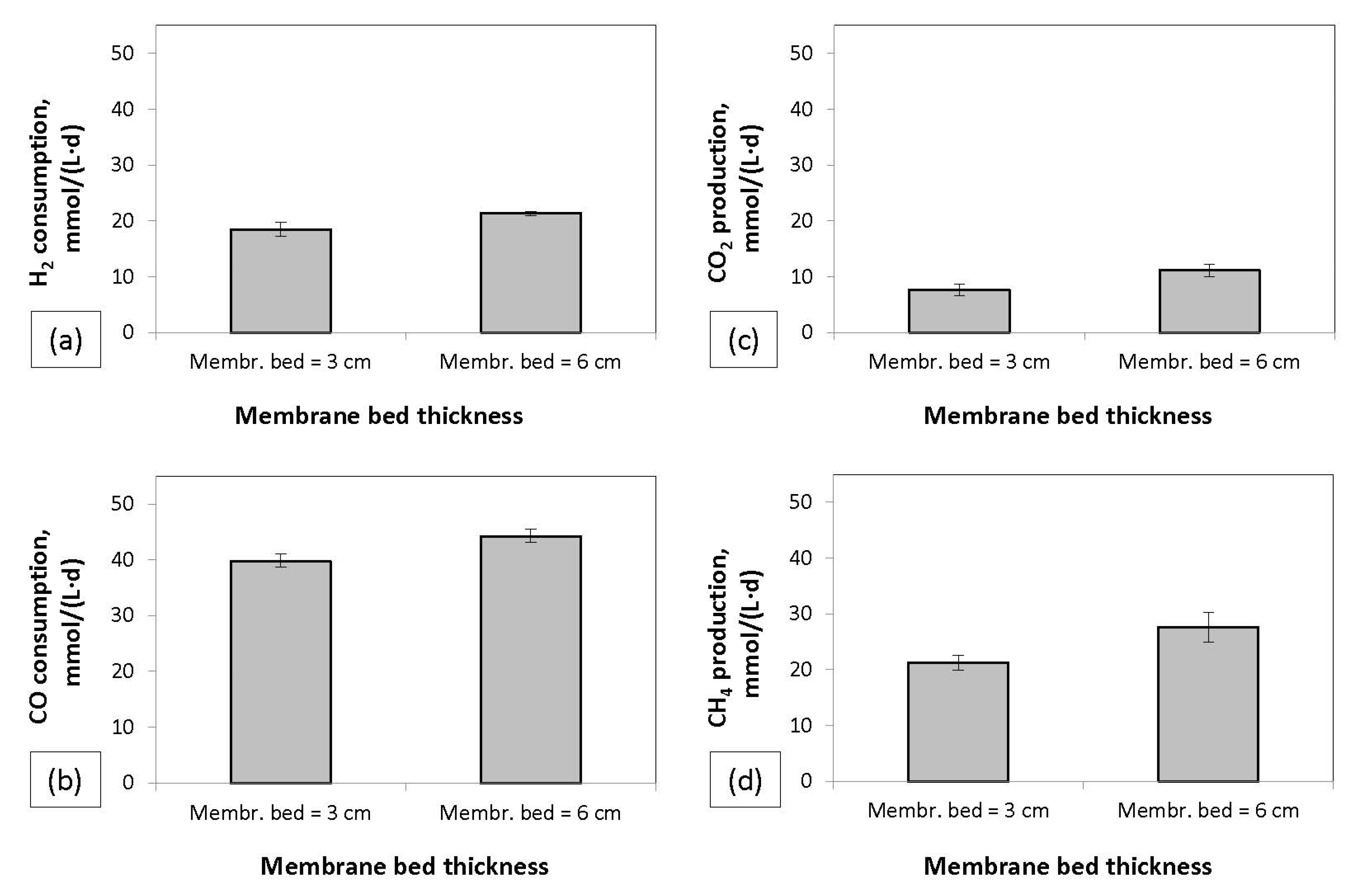
Figure 2 illustrates the different bioreactor designs that were investigated. The floating MBR was operated in parallel with a free cell bioreactor (FCBR) containing free suspended cells, a packed bioreactor (PBR) containing free cells growing on packed material, and a hybrid bioreactor (floating MBR/FCBR) containing both membrane encased cells and free cells. These bioreactors are described in more detail in the
Materials and Methods section. The effect of a thicker membrane bed was also investigated as well as the effect of a higher inoculum-to-syngas ratio (ISR). The H
2 and CO conversion rates and the CO
2 and CH
4 production rates in mmol/(L·d) were used as indicators of the reactors’ efficacy. The results showed that the use of the membrane bed resulted in higher syngas conversion rates. The increase of the thickness of the membrane bed led to higher conversion rates. In addition, higher ISRs increased the conversion rates in both batch and continuous experiments.
