Design and Performance Comparison of Methanol Production Processes with Carbon Dioxide Utilization
Abstract
:1. Introduction
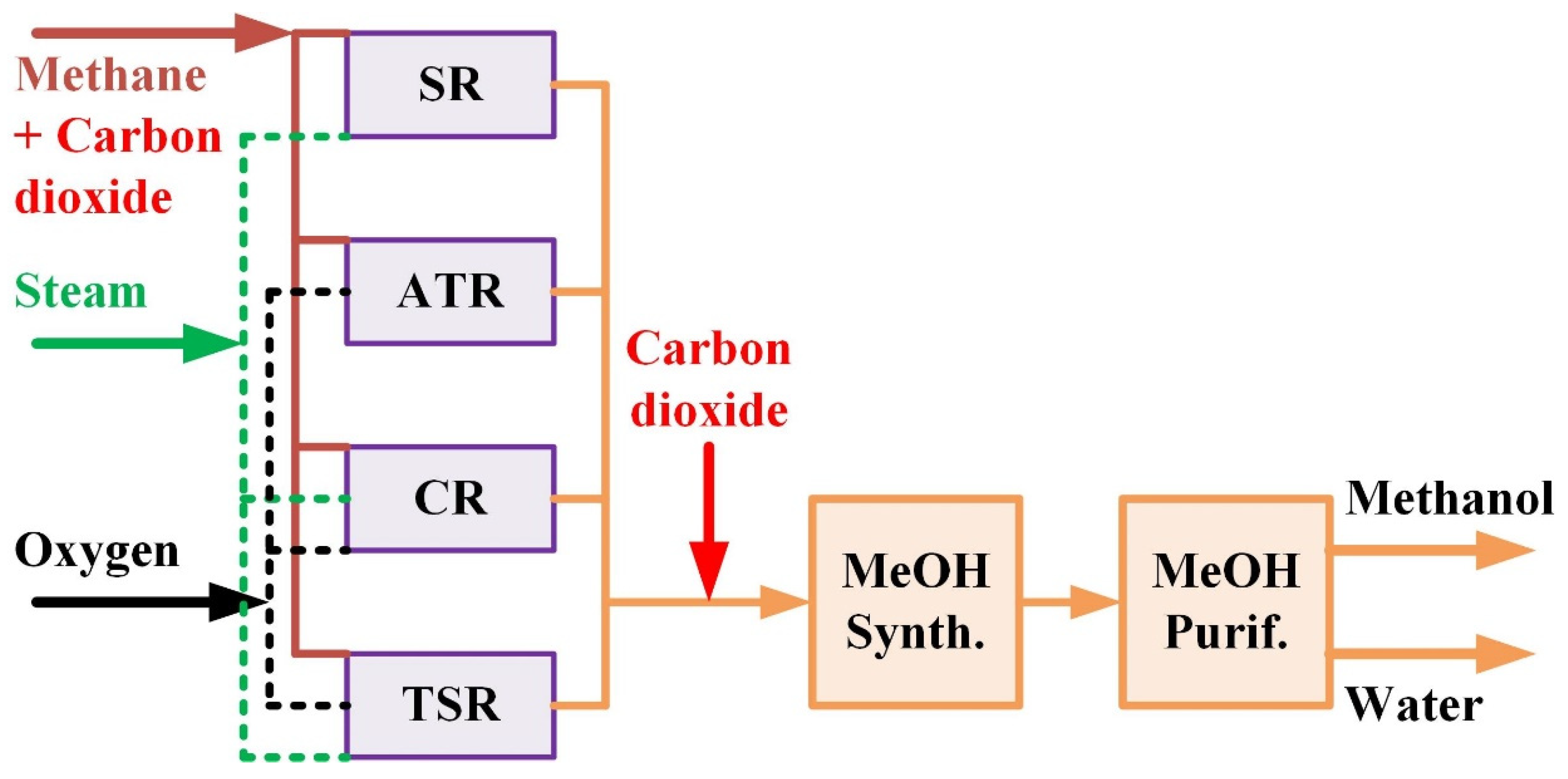
2. Process Design
2.1. Type A—Steam Reforming (A-SR) Process
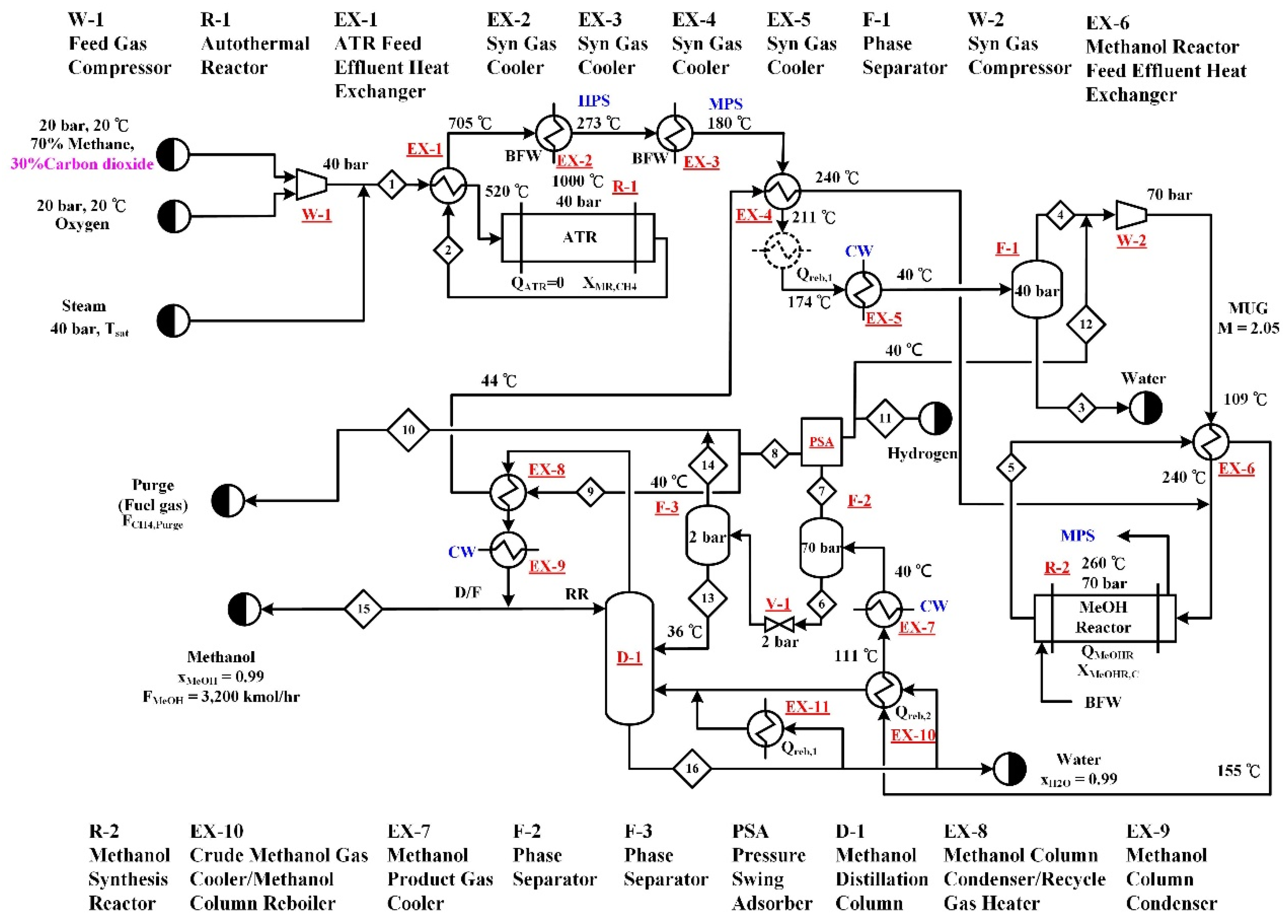
2.2. Type B—Autothermal Reforming (B-ATR) Process
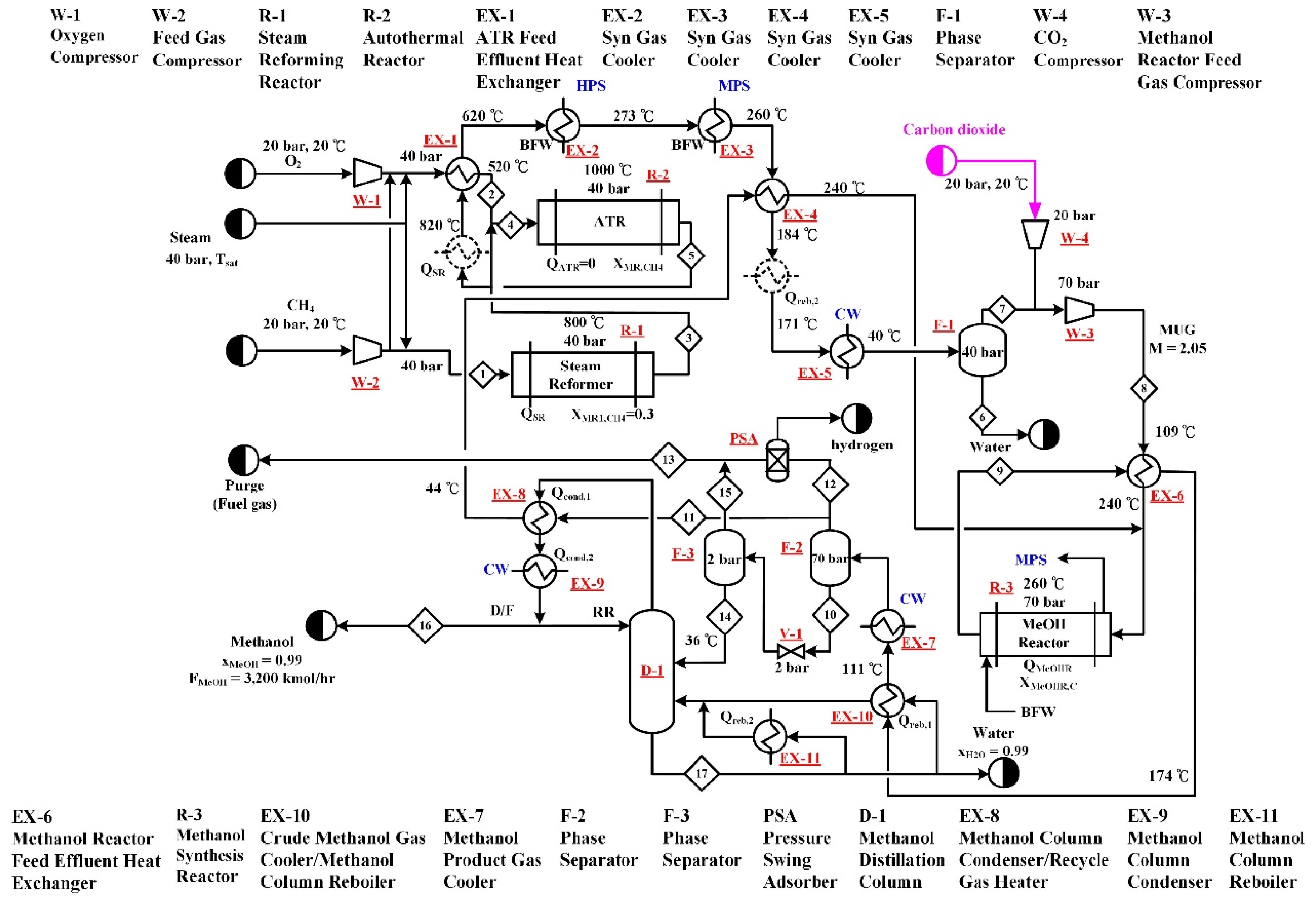
2.3. Type C—Combined Reforming (C-CR) and Two Step Reforming (C-TSR) Processes
3. Evaluation and Comparison of Performance Indicators
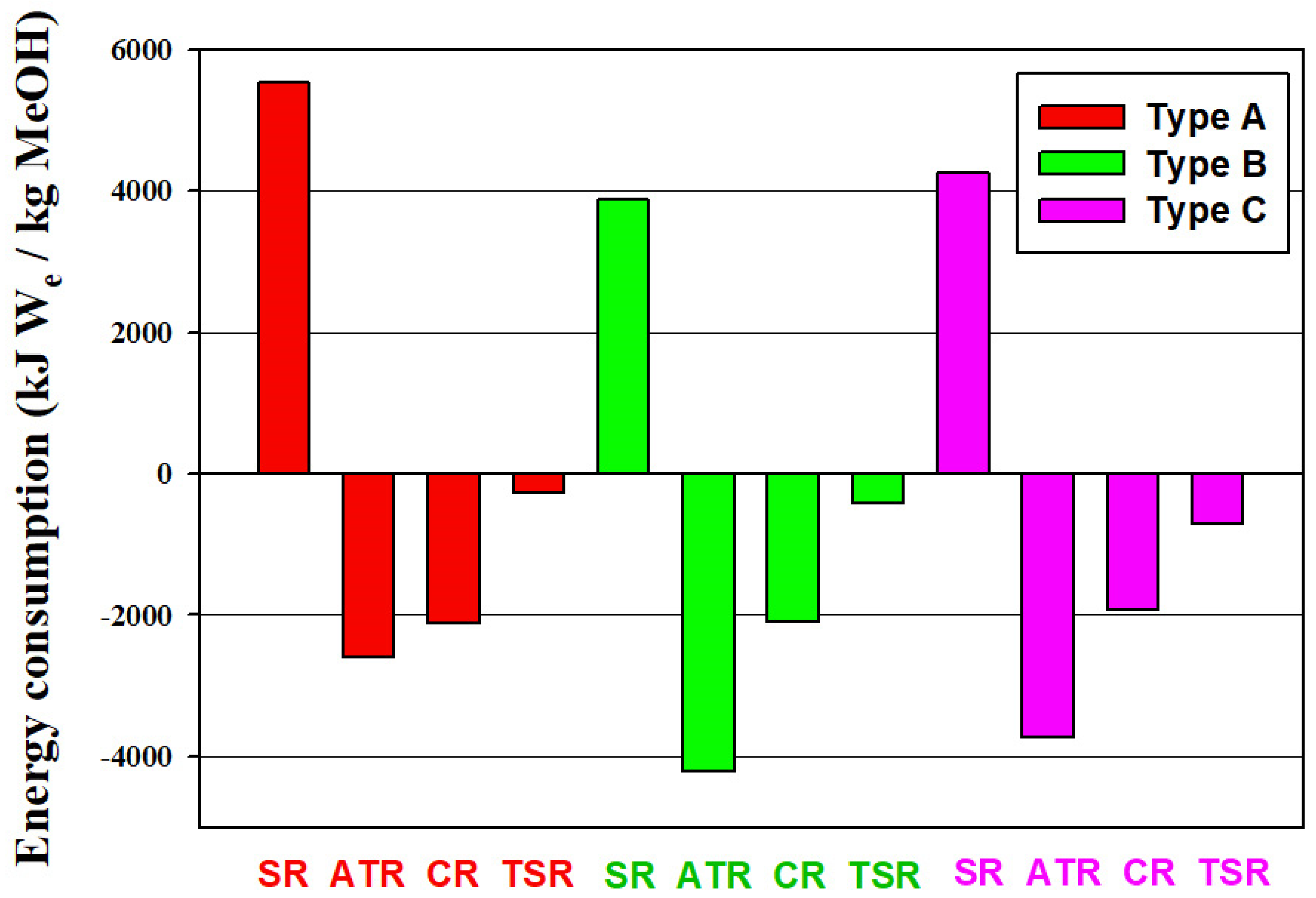
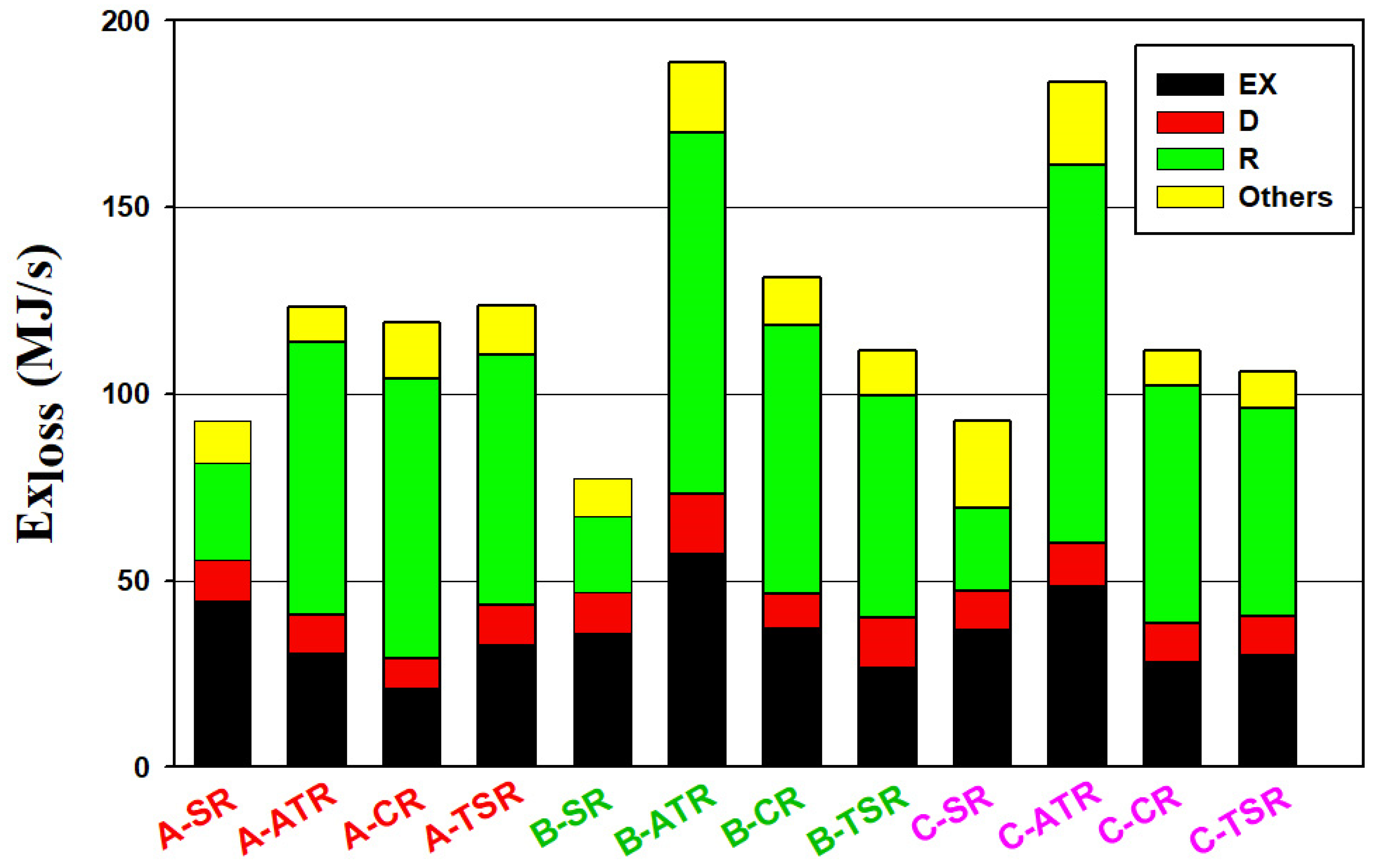
3.1. Energy Use and Exergy Loss
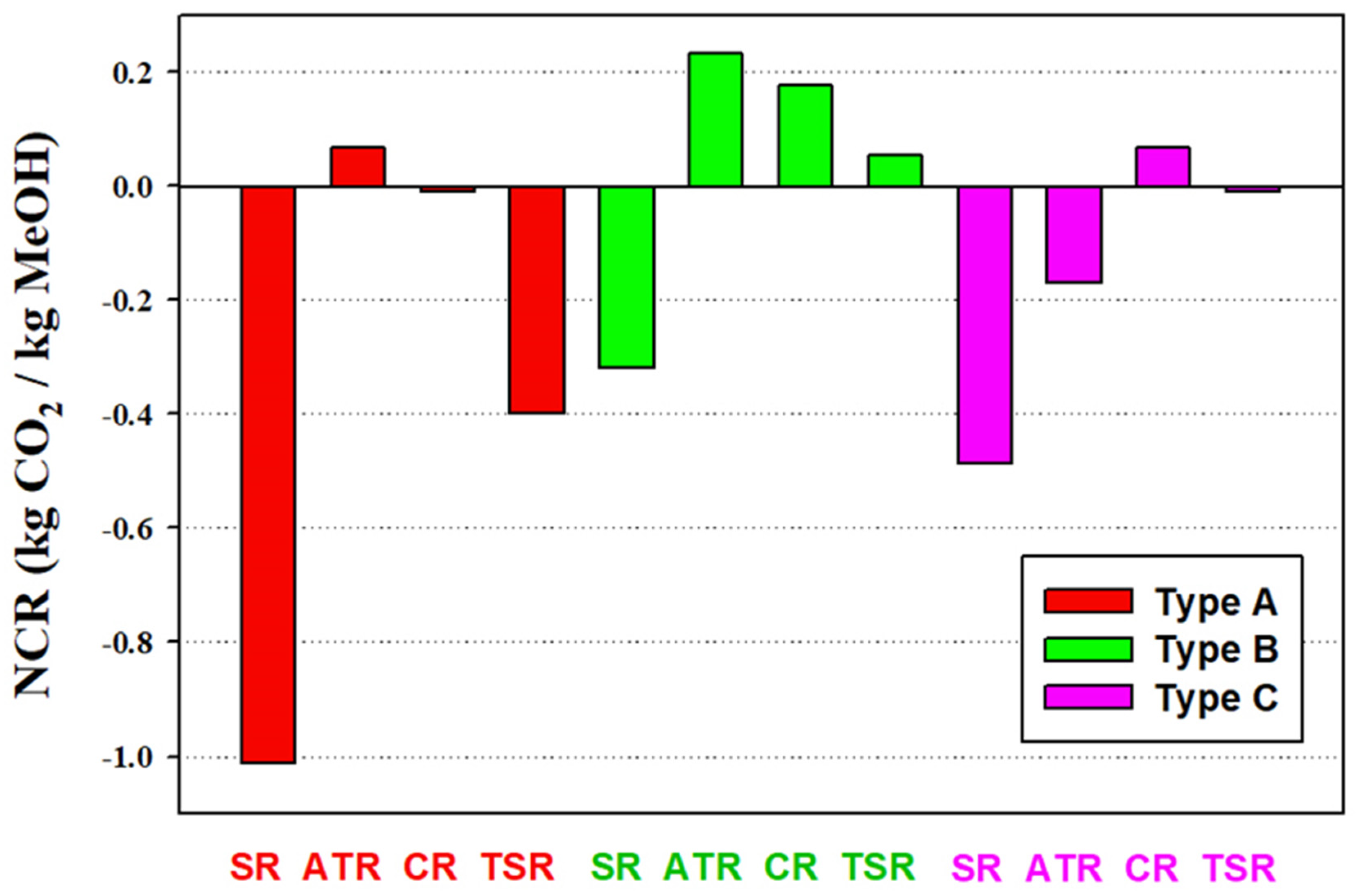
3.2. Carbon Dioxide Utilization
3.3. Cost Analysis and Economic Profit Evaluation
4. Conclusions
- With the utilization of CO2 in the methanol process, the major energy effects are on the SR and ATR processes. Less energy input to the process is needed (SR) or more energy output from the process (ATR) can be obtained.
- With the utilization of CO2 in the methanol process, the total exergy loss is increased significantly for the ATR processes, in particular from the reactors.
- The use of CO2 as part of the reforming feedstock (Type B processes) can utilize more CO2 than the direct use of CO2 as the methanol synthesis feedstock (Type C processes). The process uses the highest CO2 feed rate is B-ATR.
- The utilization of CO2 in both the reforming step and methanol synthesis step of the methanol process is beneficial to the carbon dioxide reduction. The process with the highest NCR value is the B-ATR process with a value of 0.23 kg CO2/kg methanol.
- The utilization of CO2 in methanol production does not necessarily lead to the increase of capital cost or manufacturing cost.
- The utilization of CO2 in methanol production does not necessarily lead to a reduction of the profit. The B-ATR process has the highest IRR with a value of 41%. The process with the lowest IRR is B-TSR with a value of 12.5%.
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Design Specification | Adjusted Variable |
|---|---|
| A-SR | |
| SR-methane conversion = 90% | Steam feed rate |
| Methanol production rate | Methane feed rate |
| M value of make-up-gas to methanol synthesis = 2.05 | Hydrogen purge rate from PSA-1 |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| A-ATR | |
| Adiabatic operation of ATR | Oxygen feed rate |
| Methanol production rate | Methane feed rate |
| ATR-methane conversion = 90% | Steam feed rate |
| M value of make-up-gas to methanol synthesis = 2.05 | Hydrogen purge rate from PSA |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| A-CR | |
| Adiabatic operation of ATR | Oxygen feed rate |
| Methanol production rate | Methane feed rate |
| ATR-methane conversion = 90% | Steam feed rate |
| SMR-methane conversion = 30% | Steam feed split ratio |
| M value of make-up-gas to methanol synthesis = 2.05 | Methane feed split ratio |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| A-TSR | |
| Adiabatic operation of ATR | Oxygen feed rate |
| Methanol production rate | Methane feed rate |
| ATR-methane conversion = 90% | Steam feed rate |
| SMR-methane conversion = 30% | Steam feed split ratio |
| M value of make-up-gas to methanol synthesis = 2.05 | Hydrogen purge rate from PSA-1 |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| Design Specification | Adjusted Variable |
|---|---|
| B-SR | |
| SR-methane conversion = 90% | Steam feed rate |
| Methanol production rate | Methane feed rate |
| M value of make-up-gas to methanol synthesis = 2.05 | Hydrogen purge rate from PSA-1 |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| B-ATR | |
| Adiabatic operation of ATR | Oxygen feed rate |
| Methanol production rate | Methane feed rate |
| ATR-methane conversion = 90% | Steam feed rate |
| M value of make-up-gas to methanol synthesis = 2.05 | Hydrogen purge rate from PSA |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| B-CR 1 | |