During the starting up period (first 30 days) of the continuous experiment, the bioreactors were fed with basal medium which contained micro- and macro-nutrients and syngas as the sole carbon and energy source. From the 31st day and until the 90th day, acetic acid was also introduced into the liquid medium with an OLR of 1.21 g COD/(L·d). The HRT was 34 d during the experiment.
3.1. Efficacy of the Membrane Floating Bed System
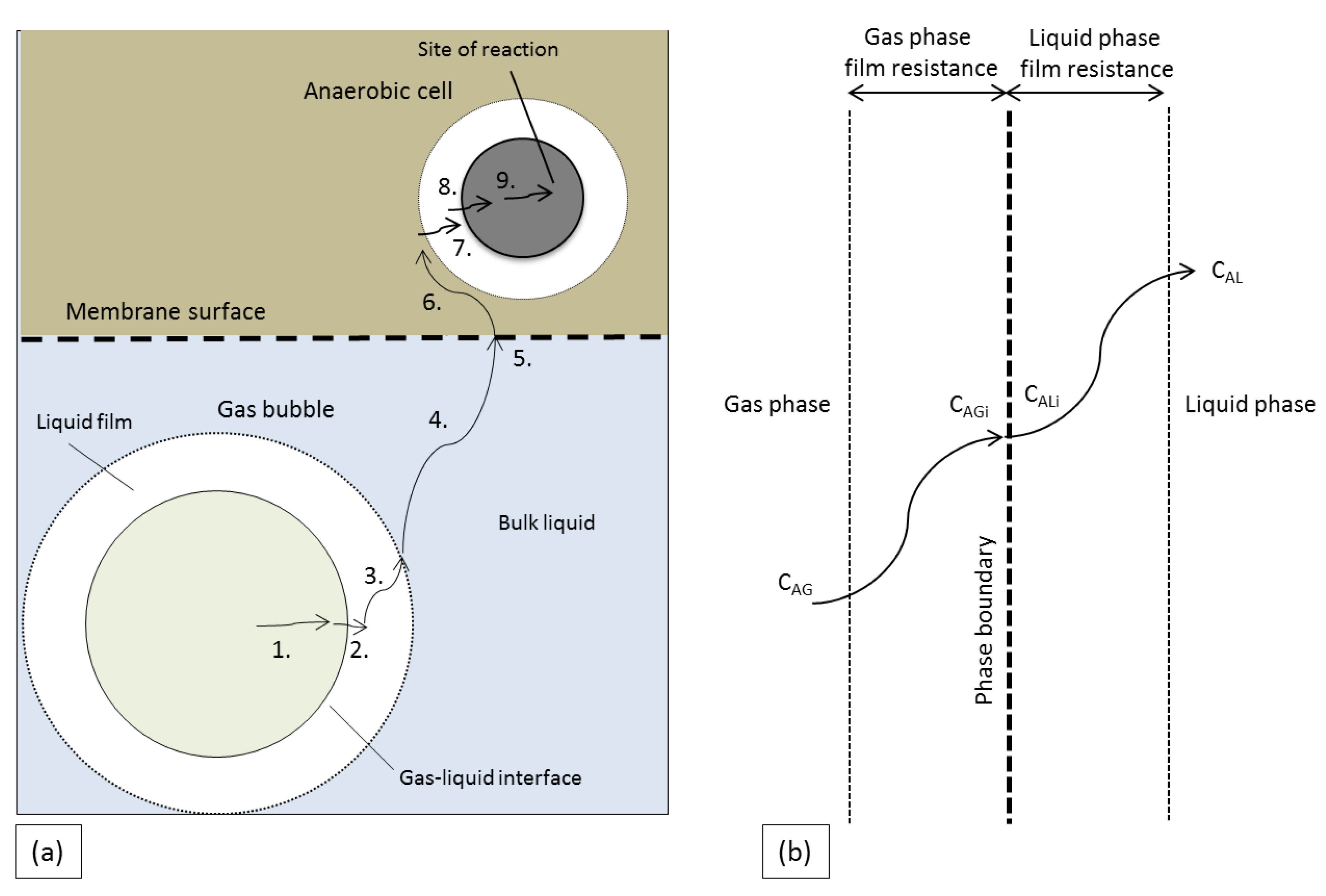
The inoculum used in this work consisted of mixed cells which take up syngas from the liquid phase. Therefore, the rate of the mass transfer and, thus, the mass transfer coefficient from the gas-to-liquid phase is of vital importance. The syngas components face several resistances during their journey from the gas phase to the cells (
Figure 3a). In the case of floating MBR, there is an extra mass transfer resistance of the membrane surface, however previous studies proved that this resistance is negligible [
15]. The liquid film surrounding the cells (
Figure 3b) is considered to be the main resistance of the gas-to-liquid mass transfer inside the investigated bioreactors, while the rest of the resistances are considered negligible [
14]. The following equations show the rate of mass transfer of component A though the gas (N
AG) and the liquid (N
AL) boundary in gmol/(m
3·s):
where k
G and k
L is the gas-phase and liquid-phase mass transfer coefficient, m/s, respectively, and α is the total interfacial area for mass transfer, 1/m. The concentration of A in the liquid bulk is C
AL and in the liquid boundary is C
Ali, while the concentration of A in the gas phase is C