| Adiabatic operation of ATR | Oxygen feed rate |
| Methanol production rate | Methane feed rate |
| ATR-methane conversion = 90% | Steam feed rate |
| SMR-methane conversion = 30% | Steam feed split ratio |
| M value of make-up-gas to methanol synthesis = 2.05 | Hydrogen purge rate from PSA-1 |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| B-TSR | |
| Adiabatic operation of ATR | Oxygen feed rate |
| Methanol production rate | Methane feed rate |
| ATR-methane conversion = 90% | Steam feed rate |
| SMR-methane conversion = 30% | Steam feed split ratio |
| M value of make-up-gas to methanol synthesis = 2.05 | Hydrogen purge rate of PSA-1 |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| Design Specification | Adjusted Variable |
|---|---|
| C-SR | |
| SR-methane conversion = 90% | Steam feed rate |
| Methanol production rate | Methane feed rate |
| M value of make-up-gas to methanol synthesis = 2.05 | CO2 feed rate |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| C-ATR | |
| Adiabatic operation of ATR | Oxygen feed rate |
| Methanol production rate | Methane feed rate |
| ATR-methane conversion = 90% | Steam feed rate |
| M value of make-up-gas to methanol synthesis = 2.05 | CO2 feed rate |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| C-CR 1 | |
| Adiabatic operation of ATR | Oxygen feed rate |
| Methanol production rate | Methane feed rate |
| ATR-methane conversion = 90% | Steam feed rate |
| SMR-methane conversion = 30% | Steam feed split ratio |
| M value of make-up-gas to methanol synthesis = 2.05 | CO2 feed rate |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
| C-TSR | |
| Adiabatic operation of ATR | Oxygen feed rate |
| Methanol production rate | Methane feed rate |
| ATR-methane conversion = 90% | Steam feed rate |
| SMR-methane conversion = 30% | Steam feed split ratio |
| M value of make-up-gas to methanol synthesis = 2.05 | CO2 feed rate |
| Overall process mass balance of methane | Split fraction of F-2 vapor outlet |
Appendix B
| Stream Number. | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| Temp (°C) | 202 | 1000 | 40 | 40 | 260 | 40 | 40 | 40 |
| Press (bar) | 40 | 40 | 40 | 40 | 70 | 70 | 70 | 70 |
| Mole flow (kmol/h) | 16,188 | 17,846 | 3442 | 14,460 | 15,719 | 3578 | 12,140 | 4290 |
| Mass flow (t/h) | 265.46 | 265.46 | 73.5 | 191 | 315 | 111 | 204 | 188 |
| Mole Flow (kmol/h) | ||||||||
| CH4 | 4641.8 | 464.2 | 49.45 | 465.7 | 467.7 | 8.4 | 461.9 | 461.9 |
| H2O | 2012.4 | 3053.7 | 3025.9 | 27.9 | 181.1 | 43.09 | 1.45 | 1.45 |
| CO | 0 | 4901.3 | 79.1 | 4822.4 | 1244.8 | 14.89 | 1236.5 | 1236.5 |
| CO2 | 1989.3 | 1090.4 | 283.4 | 807.7 | 1770.8 | 80.9 | 1638.4 | 1638.4 |
| H2 | 0 | 8336.9 | 0.0003 | 8336.9 | 8750.1 | 26.59 | 8721.7 | 872.17 |
| O2 | 2902.9 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| CH3OH | 0 | 0 | 0 | 0 | 3304.5 | 3220.7 | 80.4 | 80.4 |
| Stream Number | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 |
| Temp (°C) | 37 | 37 | 40 | 98 | 37 | 37 | 64 | 98 |
| Press (bar) | 70 | 70 | 70 | 0 | 2 | 2 | 1 | 1 |
| Mole flow (kmol/h) | 4155 | 269 | 3837 | 4012 | 3260 | 134 | 3232 | 28 |
| Mass flow (t/h) | 115 | 74 | 7.7 | 8.1 | 104.9 | 4.1 | 103.5 | 0.5 |
| Mole Flow (kmol/h) | ||||||||
| CH4 | 447.3 | 22.7 | 0 | 0 | 0.3 | 8.1 | 0.3 | 0 |
| H2O | 1.4 | 0.14 | 0 | 0 | 43.0 | 0.09 | 15.1 | 27.9 |
| CO | 1197.4 | 53.73 | 0 | 0 | 0.26 | 14.63 | 0.26 | 0 |
| CO2 | 1586.58 | 116.2 | 0 | 0 | 16.5 | 64.4 | 16.5 | 0 |
| H2 | 844.58 | 53.97 | 3837.03 | 4012.5 | 0.21 | 26.38 | 0.21 | 0 |
| O2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| CH3OH | 77.86 | 22.94 | 0 | 0 | 3200.3 | 20.4 | 3200 | 0.3 |
References
- The Global Status of CCS. 2018. Available online: https://www.globalccsinstitute.com/resources/global-status-report/ (accessed on 16 September 2019).