AG and in the gas boundary is C
AGi, in gmol/m
3.
Equations (1) and (2) can be simplified to Equations (3) and (4) [
14], respectively, where mC
AL = C
*AG is the gas-phase concentration of A in equilibrium with C
AL and C
AG/m = C
*AL is the liquid-phase concentration of A in equilibrium with C
AG, and m is the distribution factor.
Equations (3) and (4) are valid for systems in which the main mass transfer resistance is either the gas-phase film resistance or the liquid-phase film resistance. Thus, the overall mass transfer coefficients K
Gα and K
Lα were replaced by k
Gα and k
Lα [
14]. In the case of syngas fermentation, H
2 and CO are poorly soluble to the aqueous solution, which means that k
Gα is significantly larger than k
Lα and, therefore, Equation (3) is the main equation that can describe the limiting mass transfer rate in the system.
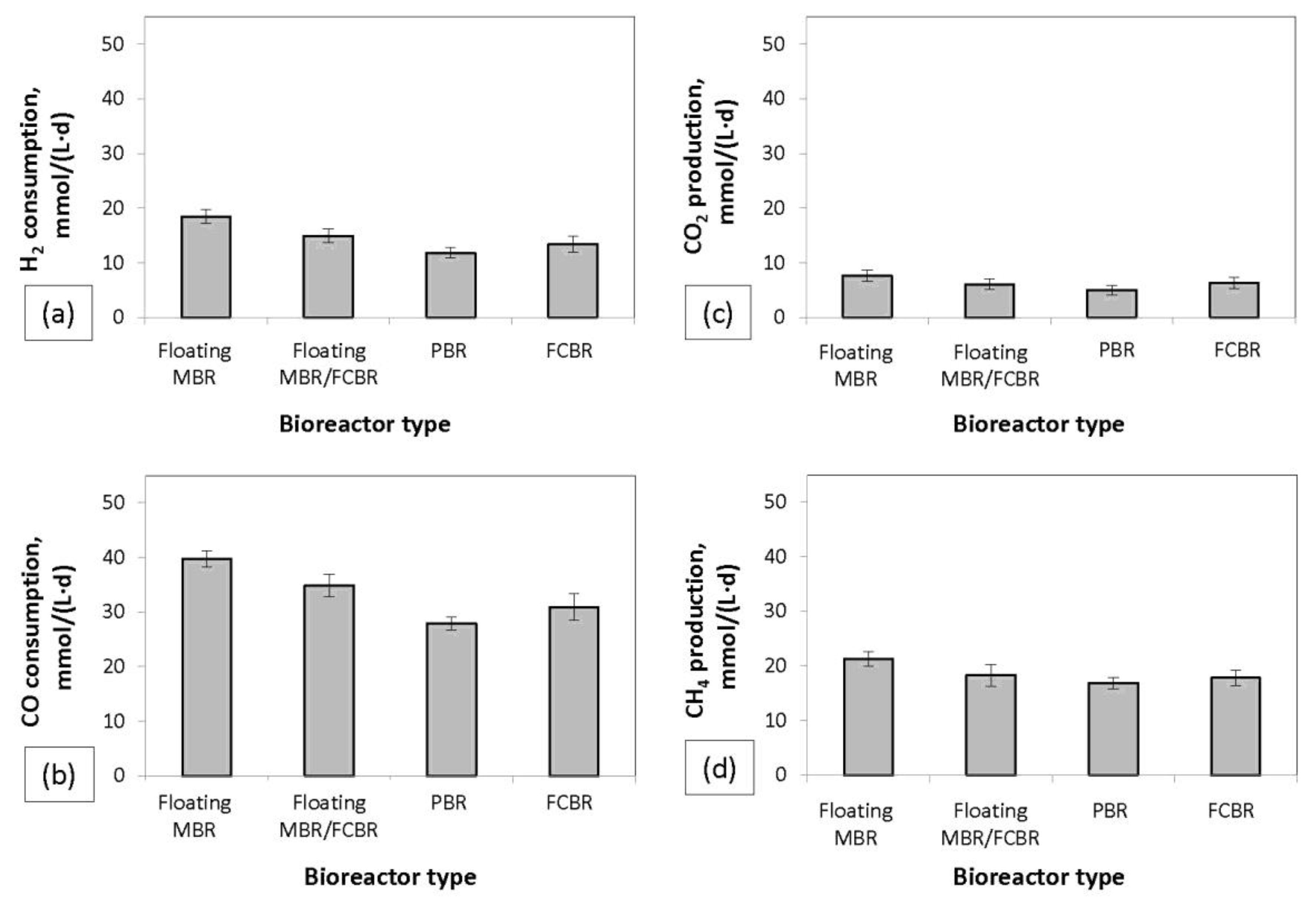
The consumption rates of H
2 and CO and the production rates of CH
4 and CO
2 in floating MBR, floating MBR/FCBR, PBR, and FCBR are presented in
Figure 4. The highest consumption rates of H
2 and CO obtained were 18.47 ± 1.25 and 39.67 ± 1.45 mmol/(L·d), respectively, in floating MBR. In the same bioreactor, the highest CH
4 production of 21.33 mmol CH
4/(L·d) was also achieved and the highest CH
4 yield of 0.36 mol CH
4/mol (H
2 + CO). The pH of the effluent was 8.2 ± 2.0 in all bioreactors during the experiment. In previous studies, a triculture (
R. rubrum,
M. barkeri,
M. formicicum) converted syngas of a similar composition into CH
4 in a trickle bed bioreactor with a CH
4 production rate of 48–72 mmol CH
4/(L·d) and yields of 0.2–0.214 mol CH
4/mol (H
2 + CO) [
16,
17]. Moreover, the same triculture converted syngas in a packed bed bioreactor with a CH
4 production of 4.8–7.2 mmol/(L·d) and a yield of 0.214 mol CH
4/mol (H
2 + CO) [
17]. Higher yields of 0.6–0.8 mol CH
4/mol (H
2 + CO) and a CH
4 production of 73 mmol/(L·d) were obtained in a multi-orifice oscillatory baffled bioreactor where granular sludge converted syngas with a gas recirculation rate of 600 mL/min [
18].
The lowest syngas biomethanation rates were observed in the PBR and the FCBR. This result contradicts with other studies, which reported that the use of packed material in the PBR significantly improved the syngas conversion rates and CH
4 production rate. For instance, Burkhardt, et al. [
19] reported a CH
4 concentration of higher than 98% in their final biogas product, achieved in a trickle bed bioreactor filled with packing material. Trickle bed bioreactors with packed material are considered effective in syngas biomethanation because of higher mass transfer coefficients and lower cost than the continuous stirred bioreactors (CSTR) [
16]. The liquid recirculation, which may be co-current or counter-current with the gas flow, is one of the main reasons for high mass transfer in the trickle bed bioreactors. However, in the present study, there was no liquid recirculation and, therefore, the lower syngas conversion rates in PBR are likely a result of inadequate substrate distribution inside the bioreactor, which is probably the reason for the lower acetic acid consumption (
Table 1) in comparison to other bioreactors.