- Centi, G.; Iaquaniello, G.; Perathoner, S. Can we afford to waste carbon dioxide? Carbon dioxide as a valuable source of carbon for the production of light olefins. ChemSusChem 2011, 4, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- Quadrelli, E.A.; Centi, G.; Duplan, J.-L.; Perathoner, S. Carbon dioxide recycling: Emerging large-scale technologies with industrial potential. ChemSusChem 2011, 4, 1194–1215. [Google Scholar] [CrossRef] [PubMed]
- Cuéllar-Franca, R.M.; Azapagic, A. Carbon capture, storage and utilization technologies: A critical analysis and comparison of their life cycle environmental impacts. J. CO2 Util. 2015, 9, 82–102. [Google Scholar] [CrossRef]
- Alper, E.; Orhan, O.Y. CO2 utilization: Developments in conversion processes. Petroleum 2017, 3, 109–126. [Google Scholar] [CrossRef]
- Wurzel, T. Lurgi MegaMethanol Technology—Delivering the building blocks for future fuel and monomer demand. In Proceedings of the DGMK International Conference Synthesis Gas Chemistry, Dresden, Germany, 4–6 October 2006. [Google Scholar]
- Bertau, M.; Offermanns, H.; Plass, L.; Schmidt, F.; Wernicke, H.-J. Methanol: The Basic Chemical and Energy Feedstock of the Future; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Aresta, M. Carbon Dioxide as Chemical Feedstock; Wiley-VCH: Weinheim, Germany, 2010. [Google Scholar]
- Lee, S. Methanol synthesis from syngas. In Handbook of Alternative Fuel Technologies; Lee, S., Speight, J.G., Loyalka, S.K., Eds.; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- König, P.; Göhna, H. Process of Producing Methanol. U.S. Patent 5,631,302, 20 May 1997. [Google Scholar]
- Joo, O.-S.; Jung, K.-D.; Moon, I.; Rozovskii, A.Y.; Lin, G.I.; Han, S.-H.; Uhm, S.-J. Carbon dioxide hydrogenation to form methanol via a reverse-water-gas-shift reaction (the CAMERE Process). Ind. Eng. Chem. Res. 1999, 38, 1808–1812. [Google Scholar] [CrossRef]
- Carbon Recycling International, CO2-to-Methanol Plant Erected in Germany. Available online: https://www.carbonrecycling.is/news/2018/11/1/cri-co2-to-methanol-plant-erected-in-germany-ck6nx (accessed on 16 September 2019).
- Aasberg-Petersen, K.; Dybkjær, I.; Ovesen, C.V.; Schjødt, N.C.; Sehested, J.; Thomsen, S.G. Natural gas to synthesis gas—catalysts and catalytic processes. J. Nat. Gas. Sci. Eng. 2011, 3, 423–459. [Google Scholar] [CrossRef]
- Ulber, D. A guide to: Methane reforming. Chem. Eng. 2015, 122, 40–46. [Google Scholar]
- Usman, M.; Wan Daud, W.M.A.; Abbas, H.F. Dry reforming of methane: Influence of process parameters—A review. Renew. Sust. Energ. Rev. 2015, 45, 710–744. [Google Scholar] [CrossRef]
- Arora, S.; Prasad, R. An overview on dry reforming of methane: Strategies to reduce carbonaceous deactivation of catalysts. RSC Adv. 2016, 6, 108668–108688. [Google Scholar] [CrossRef]
- Song, C.; Pan, W. Tri-reforming of methane: A novel concept for catalytic production of industrially useful synthesis gas with desired H2/CO ratios. Catal. Today 2004, 98, 463–484. [Google Scholar] [CrossRef]
- Noureldin, M.M.B.; Elbashir, N.O.; El-Halwagi, M.M. Optimization and selection of reforming approach for syngas generation from natural/shale gas. Ind. Eng. Chem. Res. 2014, 53, 1841–1855. [Google Scholar] [CrossRef]
- Wiesberg, I.L.; de Medeiros, J.L.; Alves, R.M.B.; Coutinho, P.L.A.; Araújo, O.Q.F. Carbon dioxide management by chemical conversion to methanol: Hydrogenation and bi-reforming. Energy Convers. Mgmt. 2016, 125, 320–335. [Google Scholar] [CrossRef]
- Pérez-Fortes, M.; Schöneberger, J.C.; Boulamanti, A.; Tzimas, E. Methanol synthesis using captured CO2 as raw material: Techno-economic and environmental assessment. Appl. Energy 2016, 161, 718–732. [Google Scholar] [CrossRef]
- Luu, M.T.; Milani, D.; Bahadori, A.; Abbas, A. A comparative study of CO2 utilization in methanol synthesis with various syngas production technologies. J. CO2 Util. 2015, 12, 62–76. [Google Scholar] [CrossRef]
- Milani, D.; Khalilpour, R.; Zahedi, G.; Abbas, A. A model-based analysis of CO2 utilization in methanol synthesis plant. J. CO2 Util. 2015, 10, 12–22. [Google Scholar] [CrossRef]
- Blumberg, T.; Morosuk, T.; Tsatsaronis, G. Exergy-based evaluation of methanol production from natural gas with CO2 utilization. Energy 2017, 141, 2528–2539. [Google Scholar] [CrossRef]
- Zhang, Y.; Cruz, J.; Zhang, S.; Lou, H.H.; Benzon, T.J. Process simulation and optimization of methanol production coupled to tri-reforming process. Int. J. Hydrogen Energy 2013, 38, 13617–13630. [Google Scholar] [CrossRef]
- Aspen Technology, Inc. Aspen Plus® V10; Aspen Technology, Inc.: Bedford, MA, USA, 2018. [Google Scholar]
- Linnhoff, B.; Mason, D.R.; Wardle, I. Understanding heat exchanger networks. Comput. Chem. Eng. 1979, 3, 295–302. [Google Scholar] [CrossRef]
- United States Energy Information Administration. How Much Carbon Dioxide Is Produced Per Kilowatthour of U.S. Electricity Generation? Available online: https://www.eia.gov/tools/faqs/faq.php?id=74&t=11 (accessed on 31 August 2019).