The floating MBR/FCBR system proved to be less efficient in terms of syngas conversion in comparison to the floating MBR system. The reason for this was probably the better gas-to-cell contact in floating MBR due to the fact that the suspended inoculum was mostly concentrated at the bottom of the floating MBR/FCBR. Likewise, in the FCBR and the PBR, the inoculum was mostly concentrated at the bottom of the bioreactors. In bubble column bioreactors with free cells, when syngas feed is introduced at the bottom of the bioreactor, the H
2 and CO levels decrease as the gas flows up the column because of cellular consumption. This causes spatial difference of dissolved syngas inside the bubble column which can affect the cellular growth and the product profile [
20]. Therefore, the aim is to establish favourable syngas concentration profiles in the liquid medium according to the desirable product [
20]. However, in the floating MBR, the cells were assembled in packed formation at the upper parts of the liquid medium so that the gas bubbles stayed longer in the liquid and were dispersed though the membrane pores. This cell-placement was probably a main factor which caused better gas-to-cell contact and, therefore, faster syngas biomethanation in the floating MBR.
The floating MBR resembles the concept of a trickle bed or a packed bed bioreactor where the packed material is replaced by membranes. Trickle bed bioreactors are preferred to other bioreactor types, such as continuous stirred bioreactors, because of their enhanced gas-to-liquid mass transfer mechanisms. A mass transfer analysis on various bioreactors claimed that the mass transfer was enhanced over three times in a trickle bed bioreactor in comparison to continuously stirred tank reactor [
21]. In addition, the optimum CO mass transfer coefficient of stirred tank bioreactors can be substantially improved in bubble column bioreactors because of higher mass transfer driving forces, which are a result of gas composition spatial profiles and longer gas hold-up [
20].
3.3. Impact of Inoculum-to-Syngas Ratio (ISR)
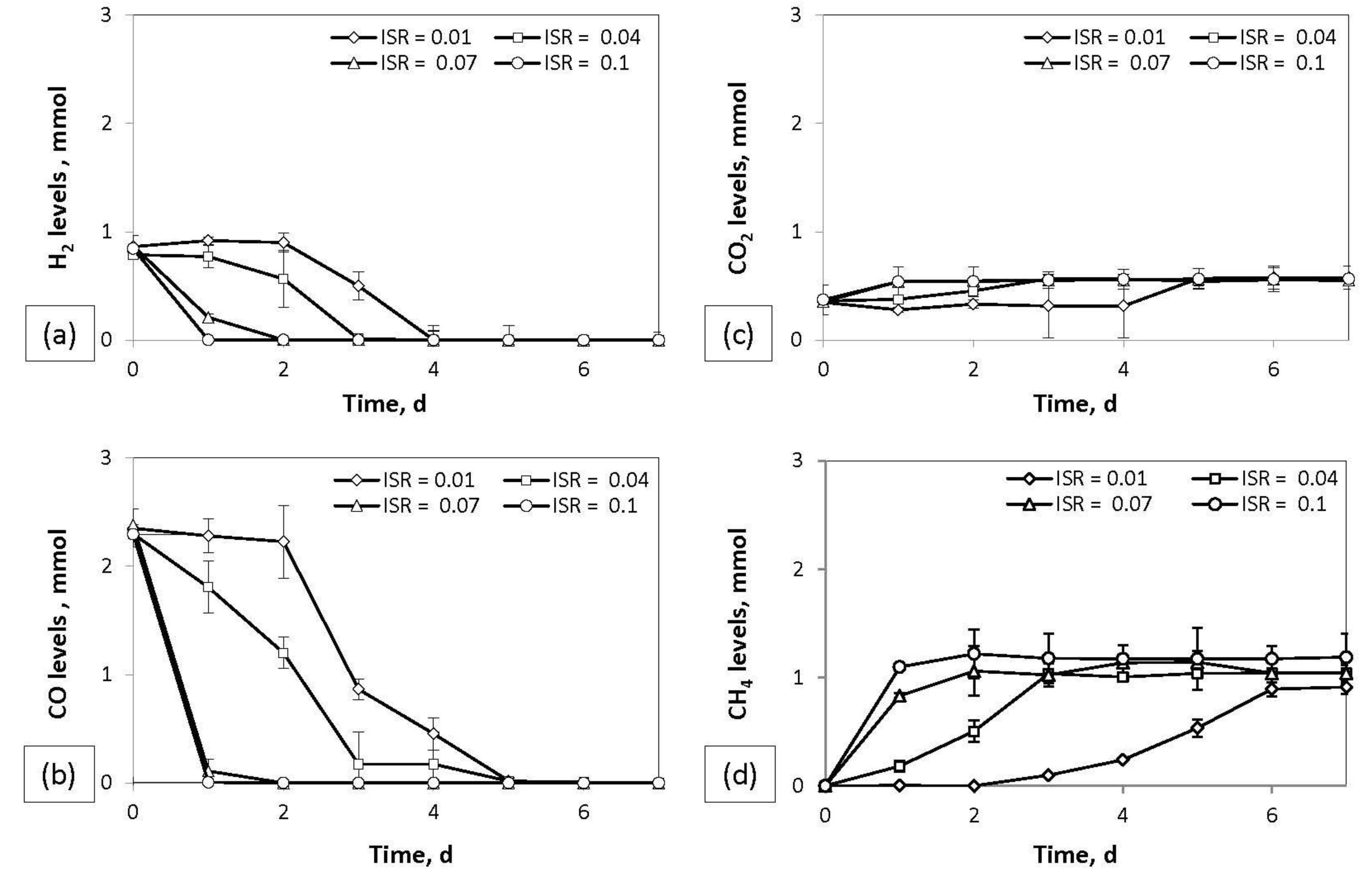
In order to study the effect of different ISRs, both batch and continuous bioreactors were operated. During the batch experiment, syngas consumption and biogas production was investigated at different ISRs (0.01, 0.04, 0.07, and 0.1 g/mL), and the results are presented in
Figure 6. According to the figure, the increase of ISR led to faster consumption of H
2 and CO. More specifically, the complete consumption of H
2 and CO took one day in bioreactors with an ISR of 0.1 g/mL, while in bioreactors with 10 times less inoculum (ISR = 0.01 g/mL), the complete consumption of H
2 and CO took four and five days, respectively. The CH
4 production followed the same trend. The CH
4 production in the bioreactor with an ISR of 0.1 g/mL was stabilized on the third day, while the CH
4 production in the bioreactor with an ISR of 0.01 g/mL reached its highest CH
4 production on the sixth day of fermentation. In bioreactors with an ISR of 0.04 g/mL, the complete conversion of H
2 and CO took three and five days, respectively. These conversion rates were lower in comparison with a previous study with a similar system (ISR = 0.38 g/mL) during which H
2 and CO were totally consumed in two days [
22]. However, in this work, the initial amount of H
2 and CO content in the bioreactors was approximately 50% and 24% higher than in the previous study. The highest CH
4 yield (at an ISR of 0.1 g/mL) obtained in the current work was 0.39 mol CH
4/mol (H
2 + CO
2), which is comparatively higher than in other similar studies. For example, the CH
4 yield achieved during the conversion of H
2/CO
2 and CO in a batch bubble column bioreactor with an ISR of 0.0037 g/mL was 0.22–0.26 mol CH
4/mol (H
2 + CO
2) and 0.25 mol CH
4/mol CO, respectively [
23]. The lower yields in the above work could have been caused by the lower ISR and the different syngas composition in comparison to the current work.
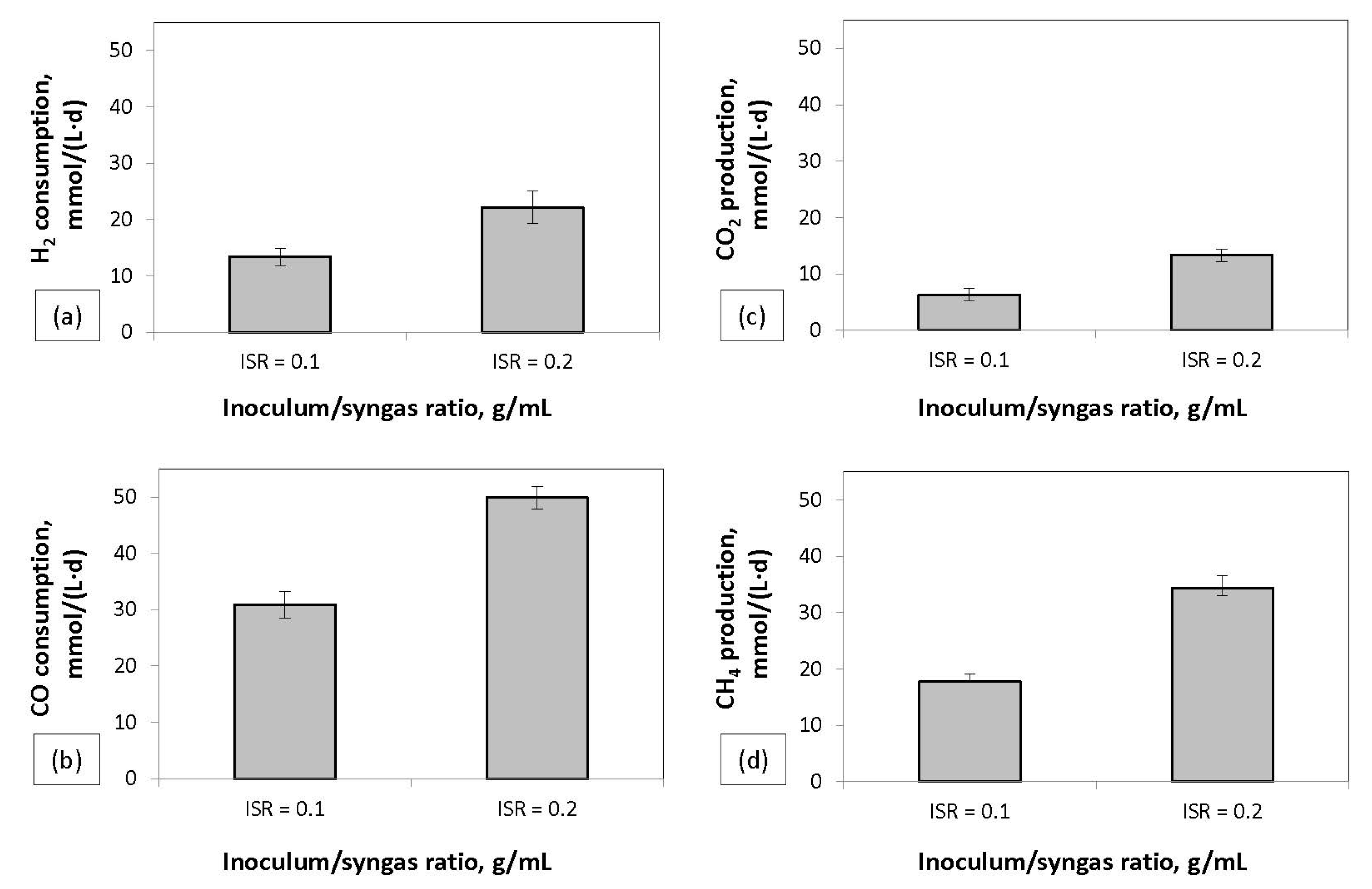