- Bolland, O.; Sæther, S. New concepts for natural gas fired power plants which simplify the recovery of carbon dioxide. Energy Convers. Mgmt. 1992, 33, 467–475. [Google Scholar] [CrossRef]
- Economic indicators. Chem. Eng. 2017, 124, 64.
- United States Energy Information Administration. Henry Hub Natural Gas Spot Price. Available online: https://www.eia.gov/dnav/ng/hist/rngwhhdm.htm (accessed on 31 August 2019).
- Turton, R.; Bailie, R.C.; Whiting, W.B.; Shaeiwitz, L.A.; Bhattacharyya, D. Analysis, Synthesis, and Design of Chemical Processes, 4th ed.; Prentice Hall: New York, NY, USA, 2012. [Google Scholar]
- Choi, S.; Park, J.; Han, C.; Yoon, E.S. Optimal design of synthesis gas production process with recycled carbon dioxide utilization. Ind. Eng. Chem. Res. 2008, 47, 323–331. [Google Scholar] [CrossRef]
- Basye, L.; Swaminathan, S. Hydrogen Production Costs—A Survey; DOE/GO/10170-T18; USDOE: Washington, DC, USA, 1997.
- Bonner, B. Current Hydrogen Cost. DOE Hydrogen and Fuel Cell Technical Advisory Committee; 30 October 2013. Available online: https://www.hydrogen.energy.gov/pdfs/htac_oct13_10_bonner.pdf (accessed on 16 September 2018).
- Methanex Methanol Price Sheet. Available online: https://www.methanex.com/our-business/pricing (accessed on 31 July 2018).
- Onel, O.; Niziolek, A.M.; Floudas, C.A. Optimal production of light olefins from natural gas via the methanol intermediate. Ind. Eng. Chem. Res. 2016, 55, 3043–3063. [Google Scholar] [CrossRef]
- National Energy Technology Laboratory. Analysis of Natural Gas-to-Liquid Transportation Fuels via Fischer–Tropsch; DOE/NETL-2013/1597; USDOE: Washington, DC, USA, 2013.
- Phillips, S.; Tarud, J.K.; Biddy, M.J.; Dutta, A. Gasoline from Wood via Integrated Gasification, Synthesis, and Methanol-to-Gasoline Technologies; USDOE Contract DE-AC36-08GO28308; National Renewable Energy Laboratory: Golden, CO, USA, 2011.
- Larson, E.D.; Jin, H.; Celik, F.E. Large-scale gasification-based coproduction of fuels and electricity from switchgrass. Biofuel. Bioprod. Biorefin. 2009, 3, 174–194. [Google Scholar] [CrossRef]





| Process | CH4 | Steam | Oxygen | CO2 |
|---|---|---|---|---|
| A-SR | 4270 | 7875 | 0 | 0 |
| B-SR | 3085 | 5056 | 0 | 1322 |
| C-SR | 3275 | 8448 | 0 | 1224 |
| A-ATR | 4052 | 2801 | 2087 | 0 |
| B-ATR | 4642 | 2012 | 2903 | 2233 |
| C-ATR | 5427 | 6728 | 3020 | 246 |
| A-CR | 3936 | 4310 | 1734 | 0 |
| B-CR | 4381 | 2600 | 1984 | 1717 |
| C-CR | 3684 | 5681 | 1737 | 402 |
| A-TSR | 4684 | 4568 | 1478 | 0 |
| B-TSR | 3732 | 5597 | 1461 | 1594 |
| C-TSR | 3531 | 5403 | 1294 | 523 |
| Material | Price | Unit | Reference |
| Methane | 132 | $/t | [30] |
| Steam (30 bar) | 28.85 | $/t | [31] |
| Oxygen 1 | 25 | $/t | [28,31] |
| Carbon dioxide 2 | 3 | $/t | [32] |
| Hydrogen | 750 | $/t | [33,34] |
| Methanol | 480 | $/t | [35] |
| Fuel gas 3 | 2.65 | $/GJ | [30] |
| Utility | Price | Unit | Reference |
| Electricity | 0.06 | $/kWh | [31] |
| Steam (LP/MP/HP) | 14.05/14.83/17.7 | $/GJ | [31] |
| Natural gas | 2.65 | $/GJ 4 | [30] |
| Cooling water | 0.354 | $/GJ | [31] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.-H.; Wong, D.S.-H.; Chen, Y.-C.; Chang, C.-M.; Chang, H. Design and Performance Comparison of Methanol Production Processes with Carbon Dioxide Utilization. Energies 2019, 12, 4322. https://doi.org/10.3390/en12224322
Chen Y-H, Wong DS-H, Chen Y-C, Chang C-M, Chang H. Design and Performance Comparison of Methanol Production Processes with Carbon Dioxide Utilization. Energies. 2019; 12(22):4322. https://doi.org/10.3390/en12224322
Chicago/Turabian StyleChen, Yih-Hang, David Shan-Hill Wong, Ya-Chien Chen, Chao-Min Chang, and Hsuan Chang. 2019. "Design and Performance Comparison of Methanol Production Processes with Carbon Dioxide Utilization" Energies 12, no. 22: 4322. https://doi.org/10.3390/en12224322
APA StyleChen, Y.-H., Wong, D. S.-H., Chen, Y.-C., Chang, C.-M., & Chang, H. (2019). Design and Performance Comparison of Methanol Production Processes with Carbon Dioxide Utilization. Energies, 12(22), 4322. https://doi.org/10.3390/en12224322