During the continuous experiment, the FCBR that had an ISR of 0.1 g/mL was operated in parallel with FCBR.2 with an ISR of 0.2 g/mL. The aim was to investigate the effect of doubling the ISR and the operation in continuous mode. The results (
Figure 7) showed an increase in the H
2 and CO consumption rate of approximately 66% and 61%, respectively, and an increase of approximately 94% in CH
4 production rate. The CH
4 production rate in the bioreactor with an ISR of 0.2 g/mL was 34.41 mmol/(L·d), the highest achieved in this work. This production rate is comparable to results from other similar studies. A CH
4 production rate of 72 mmol/(L·d) was achieved during biomethanation of syngas with a similar composition at a gas flow rate of 70 mL/min. The syngas was converted in a trickle bed bioreactor by a triculture of
R. rubrum,
M. barkeri, and
M. formicicum [
4,
16]. Another study was conducted in a packed bed bioreactor with the same triculture, similar syngas composition, and gas flow rate of 80 mL/min. In that work, a lower CH
4 production rate between 4.8 and 7.2 mmol/(L·d) was reported [
17]. In the literature, there are previous studies that have reported that higher ISRs can improve the CH
4 potential yields. These studies have mainly focused on the investigation of using different ISRs based on g VS
added, with mixed anaerobic sludge. For example, a study on the effect of ISR on the CH
4 potential of microcrystalline cellulose production wastewater reported that the fastest CH
4 production rate and highest kinetic constant were achieved at the highest ISR of 2.0 [
10]. However, extremely high ISRs may be inhibiting for the anaerobic process. Lim and Fox [
24] studied three different ISRs (1, 0.33, and 0.125) and reported that the highest CH
4 production rate was obtained at the ratio of 0.33, whereas the minimum production rate was obtained at an ISR of 0.125. The low ISR probably caused low substrate concentration and the high ISR caused high concentration of volatile fatty acids and thus, low pH [
24].